Abstract
During critical illness, patients experience significant and rapid onsets of muscle wasting and dysfunction with loss of strength, mass, and power. These deficits often persist long after the ICU, leading to impairments in physical function including reduced exercise capacity and increased frailty and disability. While there are numerous studies describing the epidemiology of impaired muscle and physical function in the ICU, there are significantly fewer data investigating mechanisms of prolonged and persistent impairments in ICU survivors. Additionally, while several potential clinical risk factors associated with poor physical recovery have been identified, there remains a dearth of interventions that have effectively improved outcomes long-term among survivors. In this article, we aim to provide a thorough, evidence-based review of the current state of knowledge regarding muscle dysfunction and physical function after critical illness with a focus on post-ICU and post-hospitalization phase of recovery.
Introduction
Clinical advances in intensive care medicine over the last several decades have significantly improved survival rates. Over 5.7 million adults in the United States are admitted to the intensive care unit (ICU) annually, and of those, approximately 4 million survive their hospitalization.1,2 We have increasingly come to understand that survivors’ struggles don’t stop once they leave the doors of the ICU and the hospital. Intensive care clinicians now recognize that patients who survive critical illness can be left with debilitating deficits in physical, cognitive, and mental health, collectively known as the “post-intensive care syndrome” or PICS. 3
The roots of PICS take hold early during a critical illness. During the first week of an ICU stay, patients, on average lose 10% or more of their muscle mass. 4 While some survivors may recover muscle mass, up to 75% still exhibit persistent muscle weakness months and years after their critical illness.5,6 As a direct consequence of the acute skeletal muscle dysfunction that develops during critical illness, up to half of survivors have impaired physical function and reduced physical activity resulting in significant performance limitations requiring endurance (eg, walking a quarter of a mile) and/or muscle strength such as lifting and carrying everyday items such as a shopping bag.7–11 These physical impairments often lead to the distinct, but related, syndromes of frailty and disability that affect over on third of survivors.12–14 Most importantly, survivors of critical illness rank physical function as one of the most important outcomes after an ICU stay. 15
Despite these alarming statistics surrounding the lack of physical recovery after the ICU and the importance placed on functional status by patients, our
Trajectories of Muscle and Physical Dysfunction due to Critical Illness
Muscle Weakness in the ICU
With the profound insult of critical illness, patients frequently develop rapid-onset muscle weakness and atrophy, with patients losing up to 2% of muscle mass per day during the first week of ICU. 21 This loss of muscle mass and strength is frequently accompanied by a symmetric, distal sensory-motor axonal polyneuropathy that affects both limb and respiratory muscles,22–25 collectively known as the clinical syndrome of ICU-acquired weakness (ICU-AW). It is estimated that one quarter to one third of mechanically ventilated patients develop ICU-AW.26,27 Common clinical presentations include flaccid quadriparesis in the limb muscles with atrophy, reduced or absent deep tendon reflexes, difficulty weaning from mechanical ventilation, and reduced peripheral sensation.28,29 ICU-AW is most commonly diagnosed using standardized clinical criteria such as manual muscle testing.30,31 While methods such as electrophysiologic testing and muscle biopsy may provide more specific evidence of neuromuscular derangements, practical limitations to such techniques such as the technical expertise required and contraindications such as coagulopathy limit the practicality of such methods for routine diagnosis. 32 Other non-invasive approaches such as handgrip dynamometry show strong correlation with manual muscle testing 33 but are frequently limited by the need for patient participation which may be limited due to sedation or severity of illness. Imaging methods such as muscle ultrasound are increasingly being used to identify muscle atrophy in the ICU and correlate well with pathological findings such as myofiber necrosis. 34 However, small studies to date have suggested that muscle ultrasound may not reliably discriminate between patients with and without ICU-AW. 35 Given the multiple limitations in diagnostic evaluation for ICU-AW, along with a lack of routine screening for pre-existing muscle weakness (ie, sarcopenia) in those with critical illness, the true prevalence and incidence of ICU-AW syndrome is unclear.
Recovery After Critical Illness
While reduced muscle strength, lower muscle mass, and neuropathic changes can be detected early during critical illness, recovery from these impairments varies considerably. For example, some patients may develop sustained impairments in physical function resulting in no recovery, while others may have some return of function, but not back to their pre-critical illness baseline having partial recovery, while impairments in others may resolve completely, allowing a patient to make a full recovery back to their pre-critical illness physical function (Figure 1).
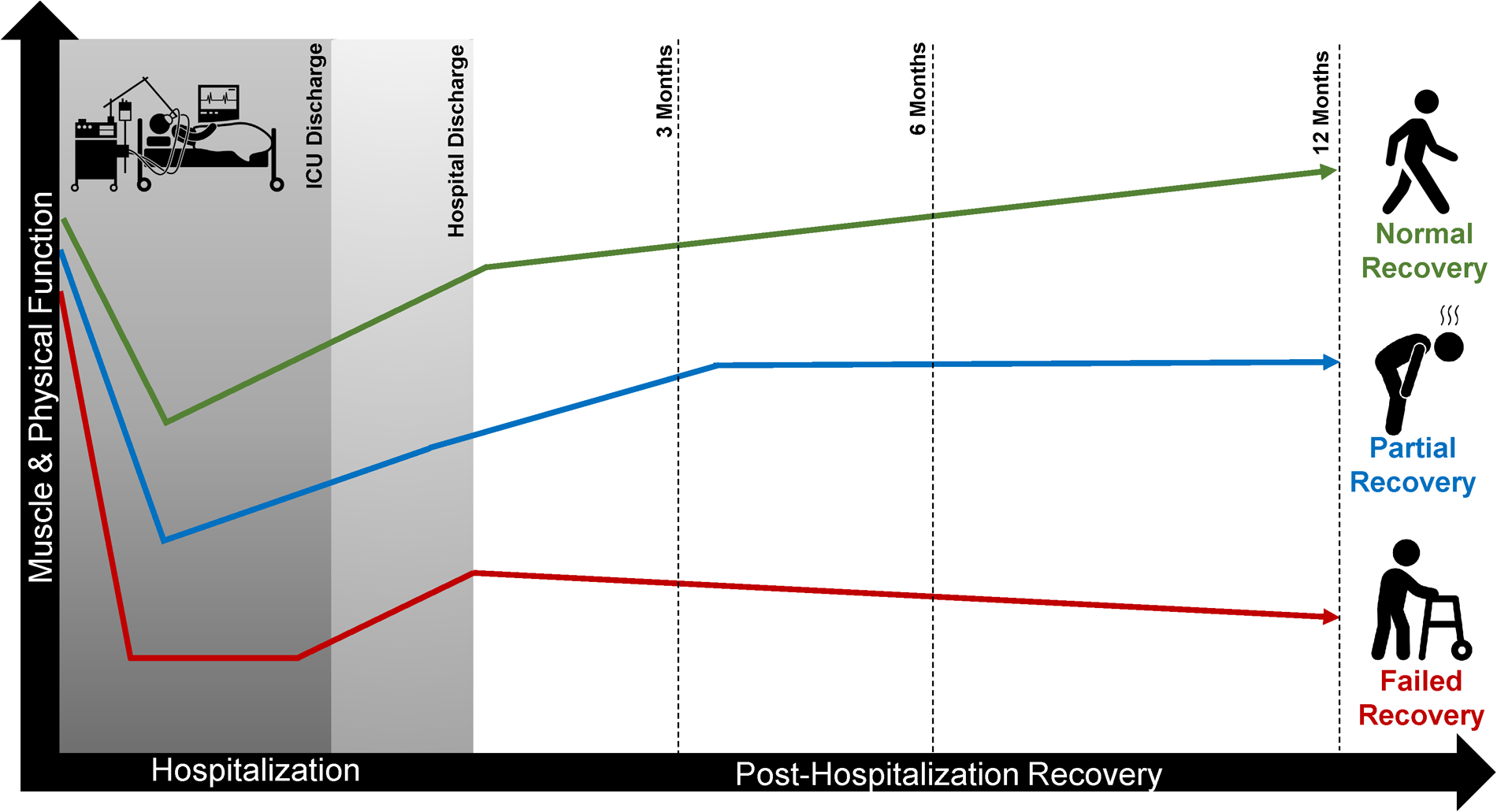
Potential recovery trajectories after critical illness. While almost all patients who are severely ill have a significant fall in both muscle and physical function during the initial phases of critical illness, some individuals will recover back to normal by 6-12 months after critical illness while others have only partial recoveries. Other individuals may completely fail to recovery and be left with significant physical impairments that lead to disability and reduced quality of life.
Illustrating the heterogeneity of recovery of physical function are data from several cohorts of survivors of critical illness who underwent serial physical function testing. Fan and colleagues found that 36% of survivors of acute respiratory distress syndrome ARDS met criteria for ICU-AW at hospital discharge, but by 1 year and 2 years post-ICU, only 14% and 9% of patients, respectively, met the same criteria for ICU-AW. 27 While these data suggest that recovery occurs in many, it should be noted that ICU-AW is a risk factor for mortality, thus, the lower prevalence of ICU-AW at follow-up could be due to weaker patients who were not assessed because they had died.
A second cohort study reported impaired exercise capacity in ARDS survivors. Herridge et al, reported the median six-minute walk distance (6MWD) in a relatively young cohort of ARDS survivors (median age 45 years) was 66% of predicted at one year after hospitalization. 7 At five year follow-up, these same ARDS survivors’ median 6MWD had only improved to 76% predicted. 8 Similarly, in a 5 year follow-up study to a randomized trial of early versus late parenteral nutrition in critically ill adults found that 38% of survivors had reduced peak oxygen consumption, a measure of cardiorespiratory fitness. 9 Finally, a recent systematic review in ICU survivors found that while 6MWD improved from 3 months to 12 months after hospitalization, it remained below predicted population norms with no further improvement out to 60 months. 36
Clinical factors that are associated with different trajectories of recovery in survivors of critical illness remain incompletely characterized. A systematic review by Parry and colleagues reported that both female sex and pre-existing comorbidities were associated with lower 6MWD, while none of the ICU-related factors were associated with this outcome. 36 Older age has also been identified as an important predictive factor for worse recovery after critical illness and lingering disability in multiple critically ill cohorts.37,38 In a trajectory analysis using data from a randomized controlled trial of standardized rehabilitation, 18 age, sex, sedation duration, and hospital length of stay (LOS) were associated with patterns of recovery using Short Physical Performance Battery scores across 6 months of follow-up. 39 Interestingly, critical illness-related factors such as severity of illness appear to have little or no impact on functional recovery9,36,39 Collectively, these data suggest that patient-related pre-existing factors (eg, older age, number of comorbidities, female sex) along with ICU practices (eg, sedation and time in the hospital) have a significant effect on functional recovery in the early months after hospitalization, but further study is needed.
Muscle Health and Recovery of Physical Function
Though a large body of research has studied ICU-AW as a primary driver of impaired physical function after critical illness, emerging data also highlight the importance of other factors such as exercise capacity, muscle power (ie, the ability to quickly generate muscle force), and muscle composition as important factors related to physical function. In a cohort of ARDS survivors, Needham and colleagues found that while impaired muscle strength improved to near- normal during 6 months of following, that 6MWD and self-reported physical function remained significantly below population norms. 40 Muscle strength accounted for a modest degree of variance in 6MWD, suggesting that other factors may effect physical performance. One of these factors may be the product of muscle force and velocity of muscle contraction, known as muscle power. 41 In one cohort study, lower muscle power was moderately to strongly related to ongoing impairments in physical function 1 month after hospital discharge. 42 Anatomic changes to muscle structure have also been studied. For example, fast-twitch myofibers are thought to be predominantly lost during the acute phase of critical illness.43–45 Fast-twitch myofibers (type IIa and IIx) generate more power than slow-twitch (ie, type I myofibers). Loss of these myofibers may lead to reductions in muscle power in those with critical illness, affecting tasks such as standing up from a chair where velocity is needed to quickly overcome gravity to achieve an upright position or achieving a faster walking speed. Given the modest discordance between commonly measures of muscle health (ie, strength) and physical function outcomes, further translational and mechanistic understanding of physical function is needed so that interventions targeting these underlying mechanisms can be used to develop and test interventions to improve recovery.
Potential Biological Mechanisms Limiting Muscle and Physical Recovery
Acute Muscle Wasting in the ICU
Muscle wasting (atrophy) occurs rapidly during critical illness with estimates of 20%–30% reduction in quadriceps size in the first 10 days, which is strongly associated with impaired physical function at hospital discharge.4,21,46 Prior data has suggested that type I and type II muscle fibers are affected in critical illness, but fiber type distribution may not significantly change.4,5,47 Early muscle wasting is likely driven by increases in proteolysis with 43 or without reductions in protein synthesis, 48 shifting muscle protein homeostasis to catabolism. Proteolytic enzyme activity and mRNA expression support increases in ubiquitin-proteasome pathway and autophagy, both catabolic pathways, 49 while indices of protein synthesis, specifically signaling of mTOR were unchanged 50 in patients with septic shock. The nexus between protein synthesis and degradation in critical illness is somewhat controversial, but at minimum, sustained catabolism likely leads to acute muscle wasting. Several additional underlying molecular and cellular mechanisms may also contribute to skeletal muscle dysfunction in the ICU, including alterations in mitochondrial function and density with oxidative stress and bioenergetic failure.51,52 Fredriksson and colleagues demonstrated that mitochondrial content in skeletal muscle was significantly reduced in patients with sepsis-induced multi-organ failure. 53 Additionally, Puthucheary et al demonstrated that mitochondrial biogenesis and lipid oxidation were impaired in mechanically ventilated ICU patients, and these bioenergetic derangements correlated with greater intramuscular inflammation and impaired anabolic signaling. 51 Iatrogenic effects from clinical practices, such as immobilization, have also been shown to reduce mitochondrial function and possibly microvascular function,54,55 which could further contribute to acute muscle wasting in the ICU. Lastly, in pre-clinical models, administration of nutrition (particularly proteins and amino acids) during critical illness may be impair autophagy in skeletal muscle, 56 and greater protein delivery has associated with increased muscle loss in critically ill patients, 4 potentially implicating nutritional factors as contributors to acute ICU muscle wasting. For further detailed review of the mechanisms of acute muscle wasting in the ICU, there are several clinical and biological reviews of ICU-acquired weakness and muscle wasting during critical illness.22,57–60
Potential Mechanisms of Post-ICU Muscle Impairment
Despite the high prevalence of impaired physical function in survivors of critical illness described above, the cellular and pathophysiological mechanisms driving persistent muscle dysfunction in patients surviving critical illness, regardless of etiology, remain unclear. Elucidating the mechanisms driving neuromuscular and physical dysfunction in the recovery phase is imperative for the development of rehabilitative, nutritional, and pharmacologic strategies to improve long-term outcomes. To date, however, there have been few studies that have examined mechanisms of muscle function through the use of muscle biopsies after critical illness.5,61
Data from ICU survivors demonstrate type-II muscle fiber atrophy, 45 reduced satellite (stem) cell content, 5 increased collagen deposition and altered mitochondrial biogenesis genes 61 ; mechanisms of skeletal muscle recovery depicted in Figure 2. Dos Santos et al, collected muscle biopsies from eight ICU survivors with heterogenous clinical characteristics and reported that those with low muscle mass and low muscle strength that persisted six months after hospitalization had decreased satellite cell content compared to those who regained muscle mass. 5 Likewise, in animal models of critical illness, including cecal ligation and puncture mouse models of sepsis, muscle atrophy with poor regeneration is driven by impaired satellite cells. 62 The restoration of muscle mass in humans, however, does not necessarily equate to better strength and function as patients with normalized size may have reduced muscle force production. 5 Together, these early data from human and animal studies suggest a potentially important role for satellite cells (muscle stem cells) in regenerating muscle function in survivors of critical illness.
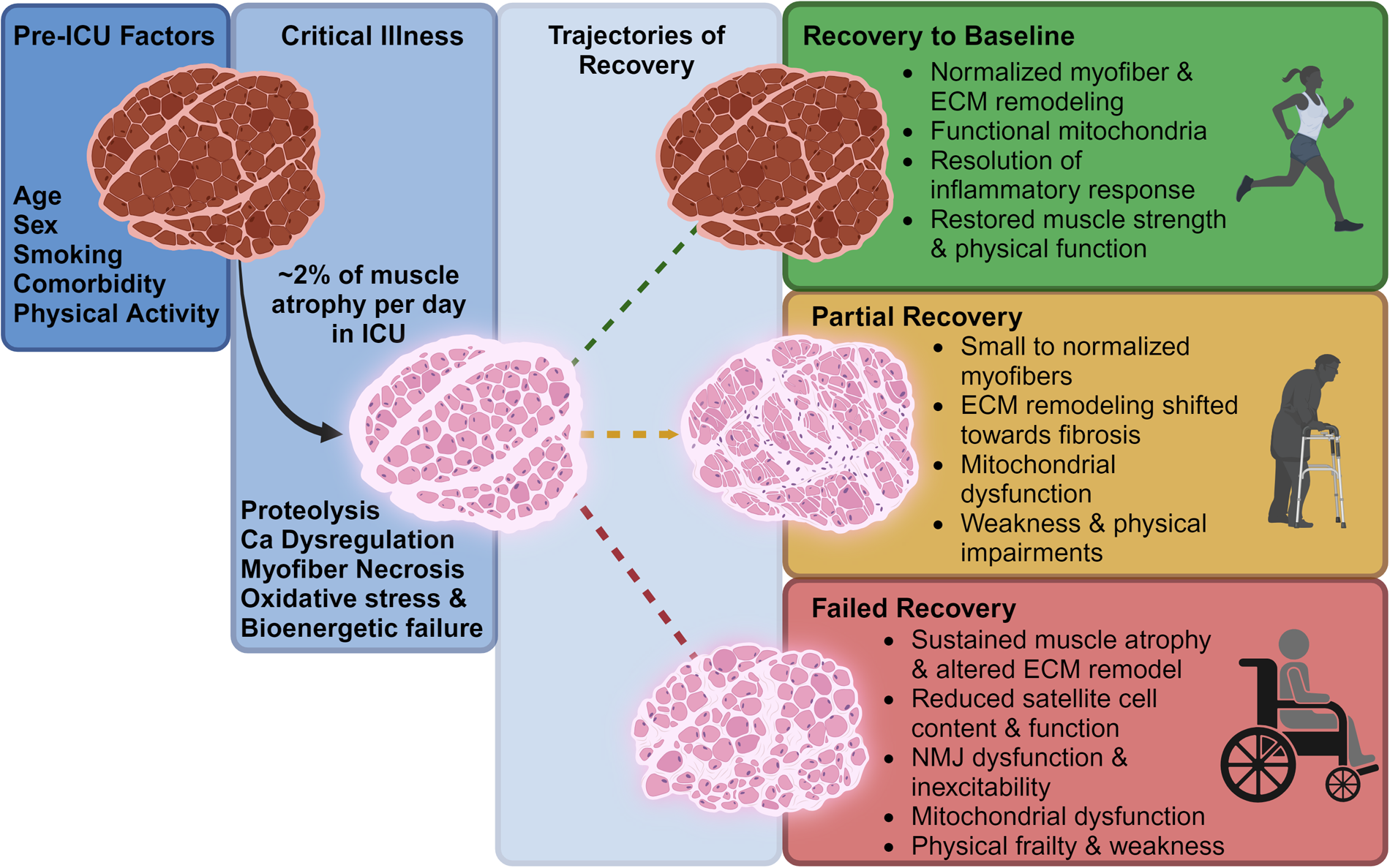
Risk factors and possible mechanisms impacting muscle and physical recovery after critical illness.
Data in human biopsies suggest that persistent weakness 6 months after hospital discharge was not explained or mediated by UPS-mediated proteolysis, muscle autophagy, or changes in mitochondrial content. 5 A secondary analysis of muscle biopsies from the Dos Santos study revealed downregulation of mitochondrial genes and increased collagen deposition, 61 which may help explain the incongruence between mass and function.
Persistent Inflammation and Bioenergetic Failure
The recovery of muscle health in survivors of critical illness is a more complex process than simply regaining muscle mass lost during an ICU admission. Rather, it represents a highly orchestrated initiation and resolution of inflammation within muscle tissue. Dysregulated inflammatory responses within muscle likely leads to significantly impaired energy metabolism (ie, “bioenergetic failure”) and mitochondrial dysfunction63,64 in addition to altered protein metabolism dynamics and extracellular matrix (ECM) remodeling.65,66 This imbalance between inflammation, bioenergetic dysfunction, and muscle regeneration is most likely altered significantly in ICU survivors where persistent inflammation may occur67–69 leading to poor muscle regeneration, though to date tissue-level data in survivors are limited with mixed evidence on the association between inflammation and muscle recovery after critical illness. 5
Data from human studies also indicate that mitochondrial dysfunction and increased oxidative damage occurs during critical illness due to endocrine downregulation and tissue-level hypoxia (Figure 3).63,64 This damage is compounded by additional insults known to worsen mitochondrial capacity in skeletal muscle, such as immobility,54,70 as well as other theorized iatrogenic insults to mitochondria such as bactericidal antibiotics, sedatives (eg, propofol), and neuromuscular blockers.71,72 Each component of this web of deranged physiology during critical illness leads to reduced mitochondrial oxidative capacity and bioenergetic failure, manifesting as multi-organ dysfunction. 73 Along this path, patients who survive and recover have evidence of mitochondrial biogenesis (ie, synthesis of new mitochondria) and improvement in function, while those who fail to recover do not.74,75 Animal models, indeed, confirm the sustained mitochondrial dysfunction even with restoration of muscle size. 76 We postulate that mitochondrial impairment in skeletal muscle likely persists after the resolution of the acute illness leading to persistent muscle dysfunction. 61 Nevertheless, the extent to which mitochondrial biogenesis occurs in survivors who experience a “full” functional recovery and to what extent this occurs by organ system remains unclear.
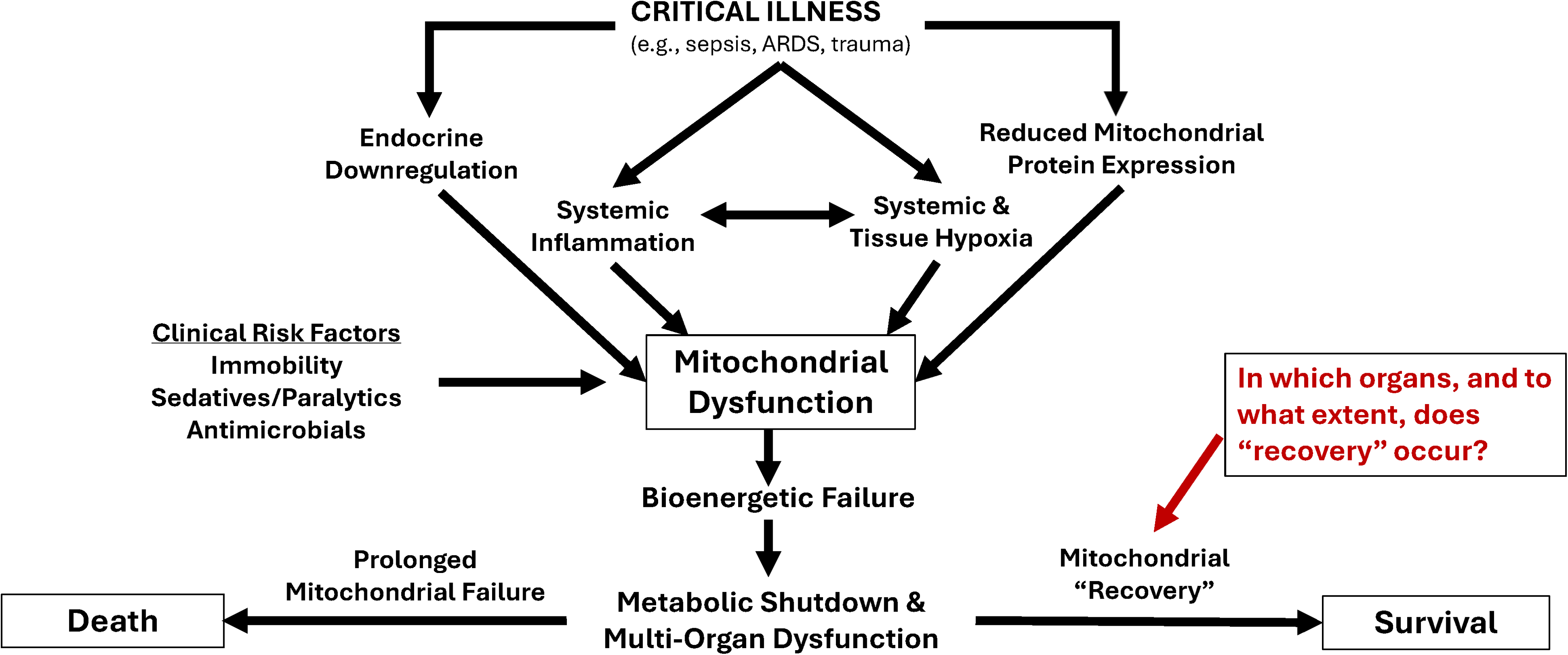
Potential pathophysiology and clinical risk factors for mitochondrial dysfunction and recovery with critical illness.
Persistent metabolic dysfunction leading to bioenergetic failure may also be a significant contributor to poor physical performance among survivor of critical illness as suggested by muscle gene expression 61 and indirect measures such as cardiopulmonary exercise testing as well as in survivors of critical COVID-19.61,77,78 Among ICU survivors with severe COVID-19, lower muscle fiber oxidative capacity (measured by succinyl dehydrogenase activity) was associated with worse six-minute walk distance and greater self-reported fatigue. 78 Future research in human studies, as well as animal models, is urgently needed to better characterize these potential mechanisms and their associations with clinical risk factors and outcomes to allow for the development of targeted therapeutics.
Clinical Risk Factors for Poor Muscle Function and Physical Recovery After the ICU
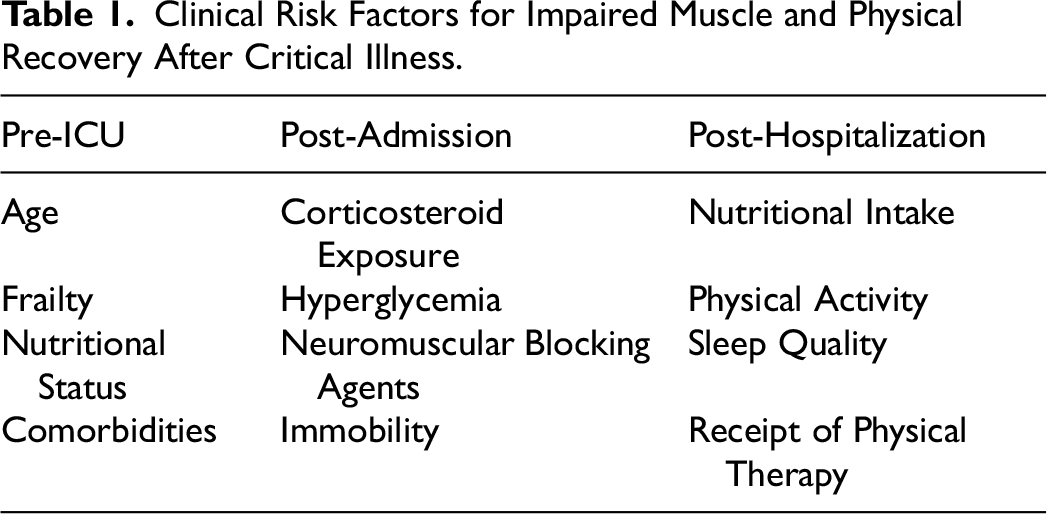
Poor muscle function and physical recovery after the ICU is hypothesized to develop in relation to a patient's underlying vulnerability to developing muscle dysfunction, the intensity of that critical illness and their underlying resilience/recovery mechanisms.79–81 In prior sections, we have described the underlying biological mechanisms hypothesized to lead to poor muscle function in survivors of critical illness. We now turn our focus to the clinical factors associated with physical dysfunction after critical illness. Together, these factors can be categorized as those related to “pre-ICU” factors (eg, age, comorbidities, frailty), “critical illness related” factors (eg, severity of illness, treatment with mechanical ventilation, deep sedation, bedrest), and post-hospitalization factors (eg, nutrition and receipt of post-hospital physical therapy) (Table 1).
Clinical Risk Factors for Impaired Muscle and Physical Recovery After Critical Illness.
Age
The incidence of critical illness increases with age. Those adults over the age of 65 comprise two thirds of intensive care unit bed days annually.82–84 In addition to the large number of older adults who become critically ill, the prevalence of sarcopenia (ie, low muscle mass) and dynapenia (ie, low muscle strength) increases with age.85–87 These age-related changes to muscle mass and function relate to increased adipose infiltration and muscle fiber necrosis resulting in fewer numbers of fibers including fewer type-IIfibers (fast-twitch fibers).88–91
Data related to age as a risk factor for the development of ICU-AW are mixed, with some studies showing an association, whereas others do not. A recent review of reviews found three systematic reviews and meta-analyses that studied age as a risk factor for ICU-AW.[Cite PMID: 39232808] Two meta-analyses showed positive associations—that older age was associated with greater risk of ICU-AW (OR 6.33; 95%CI 5.05-7.61,
Frailty
Aging results from an imbalance of damage due to environmental stressors and underlying biological repair mechanisms. With time, the homeostatic mechanisms of maintenance and repair begin to fail, leading to increased vulnerability to poor outcomes when faced with a physiologic stressor. Collectively this phenomenon is understood as the syndrome frailty.94–97 Over the past decade, the effect of pre-existing frailty on those who develop critical illness has begun to be explored.
In a study of the Precipitating Events Project, pre-existing frailty, as measured using the frailty phenotype, was associated with 41% greater disability in activities of daily living, admission to a nursing home, and death in the 6 months after a critical illness.
98
Baldwin and colleagues measured the frailty phenotype at hospital discharge in a cohort of older adults and found frailty to be associated with greater disability and mortality.
99
A large prospective study of 1040 those with medical and surgical critical illness showed that higher Clinical Frailty Scale scores were independently associated with higher mortality (
Nutritional Status in and After the ICU
Critical illness is associated with significant catabolism, protein turnover, and subsequent muscle wasting, and one previously postulated strategy to mitigate this process is through targeted nutritional provision during critical illness including increased protein intake. In one systematic review of randomized trials, higher daily protein delivery was not associated with mortality or functional outcomes, but in 5 studies that included muscle outcomes, higher protein delivery was associated with attenuated muscle loss. 100 However, increased energy and protein delivery has been not been consistently associated with reductions in muscle wasting, 101 with one study actually demonstrating a negative correlation between muscle wasting and increased protein delivery. 4 Notably, the PRECISe randomized trial, which included 935 critically ill adults, found that higher enterally delivered protein intake was associated with worse quality of life at 180 days after randomization, 102 while the EFFORT trial found a potential signal for increased mortality in mechanically ventilated patients with acute kidney injury and greater severity of illness at presentation who received greater protein delivery. 103 The potential adverse effects seen with greater protein intake in recent trials may be due to significant anabolic resistance that can’t be overcome by delivery of additional protein sources. 48
Prior critical care literature examining nutrition in critically ill patients has not specifically studied those with poor baseline nutrition at ICU admission.104–108 A nested, prospective, multicenter cohort study assessed the relationship of nutritional risk at ICU admission with disability, cognitive outcomes, and health-related quality of life outcomes and found that higher scores in the modified Nutrition Risk in Critically Ill (mNUTRIC) score, indicating greater nutritional risk, were associated with discharge to a facility (eg, inpatient rehabilitation or skilled nursing facility) with a trend towards disability 3 months after critical illness in survivors of critical illness. 109 Additionally, following an ICU admission, data suggest that 20% of survivors report decreased food intake after the ICU due to persistent symptoms and 10% report access to healthy food as barriers to recovery in the first month after hospital discharge, which may significantly influence muscle and physical function recovery. 110 Future studies are needed to better characterize how both baseline nutritional status and nutritional intake after the ICU impact long-term outcomes.
Corticosteroids
Corticosteroids are frequently administered in the ICU to treat common critical illness syndromes including septic shock, pneumonia, ARDS, and COVID-19.111–113 Though corticosteroids improve mortality, ICU length of stay, and duration of mechanical ventilation in those with critical illness, data from populations without critical illness show these drugs are strongly associated with muscle weakness. Thus, treatment corticosteroids may place survivors of critical illness at risk for developing ICU-AW and prolonged muscle failure.111–114 In contrast, because they may shorten the duration of organ failure, corticosteroids could also potentially reduce ICU-AW by shortening duration of critical illness.
Over the past 20 years, studies have sought to understand the relationship between corticosteroids and ICU-AW. This body of literature has now been analyzed in three systematic review and meta-analyses.114–116 The first of these studies included 1 randomized controlled trial (RCT) and 17 prospective cohort studies that enrolled a combined 2387 participants. 115 Overall, this analysis found that corticosteroids were associated with an 84% increase in the odds of developing ICU-AW (OR 1.84, 95%CI 1.26 to 2.67). In pre-specified subgroup analyses, the authors found that how ICU-AW was detected affected this association such that studies using the MRC scale found greater odds of developing ICU-AW (OR 2.06, 95%CI 1.27 to 3.33) whereas studies that used electrophysiologic testing found an imprecise association (OR 1.65, 95%CI 0.92 to 2.95). Interestingly, studies that enrolled those with sepsis found no association between corticosteroids and ICU-AW (OR 1.96, 95%CI 0.61-6.30). In contrast, studies that enrolled those treated with mechanical ventilation found that those who received corticosteroids were twice as likely to develop ICU-AW as those who did not (OR 2.0, 95%CI 1.23 to 3.27).
A second meta-analysis, conducted as part of a larger analysis of the effects of corticosteroids in sepsis, included 7 trials included that enrolled a total of 6178 participants. 114 There were In this meta-analysis,. being treated with corticosteroids was associated with a 21% increase in the relative risk of neuromuscular weakness (RR 1.21, 95% CI 1.01-1.45). The authors, however, cautioned that the certainty of evidence around this finding was low due to imprecision and the different types of corticosteroids studied.
Finally, a third systematic review and meta-analysis included 8 studies that examined the association between corticosteroids and ICU-AW among 888 participants. 116 In contrast to the two prior studies, this meta-analysis found no significant increase in the odds of developing ICU-AW among those exposed to steroids (OR 1.54, 95% CI 0.77-3.09).
Together, these data suggest that corticosteroids may be associated with the development of ICUAW, though the heterogeneity of these studies (eg, out of 32 studies, only 4 were included in more than one meta-analysis), the exposure to steroids (eg, type, dose, and duration) and methods used to measure ICU-AW (eg, MRC and electrophysiologic studies) reduces the certainty of this conclusion. Further work is needed to understand the effects of corticosteroids in specific patient populations such as those with sepsis and those treated with mechanical ventilation. In the meantime, the extant data suggest that the potential harms associated with corticosteroids on muscle strength should be weighed against the benefits on mortality and other outcomes (eg, ICU LOS) such that clinicians should seek to minimize corticosteroid exposure (eg, dose and duration) whenever possible.
Hyperglycemia
One mechanism by which steroids may lead to ICU-AW is through causing hyperglycemia. The harmful effects of hyperglycemia are illustrated by a systematic review and meta-analysis that reported data from 3 studies and found hyperglycemia was associated with a near tripling of the odds of developing ICU-AW (2.95, 95% CI 1.70-5.11). The effect of hyperglycemia on ICU-AW was also studied as a secondary outcome in two randomized trials comparing intensive insulin therapy to conventional insulin therapy.117,118 The first study randomized 1548 surgical intensive care unit patients treated with mechanical ventilation to intensive insulin therapy (ie, targeting glucose levels between 80-110 mg/dL) or to conventional insulin therapy (ie, targeting glucose levels between 180-200 mg/dL). 119 On day seven, there were 405 participants who were still in the ICU and underwent electrophysiologic testing. Those treated with intensive insulin therapy had a lower incidence of ICU-AW (13.8%) as compared to the conventional treatment group (21.8%). A second study of intensive insulin treatment in medical ICU patients of among medical ICU patients who remained in the ICU for 7 or more days found those managed with intensive insulin therapy likewise had an incidence of ICU-AW of 38.9% compared to 50.5% in the conventional insulin therapy group.117,120 A meta-analysis including data from these two studies found that intensive insulin therapy was associated with a 30% reduction in the risk of developing ICU-AW (Risk Ratio 0.7, 95%CI 0.6 to 0.82). 121 Thus, it may be the case that hyperglycemia serves as a modifiable risk factor for the development of ICU-AW.
A secondary analysis from a trial of early occupational and physical therapy compared the effects of this intervention along with intensive insulin therapy, titrated to achieve blood glucose levels between 80 and 12 mg/dL, on the incidence of ICU-AW.122,123 After adjusting for age and severity of illness, receiving early occupational and physical therapy was associated with an 82% reduction in the odds of developing ICU-AW (OR 0.18, 95% CI 0.06 to 0.55). Likewise, increasing doses of insulin was associated with a 99% decreased the odds of ICU-AW ((OR 0.011, [95% CI 4.6 × 10−6 to 0.2] per unit of insulin/kg/day). Interestingly, patients in the early occupational and physical therapy group required less insulin to maintain euglycemia, suggesting that these interventions act synergistically to reduce hyperglycemia and, in turn, ICU-AW. Thus, maintaining tighter glucose control through intensive insulin therapy in conjunction with higher levels of physical activity during critical illness may be a useful strategy to reduce the metabolic insult incurred by skeletal muscle due to critical illness and its treatment and could be an effective means by which to reduce downstream muscle dysfunction.
Neuromuscular Blockade
The impact that neuromuscular blocking agents (NMBA) have on post-ICU physical function remains uncertain.124,125 In a prospective cohort study of 95 patients enrolled from medical and surgical ICUs in France, De Jonghe and colleagues examined risk factors for ICU-AW and found no association between NMBA administration and ICU-AW.
26
Prior data, however, had suggested that the prolonged infusion >48 h of NMBA may be more impactful than shorter infusions.126–129 Two randomized trials of treatment with NMBAs versus placebo found similar findings. The ACURASYS trial compared NMBA to placebo in deeply sedated patients with ARDS and found no difference in the incidence of ICU acquired neuromuscular weakness at 28 days between the two groups (70.8% vs 67.5%,
A 2018 systematic review and meta-analysis that included 5 studies of risk factors for ICU-AW in mechanically ventilated patients (but which did not include the two randomized trials of NBMAs reported above) found a significant association between NMBA and ICU-acquired weakness (OR, 2.03; 95% CI, 1.22-3.40).
92
A second systematic review and meta-analysis that also studied risk factors for ICU-AW in those with mechanical ventilation, but that included 5 different studies from those included in the prior review found NMBA use was not associated with ICU-AW (OR, 1.43; 95% CI, 0.92-2.22;
Immobility
Bed rest is common in those with critical illness and is a well described risk factor for ICU-AW and increased muscle breakdown.27,132 Thus, clinicians should incorporate strategies to minimize immobility times of critically ill patients from the earliest days after ICU admission. The 2018 Pain, Agitation/Sedation, Delirium, Immobility, and Sleep disruption (PADIS) guidelines suggest performing rehabilitation or mobility as a means by which to reduce ICU-AW. 133 This suggestion was based on data from 6 randomized trials of rehabilitation and mobility that showed a clinically and statistically significant improvement in MRC scores (6.24 points [95% CI, 1.67-10.82]). Since publication of these guidelines, a large multinational, multicenter trial (TEAM trial) of 750 mechanically ventilated patients randomized patients to an enhanced early rehabilitation intervention or usual care and found no difference in the primary outcome number of days alive and out of the hospital at 180 days after randomization. 20 Likewise, there was no difference in self-reported physical function measures. A Bayesian meta-analysis of 15 RCTs found a 95% probability that early mobility interventions were associated with better self-reported physical function at 6 months post-hospitalization. 134 Together, data from over a decade and a half of studies support the use of rehabilitation and mobility interventions to reduce ICU-AW.
Despite the evidence suggesting the harms associated with bedrest and the ability of rehabilitation and mobility to reduce these harms, the number of patients across the globe (ie, Australia/New Zealand, 135 Germany, 136 Brazil, 137 Switzerland, 138 United Kingdom, 139 Saudi Arabia, 140 and USA 141 ) who achieve out of bed activities as part of routine care is approximately 20% of the total ICU stay. Moreover, actigraphy data show immobility times between 64% and 83%142,143 of the ICU stay in ventilated and non-ventilated patients; and between 96% and 98% in ventilated patients.144,145 Therefore, effective strategies to enhance uptake of rehabilitation and mobility protocols remains an important clinical and research challenge.
Post-Hospital Physical Activity, Rehabilitation, and Sleep
In addition to pre-critical illness and critical illness factors post-hospitalization factors including low physical activity, inadequate physical rehabilitation and poor sleep also affect the recovery of physical function in survivors of critical illness. Following hospital discharge, survivors of sepsis and septic shock spent approximately 90% of their waking hours sitting or lying down based on actigraphy data and over half spent less than 30 min per day walking. 10 A second cohort study of survivors of critical illness who had been treated with mechanical ventilation took around 1200 steps per day in week after hospital discharge, significantly below the 7000 to 10 000 steps per day recommended by public health experts. 146 Additionally, few patients are discharged to subacute or acute rehabilitation facilities following hospital discharge and most do not receive little outpatient rehabilitation.147,148 Lastly, survivors of critical illness report poor sleep quality despite spending over half of each day resting or sleeping. 149 Therefore, these modifiable factors may serve as attractive targets for intervention in both research studies and in clinical practice to improve long-term muscle and physical function outcomes (Table 1).
Prior Interventions
While our scientific understanding has advanced regarding the epidemiology, risks, and mechanisms of prolonged muscle and physical impairments following the ICU, effective interventions remain elusive. Both pharmacological and non-pharmacological interventions have been evaluated, though globally there is a dearth of strong evidence to support any one therapeutic approach.
Pharmacologic Therapies
A systematic review evaluated trials of drug therapies to treat loss of muscle strength in those with critical illness. 150 They identified 10 studies that evaluated therapies such as oxandrolone, propranolol, immunoglobulin, growth hormone, and glutamine. Few studies specifically evaluated the effect of these drugs on muscle strength, opting instead for secondary measures such as muscle mass, lean body mass, and muscle protein synthesis and degradation, and all studies were restricted to critically ill patients rather than survivors. Thus, data supporting the use of pharmacologic therapies to improve muscle strength are limited both in the ICU setting and after hospital discharge.
However, as described above in the discussion about hyperglycemia as a risk factor for ICU-AW, insulin therapy may be one option for preventing ICU-AW. Two studies evaluated insulin therapy were included in this meta-analysis, but only one examined ICU-AW as an outcome. This trial (N = 40 medical ICU patients) randomized trial compared treatment with insulin to maintain euglycemia (ie, 80-140 mg/dL) to usual care treatment (maintain glucose 180-200 mg/dL). The authors found that the incidence of critical illness polyneuropathy was 10% in the treatment group and 45% in the usual care group (
Non-Pharmacologic Therapies
The majority of studies of non-pharmacologic therapies to improve recovery and reduce ICU-AW have focused on prevention in the ICU setting. Three therapies, early mobility, leg cycle ergometry, and neuromuscular electrical muscle stimulation (NMES) are the most well-studied interventions. Though data are conflicting on the effects of early rehabilitation on long-term functional outcomes after the ICU, a clearer picture has emerged of the benefits of NMES and early rehabilitation on preventing ICU-AW during critical illness.18–20,123,152 A systematic review and meta-analysis of early rehabilitation included studies comparing either early mobility (eg, occupational and physical therapy, exercise) or NMES to usual care to determine effects on ICU-AW. Combined, these studies included 841 participants. Overall early rehabilitation with either early mobility or NMES was associated with a 29% reduction (OR 0.71, 95% CI 0.53 to 0.95,
A second systematic review by Garcia-Perez-de-Sevilla et al identified randomized controlled trials that compared interventions delivered in the ICU (ie, leg cycle ergometry) to usual care.
153
Two of the included studies evaluated muscle strength. In the first study, those randomized to leg cycle ergometry showed significant improvements in lower extremity strength measured using dynamometry as compared to usual care (
Finally, two additional meta-analyses explored the effects of NEMS versus usual care on ICU-AW.156,157 Both studies included five randomized trials, though only two were included in both analyses. The first found no differences in muscle strength between those treated with NMES and those treated with usual care (mean difference in MRC score 0.45, 95% CI −2.89 to 3.80). 156 The second, however, found that participants treated with NMES had a 1.78-point (95%CI 0.44 to 3.12) better MRC score than those assigned to the usual care group. 157
Collectively, these studies suggest that early mobility interventions (eg, occupational therapy, physical therapy, cycle ergometry) that are started within 72 h of ICU admission, may be effective in preventing the development of ICU-AW and may reduce muscle loss. When compared to the trial data on early rehabilitation interventions and longer-term functional outcomes, however, it is unclear if ICU-focused interventions that attempt to prevent ICU-AW translate to improved long-term physical function for survivors.
After hospitalization, there are fewer data on non-pharmacologic interventions to reduce ICU-AW and improve physical function. Studies have predominantly focused on approaches to exercise rehabilitation. These studies have consisted of two types of interventions: (1) Home-based, self-directed physical rehabilitation and (2) Short, (ie, ≤8-week) center-based interventions. Home-based interventions have been tested in 4 studies, each provided survivors of critical illness with instructional materials about exercise, with varying degrees of supervision ranging from no contact to twice weekly in-home sessions for 8 weeks.158–161 One study showed a statistically, but not clinically, improved HRQOL in the intervention group. None of these studies showed improved muscle strength.
Center-based exercise interventions have been tested in 4 studies.162–165 These studies consisted of 2-3x weekly supervised exercise sessions with an additional in-home exercise session. Each of these interventions, however, lasted <8 weeks. Exercise capacity, measured by either 6 min walk or cardiopulmonary exercise testing, improved in two studies, suggesting that intensive, center-based interventions are feasible, and may improve physical function.164,165 Neither of these studies, however, measured muscle strength. Two studies measured outcomes several months after the center-based interventions had ended. Both showed that the improvements made during the intervention did not persist over the long-term, suggesting that few survivors of critical illness continue to exercise following cessation of the formal intervention period. Thus, while studies of post-ICU exercise rehabilitation show this type of intervention is feasible and may improve mobility and physical function, these studies are limited by short duration of the interventions, limited intensity, and have not shown sustained benefits.
Pathways Forward and Future Opportunities
Given the complex nature of physical recovery after critical illness, it's clear that multiple pathways are needed to improve outcomes spanning the continuum of care from the ICU to the months and years after hospitalization. These pathways range ongoing research in the ICU on optimal strategies to prevent the development of ICU-AW including best practices for early mobilization approaches and nutritional strategies, further work to identify mechanisms of muscle dysfunction that can be targeted by pharmacologic therapy or rehabilitation approaches both during a critical illness and afterwards, and evaluating care models that may be adapted from other specialties (eg, cardiology, etc) to better serve patients as they transition from the hospital to home.
Targeting Mechanisms Through Nutrition, Drug Therapy, and Rehabilitation
Given data that support use of combined rehabilitation and nutrition interventions to improve age-related loss of physical function, further research is urgently needed to better understand molecular mechanisms at play in critical illness recovery so that targeted therapies to mitigate muscle damage in the ICU and speed recovery after hospitalization can be developed. Despite enthusiasm for providing higher amounts of protein in the ICU to preserve muscle mass, there are contradictory findings and even potentially increased muscle wasting with greater protein delivery that necessitate further investigation. 4 Drug therapies, a novel but understudied approach, targeting implicated molecular mechanisms such as the mTOR pathway and deranged autophagy, have also been proposed and deserve further investigation. 59 As described above, an important mechanism that spans the continuum of critical illness and survivorship is muscle mitochondrial dysfunction and bioenergetic impairment that leads to poor muscle recovery, exercise intolerance, and persistent fatigue among survivors. Identification and testing of drug therapies that target mechanisms of mitochondrial dysfunction including oxidative phosphorylation in skeletal muscle and other organ systems could also help speed recovery. Ultimately, the most effective interventions are likely to utilize a combination of pharmaceutical, nutritional, and rehabilitation approaches which affect multiple components of physical health.
Improving Care After the Hospital
After an ICU hospitalization, the transition from hospital to home in the early recovery and post-discharge phase is a vulnerable period for survivors. Unfortunately, care during this period is often fragmented and does not focus optimal means by which to improve physical function. One important limitation is the lack of consensus regarding standardized assessments of physical function from hospitalization to follow-up so that trajectories of recovery can be measured. Bakhru and colleagues demonstrated that measuring muscle strength, physical function, and patient-reported function was not only feasible but also was predictive of outcomes including disability, readmission, and mortality. 166 Thus, well-validated performance-based measures of physical function such as the Short Physical Performance Battery and handgrip strength can be measured in the hospital and tracked longitudinally to identify track patients’ recovery and identify those who are not progressing or are declining. 39 These data could easily be incorporated into follow-up care pathways such as ICU recovery clinics, primary care offices, and rehabilitation clinics to inform care and prompt clinicians to provide earlier interventions to improve function. Similarly, standardized rehabilitation pathways could be developed for ICU survivors similar to clinical populations with heart disease. For example, patients requiring coronary revascularization or heart failure have well defined cardiac rehabilitation pathways that are shown to improve outcomes, are strongly recommended by professional cardiology and rehabilitation societies, and are covered by insurance with.167,168 While the National Institute for Health and Care Excellence in the United Kingdom issued recommendations for ICU survivors that included post-ICU rehabilitation in 2009, no standardized care pathways have been broadly implemented and many patients continue to receive limited rehabilitation services after ICU discharge.148,169 Dedicated rehabilitation after hospitalization for critical illness may improve the recovery trajectory for survivors, including improving self-efficacy for exercise, which is associated with engagement in sustained exercise among previously hospitalized older adults. 170 Similarly, studies of nutritional interventions the hospital in the early recovery phase are needed, and dedicated nutritional evaluation by registered dieticians and other nutrition professionals could be an important part of follow-up care for ICU survivors. Therefore, we advocate for the development of standardized assessments and care pathways to improve physical function after the ICU that incorporates multiple specialties, including primary care, physical and occupational therapy, behavioral science, exercise rehabilitation, geriatrics, gerontology, social work, and physical medicine and rehabilitation, and post-ICU clinics. The cost-effectiveness of these interventions should also be studied to align financial investment with clinical outcomes.
Conclusions
While improvements in ICU care have dramatically improved survivorship following critical illness, many survivors experience ongoing impairments in muscle health and physical function. We now have a growing understanding of potential mechanisms and modifiable risk factors that contribute to the development of ICU-AW and long-term physical impairments after the ICU. Nevertheless, much work remains to determine if improving these modifiable risk factors can improve long-term outcomes. Current evidence is mixed with regard to the benefit of early mobility and rehabilitation in the ICU and post-ICU rehabilitation. Novel pharmacologic, nutritional, and rehabilitation interventions that target the underlying mechanisms and risks for poor physical recovery after the ICU, along with novel care pathways focused on improving muscle and physical health, are urgently needed to improve the lives of survivors of critical illness.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was supported in part by the Department of Veterans Affairs Office of Research & Development under award IK2RX004799-01A1, the National Institutes of Health under awards KL2TR002245, K23AR079583, K76AG054864, and R03AG083556, and the Department of Veterans Affairs Tennessee Valley Healthcare System Geriatric Research, Education and Clinical Center (GRECC). The funding sources had no role in the preparation, review, or approval of the manuscript or the decision to submit the manuscript for publication. The contents of this manuscript are solely the responsibility of the authors and do not necessarily represent those of the United States Department of Veterans Affairs, the National Institutes of Health, Vanderbilt University Medical Center, the University of Kentucky, Clínica Alemana Universidad del Desarrollo, or The Ohio State University.
