Abstract
The risk of lumbar puncture (LP) to precipitate brain herniation in acute bacterial meningitis (ABM) was reviewed in this journal in 2007. We report the case of an infant with ABM who had acute apnea requiring intubation and tonic posturing (mistaken for seizure), and then had brain herniation within four hours of an LP. The case prompted this updated narrative review, from 2007 to 2024, focused on the twelve points made in 2007. The review included 14 case reports of brain herniation shortly after LP in ABM, 23 observational studies or systematic reviews, 28 narrative reviews, and 9 guidelines, each with evidence, advice, or recommendations important for the decision to perform LP in ABM. We found evidence to support, and did not find convincing evidence to refute, the twelve points. We found five additional claims made that were meant to refute some of the original points; however, these were based upon data that did not support the claims made. Limitation of the evidence reviewed was the absence of randomized trials to prove whether those patients who herniated may have been destined to herniate regardless of whether they had an LP. Reasons why ABM may be a unique circumstance where normal CT scan cannot determine the risk of herniation after an LP were discussed. We argue that the preponderance of evidence supported the conclusion that, in a patient with strongly suspected ABM who is clinically considered at high risk for herniation, interventions to control ICP and antibiotics administration should be the priority, followed secondarily by an urgent CT scan and, even with a normal CT, not an LP. The case report emphasized that respiratory arrest or suspected tonic seizure can be due to early herniation, and indicate CT scan, and prolonged LP deferral (for 3-4 days) even with a normal CT.
Introduction
A crucial decision in practice is when to consider delaying a lumbar puncture (LP) in suspected acute bacterial meningitis (ABM) due to the risk of the LP precipitating or worsening brain herniation. 1 One of us reviewed the literature on this topic in 2007, supplemented with another related review in 2010.1,2 Twelve important points relevant to this risk were made in those two papers, summarized in Table S1.1,2 The general conclusion was that, in those considered at high risk for herniation, interventions to control intracranial pressure (ICP) and antibiotics administration should be the priority, followed secondarily by an urgent computed tomographic (CT) scan and not an LP. 1 This was because clinical signs of ‘impending herniation’ were found to be the best indicators of who should not have an LP done [even with normal CT scan] in the setting of ABM due to the risk that the LP can precipitate or complete herniation. 1 These clinical signs included significant alterations in level of consciousness (often increasing in degree), seizure within 30 min (which often precedes a terminal respiratory arrest during herniation), decerebrate or decorticate posturing (that may be mistaken for a tonic seizure), respiratory abnormalities (Cheyne-Stokes, hyperventilation, or apnea), and pupillary changes. 1 Importantly, evidence was reviewed that demonstrated that a CT scan cannot reliably detect high ICP or risk of herniation in ABM. 1
A recent case of ABM having brain herniation shortly after an LP prompted us to update the review. The objective was to determine evidence published since the 2007 review that supported or not the twelve important points previously made. The case report and updated narrative review led us to re-emphasize the important points previously made, and also to draw attention to points that were not sufficiently emphasized in the previous reviews.
Case Report
Signed informed consent to publication of this case report was provided by the patient's legal guardian. An 18-month-old previously healthy fully immunized male presented to a rural hospital emergency department (ED) with a 3-day history of fever, cough, irritability, loose stools, and emesis. He was diagnosed with bilateral acute otitis media, started on oral clarithromycin, and discharged home. Over the next day at home he experienced increasing lethargy, refusal to walk, and poor appetite, and therefore returned to the rural ED. At that time, he received 40 ml/kg of intravenous (IV) fluids for dehydration, blood culture was drawn, and ceftriaxone IV given. He was then transferred to a regional ED for further management, where, upon arrival, his HR was 190 and capillary refill 4 s. There he received another 10 ml/kg IV fluid, and an epinephrine infusion at 0.1 mcg/kg/min was started. Approximately 15 min after arrival he had sustained apnea requiring bag-valve mask ventilation, unresponsiveness, and tonic posturing thought to be a tonic seizure. He was intubated with ketamine anesthetic, with initial end tidal carbon dioxide of 70–90 mm Hg, and was loaded with levetiracetam (60 mg/kg IV). Due to the tachycardia, he was given one dose of adenosine. At this time the pediatric intensive care unit (PICU) transport team was dispatched to transfer the patient to our children's hospital.
On arrival of the transport team, he was hemodynamically stable, ventilation was optimized (pH 7.3 and PCO2 40 mm Hg on capillary blood gas), sedation was adjusted to a propofol infusion, vancomycin and acyclovir IV were administered, and he was transported to our PICU. Upon arrival to PICU the epinephrine infusion was discontinued, and the propofol stopped. Approximately 3 h after stopping propofol his Glasgow Coma Scale (GCS) was 9, with localization to stimuli, opening eyes to pain, equal reactive pupils, and no focal neurological deficits. No CT scan was performed at any point prior to the LP. An LP was done with anesthesia (using ketamine) on the first attempt approximately 17 h after the episode of apnea and intubation. Cerebrospinal fluid (CSF) analysis showed protein 1.05 g/L, glucose 1.0 mmol/L, RBC 150 × 106/L, WBC 2669 × 106/L with 72% neutrophils, and gram-positive cocci in pairs on gram stain, confirming ABM. Opening pressure was not measured. Blood Culture grew Streptococcus pneumonia sensitive to penicillin, and CSF culture was negative.
Less than 4 h after the LP he developed decerebrate posturing, decreased GCS to 4, and sunset eyes. Mannitol and 3% saline were administered. An urgent head CT performed less than 1 h later showed marked cerebellar edema concerning for ischemia with tonsillar herniation, early uncal herniation, and acute obstructive hydrocephalus due to occlusion at the foramen magnum (Figure 1).
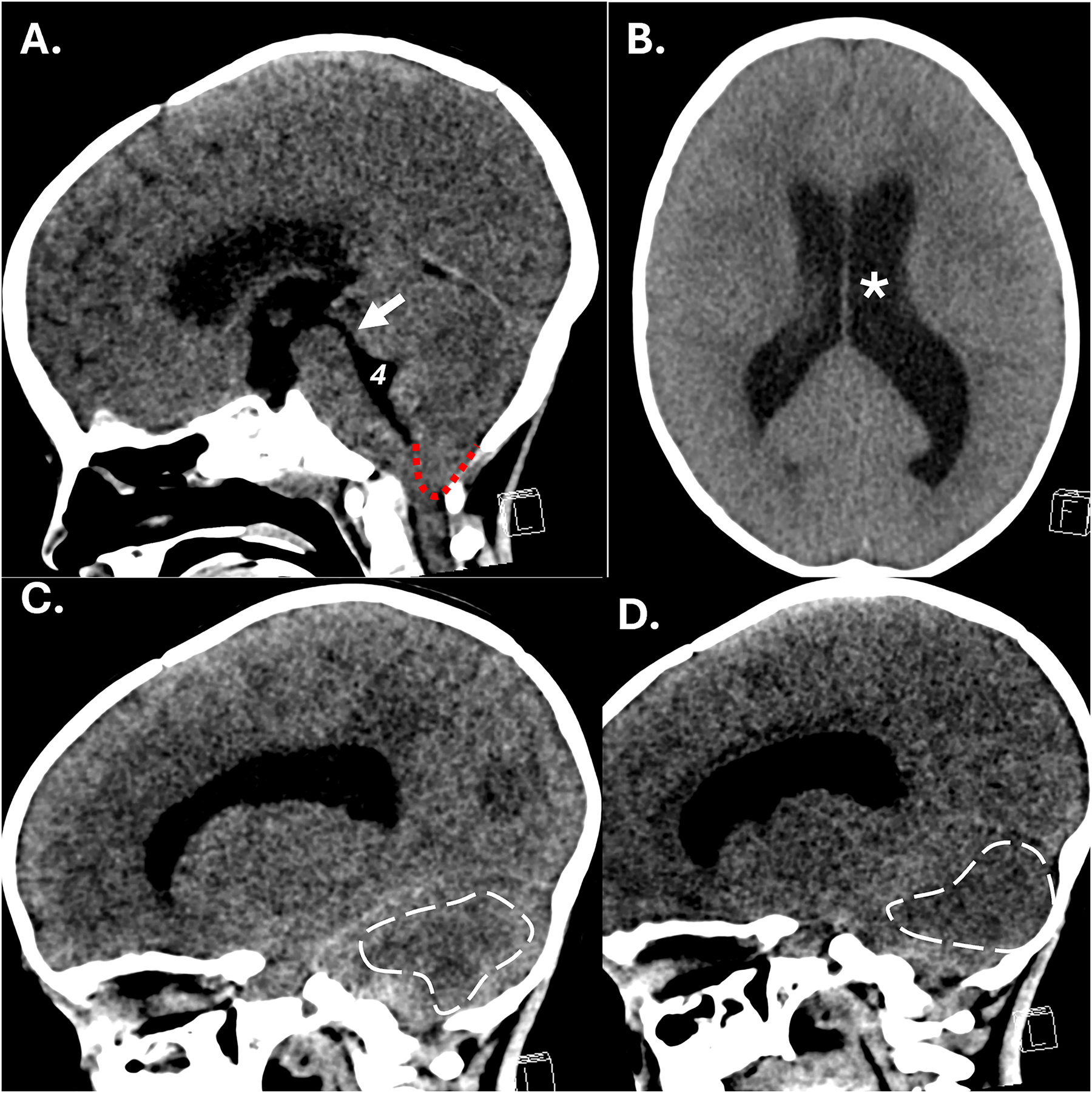
Noncontrast head CT performed just under 4 h after the LP. A) Sagittal image shows severe downward cerebellar tonsil herniation (red dotted line) that occludes the foramen magnum and causes acute obstructive hydrocephalus at the level of the foramen magnum with dilation of the fourth ventricle (4) and the aqueduct of sylvius (white arrow), as well as mass effect on the brainstem and effacement of the pre-pontine cistern. B) Axial image shows dilation of the lateral ventricles (asterisk) with effacement of the sulci/gyri along the cerebral convexities indicative of increased intracranial pressure from the acute obstructive hydrocephalus. The third ventricle was also dilated (not shown). Parasagittal cuts through the right C) and left D) cerebellar hemispheres show evolving early hypoattenuation involving the posterior inferior aspects of the cerebellar hemispheres bilaterally (dashed white line), indicative of early ischemic change in the posterior inferior cerebellar artery (PICA) vascular territories. This pattern of ischemic injury is seen specifically with acute, severe downward tonsillar herniation through the foramen magnum, which causes compression/compromise of the bilateral PICA's as they come off the high vertebral arteries.
Within one hour after the head CT, the patient underwent emergent insertion of an external ventricular drain (EVD). The ICP initially measured 25 cmH2O, and decreased to 12 cmH2O with CSF drainage. An MRI immediately after the operation showed improved but still significant tonsillar herniation and hydrocephalus, as well as diffuse ischemia in the posterior inferior cerebellar hemispheres bilaterally corresponding to the posterior inferior cerebellar artery vascular territories (Figure 2).
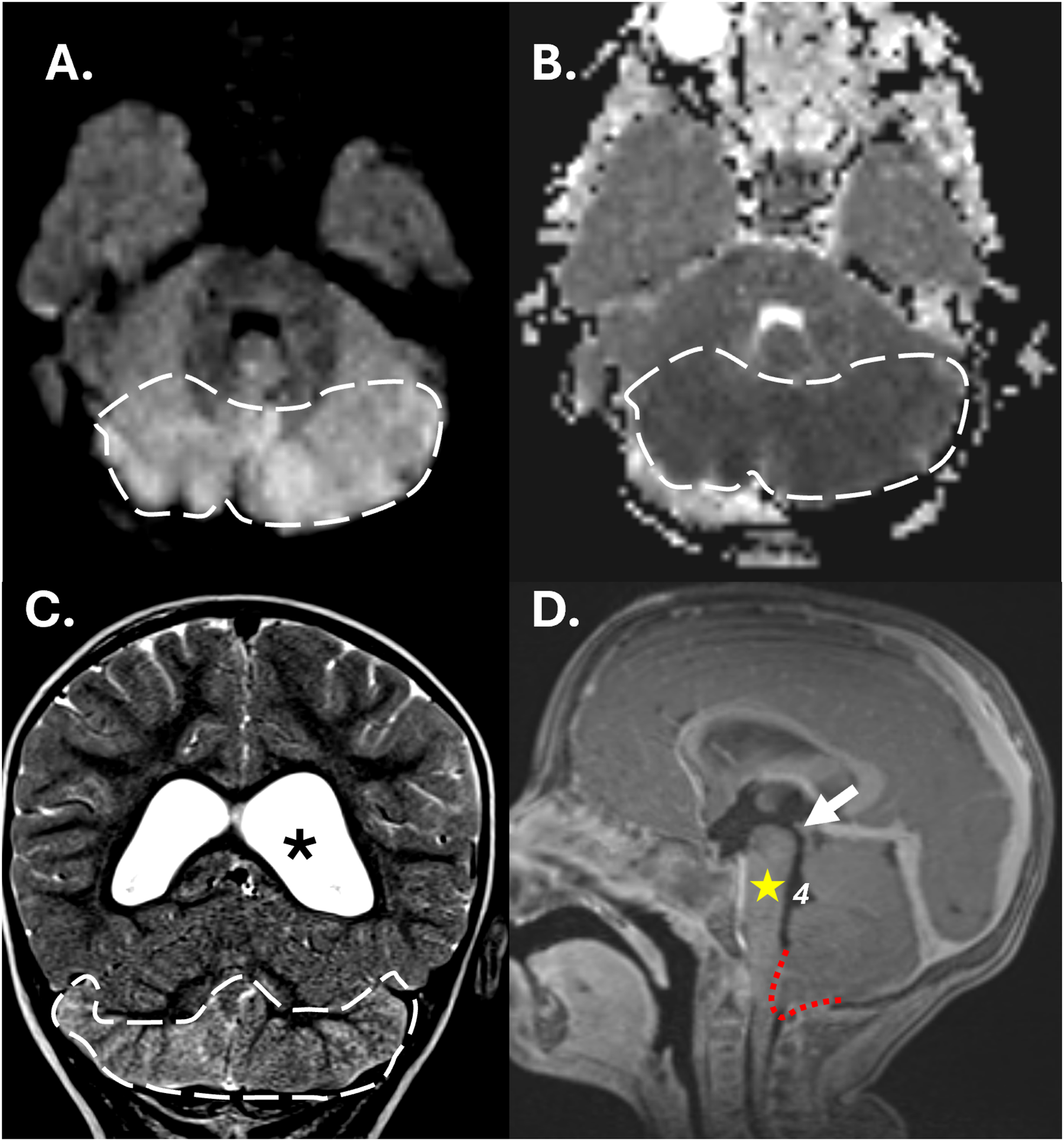
MRI of the brain with and without IV contrast performed immediately after external ventricular drain placement, 2.25 h after CT scan, and 6 h after LP. Axial diffusion-weighted imaging (DWI) (A) and corresponding Apparent Diffusion Coefficient (ADC) map (B) show diffuse signal abnormality (dashed white line) indicative of cytotoxic edema from ischemia involving the posterior inferior cerebellar hemispheres bilaterally, again corresponding to the bilateral PICA territories. Coronal T2-weighted image (C) shows ongoing dilation of the lateral ventricles (asterisk) immediately post EVD placement with corresponding abnormal T2-hyperintense signal in the cerebellar PICA territories at the sites of ischemia (white dashed line). Sagittal post-contrast T1-weighted image (D) shows improved but still severe cerebellar tonsil herniation (red dotted line), with ongoing mild mass effect on the pons (star) and pre-pontine cistern, as well as dilation of the fourth ventricle (4) and the aqueduct of sylvius (arrow). Additional minor scattered DWI signal abnormality was noted in the cortex of the bilateral parietal and occipital regions, as well as along the ventricular lining, in keeping with changes from meningitis-related sites of cerebritis and ventriculitis. No additional large territorial vascular injury related to herniation was identified.
Four days after EVD placement, upon weaning of sedation, he again developed an increased ICP (15-20 cmH2O). The GCS remained at 7–8 and there was now left hemiparesis (arm worse than leg). Repeat CT and MRI at this time demonstrated severely increased edema and bulkiness of the ischemic cerebellum with resultant significantly worsened hydrocephalus, now with trans-ependymal flow of CSF, and progressive severe compression of the brainstem (Figure 3). Within hours he underwent a second emergent neurosurgery for posterior fossa decompression, and resection of both the infarcted cerebellar tonsils and portions of infarcted cerebellum.
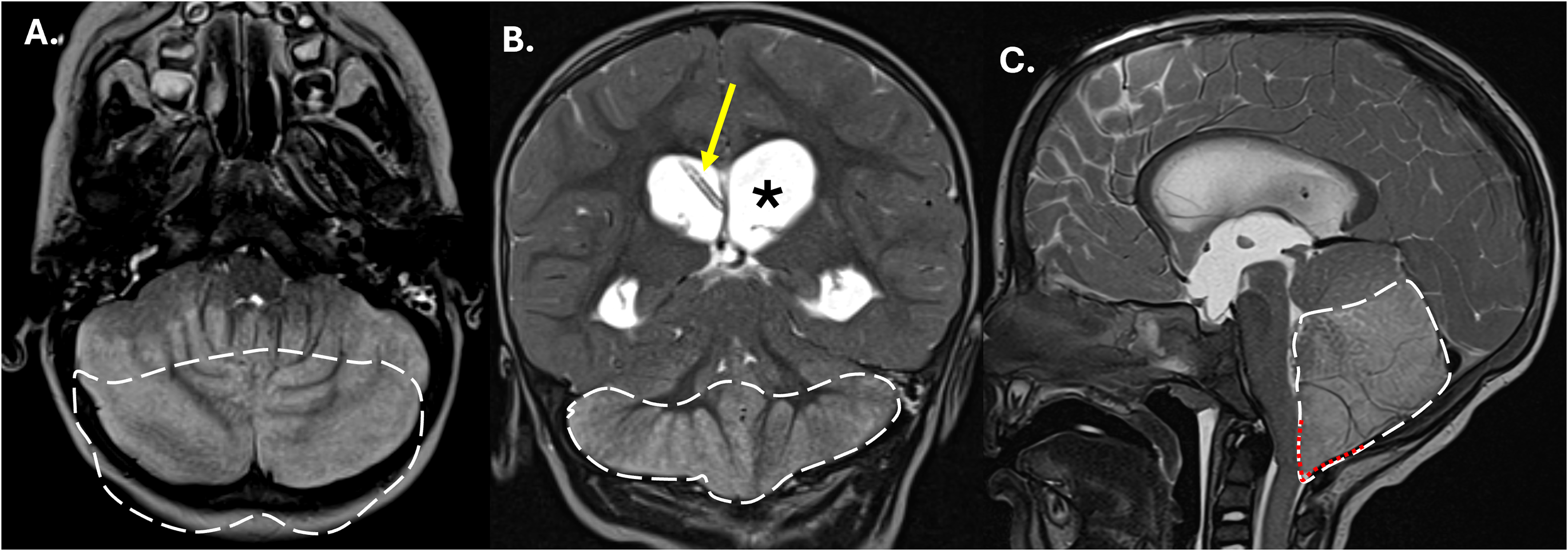
MRI of the brain performed 4 days after LP and external ventricular drain placement. Axial (A), coronal (B), and sagittal (C) T2-weighted images show worsened T2-hyperintense edema at site of posterior inferior cerebellar artery territory infract in the bilateral cerebellar hemispheres (dashed white line), with worsening mass effect in the posterior fossa where there is increasing tonsillar herniation (red dotted line), increased flattening of the brainstem, and worsening of obstructive hydrocephalus (asterisk) in spite of EVD placement (yellow arrow).
Ten days after his arrival at our PICU he was extubated. He was discharged from PICU with left hemiparesis, swallowing dysfunction, no verbal communication, and poor neck and truncal tone. Four weeks later, he was discharged to the rehabilitation hospital, where he continued to demonstrate improvements. At discharge from the rehabilitation hospital he had ataxia, was crawling, self-feeding, interactive and speaking some sentences, without residual hemiparesis.
Methods
We searched PubMed in October 2024 using the title/abstract terms ‘meningitis’ and ‘herniation’; ‘meningitis’ and ‘guideline’; and searched citations of Joffe 2007. 1 The search was from January 2007 to October 2024. Reference lists in relevant articles were screened for eligibility, and included publications’ reference lists were in turn screened for further relevant publications. We included any publication that was: i) a case report of brain herniation in ABM, or ii) a case series that presented data on brain herniation in ABM or on the risk of LP in ABM, or iii) a review article or guideline on AMB that discussed contraindications to LP. Study inclusion was determined by one of the authors after review of abstract and if necessary full text. Information from each included study was recorded according to its relevance to the previous 12 important points made. A narrative synthesis of this evidence was completed, as the outcomes (ie, relevance to each of the 12 points) were not amenable to meta-analysis. Evidence synthesis was first done by one of the authors, and then reviewed by all authors for accuracy.
Results
Case Reports of Brain Herniation in ABM
In addition to the 221 and 102 case reports of brain herniation occurring shortly after an LP in ABM previously reviewed,1,2 there were 14 more cases, detailed in Table 1.3–15 Most cases described pre-LP altered level of consciousness (10), and some reported recent seizure (1), anisocoria (1), apnea (1), repeated vomiting (6), or worsening headache (1). Pre-LP CT was normal in 5 and abnormal in 2.
Observational Studies with Data on Brain Herniation or Risk of LP in ABM
Since the original reviews, 23 relevant observational studies or systematic reviews were found.16–40 These are presented in Table 2, organized by whether and how each supported one (or more) of the 10 points made in the previous review. Two additional claims were made in these case series that may appear to be contrary to our general point 10; however, limitations in the data used to support these claims make them unsupported by data (detailed in Table 2, Comments column).
Updated Observational Studies or Systematic Reviews with Relevant Evidence About When to do a Lumbar Puncture in Acute Bacterial Meningitis.

ABM: acute bacterial meningitis; aOR: adjusted odds ratio; CNS: central nervous system; CT: computed tomography scan of head; GCS: Glasgow Coma Score; ICP: intracranial pressure; IQR: interquartile range; LP: lumbar puncture; OP: opening pressure measured on the lumbar puncture.
Narrative Review Articles on ABM That Discussed LP Contraindications in ABM
Since the original reviews, 28 relevant narrative reviews were found.41–68 These reviews are presented in Table 3, organized by whether and how each review supported one (or more) of the 10 points made in the previous reviews. Three additional claims were made in these reviews that may appear to be contrary to our points 4, 9 and 10; however, limitations in the data used to support these claims make them unsupported by data (detailed in Table 4, Comments column). The narrative reviews often gave variable advice on indications for CT prior to LP, ambiguous advice on whether normal CT was sufficient to warrant an LP in the presence of risk factors, and often did not state that normal CT does not determine the safety of LP in ABM (Table 3).
Updated Narrative Reviews Discussing Relevant Advice on When to do a Lumbar Puncture in Acute Bacterial Meningitis.

ABM: acute bacterial meningitis; CT: computed tomography scan of head; GCS: Glasgow Coma Score; ICP: intracranial pressure; LP: lumbar puncture; OP: opening pressure.
Updated Guidelines Discussing Relevant Recommendations on When to do an LP in ABM. a
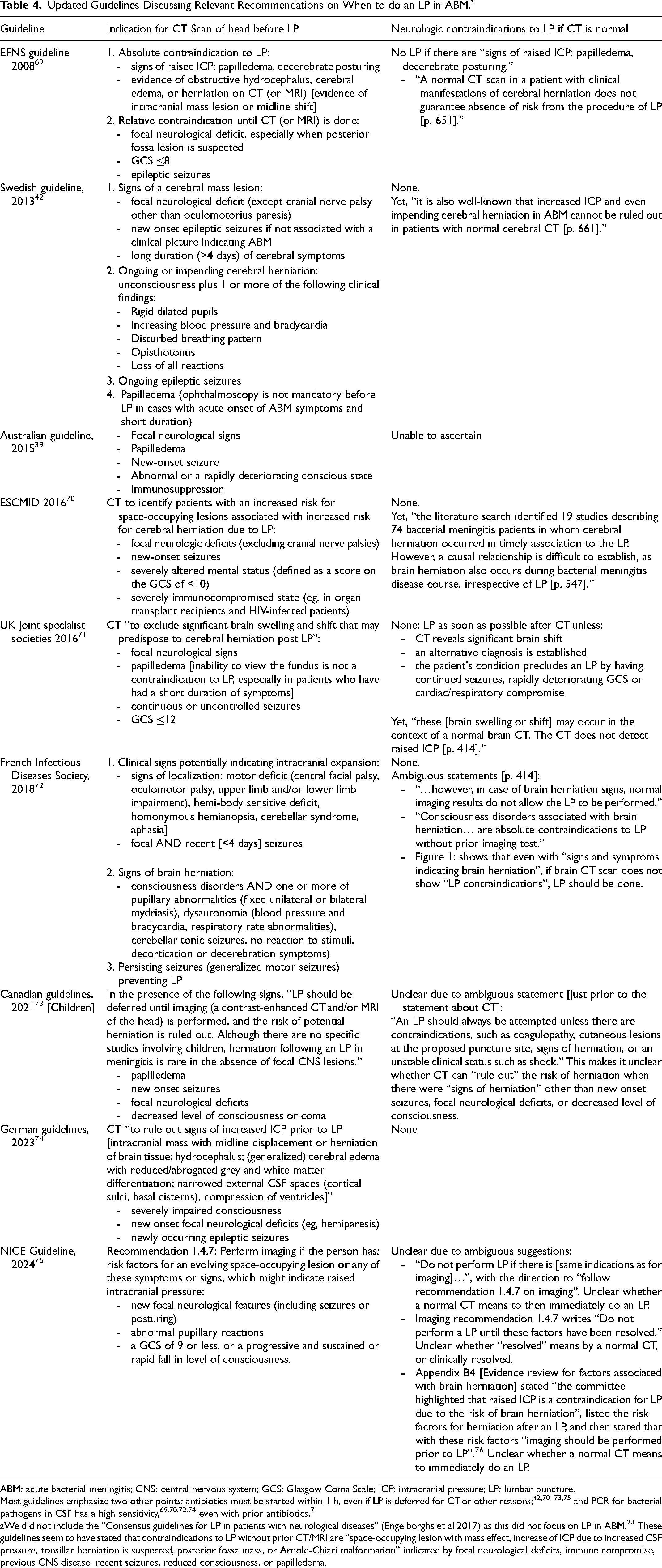
ABM: acute bacterial meningitis; CNS: central nervous system; GCS: Glasgow Coma Scale; ICP: intracranial pressure; LP: lumbar puncture.
Most guidelines emphasize two other points: antibiotics must be started within 1 h, even if LP is deferred for CT or other reasons;42,70–73,75 and PCR for bacterial pathogens in CSF has a high sensitivity,69,70,72,74 even with prior antibiotics. 71
We did not include the “Consensus guidelines for LP in patients with neurological diseases” (Engelborghs et al 2017) as this did not focus on LP in ABM. 23 These guidelines seem to have stated that contraindications to LP without prior CT/MRI are “space-occupying lesion with mass effect, increase of ICP due to increased CSF pressure, tonsillar herniation is suspected, posterior fossa mass, or Arnold-Chiari malformation” indicated by focal neurological deficits, immune compromise, previous CNS disease, recent seizures, reduced consciousness, or papilledema.
Guidelines on ABM That Explain Contraindications to LP in ABM:
There were 9 different guidelines included, presented in Table 4, and organized by recommendations for CT and whether CT without imaging contraindications indicated immediate LP.39,42,69–76 There were differences in guideline listed indications for head CT prior to LP, mostly differing in the GCS level cut-off (with two only considering low GCS if other signs of brain herniation are present),42,72 in whether immunocompromised status was relevant, and in whether recent (non-ongoing) seizure were indications (Table 4). Six of the guidelines recommended LP if head CT did not reveal an imaging contraindication to LP regardless of neurologic clinical findings,39,42,70–72,74 although one had ambiguous statements about this 72 ; two were unclear on this recommendation,73–76 and one stated that even with normal CT “signs of raised ICP: papilledema, decerebrate posturing” contraindicate an LP. 69 Three of the guidelines stated that a normal head CT does not rule out high ICP or risk for herniation,42,69,71 and two of these recommended that head CT without imaging contraindications indicated immediate LP regardless of clinical neurologic signs.42,71 In general, the different guidelines emphasized the need for urgent LP for diagnosis, and to prevent delays in starting antibiotic(s) treatment.
Discussion
Updated Literature Review Supported Previous Points
The updated literature review found further evidence to support, and did not find convincing evidence to refute, the original points made regarding the safety of performing LP in a patient with ABM (Table S1, Tables 1–4). This is important because, brain herniation accounts for a significant proportion of deaths in ABM (point 1),1,16–20 raised ICP is common in ABM (point 2),1,17,21 herniation after LP may be gradual due to ongoing CSF leakage from the LP site or cerebral vascular engorgement and edema after LP (point 3),1,22,23 and when herniation occurs in ABM patients it most often occurs shortly after an LP (point 4),1,3–15,20,21,26 together providing a compelling logical pathophysiologic explanation for the risk of brain herniation precipitated by an LP in ABM. Furthermore, the original overarching general point 10, that in those considered at high risk for herniation, interventions to control ICP and administration of antibiotics should be the priority, followed secondarily by an urgent CT scan and not an LP, remained supported by the updated literature reviewed.1,36–40 This is because the main rationales for this point were supported (beyond the pathophysiology mentioned above), that is, that a normal head CT cannot reliably detect an elevated ICP and therefore cannot determine the risk of herniation in ABM (point 5),1,21,27,28 and that clinical signs of ‘impending herniation’ are the best indicators of who should not have an LP done [even with normal CT] in the setting of ABM because of the risk that the LP can precipitate or complete herniation (point 7).1,17,20,21 Of note, in the narrative review articles these clinical signs were often framed as indications for CT prior to LP (point 6)11,47,52,54,55,58–63; however, many of these narrative reviews ignored the fact that CT cannot exclude all cases of increased intracranial pressure and brain shift (eg, diffuse brain shift with tense brain and dura starting to displace brain from one intra-cranial compartment into another) indicated by these clinical risk factors and that may contraindicate an LP even with a normal CT (point 5).11,55,59–63,68 In other words, these clinical signs that indicate head CT were not recognized as stand-alone contraindications to LP even with a normal CT.
The updated literature review found five additional claims made that were meant to refute some of the original points.36,42,46,67 These additional claims were based upon data that did not support the claims made (see Comments in Tables 3 and 4). Nevertheless, the main limitation of the data reviewed here remained the absence of any randomized controlled trial to prove whether those patients who herniated after an LP may have been destined to herniate regardless of whether they had an LP. Without an RCT, which in our view would be unethical, we are left to make the best use of observational data available. As argued above, we believe the preponderance of this evidence supports the overarching general point 10. Redetsky reviewed “fulminant” ABM where an LP “might only accelerate an otherwise inevitable process”. 43 Redetsky pointed out that fulminant meningitis may occur in up to 1% of ABM, and was “not equivalent to meningitis with clinical brain herniation.” 43 This was based on differences in the “rapidity of deterioration [sudden onset with rapid neurological deterioration],” and especially the “refractory nature of intracranial hypertension” and “irreversibility of intracranial hypertension” that “evolves swiftly despite attempts to control ICP” with death within 36–48 h. 43 These were not features in the majority of the reported cases of herniation after an LP in ABM (Table 1).1,2,4–6,9,10,13,14
The 9 guidelines reviewed made some discordant recommendations on indications for head CT prior to LP.39,42,69–76 Guidelines often (though not always) recommended immediate LP after a head CT without an identified imaging contraindication, regardless of clinical findings.39,42,70–72,74 As reviewed in Table 2, we believe the best evidence available does not support this recommendation, as head CT cannot rule out high ICP or risk for herniation after LP in ABM (as three guidelines stated).21,27,28 Reasons why ABM may be a unique circumstance for the risk of herniation after an LP despite normal CT include that purulent material may prevent the expected effacement of the extra-axial and ventricular spaces, meningeal hardening from inflammation may cause decreased meningeal compliance that then resists effacement, decreased CSF absorption may oppose the forces of cerebral edema, and, crucially, these factors may not be equally distributed throughout the brain, predisposing to unequal ICPs. 1 Again, this comes down to a judgment about the observational evidence for points 1–10, as discussed above.
The most common risk factor for herniation after LP in the described cases was a low or decreasing level of consciousness, without any other definitive clinical signs of herniation having occurred prior to an LP.6–9,13–15 Even so, as pointed out in the 2007 review, clinical risk factors identified in a minority of cases “seem to refer to patients that already have signs consistent with early uncal or central cerebral herniation… Doing an LP in that setting could complete the brain herniation. This makes pathophysiologic sense: an LP relieving pressure from below could allow a swollen brain with high ICP to develop more edema and to shift further and completely herniate.” 1
Some case series seemed to describe that rarely herniation was likely already starting prior to LP, eg, a case with decerebrate posturing, 77 dilated pupil before paralysis and intubation, 77 with obliterated basal cisterns and transient hemiparesis on admission, 78 and 4 cases with herniation “that reduced or cleared” after mannitol and then recurred after LP [no further details were provided]. 79 The 14 case reports of herniation added in Table 1 included one patient with “anisocoria”, 4 and one patient with “generalized edema causing brain shift with partial obliteration of basal cisterns [on CT]”, 11 both patients likely in the process of herniation prior to LP. Moreover, a seizure prior to LP, usually poorly described in the reported cases,2,11,15 may actually have been posturing during herniation that was mistaken for a tonic seizure. Whether the decreased level of consciousness described in other case reports was indeed due to early herniation cannot be determined; however, that CT scan cannot detect the risk for brain herniation occurring shortly after the normal CT suggests otherwise, ie, the CT did not find early herniation.9,12–15 In these cases the normal CT scan provided false reassurance, while the clinical signs suggested a high risk of herniation that subsequently occurred after LP was performed.
The Added Value of our Case Report
We reported our case because it made three points that may not have been adequately emphasized previously, or may have been misunderstood.
First, whether occurring before or after an LP, brain herniation often presents as a variable combination of apnea and quadriplegia (point 11 and 12),
2
often with a rapidly decreasing level of consciousness, and pupillary changes (unequal or fixed and dilated). These are clinical risk factors for abnormal CT,29–31,41,44–56 as well as for herniation or completion of herniation after an LP.11,17,20,21,47,52,54,55,58–63 The patient in our case report had an acute event of respiratory arrest and likely tonic (decerebrate) posturing (confused for a seizure), followed by rapid sequence intubation that later reversed these findings (ie, aborted the herniation episode). These initial findings were an indication of early herniation and indicated a head CT, which was not done and
Second, within hours of the LP there was acute deterioration in the level of consciousness, and subsequent head CT indicated tonsillar herniation and acute obstructive hydrocephalus. The LP was done less than 17 h after intubation was done for signs compatible with early herniation. When ICP is elevated in ABM it is likely that the underling cerebral edema and hyperemia causing elevated ICP does not resolve for days. 1 In our case we cannot know whether a head CT would have detected diffuse cerebral edema or acute hydrocephalus that contraindicated an LP. However, even if a CT had been done, and the CT had been normal, the clinical risk factors indicating early herniation suggested a high risk of completed herniation to occur after an LP.1,17,20,21,44,48–52,58,64–66 Therefore, our case emphasized that clinical risk factors that indicate prolonged deferral of LP include acute respiratory arrest or suspected tonic seizure as these can be due to early herniation (point 7).
Third, the initial episode of herniation was aborted with anesthesia (ie, rapid sequence intubation), mechanical ventilation, and ongoing sedation (with propofol infusion). The second completed episode of herniation was reversed with mechanical ventilation, ongoing deep sedation, hyperosmolar therapy, and decompressive surgery. The initial treatment created the misleading impression that herniation had not occurred at all, and the improved clinical findings were why an LP was done. Treatment later led to survival with hemiplegia as an outcome at acute-care hospital discharge. Therefore, our case emphasized that if herniation occurs, this should be treated aggressively as the outcome is not necessarily dismal (point 8).1,2,4–6,9,14,28
Limitations
This updated review has limitations. First, we only searched one database (PubMed), and could have missed papers indexed in other databases. The snowballing approach to reviewing references in identified publications mitigated this concern. Second, there were no randomized controlled trials, and inferences were made based on observational data. In particular, whether herniation would have occurred without having had an LP cannot be certain. Nevertheless, as argued above, we believe the preponderance of evidence supports the overarching general point 10. Third, adverse effects of delaying an LP in suspected ABM need to be considered. Early administration of antibiotics is crucial when LP is delayed. We found no updated information on potential effects of delayed LP after antibiotics on making diagnoses, and rely on point 9 in Table S1 and Table 2 to suggest this risk is very small.1,32–35 The LP may be done later when the clinical course has suggested that elevated ICP is no longer suspected (eg, after peak edema resolves in at least 2-3 days,80–84 with resolution of the clinical finding(s) that suggested elevated ICP), and PCR tests for bacterial pathogens are still reliable
The case report also has limitations. First, case reports provide a low level of evidence, and making strong inferences from a case report should be done with caution. This is why we presented the case report in the context of our previous and updated literature review. Second, how long to delay an LP when clinical risk factors are present is unknown. Since elevated ICP peaks in the first few days of ABM, this is likely for at least 2–3 days (and resolution of the clinical finding(s) that suggested elevated ICP if longer).80–84 In the setting of an initial suspected herniation event, this may be longer to ensure resolution of the peak elevated ICP.
Conclusion
An updated literature review supported the general conclusion that, in a patient with strongly suspected ABM who is clinically considered at high risk for herniation, interventions to control ICP and antibiotics administration should be the priority, followed secondarily by an urgent CT scan and [even with a normal CT] not an urgent LP. The urgent CT scan may detect a contraindication to LP; however, a normal CT scan in the setting of ABM does not rule out very high ICP with risk for brain herniation after an LP.21,27,28,41,44–57,69 We report a case that led us to emphasize three points: clinical risk factors that indicate an urgent CT in suspected ABM include acute respiratory arrest or suspected tonic seizure as these can be due to early herniation, clinical risk factors that warrant prolonged (for 3-4 days) deferral of LP [even with a normal CT] include acute respiratory arrest or suspected tonic seizure as these can be due to early herniation, and if herniation occurs it should be treated aggressively as the outcome is not necessarily dismal.
Supplemental Material
sj-docx-1-jic-10.1177_08850666251337684 - Supplemental material for Lumbar Puncture and Brain Herniation in Acute Bacterial Meningitis: An Updated Narrative Review
Supplemental material, sj-docx-1-jic-10.1177_08850666251337684 for Lumbar Puncture and Brain Herniation in Acute Bacterial Meningitis: An Updated Narrative Review by Ari R. Joffe, Fernanda de Marzio Pestana Martins, Daniel Garros and Adrienne F. Thompson in Journal of Intensive Care Medicine
Footnotes
Author Contributions
All authors made substantial contributions to conception or design of the work, and interpretation of data for the work; reviewed the work critically for important intellectual content; made final approval of the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. ARJ drafted the first version of the manuscript, FDMPM drafted the first version of the case report section, and AFT drafted the first version of the figures and figure legends.
Consent for Publication
Signed informed consent for publication of the case report was obtained from the legal guardian of the 18 months old patient.
Data Availability
All data used are given in the publication, and the original reference for that data provided.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The University of Alberta health research ethics board does not review case reports. Approval was not required for the updated review as we used only already publicly available published data for review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.