Abstract
Background
Commercial fibrin glue is increasingly finding its way into clinical practice in surgeries to seal anastomosis, and initiate hemostasis or tissue repair. Human biological glue is also being discussed as a possible cell carrier. To date, there are only a few studies addressing the effects of fibrin glue on the cell-molecular level. This study examines the effects of fibrin glue on angiogenesis and lymphangiogenesis, as well as adipose-derived stem cells (ASCs) with a focus on gene and protein expression in scaffolds regularly used for tissue engineering approaches.
Methods
Collagen-based dermal regeneration matrices (DRM) were seeded with human umbilical vein endothelial cells (HUVEC), human dermal lymphatic endothelial cells (LECs), or adipose-derived stem cells (ASC) and fixed with or without fibrin glue according to the experimental group. Cultures were maintained for 1 and 7 days. Finally, angiogenic and lymphangiogenic gene and protein expression were measured with special regard to subtypes of vascular endothelial growth factor (VEGF) and corresponding receptors using Multiplex-qPCR and ELISA assays. In addition, the hypoxia-induced factor 1-alpha (HIF1a) mediated intracellular signaling pathways were included in assessments to analyze a hypoxic encapsulating effect of fibrin polymers.
Results
All cell types reacted to fibrin glue application with an alteration of gene and protein expression. In particular, vascular endothelial growth factor A (VEGFA), vascular endothelial growth factor B (VEGFB), vascular endothelial growth factor C (VEGFC), vascular endothelial growth receptor 1 (VEGFR1/FLT1), vascular endothelial growth receptor 2 (VEGFR2/KDR), vascular endothelial growth receptor 3 (VEGFR3/FLT4) and Prospero Homeobox 1 (PROX1) were depressed significantly depending on fibrin glue. Especially short-term fibrin effect led to a continuous downregulation of respective gene and protein expression in HUVECs, LECs, and ASCs.
Conclusion
Our findings demonstrate the impact of fibrin glue application in dermal regeneration with special regard to angiogenesis and lymphangiogenesis. In particular, a short fibrin treatment of 24 hours led to a decrease in gene and protein levels of LECS, HUVECs, and ASCs. In contrast, the long-term application showed less effect on gene and protein expressions. Therefore, this work demonstrated the negative effects of fibrin-treated cells in tissue engineering approaches and could affect wound healing during dermal regeneration.
Keywords
Introduction
Fibrin glue is more and more widely used in clinical practice. 1 The formation of fibrin polymers is an ingenious trick of nature and human bodies. To prevent bleeding from injuries, our blood has a coagulation system that quickly closes the vascular defect locally without impairing perfusion.2,3 A cascade is set in motion which ultimately leads to the activation of two essential precursor factors in our blood. The first factor is called thrombin, a Na (+)-activated allosteric serine protease that plays multiple functional roles in blood pathophysiology and is synthesized in the liver. 4 The second, fibrinogen, is a large, complex, fibrous glycoprotein with three pairs of polypeptide chains linked together by 29 disulfide bonds. Fibrinogen is produced in the liver and abundantly secreted into the blood plasma, where it is a ubiquitous molecule. 5 The association of fibrinogen with thrombin creates a naturally occurring, biodegradable three-dimensional scaffold of fibrin polymers with unique properties, hence, being one of the key molecules in blood clot formation.2,3,6 This soft fibrin mesh is further stabilized by the transglutaminase factor XIIIa resulting in the formation of a stable blood clot against mechanical, chemical, and proteolytic insults. 5 Bergel et al. used fibrin first as a hemostat in 1909. 7 Several medical device manufacturers have succeeded in developing the isolation process of both factors from plasma and bringing them onto the market as a medical product. Furthermore, the Food and Drug Administration (FDA) approved a liquid fibrin sealant in 1998 and a fibrin sealant patch in 2010. 7 Fibrin glue can be fabricated from pooled or autologous human plasma. 6 Although it is an expensive medical device, for many users the advantages outweigh the disadvantages. The advantages comprise a simple application with the desired effect occurring within a few minutes. Besides, it is humanely compatible and, in many cases, shortens the operation time.8,9 Fibrin glue is clinically approved as a natural biomaterial that creates a promising sealing effect on dura mater, skin tissue, nerves, or vessels, respectively. It is widely used in surgical practice to seal small wounds,10,11 to repair bones1 12 or to perform nerve coaptations by entirely wrapping nerve transplants,13,14 to secure micro-anastomoses of blood vessels to create hemostasis and control bleeding,15,16 to treat lacerations of parenchymatous organs1 17 and to prevent leakage from colonic anastomoses.1,18 As recombinant fibrin glue evokes only minimal inflammatory response, it has been considered and tested successfully as a potential cell carrier matrix.19,20 Fibrin glue is widely used in tissue engineering approaches with inconsistent and partly contradictory results on wound healing. On the one hand supportive and on the other hand inhibitory effects on tissue regeneration are reported.6,8,21
However, to date, it is not much compelling evidence in the literature, supporting the effects of fibrin glues, necessitating a close study of fibrin glue application and its effect on the cell environment. Therefore, cells were seeded onto collagen-based scaffolds with or without fibrin glue application. During the physiological wound healing process, fibrin is ultimately degraded by proteases, so the experimental observation period extends from 24 hours to 7 days to identify possible differences. This study aims to address the effect of conventional fibrin glue on gene and protein level changes in cells that play a critical role in wound healing. Angiogenesis and lymphangiogenesis are examined on a molecular level by Multiplex-qPCR and ELISA. Included are human venous endothelial cells (HUVEC), lymphatic endothelial cells (LECs), and adipose-derived stem cells (ASC). ASCs play an important role in wound healing. The pluripotent stem cells with the ability to differentiate into different lineages and to secrete paracrine factors initiating the tissue regeneration process serve as an ideal cell type to use in therapies for the treatment of non-healing wounds. 22 LECs form the lymphatic vasculature with the task of controlling the interstitial microcirculation and removing from the extravascular space macromolecules and particles that are too large to re-enter the blood capillaries. In addition, lymphatic vessels play a critical role in immunological processes through the egress of T lymphocytes and Langerhans cells. 23 HUVEC have autocrine and paracrine functions on wound healing and achieve the restoration of perfusion conditions by establishing a vascular system.24,25
Materials and methods
Cell culture of ACSs, LECs, HUVECs
The human adipose-derived stem cells (ASC) were isolated from the subcutaneous fat tissue of a healthy donor. Cells were cultured in DMEM (Gibco, Thermo Fisher Scientific, Waltham, MA, USA), supplied with 10% FBS (fetal bovin serum, gibco, Invitrogen) 1% amphotericin B (Biochrom GmbH, Berlin, Germany) and 1% Penicillin-Streptomycin (Biochrom GmbH, Berlin, Germany). The human Dermal Lymphatic Endothelial Cells (LEC) isolated from juvenile foreskin were purchased from PromoCell in passage 2. Cells were cultured in Endothelial Cell Growth Medium MV (ECGM MV, PromoCell, Heidelberg, Germany) supplied with a supplement mix (PromoCell, Heidelberg, Germany), 1% amphotericin B (Biochrom GmbH, Berlin, Germany) and 1% Penicillin-Streptomycin (Biochrom GmbH, Berlin, Germany). The human Umbilical Vein Endothelial Cells (HUVEC) were purchased from PromoCell in passage 2. Cells were cultured in Endothelial Cell Growth Medium MV (ECGM MV, PromoCell, Heidelberg, Germany) supplied with a supplement mix (PromoCell, Heidelberg, Germany), 1% amphotericin B (Biochrom GmbH, Berlin, Germany) and 1% Penicillin-Streptomycin (Biochrom GmbH, Berlin, Germany). Co-cultures of ASCs with LECs or HUVECs were grown in Endothelial Cell Growth Medium MV (ECGM MV, PromoCell, Heidelberg, Germany).
All cell types were incubated at 37°C, 5% CO2 in a standard cell culture incubator (Thermo Fisher Scientific, Waltham, MA, USA). The medium was changed every 2–3 days. Cell number was determined using Countess 2 FL (Invitrogen, Carlsbad, CA, USA). All experiments were performed in triplicates with LECs and HUVECs of the same batch in passages 3–5, and ASCs of the same donor in passage 2.
Cell isolation and characterization of ASCs
Adipose-derived stem cells (ASCs) were harvested intraoperatively by tumescent liposuction or as tissue preparations during abdominal dermolipectomy or tummy tuck from suitable healthy donors at the Department of plastic, reconstructive and aesthetic surgery of the Ludwig-Maximilians-University Munich. All patients obtained written informed consent prior to the operations. The study was conducted in accordance with the guidelines of the Declaration of Helsinki and approved by the institutional ethics committee with registration number 17–046. The isolation protocol from Bunnell et al.2 26 was modified by not using a lysis buffer. The lipoaspirate or the adipose tissue was mixed in a ratio of 2:1 with collagenase II 0.15% (Worthington Biochemical Corp., Lakewood, NJ, USA). This was followed by incubation for 35 min at 37°C. The samples were shaken every 10 minutes. The enzyme activity was stopped by adding and mixing 5 mL of cultivation medium DMEM per tube. The suspension was centrifuged at 1200 G for 10 min. After forming three phases in the tube, the supernatant was aspirated to a residual volume of 5 mL and discarded. The remaining volume was resuspended with 3 mL of cultivation medium DMEM and filtered through a cell strainer. In order to increase the cell yield, a further 5 mL of cultivation medium (DMEM) was used for rinsing. The Falcon was centrifuged at 300 G for 5 min and decanted to a residual volume of 5 mL. The cell suspension obtained was expanded in standard culture medium DMEM, supplemented with 10% fetal bovine serum (FBS; Gibco, Thermo Fisher Scientific, USA), 1% Pen/Strep (A2213, Biochrom, Germany), and 1% Amphotericin B (A2612, Biochrom, Germany), as described above. To ensure that the cells isolated from the lipoaspirate were ASCs, their multilineage potential was confirmed by differentiating into adipogenic, osteogenic, and chondrogenic cells with StemMACS™ differentiation medium (Miltenyi Biotec, Germany).
Scaffold preparation and cell-seeding
Bilayer collagen scaffolds (IDRT, Integra© Matrix Life Science Cooperation, Plainsboro, NJ, USA) were prepared using a Ø12 mm biopsy punch (Pico Punch® P1225, Acuderm® inc., Ft. Lauderdale, FL, USA). The resulting scaffolds were separated into two groups with and without fibrin fixation. Scaffolds for fibrin fixation were air-dried for 20 min on sterile gauze and then placed in a 12-well plate. For fibrin fixation, cell suspensions were mixed with fibrinogen (TISSEEL, Baxter GmbH, Unterschleißheim, Germany) in a ratio of 1:1. Then, 50 µl thrombin solution (TISSEEL, Baxter GmbH, Unterschleißheim, Germany) were pipetted onto the scaffold, followed by 100 µl of the cell-media-fibrinogen solution to achieve a final concentration of 1 × 106 cells per scaffold and air dried it for 1 h. Scaffolds without fibrin treatment were placed in a 12-well plate and incubated for 1 hour at 37°C and 5% CO2 with 1 mL of growth medium. For seeding, a cell suspension with 50 × 106 cells/ml was prepared with the respective growth medium and 20 μL was added to a final concentration of 1 × 106 cells per scaffold. Representative cross-sections (AxioObserver, Carl Zeiss, Jena, Germany) and overview pictures of modified fibrin scaffolds were taken. Scaffolds without fibrin treatment served as a control group. The scaffolds were prepared as monocultures with LECs, HUVECs, or ASCs respectively. For building co-cultures ASCs were merged in a ratio of 1:1 with either LECs or HUVECs by retaining the final concentration of 1 × 106 cells per scaffold. The resulting scaffolds were placed in 12-well plates and cultured with 2 mL of the respective growth medium covering the scaffold for a further 30 min at 37°C and then stored at 37°C and 5% pCO2 for 4 hours. An additional monolayer control group with ASCs, using 2D cultures of each cell type instead of the scaffold, was prepared to provide a safe cell base. After 4 hours plates were incubated under normoxic conditions for 24 hours or 7 days respectively. A schematic presentation of the experimental workflow is pictured in Figure 1. Schematic representation of the experimental workflow: Living adipose-derived stem cells, endothelial and lymph endothelial cells were seeded into 3D collagen-based scaffolds and fixed either with or without fibrin glue. The control group was performed with the abandonment of a scaffold application to evaluate the effect of a 3D cell architecture. Seeded scaffolds were incubated under normoxic conditions (21% pO2) for 1 and 7 days, respectively. Upon incubation, fibrin glue affects lymphangiogenic and angiogenic protein and gene expression by inducing the downregulation of corresponding growth factors or receptors.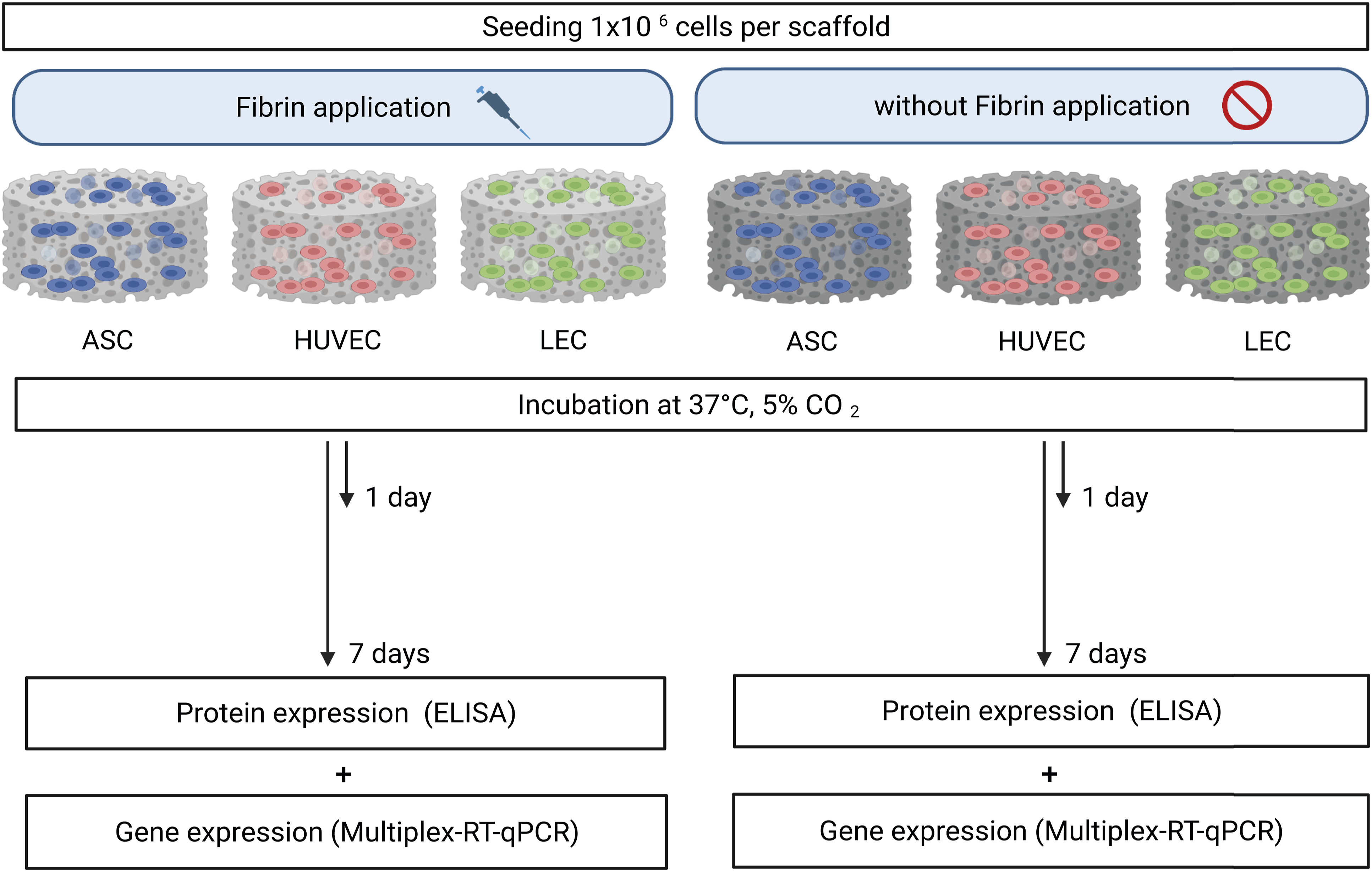
Ribonucleic acid (RNA)-extraction
For each experimental condition, 1 × 106 cells were seeded on scaffolds and cultured as described previously in triplicate for the desired duration of time. RNA extraction was performed using the RNAeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. After the respective time of incubation, for harvesting, scaffolds were collected in 2 mL Eppendorf tubes and lysed twice for 10 min at 750 rpm and 23–25°C in a thermocycler (both from Eppendorf, Hamburg, Germany) containing lysis buffer that was provided by the kit and supplemented with 1% beta-mercaptoethanol (10 μL/mL). Samples were then centrifuged for 3 min at 14,000 rpm and the supernatant was transferred to a collection tube. The amount, ratio and purity of the RNA were assessed photometrically at 260/280 nm wavelength with an Infinite TM plate reader (Infinite 200 Pro, TECAN, Männedorf, Switzerland) equipped with a NanoQuant plate (TECAN, Mänersdorf, Switzerland).
Multiplex-quantitative polymerase chain reaction (qPCR)
Primer sequences and probe design (5′-3′).
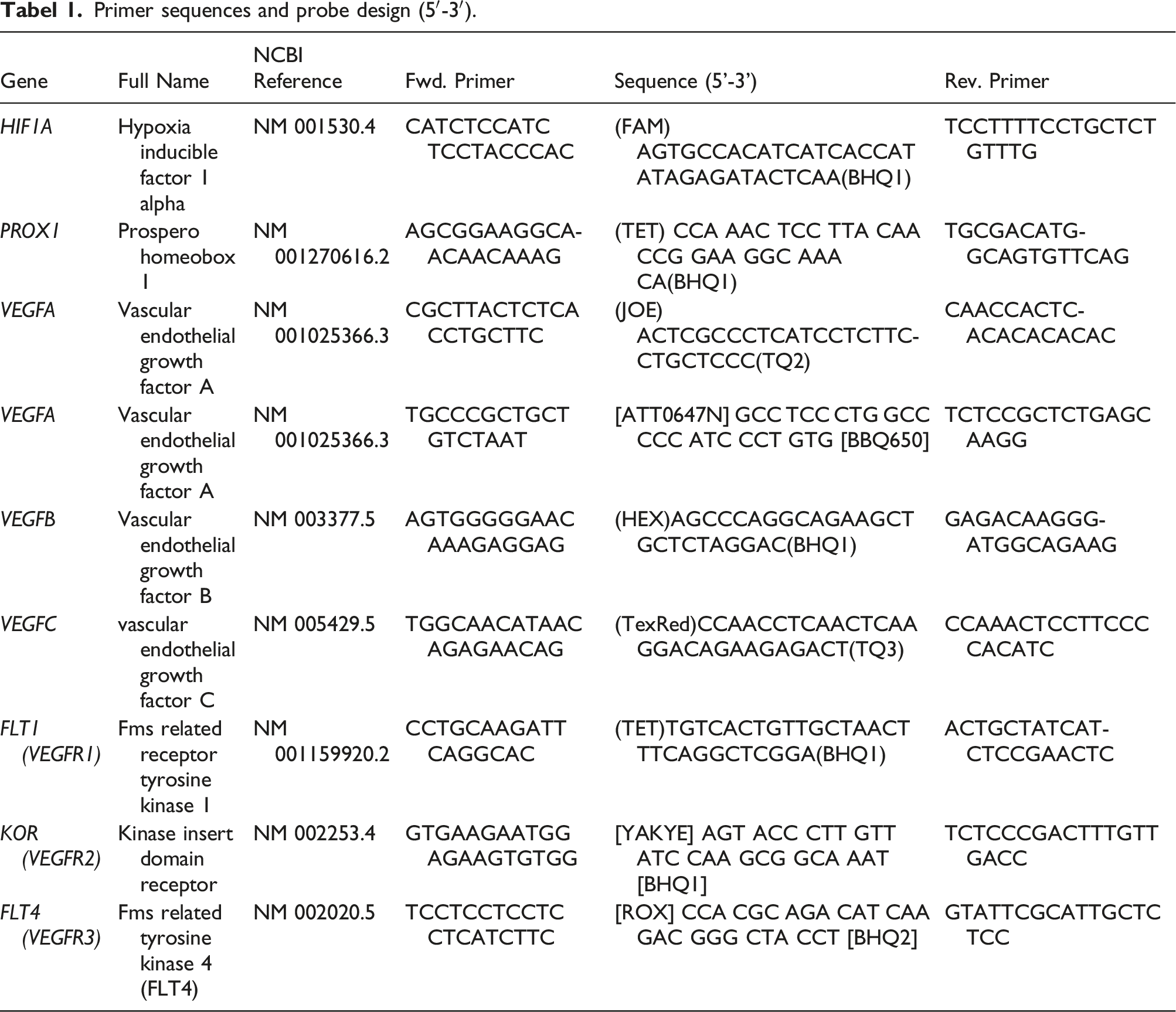
Amplification conditions.

Enzyme-linked immunosorbent assay (ELISA)
Summarized proteins and ELISA Kits.

Statistical evaluation and graph illustrations
All data were tested for Gaussian distribution by using the Shapiro-Wilk test and by visual inspection of normal q-q plots before performing a t-test. If data were normally distributed a student’s t-test was performed for comparison between two different groups. Differences among groups were considered significant if p ≤ 0.05 (ns: not significant; *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001). All results are presented as mean ± standard deviation (SD). Schematic representations were created using the platform www.BioRender.com
Results
Fibrin Glue is a human-compatible composite material that results in the rapid sealing of bleeding, tissue layers, or wounds. Because of these properties, it is widely used in clinical practice. In this work, the sealing effect is examined for possible adverse effects on wound healing after 24 hours and 7 days with a focus on angiogenic and lymphangiogenic regeneration. We evaluated in several cell types the effect of a fibrin-encapsulation on gene expression regarding genes involved in angiogenesis and lymphangiogenesis, namely vascular endothelial growth factor A (VEGFA), vascular endothelial growth factor B (VEGFB), vascular endothelial growth factor C (VEGFC), vascular endothelial growth receptor 1 (VEGFR1/FLT1), vascular endothelial growth receptor 2 (VEGFR2/KDR), vascular endothelial growth receptor 3 (VEGFR3/FLT4) and Prospero Homeobox 1 (PROX1). In addition, the hypoxia content due to the possible airtight sealing of fibrin coating was analyzed, using hypoxia-inducible factor 1 alpha (HIF1a) gene expression in enclosed cells.
Reduced angiogenic and lymphangiogenic gene and protein expression levels by fibrin glue application in scaffolds after 24 hours
First, we aimed at analyzing the effect of the use of fibrin glue on tissue regeneration in dermal replacement material, regarding angiogenesis and lymphangiogenesis. Therefore, cells were seeded on collagen matrices as described above and fixed either with (w.F.) or without fibrin glue (wo.F.). The workflow is obtained in Figure 1. Seeded scaffolds were incubated in standard human cell culture conditions at 37°C for 24 hours (Figure 1). The cross-sectional and the overview image of scaffolds with fibrin application showed a gradual distribution of fibrin glue with almost no air pockets, suggesting an airtight seal (Figures 2(a) and (b)). Scaffolds were harvested after 24 hours to evaluate short-term effects on the angiogenic and lymphangiogenic gene and protein expression profiles of the respective cell lines. (a) The cross-sectional image of a collagen-based scaffold with fibrin application after 1 day, which is intended to be in contact with the wound ground, shows a homogeneous distribution of the fibrin glue with almost no air pockets. Scale bar represents 500 μm. (b) The stereoscopic image shows a round, fibrin-coated scaffold with a diameter of 10 mm, which can serve as a multiple-cell carrier. Scale bar represents 5 mm.
The short-term effects on gene expression
The presence of an airtight seal, which is assumed to lead to hypoxic conditions, was investigated by HIF1a gene expression in cells. The hypoxia-inducible factor 1 alpha (HIF1a) is known as a very sensitive marker for low oxygen levels and is involved in the regulation of further growth factor cascades. Over an observation period of 24 hours, we assessed no markedly differences in gene expression regarding HIF1a in all cell types (Figure 3(a)), when cells are fixed with fibrin glue into the scaffold. Rather, HUVECs showed an opposite gene expression profile with a subtle upregulation of HIF1a in scaffolds without fibrin. This finding suggests no complete airtightness of the fibrin encapsulation. Use of fibrin glue reduces angiogenic and lymphangiogenic gene expression in scaffolds after 24 hours: Gene expression of endothelial and lymph endothelial cells was evaluated by Multiplex-RT-PCR and showed a significant increase after 24 hours in absence of fibrin application compared to cells fixed with fibrin glue into scaffolds. The following angiogenic and lymphangiogenic genes were analyzed: (a) Hypoxia-inducible factor 1 alpha (b) Prospero homeobox 1 (c) Vascular endothelial growth factor C (d) Vascular endothelial growth factor receptor 3 (e) Vascular endothelial growth factor A (f) Vascular endothelial growth factor B (g) Vascular endothelial growth factor receptor 1 (h) Vascular endothelial growth factor receptor 2. All experiments were repeated at least three times (ns = not significant; *p < 0.05; **p ≤ 0.01; ***p < 0.001).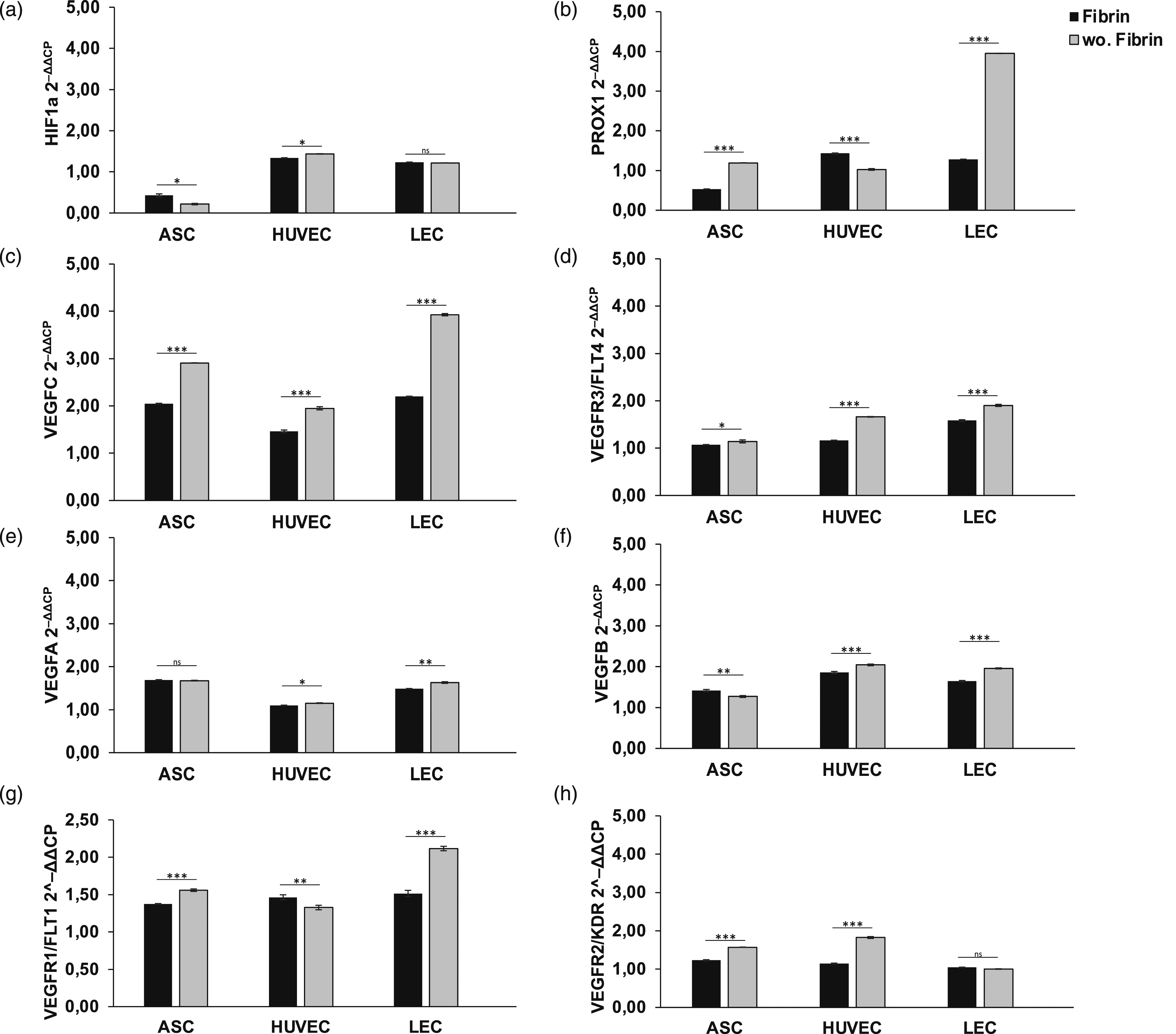
Angiogenesis
We next investigated the impact of fibrin application on endothelial gene regulation. To this end, we quantified the gene expression level of VEGFA and VEGFB as important mediators of angiogenesis (Figures 3(e) and (f)), as well as the corresponding receptors VEGFR1/FLT1 and VEGFR2/KDR (Figures 3(g) and (h)). The signal molecules VEGFA showed a significant upregulation by 5–10% regarding HUVECs and LECs (HUVEC: 1.15 ± 0.007; LEC: 1.63 ± 0.018) in absence of fibrin glue compared to scaffolds treated with fibrin glue (HUVEC: 1.10 ± 0.002; LEC: 1.49 ± 0.004) (Figure 3(e)). A similar result could also be demonstrated with VEGFB (Figure 3(f)). We could demonstrate a marked upregulation of 5–10% in HUVECs and LECs in the absence of fibrin (HUVEC: 2.04 ± 0.02; LEC: 1.96 ± 0.012), however, ASCs benefited more from the presence of fibrin glue with regard to VEGFA and VEGFB expression. Non-fibrin encapsulated scaffolds revealed significant overexpression of the corresponding receptor VEGFR1 after 24 hours, suggesting an improved angiogenic environment. We found an increase of 5% in ASCs (w.F.: 1.38 ± 0.004; wo.F.: 1.56 ± 0.016) and 20% in LECs (w.F.: 1.51 ± 0.042; wo.F.: 2.12 ± 0.029), whereas VEGFR1 was even downregulated in HUVEC (Figure 3(g)). However, VEGFR2 led to an upregulation in ASCs by 10% (w.F.: 1.23 ± 0.014; wo.F.: 1.57 ± 0.004) and HUVEC by 35% (w.F.: 1.15 ± 0.011; wo.F.: 1.83 ± 0.025) compared to cells in fibrin treated dermal replacement material (Figure 3(h)). In contrast, we assessed no significant impact on VEGFR2 expression in LECs by the application of fibrin glue. In conclusion, Multiplex-RT-qPCR detected a reduced gene expression of key angiogenic factors and receptors related to fibrin glue application, suggesting an impairment of vessel formation in skin replacement materials.
Lymphangiogenesis
Next, we assessed the effect of fibrin application in scaffolds on the lymph endothelial gene expression profile. To this end, we analyzed the potential of the growth factor VEGFC (Figure 3(c)), as well as the corresponding receptor VEGFR3/FLT4 (Figure 3(d)) and PROX1, a known lymph endothelial surface marker (Figure 3(b)). Fibrin encapsulation led to a generalized marked downregulation of PROX1, VEGFC, and VEGFR3 after just 24 hours. Respectively, PROX1 expression revealed a 2-fold upregulation in LECs and ASCs after 24 hours, when fibrin application is omitted (ASCs: 1.19 ± 0.003; LECs: 3.95 ± 0.003), compared to fibrin-containing scaffolds (ASCs: 0.53 ± 0.001; LECs: 1.28 ± 0.001) (Figure 3(b)). The signal molecule VEGFC showed a markedly increase of 50% in LECs, 10% in HUVECs, and 20% in ASCs in scaffolds not containing fibrin glue (w.F.: LECs: 2.2 ± 0.007; ASCs: 2.05 ± 0.008; HUVECs: 1.46 ± 0.027; wo.F.: LECs: 3.93 ± 0.02; ASCs: 2.91 ± 0.003; HUVECs: 1.95 ± 0.033) (Figure 3(c)). The corresponding lymphangiogenic receptor VEGFR3 was significantly upregulated in LECs by 5% (LECs: 1.9 ± 0.021) and in HUVECs by 10% (HUVECs: 1.66 ± 0.005), compared to cells in scaffolds treated with fibrin (LECs: 1.58 ± 0.017; HUVECs: 1.16 ± 0.003) (Figure 3(d)). The results suggest an inhibiting effect of fibrin encapsulation in skin replacement materials on the gene expression of lymphangiogenic factors, indicating a negative influence on lymphangiogenesis when used in wound healing.
To summarize, we demonstrated that fibrin glue application in scaffolds affected angiogenesis and lymphangiogenesis after 24 hours by downregulation of endothelial and lymph endothelial genes regarding several cell types for induction of wound healing.
The short-term effects on protein expression
To analyze the effect of the use of fibrin glue on angiogenic and lymphangiogenic gene expression, we measured the protein level by ELISA assay of the analyzed genes, described previously. To investigate the limiting effect of fibrin glue application on protein expression levels caused by an airtight encapsulation of cells inducing hypoxic conditions, we assessed the protein expression of HIF1a (Figure 4(a)). It showed a generalized low expression level in all cells with no significant differences compared to fibrinless scaffolds, pointing to sufficient oxygenation of all cells. Reduced angiogenic and lymphangiogenic protein expression level by fibrin glue application in scaffolds after 24 hours: The protein level of endothelial and lymph endothelial cells measured by ELISA showed a higher protein synthesis in HUVECs, LECs, and ASCs in absence of fibrin application, compared to fibrin containing conditions. Angiogenic and lymphangiogenic protein expression was significantly reduced in ACS, HUVECs, and LECs by the presence of fibrin glue. The following angiogenic and lymphangiogenic genes were analyzed: (a) Hypoxia-inducible factor 1 alpha (b) Prospero homeobox 1 (c) Vascular endothelial growth factor C (d) Vascular endothelial growth factor receptor 3 (e) Vascular endothelial growth factor A (f) Vascular endothelial growth factor B (g) Vascular endothelial growth factor receptor 1 (h) Vascular endothelial growth factor receptor 2. All experiments were repeated at least three times (ns = not significant; *p < 0.05; **p ≤ 0.01; ***p < 0.001).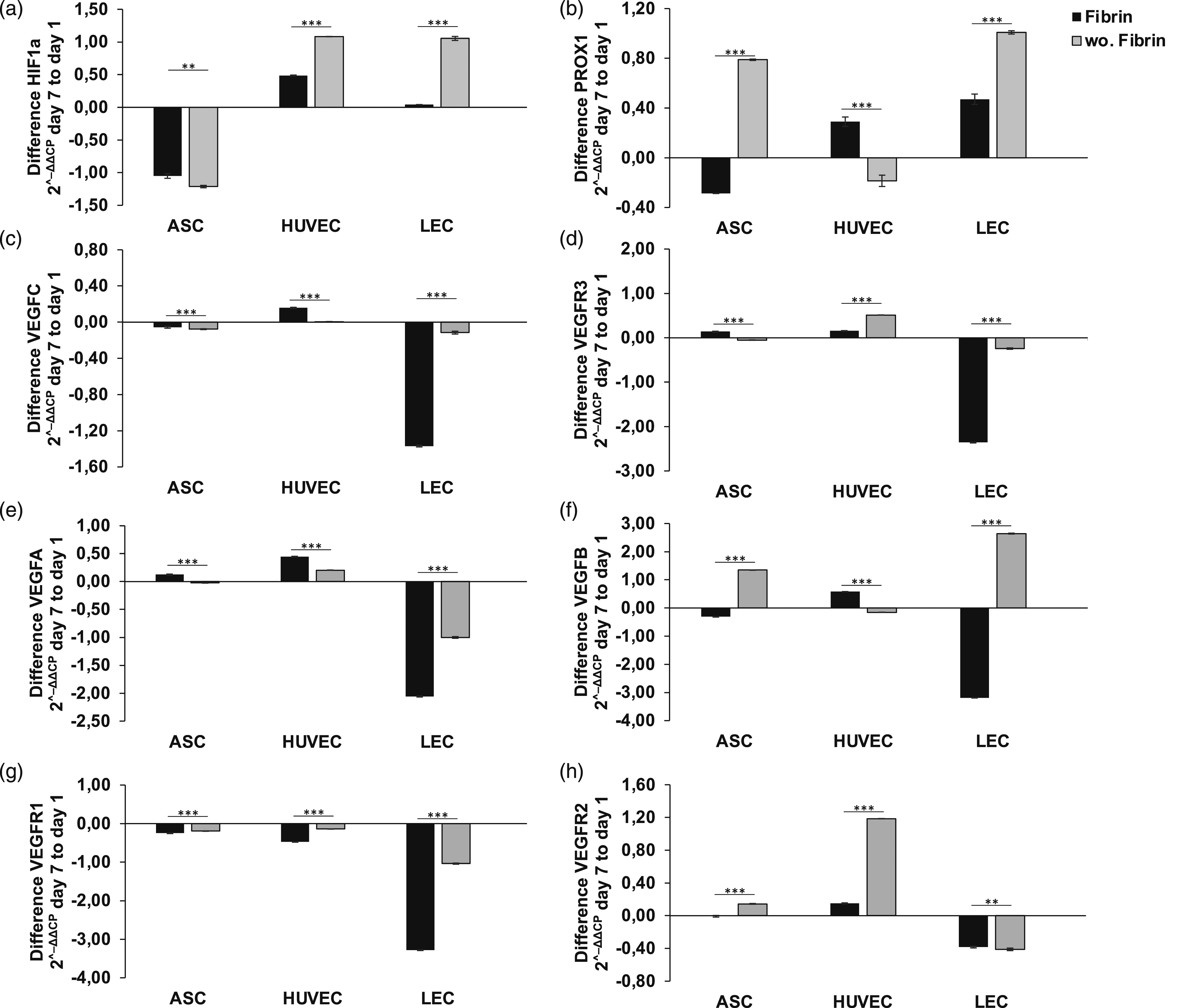
Angiogenesis
Moreover, we determined the harmful effect of fibrin application on the protein expression of endothelial cell markers, namely VEGFA, VEGFB, VEGFR1, and VEGFR2. Under fibrin-containing conditions, we assessed a generalized lower expression level, whereas protein levels were increased in absence of fibrin glue application (Figures 4(d)–(g)). The lack of fibrin glue in seeded scaffolds increased protein levels of VEGFA by >50% in ASCs, 5% in HUVEC, and 10% in LECs after 24 hours, compared to scaffolds with fibrin application, as shown in Figure 4(e). Surprisingly the peak of VEGFA expression was achieved in LECs (3.14 ± 0.013) (Figure 4(e)). We assessed enhanced protein levels of VEGFB in scaffolds without fibrin application by 20% in ASCs, 5% in HUVEC, and >30% in LECs, whereas we observed the highest VEGFB protein level in HUVEC (1.11 ± 0.013) (Figure 4(f)). Concordantly with findings in qPCR, fibrin application induced a significant reduction in protein expression of all endothelial proteins, suggesting an adverse effect on angiogenesis and related processes. Further, the protein expression-inhibiting effect of fibrin glue is emphasized in VEGFR1 and VEGFR2 expression after 24 hours. The absence of fibrin glue application in scaffolds revealed a twofold increase of protein levels of VEGFR1 in LECs (w.F.: 4.36 ± 0.008; wo.F.: 8.83 ± 0.001) and an increase of 5–10% in ASCs (w.F.: 10.23 ± 0.018; wo.F.: 12.59 ± 0.02) and HUVECs (w.F.: 10.4 ± 0.041; 12.25 ± 0.006), supporting the aforementioned findings. Further, we observed a markedly reduced upregulation of VEGFR2 in scaffolds with fibrin application, with a significant difference in protein expression of >30% in ASCs (w.F.: 0.1 ± 0.013; wo.F.: 0.16 ± 0.013) and 5–10% in LECs (w.F.: 0.09 ± 0.007; wo.F.: 0.08 ± 0.012) and HUVECs (w.F.: 0.09 ± 0.006; wo.F.: 0.1 ± 0.016) (Figure 4(h)). Peak protein expression levels of VEGFR1 and VEGFR2 were achieved in fibrinless scaffolds in ASCs (12.59 ± 0.02; 0.16 ± 0.013). Taken together, the ELISA assay confirms a possible adverse effect of fibrin glue application on protein expression of crucial angiogenic factors as early as 24 hours.
Lymphangiogenesis
Next, we evaluated the inhibitory effect of fibrin glue application on lymph endothelial cells by detecting the protein expression level of signal molecules and surface receptors, associated with lymphangiogenesis. We observed a lower protein expression of PROX1, VEGFC, and VEGFR3 in fibrin-containing scaffolds in all cell types (Figure 4(b)–(d)). 24 hours of fibrin application revealed overexpression of all three proteins in all cells, thus confirming the inducibility of gene regulation with fibrin glue. The fibrin application increased protein levels of PROX1 by twofold and 5–10% in HUVECs (w.F.: 0.05 ± 0.02; wo.F.: 0.09 ± 0.01), LECs (w.F.: 0.09 ± 0.02; wo.F.: 0.11 ± 0.02) and ASCs (w.F.: 0.09 ± 0.02; wo.F.: 0.11 ± 0.005) (Figure 4(b)), respectively, whereas the highest protein level was measured in ASCs and LECs. The growth factor VEGFC was markedly overexpressed in ASCs (0.69 ± 0.01), LECs (0.87 ± 0.01), and HUVECs (1.05 ± 0.02) in fibrinless scaffolds (Figure 4(c)), compared to fibrin containing scaffolds (ASCs: 0.37 ± 0.003; LECs: 0.75 ± 0.003; HUVECs: 0.82 ± 0.003). Surprisingly, the effect of fibrin glue application on PROX1, VEGFC, and VEGFR3 protein levels was not limited to LECs. Peak protein expression levels of VEGFC were achieved in absence of fibrin glue with HUVECs (1.05 ± 0.02). We assessed 10–30% enhanced protein levels of VEGFR3 in HUVECs, LECs, and ASCs, respectively, with significant correlation to an absence of fibrin application. Altogether, the ELISA assay provides evidence of reduced lymphangiogenic protein expression and associated lymphangiogenesis when using commercial fibrin glue in skin replacement materials.
Summarizing these findings, we demonstrated a negative influence of fibrin glue application on angiogenic and lymphangiogenic protein expression in scaffolds.
The long-term application of fibrin glue in scaffolds induces downregulation of angiogenic and lymphangiogenic genes and proteins
The long-term effects on gene expression
Having demonstrated the harmed angiogenic and lymphangiogenic gene expression profile by fibrin glue application after 24 hours we evaluated next the effect over 7 days. HIF-1 alpha showed depending on cell type only small varieties in gene expression after 7 days (Figure 5(a)). Surprisingly the fibrin encapsulation led to markedly reduced HIF-1a expression in LECs and HUVECs, suggesting paradoxically a less hypoxic condition compared to the absence of fibrin glue in dermal replacement material. These results provide evidence for no airtight sealing of a fibrin encapsulation of cells. Hence, there is no evidence of fibrin seal-induced hypoxia in scaffolds with adverse effects on wound healing. Long-term application of fibrin glue in scaffolds induces downregulation of angiogenic and lymphangiogenic genes: Relative angiogenic and lymphangiogenic gene expression in ASCs, LECS, and HUVECs analyzed by Multiplex-RT-qPCR after 7 days of fibrin application in scaffolds revealed a reduced expression profile compared to cells in fibrinless scaffolds. Following angiogenic and lymphangiogenic genes were analyzed: (a) Hypoxia-inducible factor 1 alpha (b) Prospero homeobox 1 (c) Vascular endothelial growth factor C (d) Vascular endothelial growth factor receptor 3 (e) Vascular endothelial growth factor A (f) Vascular endothelial growth factor B (g) Vascular endothelial growth factor receptor 1 (h) Vascular endothelial growth factor receptor 2. All experiments were repeated at least three times (ns = not significant; *p < 0.05; **p ≤ 0.01; ***p < 0.001).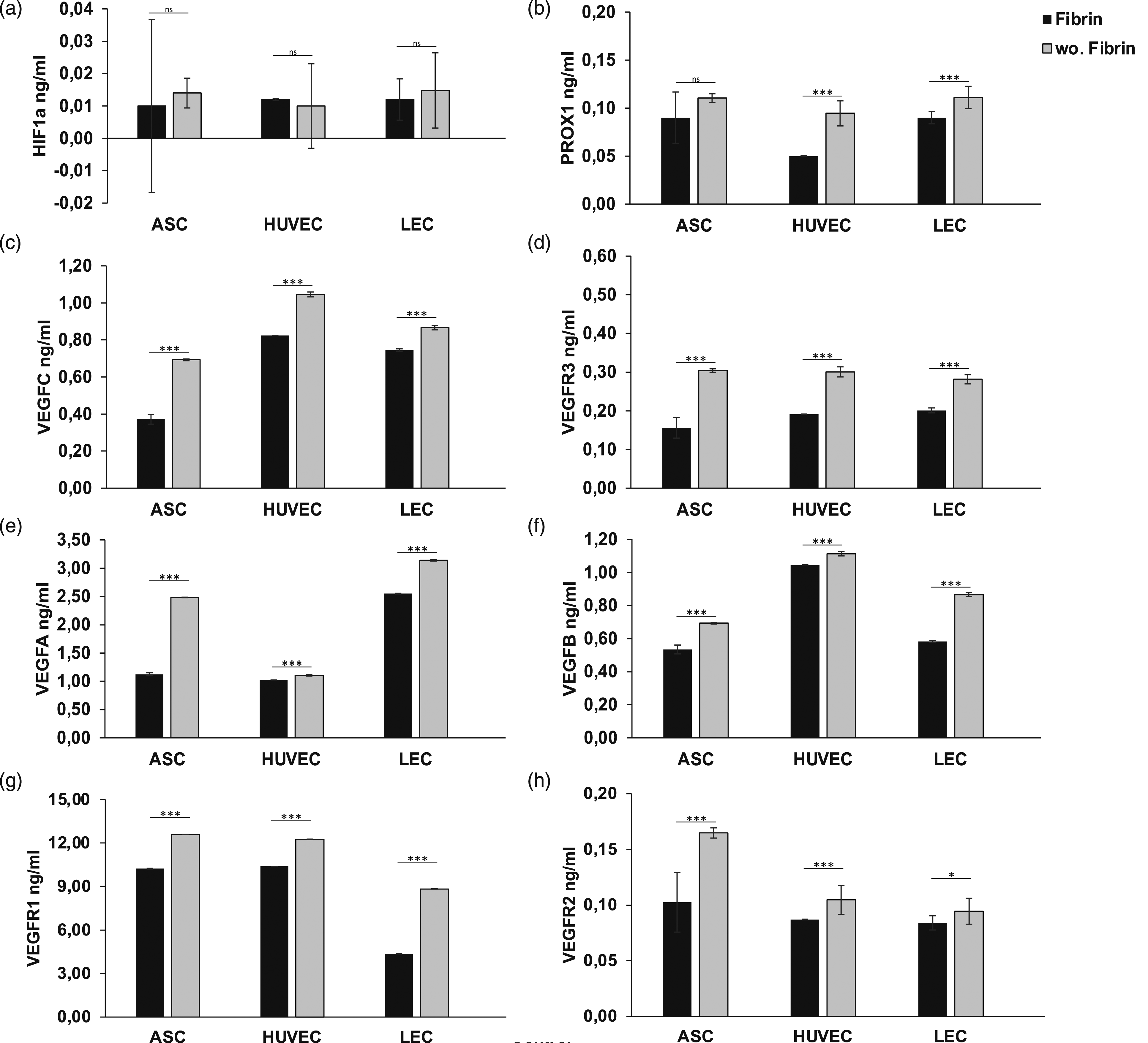
Angiogenesis
Further, we investigated the influence of the long-term application of fibrin glue on angiogenesis by analyzing the difference in endothelial gene expression after 7 days in seeded dermal replacement material. The growth factor VEGFA is upregulated over the 7-day period in HUVEC and ASCs, especially in fibrin-treated scaffolds (Figure 5(e)). In LECs, on the other hand, there is a marked downregulation, which, however, turned out to be >50% less without the use of fibrin (Figure 5(e)). Rather we observed similar results by increasing VEGFB expression in HUVEC with fibrin applications. However, the VEGFB expression revealed a significant downregulation in LECs (−3.19 ± 0.107) and in ASCs (−0.13 ± 0.036) in the presence of fibrin glue (Figure 5(f)). In ASCs, the absence of fibrin glue led to an increase in VEGFB expression after 7 days by 1.35 ± 0.04 points, in LECs even by 2.64 ± 0.056 points (Figure 5(f)), pointing to a high angiogenic impact and demanding a reevaluation of further use of fibrin in clinical and experimental applications. Surprisingly, there is a general downregulation of the expression of the receptor VEGFR1 in all cell types, regardless of fibrin treatment. But scaffolds without fibrin application revealed a reduced downregulation in HUVECs and LECs (Figure 5(g)). This difference became evident in HUVEC and LECs with >50% lower loss of expression of the receptor after 7 days when fibrin glue was avoided (Figure 5(g)). After 7 days of incubation, we evaluated a significant increase in gene expression regarding VEGFR2. The receptor expression revealed the strongest increase in HUVECs with 1.19 ± 0.0563 and ASCs with 0.14 ± 0.003 points (Figure 5(h)), suggesting an angiogenesis-promoting effect of the absence of fibrin glue in tissue engineering approaches. Taken together, fibrin glue leads to partial inhibitory effects on genes for hemangiogenesis after 7 days, since growth factors such as VEGFA and B are even upregulated in HUVEC after 7 days of adapting to fibrin-containing conditions. However, there is a tendency for increased angiogenic gene expression in untreated fibrinless scaffolds, suggesting an advantage of an absence of fibrin in the wound ground and judicious use in clinical practice.
Lymphangiogenesis
Next, we investigated the long-term effects of fibrin application on lymph endothelial gene expression in seeded scaffolds. Due to the characteristics as a lymphatic surface marker, a strong increase of PROX1 expression level could be evaluated in LECs after 7 days regardless of the fibrin application (Figure 5(b)). The absence of fibrin glue in scaffolds induces a significant upregulation of PROX1 by >50% in LECs (1.01 ± 0.045) and >100% in ASCs (0.79 ± 0.005), compared to fibrin-containing scaffolds (LEC: −0.47 ± 0.041; ASC: −0.29 ± 0.005), suggesting a high impact in growth-inhibiting of fibrin glue on lymphangiogenesis. As part of the cascade, the docking of VEGF-C to the complementary lymphatic receptor VEGFR3/FLT4 induces lymphangiogenesis. The growth factor VEGFC and corresponding receptor VEGFR3 showed only minor changes in gene expression regarding all cell types, except LECs (Figures 5(c) and (d)). We evaluated a markedly decreased gene expression profile of VEGFC (−0.12 ± 0.083) and VEGFR3 (−0.24 ± 0.026) in LECs after 7 days in fibrin-containing scaffolds, compared to not fibrin encapsulated ones (VEGFC: −1.37 ± 0.014; VEGFR3: −2.36 ± 0.058), suggesting a growth-inhibiting effect in lymphangiogenesis by fibrin glue application. Further, we observed a significant increase of VEGFR3 expression level in HUVECs (w.F.: 0.16 ± 0.005; wo.F.: 0.52 ± 0.042) by > 50% in fibrinless scaffolds (Figure 5(d)), whereas an increased expression of VEGFC under fibrin containing conditions after 7 days (Figure 5(c)). Surprisingly, the fibrin application induces no significant changes in the gene expression profile of VEGFC and VEGFR3 of ASCs. In conclusion, after 7 days, these results provide evidence of an inhibitory effect on the gene expression of lymphatic endothelial cells by fibrin glue application in tissue engineering, referring to impaired lymphangiogenesis and lymphatic regeneration.
Altogether, we demonstrated a harmful effect on angiogenic and lymphangiogenic gene expression by long-term application of fibrin glue with a possible impact on impaired wound healing and tissue regeneration.
The long-term effects on protein expression
Having demonstrated the inhibiting effects of the presence of fibrin glue in skin replacement material on protein expression levels, we investigated the potential of a long-term treatment over 7 days. The protein level of HIF1a showed no significant differences after 7 days of treatment in ASCs and LECs, except HUVECs (Figure 6(a)). Fibrin glue application induces an isolated markedly increase of HIF1a expression in HUVEC (0.029 ± 0.005), pointing towards a lack of oxygen after 7 days for these cells. Long-term application of fibrin glue in scaffolds reduces angiogenic and lymphangiogenic protein expression in scaffolds: Quantification of protein levels by ELISA after continuous fibrin treatment over 7 days showed significant downregulation of VEGFA, VEGFB, VEGFC, VEGFR3 levels regarding ASCs, LECS, HUVECs in scaffolds. The lack of fibrin glue was capable to stimulate protein expression in all cell types over 7 days and thus promoting angiogenesis and lymphangiogenesis in scaffolds. The following angiogenic and lymphangiogenic genes were analyzed: (a) Hypoxia-inducible factor 1 alpha (b) Prospero homeobox 1 (c) Vascular endothelial growth factor C (d) Vascular endothelial growth factor receptor 3 (e) Vascular endothelial growth factor A (f) Vascular endothelial growth factor B (g) Vascular endothelial growth factor receptor 1 (h) Vascular endothelial growth factor receptor 2. All experiments were repeated at least three times (ns = not significant; *p < 0.05; **p ≤ 0.01; ***p < 0.001).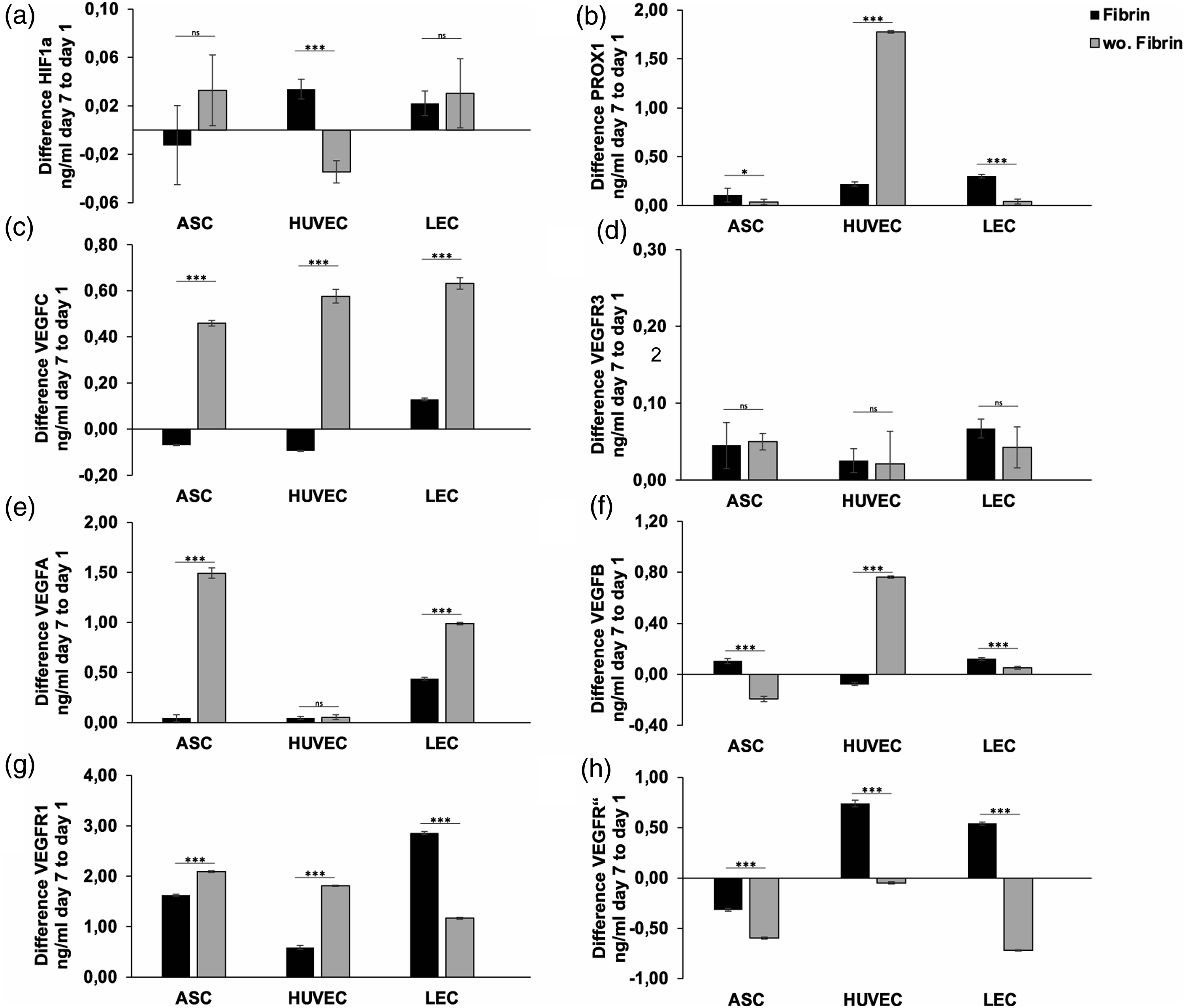
Angiogenesis
Next, we evaluated the influence of long-term fibrin glue treatment on angiogenic protein expression. No fibrin glue application significantly increased protein levels by ten and twofold of VEGFA in ASCs (w.F.: 0.04 ± 0.005; wo.F.: 1.49 ± 0.003) and LECs (w.F.: 0.44 ± 0.001; wo.F.: 1.26 ± 0.002), respectively (Figures 6(c) and (d)), detecting no significance only in HUVECS. Regarding VEGFB we evaluated a markedly increase by > 100% in the absence of fibrin glue limited to HUVECs (Figure 6(f)). The 7 days of absence of fibrin application revealed effectiveness in protein expression of VEGFR1 with sufficient significance in ASCs and HUVECs (Figure 6(g)). We assessed an overexpression of VEGFR1 by 10% in ASCs (w.F.: 1.62 ± 0.003; wo.F.: 2.09 ± 0.0002) and >50% in HUVECs (w.F.: 0.059 ± 0.001; wo.F.: 1.81 ± 0.002), confirming the effectiveness of the lack of fibrin application. However, protein levels of VEGFR2 showed a marked regression in ASCs, HUVECs, and LECs after 7 days (Figure 6(h)), inconsistent with our previous results.
Lymphangiogenesis
To investigate the impact of fibrin glue on lymphangiogenic protein expression, we analyzed the protein levels of PROX1, VEGFR3, and VEGFC. Surprisingly, we observed a tenfold increase of PROX1 expression, limited to HUVECs (w.F.: 0.23 ± 0.002; wo.F.: 1.89 ± 0.001) in the absence of fibrin glue and an increase of PROX1 expression in fibrin containing conditions regarding ASCs and LECs (Figure 6(b)), pointing towards a limited inhibiting effect of fibrin application depending on cell-type. However, after 7 days of hypoxic treatment, we assessed a significant increase of VEGFC protein levels by > 100% in all cells (ASC: w.F.: −0.09 ± 0.001; wo.F.: 0.45 ± 0.002; HUVEC: w.F.: −0.11 ± 0.001; wo.F.: 0.52 ± 0.003; LEC: w.F.: 0.18 ± 0.002; wo.F.: 0.66 ± 0.004) (Figure 6(c)), concordantly suggesting fibrin glue as a crucial gene regulator with high impact on lymphangiogenesis. In contrast to these results, we observed no significance in VEGFR3 expression to the lack of fibrin application regarding all cell types. We evaluated the strongest increase in protein levels of VEGFC and PROX1 after 7 days in HUVEC and LEC monocultures.
Taken together, the results of protein expression after 7 days of fibrin application demonstrated a long-term impact on angiogenesis and lymphangiogenesis in scaffolds.
Discussion
Fibrin glue is a composite material commonly used in clinical practice with a wide range of applications. It is used, for example, in neurosurgery to seal the dura mater 16 and in plastic reconstructive surgery to stop bleeding and improve wound closure.27–30 When thrombin is added, the synthetically manufactured fibrin combines to form a polymer that promises a rapid sealing effect. In addition, it is a human-compatible substance that is metabolized by the human organism over time without leaving residues of foreign material. These ideal properties explain the frequent clinical use of fibrin glue, but its application was rarely questioned, especially with regard to wound healing. Our group discovered the potential of fibrin glue early on and presented first-time a cell fixation method for algae and cyanobacteria in skin replacement materials with fibrin glue.31,32 We analyzed the permeability of the fibrin scaffold and found that nutrients could pass into the fibrin scaffold and growth factors produced by microorganisms, as well as oxygen to the outside. According to our findings, we assume fibrin glue is an ideal material and cell carrier for wound healing. But the question arises whether the densely formed fibrin polymers allow the formation of vessels and lymphatic vessels, promote them, or even hinder them. To ensure that fibrin glue does not have an adverse effect on wound healing, we fixed cells, which play a key role in the wound healing process, into regular skin substitutes, either with or without fibrin glue. These were human venous endothelial cells (HUVEC), lymphatic endothelial cells (LEC), and adipose-derived stem cells (ASC). Subsequently, changes in the angiogenic and lymphangiogenic genetic profiles were analyzed. In order to be able to rule out a hypoxic encapsulation, the hypoxia-inducible factor 1 alpha (HIF1a) was also examined.
First, we analyzed the fibrin construct applied onto the scaffold macroscopically and microscopically. We demonstrated a continuous distribution of fibrin within the skin substitute with no formation of air bubbles or clots (Figure 2). Due to the sealing effect of the fibrin polymers, we suspected a possible insufficient supply of nutrients and oxygen within the fibrin structure. Previous analysis of the permeability of the fibrin construct showed a direct correlation between the structural permeability and pore size of the final 3D fibrin construct to fibrinogen and thrombin concentration. 33 Chiu et al. found a significant restriction of the flow speed and diffusive property was already found at 15 UI/ml thrombin and at 15 mg/mL fibrinogen concentrations. 33 Fibrin glue from regular clinical practice contains a concentration of 500 UI/ml thrombin and 91 mg/mL fibrinogen and creates a very strong sealing effect for e.g. restoration of hemostasis or elimination of anastomotic leaks without considering the consequences for the cellular environment.
Accordingly, the scaffolds were seeded with different cell types and the vitality was assessed after 24 hours and 7 days on basis of gene and protein expression. With continuous gene and protein expression even after 7 days, we assume a high vitality and preserved metabolism of the cells. Previous studies have promised the survival of cells encapsulated in fibrin. Krug et al. cultured adipose-derived multipotent stem/progenitor cells over 14 days in fibrin gel with high viability, metabolic, and remodeling activities during the cultivation period. 6 Gilles et al. studied the migration pattern, morphology, and viability of cells suspended in five different fibrin glues and found a significantly higher TGF-beta 1 and 2 concentration in partial autologous fibrin sealant compared to their commercial counterparts. Further, no ingrowth of cells was seen in any of the experimental conditions. 34 Hopfner et al.3 35 Chavez et al.3 36 and Schenck et al.3 32 used fibrin glue to fix photosynthetic microorganisms and showed high metabolic activity and proliferation of these in the scaffold over 7–14 days. However, these cultures had the property of producing oxygen self-sufficiently by photosynthesis and can therefore only confirm a sufficient nutrient supply through the medium. Adequate oxygen supply is vital for a healthy cell growth environment. An airtight sealing effect of fibrin encapsulation would result in significant growth restrictions and apoptosis. Previously Volkmer et al.3 37 showed that hypoxia may be a serious problem in scaffold-based approaches. Our results showed no significant differences between fibrin-bonded and non-bonded scaffolds with regard to the hypoxia-induced factor HIF1a as a significant marker of hypoxic cell metabolism. This finding suggests no complete airtightness of the fibrin encapsulation creating no hypoxic conditions within the scaffold accompanied by no reduced gene expression in scaffolds. Thus, we were able to confirm preserved cell vitality within the fibrin glue used in regular clinical practice.
In recent studies, our working group has already introduced a new method for fixing bacteria or algae in collagen-based scaffolds using fibrin glue.31,32,35 The scaffolds applied with fibrin should then be placed on the wound bed and provide an optimized platform for vascular and skin tissue ingrowth.31,32 Therefore, there is great interest in investigating the effect of fibrin on human endothelial cells at the molecular level. In our preparatory studies, cells were always vital with activated proliferation, but in contrast, these were not human cells, but aquacultures with high adaptability and autotrophic oxygen production through the property of photosynthesis. This allowed these organisms to proliferate even in oxygen-poor scaffolds compacted with fibrin. In addition, fibrin glue is used in various operations with the aim of sealing vessels and initiating hemostasis. 38 It is therefore obvious that endothelial cells in a fibrin clot have no tendency to proliferate and migrate. This also confirms the molecular interactions. By binding to the ICAM-1 receptor, fibrinogen causes activation of the signaling molecule ET-1, which produces vasoconstriction and endothelial fibrosis. 39 At the same time, ERK 1/2 promotes apoptosis and pro-inflammatory cytokine secretion, which on the contrary does not promote wound healing. Our results showed significantly higher gene and protein expression in the absence of fibrin glue after 24 hours. This goes in line with the results of previous studies, which introduced modified fibrin glue containing growth factors.40–42 These studies already addressed the problem of missing stimulating factors and impaired or delayed wound healing caused by commercial fibrin glue. One group introduced fibrin glue from autologous plasma containing growth factors such as TGF-β and VEGF that promote the reconstruction of the mucous membrane by stimulating fibroblast proliferation and angiogenesis. 40 Shi et al. developed a clinical-grade platelets exosome product (PEP), which was incorporated with injectable surgical fibrin sealant, to promote chronic wound healing and complete skin regeneration. 41 Both groups showed that modified fibrin glue is predicted to improve healing better than commercial fibrin glue. Further the application of fibrin glue to reinforce the gastric staple-line after sleeve gastrectomy revealed no histopathological effects on inflammation, fibroblastic activity, and neo-angiogenesis, compared to the absence of fibrin glue. 43 Yeung et al.8 8 discovered a delayed epithelial cell migration onto the corneal surface caused by fibrin glue acting as a physical barrier. In contrast, previous studies by Hopfner et al.2 21 showed an enhanced secretion of key cytokines (SDF-1, b-FGF) of human ASCs by fibrin glue application. However, in this case, cells and fibrin glue were cultivated for at least 1 week, going in line with our results, as the effect was only partially inhibitory on genes for hemangiogenesis after 7 days. Surprisingly, the growth factors VEGFA and VEGFB were markedly upregulated in HUVECs in fibrin-containing scaffolds. The reason for this discrepancy could be explained by the short half-time of fibrin polymers or bias such as the influence of collagen scaffolds or the cell medium and requires further clarification.44,45 Further, Krug et al.6 6 discovered a higher mortality rate of cells encapsulated with fibrin after 24 hours, compared to those cultured over 7 days by performing a live-dead assay, supporting the hypothesis of a toxic or harming effect of fibrin polymers in the first hours of application.
Taken together commercial fibrin glue itself suggests no promising impact on building new vessels in the observation period. In our study, we even revealed a negative influence on gene and protein expression regarding angiogenesis. Hence, these results provide evidence that the application of fibrin glue could have a harmful effect on skin regeneration and proper wound healing, which requires unrestricted vascular vessel formation.
Furthermore, we investigated the effect of fibrin glue on gene and protein expression in lymphatic cells. We analyzed the expression level of PROX1, VEGFC, and the corresponding receptor VEGFR3, as well-known lymphatic markers and important molecular factors of lymphatic vessel formation. VEGFC is a growth factor that significantly induces lymphangiogenesis by binding to VEGFR3. To date, there is little literature examining the effect of fibrin on lymphangiogenesis. Our group is the first to evaluate the impact of fibrin glue on a genetic level. We showed significant downregulation of gene and protein expression, especially with regard to VEGFC and VEGFR3 after 24 hours. Other studies have so far been limited to a purely histological evaluation and have shown inconsistent results. The study of Ritschl et al.4 46 showed increased lymphangiogenesis and neovascularization with the use of fibrin glue in clinical practice to seal vascular anastomoses. However, Cho et al.4 47 detected no histological differences in the group with fibrin glue application in the formation of microvascular anastomoses compared to those without fibrin, which largely supports our results. We have demonstrated a significant increase in PROX1, VEGFC, and VEGFR3 gene and protein expression after 24 hours, especially regarding LECs, but also in HUVECs and ASCs when fibrin glue was dispensed with. This also supports an inhibiting effect on lymphangiogenesis, which could impair adequate wound healing. After 7 days, this effect was only detectable in LECs. Fibrin application no longer showed a negative effect on VEGFC and VEGFR3 expression in ASCs. With regard to HUVECs, an upregulation of VEGFC was even evident in scaffolds containing fibrin glue. This may be due to the fact that fibrin glue was already partially degraded after 7 days with limited effect on cells. Commercial fibrin glue has a short half-time and is resorbed in approximately 10–14 days,44,45 according to the manufacturer. Krug et al. reported that the fibrin matrix was completely degraded after 14 days of incubation. 6 Thereby it is not clear whether it is a degradation process of fibrin itself or the cells actively induced fibrin degradation. 6
In summary, the application of fibrin glue could negatively affect cellular lymphatic gene and protein expression in the first 24 hours. Further, we confirm a harmful effect of fibrin application in protein and gene expression limited to adipose, endothelial, and lymph endothelial cells. After 7 days the effect is reduced.
Conclusion
This study indicates an inhibitory impact at the molecular genetic level on angiogenesis and lymphangiogenesis, especially in the first 24 hours. The increased protein and gene expression profile in absence of fibrin glue provides evidence of orchestrating the process of angiogenesis and lymphangiogenesis to reinstate perfused environment in a wound and could permit the progress of wound healing. Further in vitro and in vivo studies are necessary with regard to the proliferation and migration behavior of these cells in order to be able to make a reliable statement. The clinical application with regard to wound healing should be critically questioned. However, our group proposes a strategic use of glue, to seal anastomosis or initiate hemostasis, but appeals to avoid excessive consumption, especially in the wound area.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
