Abstract
Background
Traumatic brain injury (TBI) is a leading cause of death and permanent disability, with the burden being higher in low- and middle-income countries (LMICs). Effective management during the acute phase is critical for improving survival and long-term outcomes. For this reason, evidence-based decision-making is essential to delivering consistent, high-quality care. The objective of this review is to assess the safety and effectiveness of standardized care in adults with moderate or severe TBI in LMICs.
Materials and Methods
A literature search was conducted in MEDLINE/PubMed, Embase, CENTRAL, LILACS, ClinicalTrials.gov and WHO-ICTRP. Conference proceedings from NCS, SCCM, and ESICM were searched for unpublished studies. Randomized controlled trials (RCTs) and non-randomized (NRSs) controlled studies comparing protocolized with non-protocolized care for patients 14 years or older with acute moderate or severe TBI in LMICs were included. Studies were independently assessed for inclusion, data extraction, and risk of bias. Study flaws were assessed using the Cochrane risk of bias tool, and quality-of-evidence using the GRADE approach.
Results
Seven studies were included, involving a total of 1821 participants. Five of these employed a quasi-experimental before-and-after design, while two used a quasi-experimental design with a non-equivalent control group. NRS recruited participants from Cuba, Argentina, Bolivia, Ecuador, Venezuela, Uruguay, Colombia, Brazil, Egypt, and Thailand. All included studies were deemed to have a high risk of bias. Very low to low-quality evidence suggests that protocolized care may provide benefits for adults with moderate to severe TBI in LMICs, including reduced mortality, improved cognitive outcomes, decreased hospitalization-related complications, and increased satisfaction with the care process. However, there appears to be little or no effect on quality-of-life scores and length of hospital stay. The impact of standardized care on functionality, language processing abilities, and ICU stay remains uncertain.
Conclusions
Very low-quality evidence suggests that protocolized care may provide benefits for adults with moderate to severe TBI in LMICs. Higher-quality research is imperative to rigorously assess the safety and effectiveness of this intervention.
PROSPERO registration number
CRD 420251074998
Keywords
Background
Traumatic brain injury (TBI) is a major global public health problem and one of the leading causes of death and disability among young adults worldwide. 1 It is estimated that approximately 69 million people suffer TBI annually, with nearly 90% of this burden occurring in low- and middle-income countries (LMICs). 2 In LMICs, the management of moderate (mTBI) and severe (sTBI) is particularly challenging due to limitations such as shortages of specialized healthcare professionals, inadequate infrastructure, and fragmented healthcare systems.3,4 These factors are closely linked to poor clinical outcomes, including elevated mortality rates, a higher incidence of complications, and long-term disability.3,4
Additionally, the lack of standardized care protocols and the inconsistent implementation of evidence-based clinical guidelines further exacerbate these disparities in care delivery in LMICs. 4 This is particularly evident in critical aspects of care such as intracranial pressure monitoring, initial hemodynamic stabilization, management of intracranial hypertension, and other time-sensitive therapeutic decisions during the acute phase. 5 A meta-epidemiological study involving 59,197 patients across 31 LMICs reported a pooled TBI mortality rate of 16.7%, three to four times higher than in high-income countries, highlighting how inconsistent implementation of evidence-based care contributes to poor clinical outcomes. 6
In contrast, experiences from high-income countries provide compelling evidence that implementing structured strategies—such as clinical bundles, multidisciplinary protocols, clinical guidelines, or structured care pathways—can significantly reduce care variability and improve outcomes in patients with TBI. 7 For instance, adherence to the Brain Trauma Foundation (BTF) guidelines in the United States has been associated with reduced mortality, shorter hospital stays, and better functional recovery. 8
Standardized or protocolized care in the management of TBI can reduce practice variability, improve timeliness of clinical decisions, and optimize multidisciplinary coordination in neurocritical care.5,9,10 However, its impact in LMICs remains uncertain. Structural and financial constraints, as well as differences in health system organization, culture, and human resource availability, may limit both feasibility and impact of these strategies in lower-resource contexts.4,5,11 This highlights the need for context-sensitive evaluations of standardized care models in LMICs, particularly for moderate and severe TBI, where the burden is greatest, and the evidence is scarcest.4,5,11
Given the substantial burden of moderate and severe TBI in LMICs, and the proven benefits of standardized care in high-income settings, evaluating the applicability and impact of such strategies in resource-limited environments is both timely and necessary. 4 The findings from this review could inform the development and contextual adaptation of clinical guidelines, care bundles, and feasible implementation strategies tailored to the realities of LMICs. Moreover, they may support evidence-based clinical decision-making, guide health policy formulation, and promote more efficient use of healthcare resources, generating tangible benefits for patients, providers, and health systems in these countries. Therefore, the objective of this review is to assess the safety and effectiveness of standardized care for the adult population with moderate or severe TBI in LMICs. The results may also help identify priority areas for future research and contribute to global efforts aimed at reducing mortality and disability from TBI in vulnerable settings.
Methodology
The protocol for this systematic review was designed in accordance with the recommendations of the Cochrane Handbook for Systematic Reviews, 12 and was registered in Prospero (https://www.crd.york.ac.uk/PROSPERO/view/CRD420251074998). The results are presented as per the PRISMA statement, 13 and given the nature of this study, ethics committee approval was not required.
Electronic searches were conducted in the MEDLINE/PubMed, Embase, CENTRAL, and LILACS databases, and for ongoing studies ClinicalTrials.gov and WHO International Clinical Trials Registry Platform (WHO- ICTRP) were queried. Gray literatur were identified in the IBECS, BINACIS, CUMED, WPRIM, BDENF, LIPECS and LIS platforms using the LILACS interface, and conference proceedings from the NCS Annual Meeting, SCCM Critical Care Congress, and the European Society of Intensive Care Medicine were searched for unpublished studies. Finally, the reference lists of all included studies and expert content were examined to identify additional relevant publications. There were no language or date restrictions. All databases were searched from inception until June 16, 2025 (Supplemental material, Table A).
Randomized controlled trials (RCTs) and non-randomized (NRSs) controlled studies comparing protocolized versus non-protocolized TBI care for patients 14 years or older who presented with moderate or severe TBI within 24 h of injury or deteriorating to that level within 48 h of injury in lower- and middle-income countries were included. Severity of TBI was defined using the Glasgow Coma Scale (GCS), 14 the GCS motor or eye (GCSm-e) component if intubated, 15 or through the Abbreviated Injury Score (AIS). 16 Studies that recruited participants with GCS of 3 with bilateral fixed and dilated pupils on admission, with injuries otherwise considered non-survivable, re-admissions for the same head injury, and patients with clear non-traumatic causes of decreased brain function (eg alcohol, drugs, sedatives used in the receiving hospitals before transport to trauma center, shock, extreme hypoxia, hypoglycemia, and metabolic acidosis) were excluded.
For this review, protocolized care was defined as the implementation 17 of a clinical practice guideline, consensus-based management protocol, clinical pathway, patient care bundles, clinical trial protocol, or any other directive designed to standardize 18 the acute care of patients with TBI. Non-protocolized care was defined as usual care (eg, standard practice without a system of rules that explain the correct conduct and procedures to be followed in patients with TBI). Primary outcomes were mortality at the end of the follow-up period; functional and cognitive status at discharge, 3, 6, and 12 months; incidence of hospitalization-related complications, and quality of life. Secondary outcomes included the use of general or specific interventions directed at managing intracranial hypertension, in-hospital length of stay (LOS), intensive care unit length of stay (ICU-LOS), costs, and satisfaction related to the care process.
Two authors independently screened all the titles and abstracts retrieved from the search strategy to determine which studies should be assessed further, and independently investigated the full-text articles of all potentially relevant articles. 19 Any disagreements were resolved through discussion. All excluded articles after full-text assessment, and the reasons for exclusion, are presented in the characteristics of excluded studies table. A data extraction form was designed to obtain the relevant information from the included studies. 19 Two authors independently extracted the data, and disagreements were solved through discussion until a consensus was reached. 19 Extracted data were location of the study and setting; study design; power calculation performed; inclusion and exclusion criteria; baseline information of the participants; type of intervention and components; type of comparison; number of participants enrolled, randomized, excluded after randomization, and analyzed; adherence to the planned intervention and other interventions in the group under evaluation; outcomes definition; time of follow-up of participants to measure outcomes; funding sources; and ethical issues. When information regarding any of the above was unclear, the authors of the original trial reports were contacted. 19
Two review authors independently assessed the risk of bias for included studies using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (RoB-2 tool) 20 or the risk of bias ROBINS-I tool for non-randomized controlled studies (NRS). 21 Disagreements were resolved by consulting a third review author. The review authors who assessed the risk of bias in the included studies were experts in the theme and methodology. To estimate the magnitude of the effect, risk ratios (RRs) with corresponding 95% confidence intervals (CIs) for dichotomous outcomes, and mean differences (MDs) or standardized mean differences (SMDs) for continuous outcomes were planned. For data synthesis, random-effects meta-analysis was considered if substantial statistical heterogeneity was detected, guided by both clinical sources of heterogeneity and statistical measures, including I2, Tau2, and the chi-square test. 22
However, based on the nature of the available evidence and the methodological designs of the included studies—and given that meta-analyses of pre-post values are generally discouraged due to their potential for bias—it was not feasible to carry out the planned analysis. 23 Pre-post comparisons assess within-group changes from baseline to post-intervention but are problematic because the two measurements are not independent. 23 Furthermore, such changes may reflect natural progression or contextual factors unrelated to the intervention, making it difficult to isolate its true effect. 23 Consequently, we conducted a qualitative and quantitative data synthesis, 24 in accordance with the Synthesis Without Meta-analysis (SWiM) guideline, to ensure a transparent and valid presentation of the intervention's effect. 24
For meta-analyses involving more than 10 studies, the assessment of funnel plot asymmetry and the implementation of a formal test were planned to explore publication bias. 22 Otherwise, an exploratory analysis was done through visual inspection. Subgroup and sensitivity analyses were planned according to the setting (low vs middle-income countries; clinical practice guideline vs other directives) and the risk of bias of the included studies (low vs unclear or high risk of bias), respectively. 22 The GRADE approach was implemented to assess the evidence quality and produce a ‘Summary of findings’ table for each outcome. 25 This methodology downgraded evidence quality depending on the presence of the factors, such as study limitations, inconsistency of results, indirectness of the evidence, imprecision, and publication bias. 25 ‘Summary of findings’ tables can be found in the Supplemental material.
Results
A total of 2444 references were retrieved and 2243 studies were screened after duplicates were removed. Full-text reviews were carried out for 14 studies (Figure 1). Seven studies met the inclusion criteria,26–32 and seven33–39 were excluded because the studied population primarily consisted of participants with mild TBI37–39 or because they examine the effect of ICU adherence rate on in-hospital mortality without a formal comparison group, 36 assessed effects of a neurocognitive intervention during the late phase of mTBI 32 or were duplicated publications.34,35 Included and excluded studies are described in Supplemental material, Table B.
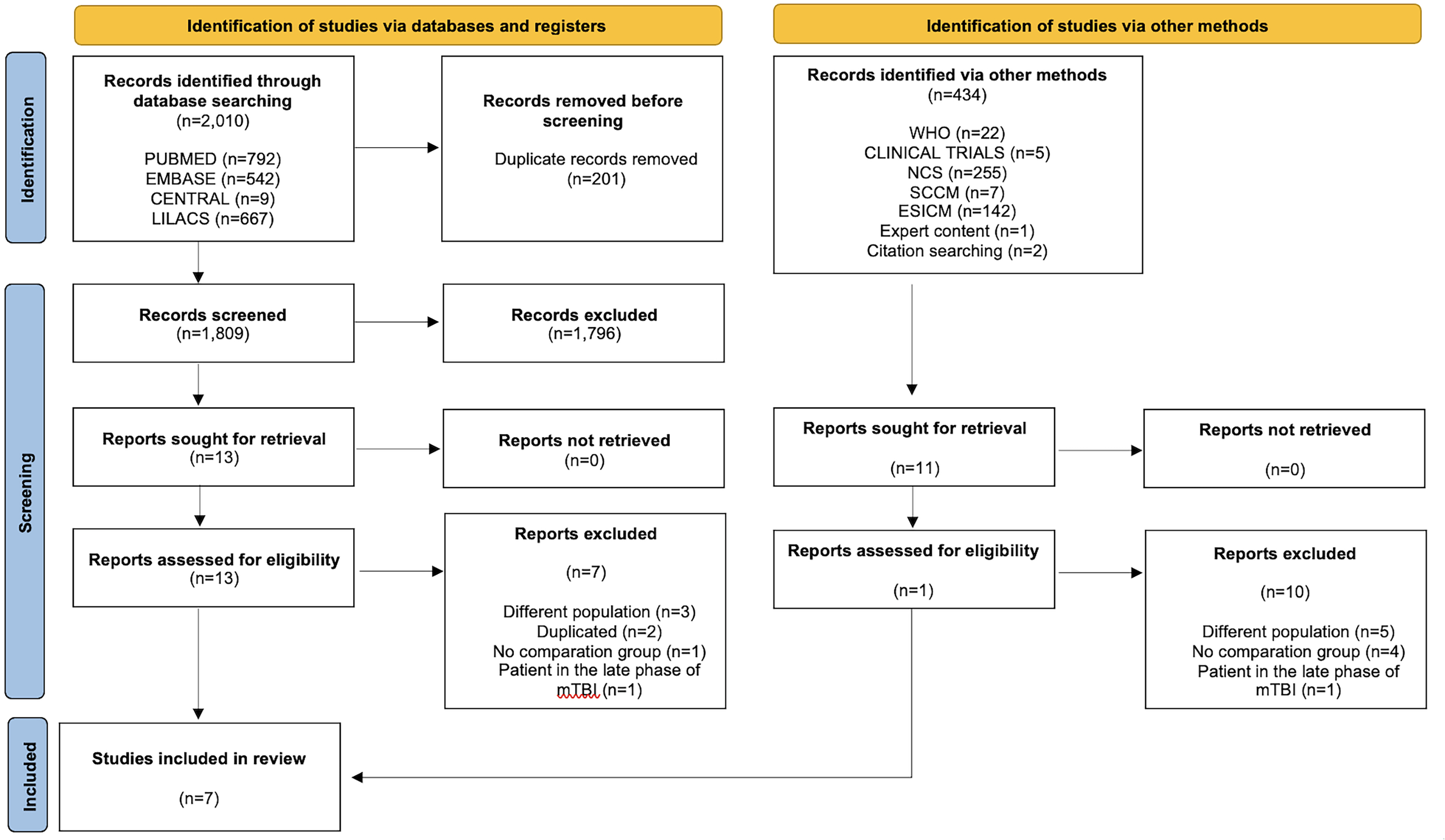
Study flowchart. Inclusion of studies at different stages for this systematic review.
No RCTs were identified. All studies that met the inclusion criteria were NRS, providing a sample size of 1821 participants. Five studies employed a quasi-experimental before-and-after design,28–32 and two used a quasi-experimental design with a non-equivalent control group.26,27 Six NRS mentioned protocol approval by an ethics committee,26–31 and only three calculated the sample size a priori.26,27,31 All studies were published in English (2010 to 2024), three were funded by academic entities,28–30 and two by a non-governmental organization.26,27 The NRS recruited participants from high-complexity institutions located in Cuba, 32 Argentina, Bolivia, Ecuador, Venezuela, Uruguay,26,27 Colombia,26,27,30 Brazil, 28 Egypt, 31 and Thailand, 29 some of which were university hospitals.26,27,30,32 All but two studies26,27 were conducted at a single center.
The study population was characterized by being predominantly male (77%–93%), older than 14 years (age range 29-92 years), with a diagnosis of moderate28,32 or severe26–32 non-penetrating head trauma in the acute phase (<24 h). All studies used the GCS as a tool to grade trauma severity (eg moderate 9-12 and severe ≤ 8), except for two that allowed the entry of participants with a GCS ≤ 5 in the motor component in case of orotracheal intubation26,27 and another NRS that implemented the combination of an AIS score ≥ 3 in the presence of GCS ≤ 8 30 as a selection criterion. Participants who died within 48 h of admission 32 had a GCS of 3 and fixed pupils at enrollment,26,27 died as a direct result of other traumatic injuries,26,27,32 or had another reasonably documented cause of altered consciousness 30 were excluded. Twenty-five to fifty-five percent of participants were transported directly to the participating institution after the trauma occurred. Motor vehicle accidents (48-90%), falls (5-20%), or being pedestrian (6-16%) were the most common causes of injury, with CT findings classified as Marshall V (0-50%), IV (0-50%), III (12-34%), II (20-35%), or I (0-35%).26,32 At the time of assessment, 46% to 93% of participants had other moderate-to-severe associated injuries (mean ISS range 16 to 45 points)26,27,32 (Supplemental material, Table B).
Six of the retrieved studies27–32 focused on the surveillance of patients with moderate to severe TBI, using primarily clinical and radiological monitoring. Only one study employed intracranial pressure (ICP) measurement in selected cases. 26 Overall, the interventions addressed in these studies included advanced airway management,26–32 continuous monitoring of vital parameters such as heart and respiratory rate,26–32 arterial oxygen saturation,27,30 partial pressure of CO₂,27,28,30 and blood pressure. 27 One study also monitored central venous pressure, targeting values between 10 and 12 cmH₂O. 27 Neurological assessments were performed hourly, with an emphasis on the Glasgow Coma Scale, and pupillary size and reactivity.27,28,30,32 Brain imaging via tomography was conducted upon admission, at 48 h, and between 5 and 7 days after hospital admission.26–28 One study recommended follow-up CT scans upon admission, at 12 h if the initial scan was within 4 h of injury, and then at 24 and 48 h. 32 All studies allowed for repeat imaging in cases of clinical deterioration, regardless of timing.26–32
General care measures for TBI patients included elevating the headrest to 30 degrees,27,30 maintaining a neutral head and neck position,27,30 preventing and managing hyperthermia,27,28,32 initiating early enteral nutrition,27,29 providing anticonvulsant prophylaxis,27,28,32 administering gastric protection agents, 27 moisturizing the skin, and starting antithrombotic therapy.27,29 Some protocols aimed to maintain arterial oxygen saturation above 90% 27 or 95%, 28 with partial pressures of oxygen above 60 mm Hg 27 or 100 mm Hg. 28 Hemoglobin levels were targeted above 7 g/dL.27,28 Vasopressor support was employed as needed to maintain systolic blood pressure above 90-100 mm Hg 28 or mean arterial pressure (MAP) above 70 mm Hg, 27 with vasopressors being permitted when necessary.27–29,32 All studies emphasized maintaining partial pressure of oxygen within 35 to 40 mm Hg,27,28 and consulting neurosurgery for intervention in cases of space-occupying lesions.26–28,32 One study implemented strict glycemic control, maintaining blood glucose levels within the range of 135-145 mg/dL, 28 sodium levels between 135-145 mEq/L, 28 and pH values between 7.35 and 7.45. 28 Sedation and analgesia were administered appropriately across all NRS levels.27–30,32 In cases of intracranial hypertension, hyperventilation maneuvers were encouraged,27,28,32 and hyperosmolar therapy with hypertonic saline or mannitol was used according to the patient's clinical profile.27,28,32 Additional measures included barbiturate infusions and neuromuscular blockade as a last resort 32 (Supplemental material, Table C).
Four studies26–2830 used an appropriate analysis method, controlling the association of interest for the presence of confounding variables, were measured in a valid and reliable manner, hence the unlikely risk of confounding bias. The remaining three studies29,31,32 did not implement an appropriate analysis and were judged to have a high risk of bias. Regarding the participant selection domain, all studies26–32 were graded as low risk of bias since participant selection was not based on their characteristics and because the start of follow-up and the beginning of the intervention were relatively homogeneous for the entire study population. Five NRS26–29,31 were appraised as low risk of bias for the intervention classification domain since they provided detailed information regarding the different components of the management protocol (eg, the intervention was clearly defined). Two NRS30,32 did not provide sufficient information in this regard.
Only one study 31 reported the percentage of adherence to the protocol implemented in the participating population, in which deviations were found to be balanced across the groups, hence the low risk for the intervention deviation domain. The remaining six studies26–30,32 did not provide sufficient information to analyze this aspect. Two NRS26,27 were graded as high risk of bias for the incomplete data domain because the outcomes were available only for a portion of the participants, creating uncertainty as to whether the results are robust to the presence of missing data. The remaining studies28–32 were assessed as low risk of bias for this domain. Two NRS29,31 were appraised as prone to outcome measurement bias, given that the measurement of the results could have been influenced by the knowledge of the intervention received. The other studies were evaluated as low risk of bias.26–28,30,32 Finally, selective reporting bias was considered unlikely in all NRS,26–32 as it is possible that the effect estimators were not selected based on multiple measurements of outcomes, differences between subgroups, or as result of multiple comparisons (Figure 2).
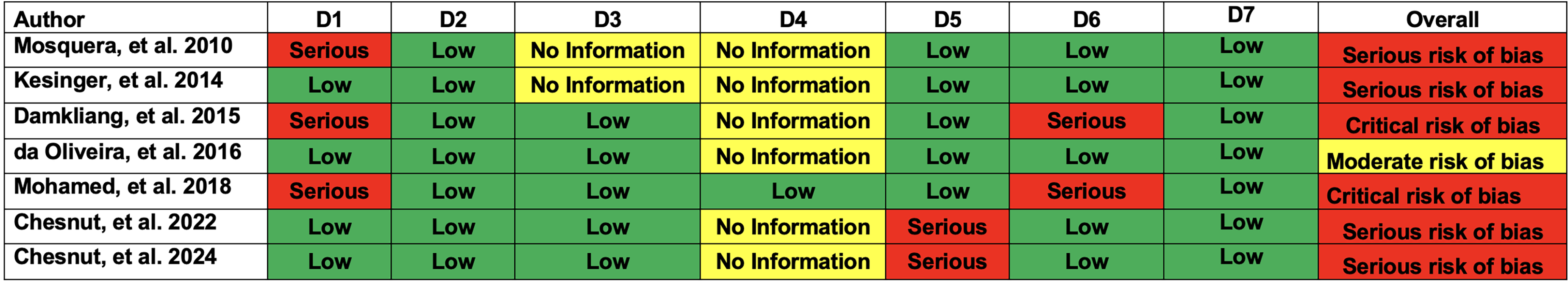
Risk of bias in included studies.
Intervention Effect: Primary Outcomes
Mortality at the End of the Follow-Up Period
There was very low-quality evidence (downgraded for serious risk of bias) for this outcome, reported by five studies involving 1552 patients.26–28,30,32 The implementation of protocolized care may reduce mortality at the end of the study period. One study 32 documented a difference between treatment groups for patients with mTBI [33% pre- versus 17% post-intervention; p = .001] and sTBI [97% pre- versus 82% post-intervention; p = .001]. Three studies found reduced mortality in patients with sTBI when protocol-based care was instituted, even when adjusted for confounding variables [36% pre- versus 21% post-intervention; p < .01, aHR 0.59, 95% CI 0.46–0.76 27 ; 12.6% pre- versus 7.3% post-intervention; p < .05, aOR 0.40, 95% CI 0.20–0.81 28 ; and 38% pre- versus 18% post-intervention; p = .02, aOR 0.24, 95% CI 0.07–0.83 30 ]. Finally, a fifth study reported a trend toward lower 6-month mortality in patients with sTBI (36% vs 25%); however, this difference did not reach statistical significance and was associated with an imprecise effect estimate [HR 0.69, 95% CI 0.43–1.10] 26 ] (Figure 3).
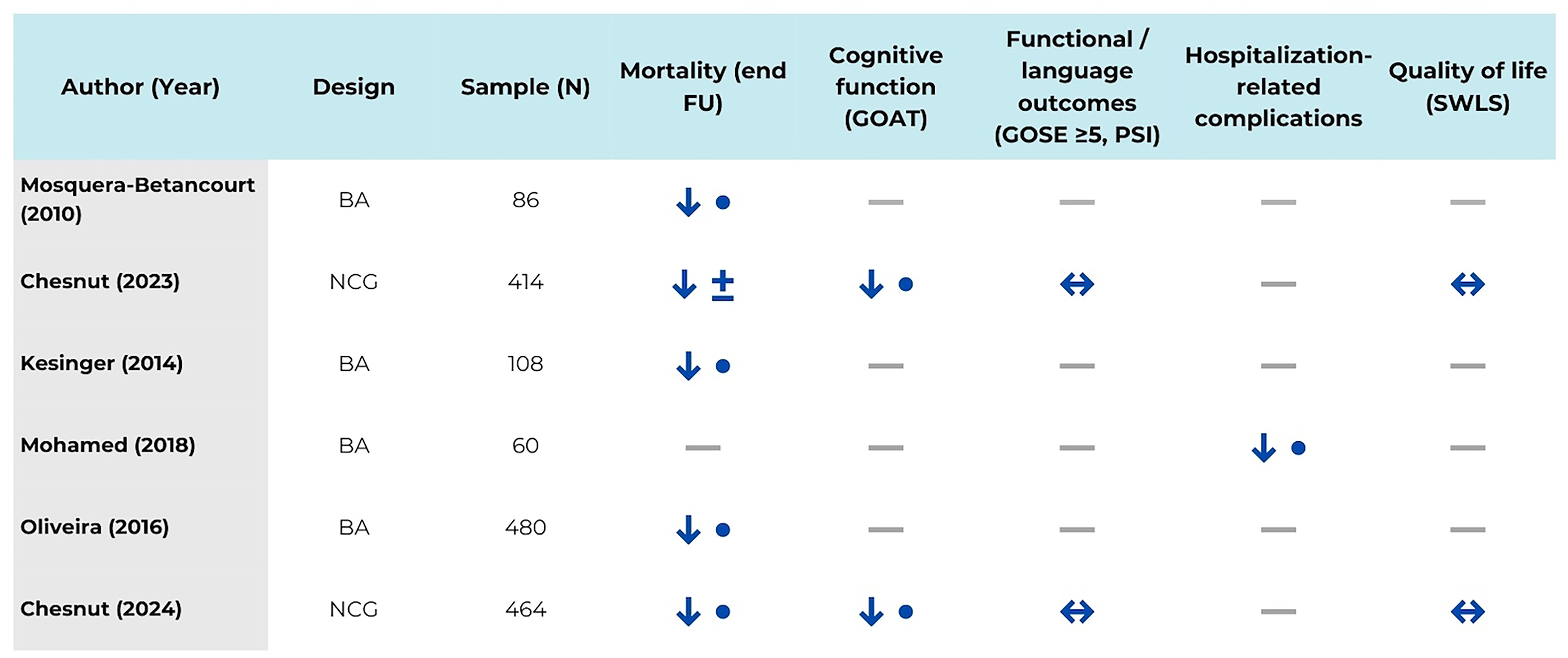
Matrix of primary outcomes across included studies. Direction of effect is shown for each outcome as favoring protocolized care
Functional and Cognitive Status at Discharge, 3, 6, and 12 Months
There was very low-quality evidence (downgraded for serious risk of bias, imprecision, and inconsistency) for these outcomes, reported by two NRS with 878 participants.26,27
Cognitive outcomes were assessed at hospital discharge and at 6 months of follow-up. At discharge, protocolized care was associated with higher cognitive performance, as measured by the Galveston Orientation and Amnesia Test (GOAT), with improvements observed in both studies (5.5 points pre-intervention vs 24.5 post-intervention, p = .0126; and 5.5 vs 21.4 points, p = .0127). Similarly, at 6 months of follow-up, higher GOAT scores were reported in patients managed with standardized care (33.5 vs 51 points, p = .0426; and 33.5 vs 47.8 points, p = .0227) (Figure 3).
In contrast, functional and language-related outcomes assessed at 6 months showed inconsistent findings. Functional status evaluated using the Glasgow Outcome Scale–Extended (GOSE ≥5), improved in one study (43% pre-intervention vs 59% post-intervention, p = .0126) but not in the other (43% vs 51%, p = .4227). Likewise, language processing speed, assessed using the Psychological Status Inventory (PSI), showed discordant results between studies (57.2 vs 63.3 points, p = .0126; and 57.2 vs 59.7 points, p = .5027). Neither study reported functional or cognitive outcomes beyond 6 months of follow-up, and no data were available at 12 months (Figure 3).
Hospitalization-Related Complications
Very low-quality evidence (downgraded for a very serious risk of bias and imprecision) was reported in one NRS with 60 participants. 31 Standardized care might reduce the incidence of hyperglycemia [43% pre- versus 13% post-intervention; p < .05], pressure ulcers [16% pre- versus 0% post-intervention; p < .05], hyperthermia episodes [80% pre- versus 40% post-intervention; p < .05], and procedure-related pain [44% versus 0% post-intervention; p < .05] in patients with sTBI. No cases of deep vein thrombosis were reported 31 (Figure 3).
Quality-of-Life (QoL)
Very low-quality evidence (downgraded for serious risk of bias and imprecision) for the QoL outcome was reported in two NRS with 878 participants.26,27 Patients with protocolized care may have little or no difference in QoL scores measured with the Satisfaction with Life Scale (SWLS) [14.7 points pre- versus 15.9 points post-intervention; p = .17, 26 and 23.8 points pre- versus 20.9 points post-intervention; p = .21 27 ] in sTBI population (Figure 3).
Intervention Effect: Secondary Outcomes
Treatments Directed to Management of Intracranial Hypertension (TDIH)
There was very low-quality evidence (downgraded for serious risk of bias and imprecision) for this outcome, reported by four studies involving 1031 patients.26,27,29,30 Protocolized care could increase the use of head of bed elevation at 30 degrees [6.3% pre- versus 75% post-intervention; p = .00], 29 with continuous CO2 monitoring [0% pre- versus 56% post-intervention; p = .00 29 ], blood transfusion [3% pre versus 13% post-intervention; p = .00 30 ], and hypertonic fluid administration [38% pre- versus 63% post-intervention; p = .01 30 ] in patients with sTBI (Figure 4).
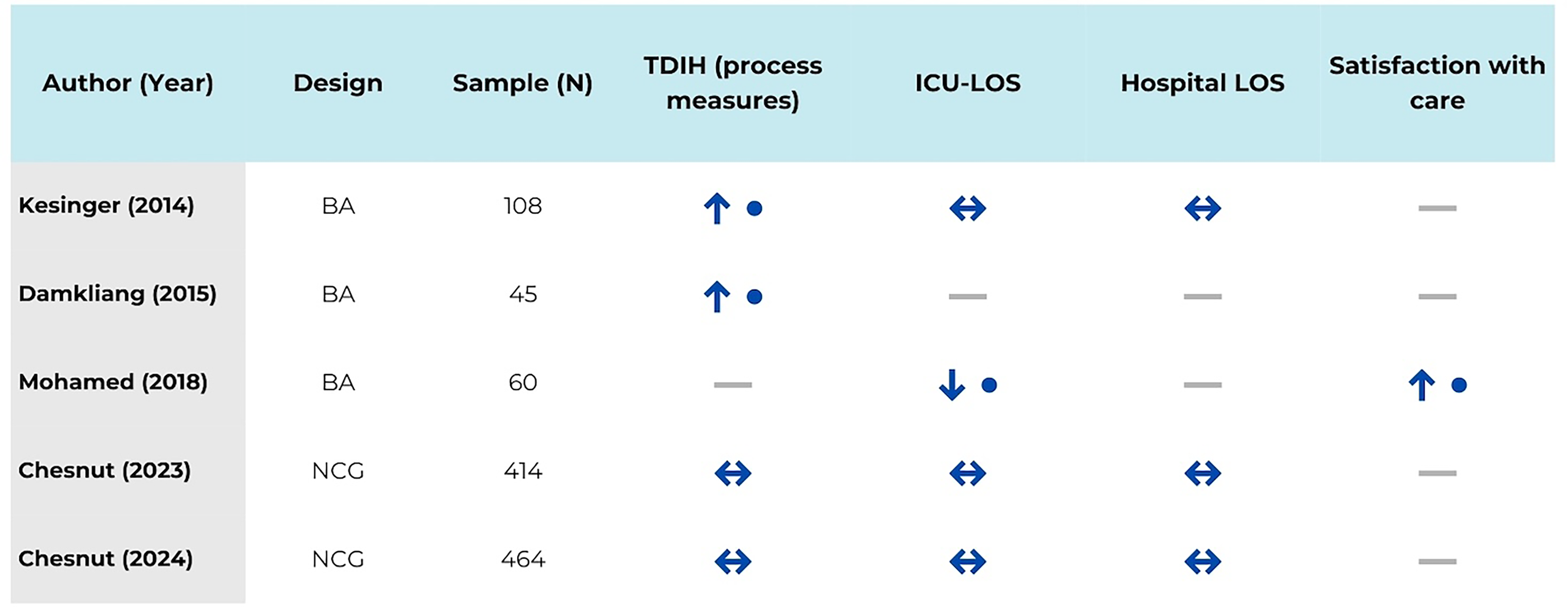
Matrix of secondary outcomes across included studies. Direction of effect is shown for each outcome as favoring protocolized care
Very low-quality evidence suggests that protocolized care may have little or no difference in the proportion of participants with sTBI who are maintained in neutral alignment [85% pre- versus 100% post-intervention; p = .08 29 ] or given prophylactic antibiotics [16% pre- versus 28% post-intervention; p = .16 30 ]; sedation days [3.3 days pre- versus 1.3 days post-intervention; p = .41, 26 and 3.3 days pre- versus 1.7 days post-intervention; p = .06 27 ]; or the use of hyperventilation maneuver [point estimates not reported in the first study, p = .27 26 ; and 3.0 days pre- versus 1.9 days post-intervention; p = .28 27 ]. The included studies did not provide additional information regarding other interventions for the management of intracranial hypertension (Figure 4).
In-Hospital Length of Stay (LOS)
There was very low-quality evidence (downgraded for serious risk of bias) for the LOS outcome, reported in three NRS with 986 participants.26,27,30 Protocolized care has little or no effect on LOS in patients with sTBI [28.3 days pre- versus 27.1 days post-intervention; p = .98, 26 28.6 days pre- versus 30.2 days post-intervention; p = .82, 30 and 27.2 days pre- versus 25.2 days post-intervention; p = .25 27 ] (Figure 4).
Intensive Care Unit Length of Stay (ICU-LOS)
Very low-quality evidence (downgraded for serious risk of bias, and inconsistency) for the outcome of ICU stay was reported in four NRSs with 1046 participants.26,27,30,31 The effect of standardized care on ICU stay in patients with sTBI is uncertain. Three studies documented that standardized care has little or no effect on ICU-LOS [14.8 days pre- versus 15.9 days post-intervention; p = .57 26 ; 15.9 days pre- versus 16.2 days post-intervention; p = .33 27 ; and 13.3 days pre- versus 15.5 days post-intervention; p = .38 30 ]. However, a fourth study reported that patients receiving standardized care might have a shorter ICU length of stay compared to the control group [16.5 days pre- versus 14.8 days post-intervention; p = .00 31 ] (Figure 4).
Satisfaction with the Care Process
There was very low-quality evidence (downgraded for serious risk of bias and imprecision) for this outcome, reported in one NRS with 60 participants. 31 Protocolized care could increase the overall level of patient/family satisfaction, at the expense of higher scores in domains such as respect for dignity (12.8 ± 1.0 points pre- vs 18.3 ± 0.9 points post-intervention; p = .00), caring licensed practical nurses (13.3 ± 1.6 points pre- vs 17.0 ± 1.5 points post-intervention; p = .00), caring nurses (5.9 ± 0.7 points pre- vs 8.5 ± 0.7 points post-intervention; p = .00), caring physicians (6.9 ± 1.0 points pre- vs 8.4 ± 0.7 points post-intervention; p = .00), and hospitalization outcome (12.9 ± 1.0 points pre- vs 19.1 ± 0.9 points post-intervention; p = .00). Results were evaluated using the patient/family satisfaction scale, which assesses perceptions regarding the quality of healthcare service, designed by Mohamed et al 2018 31 (Figure 4).
Cost Related to the Care Process
None of the included studies assessed this outcome.
Exploring Sources of Heterogeneity and Sensitivity Analysis
Given the limitations of the evidence retrieved under this review, a formal exploration of sources of heterogeneity was not feasible. Similarly, since none of the retrieved studies were classified as low risk of bias, the proposed sensitivity analysis was not viable. The certainty of the evidence, along with the corresponding “Summary of Findings Table” is provided in the Supplemental material (Supplemental material, Table D).
Discussion
TBI is a major global health challenge and a leading cause of death and permanent disability worldwide. 40 The burden of TBI is disproportionately higher in LMICs, highlighting significant health inequities.6,40 In these regions, the risk of death from TBI is nearly twice that of high-income countries—with mortality rates as high as 71% in Sri Lanka, 52% in Tanzania, and 41% in Egypt. 6 Geographic, political, and financial constraints often limit timely access to high-quality neurosurgical and neurocritical care. 41 Effective management during the acute phase of TBI is critical to improving survival and long-term outcomes. In this context, evidence-based decision-making is essential to delivering consistent, high-quality care and narrowing the health equity gap. 17 Furthermore, protocolized care supports a coordinated, multidisciplinary approach to managing critically ill patients safely and efficiently in life-threatening situations. 18
Although the certainty of the evidence remains very low to low, this systematic review suggests that protocolized care may reduce mortality, improve cognitive outcomes, reduce complication rates, promote timely interventions, and enhance satisfaction with the care process among adults with moderate-to-severe TBI in LMICs. These findings are consistent with those of a previous systematic review. In 2013, English et al. 42 reported that protocol-based care, compared to usual care, significantly reduced mortality (OR 0.33; 95% CI 0.13-0.82) and increased the proportion of patients with favorable neurological outcomes (OR 3.84; 95% CI 2.47-5.96, assessed by GOS > 4) at six months of follow-up in high-income country settings. Notably, there was little or no difference in ICU or in-hospital length of stay among patients with severe TBI. 42 While differences in clinical context and resource availability exist between LMICs and high-income countries, the similarity in outcomes may be attributed to the focus on a comparable patient population (ie, individuals with moderate to severe TBI in the acute phase) and shared core components of the intervention across settings.
When defining minimum quality standards for the care of patients with TBI, both the studies included in this review as well as those analyzed by English et al 42 rely on care processes grounded in the best available evidence. This evidence is typically identified through systematic literature searches or through the adoption of management guidelines developed by expert scientific bodies such as the Brain Trauma Foundation (BTF) 43 and the American College of Surgeons (ATLS). 44 These organizations use formal consensus processes to define structured care pathways—such as Tier 1, Tier 2, and Tier 3 interventions—for managing TBI. 45 This shared foundation results in consistent, evidence-based, and applicable recommendations, which likely contributes to the similarities observed in outcomes across different healthcare settings. 9
One of the most notable differences between the two reviews is the use of invasive intracranial pressure (ICP) monitoring as a therapeutic guide. This intervention was reported in nearly all studies conducted in high-income countries 42 but appeared in only one study included in this review—used in fewer than 10% of patients. 32 While it is beyond the scope of this review to assess whether these findings support the conclusions of the BEST:TRIP trial 46 —which found that ICP-targeted care (maintaining ICP at or below 20 mm Hg) was not superior to management based on imaging and clinical examination—it is difficult to ignore the controversy surrounding the role of invasive monitoring in improving mortality and functional outcomes in TBI patients. It is important to recognize that, in the context of complex care pathways, analyzing individual components in isolation can obscure their true value. 17 The utility of ICP monitoring, for example, may appear limited if it is not integrated with other essential aspects of care (eg, Tier 1, 2, or 3 interventions). 45 Advanced monitoring offers little benefit if data are not interpreted within a comprehensive understanding of cerebral perfusion, metabolism, and oxygen dynamics—or if the clinician lacks the expertise to contextualize the findings within the broader clinical picture. Therefore, whether invasive monitoring is used or not, protocolized care clearly adds value, especially in resource-limited settings, by establishing minimum standards for high-quality treatment. These benefits may be further enhanced if greater resources become available to support comprehensive TBI management (eg multimodal monitoring).
Regarding other outcomes, this review agrees with the aspects pointed out by English et al regarding the impact of the protocolized care on overall hospital stay and ICU stay, as the analysis of the retrieved evidence shows little to no effect from the intervention. Another relevant finding is the fact that standardized management may reduce the incidence of hospitalization-related complications, while perhaps increasing satisfaction with the care received. These outcomes, unfortunately, could not be analyzed as part of the review by English et al, given inconsistent or absent reporting in the literature. Finally, the observed results also coincide with the opinions of some experts 47 and the results of other research 41 : the implementation of algorithms based on neurological examination and imaging increases the use of brain-specific treatments. This aspect is probably explained by the fact that protocolized care reduces unjustified variability,18,48 increasing the adherence to effective interventions for the management of patients with intracraneal hypertension (eg, administration of mannitol or hypertonic saline solution). 41
This study has limitations, stemming primarily from its reliance on quasi-experimental before-and-after designs. While these designs offer some insight into an intervention's effect size, they lack the robustness of randomized controlled trials (RCTs). Quasi-experimental studies are prone to spurious associations because researchers cannot control all prognostic variables influencing the outcome. Without a concurrent control group, observed changes might also be attributed to environmental, social, political, educational, technological, or cultural shifts, making it difficult to conclusively prove the impact of the intervention per se. Further limitations relate to the precision and applicability of the evidence. The results suffer from imprecision, often due to the low frequency of some events and suboptimal sample sizes across most studies, potentially leading to a lack of power to detect the intervention's effect on certain outcomes. Regarding applicability, studies frequently failed to report adherence rates to care protocols, or the methodologies used for continuous quality improvement. These omissions hinder the replicability of findings.
This review exhibits several methodological strengths. 49 Foremost, it was predicated on a precisely formulated research question and the application of rigorous inclusion and exclusion criteria, with a specific focus on patient-critical outcomes.17,50 The review protocol was prospectively registered, detailing all relevant aspects of its design and conduct. A comprehensive and unrestricted literature search was undertaken, encompassing diverse sources such as gray literature and ongoing studies, without language or publication date limitations. Transparency was maintained through the provision of a detailed roster of both included and excluded studies, with explicit documentation of exclusion rationales. The risk of bias for individual studies was assessed using a validated instrument. Given the impracticality of data pooling, a transparent and robust qualitative synthesis method, adhering to The Synthesis Without Meta-analysis (SWiM) guideline, was employed to report the findings. Additionally, the overall quality of the body of evidence was systematically evaluated (GRADE approach), 50 ensuring that inherent limitations were duly considered during result interpretation. Finally, funding sources for all included studies were meticulously investigated, and the review authors proactively declared all potential conflicts of interest.
Despite its limitations, this review has important implications for both clinical practice and future research. Although the evidence is of very low-to-low quality, it suggests that protocolized care may reduce mortality and enhance cognitive outcomes in adults with moderate to severe TBI in LMICs. This potential benefit appears to be consistent despite variations in geographic, political, and financial contexts. However, the role of routine monitoring of intracranial pressure or other critical physiological parameters—whether through invasive or noninvasive methods (such as transcranial Doppler ultrasound, optic nerve sheath diameter measurement, or quantitative pupillometry)—remains an unresolved issue. Future studies should adopt more rigorous methodological approaches to address current evidence limitations, including biases related to selection, detection, performance, attrition, and reporting. Emphasis should be placed on critical patient-centered outcomes and decision-making metrics. Additionally, it is essential to compare the safety and effectiveness of various management strategies for TBI in resource-constrained settings, including guidelines such as the Brain Trauma Foundation (BTF), 43 the Consensus Revised Imaging and Clinical Examination (CREVICE) protocol, 51 and the 2020 recommendations for the general approach to severe TBI management.52,53
Conclusion
Very low-quality evidence suggests that protocolized care may offer benefits for adults with moderate-to-severe TBI in LMICs, including reduced mortality, improved cognitive status, decreased hospitalization-related complications, and greater satisfaction with the care process. However, these findings must be interpreted with caution due to limited data and the inherent uncertainties surrounding evidence quality. Higher-quality research is imperative to rigorously assess the safety and effectiveness of this intervention, particularly concerning key patient and decision-maker outcomes like functionality, quality of life, and resource utilization.
Supplemental Material
sj-docx-1-jic-10.1177_08850666261424878 - Supplemental material for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review
Supplemental material, sj-docx-1-jic-10.1177_08850666261424878 for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review by Carlos Fernando Grillo-Ardila, Malena Grillo-Ardila, Javier Andrés Mora-Arteaga, Iván Riaño and Miguel Gómez-Hernandez in Journal of Intensive Care Medicine
Supplemental Material
sj-docx-2-jic-10.1177_08850666261424878 - Supplemental material for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review
Supplemental material, sj-docx-2-jic-10.1177_08850666261424878 for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review by Carlos Fernando Grillo-Ardila, Malena Grillo-Ardila, Javier Andrés Mora-Arteaga, Iván Riaño and Miguel Gómez-Hernandez in Journal of Intensive Care Medicine
Supplemental Material
sj-docx-3-jic-10.1177_08850666261424878 - Supplemental material for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review
Supplemental material, sj-docx-3-jic-10.1177_08850666261424878 for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review by Carlos Fernando Grillo-Ardila, Malena Grillo-Ardila, Javier Andrés Mora-Arteaga, Iván Riaño and Miguel Gómez-Hernandez in Journal of Intensive Care Medicine
Supplemental Material
sj-docx-4-jic-10.1177_08850666261424878 - Supplemental material for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review
Supplemental material, sj-docx-4-jic-10.1177_08850666261424878 for Safety and Effectiveness of Standardized Care for Adult Population with Moderate or Severe Traumatic Brain Injury in Lower- and Middle-Income Countries: A Systematic Review by Carlos Fernando Grillo-Ardila, Malena Grillo-Ardila, Javier Andrés Mora-Arteaga, Iván Riaño and Miguel Gómez-Hernandez in Journal of Intensive Care Medicine
Footnotes
Acknowledgments
Heartfelt gratitude to PG-M for their unwavering support and motivation. To ASM-K and Heart-Brain foundation for their encouragement to keep pushing forward.
Authors’ Contributions
CFG-A contributed to study concept, design, acquisition, statistical analysis, data interpretation, and drafting of the manuscript. MG-A and JAM-A contributed to study acquisition, data interpretation, statistical analysis, and study supervision. IR-F and MG-H contributed to study acquisition, data interpretation, and drafting of the manuscript. All of the authors read and approved the final manuscript and agreed to be accountable for all aspects of the work. All of the authors made sure that the questions related to the accuracy or integrity of any part of the work were appropriately investigated and resolved. All of the authors read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Open access funding provided by Universidad Nacional de Colombia, Bogotá Campus.
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary file.
Supplemental Material
Supplemental material for this article is available online.
