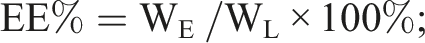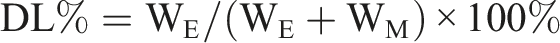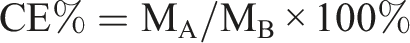Abstract
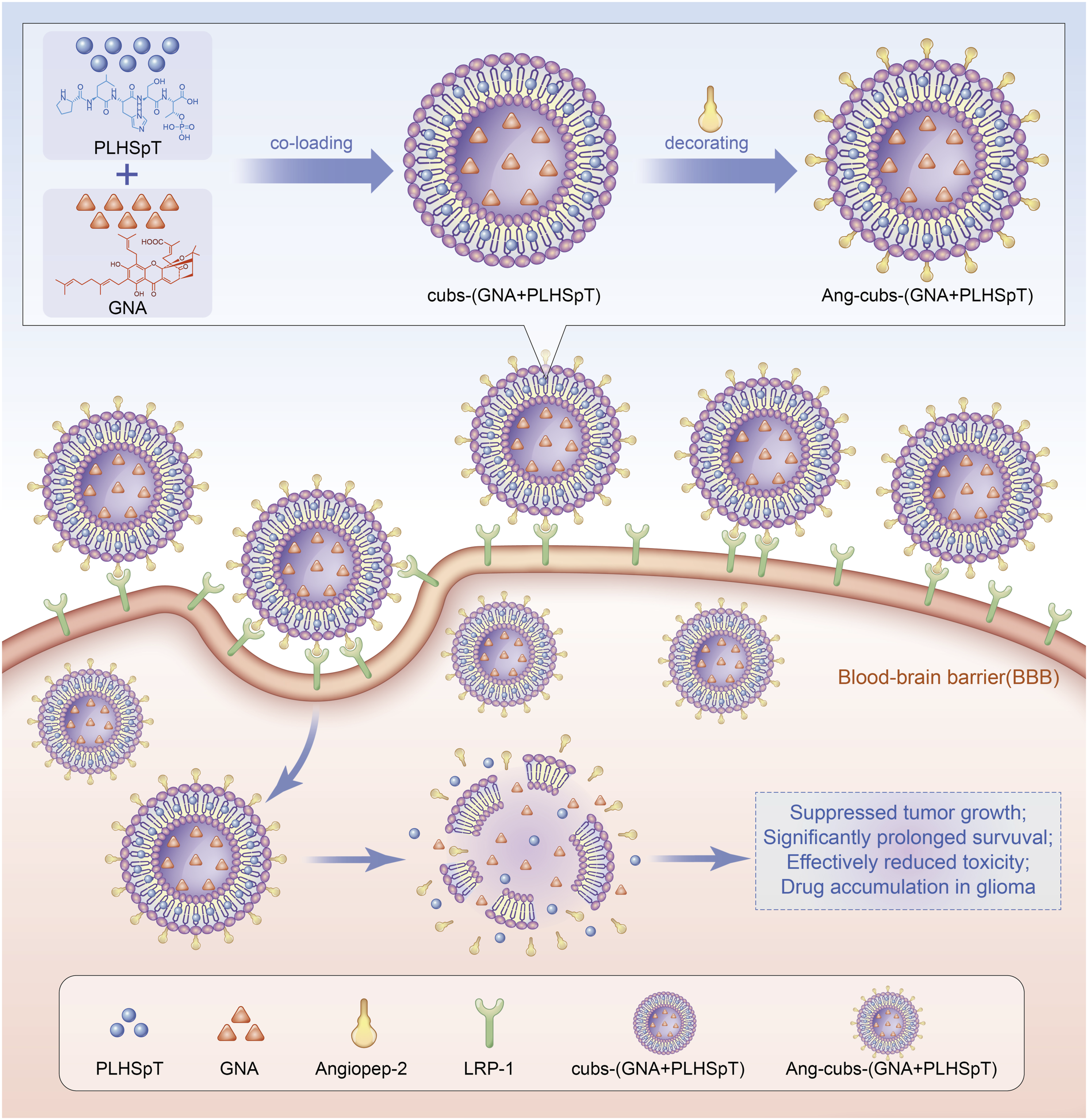
3Glioblastoma multiforme is the most aggressive malignant brain tumor. However, the treatment of glioblastoma multiforme faces great challenges owing to difficult penetration of the blood-brain barrier. Therefore, more effective treatment strategies are desired quite urgently. In our study, a dual-targeting drug delivery system for co-loading with hydrophobic Gambogenic acid and hydrophilic PLHSpT was developed by cubosomes with angiopep-2 decorating. The Ang-cubs-(GNA + PLHSpT) was prepared by high-temperature emulsification-low-temperature solidification demonstrating excellent physical properties.Transmission electron microscopy revealed that Ang-cubs-(GNA + PLHSpT) was nearly spherical with a “core-shell” double-layer structure. Differential scanning calorimetry suggested that a new phase was formed. Small-angle X-ray scattering also verified that Ang-cubs-(GNA + PLHSpT) retains the Pn3m cubic. Moreover, laser confocal indicated that Ang-cubs-(GNA + PLHSpT) was capable of crossing BBB via binding to lipoprotein receptor-related protein-1, likely suggesting the potential tumor-specific targeting characteristic. Compared to free drug and cubs-(GNA + PLHSpT), Ang-cubs-(GNA + PLHSpT) was easily taken up by C6 cell and exhibited better anti-glioma effects in vitro. Importantly, GNA and PLHSpT co-loaded Ang-cubs could suppress tumor growth and significantly prolong survival in vivo. In conclusion, Ang-cubs-(GNA + PLHSpT) acts as a new dual-targeting drug delivery system for the treatment of GBM.
Introduction
Glioblastoma multiforme (GBM), a subtype of glioma, is the most frequent and devastating primary brain tumor characterized by a high rate of proliferation cell and poor prognosis. 1 Worldwide, approximately 3.23 per 100,000 population were diagnosed per year with this fatal cancer and the incidence rate in men was higher than in women. 2 Histological analysis suggested the high mortality of GBM is owing to invasive peripheral growth and highly resistant cell subpopulations. The current standard therapeutic approaches, including surgery, radiotherapy, and chemotherapy, play a pivotal role in enhancing the quality of life and extending the survival of patients diagnosed with gliomas. Nevertheless, the prognosis for individuals with GBM remains unfavorable, with a median overall survival (OS) of less than 15 months and a 5-years survival rate of merely 4.6%.3–5 Numerous scholars argue that the treatment of GBM is somewhat limited by the blood-brain barrier (BBB), which serves as the primary barrier characterized by tight epithelial-like junctions and lack of fenestration, thereby restricting and regulating the substances from the blood flow to brain.6,7 Moreover, the vascular network surrounding brain metastases, known as the blood-tumor barrier (BTB), acts as a cellular barrier regulating the entry and exit of drugs into the brain.8,9 Consequently, there is a pressing demand for enhanced therapeutic approaches for glioblastoma (GBM) that prioritize the penetration of both the blood-brain barrier (BBB) and the BTB, while selectively targeting tumor cells without harming healthy tissue. In recent years, researchers have concentrated on “targeting” strategies for GBM patients, encompassing passive and active targeting methods that can be achieved directly or indirectly. 10 The targeting idea is based on the fact that the passive targeting is attributed to enhanced permeability and retention (EPR) effect. 11 Compared with passive targeting, active targeting is make use of targeting ligands, such as antibodies and penetrating peptides, which are proven to be able to actively recognize specific receptors on BBB and then leading to targeting delivery to the brain.12,13 Consequently, both passive targeting and active targeting complement each other by enhancing the specificity of drug delivery to brain tumor cells.
Cubosomes (cubs), a recently introduced nano-particle delivery system, have garnered significant scholarly attention in recent years.14,15 These cubs possess twisted and contorted lipid bilayers, resulting in three-dimensional structures that exhibit continuous hydrophobic and hydrophilic regions. This unique architecture offers potential benefits such as increased drug stability by shielding them from degradation.16,17 However, it is crucial to address the issue of nonspecific targeting of cubs, which may arise due to the spontaneous release of drugs in vivo. The potential limitations of BBB penetration by cubs and its implications for GBM research have been the subject of extensive debate. It is worth noting that the low-density lipoprotein receptor-related protein (LRP-1) is found to be overexpressed on brain endothelial cells of the BBB, but exhibits weak expression in normal brain parenchyma. 18 Numerous studies have demonstrated that the binding of ligands to LRP-1 facilitates the transport of cubs across the BBB through internalization, ultimately reaching the brain parenchyma. 19 Angiopep-2 (Ang, TFFYGGSRGKRNNFKTEEY, 2.4 kDa), a 19 amino acid peptide ligand, has been identified as a ligand for LRP-1. 20 Multiple investigations have provided evidence supporting the role of angiopep-2 in BBB penetration. Several laboratories used angiopep-2 conjugated nano-particles that could effectively improve the ability of chemotherapeutic drugs to cross BBB, increase the accumulation in GBM tumors.21–24
Hydrophobicity Gambogenic acid (GNA) and hydrophilic PLHSpT (Sequence:Pro-Leu-His-Thr(HPO3Bzl)-Thr) are regarded as model drug in the current study. Gambogenic acid (GNA, C38H47O8, Figure 1(a).) is a main active component isolated from Gamboge.
25
Available evidence suggests that GNA has shown to have strong anti-tumor activity in vitro and in vivo.26,27 However, GNA has not been approved to the clinical usefulness mainly due to its severe clinical side effects, primarily associated with water-insoluble, excessive irritation to blood vessel, and poor bioavailability.28,29 In our study, the other model elements PLHSpT (Figure 1(b).) is a specific inhibitor for Polo-like kinase 1 (Plk1). The Plk1 is abundantly expressed in tumor cells, but not in normal cells, and is also associated with adverse prognosis, metastatic potential and cancer progression.30,31 Structure of GNA (a) and PLHSpT (b).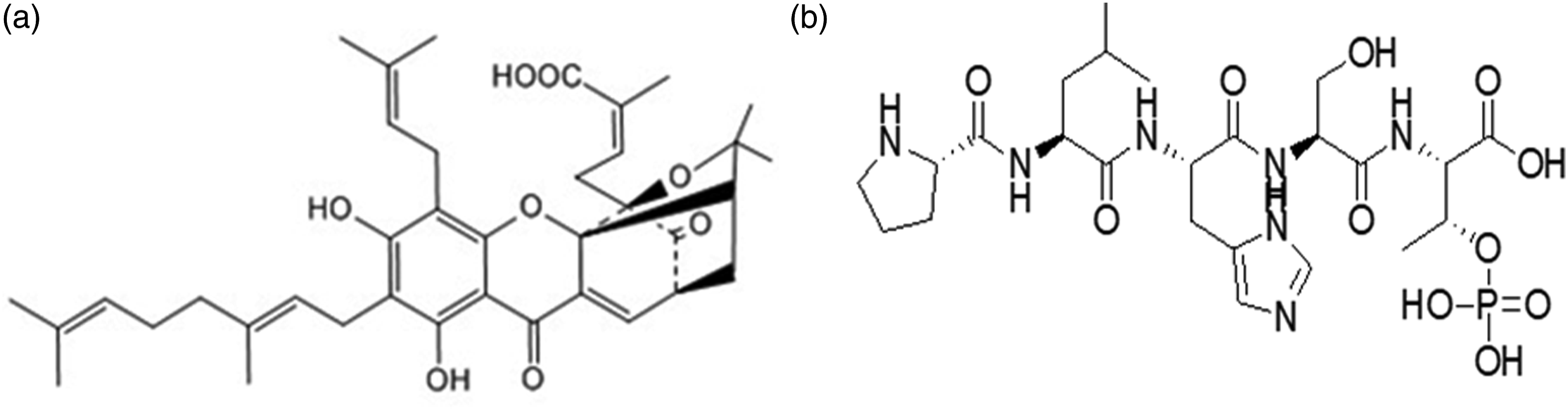
In this study, we employed the conjugation of “triple-interaction” angiopep-2 decorated novel cubs systems (Ang-cubs-(GNA + PLHSpT)). The physicochemical properties of Ang-cubs-(GNA + PLHSpT), including particle size, Zeta potential, transmission electron microscopy (TEM), entrapment efficiency (EE%), drug loading (DL%), differential scanning calorimetry (DSC), small-angle X-ray scattering (SAXS) and ligation efficiency were comprehensively investigated. Subsequently, the impact of Ang-cubs-(GNA + PLHSpT) on cellular uptake, cell proliferation, cell apoptosis, and blood-brain barrier (BBB) permeability was assessed in vitro. In addition, the anti-tumor effect of Ang-cubs-(GNA + PLHSpT) on tumor-bearing mice was analyzed in vivo.
Materials and methods
Materials
GNA (purity ≥ 98.0%) was sourced from Gamboges in author’s laboratory. GMO was purchased from Tianjin heowns Biochemical Technology Co., Ltd. (China). Tween-20 and Tween-80 were supplied from Tianjin Chemical Reagent Company (China). F127 was obtained from BASF (Germany). 9-Fluorenylmethoxycarbonyl (Fmoc)-protected L-α-amino acids, Fmoc-N-Me-Leu-OH, Fmoc-Thr-(HPO3Bzl)-OH, Fmoc-Phe(4-NO2)-OH, Fmoc-Phe(4-Cl)-OH, Fmoc-Phe(4-F)-OH Fmoc-Tyr(Me)-OH, Fmoc-Phe(3,4-Cl)-OH, Hydroxybenzotriazole(HOBT), O-(Benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate (TBTU), o-(7-Azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate (HATU), o-(Benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate (HBTU) and Rink-amide-MBHA resin were purchased from GL Biochem Ltd (China). Dimethylformamide(DMF), piperidine, diethyl ether, dichloromethane (DCM), triisopropylsilane (TIPS), diisopropylethylamine (DIPEA), Ethylene Mercaptan (EDT), trifluoroacetic acid (TFA) and phenol were purchased from Sinopharm Chemical Reagent Company (China). Sephadex gel G-50 was purchased from Beijing Ruida Henghui Science &Technology Development Co., Ltd. (China). Dimethyl sulfoxide (DMSO), trypsin, (EDTA) and streptomycin/penicillin were obtained from Sigma (USA). DAPI and Annexin V-FITC/PI were obtained from Bestbio Biotechnology (China). 1.1-dioctadecyltetra-methyl indotricarbocyanine iodide (DiR) and coumarin 6 were commercially obtained from Biotium Inc (USA). Fetal bovine serum (FBS) and 0.25% trypsin were obtained from Thermo Fisher Scientific Co., Ltd (China). The GBM (C6 cell) cell line was obtained from Institute of Biochemistry and Cell Biology, Institute for Biological Sciences, Chinese Academy of Science (China). All other chemicals and solvents were analytical purity grade. Water used was doubly distilled and deionized.
Synthesis of angiopep-2 and PLHSpT
Synthesis of Angiopep-2 was based on conventional Fmoc-solid phase peptide synthesis (SPPS. 32 Our laboratory has explored efficient means for synthesizing PLHSpT which contains non-natural amino acids. The synthesis method for PLHSpT which contains non-natural amino acids was based on Lin et al. 33 The Fmoc-Thr (HPO3Bzl)-OH of PLHSpT with spatial steric hindrance should be three times coupling reactions by treatment with different coupling reagents. Reaction completion was checked by means of the Kaiser tests. The phosphopeptide was cleaved from the rink amide resin with simultaneous removal of side chain protecting groups from Fmoc amino acid by the optimized mixture of TFA-H2O-EDT-TIS (95:1:2:2) for 3 h at ambient temperature. The filtrate was collected and then precipitated with ice-cold ether. Additionally, crude phosphopeptides above were purified by the preparative RP-HPLC (XBridge™, Prep C18, 50 × 250 mm, 10 μm), and then the characterization of Angiopep-2 and PLHSpT was confirmed by TOF-MS and analytical by High-Performance Liquid Chromatography (HPLC) equipped with Waters 1525-2489 HPLC system (Waters 1525 Binary HPLC Pump, Waters 2489 UV/Visible detector). Samples were analyzed through COSMOSIL C18 column (4.6 × 250 mm, 5 μm; Nacala Inc., Japan) at 30°C. The mobile phase methanol/0.1% phosphoric acid solution (90:10, vol/vol) was used for chromatographic separation at a flow rate of 1.0 mL/min.
Preparation of Ang-cubs-(GNA + PLHSpT)
Based on preliminary research in our laboratory, cubs-(GNA + PLHSpT) was prepared by emulsification evaporation and low-temperature solidification metho. 34 We determined the optimal prescription by single-factor experimental design and orthogonal array. In short, GNA (5.0) mg and GMO (100.0) mg were completely dissolved into anhydrous ethanol (5.0) mL in water at 60°C to obtain the organic phase. The 40.0 mL water solution containing p407 (12.0) mg and PLHSpT (0.5) mg were heated to same temperature. Then the organic solution was slowly and uniformly injected into the aqueous solution and the stirring continued for 2 h until the organic solvent volatilized completely to obtain the uniform emulsion. Following that, the obtained primary emulsion was quickly dispersed intocold distilled water (0-4) °C under stirring at 1000 r/min for 1 h. Finally, a certain proportion was injected into angiopep-2 (0.5) mg incubated at 25°C for 1 h. Angiopep-2 was connected to cubs-(GNA + PLHSpT) through esterification reaction.
Characterization of Ang-cubs-(GNA + PLHSpT)
Particle diameter and Zeta potential of Ang-cubs-(GNA + PLHSpT)
The particle diameter, polydispersity index (PDI) and Zeta potential of Ang-cubs-(GNA + PLHSpT) were evaluated by dynamic light scattering (Malvern Instrument, UK). Ang-Cubs-(GNA + PLHSpT) was diluted with deionised water for the measurements. Each sample was carried out in triplicate at room temperature.
Morphology of Ang-cubs-(GNA + PLHSpT)
A total of 10.0 µL aliquots of Ang-cubs-(GNA + PLHSpT) suspension were applied to carbon coated copper grids. After 3 min, negatively stained with 2% sodium phosphotungstate solution. The morphology of Ang-cubs-(GNA + PLHSpT) was characterized using a transmission electron microscope (JEM1400, JEOL Ltd., Tokyo, Japan. 35
Small-angle X-ray scattering of Ang-cubs-(GNA + PLHSpT)
Small-Angle X-ray scattering (SAXS) is a well-established and versatile technique to obtain information about the nanoscale structures and dimensions. The internal crystallographic structure of Ang-cubs-(GNA + PLHSpT) was assayed using SAX. 36 And the resulting data were processed using DAWN software.
Differential scanning calorimetry of Ang-cubs-(GNA + PLHSpT)
The crystalline structure of Ang-cubs-(GNA + PLHSpT) was determined using DSC Q2000 (TA Instrument, New Castle, DE). The Ang-cubs-(GNA + PLHSpT) (5.0–7.0) g were accurately weighed in the aluminum pans then hermetically sealed. The empty pans were used as a reference. The thermal behavior was obtained at a scanning rate of 10°C/min in the temperature of (20–300) °C under a nitrogen purge 50 mL/min.
Entrapment efficience and drug loading of Ang-cubs-(GNA + PLHSpT)
Entrapment efficiency (EE%) and drug loading (DL%) of Ang-cubs-(GNA + PLHSpT) was determined by measuring the amount of GNA or PLHSpT entrapped in Ang-cubs-(GNA + PLHSpT) system. The total drug amount using HPLC nonencapsulated GNA and PLHSpT were separated from Ang-cubs-(GNA + PLHSpT) by size exclusion chromatography using a minicolumn centrifugation method and purified water as the eluent. The EE% and DL% were determined as follows equation:
WE is the actual amount of GNA or PLHSpT entrapped in Ang-cubs-(GNA + PLHSpT) system, WL is theoretical amount GNA or PLHSpT in nanoparticles drug delivery system, while WM is the materials amount. Each experiment was carried out in triplicate.
Connection efficiency of angiopep-2 with cubs-(GNA + PLHSpT)
To determine ligation efficiency, consider measuring the amount of protein (i.e. angiopep-2) in the wash solutions using BCA. Free Angiopep-2 was removed by centrifugation using an ultrafiltration tube. First, the protein reference substance 1.0 mg/mL in the kit was diluted to different concentrations and added to a 96-well plate, and three replicate wells were set for each concentration. Incubate at 37°C for 30 min, measure absorbance of each well with a microplate reader at 570 nm, calculate the average of three replicate wells at each concentration, and draw a standard curve between protein concentration and absorbance. Then add BCA reagent to the sample solution Ang-cubs-(GNA + PLHSpT) according to the instructions, measure its absorbance using a microplate reader, and substitute it into the standard equation to calculate the concentration of Angiopep-2. The calculation formula of connection efficiency (CE%) of Angiopep-2 is as follows:
Cell culture
In this study, we used C6 cell, which was maintained in DMEM media (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS), 100 units/mL penicillin and 100 units/mL streptomycin (Sigma, St. Louis, Mo, USA) under a humidified atmosphere containing 5% CO2 at 37°C. The C6 cell were harvested from the confluent cultures by trypsinization and collected by centrifugation at 1000 g for 5 min. And re-suspended cell into a new culture dish with fresh culture medium maintained at 37°C and 5% CO2. All procedures were performed on sterile conditions.
In vitro cell cytotoxicity assay of Ang-cubs-(GNA + PLHSpT)
The cytotoxicity of Ang-cubs-(GNA + PLHSpT) was assessed using MTT assay. C6 cell were grown in a 96-well culture plate at density of 6 × 104 cells and allowed to attach for 24 h. After discarding the old medium, the C6 cell were then incubated with GNA-solutions, PLHSpT-solutions, blank cubs, cubs-GNA, cubs-PLHSpT and cubs-(GNA + PLHSpT) as well as Ang-cubs-(GNA + PLHSpT) at various concentrations (0.5, 1.0, 2.0, 4.0, 6.0, 8.0, 10.0 and 20.0) μg/mL for 24 h. Next, 50.0 μL MTT (3-(4,5-Dimethylthiazol2-yl)-2,5-Diphenyltetrazolium Bromide) reagent was added to each well for an additional incubation for 4 h to form violet crystals. The supernatant was then removed and 100.0 μL DMSO was added to dissolve any formed formazal crystals. Cell viability was calculated according to the formula:
Cell viability (%) = (OD emodin treated cells-OD emodin medium control) / (OD untreated cells-OD medium control) × 100%. The IC50 is calculated using GraphPad software. The study was carried out in triplicates and cell viability assay was presented as the Mean ± S.D.
In vitro cell apoptosis assay of Ang-cubs (-GNA + PLHSpT)
The Annexin V-FITC/PI staining and flow cytometer was used to evaluate cell apoptosis for each sample in our study. Briefly, C6 cell were plated in a 6-well culture plate at cell density of 1.0 × 106 cells/dish. Different concentrations (1.0,2.0,4.0) μg/mL of GNA-solution, PLHSpT-solution, cubs-PLHSpT, cubs-GNA, cubs-(GNA + PLHSpT), Ang-cubs(GNA + PLHSpT) were added to the 6-well plate and incubated for an additional 24 h, respectively. In control group, 3.0 mL of blank culture medium without drug was added instead. After incubated for 24 h, the C6 cell were collected and rinsed three times with PBS. According to manufacturer’s instructions, the cell apoptotic assay was identified by double supravital staining with recombinant fluorescein isothiocyanate (FITC)-conjugated Annexin-V and PI, using the Annexin V-FITC apoptosis detection kit (Becton Dickinson, Canaan, CT, USA) at room temperature in the dark. Ten thousand events were collected for each sample and analyzed using BD FACS Calibur flow cytomete−38. 37
In vitro wound-healing assay of Ang-cubs (-GNA + PLHSpT)
The C6 cell were seeded in a 6-well plate at a density of 5 × 105 cells/well, and three replicate wells were set in each group. Inter-secting scratches draw a straight line the center of the well with a pipette tip and then carefully rinse the surrounding cell debris with sterilized PBS buffer. Observe and take pictures under microscope (recorded as the result of 0 h), then C6 cell were add with the concentration of 2 μg/mL GNA-solution, PLHSpT-solution, cubs-PLHSpT, cubs-GNA, cubs-(GNA + PLHSpT) and Ang-cubs(GNA + PLHSpT). After culturing for 24 h, observe and take pictures inverted microscope (Olympus Optical Co., Ltd, Tokyo, Japan). Image J V1.8.0 analyzes the experimental results, and the cell migration rate was calculated according to the width of the cell scratch area within 24 h. Cell migration rate (%) was calculated according to below formula:
Cell migration rate (%) = (scratch area at 0 h - scratch area after 24 h of culture)/(scratch area at 0 h) × 100%
Establishment of the blood-brain barrier model in vitro
The BMECs with higher positive rate were used as the BBB model and C6 as tumor cells to establish BMECs-C6 co-culture model in vitro.First, BMECs were seeded in the upper chamber of transwell at 1 × 105 cells/well then blank medium was added to the lower chamber, and incubated in a 37°C, 5% CO2 incubator. The C6 cell were seeded into other 6-well plate at 1 × 105 cells/well, and were routinely cultured for 24 h. Measure the transmembrane resistance value of BMECs and C6 cell, and select a transwell chamber with a value greater than 200 cm2 then combined the upper and lower chambers, and continue to culture for 24 h to establish an BMECs-C6 co-culture model in vitro.
In vitro cell uptake of Ang-cubs-(GNA + PLHSpT)
To study cellular uptake of Ang-cubs-(GNA + PLHSpT) by laser confocal inverted fluorescence microscope, C6 cell were cultured in DMEM with 10.0% FBS 2.0 mM glutamine and 100 U penicillin with 0.1 mg/mL streptomycin for 24 h. Remove the original medium was then added same volume fresh medium into each well. The concentration of 2.0 μg/mL with Courmarin 6 -solution, cubs-C6, cubs-(Courmarin 6+PLHSpT) and Ang-cubs-(Courmarin 6+PLHSpT) were added to each well and incubated for 4 h, respectively. The 4.0% paraformaldehyde was added and left after 10 min. The nuclei were stained with DAPI, and the coverslips removed then washed three times with PBS before mounting on microscope slides (ImmuHistoMount). Confocal images were obtained with Zeiss Axio Observer with Zeiss LSM 800 confocal module (Germany) using sequential scanning. DAPI visualization 405 nm laser was used for excitation and Rhodamine 561 nm excitation laser.
In vivo imaging of Ang-cubs-(GNA + PLHSpT)
Eight sprague-dawley rats randomly divided into two groups (I and II). The cubs-(DiR + PLHSpT) and Ang-cubs-(DiR + PLHSpT) solutions (400.0 μL/rat) were injected by tail vein, respectively. The rats were anesthetized with isoflurane at (2, 4, 12, 24) h, and the fluorescence distribution and intensity of DiR in rats was observed by in vivo imaging system (IVIS). After 24 h, the rats were sacrificed then the brain tissue was taken and photographed.
In vivo anti-tumor assay of Ang-cubs-(GNA + PLHSpT) 38
Sprague-Dawley rats (200 ± 20) g were obtained from the Laboratory Animal Center of Anhui Medical University (Hefei, Anhui, China). All animal experimental protocols were approved by the Anhui University of Chinese Medicine. C6 cell (5 × 106) were suspended in PBS with 50.0% matrigel and inoculate 400.0 μL of tumor cells in tail vein for each rat. The 32 animals with better survival status were selected and randomly divided into 4 groups (saline control group, GNA-solution group, cubs-(GNA + PLHSpT) group and Ang-cubs-(GNA + PLHSpT) group) when the tumors on the brain grew into about 150 mm3. Eight rats for each group (half male and half female) were treated via tail vein injection at dose of 2.0 mg/kg. Dosing every other day, a total of ten times. The body weights of glioma rats were measured every 2 day. The survival times were recorded and median survival curves were analyzed using Kaplan-Meier method. At the end of experiment, the glioma rats were sacrificed. The heart, liver, spleen, lung, kidney and brain tissue were removed and processed for Haematoxylin and eosin (H&E) staining analysis.
In vivo histological evaluation of Ang-cubs-(GNA + PLHSpT)
The heart, liver, spleen, lung, kidney and brain tissue were removed from sacrificing glioma rats, fixed in 4.0% paraformaldehyde for 24 h, progressively dehydrated. Above tissue were embedded into the paraffin and then cut into 5.0 μm sections. The sections were incubated with indicated antibodies. H&E staining was carried out according to the standard protocol with H&E to indicate nucleus and cytoplasm, respectively.
Statistical analysis
Each assay was repeated at least three independent times except for animal experiments. All data were expressed as mean ± S.D. The two groups data was analyzed by Student t-test using SPSS 22.0 statistical software. The Anova and a posthoc test should be used instead for more than 2 groups. p* < .05, p** < .01, p*** < .001.
Results and discussion
Physicochemical characterization of optimal Ang-cubs-(GNA + PLHSpT)
The optimal prescription particle size and Zeta potential of Ang-cubs-(GNA + PLHSpT)
The optimal prescription of Ang-cubs-(GNA + PLHSpT).

As known that the size and Zeta potential of Ang-cubs-(GNA + PLHSpT) are very important for studying the interaction between the cell membrane and nano-particles. In our study, increasing the concentration of angiopep-2 resulted in an increase of Ang-cubs-(GNA + PLHSpT) particle size (support information-table 1). This could be attributed to an increase in viscosity of the organic phase with increasing angiopep-2 leads to a reduction in the evaporation rate of the organic phase thereby forming larger particles. On the other hand, increasing nano-carrier material ratio (GMO: p407) resulted in a decrease in particle size upon fixing the amount of drug and angiopep-2, which could be explained by the fact that reduction of the interfacial tension between dispersed organic phase and dispersion media (aqueous phase), leading to smaller physically stabilized particles with tighter surfaces (support information-table 2). Overall, the size of Ang-cubs-(GNA + PLHSpT) is highly dependent on the ratio of GMO with p407 and the angiopep-2 concentration. In the current study, the average size of optimal Ang-cubs-(GNA + PLHSpT) was (128.70 ± 1.04) nm, suggesting acceptable size distribution (Figure 2(a)). In general, the nano-carriers of (100–400() nm size are expected to accumulate in the tumor site by the EPR effect.
39
Additionally, the PDI of Ang-cubs-(GNA + PLHSpT) was below 0.2, showing a uniform nano-system. Regarding Zeta potential, an important parameter for nano-particles reflects the extent of repulsion between nanoparticles. The Zeta potential of optimal Ang-cubs-(GNA + PLHSpT) was higher than |30| mV which demonstrates that all cubs were relatively stable in the dispersion system (Figure 2(b)).A possible explanation for these results that the angiopep-2 adsorbed on the colloidal systems surface could remain stable for in vitro storage due to electrostaric repulsion. Particle size and Zeta potential of Ang-cubs-(GNA + PLHSpT). Particle size (a) and Zeta potential (b) of Ang-cubs-(GNA + PLHSpT).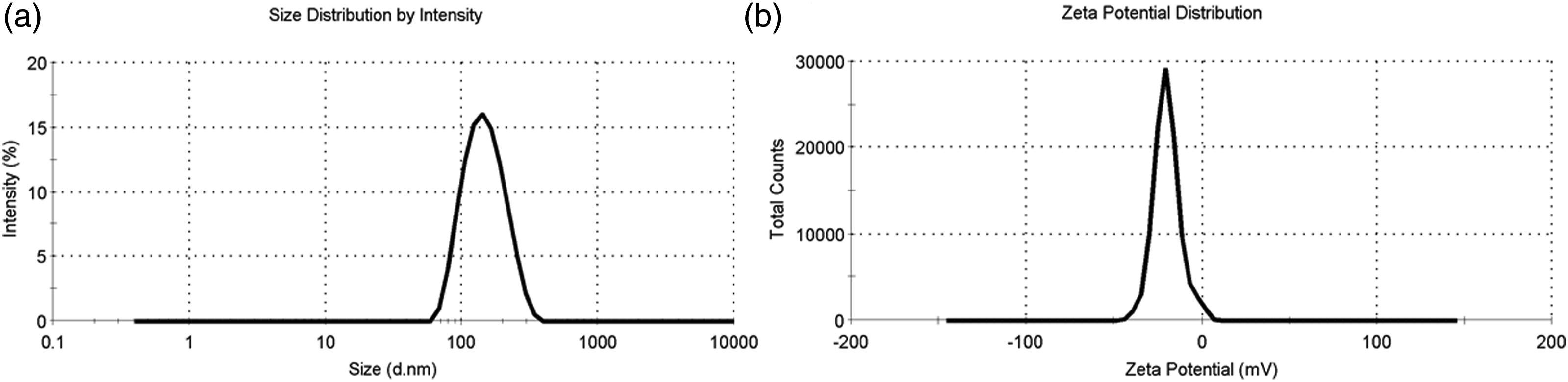
And the electrostaric repulsion will lead to the higher negative surface charges.
Transmission electron microscopy, differential scanning calorimetry and SAXS measurement of Ang-cubs-(GNA + PLHSpT)
The Ang-cubs-(GNA + PLHSpT) morphology was assessed by TEM. TEM observations showed that Ang-cubs-(GNA + PLHSpT) was approximately cube shape, slightly three-dimensional, with no adhesion between particles, and the average particle size of Ang-cubs-(GNA + PLHSpT) was near 200 nm (Figure 3(a)), which was consistent with the DLS results. In addition, a characteristic oval coffee-bean shape was obviously observed in Ang-cubs-(GNA + PLHSpT), indicating that model drugs of GNA and PLHSpT were not accumulated on the surface of the cubs rather than successfully encapsulated into nano-particles. (a): Transmission electron microscopy of Ang-cubs-(GNA + PLHSpT); (b): (a) DSC analysis of Ang-cubs-(GNA + PLHSpT); (b) cubs-GNA; (c) cubs-(GNA + PLHSpT); (d) GNA; (e) p407; (f) PLHSpT; (g) Angiopep-2; (h) GMO; (i) physical mixture. (c): The SAXS profiles of Ang-cubs-(GNA + PLHSpT).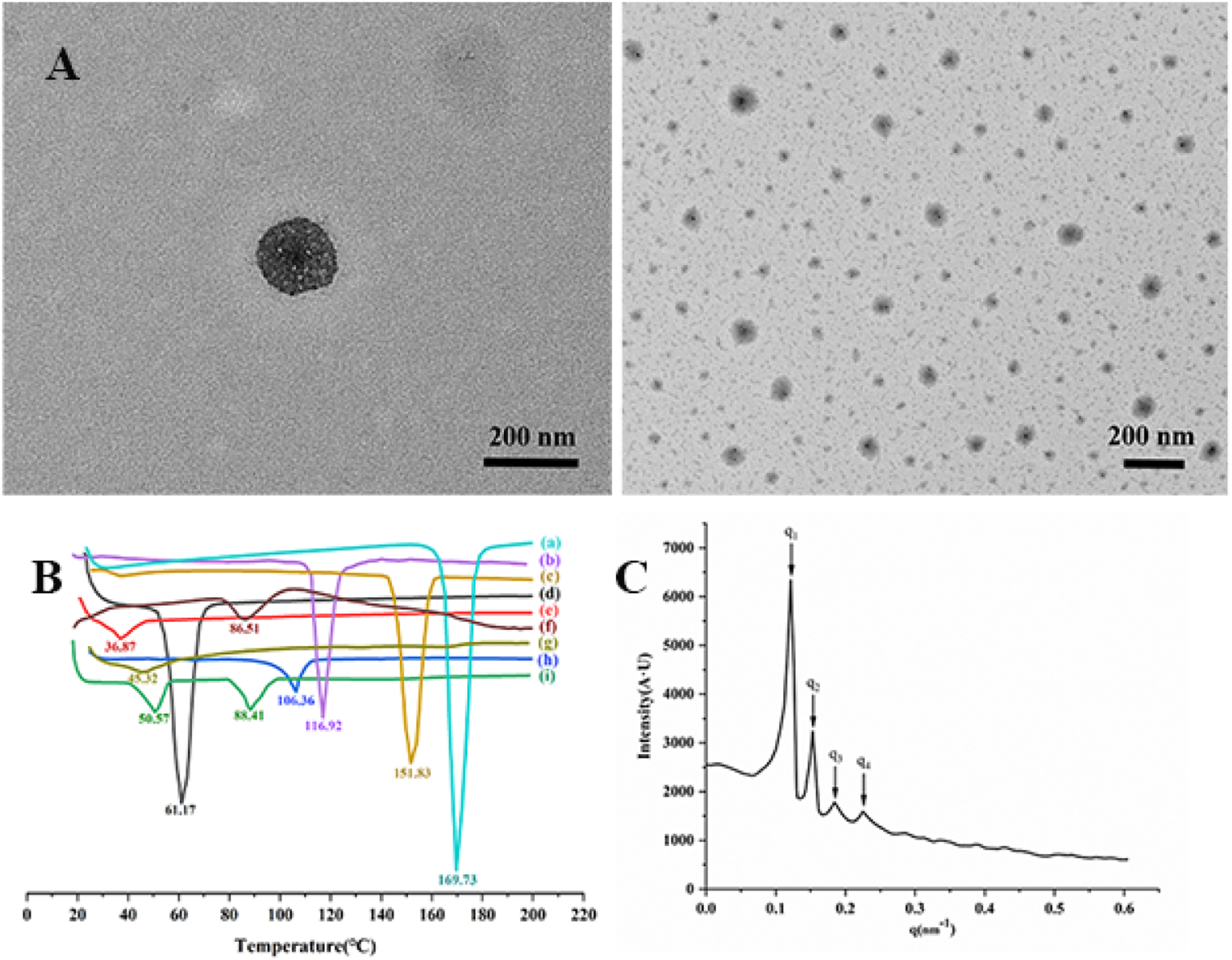
The Ang-cubs-(GNA + PLHSpT) was successfully prepared by emulsification evaporation and low-temperature solidification method. Under evaporation and solidification, a violent physical and chemical reaction occurred, which broke the chemical bonds of material and drug and formed new bonds between drug and excipient. The DSC curve could reflect thermodynamic properties of Ang-cubs-(GNA + PLHSpT). The Figure 3(b). reveals that there was a sharper characteristic peak at 61.17°C for GNA, blunt characteristic peak at 86.51°C of PLHSpT, respectively. At 36.87°C and 106.36°C showed relatively peak with p407 and GMO, respectively. However, the characteristic peaks of cubs-GNA and cubs-(GNA + PLHSpT) appeared at 151.83°C, 116.92°C, respectively. Besides, the physical mixture observed two characteristic peaks at 50.57°C and 88.41°C, respectively. The last but not least that a sharp peak at 169.73°C of Ang-cubs-(GNA + PLHSpT) was found. The characteristic peaks of the physical mixture and other composition disappeared, which further indicated that a new phase was formed of Ang-cubs-(GNA + PLHSpT).
The lattice analysis of Ang-cubs-(GNA + PLHSpT) was shown in Figure 3(c). The SAXS spectrum of Ang-cubs-(GNA + PLHSpT) has four main scattering peaks, which positions at 0.12, 0.15, 0.18, 0.22 nm−1, and the ratio was q1:q2:q3:q4 =
Determination of drug entrapment, loading efficiency and ligation efficiency of Ang-cubs-(GNA + PLHSpT)
The EE% and DL% are usually regard as indicators for evaluating the drug entrapment capability. The greater the values, the better entrapment of each drug in the nano-system. In our study, we used Ang-cubs to co-load hydrophilic PLHSpT and hydrophobic GNA. The EE% of GNA and PLHSpT was (89.04 ± 0.72)% and (81.5 ± 1.24)%, respectively. And (10.6 ± 0.85)%, (9.3 ± 0.45) % of DL% for GNA and PLHSpT, respectively. For GNA and PLHSpT, there was no statistical difference between EE% and DL%. The greater EE% and DL% values of GNA probably benefit from its high lipophilicity, which enabled effective encapsulation in hydrophobic nanocapsules core, obliging it to reside in hydrophobic nanocapsules and refuse migration to hydrophilic channel. Moreover, strong hydrogen bonds formed via hydroxyl group of GMO molecules with carboxyl group of angiopep-2, leading to its gelatinization at oil/water interface during cubs formation process. However, the higher EE% and DL% of PLHSpT might be attributed to higher adsorptive hydrophilic structure. It is also closely related to the abundant aromatic ring with PLHSpT, which could through π-π stacking to obtain a stable style in the hydrophilic channel.
In our study, the ligation efficiency of angiopep-2 with cubs-(GNA + PLHSpT) was examined by BCA method. The standard curve of the protein reference substance obtained by BCA protein quantification kit was: A = 0.0013 C + 0.2061. The ratio of cubs-(GNA + PLHSpT) with angiopep-2 was 10:1, the ligation efficiency of angiopep-2 was (43.11 ± 1.82)%. The result confirmed that targeting molecule of angiopep-2 has been attached to the surface of cubs, which have brain targeting functions in theory.
The cell viability assay of Ang-cubs-(GNA + PLHSpT) in vitro
The MTT assay was applied to investigate the cytotoxicity properties of free drug, blank cubs, cubs-GNA, cubs-PLHSpT, cubs-(GNA + PLHSpT) and Ang-cubs-(GNA + PLHSpT) against C6 cell, which was evaluated at various drug concentrations for 24 h. The Figure 4 clearly showed that blank cubs were non-toxic to C6 cell. However, the free drugs, drug-loaded cubs and decorated with angiopep-2 cubs exhibited obviously anti-tumor efficacy. The IC50 suggested that drug-loaded cubs showed significantly higher cytotoxicity compared with the free drug group. Moreover, dual drug-loaded cubs had better anti-tumor efficacy against C6 cell in dose-dependent manner, followed by the cubs-GNA and cubs-PLHSpT group, which may attribute to the synergistic effect of two drugs with different mechanisms co-encapsulated in cubs drug delivery system (Table 2). It is known that GNA and PLHSpT act on different stages for tumor cell mitosis. GNA can directly kill tumor cells in G0/G1 phase, while PLHSpT could block the growth of tumor cells in the G2/M phase. More importantly, ang-decorated cubs drug system exhibited the best inhibitory activity on C6 cell viability than that non-decorated cubs. Moreover, we confirmed the anti-tumor of the dual drug-loaded Ang-cubs system through in vitro cell studies and in vivo animal experiments. The effect of different concentrations of blank carrier and nano-carriers on the viability of C6 cell (Mean ± SD, n = 3). Particle size (a) and Zeta potential (b) of Ang-cubs-(GNA + PLHSpT). IC50 of GNA formulations (Mean ± SD, n = 3). PS: Indicates that the IC50 of cubs-(GNA + PLHSpT) and ang-cubs-(GNA + PLHSpT) are significantly different compared with GNA, **p < .01.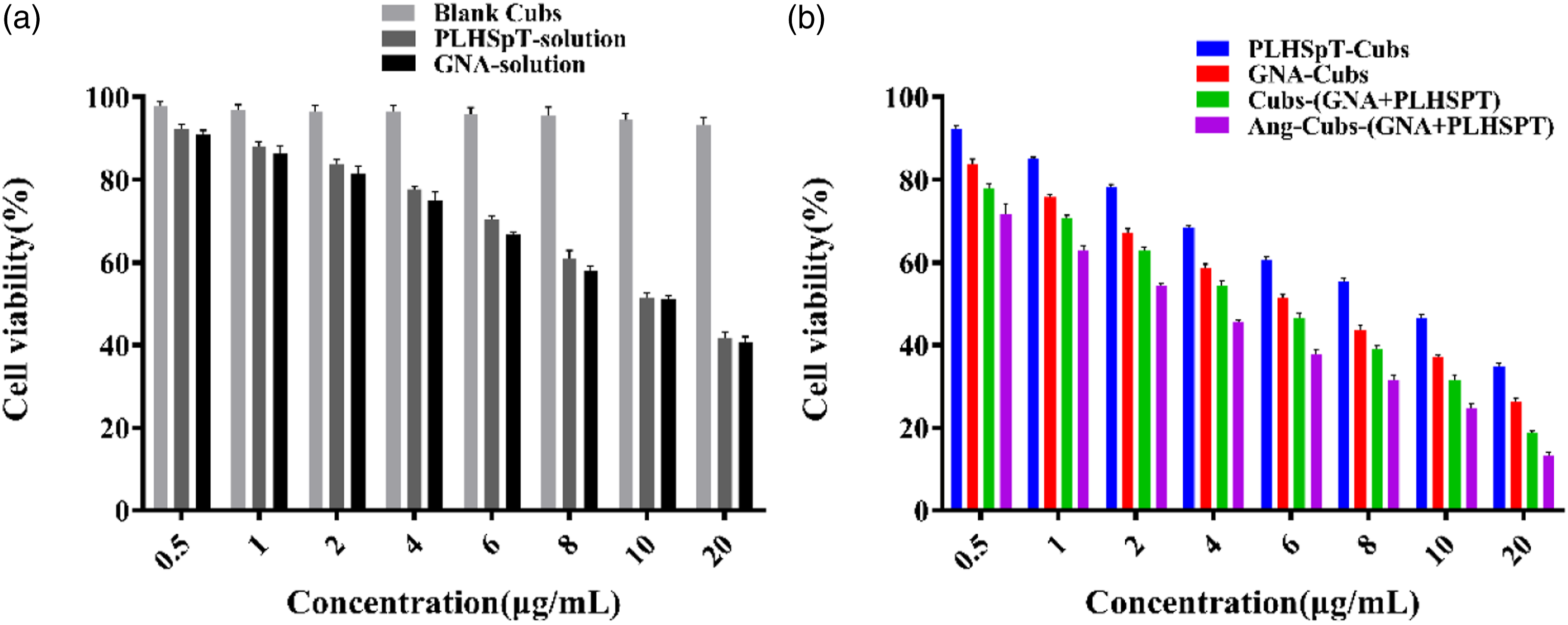

Ang-cubs-(GNA + PLHSpT)-induced cell apoptosis in C6 cell in vitro
It is known that cell apoptosis could be regarded as an important obstacle from damaged cell to malignant tumors. To evaluate the apoptosis mechanism, we used Annexin V-FITC double-stained C6 cell in our study. The relative apoptosis proportions of each type could be rapidly quantified by flow cytometry. Different concentrations (1.0,2.0,4.0) μg/mL of GNA-solution, PLHSpT-solution, cubs-PLHSpT, cubs-GNA、cubs-(GNA + PLHSpT), Ang-cubs(-GNA + PLHSpT) incubated C6 cell for 24 h. As Figure 5(a)and (b) shows that the apoptosis percentage of cubs-(GNA + PLHSpT) and Ang-cubs-(GNA + PLHSpT) group were greatly higher than cubs-GNA, cubs-PLHSpT, and also excellent than the free drug group. These data revealed that the apoptotic effects of cubs-(GNA + PLHSpT) and Ang-cubs-(GNA + PLHSpT) were stronger than the single drug delivery system, and Ang-cubs-(GNA + PLHSpT) significantly increased the cellular apoptosis in a concentration-dependent manner. In summary, these results again demonstrated that remarkable tumor cells apoptosis effect of the dual drug-loaded Ang-cubs delivery system. Annexin-V PI double staining to detect the induction of different concentrations of GNA-solution, PLHSpT-solution, cubs-GNA, cubs-PLHSpT, cubs-(GNA + PLHSpT), and Ang-cubs-(GNA + PLHSpT) in C6 cell at 24 h (a). C6 cell apoptosis by histogram analysis (b). p* < .05, p** < .01, p*** < .001.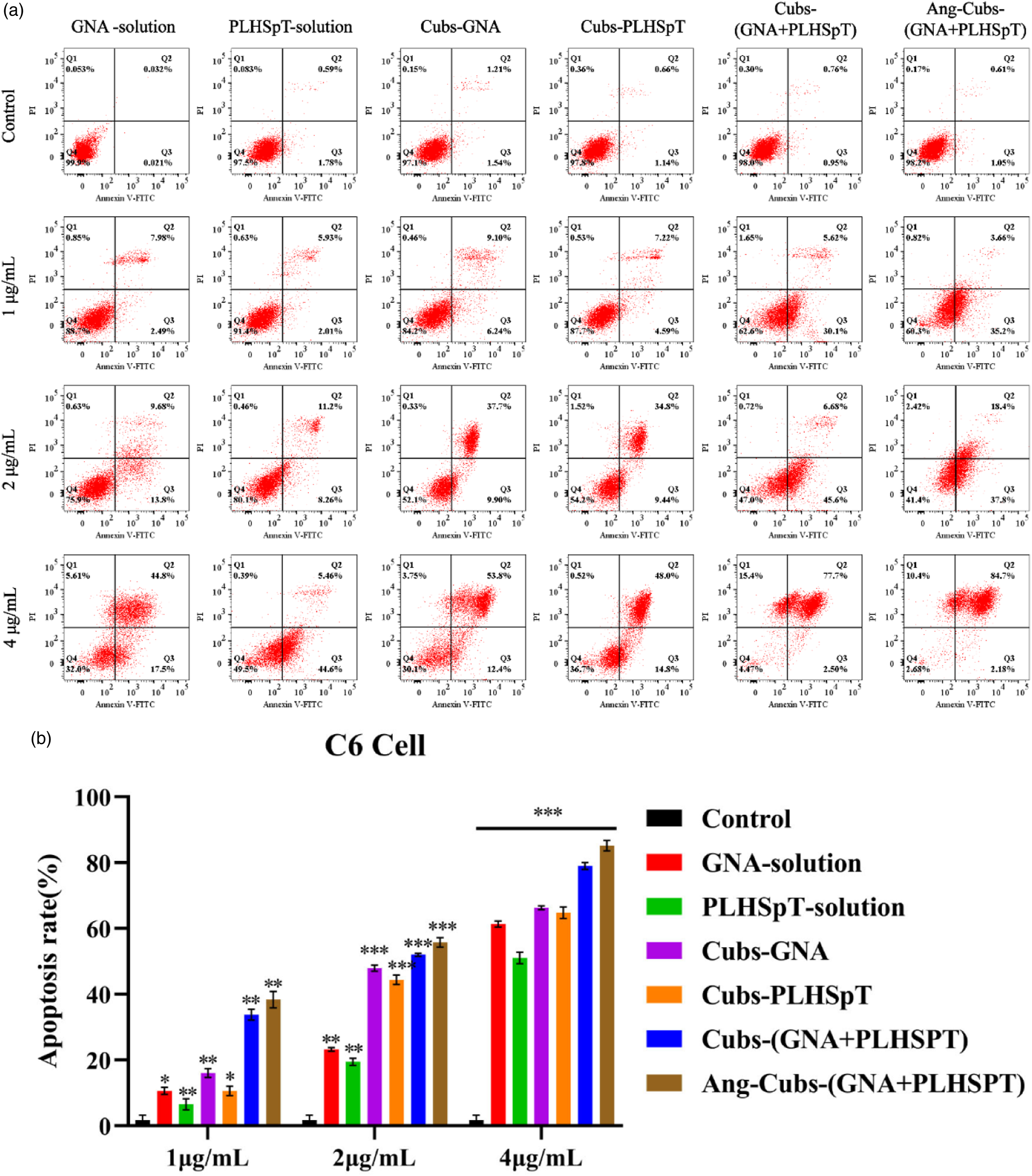
Ang-cubs-(GNA + PLHSpT) suppressed migration and invasion in vitro
To determine whether Ang-cubs-(GNA + PLHSpT) could suppress the aggressiveness of C6 cell, we performed a cell scratch experiment to intuitively evaluate cell migration and invasion. From Figure 6, it is demonstrated that C6 cell migrated and the scratch area shrank from 0 h to 24 h, whereas the migration distance treated with Ang-cubs-(GNA + PLHSpT) was significantly shorter than control cells. More importantly, the migration rates of cubs-(GNA + PLHSpT) and Ang-cubs-(GNA + PLHSpT) groups were lower than other groups within 24 h. The cell migration rates of each group were significantly different and the differences were reported to be statistically significant (p < .001). C6 cell (b) cell scratches (×400, 8bit) and cell migration results (Mean ± SD, n = 3, p* < 0.05, p** < 0.01, p*** < 0.001).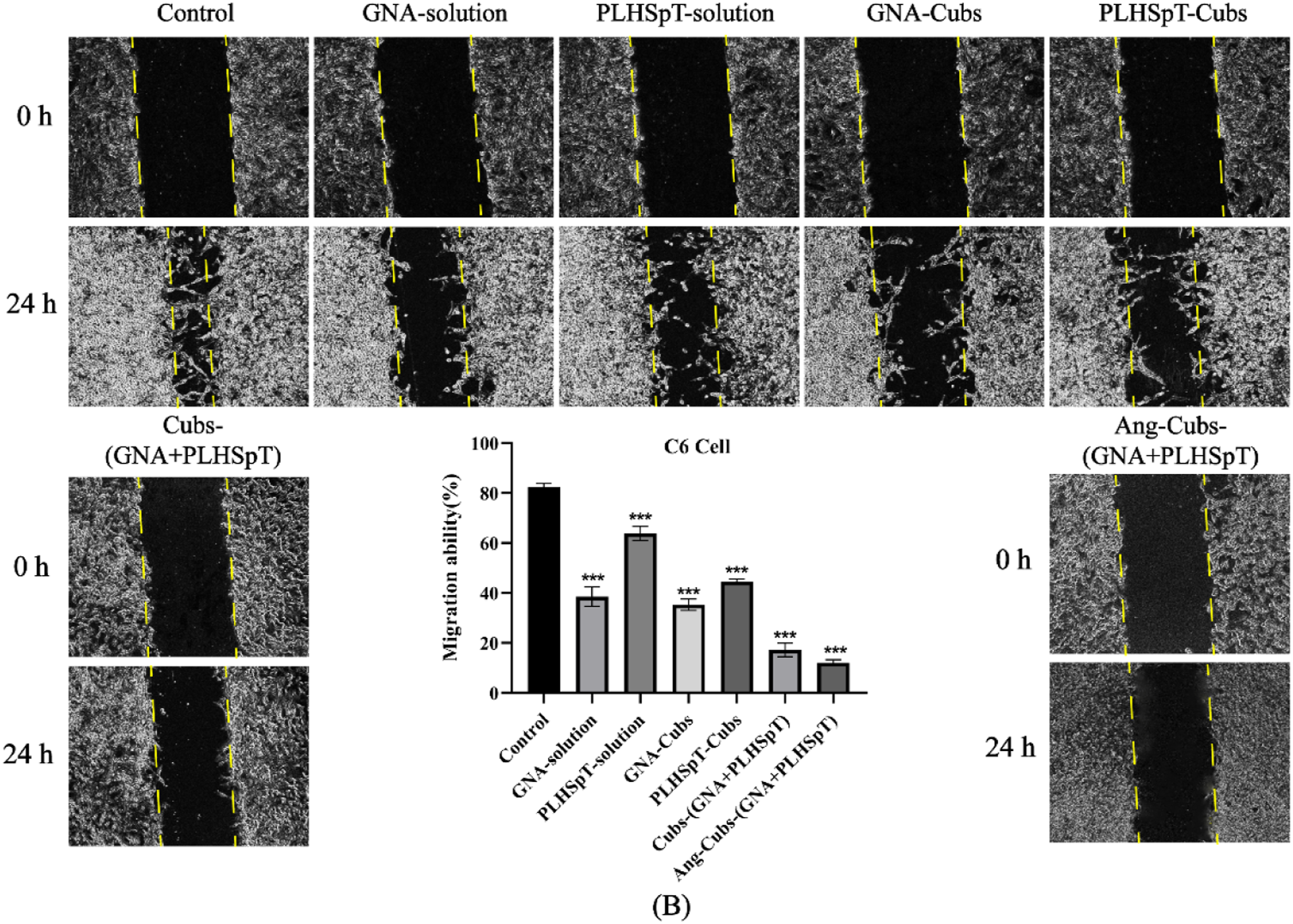
The penetrate blood-brain barrier model of Ang-cubs-(GNA + PLHSpT) in vitro
It is now necessary to assess the BBB penetration before fulfilling cellular uptake in vitro. As known, LRP-1 overexpression in BMECs and C6 cell, the co-culture system of BMECs-C6 was chosen as BBB model to study the brain-targeting effect for angiopep-2 functionalized-cubs in vitro (Figure 7(a)). Strong green fluorescence was observed of Ang-cubs-(GNA + PLHSpT) system in Figure 7(b). In other words, Ang-cubs-(GNA + PLHSpT) exhibited efficient penetration property compared with cubs-(Courmarin 6 +PLHSpT) group. Small amount of cubs-(Courmarin 6 +PLHSpT) could penetrate the BBB, which may be due to the smaller particle size of cubs-(Courmarin 6 +PLHSpT). This was a rather remarkable result which verified the fact that the novel penetration peptide of angiopep-2 significantly increased the transport ratio of cubs-(Courmarin 6 +PLHSpT) across the BBB into the glioma. BMECs-C6 cell transwell model (a); In vitro blood-brain barrier penetration ability of Coumarin 6-loaded nanoparticles (b).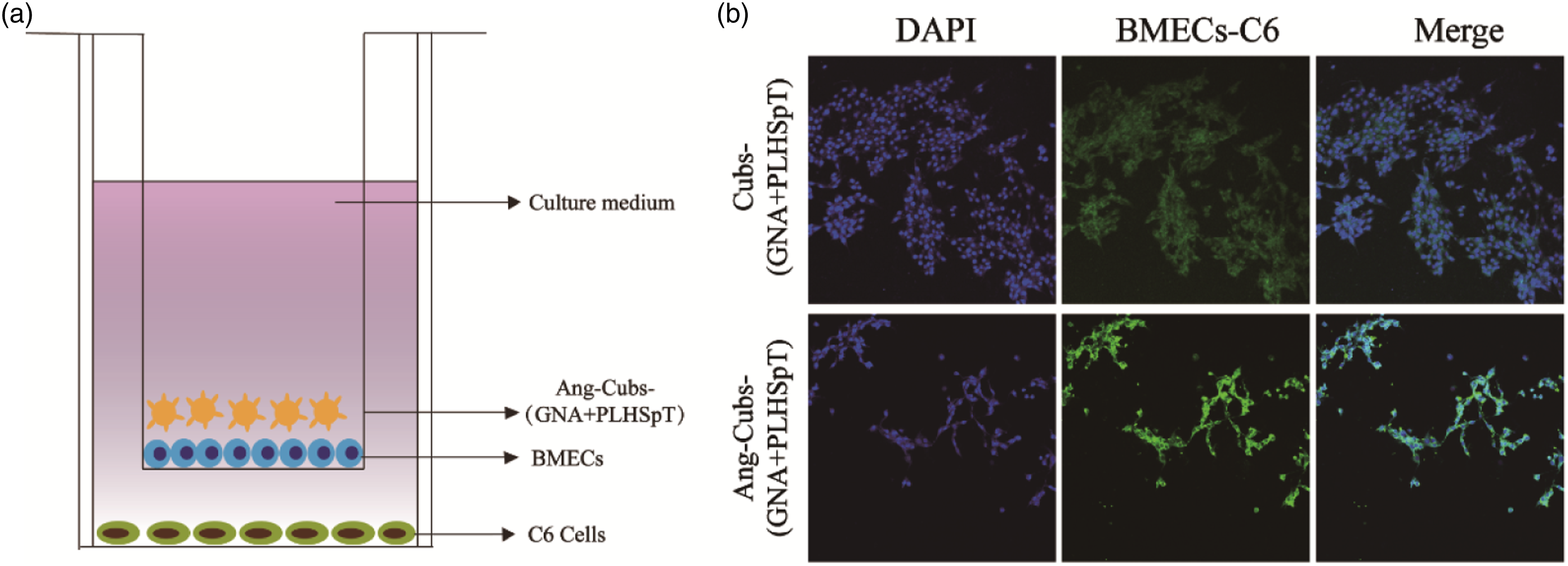
The cell uptake assessment of Ang-cubs-(GNA + PLHSpT) in vitro
To verify that the active targeting was specifically mediated by the LRP-1 receptor, we used confocal laser microscopy (CLSM) to investigate the in vitro cellular uptake behavior and intracellular location after angiopep-2 decorated cubs system in C6 cell. The C6 cell incubated separately with Courmarin 6-solution, cub-Courmarin 6, cub-(Courmarin 6+PLHSpT) and Ang-cubs-(Courmarin 6+PLHSpT) for 4 h. After 4 h incubation, the cell uptake of single drug delivery groups and unmodified cubs system were not ideal compared to Ang-cubs-(GNA + PLHSpT) according to the CLSM results in Figure 8. There was a smaller uptake in cub-Courmarin 6 and cub-(Courmarin 6+PLHSpT) group may be due to the smaller particle size rather than the penetration properties. In contrast, the abundance fluorescence intensity of C6 cell in Ang-cubs-(GNA + PLHSpT) was higher than that of other group, implying that the function of presence of ligand angiopep-2 effectively increased cubs cell uptake and enhances its brain-targeting abilities. Above all, the results of cell uptake strongly support our assumption that angiopep-2 was a very key factor, which determined cell recognition, accelerate cubs drug delivery system transport and promote cubs drug delivery system to exert anti-glioma effects. More importantly, based on the cell uptake of Ang-cubs-(GNA + PLHSpT) in vitro, it was valuable to explore the dual brain targeting for Ang-cubs-(GNA + PLHSpT) in vitro and in vivo. Cell uptake of C6 cell treatment with Courmarin 6, Courmarin 6, cubs-(Courmarin 6+PLHSpT), Ang-cubs-(Courmarin 6+PLHSpT).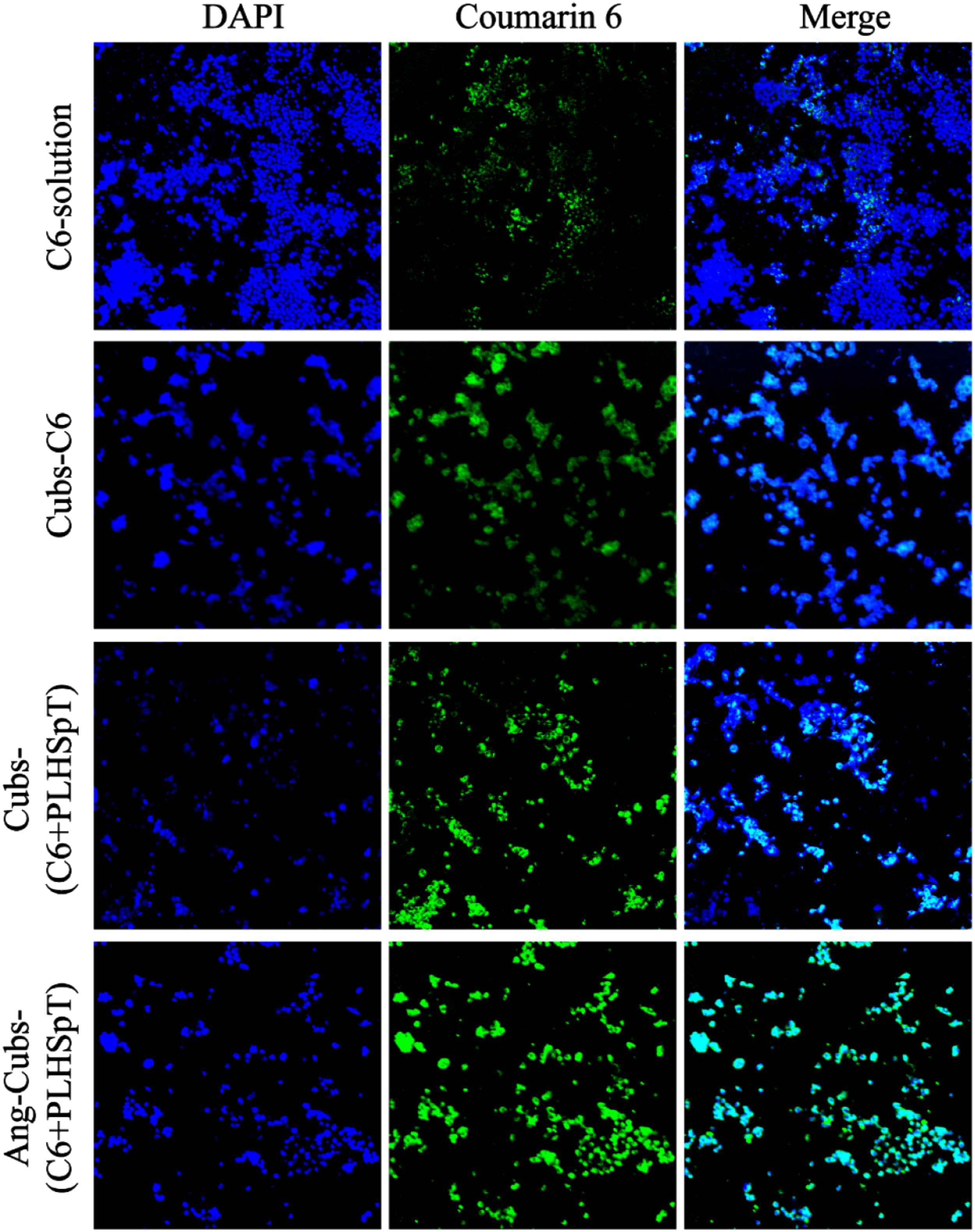
Brain targeting of ang-cubs-(GNA + PLHSpT) by live fluorescence imaging technology in vivo
To verify the BBB penetration and the brain targeting efficiency of Ang-cubs-(GNA + PLHSpT) in vivo, the fluorescence signals in brain were detected by IVIS. The fluorescence intensity in the brains of the DiR/Ang-cubs group was detected at initial time 1 h and reached peak at 24 h as depicted the real-time in vivo images (Figure 9). It was also concluded that the Ang-cubs delivery system significantly increased cell penetration in a time-dependent manner. In contrast, the brain of rat injected with cubs-(Dir + PLHSpT) showed completely no fluorescence, which was mainly distributed among macrophage-related organs, such as the liver and the spleen. The results indicated that the angiopep-2-decorated cubs drug delivery system effectively targets the brain. Those results were consistent with cellular uptake assay in vitro. Fluorescence distribution in rats after (2,8,12,24) h of tail vein injection of cubs-(DiR + PLHSpT) and Ang-cubs-(DiR + PLHSpT) (Mean ± SD, n = 3).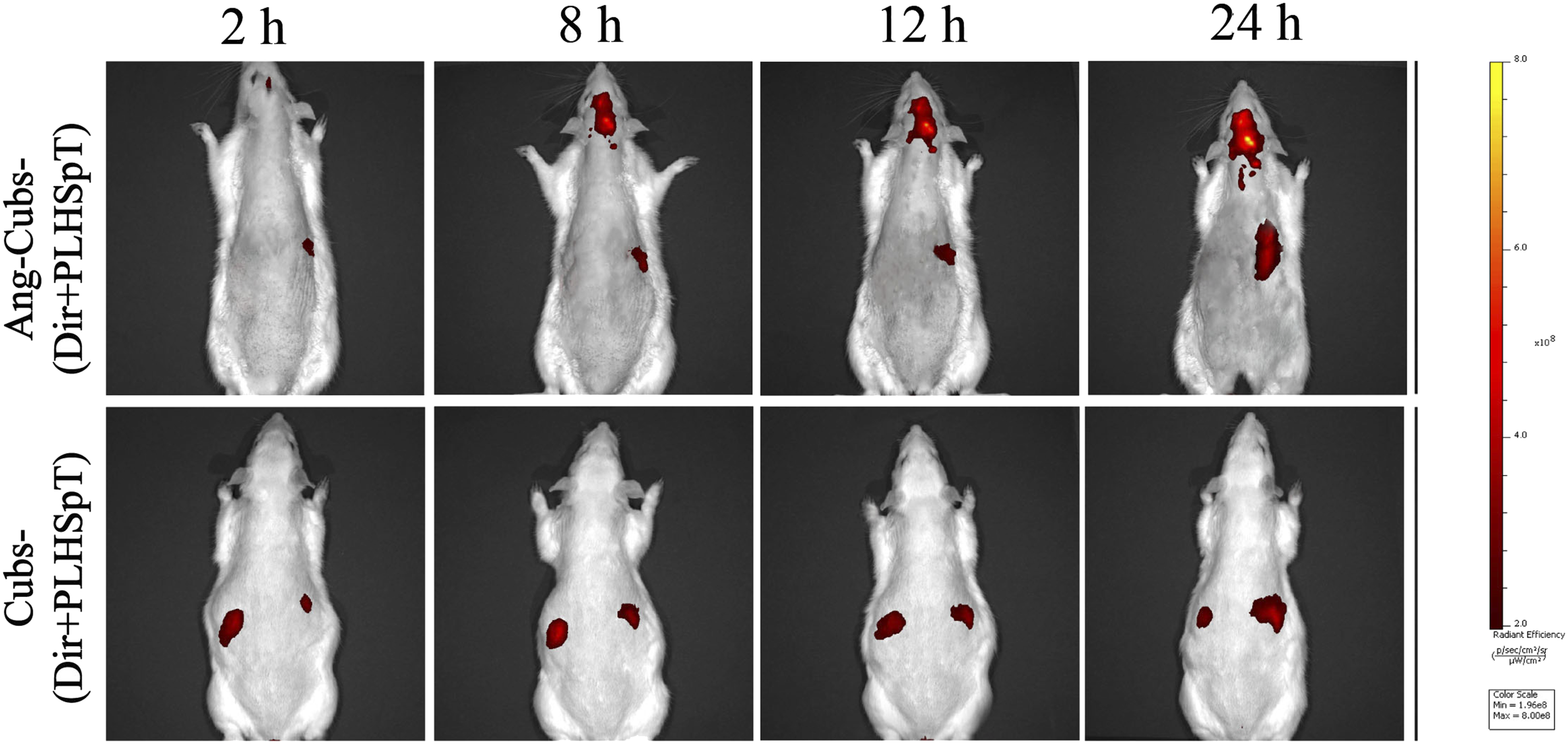
In vivo antitumour efficacy of Ang-cubs-(GNA + PLHSpT)
It is known that the most important aim of drug discovery is to output drugs with high anti-cancer efficacy and low toxicity. Considering the favorable in vitro anti-tumorigenesis and effective brain tumor targeting by Ang-cubs-(GNA + PLHSpT). We further evaluated the in vivo anti-tumor activity for Ang-cubs-(GNA + PLHSpT) in the orthotopic GBM model. Seven days after C6 cell implantation, glioma rats were randomly groups (see details 2.15). Glioma rats were tail vein injection at a dose of 2.0 mg/kg with saline, GNA-solution and cub-(GNA + PLHSpT) as well as Ang-cubs-(GNA + PLHSpT) on alternate dates for a period of 10 times according to treatment schedule. We recorded the median survival and behavioral performance of each group of glioma rats within 30 days.
The glioma rats in the normal saline group showed the most prominent behavioral performance, with reduced foraging behavior, staggered walking and sluggishness as well as hemiplegia in the later stage, and their median survival time was only 15 days. The median survival results, as shown in Figure 10(a), indicate that there was a slight increase of median survival in GNA-solution and cub-(GNA + PLHSpT) group. The most striking aspect of this graph was the median survival of Ang-cubs-(GNA + PLHSpT) treatment was significantly prolonged compared to other treatment group. The order of median survival was as follows: Ang-cubs-(GNA + PLHSpT) (29 days) > cub-(GNA + PLHSpT) (27 days) > GNA-solution (22 days) > saline (15 days). There was a statistical difference between the control group and the treatment group (p < .05). (a): Median survival time of glioma rats in saline, GNA-solution, cubs-(GNA + PLHSpT) and Ang-cubs-(GNA + PLHSpT) groups (Mean ± SD, n = 8). (b): Body weight change curve of glioma rats after administration of saline, GNA-solution, cubs-(GNA + PLHSpT) and Ang-cubs-(GNA + PLHSpT) for 10 days, respectively (Mean ± SD,n = 8). p* < 0.05, p** < 0.01, p*** < 0.001.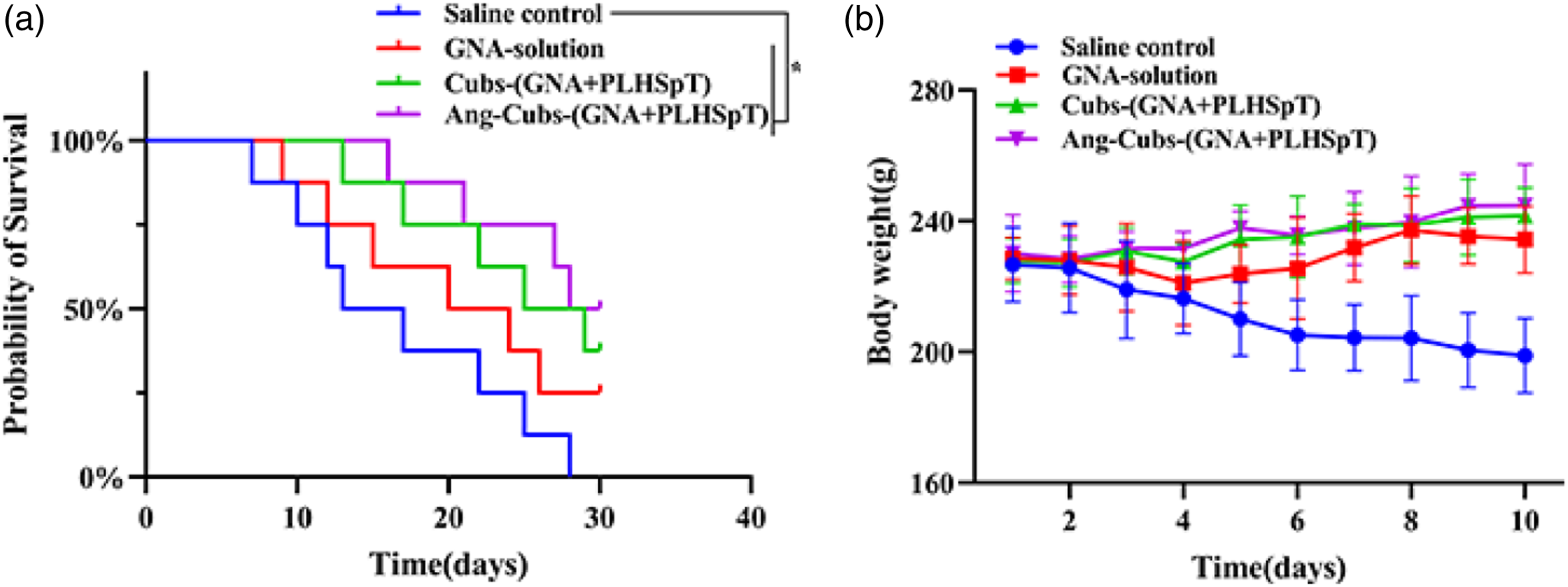
Moreover, body weights of glioma rats in the different treatment group was conducted. The body weight of glioma rats from the nano-particle conditioning drug delivery system increased slightly in the study period. However, there was steadily decreased in control group, suggesting the cubs and novel Ang-cubs drug delivery system produced less systematic toxicity (Figure 10(b)). Thus, in vivo results definitely indicated that Ang-cubs-(GNA + PLHSpT) significantly reduced glioma rats burden and improved median survival time. However, it is necessary to further confirmed their reliability for patients in subsequent clinical experiments before clinical practice.
Conclusion
In summary, we developed BBB-penetrating peptide (angiopep-2) modified novel dual-targeted drug delivery system to co-delivery hydrophobicity GNA and hydrophilic PLHSpT for anti-glioma therapy. Our successfully designed Ang-cubs-(GNA + PLHSpT) exhibited ideal particle size with a narrow size distribution, spherical shape with a surrounding corona on the surface, no adhesion between particles, high encapsulation efficiency and loading efficiency, stable Zeta potential and sustained drug release. Compare with the non-angiopep-2 decorated cubs delivery system and single drug-loaded cubs system, Ang-cubs-(GNA + PLHSpT) exhibited significantly cellular uptake, cell apoptosis and profound tumor inhibition ability in vitro. More importantly, Ang-cubs-(GNA + PLHSpT) could delay weight loss, inhibited tumor growth and prolonged the median survival time, superior BBB penetrating efficiency with orthotopic GBM model in vivo anti-tumor experiments. In conclusion, our research suggested that novel Ang-cubs-(GNA + PLHSpT) regarded as a dual-targeting drug delivery system for GBM treatment.
Supplemental Material
Supplemental Material - Novel dual targeting cubosomes modified with angiopep-2 for co-delivery GNA and PLHSpT to brain glioma
Supplemental Material for Novel dual targeting cubosomes modified with angiopep-2 for co-delivery GNA and PLHSpT to brain glioma by Tongyuan Lin, Qing Wei, Huamin Zhang, Yong Yang, Bo Jiang, Zhangyi Wang, Siyuan Li, Qiang Wang, Mengru Hu, Weidong Chen, Lei Wang and Baijing Ding in Journal of Biomaterials Applications
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.Tongyuan Lin and Qing Wei contributed equally to this work. Wei dong Chen, Lei Wang, Bai Jing Ding is corresponding author.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the scientific research project of Anhui Provincial Health Commission in 2021 (Grant/Award Number: AHWJ2021b045). The key Research Projects of Wuhu Science and Technology Bureau in 2021 (Grant/Award Number: 2022yf69), The Anhui Provincial Department of Education Natural Science Research Key Program (Grant/Award Number: KJ2019A1082) and The key Research and Translation Projects of Wuhu Science and Technology Bureau in 2022 (2023bc0126).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.