Abstract
Cardiac surgery-associated acute kidney injury (CSA-AKI) is a common complication following cardiac surgery. Despite growing awareness of sexual dimorphism in cardiovascular and chronic kidney disease, little is known about sexual dimorphism in CSA-AKI. In this narrative review, we will first discuss sex differences and the potential role of sex hormones in the CSA-AKI pathophysiology. Women may tolerate hypoperfusion and hypoxia better, have a different neurohumoral response and experience less inflammation and oxidative stress. Next, we discuss the conflicting evidence on the difference in incidences of CSA-AKI between sexes and suggest that female sex might not be a risk factor. Finally, we focus on sex differences in preventative treatment strategies for CSA-AKI. Analysing sex differences in interventional studies is challenging due to the relative minority of women within the cardiac surgery population. Unravelling sex differences can improve personalized medicine and improve future patients’ outcomes, for both sexes.
Introduction
Cardiac surgery-associated acute kidney injury (CSA-AKI) is a common and serious complication associated with cardiac surgery. CSA-AKI is typically defined as a postoperative increase in serum creatinine concentrations and/or a decrease in urinary output.1,2 The reported incidence varies in the literature, but CSA-AKI is generally estimated to occur in 20-40% of patients undergoing cardiac surgery.1,3 Factors influencing incidence include the type of cardiac surgery, isolated coronary artery bypass graft (CABG) carries a lower risk compared to valve and aortic surgery, the characteristics of the study population, particularly the number of risk factors present in the population, and the definition of AKI, especially whether urine output is taken into account.4,5 CSA-AKI is associated with an increased risk of short-term complications, such as infections, strokes, myocardial infarctions and mortality.6-8 In addition, it has also been linked to a higher long-term risk of developing chronic kidney disease (CKD) and end-stage kidney disease, even when kidney function appears to have fully recovered at hospital discharge.9,10 Structural injury to the kidneys after cardiopulmonary bypass was demonstrated after recovery of functional parameters such as creatinine concentrations.11,12 Given these short-term and long-term sequelae of CSA-AKI, preventing CSA-AKI by pharmacological and other interventions has been a topic of intensive investigation. However, with many of the conducted studies showing no clinical benefit, effective strategies to prevent or reduce CSA-AKI remain limited, highlighting a need for further mechanistic insight into this common complication of cardiac surgery. 13
In recent years, it has become increasingly evident that sexual dimorphism influences both cardiovascular and CKD, with women, particularly premenopausal women, being considered protected. 14 For example, it is known that coronary artery disease (CAD) is more prevalent in men, while the risk in women starts to increase after menopause. 15 Regarding CKD, women experience slower disease progression and lower rates of end-stage kidney disease than men.16,17 Additionally, a prospective, observational study (n = 3534) concluded that women appear to have a lower risk of AKI due to other causes than cardiac surgery compared with men, with an incidence of 11.3% in men versus 7.1% in women (P < .001). 18
Differences between men and women can arise from biological sex differences and gender-related constructs, such as lifestyle choices, as well as social and economic disparities. 19 The biological differences between men and women stem from variations in sex chromosomes, and the impact of sex hormones concentrations, most notably the female sex hormone estrogen and the male sex hormone testosterone. Sex hormones have profound effects beyond their influence on reproductive organs and secondary sexual characteristics; their receptors are distributed throughout the body, including the cardiovascular system and the kidneys. 20 However, sex differences, and the influence of sex hormones in CSA-AKI, specific, remain unclear. It is important to explore whether sex differences in pathophysiology and incidence of CSA-AKI exist as this might indicate whether sex-tailored prevention strategies for CSA-AKI could offer benefit. To address this issue, this review focuses on sex differences, and the influence of sex hormones, in the pathophysiology, incidence and preventive therapies of CSA-AKI. Throughout this review, references to men and women refer to cisgender individuals, meaning their gender identity corresponds with their sex assigned at birth.
Sex Differences in the Underlying Pathophysiology of CSA-AKI
While the etiology of CSA-AKI is complex, multifactorial and incompletely understood, a number of key pathways are involved. These include (regional) kidney hypoperfusion, altered kidney oxygen availability, kidney inflammation, oxidative stress, systemic neurohumoral activation and exposure to nephrotoxic stimuli. The influence of these pathways is modulated by whether cardiopulmonary bypass is used during cardiac surgery. While the use of cardiopulmonary bypass is associated with increased inflammation and hemodilution, off-pump cardiac surgery tends to result in greater hypoperfusion.21,22 Also, patient-specific and surgery-specific risk factors, such as the type and duration of surgery, affect the risk of CSA-AKI (Figure 1).13,23 Below, we will review the evidence for potential sex differences in these pathophysiologic drivers of CSA-AKI (Figure 2).
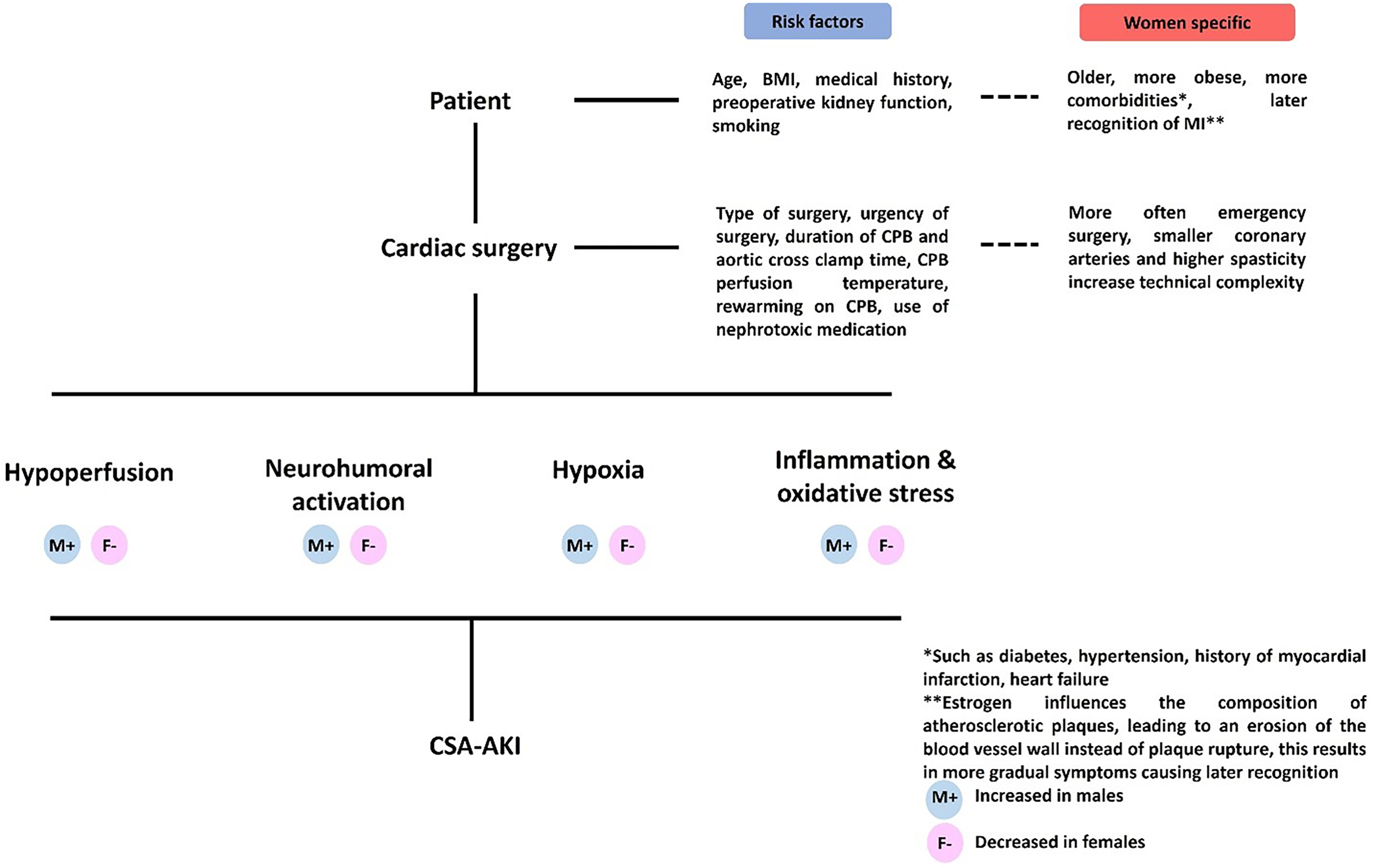
Summary of pathophysiology, risk factors and sex differences in dardiac surgery-associated acute kidney injury. BMI = Body Mass index; CPB = Cardiopulmonary Bypass; CSA-AKI = Cardiac Surgery-Associated Acute Kidney Injury; MI = Myocardial Infarction.
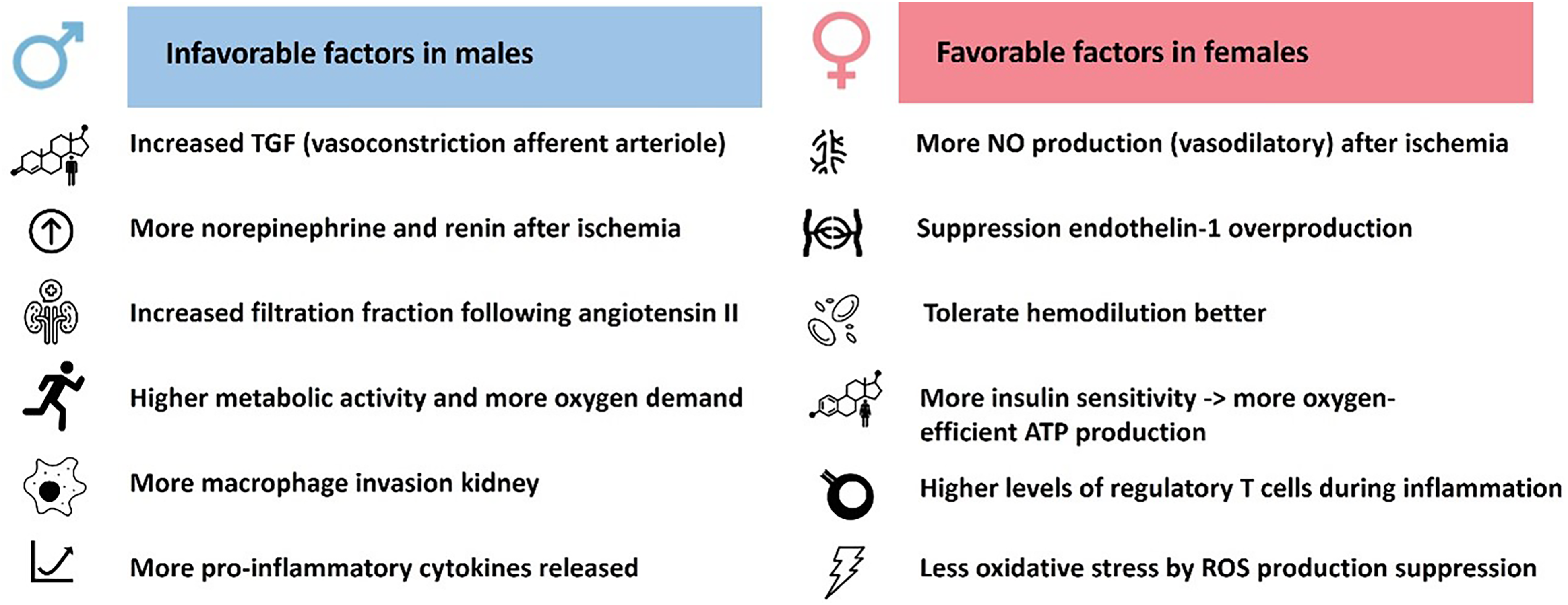
Potential sex differences in the pathogenesis of cardiac surgery-associated acute kidney injury. ATP = Adenosine Triphosphate; NO = Nitric Oxide; ROS = Reactive Oxygen Species; TGF = Tubuloglomerular Feedback.
Kidney Hypoperfusion
Kidney perfusion refers to the oxygen-rich blood supply to the kidneys, which can be compromised during cardiac surgery. 24 The use of cardioplegia and cardiopulmonary bypass or mechanical stress on the heart during off-pump cardiac surgery reduces systemic blood pressure and blood flow. 25 The kidney can regulate the blood flow in the kidney through changes in pre-glomerular vascular tone by the afferent glomerular arterioles, the major resistance vessels in the kidney.26,27 Tubuloglomerular feedback (TGF) is one of the key mechanisms that helps regulate glomerular filtration rate (GFR) during periods of low kidney perfusion, primarily by promoting afferent arteriolar dilatation and activating the renin-angiotensin system (RAAS) to induce efferent arteriolar vasoconstriction. 28 The TGF response relies on cells in the macula densa of the kidney that monitor the concentration of sodium chloride (NaCl) passing through the distal convoluted tubules.29,30 When these cells detect a decrease in NaCl concentrations, the feedback system is inhibited, causing afferent arteriolar vasodilation and efferent arteriolar vasoconstriction, thereby maintaining GFR. 29 During cardiac surgery, kidney perfusion is temporarily reduced, which will inhibit TGF, leading to afferent vasodilation and efferent vasoconstriction. In addition to TGF, vasoactive mediators such as nitric oxide, endothelin-1, and angiotensin II, as well as smooth muscle cells in the afferent and efferent arterioles and the sympathetic nervous system, also help regulate kidney perfusion under dynamic conditions through direct effects on the vascular endothelium. 26 Although above described mechanisms regulate kidney perfusion to maintain GFR, cardiac surgery challenges the kidney's autoregulation. 28 This is illustrated by an observational study showing a 40% decrease in kidney blood flow during hypothermia, despite a pump flow of 1.45-1.65 L/min/m2, which is considered adequate under such conditions.25,31 These findings suggest that the kidney's autoregulation may fail due to hypothermia, low, non-pulsatile blood flow during cardiopulmonary bypass, hemodilution or medication that affect autoregulatory mechanisms. 25
Several studies have suggested that sex hormones might influence kidney perfusion and the response to low perfusion by affecting TGF, and vasoactive mediators. As such, testosterone has been shown to increase TGF via androgen receptors in the macula densa, leading to vasoconstriction in the afferent arterioles. 29 Additionally, in a rodent study, female rats produced more nitric oxide (NO) compared with male counterparts after ischemia, ensuring sufficient perfusion of the kidney through the vasodilatory actions of NO on the afferent arteriole.32,33 A study in healthy men and women observed that menopause and male sex were associated with lower concentrations of NO and two weeks of estrogen replacement restored NO concentrations to premenopausal levels. 34 Another vasoactive agent is endothelin-1, which causes both afferent and efferent arteriolar vasoconstriction via the endothelin A receptor, while also stimulating efferent vasodilation via the endothelin B receptor. 35 Increased concentrations of endothelin-1 have been linked to the onset of AKI. 36 In male rats after ischemic injury, estrogen treatment resulted in a decrease of tissue injury, possibly through suppression of endothelin-1 concentrations. 37 In healthy men and women undergoing gender-affirming hormone therapy, estrogen reduced endothelin concentrations whereas testosterone increased endothelin concentrations. 38 Overall, compared with men, women may be less susceptible to kidney injury after hypoperfusion due to the vasodilative properties of estrogen in the kidney, and the lower concentrations of testosterone, which may have vasoconstrictive effects.
Neurohumoral Activation
Neurohumoral responses regulating kidney perfusion are affected by cardiac surgery. For example, the arterial baroreceptors activate the sympathetic nervous system when they detect low pressure. 39 Norepinephrine, the main neurotransmitter of the sympathetic nervous system causes vasoconstriction, decreasing kidney perfusion.40,41 A study in ewes showed norepinephrine increased ischemia in the kidney medulla. 42 Another hormonal system active in regulation of blood pressure, electrolyte and fluid balance is the renin-angiotensin aldosterone system (RAAS). 43 RAAS is activated when blood pressure, blood volume or sodium concentration is low. 44 In summary, renin is released by the kidneys and cleaves angiotensinogen into angiotensin I, which is then converted into angiotensin II by angiotensin converting enzyme, causing vasoconstriction and sodium retention.44,45 Important sex differences exist in the adrenergic and RAAS. 45
In rats, when AKI was induced by clamping the renal artery, kidney sympathetic nerve activity and plasma norepinephrine concentrations increased in both sexes. 46 However, one day after the injury, plasma norepinephrine concentrations, as well as plasma creatinine concentrations and histological kidney damage, were higher in male rats and ovariectomized female rats compared with female rats with intact ovarian function.46,47 The lower norepinephrine concentrations in female rats coincided with reduced rates of ischemic injury.46,47 Male rats exhibit higher baseline renin concentrations compared with female rats, and following kidney ischemia, the increase in renin was greater in males and persisted longer than in female rats. 48 This is beneficial for females as lower renin concentrations are associated with reduced apoptosis and kidney fibrosis. 48 In humans, men, from the age of 15 years, have higher renin concentrations compared with women and oral estrogen treatment reduced renin concentrations in women.49,50 Sexual dimorphism is also present in the response of healthy men and women to angiotensin II administration. Angiotensin II causes efferent vasoconstriction and, to a limited degree also afferent vasoconstriction. 51 This leads to an increase in GFR and a reduction in effective kidney plasma flow, thus resulting in an elevated filtration fraction. 51 In both sexes effective kidney plasma flow reduced after infusion of angiotensin II. 52 However, in men, GFR remained stable, which led to increased filtration fraction, whereas in women GFR decreased and filtration fraction remained stable. 52 As high filtration fraction is associated with increased glomerular pressure and kidney damage, the stable filtration fraction after angiotensin II infusion in women suggest a kidney protective effect. 53 To conclude, adrenergic and RAAS control differ between sexes with evidence for more beneficial kidney responses in (premenopausal) women.
Altered Kidney Oxygen Availability
The kidneys have a remarkably high oxygen demand, surpassed only by the heart in terms of oxygen consumption normalized to tissue mass. 54 As a result, the kidneys are vulnerable to hypoxia, which occurs when there is an imbalance between oxygen delivery and oxygen demand. Particularly vulnerable is the medulla, where oxygen concentrations are lower compared to the cortex (10-20 mm Hg compared to 50 mm Hg in the cortex). 39 Despite this, the medulla is responsible for most of the active sodium reabsorption, a process that requires a significant amount of oxygen (approximately 80% of the total oxygen consumption of the kidney).55,56 Two possible causes of impaired oxygen delivery during cardiac surgery, are hypoperfusion, as discussed in more detail above, and iatrogenic hemodilution. Hemodilution is necessary for the use of cardiopulmonary bypass during cardiac surgery and decreases the oxygen-carrying capacity of blood.
Regarding sex differences in the oxygen-carrying capacity, women are known to have overall lower hematocrit, but also to have a greater hematocrit drop during cardiopulmonary bypass. 57 Although women have lower hematocrit, they seem to tolerate hemodilution better than men; when hematocrit was below 26%, women had a lower risk of developing severe CSA-AKI compared with men. 57 A possible explanation is that women possess more adaptive mechanisms to cope with lower hemoglobin concentrations. 57 An example of adaptation is that in women, the hemoglobin-oxygen dissociation curve is shifted to facilitate greater oxygen delivery to tissues. 58
In addition to impaired oxygen delivery, an oxygen demand that exceeds supply can also contribute to hypoxia during cardiac surgery. Oxygen demand in the kidney is driven by active sodium reabsorption, a process that requires the production of adenosine triphosphate (ATP), which in turn is oxygen consuming. A rodent study showed that the metabolic activity, assessed using hyperpolarized pyruvate magnetic resonance imaging (MRI), in the kidney was higher in males versus females, even after correction of kidney weight, 59 suggesting that male kidneys have a higher oxygen demand compared with female kidneys. A possible explanation for this might be that testosterone is associated with increased expression of the sodium-hydrogen exchanger isoform 3 (NHE3) in the proximal tubule, leading to increased active sodium reabsorption and consequently increased kidney workload. 60 In line, analysis of single-cell profiling of human proximal tubule cells showed that men have increased mitochondrial aerobic metabolism compared with women, and a higher oxygen consumption rate. 61 Another factor important for kidney oxygen consumption is kidney insulin sensitivity as insulin resistance decreases oxygen-efficient ATP generation. 54 Estrogen is known to enhance insulin sensitivity in women, a benefit that diminishes after menopause. 62 In conclusion, compared with men's, women's kidneys might be better protected against a oxygen supply and demand mismatch through adaptions to lower hematocrits, a lower kidney oxygen demands and more oxygen-efficient ATP generation.
Inflammation and Oxidative Stress
Inflammation is known to be increased during and after cardiac surgery and contribute to the development of AKI. 63 This inflammatory response may last weeks after surgery, a study in female sheep showed that the macrophage infiltration to the kidneys lasted for four weeks after cardiopulmonary bypass. 12 Various factors contribute to this activation of kidney inflammation, including tissue hypoxia, ischemia-reperfusion injury, endothelial damage, exposure of blood to artificial surfaces such as the cardiopulmonary bypass, gaseous micro-emboli, and surgical trauma.64-66 While inflammatory and oxidative stress responses contribute to AKI, these are also affected by sex hormones. In a study, where AKI was induced by clamping the renal artery, tissue macrophage infiltration was reduced in male mice that were castrated, and increased following testosterone suppletion. 63 This was contrary to the finding in female mice, where macrophage infiltration was higher in ovariectomized mice and reduced after estrogen suppletion. 63 These findings were in line with another rodent study that observed that estrogen receptor activation reduced the macrophage invasion in the kidney. 67 The increase of pro-inflammatory factors tumor necrosis factor (TNF)-α, monocyte chemotactic protein (MCP)-1, interferon (IFN)-γ in the kidney was also more prominent in the male mice compared with female littermates. 63 In rodents, the number of anti-inflammatory regulatory T cells in females was higher compared with males, which may protect against kidney inflammation. 68 Another sex difference may arise from variations in response to oxidative stress, which is defined as an imbalance between the production and elimination of reactive oxygen species (ROS). 69 Oxidative stress can alter lipid, protein and DNA structures, potentially leading to cell death, tissue damage and inflammation. 70 In mice, testosterone was associated with an increased ROS production in the kidneys, whereas estrogen was associated with a protective antioxidant response. 69 In summary, testosterone is associated with more pro-inflammatory factors and ROS compared with estrogen. This influences not only AKI pathophysiology but potentially also the transition of AKI to CKD. In humans, women show better recovery of kidney function after AKI compared to men. 71 Preclinical studies investigating potential underlying mechanisms have shown inconsistent results: one study reported that female mice developed less fibrosis and were protected against CKD progression 72 whereas another study found greater fibrosis in female mice compared to males. 73 In the future, more preclinical research into sexual dimorphism in the AKI to CKD transition is needed to elucidate sex differences and the underlying mechanisms.
In conclusion, regarding the evidence for sexual dimorphism in CSA-AKI; Although the number of studies is limited, and predominantly based on rodent models, these findings suggest that estrogen might have a protective effect on the kidneys during acute injury. Premenopausal women may be better protected during hypoperfusion due to more vasodilatation of the afferent arteriole. They may also better tolerate low oxygen supply and have reduced oxygen demand, likely due to lower metabolism and greater insulin sensitivity. Furthermore, women may experience less inflammation, oxidative stress, and hyperfiltration. Considering these mechanistic insights, it could be hypothesized that women may have a lower risk of developing CSA-AKI.
Sex Differences in the Incidence of CSA-AKI
There are conflicting reports regarding the risk of CSA-AKI in women compared with men. A large meta-analysis showed that when including all patients (n = 1,057,412) of the 64 studies that provided sex-specific data regarding AKI incidence, women had a higher likelihood of developing CSA-AKI (odds ratio (OR), 1.21; 95% CI 1.09 to 1.33). 74 In a sub-analysis of studies that used kidney replacement therapy to define CSA-AKI, the risk difference between the sexes was even more pronounced, with again higher risks observed in women (OR, 1.70; 95% CI 1.19 to 2.44). 74 This observation has led to the conclusion of several studies that women are at risk to develop a more severe stage of CSA-AKI.75-78 However, when the abovementioned meta-analysis restricted the analysis to studies that included multivariate analysis to correct for covariates (most notably age and medical history), the CSA-AKI risk was not influenced by sex (OR 1.04; 95% CI 0.92 to 1.19). 74 This finding supports the idea that the higher incidence of CSA-AKI observed may be attributed to women's greater comorbidity burden and older age at time of surgery, rather than female sex itself being a risk factor for CSA-AKI. Earlier reviews indeed reported on sex differences in the cardiac surgery population: on average, women undergoing cardiac surgery are three to four years older and more often undergo emergency surgery, compared with men. 79 Furthermore, women undergoing cardiac surgery are more obese and more frequently present with other comorbidities, such as type 2 diabetes, hypertension, history of myocardial infarction, heart failure and more often use anticoagulants preoperatively. 80 Age, BMI, medical history, and emergency surgery are all established risk factors for CSA-AKI. 1 Interestingly, a retrospective cohort study on CSA-AKI incidence (n = 781) divided the study population in younger and older (cut-off 60 years) participants, and observed in the group below the age of 60, that the incidence of stage 1 CSA-AKI was lower in women compared with men.81,82 There were no sex differences in stage 2 and 3 CSA-AKI observed. 81 This significant difference between men and women disappeared in the men and women above 60 years of age. 81 This supports the hypothesis that when corrected for age, there is no sex difference in CSA-AKI incidence and that young women might even be protected from CSA-AKI. The role of estrogen in this finding in unclear but it could play a role as younger, premenopausal women typically have higher estrogen concentrations. This hypothesis is also supported by a prospective study (n = 4782) that observed that the incidence of CSA-AKI was lower in women receiving hormonal replacement therapy (HRT) compared with women without HRT or men (incidences respectively, 0.7% vs 5.3% vs 4.8%; P = .06). 83
In summary, female sex has frequently been considered an independent risk factor for CSA-AKI and is included as a risk factor in prediction models for CSA-AKI. 1 However, based on the aforementioned studies, the association between female sex and CSA-AKI appears to be influenced by factors such as age and comorbidities. After adjusting for these factors, the incidence was not higher in women. In contrast, there is evidence suggesting that women below the age of 60 years old, and women on HRT may have a lower incidence of CSA-AKI.
Sex Differences in Preventative Treatment Strategies for CSA-AKI
As discussed, the pathophysiological mechanisms underlying CSA-AKI may vary between sexes. Consequently, the optimal preventative approach may also differ between sexes. Therefore, it is important to investigate whether there are any notable sex-based differences in outcomes of clinical trials. However, the cardiac surgery population consists mostly of men, among patients who underwent CABG between 2011 and 2020, 25% were women. 84 This also results in fewer women being included in studies in the cardiac surgery population, which hampers the investigation of sex-specific treatment effects. Previous studies investigated the effects of furosemide,85-88 spironolactone, 89 fenoldopam, 90 levosimendan,91,92 erythropoietin,93-97 sodium bicarbonate,98-102 N-acetylcysteine,103-111 dexmedetomidine,112-115 methylprednisolone 116 and dexamethasone 117 on the prevention of CSA-AKI in both men and women. The fact that women were a minority in most of these trials, complicates drawing conclusions about the treatment effect in women (Table 1). This may have led to interventions being erroneously classified as ineffective for women, as evidence in women is lacking. Furthermore, the few positive studies did not include analyses to investigate sex differences in the treatment effect.97,109,112,114
Overview of Women Representation in Clinical Trials of Preventative Strategies for CSA-AKI.
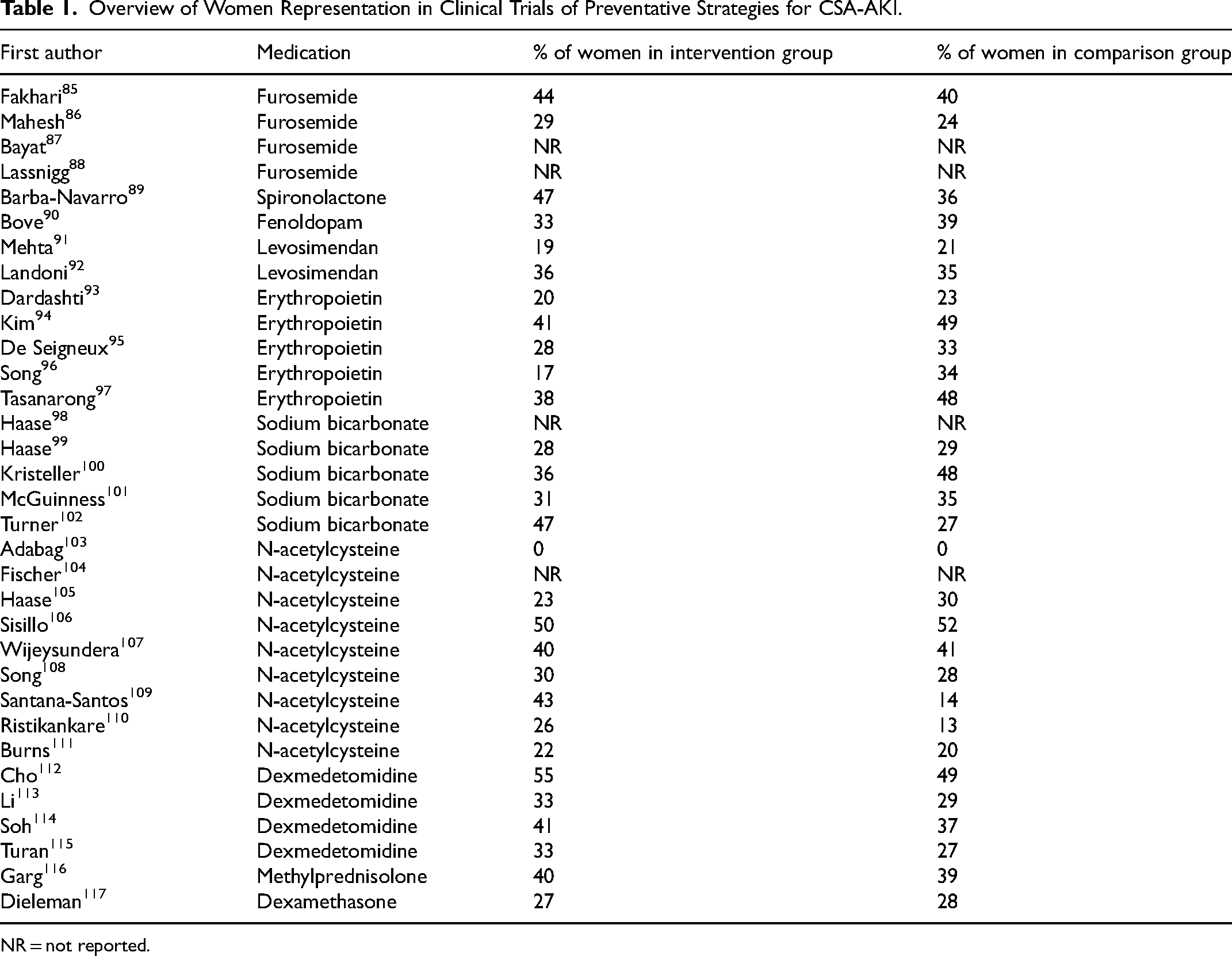
NR = not reported.
A new study with positive results to prevent CSA-AKI has recently been published. This study reported the beneficial effects of infusion of amino acids from the start of surgery to increase kidney perfusion and showed a lower incidence of CSA-AKI. 118 1057 out of 3511 participants (30%) were women in this trial. 118 Subgroup analysis showed that treatment effect was only significant in men (relative risk (RR) in men, 0.84; 95% CI 0.74-0.95, RR in women 0.89; 95% CI 0.73-1.08). 118 This could be due to the lower number of women in the study, which could have limited the statistical power to detect a significant difference. Additionally, no post hoc analysis including a sex-by-treatment interaction term was conducted. Alternatively, amino acid infusion may be more effective in men, as they experience greater vasoconstriction, leading to reduced kidney blood flow, and therefore may benefit more from amino acids.
To conclude, until now, much remains unknown about sex differences in the preventative strategies for CSA-AKI. Future studies should be designed to investigate treatment effects in women and to determine whether sex differences in the underlying mechanisms of CSA-AKI are reflected in the efficacy of preventive strategies.
Conclusion and Future Perspectives
This review explored sex differences in CSA-AKI regarding underlying pathophysiology, incidence and responsiveness to preventative strategies. While sex differences and the role of sex hormones in cardiovascular disease and CKD are well-documented, their role in CSA-AKI is still unknown. The underlying pathophysiology suggests that estrogen may have a protective role. Until today, it remains unclear whether estrogen also reduces the incidence of CSA-AKI in women. The cardiac surgery population consists mainly of men and previous research showed that women undergoing cardiac surgery are on average older and have more comorbidities, which precludes an equal comparison between men and women. However, the fact that the incidence of CSA-AKI was not higher in women after adjusting for age and comorbidities, and that, when including younger individuals (<60 years old), women had a lower incidence compared with men, indicates that female sex itself does not appear to be factor for CSA-AKI. Additionally, this review examined sex differences in new strategies to prevent CSA-AKI. However, the small number of women in the cardiac surgery population makes it challenging to determine whether these therapies are equally (in)effective for women.
In order to better understand the role of sex hormones in CSA-AKI, future research should investigate the CSA-AKI incidence in women before and after menopause, women with estrogen therapy, women after ovariectomy, men after orchidectomy and men and women with gender-affirming hormone therapy. Additionally, clinical trials should prioritize adequate representation of women to ensure that future interventions are also adequately studied in women. Understanding sex differences in CSA-AKI has the potential to improve personalized medicine and patient outcomes in the future.
Footnotes
Acknowledgements
YRL was supported by an Emerging Leader Investigator Grant by the National Health and Medical Research Council of Australia (GNT2025266) and a Future Leader Fellowship by the National Heart Foundation of Australia (FLF105666). AHH was supported by an European Union's Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement (No 101024833). DHvR is supported by a senior fellowship of the Dutch Diabetes Foundation (No 2021.82.010) and a Kolff fellowship from the Dutch Kidney Foundation (24OK2091).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
