Abstract
Patients in the intensive care unit often develop anion gap metabolic acidosis most commonly from lactic acidosis. Clinicians routinely measure serum lactate levels, but it is not clear if one is able to use the measured serum lactate level to determine if there is a second cause for anion gap metabolic acidosis in these critically ill patients. In this report, 503 episodes of lactic acidosis with serum lactate levels greater than 5 mmol/L at a single institution over two years were analyzed. The average serum anion gap minus serum lactate level in these patients was 6.9 ± 0.21 mEq/L, the average corrected anion gap was −1.3 ± 0.23 mEq/L and the average strong ion gap was 5.1 ± 0.3 mEq/L. The majority of the episodes with anion gap—serum lactate concentration >8.0 mEq/L were explained by elevated serum albumin concentration, elevated serum phosphorus concentration, presence of ketones in the urine or high β-hydroxybutyrate concentrations. The above data suggest that one can use the measured serum lactate concentration and subtract from the serum anion gap to determine if there is a second reason for anion gap metabolic acidosis.
Introduction
The question of whether one can use the serum lactate concentration and the serum anion gap to determine if there is a second acid-base disorder in addition to lactic acidosis has not been investigated directly to the best of our knowledge. Several studies have shown that the rise in serum anion gap is more than the rise in serum lactate levels, but this has been more in the setting of studies trying to determine if one can use the serum anion gap as a surrogate to measuring serum lactate levels.1–7 It has also been found that lactate levels up to 5 mmol/L may still be associated with normal anion gaps.1,7,8
As previous studies have shown, one should not just rely on the serum anion gap for establishing the diagnosis of lactic acidosis in cases of metabolic acidosis but actually measure serum lactate levels.1–7 The ratio between the change from normal in serum anion gap over the change from normal in serum bicarbonate is used to study whether there are other acid-base disorders in cases of lactic acidosis. 9 In this report, we studied whether the serum lactate level will allow one to determine if there is a second reason for high anion gap metabolic acidosis in addition to lactic acidosis.
Methods
We conducted a retrospective cross-sectional study using electronic health record data from a single urban academic medical center between January 2016 and December 2017. We identified all patient admissions having at least one serum lactate level greater than 5 mmol/L. For each admission, we identified the highest lactate value and extracted available lab values collected within ±24 h of this measurement, including serum sodium, potassium, magnesium, chloride, phosphorus, total CO2 (TCO2), calcium, albumin, total protein, β-hydroxybutyrate, and urinary ketones.
First, we calculated the serum anion gap—serum lactate difference (AG-L) to determine if the serum lactate level accounted for the change in serum anion gap from normal. Patients with an average AG-L difference of ≥8 mEq/L were analyzed separately to determine if there was evidence of other measured anions (albumin, phosphate, and ketones) that could contribute to the higher-than-expected difference.
To examine whether incorporating other measured anions beyond lactate could quantitatively explain the rise in anion gap, we examined the corrected anion gap (cAG) and the strong ion gap (SIG) among episodes having sufficient data for these calculations. The cAG was defined in mEq/L by the formula: corrected anion gap = [Na+] + [K+] − {[Cl−] + [TCO2] + [lactate] + [phosphate] + [total protein (mmol/L)]}, all in mEq/L except total protein (Formula 1). Phosphate concentration in mEq/L was calculated using formula phosphate = C × (10× [P in mg/dL]/31) where C = 1 + (10pH−6.8/10pH−6.8 + 1). The net charge contribution of total protein was calculated using Van Leeuwen's formula total protein (mmol/L) = 10.3 × (pH-5.66) × 0.1 × (total protein in g/dL). 10 Finally, the SIG was used to calculate charge balance: [Na+] + [K+] + [Ca++] + {Mg++] − [Cl−] [lactate−] 2.46 × 10pH−8 × PCO2 − albumin × (0.123 × pH − 0.631) – phosphate x (0.309 × pH – 0.469) all values being in mEq/L, except for albumin (g/dL), phosphate (mmol/L) and pCO2 in mm Hg. 11
The study was approved by the University of California, San Francisco Institutional Review Board (#18-24882). Data were analyzed using Microsoft Excel statistical-use software (Microsoft Corporation, Redmond, Washington, USA) and expressed as average ± standard deviation/standard error of the mean. We performed multivariate regression analysis to identify significant contributors to the anion gap beyond lactate. The dataset included 461 unique patients selected from 503 episodes by choosing the most recent lactate >5 mmol/L measurement per patient to ensure statistical independence. First, simple linear regression established the baseline relationship between lactate and anion gap (n = 461). Subsequently, we analyzed residual anion gap (observed minus lactate-predicted) using three nested multivariate models based on data availability: albumin and phosphorus (<10% missing, n = 417), then adding creatinine, pH, and pCO2 (<30% missing, n = 322), and then incorporating ketones and β-hydroxybutyrate (>65% missing, n = 104). Additionally, we evaluated the Strong Ion Gap contribution by comparing models with and without SIG (n = 345). All analyses used robust standard errors (HC3) to account for heteroscedasticity. Statistical significance was set at p < .05. For relationship between lactate and anion gap simple linear regression was performed with anion gap as the dependent variable and serum lactate as the independent variable (n = 461). Model assumptions were evaluated using residual plots, the Shapiro-Wilk test for normality of residuals, the Breusch-Pagan test for heteroscedasticity (α = .05), and the Durbin-Watson statistic for independence. Influential observations were identified using Cook's distance (threshold >4/n). Robust standard errors (HC3) were calculated to account for any residual heteroscedasticity.
Results
In total, 503 episodes of lactic acidosis were included in the analysis. Average age was 57 ± 10.23 (mean ± SD) years; 33.3% of the patients were female. Of the 503 patients, 10 were younger than 18 years of age, including 7 newborns, one 2-year-old, one 10-year-old, and one 17-year-old. A small number of patients (<10) contributed multiple episodes due to having multiple admissions with lactic acidosis. Out of the 503 lactic acidosis episodes, the average lactate level was 9.8 ± 0.4 mmol/L (average ± standard error of the mean). The median was 7.9 mmol/L). The average anion gap in these patient episodes was 17.0 ± 0.8 mEq/L and the AG-L was 6.9 ± 0.2 mEq/L (Table 1). All the newborns except two had an AG-L of less than 0. None of them had enough data to calculate SIG or the corrected anion gap.
Mean Laboratory Values and Standard Deviations of the Cohort of Patients.
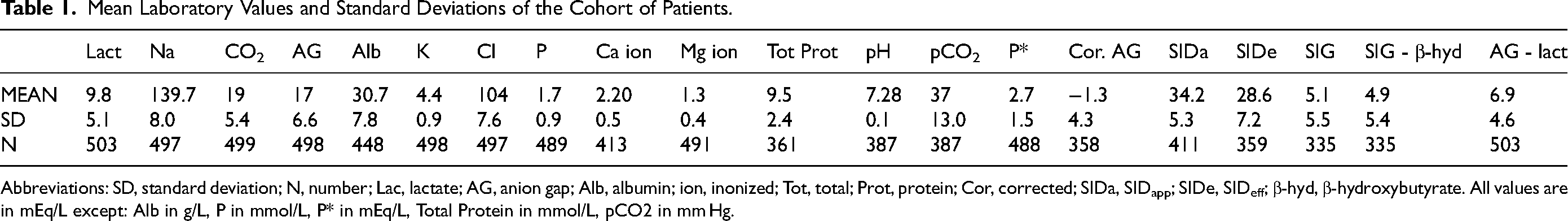
Abbreviations: SD, standard deviation; N, number; Lac, lactate; AG, anion gap; Alb, albumin; ion, inonized; Tot, total; Prot, protein; Cor, corrected; SIDa, SIDapp; SIDe, SIDeff; β-hyd, β-hydroxybutyrate. All values are in mEq/L except: Alb in g/L, P in mmol/L, P* in mEq/L, Total Protein in mmol/L, pCO2 in mm Hg.
Out of the 503 lactic acidosis episodes, 170 (34%) had a serum AG-L level of ≥8.0, 102 (20%) had a serum anion gap minus serum lactate level of ≥10 mEq/L, and 5 (1%) had a serum anion gap minus serum lactate level of ≥20 mEq/L. Out of the 170 episodes with the difference ≥8.0 mEq/L, 140 episodes (82%) could be at least partially explained by high serum albumin concentration (4.1-5.0 g/dL), high serum phosphorus concentration (6.0-14.5 mg/dL), presence of ketones in the urine, high serum β-hydroxybutyrate concentration (0.18-15.59 mmol/L) or a combination of the above (Table 2). In the 30 (18%) remaining episodes, the AG-L level remained unexplained; however, most of these patients did not have β-hydroxybutyrate or ketone measurements (Table 3). Twelve of these patients had negative urine ketone measurements (all others did not have a measurement), and none of the patients had a serum β-hydroxybutyrate level measured
Explanation for the 171 Patients with Explained Anion Gap Minus Lactate Level >8.0 mEq/L.

Phosphorus in mg/dL, Albumin in g/dL, β-hydroxy = β-hydroxybutyrate in mEq/L, AG = Anion gap, AG – lactate is in mEq/L, *urine ketones were measured as 1+, 2+ or 3+.
Available Lab Values for the 31 Patients with Anion Gap Minus Serum Lactate Level >8 Whose Value Remains Unexplained.
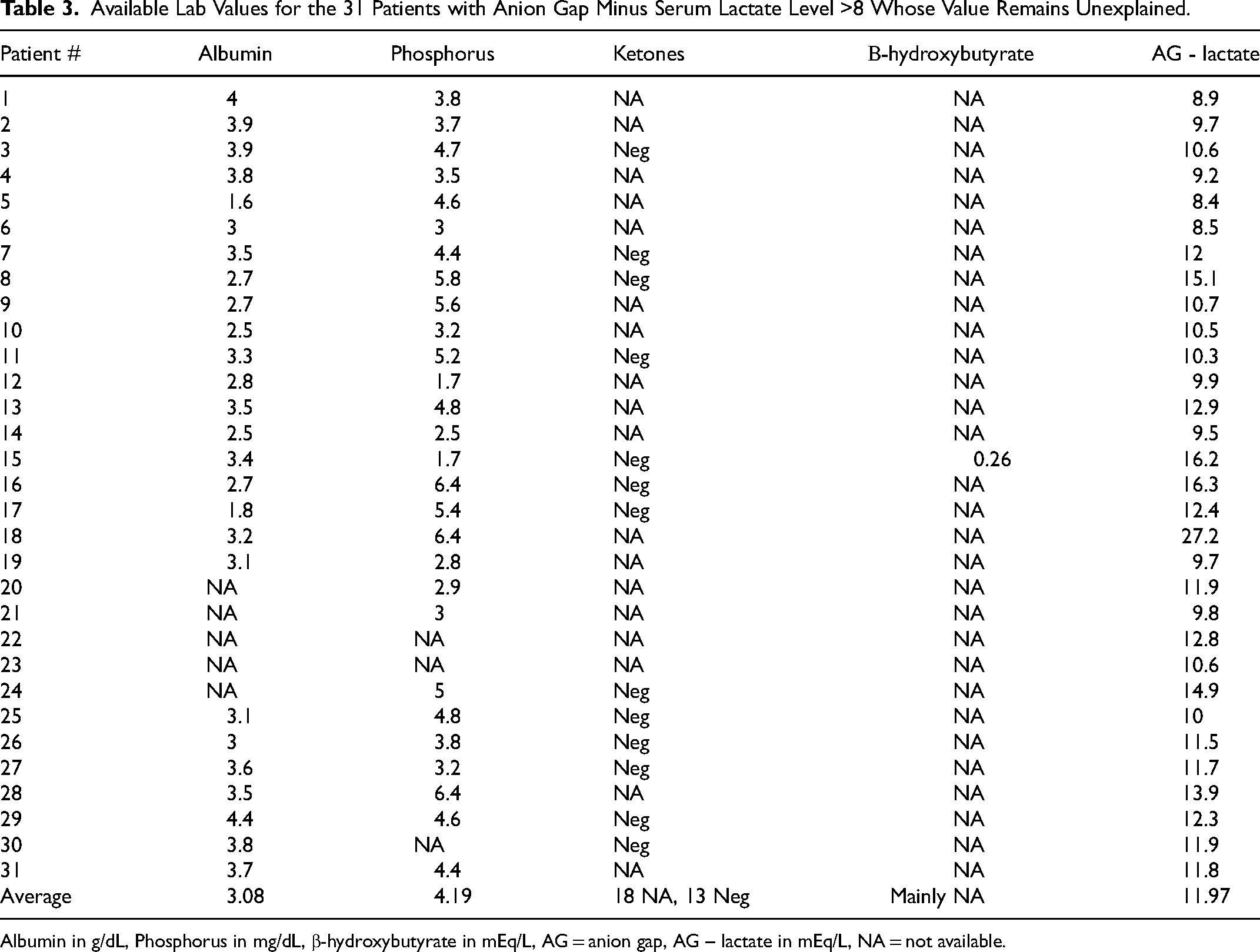
Albumin in g/dL, Phosphorus in mg/dL, β-hydroxybutyrate in mEq/L, AG = anion gap, AG – lactate in mEq/L, NA = not available.
Out of the 140 episodes with at least partially explained AG-L difference, high serum albumin concentration was present in 5 (3.6%), high serum phosphorus concentration in 75 (54%), positive urine ketones in 8 (5.7%), elevated β-hydroxybutyrate in 5 (3.6%) and a combination of the above in 39 (27.9%).
Among the 358 episodes with available data for calculation, the average cAG was −1.3 ± 0.23 mEq/L (median cAG was −1.2 mEq/L). This value indicates that the available anion and cation measurements were balanced and there was no indication that there was an extra anion or cation that contributed to an anion or cation gap.
In 334 patients having available data to calculate the strong ion gap, the average SIG was 5.1 ± 0.3 mEq/L (median 5.2 mEq/L). Strong ion gap is made up of two parts
Average Corrected Anion Gap and Anion Gap Minus Serum Lactate Concentration According to the Serum SIG Level.

Abbreviations: SIG, strong ion gap; AVG, average; AG, anion gap; SIG is in mEq/L, AG and AG – lactate are in mEq/L.
Among 31 episodes with available serum β-hydroxybutyrate, additional correction of the SIG for β-hydroxybutyrate did not significantly change the mean SIG level (4.9 ± 5.4 mEq/L vs 5.1 ± 5.5 mEq/L).
A total of 48 patients out of the 334 patient episodes had urinary ketone measurements. Urine ketone levels were negative in the 2 patients who had SIG < 2 mEq/L, 3 patients had urinary ketones determination in the SIG 2–5 mEq/L range, 2 were negative, and one had 1 + ketones. Forty-three had ketone measurements in the SIG > 5 mEq/L group, 24 negative, 3 trace, 11 with 1+, 2 with 2+, and 3 with 3+ (Table 5).
Urinary Ketone Levels Based on the SIG Group Classification.
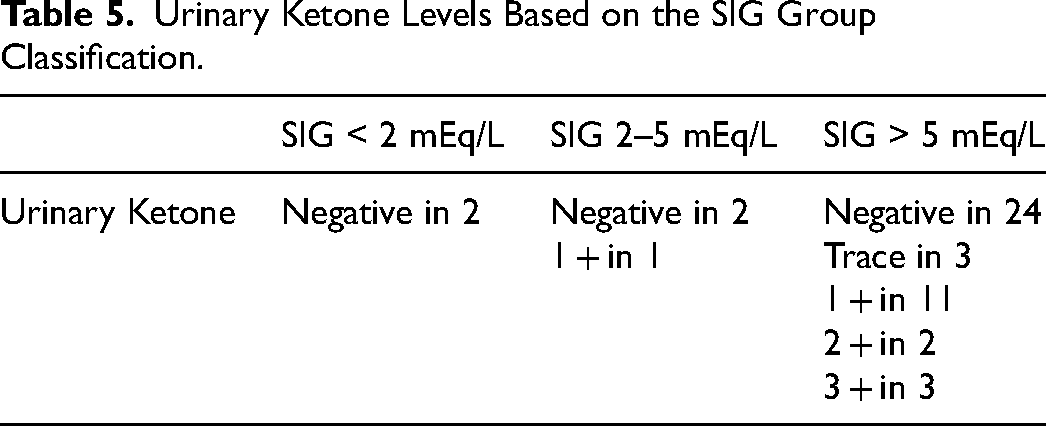
In order to determine if the discrepancy between SIG and cAG is explained by renal failure, the serum creatinine, eGFR and phosphorus levels were graphed as a function of SIG – cAG (Figure 1). There did not seem to be any relationship between any of the three variables and the SIG – cAG, suggesting that the difference is present not only in patients with renal failure but also in patients who did not have renal failure.
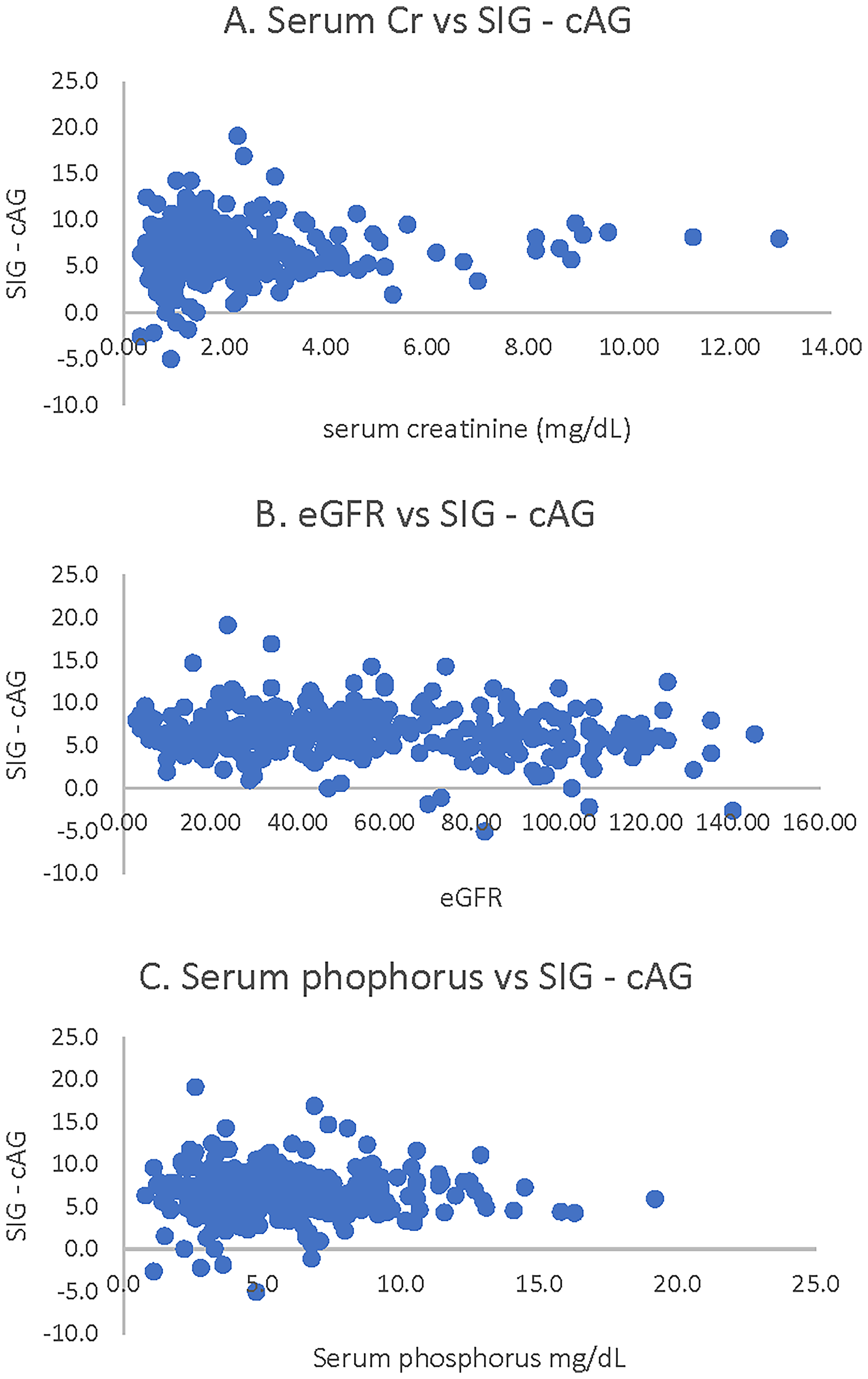
Relationship between serum creatinine (A), eGFR (B) and serum phosphorus (C) and the difference between SIG and cAG.
Multivariate Regression Analysis
Lactate alone explained 60.4% of anion gap variance (R2 = 0.604, n = 461). Analysis of residual anion gap identified additional significant contributors. Albumin (β = 1.37, 95% CI: 0.79-1.95, p < .001), phosphorus (β = 0.26, 95% CI: 0.02-0.50, p = .04), creatinine (β = 0.56, 95% CI: 0.33-0.79, p < .001), and pCO2 (β = −0.11, 95% CI: −0.14 to −0.07, p < .001) collectively explained 26.1% of residual variance (n = 322). In a separate analysis (n = 345), adding Strong Ion Gap to the lactate model improved R2 from 0.643 to 0.763 (ΔR2 = 0.120, p = .007).
Lactate explains most (60%) but not all of the anion gap elevation (Figure 2). The relationship is nearly 1:1—each 1 mmol/L increase in lactate raises AG by about 1 mEq/L. This strong relationship supports using lactate to “correct” the anion gap, but 40% of AG variance remains unexplained.
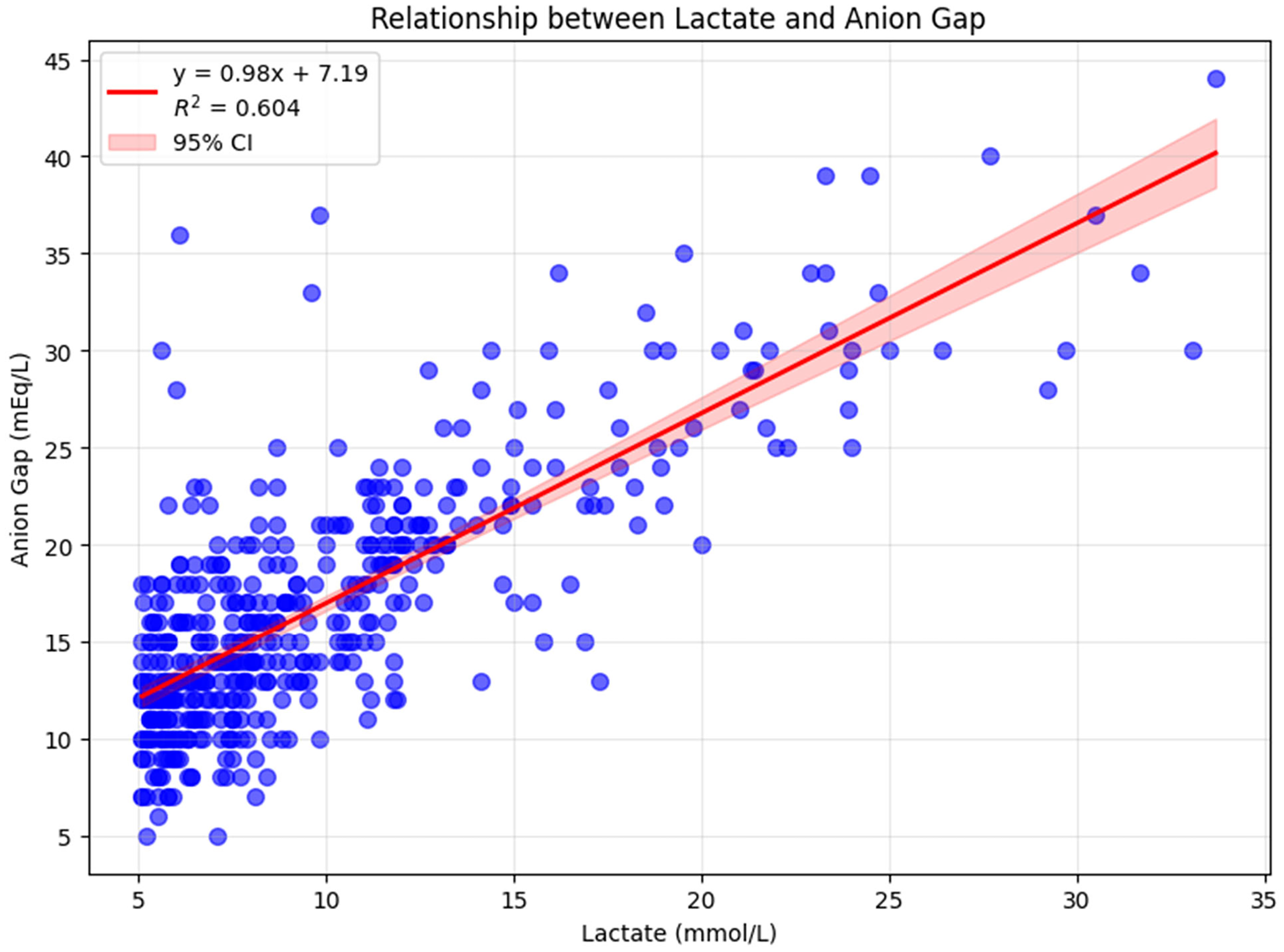
Relationship between serum lactate (mmole/L) and serum anion gap (mEq/L).
The relationship between lactate and anion gap was strong and linear (R2 = 0.604, p < .001). Each 1 mmol/L increase in lactate corresponded to a 0.98 mEq/L increase in anion gap (95% CI: 0.89-1.06), demonstrating a nearly 1:1 relationship. The model showed good calibration with (((mean residuals of 0.00 mEq/L (SD = 4.12), though individual residuals ranged from −11.12 to 22.84 mEq/L, indicating substantial patient-level variation.
Multiple regression analysis of residual anion gap (observed minus lactate-predicted) identified four significant predictors (n = 322) (Table 6). Albumin demonstrated the strongest association (β = 1.37 per g/dL, 95% CI: 0.79-1.95, p < .001), followed by creatinine (β = 0.56 per mg/dL, 95% CI: 0.33-0.79, p < .001), phosphorus (β = 0.26 per mg/dL, 95% CI: 0.02-0.50, p = .04), and pCO2 (β = −0.11 per mm Hg, 95% CI: −0.14 to −0.07, p < .001). These variables collectively explained 26.1% of the variance in residual anion gap (adjusted R2 = 0.250, F = 17.46, p < .001). In the complete model (n = 104), ketones (β = 0.62, p = .22) and β-hydroxybutyrate (β = 0.36, p = .23) did not achieve statistical significance despite theoretical relevance probably due to low number of values available.
Factors Predicting Residual Anion Gap After Accounting for Lactate.
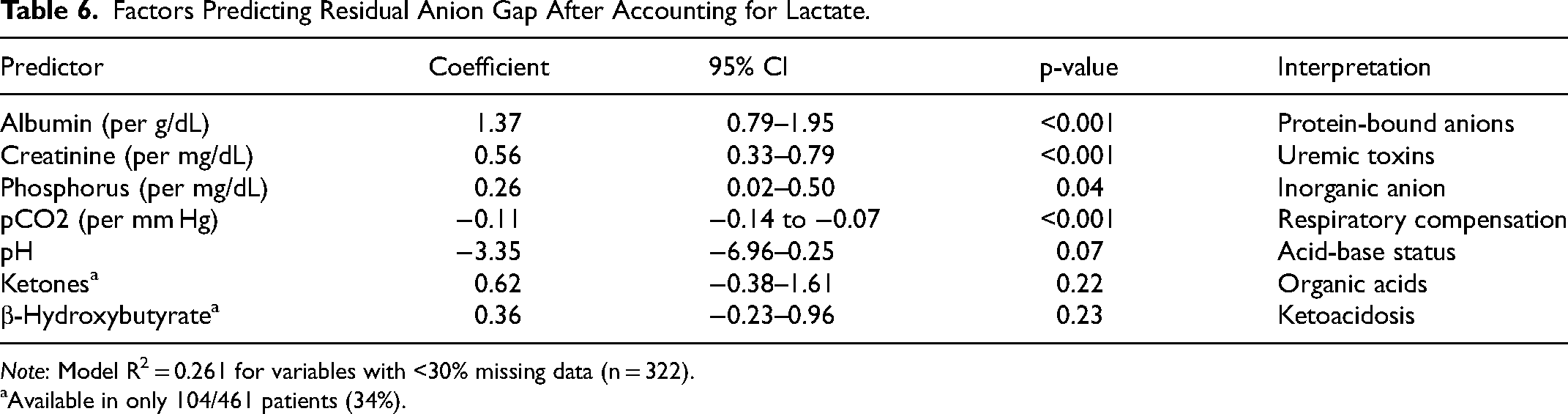
Note: Model R2 = 0.261 for variables with <30% missing data (n = 322).
Available in only 104/461 patients (34%).
To further determine the specific cause of the discrepancy between SIG and cAG, we measured the individual constituents of the calculations (Table 7). About half of the discrepancy is explained by the absence of serum Magnesium and Calcium in the cAG calculation but their presence in the SIG calculation. The other half was explained by the contribution of serum total CO2 and bicarbonate and the protein component (in cAG calculation total protein is used while with SIG only serum albumin is utilized).
The Average Contribution of Certain Cations and Anions to the Value of cAG and SIG.
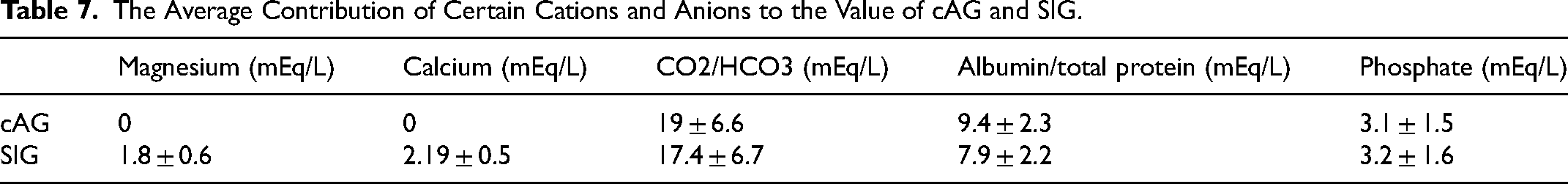
Discussion
There are two important findings of this study. First, the serum lactate concentration can be used to determine if there is a second reason for high anion gap metabolic acidosis in addition to lactic acidosis. The average serum AG-L concentration of 6.9 ± 4.6 mEq/L was well within the normal reference range for serum anion gap with 66% of episodes having anion gap <8.0 mEq/L, suggesting that rises in serum anion gap could be fully explained by lactate in most cases. Only 34% of those patients had a difference >8.0 mEq/L, and 20% had a difference >10 mEq/L to suggest the presence of additional uncounted anions contributing to the anion gap acidosis or the presence of other conditions increasing the anion gap. In 82% of these cases, the difference could be at least partially explained by either a high serum albumin concentration, a high serum phosphorus concentration, the presence of ketones in the urine, a high serum β-hydroxybutyrate concentration, or a combination of the above. Of the remaining 18%, there was not enough data available on one or more of the above factors to determine if there was an explanation for the persistence of the gap. In addition to the conditions mentioned above, the causes of a high serum anion gap include laboratory error. 12 Falsely low values for both bicarbonate in blood gases 13 and TCO2 in serum, 14 and therefore falsely elevated anion gap values, have been described by endogenous substances interfering with the assays.
The second finding of the study was that the serum AG-L correlated well with the corrected anion gap formula (formula 1 above), but the SIG was significantly higher within all groups. The discrepancy is more than 50% explained by the accounting of the charge of magnesium and calcium with the SIG calculation and not with the cAG calculation. The other 50% is explained by different formulas used to calculate the charge contribution of bicarbonate and serum proteins by the two methods.
The rise in anion gap mirrors the rise in serum lactate in an approximately one-to-one manner when mild lactic acidosis is induced by extreme exercise in healthy individuals. 15 In a study by Mecher et al, 30 patients with lactic acidosis from sepsis were analyzed, and the corrected anion gap in these patients ranged from −5.4 to 11.3 mmol/L. 10 Magnesium and calcium were not taken into account in this study and 4 patients also had ketones in their urine. In 80% of the patients, there was an increase in anion gap beyond that explained by lactate, phosphate, total serum protein, and urate level, leading the authors to conclude that there is an unmeasured anion in cases of septic shock. In addition, the regression line for estimating the corrected anion gap from the anion gap had a slope of 0.5, indicating that, on average, lactate contributed to half of the increase in the anion gap. However, approximately half the study participants had a serum lactate level below 5 mEq/L, which is in contradiction to the well-documented literature that argues lactate levels below 5 mEq/L may not be associated with an increased anion gap at all in 50% of the patients. 16
Previous studies have shown that amino acids, uric acid, and organic acids together only account for 7.9% of the SIG in intensive care unit patients. 11 Thus, even though the SIG level remains high after accounting for lactate, after searching, an unmeasured anion was not found.
Forni et al looked at patients with elevated anion gap and found that certain low molecular weight anion levels are elevated in some of these patients. 17 They only studied five patients with lactic acidosis and found that isocitrate, α-ketoglutarate, malate, and d-lactate combined accounted for ∼2.2 mEq/L of the increased anion gap.
Another study found that only 44% of patients with a serum lactate level over 2.5 mmol/L had an elevated anion gap. 16 As the authors concluded, this probably reflects that a sick intensive care unit patient frequently has a low serum albumin concentration, and as a result, their baseline serum anion gap may be as low as 5 mEq/L. If they develop lactic acidosis with a serum lactate level of 4 mmol/L, their anion gap would only increase to 9 mEq/L, which could still be considered normal.
Contrary to previous studies, our study found that the serum lactate level did correlate well with the serum anion gap. An explanation for the above finding is that we only included patients with serum lactate levels of >5 mmol/L, whereas previous studies included patients with mildly elevated serum lactate levels, where serum lactate and anion gap values do not correlate as closely.1,10,18 In fact, if one looks at their figures and excludes patients with mildly elevated serum lactate levels in previous studies the correlation between serum lactate and serum anion gap would seem to be much more robust.
Our findings suggest that serum lactate levels combined with serum albumin and phosphorus concentration can be used to determine if there is an additional cause for anion gap metabolic acidosis in patients with a serum lactate level of >5 mmol/L. However, there was a discrepancy between the cAG and serum SIG levels in this study. The SIG level seems a more thorough method as it also considers the contribution of magnesium and calcium to the charge. However, one should be wary as the serum SIG level calculation may not be as accurate in critically ill patients with septic shock as compared with the normal population from which the formula was derived. For example, in the calculation of SIG one uses the ionized serum calcium and magnesium levels. However, the non-ionized serum calcium and magnesium are mainly bound to albumin, which is also a polar interaction. Thus, it may be more accurate to use the total serum calcium and magnesium levels. However, in that instance the SIG level would even be higher. The contribution of CO2/bicarbonate and albumin/total protein each is also ∼1.5 mEq/L different between the two formulas. The CO2/bicarbonate difference is likely explained by the fact that we used total CO2 for cAG formula and bicarbonate for the SIG formula. Whether one should use albumin concentration (as in SIG formula) or total protein concentration (as in cAG formula) is to be determined but it is true that some immunoglobulins are positively charged while others are negatively charged. With renal failure and high serum phosphorus concentrations, the phosphorus itself explains only about half of the increase in anion gap. Since a significant portion of critically ill patients have renal failure and some have high phosphorus levels, the renal failure itself may account for the excess increase in SIG. However, we did not find a correlation between serum creatinine, eGFR or serum phosphorus and the SIG – cAG. It seems more likely that SIG is a more accurate formula indicating the presence of another anion with lactic acidosis. However, despite extensive search this unmeasured anion has not been found for the most part.10,11,17 In either case, it appears that in most cases of severe lactic acidosis, the rise in anion gap is largely explained by the rise in lactate and that the presence of additional cause for anion gap metabolic acidosis can be assessed by subtracting serum lactate levels from the serum anion gap in this subset of critically ill patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
