Abstract
Adding dental pulp stem cells (DPSCs) to vascular endothelial cell–formed vessel-like structures can increase the longevity of these vessel networks. DPSCs display pericyte-like cell functions and closely assemble endothelial cells (ECs). However, the mechanisms of DPSC-derived pericyte-like cells in stabilizing the vessel networks are not fully understood. In this study, we investigated the functions of E-DPSCs, which were DPSCs isolated from the direct coculture of human umbilical vein endothelial cells (HUVECs) and DPSCs, and T-DPSCs, which were DPSCs treated by transforming growth factor beta 1 (TGF-β1), in stabilizing blood vessels in vitro and in vivo. A 3-dimensional coculture spheroid sprouting assay was conducted to compare the functions of E-DPSCs and T-DPSCs in vitro. Dental pulp angiogenesis in the severe combined immunodeficiency (SCID) mouse model was used to explore the roles of E-DPSCs and T-DPSCs in vascularization in vivo. The results demonstrated that both E-DPSCs and T-DPSCs possess smooth muscle cell–like cell properties, exhibiting higher expression of the mural cell–specific markers and the suppression of HUVEC sprouting. E-DPSCs and T-DPSCs inhibited HUVEC sprouting by activating TEK tyrosine kinase (Tie2) signaling, upregulating vascular endothelial (VE)–cadherin, and downregulating vascular endothelial growth factor receptor 2 (VEGFR2). In vivo study revealed more perfused and total blood vessels in the HUVEC + E-DPSC group, HUVEC + T-DPSC group, angiopoietin 1 (Ang1) pretreated group, and vascular endothelial protein tyrosine phosphatase (VE-PTP) inhibitor pretreated group, compared to HUVEC + DPSC group. In conclusion, these data indicated that E-DPSCs and T-DPSCs could stabilize the newly formed blood vessels and accelerate their perfusion. The critical regulating pathways are Ang1/Tie2/VE-cadherin and VEGF/VEGFR2 signaling.
Keywords
Introduction
Angiogenic sprouting initiated by vascular endothelial growth factor (VEGF) and angiopoietin 2 (Ang2) triggers the detachment of pericytes from the endothelium and promotes the proliferation and migration of endothelial cells (ECs). After reassembled, ECs deposit the new basement membranes and further secrete cellular factors, such as platelet-derived growth factor (PDGF), which can recruit supporting mural cells (smooth muscle cells (SMCs) and pericytes) to stabilize the newly formed vessels (Armulik et al. 2005). The newly formed vessels are prone to be leaky and regressive quickly if they do not become stable and mature (Gonçalves et al. 2007). Sprouting is the first step of angiogenesis and a sign of vascular instability. The adhesions between mural cells and ECs in sprouting are essential for maintaining the stability of neovasculature (Thomas and Augustin 2009). For in vitro studies, 3-dimensional (3D) coculture spheroidal sprouting assay is widely used to assess vascular stability and to explore the interactions between human umbilical vein endothelial cells (HUVECs) and other cell types, such as SMCs, pericytes, and mesenchymal stem cells (MSCs) (Vorwald et al. 2020).
Ang1/Tie2 signaling is involved in the regulation of vascular stability. There are four ligands in the Ang family, Ang1, Ang2, Ang3, and Ang4, and two corresponding tyrosine kinase receptors, Tie1 and Tie2 (Lee et al. 2004). Tie1 and Tie2 are mainly expressed on the membrane of ECs, while Ang1 is primarily secreted by mural cells. Ang1 activates Tie2 and its downstream signaling after binding to Tie2. A study reported that the activated Ang1/Tie2 signaling promotes HUVEC survival and enhances vascular stability by activating the phosphatidylinositol 3-kinase (PI3K)–AKT pathway (Kim et al. 2000). Furthermore, Ang1 improves the adhesions of ECs and decreases vascular permeability by regulating vascular endothelial (VE)–cadherin (Lee et al. 2011). Tie2 also associates with vascular endothelial protein tyrosine phosphate (VE-PTP) to maintain the stability of blood vessels. Inhibition of VE-PTP activity induces Tie2 activation and its downstream signaling pathways, including extracellular signal-regulated kinase (ERK), endothelial nitric oxide synthase (eNOS), and AKT, stabilizing blood vessels (Shen et al. 2014). In addition, VE-PTP downregulates VE-cadherin phosphorylation, suppressing vascular permeability (Saharinen et al. 2008).
Dental pulp stem cells (DPSCs) isolated from dental pulp tissue can differentiate into various cell types, including odontoblasts, chondrocytes, osteoblasts, ECs, and SMCs (Gronthos et al. 2000; Zhang et al. 2021). DPSCs promote vascular stability by acting as mural-like cells (Dissanayaka et al. 2012), but the mechanisms are unclear. Our recent studies demonstrated that DPSCs isolated from HUVEC + DPSC direct cocultures (E-DPSCs) highly expressed SMC-specific markers (Zhang et al. 2023), which was similar to DPSCs treated by transforming growth factor β1 (TGF-β1) (T-DPSCs) (Zhang et al. 2021). These studies suggest that DPSCs dynamically alter their phenotypes from stem cells to SMC-like cells when interacting with ECs or treated with TGF-β1. Therefore, this study aimed to investigate the functions of SMC-like cells, E-DPSCs, and T-DPSCs in maintaining the stability of blood vessels and underlying mechanisms in vitro and in vivo.
Materials and Methods
Real-time quantitative polymerase chain reaction (RT-qPCR), Western blotting, and immunofluorescence were used to evaluate the SMC-specific marker expression in E-DPSCs and T-DPSCs. The 3D sprouting assay was used to assess the functions of E-DPSCs and T-DPSCs on HUVEC sprouting. The expression of Ang1 and VEGF in E-DPSCs and T-DPSCs was detected by RT-qPCR, Western blotting, and enzyme-linked immunosorbent assay (ELISA). Tooth slice angiogenesis in the severe combined immunodeficiency (SCID) mouse model was conducted to evaluate the effects of E-DPSCs, T-DPSCs, Ang1, and VE-PTP inhibitor on vascularization in vivo. The hematoxylin and eosin (H&E) staining and immunohistochemistry were performed to examine the structure and morphology of the newly formed vasculatures. All the animal experiment protocols were approved by the committee on the use of live animals in teaching and research (approved No. 202207S083) of Xuzhou Medical University, Jiangsu, China. All animal handling was carried out under the guidance and supervision of the Laboratory Animal Unit at Xuzhou Medical University and the ARRIVE (Animal Research: Reporting of In Vivo Experiments) 2.0 guidelines. The details of the methods are found in the Appendix.
Results
E-DPSCs and T-DPSCs Inhibit HUVEC Sprouting by Activating Tie2 Signaling
E-DPSCs were assessed by flow cytometry and immunofluorescent staining to confirm there were no HUVECs left in E-DPSCs (Appendix Fig. 1). RT-qPCR and Western blot results showed the messenger RNA (mRNA) and protein expression of α–smooth muscle actin (α-SMA), smooth muscle 22α (SM22α), and calponin was significantly increased in E-DPSCs and T-DPSCs compared to that in DPSCs (Appendix Fig. 2). In line with Western blot results, the fluorescence intensity of α-SMA, SM22α, and calponin in E-DPSCs and T-DPSCs was much higher than in DPSCs (Appendix Fig. 3). In addition, E-DPSCs and T-DPSCs markedly suppressed HUVEC sprouting in the 3D spheroidal coculture model (Fig. 1A). To explore how E-DPSCs and T-DPSCs inhibit HUVEC sprouting, the expression of Ang1 and VEGF in E-DPSCs and T-DPSCs was analyzed by RT-qPCR, Western blot, and ELISA. The mRNA (Appendix Fig. 4) and protein expression of Ang1 was much higher in E-DPSCs and T-DPSCs compared to that in DPSCs, as well as the concentrations of Ang1 in E-DPSC–conditioned media (CM) and T-DPSC-CM (Fig. 1B, 1C). However, VEGF had no significant difference. Furthermore, the fluorescence intensity of p-Tie2 in spheroids of HUVEC + E-DPSC and HUVEC + T-DPSC was much higher than that in spheroids of HUVEC + DPSC (Fig. 1D).
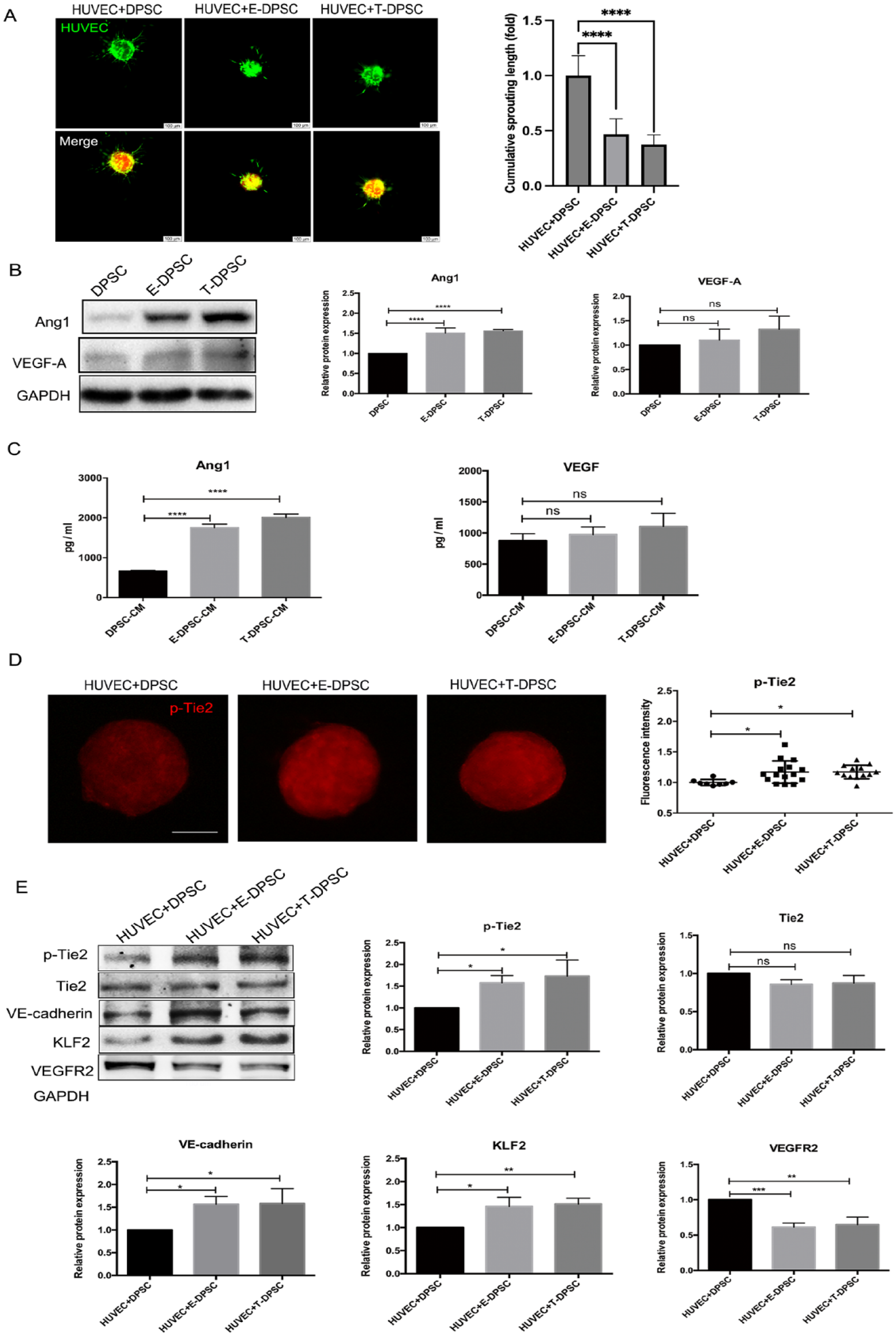
E-DPSCs and T-DPSCs activate Tie2 signaling and its downstream effectors in HUVECs. (
To explore whether the secreted Ang1 in E-DPSC-CM and T-DPSC-CM can activate Tie2 and its downstream signaling pathways in HUVECs, HUVECs were treated with E-DPSC-CM and T-DPSC-CM for different time points. The expression of p-Tie2 and VE-cadherin was significantly increased, while VEGFR2 was markedly suppressed with Kruppel-like factor 2 (KLF2) upregulation after treatment with E-DPSC-CM and T-DPSC-CM (Appendix Fig. 5). To further investigate the interactions of HUVECs and DPSCs, E-DPSCs, and T-DPSCs, the expression of p-Tie2 and its downstream effectors was analyzed in HUVEC + DPSC, HUVEC + E-DPSC, and HUVEC + T-DPSC cocultures. The p-Tie2 and VE-cadherin were significantly increased, while VEGFR2 was notably decreased with KLF2 upregulation in HUVEC + E-DPSC and HUVEC + T-DPSC cocultures (Fig. 1E). These results indicated that E-DPSCs and T-DPSCs inhibit HUVEC sprouting through activating Tie2 and its downstream signaling.
Activated Tie2 Signaling Inhibits HUVEC Sprouting
To further verify the activated Tie2 signaling was responsible for suppressing HUVEC sprouting, human recombinant Ang1 and VE-PTP inhibitor were used to induce Tie2 phosphorylation. The cumulative sprouting length was significantly suppressed in HUVEC + DPSC spheroids when treated with Ang1 or VE-PTP inhibitor (Fig. 2A). The fluorescence intensity of p-Tie2 in HUVEC + DPSC spheroids was much higher after stimulation with Ang1 or VE-PTP inhibitor compared to the control group (Fig. 2B). To explore how the activated Tie2 signaling inhibits HUVEC sprouting, the expression of p-Tie2 and its downstream effectors in HUVEC + DPSC cocultures was analyzed by Western blot. It was found that Ang1 and VE-PTP inhibitor upregulated the expression of p-Tie2, VE-cadherin, and KLF2 but downregulated the expression of VEGFR2 (Fig. 2C). In contrast, Tie2 inhibitor enhanced the HUVEC sprouting in HUVEC + E-DPSC and HUVEC + T-DPSC coculture spheroids (Fig. 3A, 3B). In addition, when treatment with Tie2 inhibitor, the expression of p-Tie2, and VE-cadherin were decreased, and VEGFR2 was significantly increased with KLF2 downregulation in HUVEC + E-DPSC and HUVEC + T-DPSC cocultures (Fig. 3C, 3D). These results demonstrated that the activated Tie2 signaling inhibits HUVEC sprouting by regulating VE-cadherin and VEGFR2 expression.
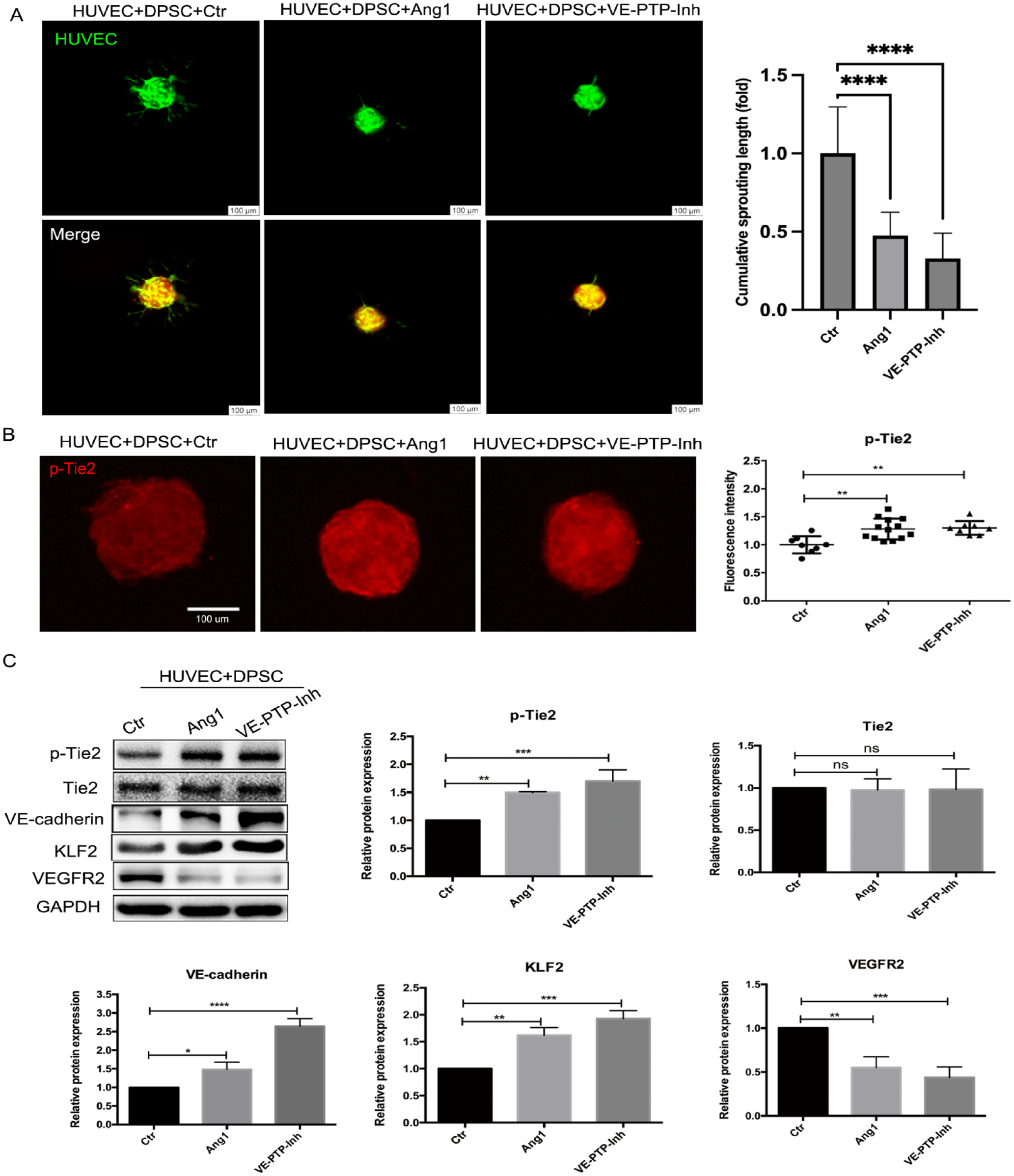
Tie2 signaling was activated by Ang1 and VE-PTP inhibitor in HUVEC + DPSC cocultures. (
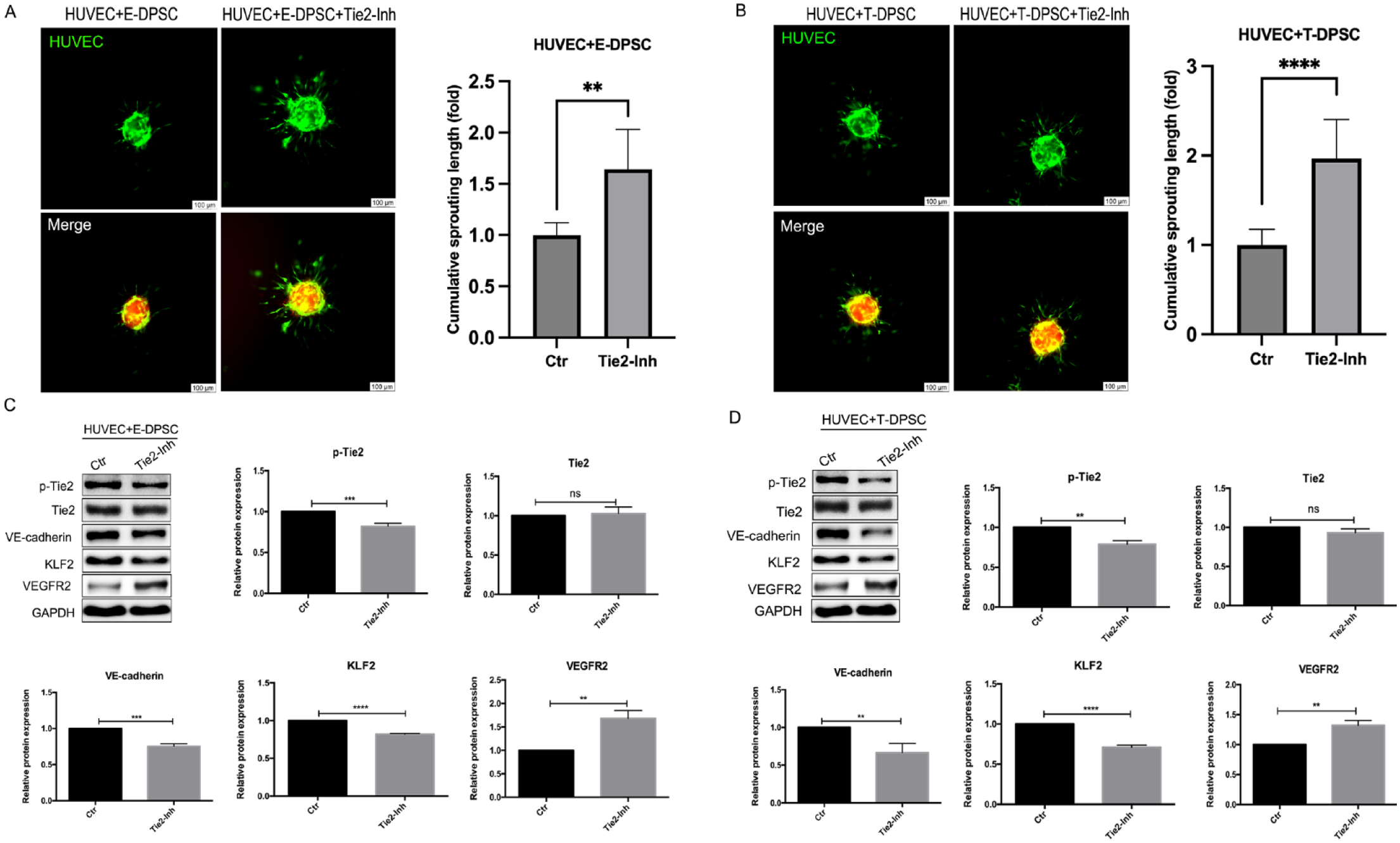
The effects of Tie2 inhibitor on HUVEC sprouting and its downstream effectors. (
Dental Pulp Angiogenesis in the SCID Mouse Model
To investigate the roles of E-DPSCs, T-DPSCs, Ang1, and VE-PTP inhibitor in angiogenesis in vivo, HUVECs and DPSCs, E-DPSCs, or T-DPSCs were encapsulated in GelMA hydrogel and injected into the root segments. The root segments with cells were cultured in EGM2 medium with or without Ang1 and VE-PTP inhibitor for 1 wk and then implanted subcutaneously into the dorsum of SCID mice for 4 wk (Appendix Fig. 6). H&E staining showed there were more perfused and total blood vessels in HUVEC + E-DPSC, HUVEC + T-DPSC, HUVEC + DPSC + Ang1, and HUVEC + DPSC + VE-PTP-Inh groups compared to the HUVEC + DPSC group (Fig. 4A, 4B).
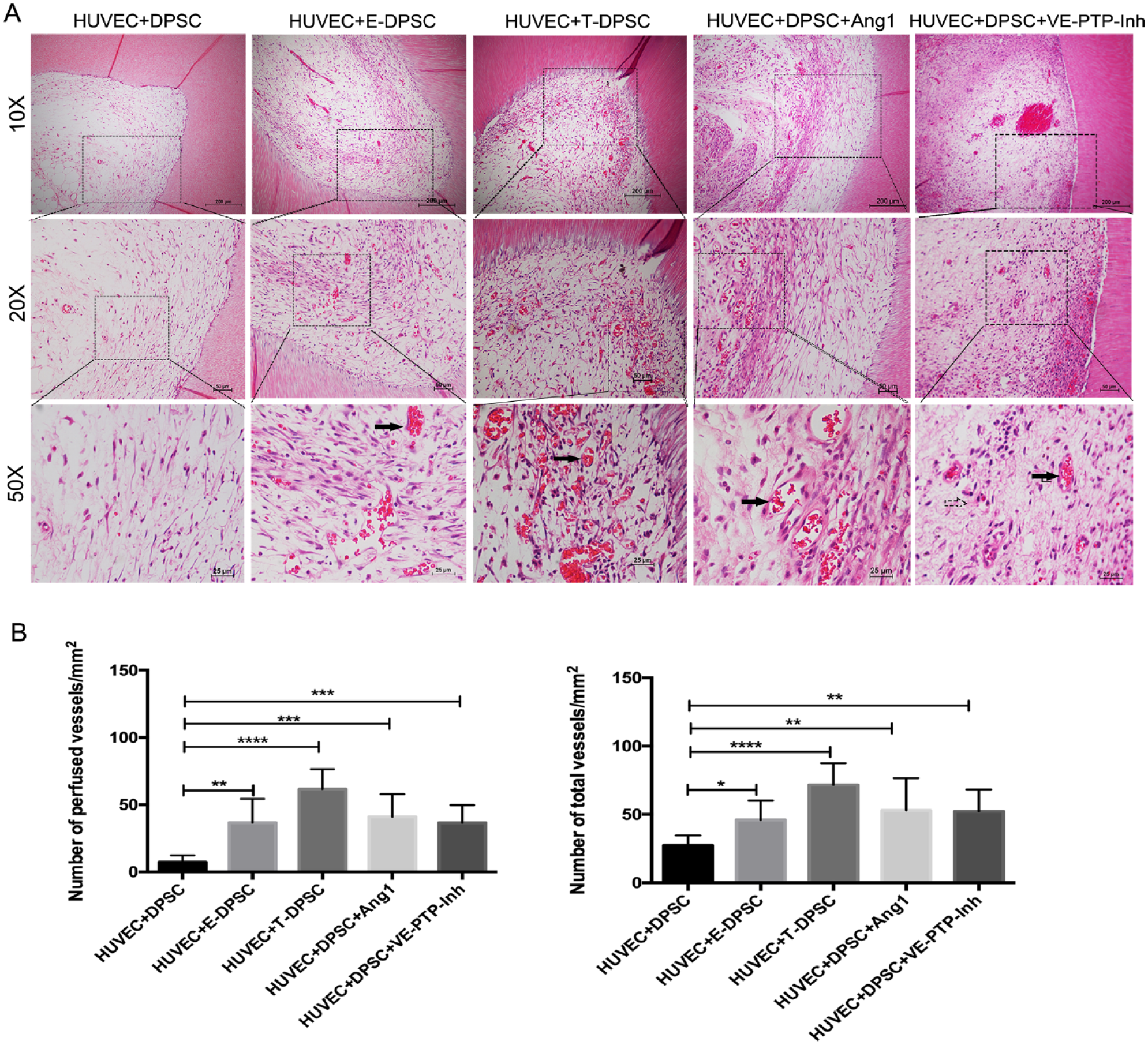
E-DPSCs, T-DPSCs, Ang1, and VE-PTP inhibitor on blood vessel formation in vivo. (
The immunohistochemistry results showed that most of the lumen structures in the sections were stained positive for human CD31 (Fig. 5A), indicating that the implanted HUVECs participated in the blood vessel formation in vivo. In addition, there were more CD31-positive vessels in HUVEC + E-DPSC, HUVEC + T-DPSC, HUVEC + DPSC + Ang1, and HUVEC + DPSC + VE-PTP-Inh groups (Fig. 5C). To further explore the role of the implanted DPSCs in angiogenesis in vivo, the sections were stained with human-specific anti-SM22α antibody to determine the location of the implanted DPSCs. It was found that the SM22α-positive cells enclosed the newly formed lumens, and more SM22α-positive vessels were observed in HUVEC + E-DPSC, HUVEC + T-DPSC, HUVEC + DPSC + Ang1, and HUVEC + DPSC + VE-PTP-Inh groups (Fig. 5B, 5D), which suggest that the implanted DPSCs functioned as the pericyte-like cells in stabilizing the newly formed blood vessels.
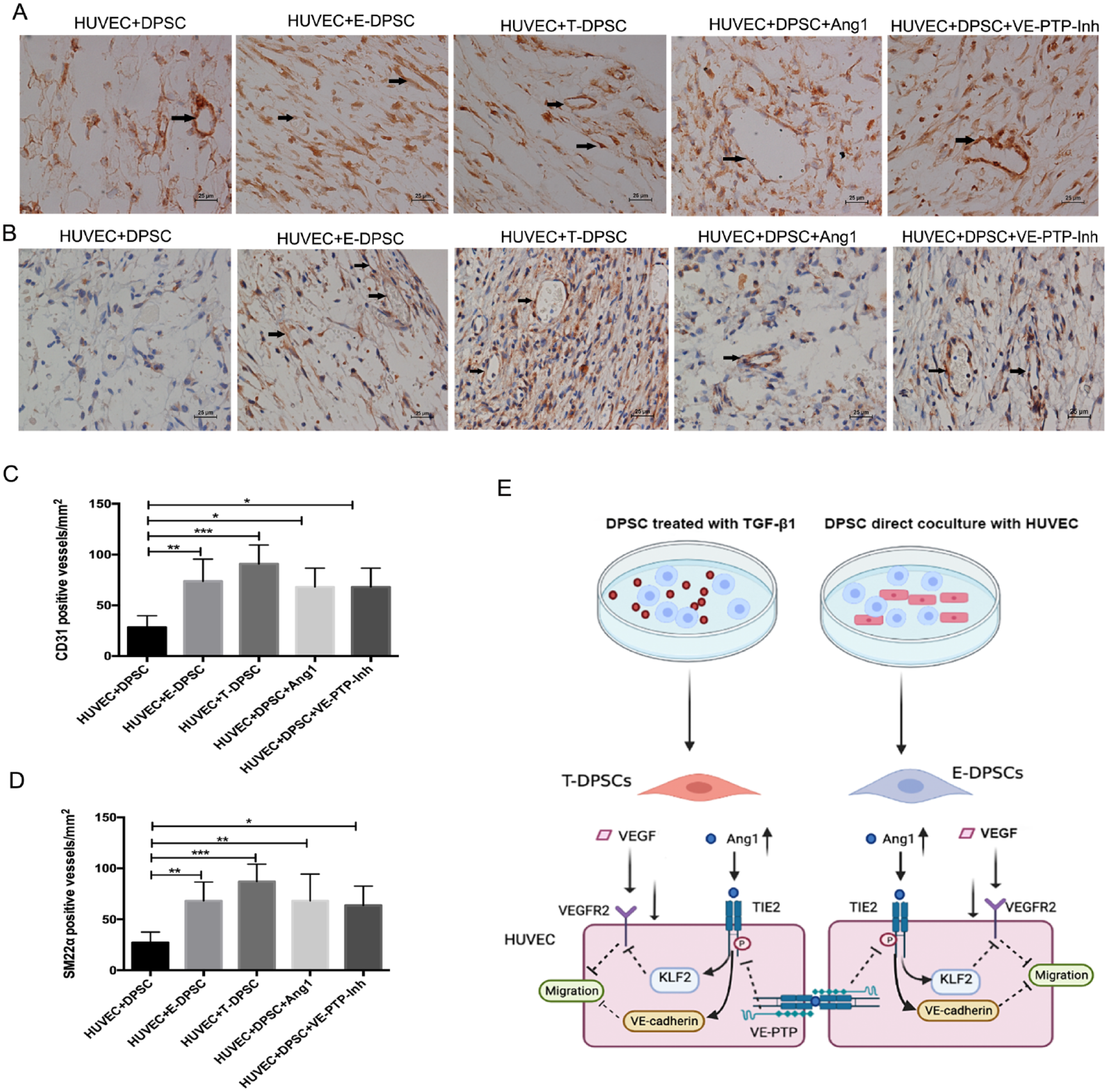
Immunohistochemistry staining for human CD31 and SM22α. (
Discussion
Many strategies have been developed to facilitate angiogenesis in vascular tissue engineering, including the addition of growth factors, such as fibroblast growth factor, VEGF, and Ang1; transplantation of endothelial progenitor cells or stem cells; and cotransplantation of ECs and stem cells (MacAskill et al. 2018; Hage et al. 2022). Although these methods can promote new blood vessel formation, the new vasculatures are prone to be leaky or regressive quickly if not progressing toward maturation (Gonçalves et al. 2007). Therefore, enhancing the stability and increasing the longevity of new blood vessels are critical issues in vascular tissue engineering.
DPSCs are dental pulp–derived MSCs with strong angiogenic potential (Yong et al. 2022). DPSCs promote vascular formation by increasing EC proliferation and migration through secreting VEGF and enhancing the stabilization of newly formed blood vessels by acting as pericyte-like cells (Dissanayaka et al. 2015). However, the mechanisms for the latter part remain largely unknown. Elucidating the mechanisms may unravel the roles of DPSCs in the vascular maturation process and bring much-needed insight into dentin/pulp regeneration.
TGF-β1 treatment or coculture with HUVECs has been verified to induce DPSC differentiation toward SMCs (Zhang et al. 2021, 2023). This study found that SMC-specific markers’ mRNA and protein expression were significantly upregulated in E-DPSCs and T-DPSCs compared to DPSCs. Studies have reported that bone marrow–derived MSCs and adipose stromal cells (ASCs) were differentiated into SMCs when directly cocultured with ECs (Goerke et al. 2012; Merfeld-Clauss et al. 2014). To explore the functions of E-DPSCs and T-DPSCs on HUVEC sprouting, the 3D coculture spheroidal sprouting assay was conducted. The results showed that E-DPSCs and T-DPSCs markedly suppress HUVEC sprouting in 3D coculture spheroids. Similarly, a previous study reported that SMC inhibited EC spouting in 3D coculture spheroids (Korff et al. 2001). These results suggest that E-DPSCs and T-DPSCs enhance HUVEC quiescence and function similarly to SMCs in angiogenic sprouting, offering that E-DPSCs and T-DPSCs may replace limited primary SMCs in vascular tissue engineering.
To explore how E-DPSCs and T-DPSCs inhibit HUVEC sprouting, Ang1 and VEGF expression was analyzed in E-DPSCs and T-DPSCs. Ang1, a proangiogenic factor, can activate its receptor, Tie2, via phosphorylation (Babaei et al. 2003). Ang1/Tie2 signaling regulates EC survival through the PI3K-Akt pathway and is the key contributor to stabilizing new blood vessels (DeBusk et al. 2004). VEGF, which is mainly secreted by mural cells, promotes the proliferation and migration of ECs and further induces angiogenesis when bound to its specific receptor, VEGFR2, which is expressed on the membrane of ECs (Matsumoto and Ema 2014). In this study, the total and secreted Ang1 were markedly upregulated in E-DSPCs and T-DPSCs, while there was no significant difference in VEGF expression and secretion among DPSCs, E-DPSCs, and T-DPSCs. To further reveal whether Tie2 can be activated by Ang1 secreted from E-DPSCs and T-DPSCs, the phosphorylation of Tie2 was analyzed by immunofluorescence staining in 3D coculture spheroids. The fluorescence intensity of p-Tie2 was much higher in spheroids formed by HUVEC + E-DPSC and HUVEC + T-DPSC than in HUVEC + DPSC. Therefore, we infer that the upregulated Ang1 in E-DPSCs and T-DPSCs activated Tie2 signaling and further inhibited HUVEC sprouting.
To further investigate how Ang1/Tie2 signaling inhibits HUVEC sprouting, the expression of p-Tie2 and its downstream signaling effectors was analyzed in HUVECs. Western blot results showed that the expression of p-Tie2 and VE-cadherin was significantly upregulated, while VEGFR2 was markedly decreased along with KLF2 upregulation after treatment with E-DPSC-CM and T-DPSC-CM, as well as in HUVEC + E-DPSC and HUVEC + T-DPSC cocultures. VE-cadherin is an EC-specific adhesion protein that maintains vascular permeability and stability (Giannotta et al. 2013). Activation of Ang1/Tie2 signaling induces RhoA activation, leading to the association of mDia with Src, which blocks the binding of Src to VEGFR2, ultimately preventing VE-cadherin internalization and vascular permeability (Gavard et al. 2008). In addition, Ang1 induced Tie2 trans-association at cell–cell contacts, and the translocated Tie2 formed a complex with VE-PTP to enhance cell adhesions and inhibit paracellular permeability (Saharinen et al. 2008). Studies reported that KLF2 inhibits VEGFR2 expression through binding to the VEGFR2 promoter by competing with Sp1, a transcription factor belonging to the KLF family (Bhattacharya et al. 2005). The downregulated VEGFR2 in HUVECs attenuated the effect of VEGF on EC proliferation and migration, resulting in a more stable vascular structure (Matsumoto and Ema 2014).
VE-PTP, a receptor-type protein tyrosine phosphatase, is mainly expressed in vascular ECs (Vestweber 2021). Inhibition of VE-PTP leads to the phosphorylation of Tie2 and enhances EC junctions (Braun et al. 2019). To investigate the roles of Tie2 signaling and VE-PTP in vascular sprouting, Ang1 and VE-PTP inhibitor were used to induce Tie2 phosphorylation in this study. The results showed that both Ang1 and VE-PTP inhibitor significantly inhibit HUVEC sprouting in 3D coculture spheroids. The fluorescence intensity of p-Tie2 in HUVEC + DPSC spheroids treated with Ang1 or VE-PTP inhibitor was higher than in the control group. In addition, both Ang1 and VE-PTP inhibitor upregulated the expression of p-Tie2, VE-cadherin, and KLF2 and downregulated the expression of VEGFR2 in HUVEC + DPSC cocultures. Inhibition of VE-PTP induces Tie2 activation and the phosphorylation of FGD5 (FYVE, RhoGEF, and PH domain containing 5), a GTP-exchange factor. It promotes FGD5 translocation to EC contacts, which is essential for stabilizing cell–cell junctions (Braun et al. 2019). In addition, VE-PTP can associate with VE-cadherin via extracellular domains and reduce VE-cadherin phosphorylation and cell layer permeability (Nawroth et al. 2002).
The tooth segments model is widely used in vivo to study the vascularization in dental pulp (Nakashima et al. 2019). In vitro studies have verified that both E-DPSCs and T-DPSCs have similar functions and properties to SMCs. The in vivo study further demonstrated the roles of DPSCs in stabilizing newly formed blood vessels. More perfused vessels were observed in HUVEC + E-DPSC and HUVEC + T-DPSC groups, and both the implanted HUVECs and DPSCs were found to participate in the formation of blood vessels in vivo. Most important, human SM22α-positive cells were observed to enclose the perfused lumen structures, suggesting the pericyte-like functions of DPSCs during vascular maturation. Besides, Ang1 and VE-PTP inhibitor pretreatment groups had more perfused blood vessels. The newly formed blood vessels are prone to regress quickly if not progressing toward maturation (Gonçalves et al. 2007). Mural cells are essential to maintain the stability and extend the longevity of newly formed blood vessels. E-DPSCs and T-DPSCs have similar functions to SMCs, which may support vascular stability and sustain vascular longevity. In vitro studies have shown that besides E-DPSCs and T-DPSCs, Ang1 and VE-PTP inhibitor also suppressed HUVEC sprouting and induced Tie2 phosphorylation, implying that the activated Tie2 signaling contributes to stabilizing newly formed blood vessels. On the other hand, the fragile immature vessels may collapse under shear pressure after transplantation and become less conducive to blood flow (White et al. 2012). Blood flow in tissue constructs with immature vasculatures may cause turbulence that leads to the endothelium and platelets forming blood clots, obstructing blood circulation in the vessels (Ben-Shaul et al. 2019). In addition, mature prevascularized constructs could facilitate quicker anastomosis with the host tissue vasculatures (Chen et al. 2009). These could explain why HUVEC + E-DPSC, HUVEC + T-DPSC, Ang1, and VE-PTP inhibitor pretreatment groups had more perfused blood vessels.
Obtaining sufficient blood perfusion in engineered tissue is critical for tissue survival in stem cell–based tissue regeneration. Prevascularization of grafted tissue is a promising strategy to speed up the anastomosis of transplanted tissue and host vasculatures. HUVECs and human bone marrow stromal cells (BMSCs) cocultured for 7 d prior to implantation generated more bone and vascularity than the non-prevascularized group (Subbiah et al. 2021). In addition, a study on in vitro coculturing ECs and fibroblasts for different time points (1, 7, and 14 d) before implantation revealed more blood perfusion and graft–host vascular anastomosis in the long-term (14 d) cocultured grafts, indicating the vessel maturity in grafted tissues is critical for blood perfusion and anastomosis with host vasculatures (Ben-Shaul et al. 2019). Atlas et al. reported that prevascularized constructs with EC and stem cells from human exfoliated deciduous teeth (SHED) enhanced blood perfusion in grafted tissue compared to EC or SHED-alone culture. In this process, SHED was recruited by PDGF-BB released from ECs and functioned as pericyte-like cells in stabilizing capillaries, which enhanced blood perfusion within the grafted tissues (Atlas et al. 2021). To enhance the SMC-like properties, DPSCs were treated with TGF-β1 (T-DPSCs) or cocultured with ECs (E-DPSCs) in vitro in this study. Indeed, E-DPSCs and T-DPSCs more closely simulate SMCs than DPSCs, with higher SMC-specific markers’ expression. E-DPSCs and T-DPSCs can function as pericyte-like cells in the 3D sprouting assay. Our in vivo study further revealed that pre-coculture of HUVECs and E-DPSCs or T-DPSCs could enhance vessel stability and promote blood perfusion in grafted tissues in vivo.
In summary, this study demonstrated the functions and properties of E-DPSCs and T-DPSCs in stabilizing blood vessels in vitro and in vivo. Ang1/Tie2/VE-cadherin and VEGF/VEGFR2 signaling pathways were involved in the vascular stabilization induced by DPSCs (Fig. 5E). The two approaches to differentiate DPSCs into SMC-like cells may be helpful for the development of new dental pulp regeneration strategies.
Author Contributions
Y. Zhang, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; S. Lin, J. Liu, Q. Chen, J. Kang, contributed to conception, design, data critically revised the manuscript; J. Zhong, M. Hu, M. S Basabrain, Y. Liang, contributed to data acquisition, analysis, and interpretation, critically revised the manuscript; C. Yuan, C. Zhang, contributed to conception, design, data interpretation, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345231210227 – Supplemental material for Ang1/Tie2/VE-Cadherin Signaling Regulates DPSCs in Vascular Maturation
Supplemental material, sj-docx-1-jdr-10.1177_00220345231210227 for Ang1/Tie2/VE-Cadherin Signaling Regulates DPSCs in Vascular Maturation by Y. Zhang, S. Lin, J. Liu, Q. Chen, J. Kang, J. Zhong, M. Hu, M.S. Basabrain, Y. Liang, C. Yuan and C. Zhang in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (82170938, 81970934), a grant from Shenzhen Science and Technology Innovation Committee (JCYJ20210324135806017), and the RGC General Research Fund, Hong Kong (2022/2023) (Ref No. 17105422) to C. Zhang.
Data Availability Statement
The data of this study are available from the corresponding author upon reasonable request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
