Abstract
Quaternary ammonium compounds (QACs) are recognized by the World Health Organization as a useful disinfectant against microbes. The synergistic effect of zwitterionic polymers with QACs as antimicrobial agents rather than QACs alone is yet to be investigated. A potential strategy is the use of covalent bonding to halt the release of minute antibacterials and a hierarchy of functional layers to detain and annihilate microbes. The strategy was tested on a polydimethylsiloxane (PDMS) surface on which quaternized poly(2-dimethylaminoethyl methacrylate) (qDMA+) and sulfobetaine (SBMA) were hierarchically functionalized. Attenuated total reflectance Fourier transform infrared analysis confirmed the quaternization of DMA to qDMA+, grafting of qDMA + on PDMS (PDMS-qDMA+), and grafting of the SBMA overlayer on PDMS-qDMA+ (PDMS-qDMA+-SB). Contact angle measurement showed that PDMS-qDMA + exhibited the lowest contact angle (26.2 ± 2.9°) compared with the hydrophobic PDMS (115.2 ± 1.6°), but that of PDMSqDMA+-SB increased to 56.3 ± 1.3°. The Escherichia coli survival count revealed that PDMS-qDMA+ and PDMS-qDMA+-SB exhibited significantly greater bactericidal ability than PDMS. Confocal laser scanning microscopy revealed fewer dead bacteria on PDMS-qDMA+-SB than on PDMS-qDMA+. Scanning electron microscopy demonstrated that E. coli was disintegrated on the functionalized surface via dual-end cell lysis. To the best of our knowledge, this is the first observation of this type of process. The results confirmed the potent antibacterial and cell disruption activities of the qDMA+ and SBMA modified PDMS surface.
Introduction
Polydimethylsiloxane (PDMS) is an organosilicon elastomer with distinctive characteristics, such as a low glass transition temperature, favorable thermal stability, high flexibility, optical transparency, resistance to biodegradation, chemical inertia, and excellent biocompatibility. 1 It has been used in a wide range of fields including shampoos, food, coatings, sealants, microfluidic systems, sensors, electronic components, and drug release adhesives. 2 The US Food and Drug Administration has approved PDMS for use in medical devices. In biomedical applications, 3D printable acrylate PDMS has proven more suitable for in vitro drug testing and cell culturing of lung and skin epithelium compared with PDMS. 3 A porous titanium coating on PDMS, applied using the cold spray technique, enhances cell adhesion and proliferation, alkaline phosphatase production, and calcium deposition of MG63 cells. 4 Carbon implantation on PDMS using plasma processing facilitates covalent protein immobilization, thereby enhancing the attachment and spread of MG63 osteosarcoma cells. 5
PDMS has also been used for medical devices. For example, a PDMS bioscaffold-intravascular catheter has been shown to maintain in vitro islet viability and function for potential treatment of Type-1 diabetes. 6 In addition, membranes composed of PDMS have been used for extracorporeal membrane oxygenation in a biohybrid lung assist device. 7 However, the hydrophobicity of PDMS elicits nonspecific protein adsorption after contact with biological fluids and then increases the risks of microbial infection and persistent biofilm formation. The life span of the implant is consequently shortened and a replacement is ultimately required. Therefore, modification of PDMS material surfaces is required to combat biofilm formation.
Quaternized poly(2-dimethylaminoethyl methacrylate) (qDMA+) is a positive charge-bearing polymer that exhibits antibacterial capability against Gram-negative and Gram-positive bacteria. The mechanism is postulated to function via qDMA+ attaching to bacteria and diffusing throughout the cell wall, followed by disruption of the cytoplasmic membrane and bacterial death. 8 Therefore, using qDMA+ for biofouling prevention is classified as a contact killing strategy. Another study showed that low-density polyethylene and silicone rubber grafted with qDMA+ inhibited Candida albicans and Staphylococcus aureus biofilm formation. 9 qDMA+ copolymerized with a diazaborine-based polymer has been suggested for paints used in hospitals because of its antimicrobial ability against S. aureus, Escherichia coli, Pseudomonas aeruginosa, and C. albicans. 10 Although the effectiveness of qDMA+ against bacteria has been verified, PDMS grafted with qDMA+ and its antimicrobial ability remain to be investigated.
Another strategy is to resist bacterial adhesion via surface modification of hydrophobic PDMS to render it hydrophilic. Among hydrophilic materials, poly(ethylene glycol) (PEG) has been widely studied, 11 but oxidative degradation in complex media is a major drawback and long-term use is unsuitable. 12 Moreover, PEG is ineffective at preventing bacterial adhesion and colonization. 13 By contrast, zwitterionic polymers exhibiting strong hydration capacity and higher stability than PEG, and are thus considered as a potential antifouling agent. For example, zwitterionic phosphorylcholine-based polymers grafted onto PDMS surfaces using the matrix-assisted laser evaporation technique significantly reduce E. coli and S. aureus adhesion. 14 [2-(methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl)ammonium hydroxide (SBMA) grafted onto PDMS surfaces using oxygen plasma treatment could reduce salt deposition for urinary applications. 15
To enhance the ability of materials against biofouling, surface modification using both bactericidal polymer brushes and nonfouling zwitterionic polymer brushes has been studied. Ye et al. sequentially grafted sulfobetaine methacrylate (SBMA) and a [2-(methacryloyloxy)ethyl]trimethylammonium chloride solution on thin-film composite polyamide membranes using activators regenerated via the electron transfer−atom transfer radical polymerization technique. 12 Their results showed that this polymer architecture could effectively prevent bovine serum albumin adsorption and E. coli biofilm formation. Another study used 1,3-propanesultone for betainization of qDMA+ and the results demonstrated that 12% of quaternized and 85% of betainized units exhibited antibacterial and antifouling ability against E. coli. 16
An intervention/control strategy was adopted in this study to modify the PDMS surface through which pathogens are transmitted into an exterminator against them. The modification required covalent bonding of antibacterial agents (qDMA+ and SBMA) on the treated surface to halt their release at any level. The core of the design was a hierarchy of chemical layers on the modified surface. The covalent binding of qDMA+ and SBMA to the PDMS surface was characterized using attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy. The antibacterial power of the modified PDMS against E. coli was assessed using the spread plate method, confocal laser scanning microscopy (CLSM), and scanning electron microscopy (SEM).
Materials and methods
The aforementioned intervention/control strategy was tested using PDMS as the model substrate. PDMS discs were fabricated from a commercially available PDMS elastomer kit (Sylgard 184; Sil-More Industrial Ltd., New Taipei, Taiwan). The kit consisted of a silicon elastomer base and a curing agent. They were mixed thoroughly at a ratio of 10:1 (w/w; base: curing agent) until a homogenous phase was reached. The mixture was then de-gassed in a vacuum for 1 h before curing inside of a laminar flow hood at 80°C for 4 h to produce 0.5 cm dia. and approximately 0.5-mm-thick discs. The obtained PDMS discs were sonicated successively in deionized water, ethanol, acetone, hexane, and acetone again before drying and storage in a vacuum.
Materials
The PDMS discs were grafted using 1,1,4,7,10,10-hexamethyltriethylenetetramine (HMTETA, 97%); 2,2′-bipyridyl (bpy, 99%); 2,2-dimethoxy-2-phenylacetophenone (DMPA, 99%); (2-dimethylaminoethyl) methacrylate (DMA, 99%); [2-(methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl)ammonium hydroxide (SBMA, 97%); (3-mercaptopropyl)trimethoxysilane (MTS, 95%); allyl 2-bromo-2-methylpropionate (ABrMP, 99%); bromoethane (C2H5Br, 98%); copper(I) bromide (CuBr, 99.999%); copper(II) bromide (CuBr2, 99%); ethyl α-bromoisobutyrate (EBiB, 98%); potassium hydroxide (KOH, ca. 85%); acetonitrile (CH3CN, 99.5%); acetone (CH3COCH3, 99%); ethanol (C2H5OH, 99.9%); and methanol (CH3OH, HPLC grade).
Silanization of PDMS with MTS
To activate PDMS for covalent bonding, a layer of MTS was silanized onto PDMS by adding, at 50°C, MTS (0.75 M) dropwise to a KOH (0.150 M)-methanol mixture containing PDMS samples,
17
and the resulting solution was left at 50°C for 6 h (Step (a), Scheme 1). After heating, the activated, thiol-functionalized PDMS (hereafter PDMS-S) was rinsed with and sonicated in 95% ethanol to completely remove physisorbed MTS and KOH molecules. Surface grafting strategy for developing hierarchically modified PDMS as a potential approach to combate biofilm formation.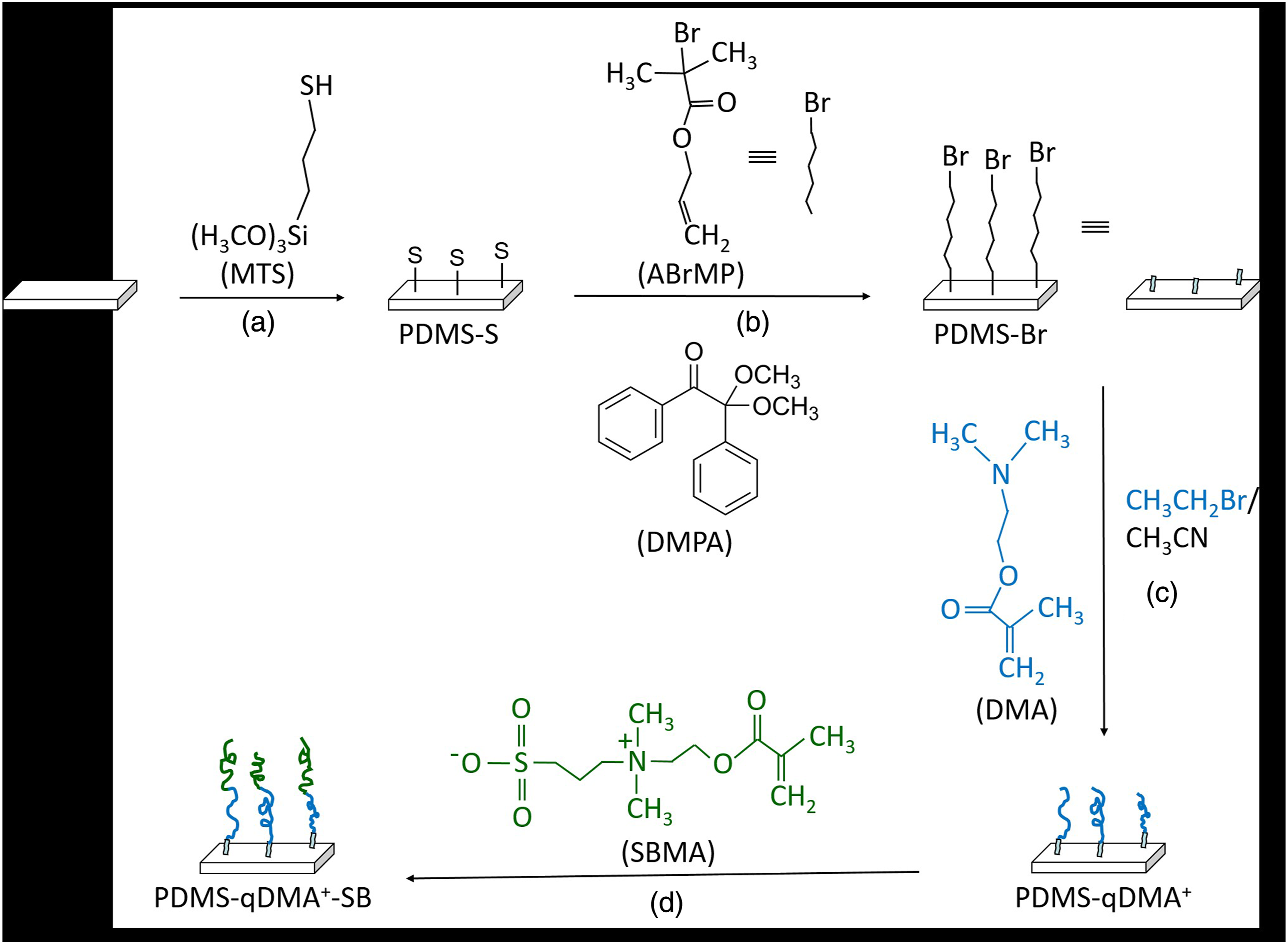
Grafting of ABrMP onto PDMS-S
The polymerization of 2-dimethylaminoethyl methacrylate (DMA) on PDMS-S was achieved by first depositing the initiator ABrMP on PDMS-S. As shown in Step (b) of Scheme 1, the deposition was carried out through a thiol-ene reaction 18 occurring on the PDMS-S sample placed in a glass tube containing ABrMP (463 M), DMPA (9.3 M), and ethanol. The tube was then purged with nitrogen and exposed to UV irradiation at λ = 365 nm for 2 h in a rotary photochemical reactor. The resulting ABrMP-initialized PDMS-S is hereafter referred to as PDMS-Br.
Polymerization of qDMA+ on PDMS-Br
DMA was grafted via atom transfer radical polymerization on PDMS-Br using a modified Huang’s procedure. 19 In brief, DMA (4.6 M) (total volume in this experiment was 1.0 mL of acetone + 4.0 mL of DMA + 8.7 µL of EBiB, for a DMA molarity of 23 mmol/(1.0 + 4.0+0.0087) mL = 4.6) and sacrificial initiator EBiB (0.012 M) in acetone were introduced into a Schlenk flask (Step (c), Scheme 1). After freeze–pump–thaw cycles, the mixture was transferred via a cannula into another Schlenk flask that contained CuBr and CuBr2 catalysts (3.2/1, w/w) and had undergone evacuate/backfill cycles to replace oxygen with nitrogen. After being stirred for 5 min, the new mixture was transferred again via another cannula into a third Schlenk flask, which had undergone evacuate/backfill cycles after PDMS-Br samples were placed into it. After the mixture was stirred at room temperature, HMTETA (0.057 M) was added to start the DMA polymerization. The polymerization lasted for 90 min before being terminated by exposing the system to the air to obtain DMA-grafted PDMS-Br (hereafter referred to as PDMS-DMA). To impart antibacterial properties, PDMS-DMA was immersed in a mixture of bromoethane and acetonitrile (1/1, v/v; 6.7 M) at 40°C for 24 h to obtain quaternized PDMS-DMA (hereafter referred to as PDMS-qDMA+).
Grafting of SBMA onto PDMS-qDMA+
The biocidal potency of the PDMS-qDMA+ polymer layer was tuned with an SBMA overlayer. Using Zhang’s procedure, 20 the overlayer’s optimal density of SBMA polymer chains was obtained as follows. SBMA (0.38 M) and bpy (0.10 M) were mixed into a solution of water and methanol (1:1, v/v) in a Schlenk flask (Step (d), Scheme 1). After freeze–pump–thaw cycles, the mixture was transferred into another Schlenk flask that contained the CuBr catalyst and had undergone evacuate/backfill cycles. After being stirred for 10 min, the mixture was transferred again into a third Schlenk flask, which had undergone evacuate/backfill cycles after PDMS-qDMA+ samples were placed into it. SBMA polymerization started when the mixture was stirred at room temperature. After 24 h, the polymerization was terminated by exposing the system to the air. The resulting samples are hereafter referred to as PDMS-qDMA+-SB.
Attenuated total reflectance Fourier transform infrared analysis
The surfaces of PDMS-S, PDMS-Br, PDMS-qDMA+, and PDMS-qDMA+-SB (n = 3) were examined using attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy (FTIR-4200; Jasco International Co., Ltd, Tokyo, Japan). FTIR spectra were recorded by pressing the samples against the ZnSe ATR crystal at a slow scan rate. The wavelength used was in the range of 4000–650 cm−1.
Contact angle measurements
Static contact angles were measured with the sessile drop method of water drops at room temperature using FTA125 (First Ten Ångstroms, Inc., VA, USA). Each specimen was mounted at a height sufficiently close to the delivery needle of a syringe, and water droplets (approximately 3 μL) were delivered to different points of each modified PDMS surface (n = 3). An image was then captured and the contact angle was calculated using the tangent method. All measurements were repeated at least three times for each sample.
Bacterial culture
Gram-negative E. coli stored at −80°C were separately cultured on tryptic soy agar (TSA; BD Biosciences, NJ, USA) at 37°C overnight. A strain of a single colony on TSA was then cultured in a 10-mL tryptic soy broth (TSB; BD Biosciences) at 37°C with an orbital shaker incubator at 220 r/min for 16 h. After 16 h of culture, the E. coli strains were harvested by centrifugation at 3000 r/min for 10 min. The resultant bacterial pellet was washed three times with sterile phosphate buffered saline (PBS) and then adjusted to a concentration of 107 colony-forming units (CFU)/mL (OD600 = 0.1) before use.
Bacterial survival test
The pristine and modified-PDMS samples (n = 6) were sterilized using 75% alcohol for 5 min and UV irradiation for 30 min. The bacterial suspension (OD600 = 0.1, 30 μL) was dropped onto the sample surfaces. It covered the surface without spilling over so as to eliminate the edge effect. The samples dropped with bacteria were incubated at 37°C for 4 h. The suspension was then drawn off using a micropipette, and the total live bacteria in the suspension were enumerated using the spread plate method. 21
Confocal laser scanning microscopy (CLSM) examination
For examination using CLSM, the pristine PDMS, PDMS-qDMA+, and PDMS-qDMA+-SB samples (n = 3) after the bacterial survival test were dyed using a LIVE/DEAD BacLight Bacterial Viability Kit (Molecular Probes, OR, USA) consisting of propidium iodide (PI) and SYTO® 9. Green fluorescing SYTO® 9 can label live bacteria and red fluorescing PI can label dead bacteria. A Zeiss LSM 880 CLSM (Carl Zeiss Microscopy, Jena, Germany) at 40× magnification was used at excitation wavelengths of 488 nm and 561 nm for SYTO® 9 and PI, respectively.
Scanning electron microscopy (SEM) examination
The instrument used for SEM was a field emission microscope (JSM-7600F, JEOL Ltd, Tokyo, Japan), which was operated under approximately 10−5 torr at a 10-kV accelerating voltage at various magnifications. The pristine PDMS, PDMS-qDMA+, and PDMS-qDMA+-SB samples (n = 3) were adhered to 15-mm aluminum stubs using a carbon conductive tape and sputter-coated with an approximately 10-nm layer of Pt to improve conductivity.
Results and discussion
FTIR characterization
The FTIR spectra taken from the PDMS-S, PDMS-Br, PDMS-qDMA+, and PDMS-qDMA+-SB surfaces, along with the qDMA+ and SBMA spectra, are displayed in Figures 1 and 2. Pristine PDMS typically has two main chemical groups: Si–CH3 and Si–O–Si. Because of the C–H stretching vibrations of Si–CH3, PDMS absorbed at 2962 and 2838 cm−1. Furthermore, the PDMS spectrum displayed a characteristic absorption band at 1256 cm−1, which was attributed to the deformations of C–H bonds in Si–CH3,
22
and a broad band encompassing the region between 1000 and 1100 cm−1, consisting of absorption resulting from the stretching of Si–O–Si bonds of different angles in a cage structure.
23
For PDMS-S, a very weak peak appeared at 2838 cm−1 (inset of Figure 1), which was attributed to the symmetric stretching of the methoxy group (Si–OCH3),
24
indicating the bonding of MTS molecules on PDMS. Although the peak became relatively intense at high initial concentrations of MTS, MTS was immobilized under mild conditions to avoid severe cracking on the substrate surface.
17
A peak attributed to the C = O stretching of the ABrMP ester group (O–C = O) indicated the grafting of ABrMP on PDMS-S at 1738 cm−1 in the PDMS-Br spectrum (Figure 1). The amount of ABrMP grafted to PDMS-S was small because MTS was immobilized under mild conditions. The grafted ABrMP served as the initiator for the atom transfer radical polymerization of DMA onto PDMS-Br before quaternization of the polymerization product with ethyl bromide. The spectrum of the resulting product, PDMS-qDMA+, exhibited ester absorption peaks of the methacrylate group in qDMA+ at 1145 and 1725 cm−1 (Figure 1). These peaks were ascribed to the acyl C–O and C = O stretching vibrations.
25
The absence of absorptions resulting from C = C stretching at 1637 cm−1,
26
= CH2 in-plane bending at 1318 and 1295 cm−1,
26
= CH2 out-of-plane bend or wagging vibration at 939 cm−1,
27
and = CH2 twisting vibration at 815 cm−1 indicated that no unreacted DMA or qDMA+ monomer was physically attached to the PDMS-qDMA+ surface.
28
Broad peaks were observed around 3430 and 1634 cm−1, which were attributed to the O–H stretching and bending of water adsorbed on the highly hydrophilic qDMA+ polymer brushes.
29
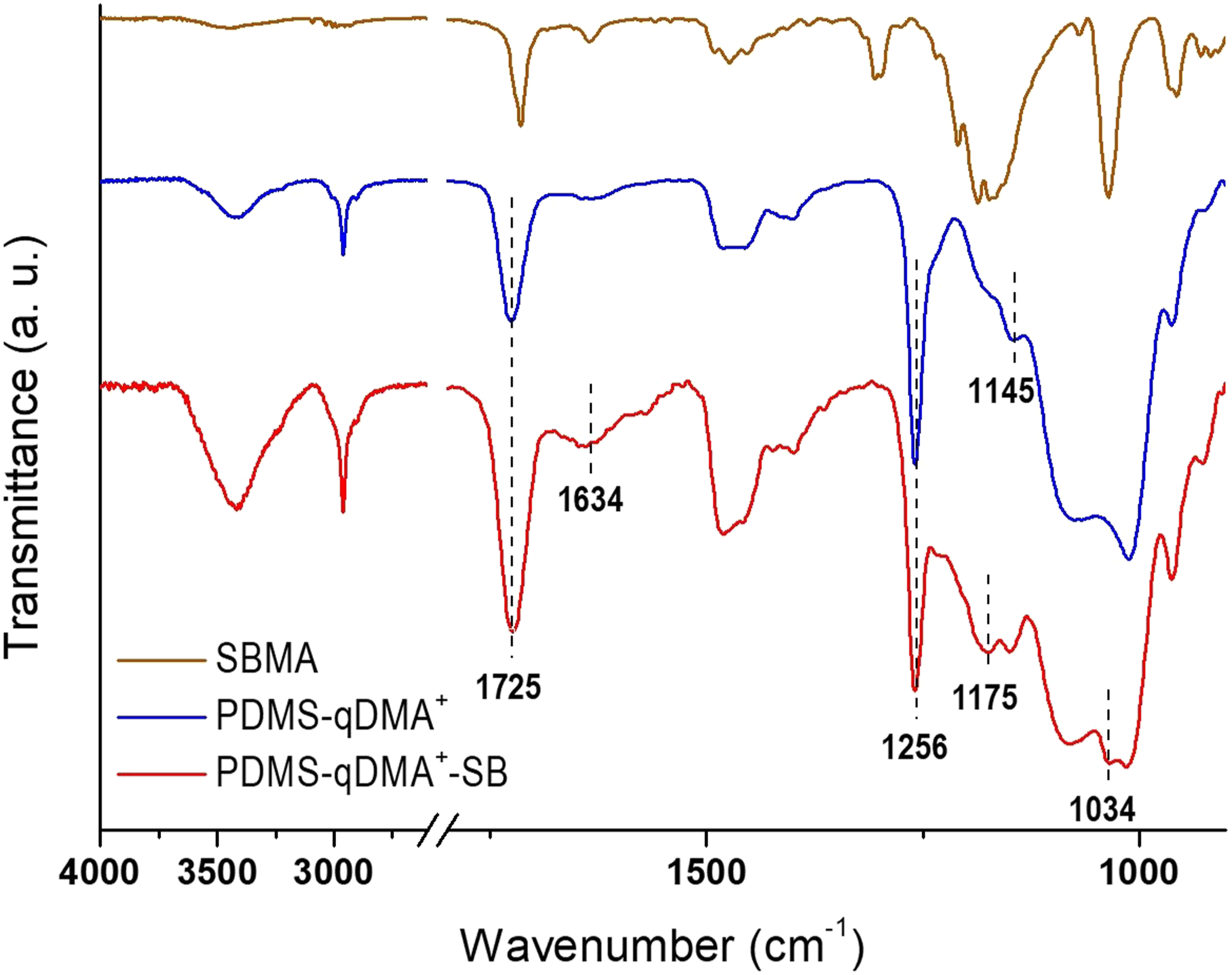
These peaks confirmed the quaternization of DMA to qDMA+. Tuning the biocidal potency of qDMA+ by grafting the polymer chains of the SBMA overlayer resulted in the absorption band growth of C = O at 1725 cm−1 and the growth of sulfonate at 1175 and 1034 cm−1 in the PDMS-qDMA+-SB spectrum (Figure 2). FTIR spectra of PDMS-S, PDMS-Br, and PDMS-qDMA+ compared to the spectrum of qDMA+. Inset: FTIR spectra of PDMS and PDMS-S with high MTS coverage. FTIR spectrum of PDMS-qDMA+-SB compared to the spectra of PDMS-qDMA+ and SBMA. The PDMS-qDMA+ spectrum is reproduced from Figure 1 to aid visual inspection.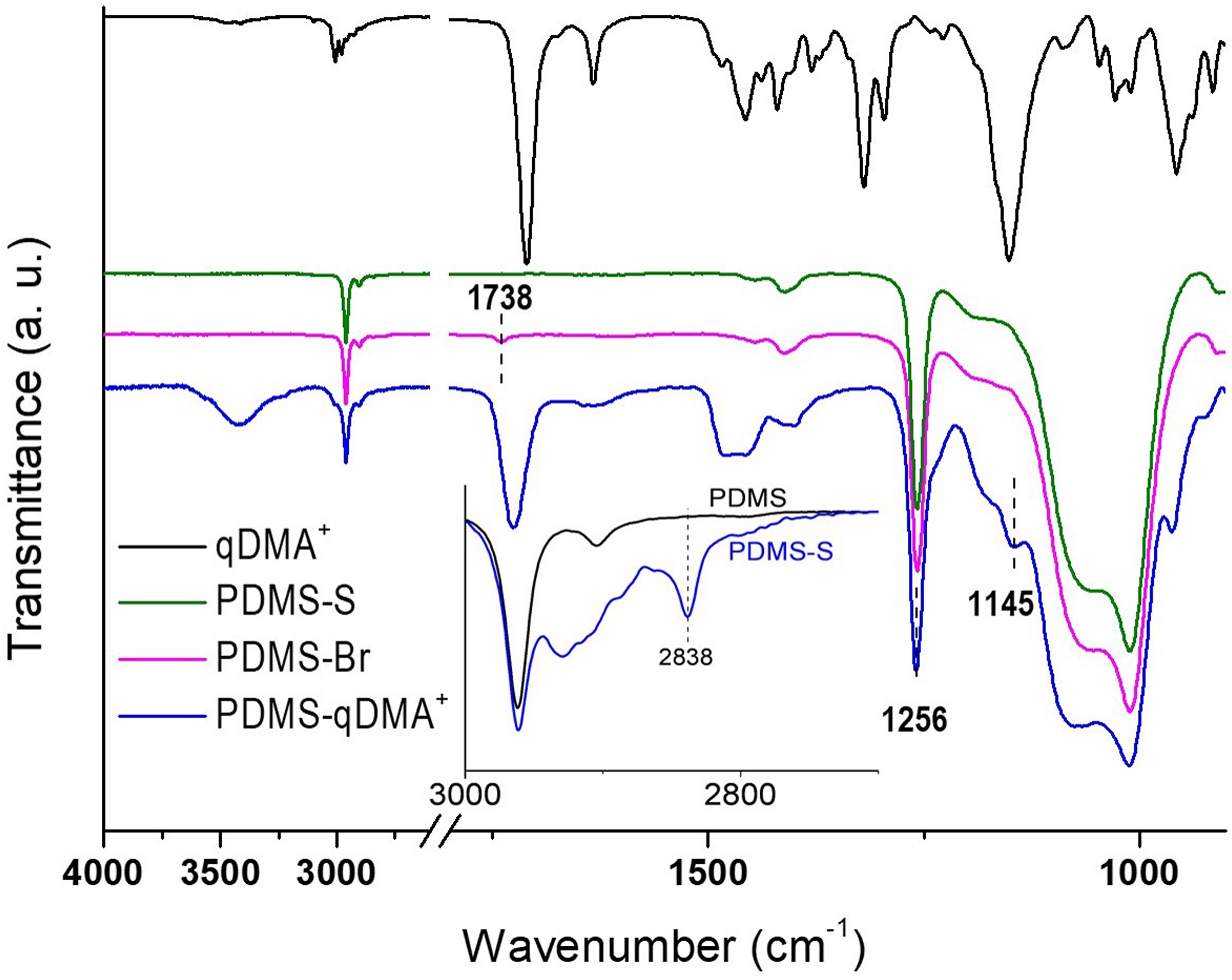
Contact angle measurement
Analysis of the surface hydrophilicity of the modified PDMS surfaces supported the intended hierarchical surface modification. The static contact angle with water of the pristine PDMS surface was 115.2 ± 1.6° (Figure 3),
30
indicating the surface to be hydrophobic. After MTS and ABrMP treatments, PDMS-S and PDMS-Br exhibited slightly higher contact angles of 129.0 ± 2.9° and 126.4 ± 1.5°, respectively. Because each ABrMP molecule contains an ester group and the ester monolayer typically displays contact angles in the range of 70–80°,
31
the high measured contact angle of 126.4° indicated that PDMS-Br was not fully covered with ABrMP. The grafting of DMA on PDMS-Br caused the measured contact angle to decrease sharply from 126.4 ± 1.5° to 78.2 ± 4.5°, and the PDMS sample surface was transformed from hydrophobic to hydrophilic.
32
The quaternization of grafted DMA to produce PDMS-qDMA+ further reduced the surface contact angle to the low value of 26.2 ± 2.9°. The large decrease was in agreement with the expected electrostatic interaction between the positive charge on the quaternary amine of PDMS-qDMA+ and the polar molecule of water.
33
Grafting zwitterionic SB to PDMS-qDMA+ led to an increase in the surface contact angle to 56.3 ± 1.3°. The changes in contact angle thus indicated the successful synthesis of the hierarchical surface. Water-contact angle measurements on pristine PDMS, PDMS-S, PDMS-Br, PDMS-qDMA+, and PDMS-qDMA+-SB sample surfaces.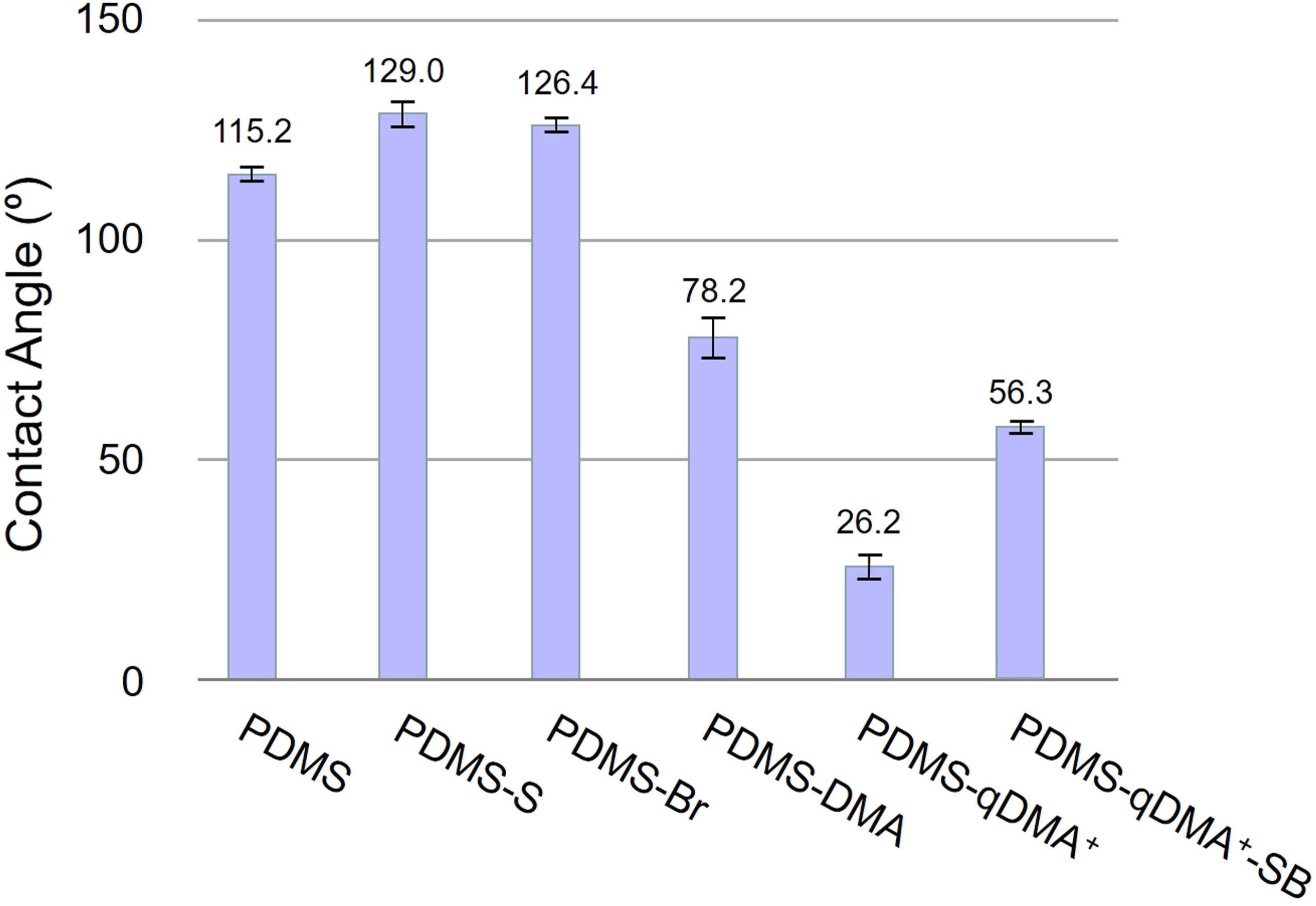
Bacterial survival count
The effectiveness of the synthesized hierarchical surface in terms of biocidal interaction to disintegrate microbes and produce signature fragments was tested using E. coli cells as the model microbes. A diluted bacterial suspension (OD = 0.1) was inoculated on pristine PDMS (which served as the control), the qDMA+-grafted PDMS, and the hierarchical surface. After inoculation for 4 h, the suspension was drawn from the surface and its bacterial survival count was determined using the spread plate method. As shown in Figure 4, the pristine PDMS surface, with a bacterial count of 1.77 × 106 CFU/mL, did not exhibit substantial bactericidal ability.
34
In comparison, the bacterial counts of the suspensions drawn from the PDMS-qDMA+ and the PDMS-qDMA+-SB surfaces were nil. Bacterial survival count in the E. coli inoculum (OD = 0.1) on pristine PDMS, PDMS-qDMA+, and PDMS-qDMA+-SB sample surfaces after 4 h of inoculation.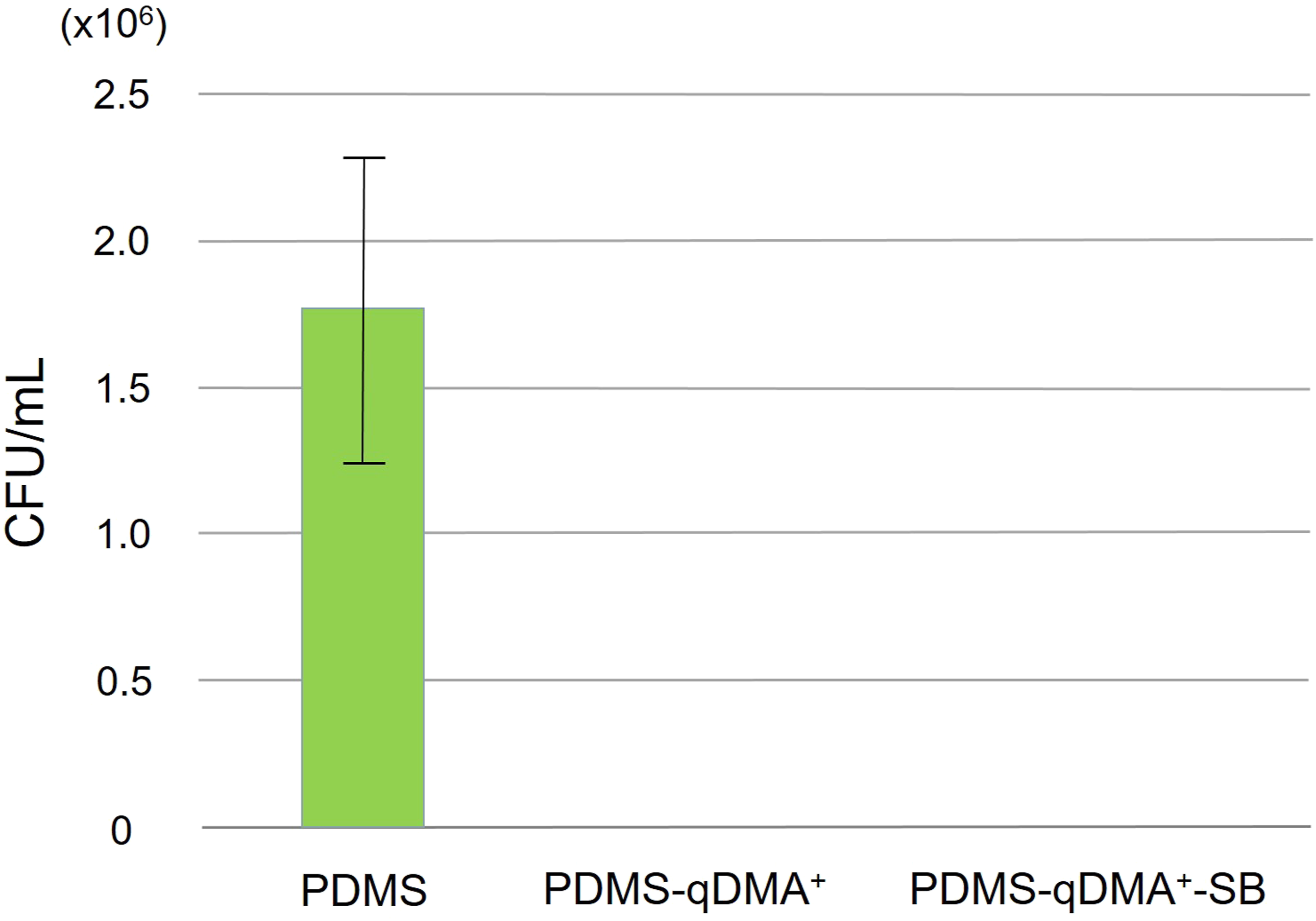
CLSM imaging
On the synthesized hierarchical surface, the overlayer of SBMA above the disinfectant qDMA+ served to optimize the biocidal interaction of qDMA+ with microbes for concurrent areal disruption across the whole microbe with which it was in contact. The SBMA polymer has positive and negative charges; theoretically, the observed nil count may have resulted from these charges forming a hydrophilic layer that trapped water molecules in an aqueous solution, leading to a large steric repulsive force against approaching bacterial cells such that no cells were attached to the surface.
35
CLSM was thus employed to observe the biofilms formed on the hierarchical surface to determine whether bacterial cells existed and if they were viable. The observation was made possible by first staining the bacteria suspended in sterilized PBS solution with SYTO 9 green-fluorescent dye, which can penetrate cells with both intact and damaged membranes, and then removing the free SYTO 9 dye from the solution through cycles of centrifugation and re-suspension. The diluted suspension (OD600 = 0.1) of the stained bacteria was then prepared for inoculation. The stained bacteria were then inoculated onto both the synthesized DMA-quaternized and the hierarchical surfaces for 4 h. After the sample was removed from the bacterial suspension and washed by backfilling and drawing of sterile PBS buffer to remove the non-attached bacteria, it was immersed in a PBS solution of propidium iodide in the dark for 15 min before being washed with PBS and undergoing CLSM analysis. As shown in Figure 5, the hydrophilic layer on the hierarchical surface prevented bacterial cells from approaching and resulted in far fewer dead bacteria on the SBMA overlayer than on the DMA-quaternized surface. However, some dead bacteria adhered to the hierarchical sur-face, indicating that the SBMA overlayer did not completely prevent bacteria from coming into contact with the surface; according to Halperin’s model,
36
the conformation of the SBMA polymer chains on the synthesized PDMS-qDMA+-SB may be between brush and coil shapes. However, no bacteria cells adhering to the surface remained alive, thus sup-porting the bacterial survival count determined using the spread plate method (Figure 4). That is, the disinfectant qDMA+ present underneath the overlayer of the protein-repellent SBMA in the synthesized sample was still capable of disrupting the cells. CLSM images of the pristine PDMS, PDMS-qDMA+, and PDMS-qDMA+-SB surfaces after 4 h of inoculation with the diluted suspension of SYTO 9 dye-stained E. coli (OD600 = 0.1) in PBS followed by 15-min immersion in a PBS solution of propidium iodide. The white scale bar is 10 μm.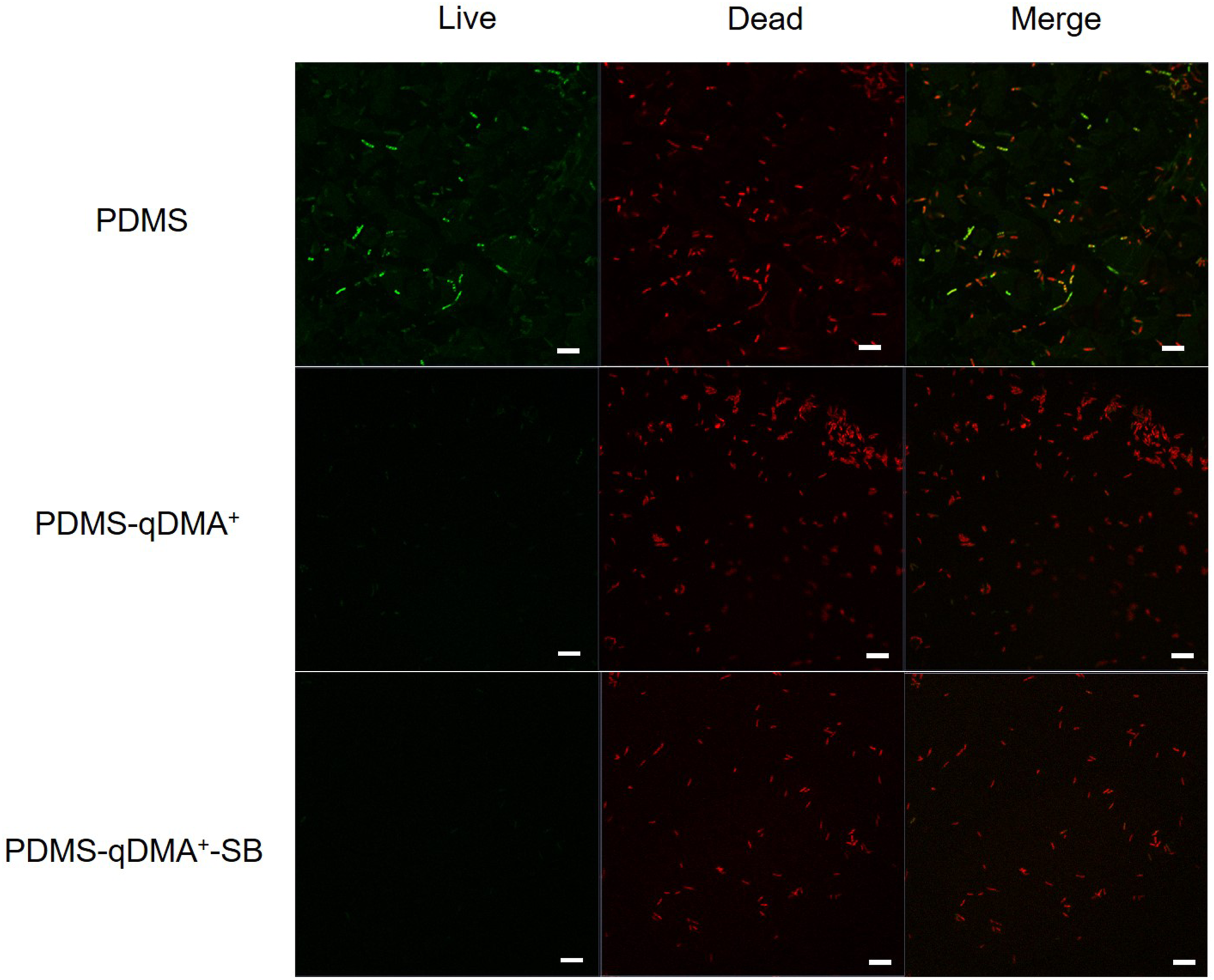
SEM imaging
The ability of the fabricated PDMS-qDMA+-SB hierarchical surface to disrupt microbes was examined after the surface was inoculated with a diluted E. coli suspension (OD = 0.1) for 4 h and, after being removed from the suspension and washed, incubated in TSB for 20 h. SEM images of the resultant surface (Figure 6(e)–(h)), compared with those of the pristine PDMS (Figure 6(a) and (b)) and PDMS-qDMA+ (Figure 6(c) and (d)) surfaces, showed that most bacteria adhering to the surface were surrounded by rod-shaped or oval “clouds” of substance. The differences in brightness indicated that the elevation of the “cloud” on the periphery of the bacterium might be higher than the surface and most parts of the bacterial cell. The “cloud” was not observed when the sample was not hierarchical (i.e., was fabricated with only a layer of qDMA+ or SBMA on PDMS-Br). As mentioned, the hierarchical surface had a two-layered architecture comprising quaternary ammonium and zwitterionic polymer layers. The underlying quaternary ammonium layer had a strong interaction with the bacterial cell, and the zwitterionic overlayer of SBMA polymer chains with a brush-coil shape may have retained the cell. Once the bacterial cell came into contact with the hierarchical surface, it became permanently adhered; the cohesive attraction of the surface was so strong that the membrane of the retained cell was spread widely. In addition to the outward spread, the membrane was also pulled downwards by the attractive interaction of the qDMA+ underlayer. The long SBMA polymer chains immediately underneath the outwardly spread membrane were wrapped and squeezed together by the downward pull of qDMA+. This squeeze caused the membrane to bulge higher than the main body of the bacterium, thus forming the “cloud.” SEM images of the (a and b) pristine PDMS, (c and d) PDMS-qDMA+, and (e–h) PDMS-qDMA+-SB surfaces after incubation in TSB for 20 h following the 4 h of inoculation of the E. coli suspension (OD = 0.1) and washing. The blue arrow indicates the “cloud” formation resulting from bacteria half-buried in the SBMA layer and strongly attracted by the underlying qDMA+ layer, causing the membrane to spread widely. Yellow arrows highlight the cell lysis occurring at the two ends of the cell, with different cells at different cell lysis stages.
The differences in brightness displayed in Figure 6(e)–(h) also indicated that the two ends, which were brighter, of almost all bacterial cells and their segments amid the “clouds” were higher than the corresponding center parts, which were dimmer. The differences in brightness suggested that the cell body on the PDMS-qDMA+-SB surface may have been severely bent into a boat shape, the center portion of which lay somewhat flat on the surface with the two ends bent upwards. No height dominance was noted for any particular parts of the cells if they were inoculated on the surface fabricated with only one layer (qDMA+ or SBMA). Thus, instead of bacterial scathe and corruption occurring randomly around the cells, Figure 6(e)–(h) show that the bacterial cells tended to always disrupt at approximately the same time at the two terminal ends of the cell first, regardless of the orientation at which the cells initially approached the surface. This dual-end lysis occurred even when the cells had been disintegrated to a very short length, as demonstrated by the cell segment on the lower right side of Figure 6(h). However, the brighter part of the cell in the SEM image was further away from the biocidal qDMA+ polymer layer. Its association with more extensive disruption than the dimmer part of the cell can be explained as follows. SBMA is non-bactericidal; as the top layer of the hierarchical surface, the SBMA polymers restrained the approaching cell from instantly coming into direct contact with the underlying qDMA+. Furthermore, during incubation, some bio-ingredients of TSB may have deposited on the surface and modified its properties such that some local sites on the top layer were more microbicidal than others. Thus, if the bacterial cells reached the hierarchical surface, their membranes were not lysed instantly at the contact points. Instead, the surface attracted the cells to lay flat on it. Because of the larger net interaction of qDMA+ polymer chains with the two ends of the cell than the middle parts, the ends then experienced higher stress. This stress may have induced the two ends to bleb at approximately the same time,37,38 resulting in the similar differences in brightness observed in the SEM images (Figure 6(e)–(h)) at the two ends of most bacteria or their disrupted segments. As the cell ends were disintegrated and the cytoplasmic content was exuded, the stress and blebbing gradually shifted from the cell termini toward the center. The shift caused the cell lysis to continue shortening the cell length. Although the cell segment remaining on the surface was only a small fraction of the intact cell, the stress and blebbing still caused the segment’s two ends to appear brighter than the center (lower right bacterial segment in Figure 6(h)). To the best of our knowledge, this is the first report of cell lysis that occurs simultaneously at two ends of a cell on bactericidal surfaces. The genetic material is removed from the end-lysed cell as the cytoplasmic content is exuded.
The concern for hydrophobic recovery of PDMS surfaces arises because PDMS has many applications such as in health and biomedical fields that need wetting. In order to make hydrophobic PDMS material hydrophilic, the material surface is often exposed to UV radiation and O2 plasma 39 or undergoes ultraviolet/ozone (UVO) treatment in water media. 40 The resulting hydrophilic PDMS surface is unstable, however, because the radiation/plasma treatment converts the chemically inert PDMS into a nonequilibrium state. Metastable radical-ion complexes, such as radical silanol groups, 41 may thus be formed on the PDMS material, causing its low molecular weight chains to diffuse from the bulk of the material and cover up the thermodynamically unstable surface. 42 The maximum lifetime is often on the order of days to weeks. 43
The PDMS sample used in this study, however, was never treated by O2 plasma or exposed to UV. Instead, our PDMS surface was activated by thermally treating the surface with MTS in a KOH-methanol solution. In addition, covalent bonding occurred in all the subsequent synthesis processes, including grafting of ABrMP on PDMS-S and polymerization of qDMA+ and SBMA zwitterionic polymers on PDMS-Br. The zwitterionic polymer has been reported to effectively suppress the hydrophobic recovery of PDMS when the polymer is coated by conventional radical polymerization on O2-plasma-treated PDMS specimens. 44
Because of the covalent bonding occurring in the fabrication of our products, the suppression of PDMS hydrophobic recovery by zwitterionic polymers, and the lack of use of radiation/plasma treatment in this study, the hydrophilic unstability of our PDMS was not perceived. No study was thus done on the hydrophobic recovery of our synthesized PDMS products.
Conclusions
This study demonstrated the potential usefulness of controlling the bactericidal activity of a qDMA+-protected solid surface with an overlayer of protein-repellent SBMA polymer chains grafted onto PDMS. Both PDMS-qDMA+ and PDMS-qDMA+-SB demonstrated excellent antibacterial ability against E. coli adhesion. Moreover, a unique characteristic was discovered in SEM images of the PDMS-qDMA+-SB group; E. coli were surrounded by an oval substance and partially submerged in the SBMA layer. The underlying qDMA+ layer simultaneously strongly attracted E. coli, causing cell lysis to occur at the two ends of the cell.
Nomenclature
polydimethylsiloxane
quaternary ammonium compounds
sulfobetaine
(3-mercaptopropyl)trimethoxysilane
2-(dimethylamino)ethyl methacrylate
allyl 2-bromo-2-methylpropionate
thiol-functionalized PDMS
ABrMP-initialized PDMS-S
DMA-grafted PDMS-Br
quaternized PDMS-DMA
SB-grafted PDMS-qDMA+
confocal laser scanning microscopy
scanning electron microcopy
tryptic soy broth
phosphate buffered saline
Footnotes
Acknowledgements
Imaging and spectral works were performed at National Taiwan University Hospital and the Instrumentation Center of National Taiwan University.
Author contributions
Conceptualization, C.-C.C.; methodology, Z.-T.C., B.-S.L., and C.-C.C.; validation, Z.-T.C., C.-C.C., B.-S.L., and Y.-T.C.; formal analysis, Z.-T.C., B.-S.L., and C.-C.C.; investigation, Z.-T.C and T.-H.T.; re-sources, B.-S.L., Y.-T.C. and C.-C.C.; data curation, Z.-T.C. and C.-C.C.; writing—original draft preparation, Z.-T.C. and C.-C.C.; writing—review and editing, Z.-T.C., B.-S.L., Y.-T.C., and C.-C.C.; visualization, Z.-T.C., B.-S.L., and C.-C.C.; supervision, C.-C.C., B.-S.L., and Y.-T.C.; project administration, B.-S.L. and C.-C.C.; funding acqui-sition, C.-C.C., B.-S.L., and Y.-T.C. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Taiwan University Hospital, grant number 111-S0191, and the National Science and Technology Council, Taiwan, grant numbers 105-2113-M-002-011, 109-2314-B-002-032-MY3, 109-2113-M-002-019-MY3, and 112-2314-B-002-081-MY3
