Abstract
Background
Muscle ultrasound represents a promising approach to aid diagnoses of neuromuscular diseases in critically ill patients. Unfortunately, standardization of ultrasound measurements in clinical research is lacking, making direct comparisons between studies difficult. Protocols are required to assess qualitative muscle changes during an ICU stay in patients at high risk for the development of neuromuscular acquired weakness (ICUAW).
Methods
We conducted a retrospective, observational analysis comprised of three prospective observational studies with the aim of diagnosing muscle changes by ultrasound measurement of the quadriceps muscle. Different protocols were used in each of the three studies. In total, 62 surgical, neurocritical care and trauma intensive care patients were serially assessed by different ultrasound protocols during the first week of critical illness. The relative change in ultrasound measurements was calculated for all possible locations, methods and sides. Comparison was obtained using mixed effect models with the location, the height and the side as influencing variables and patients as fixed effect. The relationship between variables and outcomes was assessed by multivariable regression analysis.
Results
Ultrasound methods and measurement sites of the quadriceps muscles from all protocols were equally effective in detecting muscle changes. During the first week of an ICU stay, two groups were identified: patients with decreased muscle mass on ultrasound (n = 42) and a cohort with enlargement (n = 23). Hospital mortality was significantly increased in the cohort with muscle swelling (8 (19%) versus 12 (52%),
Conclusions
Different approaches of ultrasound measurement during critical-illness are equally able to detect muscle changes. While some patients have a decrease in muscle mass, others show swelling, which may result in a reduced probability of surviving the hospital stay. Causative reasons for these results still remain unclear.
Introduction
Survivors of critical illness frequently experience limitations in activities of daily living, decline in cognitive function and therefore reduced quality of life well beyond hospital discharge.1,2 Here, factors such as immobilization, inflammation, and oxidative stress lead to a state of muscular atrophy dominated by increased protein breakdown and decreased protein synthesis. 3 This can contribute to the development intensive care unit- acquired weakness (ICUAW), 4 which is associated with prolonged mechanical ventilation, 5 longer hospital stays and increased mortality. 6
Different methods of assessing muscle mass in critically ill patients have been evaluated to monitor the development of ICUAW and consequently predict clinical outcomes. While there are many techniques to quantify skeletal muscle mass (such as bioelectrical impedance analysis (BIA), dual energy x-ray absorptiometry (DXA), computer tomography (CT), and magnetic resonance imaging (MRI)), not all modalities are feasible in a critically ill population.7,8 In addition, invasive muscle biopsy is increasingly performed to complement clinical diagnosis and understand the underlying factors of muscle-metabolism. 3 Ultrasound as a non-invasive tool to assess muscle status especially in the early course of critical illness has been discussed with growing interest. It represents a non-invasive and repeatable diagnostic tool, that is readily available at the bedside.9–11 Assessment of the quadriceps muscle has been used extensively to assess muscle wasting with ultrasound in the critically ill. 12 The rectus femoris muscle is often used to evaluate muscle cross-sectional area as a surrogate for whole-body skeletal muscle.13,14 Either the rectus femoris or quadriceps (sum of rectus femoris and vastus intermedius muscle thickness) is frequently measured for muscle thickness. Both CSA and thickness measurements have been shown to decline early during the course of critical illness and have been inconsistently associated with impairment of strength and physical function.15,16 Unfortunately, standardization of ultrasound measuring protocols in clinical research is lacking, making direct comparisons between studies difficult. 17 Several factors (positioning of the patient, the ultrasound probe including the angle, the height of the measurement in relation to the patella, the amount of compression applied, as well as the image evaluation procedure) influence reproducibility of ultrasound data. 18 It remains unclear which of these factors has the strongest impact on the prediction of muscle mass and can therefore be considered as a monitoring parameter.
The aim of this retrospective study was to investigate the method of measurement using ultrasound that best correlates with patient condition and predicts the development of muscle change. We hypothesized, that use of one ultrasound method and serial repetition is superior in the evaluation of muscle mass and the prediction of ICUAW, to a single measurement.
Methods and Materials
Study Design, Setting and Participants
This study is a secondary analysis of ultrasound assessments of 3 observational studies of two interdisciplinary surgical intensive care units of the Department of Anaesthesiology and Intensive Care at Klinikum rechts der Isar, School of Medicine, Technical University of Munich, Germany between April 2017 and May 2019. The data is comprised of three study populations with serial ultrasound assessments: trauma patients included in the Miracle II Study (Metabolomics pilot study on intensive care acquired muscle weakness) NCT03819959, stroke patients from the NICU study (Effects of mobility dose on discharge disposition in critically ill stroke patients, NCT03347656) and general surgical ICU patients from the SICU-SOMS2 study (Effects of mobility dose in surgical intensive care unit patients on muscle wasting and adverse outcomes, NCT01363102). This secondary analysis was approved by the Ethics Committee of the Faculty of Medicine, Technical University of Munich (Grillparzer Str. 16, 81675 Munich, Chairperson Prof. Dr G. Schmidt, Reference150/22-S). For all patients, ultrasound measurements were performed within 48 h of admission to the ICU and were repeated weekly or within one day of ICU discharge.
The MIRACLE II, NICU and SICU-SOMS studies
All patients analyzed from the Miracle II Study had experienced trauma in the 24 h prior to inclusion, had a pre-operative physical function score of ASA I or II prior to admission (healthy or mild systemic disease), and were expected to remain in the ICU for no less than three days; patients were excluded from the study, if CPR was performed following trauma and prior to admission or, if treatment was deescalated to comfort care. Patients analyzed from the NICU study all experienced either an ischemic stroke or non-traumatic intracerebral hemorrhage, had been admitted to the ICU no longer than 48 h prior to inclusion, and had functional independence exhibited by a Barthel Index >70 two weeks prior to hospital admission; patients were excluded if transferred from another institution (other hospital, rehabilitation center, skilled nursing facility, etc), had exclusive or clinically dominant posterior circulation stroke, subarachnoid hemorrhage, subdural or epidural hemorrhage, absence of lower limbs, or if treatment was deescalated to comfort care. Lastly, patients analyzed from the SICU-SOMS study were all functionally independent with a Barthel Index >70 prior to hospital admission, had been admitted to the ICU no longer than 48 h prior to inclusion, and had an expected stay of at least three days; patients were excluded if transferred from another institution, had amputated lower limbs, were pregnant, had a shift in treatment strategy to comfort care, showed high risk for persistent brain injury (GCS <5), ischemic stroke, or non-traumatic intracranial hemorrhage. Age requirement for all three study populations was 18 years or over. Informed consent was obtained from patients directly, and if not possible, via direct proxy. Please find the Consort Diagram in the Supplement (Table S1).
Data Collection
Data included upon admission were basic demographics, location prior to ICU admission, admission category, admission diagnosis (eg sepsis or trauma upon admission) and several scores to characterize the cohort: baseline Glasgow Coma Scale (GCS), Charlson Comorbidity Index, 19 Sequential Organ Failure Assessment score (SOFA) 20 as well as standard laboratory and hemodynamic parameters. Upon ICU discharge, data was obtained regarding ICU length of stay and mortality, as well as data about ICU-related therapy (eg fluid administration, nutrition, dialysis and laboratory parameters). Upon hospital discharge, data on length of stay and discharge disposition (prior residence, nursing home, rehabilitation clinic, etc.) as well as mortality was obtained. This is a retrospective evaluation of the data collected in these studies.
Bedside Ultrasound
Quadriceps Muscle Ultrasound
Two common measurement techniques were used: muscle CSA and muscle thickness. Within 48 h of ICU admission, ultrasound image acquisition of the quadriceps muscle was performed using a 2–6 MHz curvilinear probe on the Sparq Ultrasound System (Philips, Bothell, WA, USA). Prior to assessment, patients were placed into the supine position with their upper body raised to a 30° angle and their legs relaxed and extended outright in bed. First, the cross-sectional area and thickness measuring sites were identified by marking 3/5 of the distance from the superior anterior iliac spine to the mid-upper border of the patella with a pen. 11 Except for the cross-sectional area measurement in all SICU-SOMS patients (2/3 distance), cross-sectional area and thickness were all taken at 3/5 distance. 21 After applying ultrasound gel, the probe was placed at a 90° angle perpendicular to the longitudinal axis of the thigh. Two measurement techniques, one to acquire the cross-sectional area of the rectus femoris muscle (cm2), and the other to obtain the thickness of the rectus femoris and quadriceps muscle (rectus femoris and vastus intermedius in cm), were performed to yield three values per leg. For the SICU-SOMS cohort, the quadriceps muscle of only one leg was measured. The measurement of the cross-sectional area was acquired by applying minimal pressure with the probe, with images ideally capturing a centered muscle with visualization of the femur bone. For muscle thickness, as described previously, maximum compression was applied with the probe to capture images. 18 All images were saved and exported to a USB device for later assessment.
Image Assessment
All Images were imported into HOROS, a free, open-source medical image viewing software (LPGL-3.0; Lesser General Public License, Version 3.3.6). Prior to measurement, quality control of the images was performed to ensure, the muscle was centered and that the left and right borders as well as the hyperechoic fascia encircling the rectus femoris muscle were visible. Similarly, quality of the muscle thickness image was assessed, ensuring the rectus femoris and vastus intermedius muscles were centered underneath the probe and that the muscle was not squeezed to one side during maximum compression. Images, that did not meet these criteria, were excluded from assessment, blinded to outcome data. First, with the use of a free hand tracing tool, the cross-sectional area was marked along the hyperechoic fascia of the rectus femoris muscle (cm2) (Supplement Figure 1a). Next, the thickness of the rectus femoris and quadriceps muscle (rectus femoris and vastus intermedius in cm) was measured on the maximally compressed images along the shortest imaginary line from the exact top-center of the image down to the muscle-bone interface 18 (Supplement Figure 1b). Thus, one distance was measured along this imaginary line (rectus femoris), before a second line was placed over the quadriceps, to yield two thickness values. 22 Please find a supplementary video of the ultrasound methodology (Supplement Media 1) as well as two supplementary figures showing the trajectory of patients over the course of the first seven days on ICU (Supplement Figure 2a and b).
Statistical Methods
Analyses were conducted using R (Version 4.1.0., Foundation for Statistical Computing, Vienna, Austria). Continuous variables are represented as median with interquartile range, discrete values as absolute and relative numbers. A
Results
Between April 2017 and May 2019, we included 65 patients with serial ultrasound assessment comprised of three study populations (Table 1). The median age was 65 [52-77] years, of which 41 patients (63%) were male. Further patient characteristics are presented in Table 1. Please find the Consort Diagram in the Supplement (Table S1).
Baseline Characteristics.
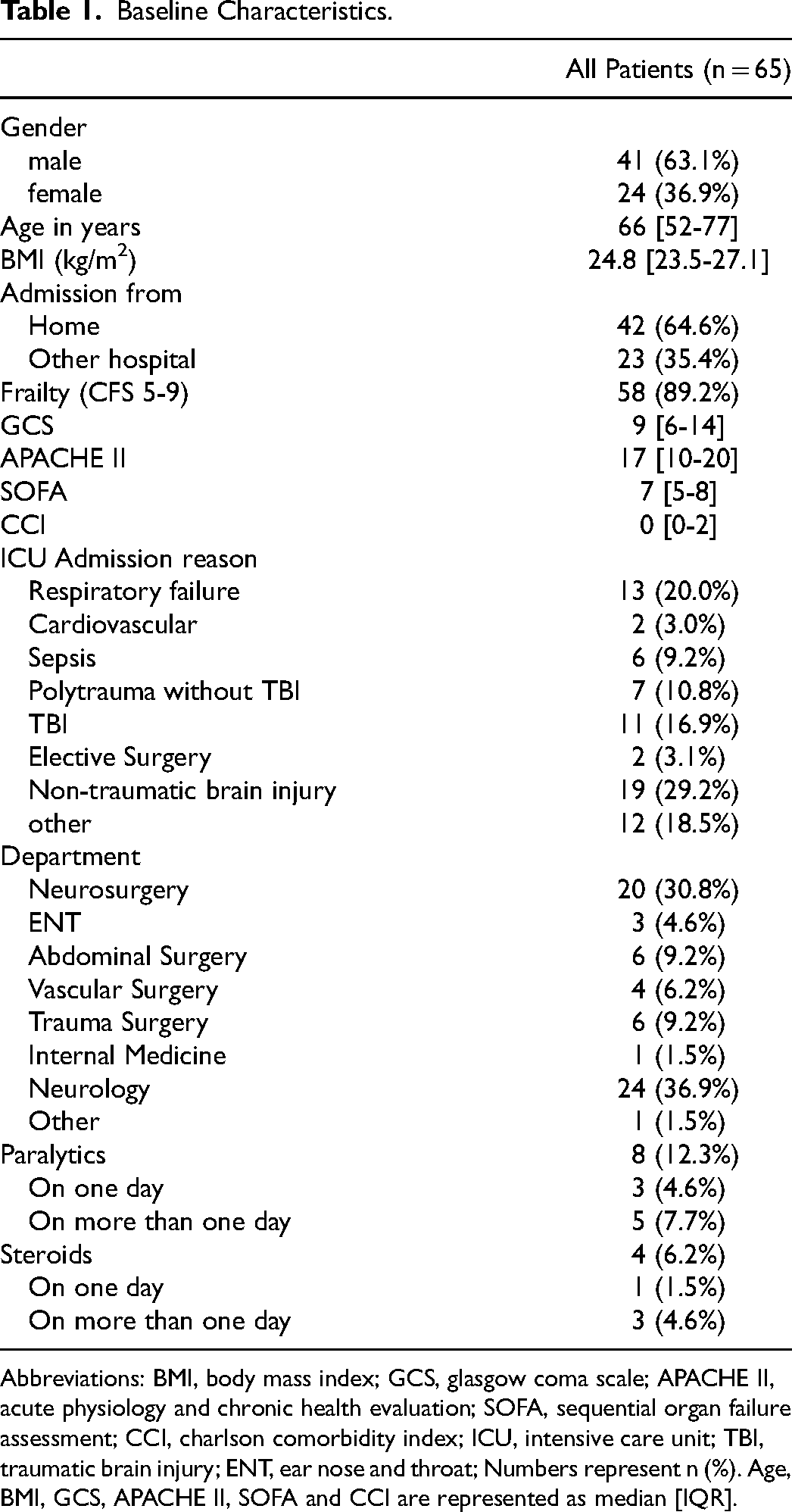
Abbreviations: BMI, body mass index; GCS, glasgow coma scale; APACHE II, acute physiology and chronic health evaluation; SOFA, sequential organ failure assessment; CCI, charlson comorbidity index; ICU, intensive care unit; TBI, traumatic brain injury; ENT, ear nose and throat; Numbers represent n (%). Age, BMI, GCS, APACHE II, SOFA and CCI are represented as median [IQR].
Primary Outcome
Table 2 shows the characteristics of the different measurements in the study population. Measurement methods were equally frequent, whereas side and height differed among the cohort (Table 2). The relative change in ultrasound measurements was calculated as (measure day 7-measure day 1)/ measure day 1 for all possible locations. The mixed model shows no difference between the relative decrease in muscle mass regarding method (
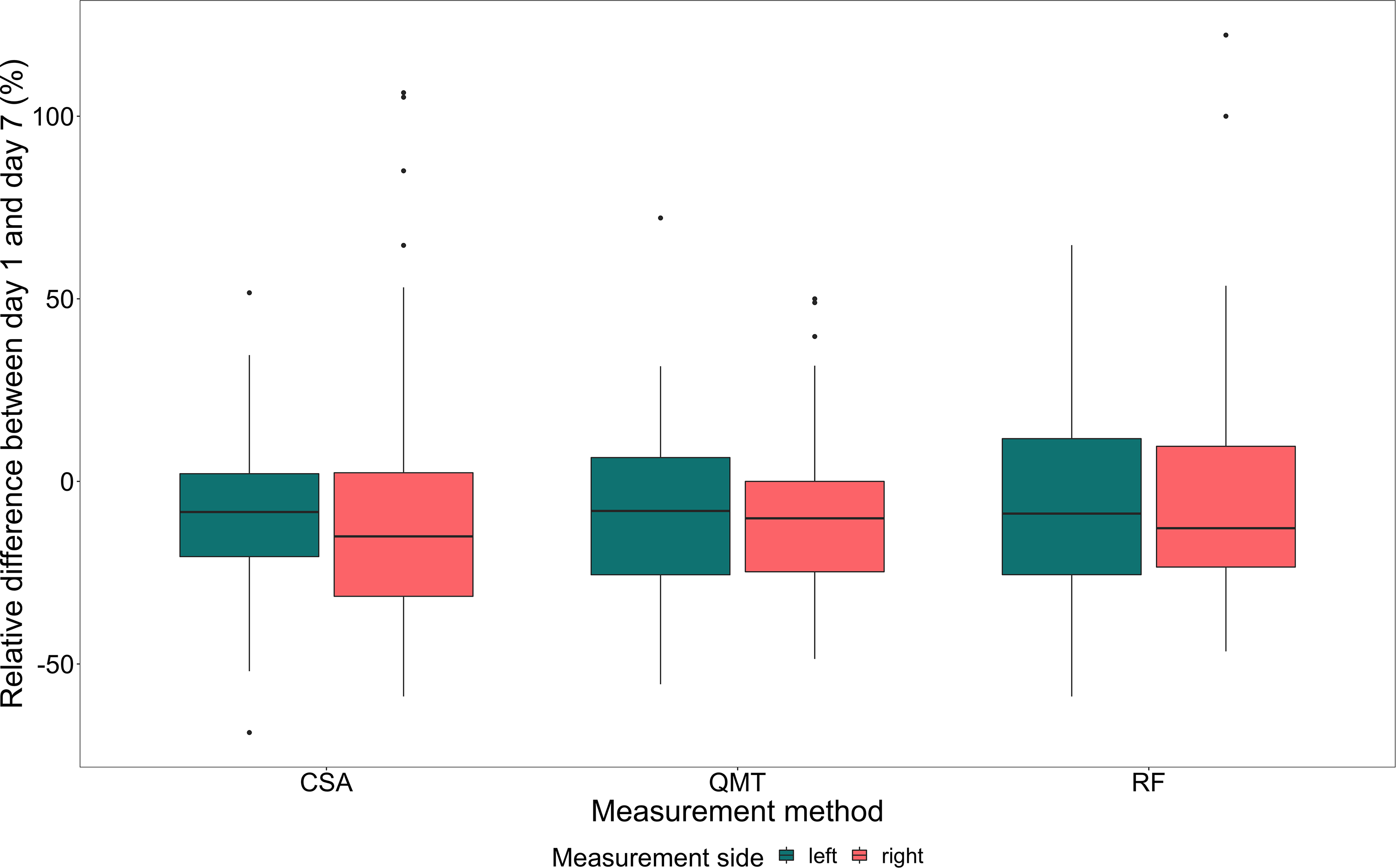
Relative difference between day 1 and day 7 (%) between the different measurement methods and measurement side. Abbreviations: CSA, Cross Sectional Area; QMT, Quadriceps Muscle Thickness; RF, Rectus Femoris Muscle.
Characteristics of the Different Ultrasound Measurements.
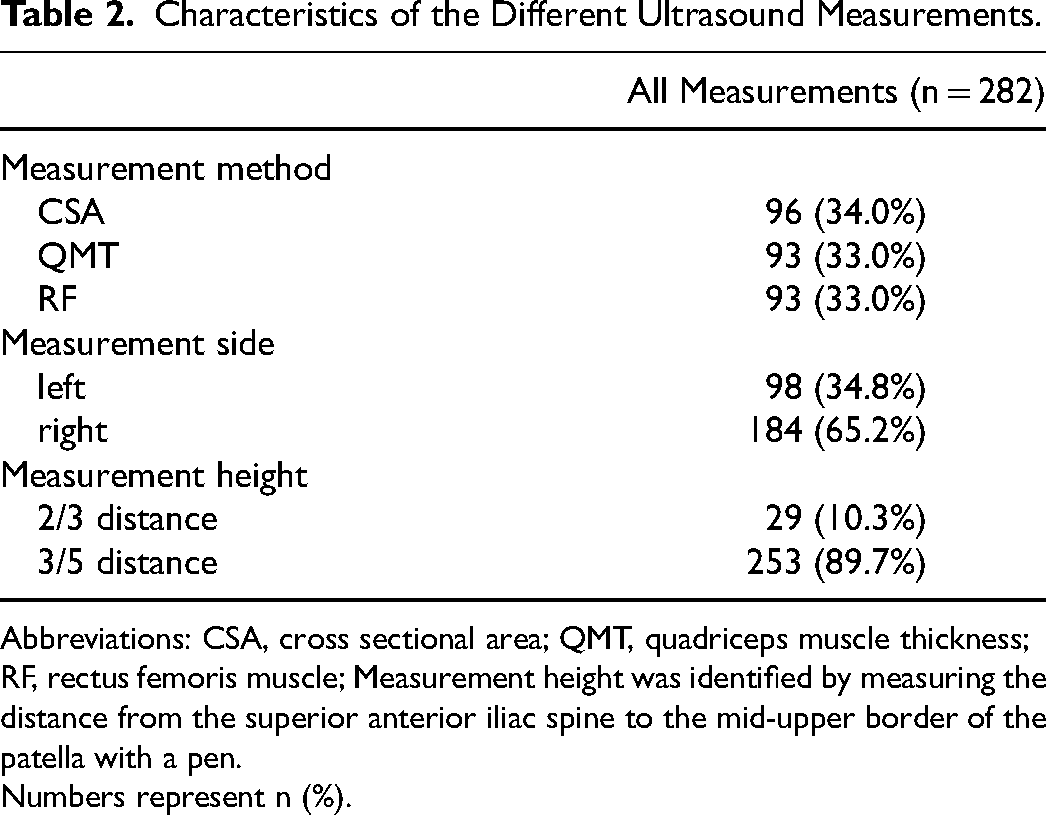
Abbreviations: CSA, cross sectional area; QMT, quadriceps muscle thickness; RF, rectus femoris muscle; Measurement height was identified by measuring the distance from the superior anterior iliac spine to the mid-upper border of the patella with a pen.
Numbers represent n (%).
Mixed Effect Model for Relative Differences Between Day 1 and Day 7.
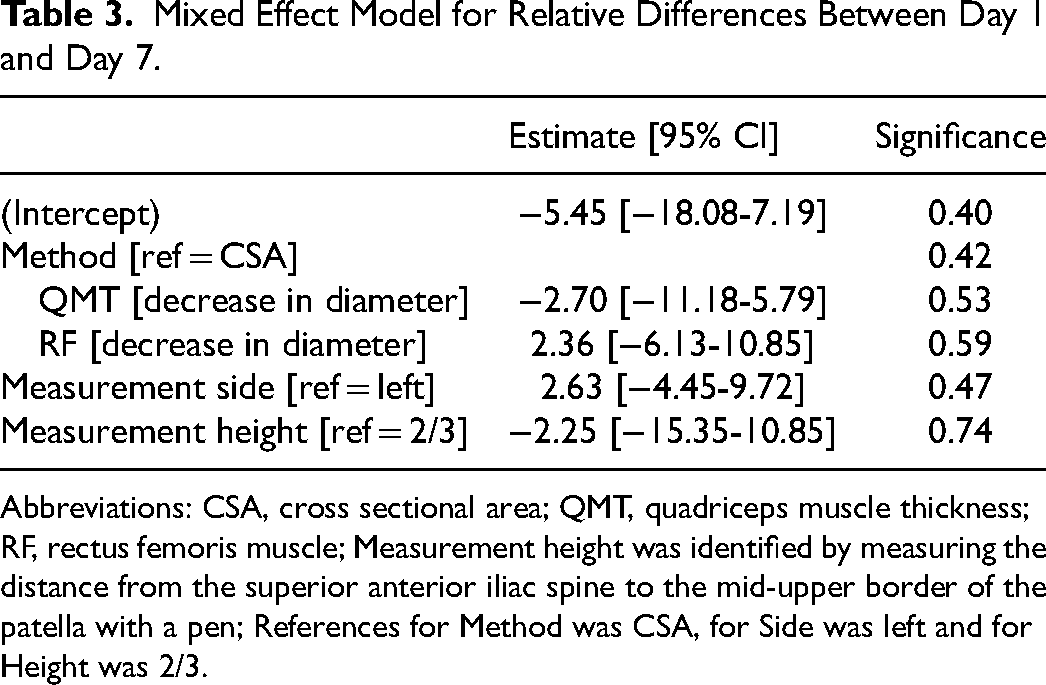
Abbreviations: CSA, cross sectional area; QMT, quadriceps muscle thickness; RF, rectus femoris muscle; Measurement height was identified by measuring the distance from the superior anterior iliac spine to the mid-upper border of the patella with a pen; References for Method was CSA, for Side was left and for Height was 2/3.
Secondary Outcomes
All measurements were averaged for one patient and divided by decrease or increase resulting in atrophy or swelling. Figure 1 shows the relative changes in muscle mass over the course of the first 7 days. There is no difference between the groups of patients with atrophy and swelling regarding clinical characteristics and treatment during critical illness, as shown in Table 4. Especially, the cumulative fluid balance over 7 days showed no difference (3127 ml [−667-6314] vs 4875 ml [2480-6388],
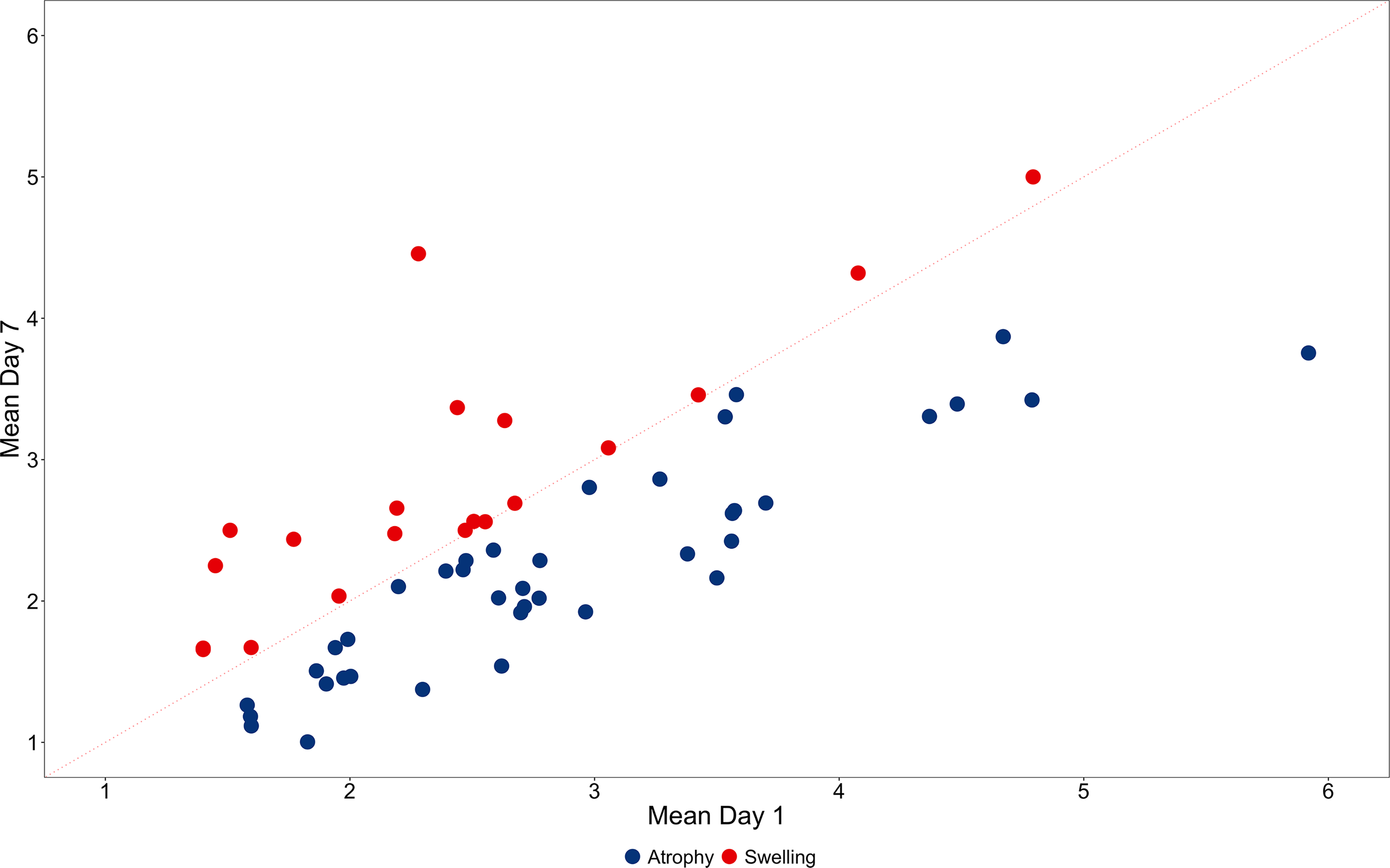
Relative changes in ultrasound measurements between day 1 and 7 after ICU admission. All measurements were averaged for one patient and divided by decrease or increase resulting in atrophy or swelling. Red dots indicate a relative increase, ie swelling; blue dots indicate a relative decrease, ie atrophy. The red dotted line indicates that there was no change between day 1 and 7.
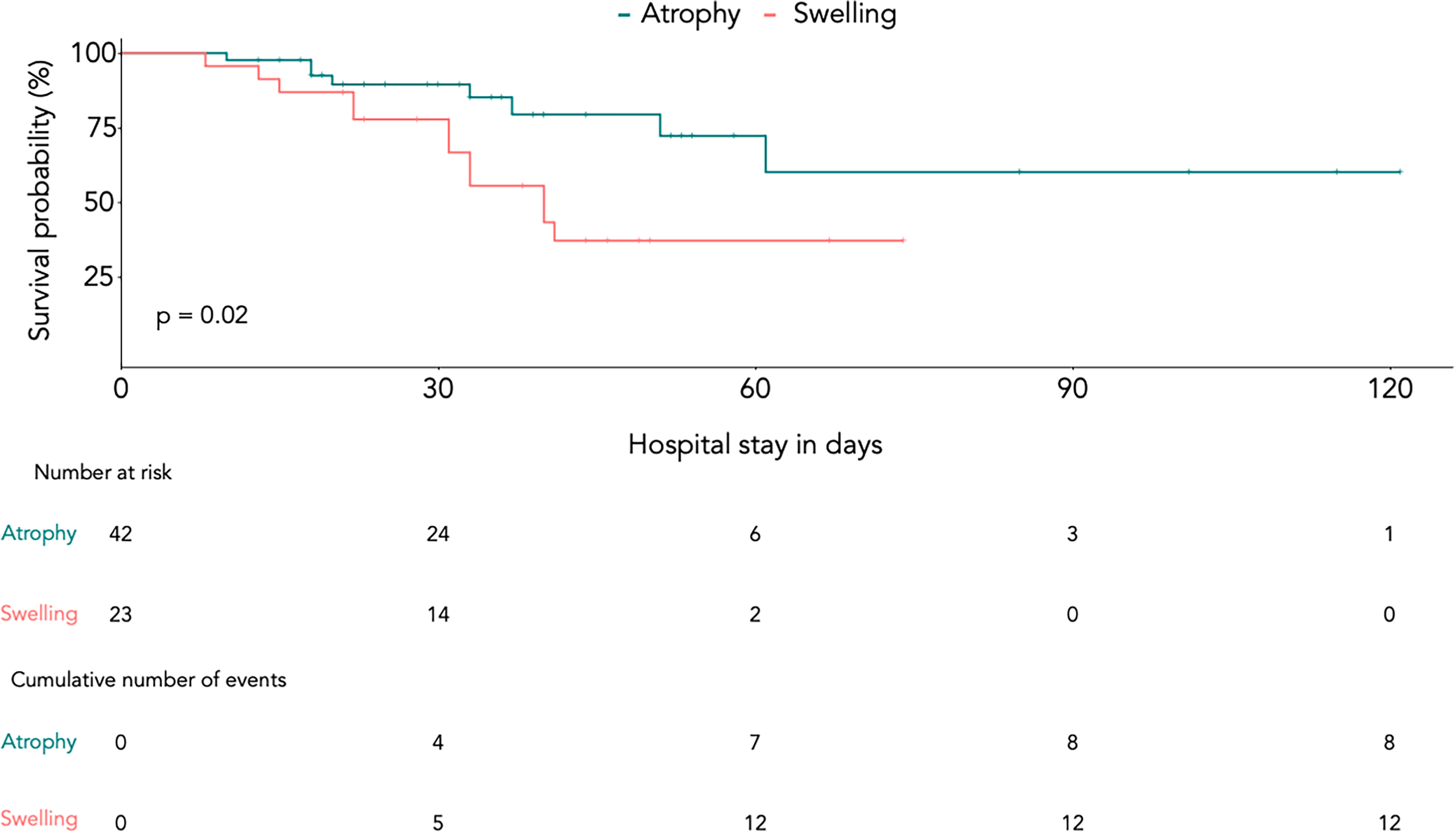
Survival probability of patients with atrophy and swelling presented by Kaplan Meier curve. Red line indicates patients with swelling, green line indicates patients with atrophy. Log rank test between patients with atrophy and patients without atrophy or swelling showed a significant difference in hospital survival (
Patient Characteristics Stratified by Change in Mean Ultrasound Measurements Between Day 1 and 7 After ICU Admission.
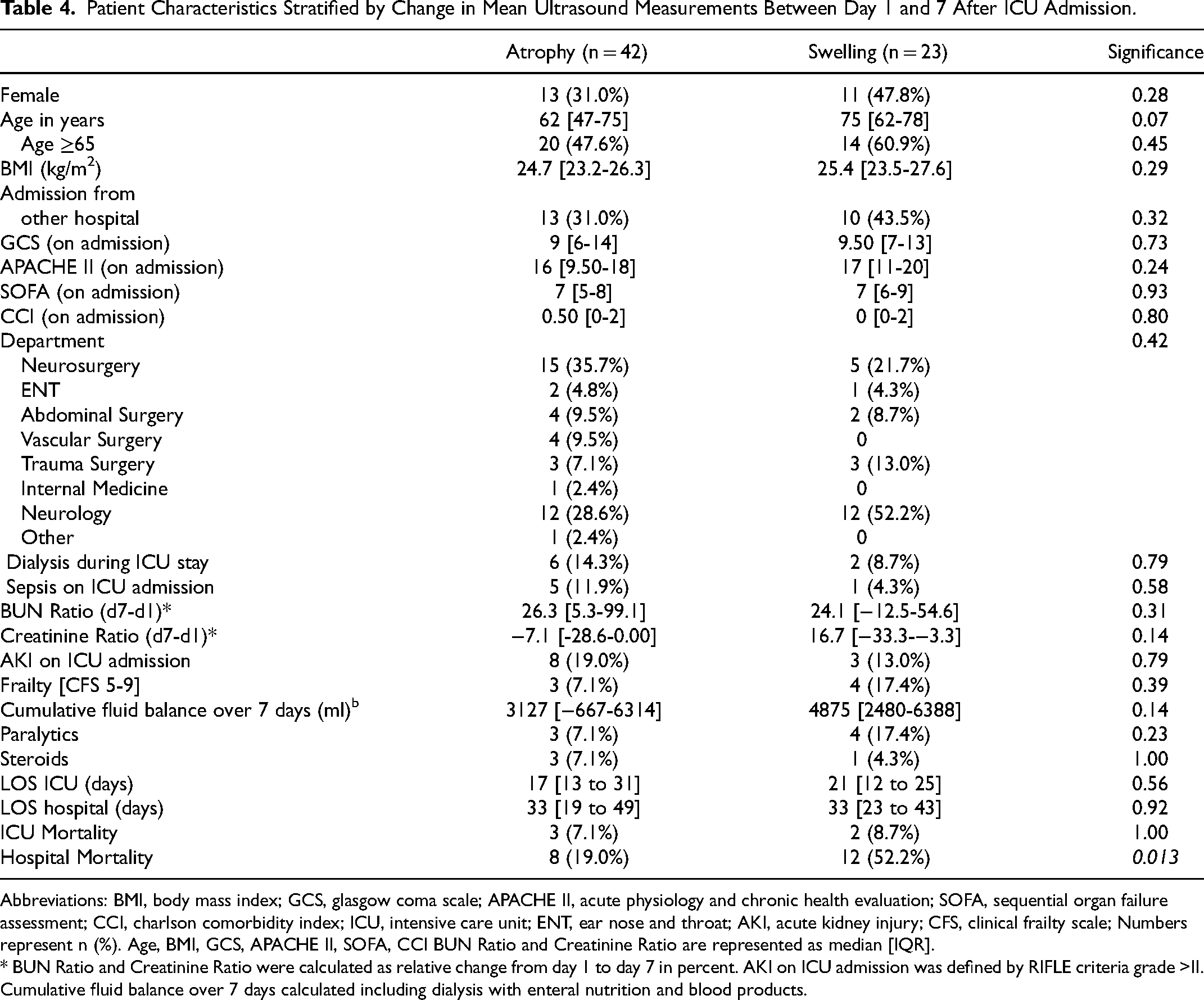
Abbreviations: BMI, body mass index; GCS, glasgow coma scale; APACHE II, acute physiology and chronic health evaluation; SOFA, sequential organ failure assessment; CCI, charlson comorbidity index; ICU, intensive care unit; ENT, ear nose and throat; AKI, acute kidney injury; CFS, clinical frailty scale; Numbers represent n (%). Age, BMI, GCS, APACHE II, SOFA, CCI BUN Ratio and Creatinine Ratio are represented as median [IQR].
* BUN Ratio and Creatinine Ratio were calculated as relative change from day 1 to day 7 in percent. AKI on ICU admission was defined by RIFLE criteria grade >II. Cumulative fluid balance over 7 days calculated including dialysis with enteral nutrition and blood products.
Multivariate Full Model with Selected Confounding Variables on Hospital Mortality.

Abbreviations: GCS, glasgow coma scale; SOFA, sequential organ failure assessment; CFS, clinical frailty scale;
*Cumulative fluid balance over 7 days calculated including dialysis with enteral nutrition and blood products.
Discussion
We prospectively studied different ultrasound methods of measurement in an heterogenous cohort of critically ill patients at risk of prolonged ICU and hospital stay or death. Most of the studied patients were severely injured or septic, which are well-recognized risk factors for ICUAW. We have found that all the investigated ultrasound methods and measurement sites for the thigh muscles published in different assessment protocols are equally effective in detecting muscle changes. Over the course of the first seven days in the ICU, two groups were identified: those whose muscle mass decreased in ultrasound measurement and another cohort in which there was an increase in muscle mass or swelling, respectively. Above that, we were able to show that mortality was significantly increased in the cohort with muscle swelling. As far as we know, this is the first time that techniques from different studies have been compared in critically ill patients and differentiation into distinct types (atrophy and swelling) has been performed. Puthucheary was one of the first to demonstrate the relevance of the first week of critical illness to muscle loss and the development of ICUAW. 25 Here, 63 ICU- Survivors experienced significant skeletal muscle weakness and physical disability. 23 Though this can be suggested by these data, severe muscle loss, as indicated by ultrasound, during critical illness does not occur in all of our patients. Moreover, this effect was independent of the type and severity of the disease and of the therapeutic measures taken in the intensive care unit in our cohort. While the number of patients is similar in both studies, the APACHE II Score 23.5 (95% CI, 21.9-25.2) was higher than in our analysis 17 [10-20]. Annetta et al observed a significant 45% decrease in rectus femoris cross-sectional area over a 20-day ICU stay in a cohort of 38 young trauma patients, meanwhile tibialis anterior CSA reduction of 22% was non-significant. 24 The authors discussed whether different functions and muscle fiber types played a role in the different magnitudes of change for these muscles. The rectus femoris is an extensor with Type II fast fiber predominance and the tibialis anterior a flexor with Type I slow fiber predominance. The causative mechanism responsible for these differences in reduction rates among different muscle groups remains unclear. 24 Although our analysis did not examine different muscle groups, there was a detailed assessment of the thigh muscle, which represents common practice. In comparison, no differentiation was made between an increase or reduction of muscle mass in either of the above-mentioned studies. As inflammatory and molecular pathways at the level of the muscle fiber remain one possible cause, it can only be assumed, that swelling of the muscle with consecutive edema, necrosis, and muscle breakdown is the cause of the observed difference in mortality between the two groups. Hernández-Socorro et al detected significantly greater levels of subcutaneous edema and fluid in critically ill patients as well as reduced microvascular angiogenic activity compared to a group of healthy controls. 25 Above that, their researched novel high-quality ultrasound tool showed a significant increase in muscle stiffness indicating a significant increase in muscle fibrosis. Although measurement of intrafascial or intramuscular fluid was not part of our study protocols, a different ultrasound technique would nevertheless have been necessary to quantify muscle perfusion or edema formation. However, the aim of their study was the detection of muscle changes with different innovative approaches in comparison to healthy control patients and did not look at outcome or mortality in the critically ill patients. This makes comparison with our study difficult. Apart from increasing fibrosis of the muscle, there is further remodeling and change in echogenicity over the course of the disease with increased fatty infiltration of the tissue. 26 As a consequence, muscles may be enlarged by the infiltration of fat, which is defined by the term pseudohypertrophy or false enlargement. 27 Although this phenomenon is more apparent in neurological diseases such as myotonia, muscular dystrophies or motor neuron disease, it is associated with muscular weakness and thus represents a reasonable explanation for our observation. 27 Another rationale for our results could be the underlying disease, that lead to critical illness. Trauma patients as well as patients with severe sepsis seem to develop a higher protein breakdown than patients with other pathologies. 4 However, there was no significant difference regarding admission reason or department in our cohort, which is composed of patients with a wide variety of medical conditions (eg polytrauma, neurocritical care or sepsis).
Fluid overload due to an imbalance of volume requirements and renal function, which often occurs during the first week of critical illness, is also one of the reasons we considered as a possible cause for the increase in muscle on ultrasound measurement. However, the statistical analysis did not show any difference regarding fluid balance or the need for renal replacement therapy. Grimm et al. examined 28 patients with either septic shock or severe sepsis on days 4 and 14 after onset of sepsis. In their study, mean cumulative fluid balances at day 4 and day 14 were similarly positive. The authors conclude, that changes in superficial tissues due to edema may affect the interpretation of muscle images and may also cause artificial amplitude reductions in nerve conduction measurements. 26 However, they state that the significance of tissue edema in the assessment of muscle echogenicity may be overestimated, since tissue edema cannot alter the bone signal in contrast to fibrous tissue. Although echogenicity was not determined in our study, careful quality assessment of the images was performed to ensure high quality and delineation of the muscle from surrounding tissue. All these aspects to be discussed can only be adequately addressed by a muscle biopsy, which is an invasive procedure. A strength of this study is the wide variety of patients included with continuous ultrasound monitoring during the course of the first 7 days. This resulted in a large number of measurement points, that allowed for good comparability. Although this was a single center study, our cohort showed a heterogenous group of adult patients regarding diagnosis or prognosis of the disease. Assessing the relative changes in all ultrasound measurements between day 1 and 7 after ICU admission makes this study unique compared to others and offers new perspectives in understanding the role of different ultrasound methods to assess muscle mass of critically ill patients. However, the cohort of patients was retrospectively assembled from 3 different studies, which is a limitation. Yet, it should be noted, that the different study protocols from the different studies have finally raised the question of this analysis; which is the best method to detect changes in muscle mass in critically-ill patients. Although we implemented detailed and ongoing training of our study staff, the assessment using ultrasound highly depends on the examiner. This important problem has not been satisfactorily solved yet. To address this bias, we performed a quality control of the images before data entered the evaluation. Images that did not meet specific criteria, were excluded from assessment. However, this has to be mentioned as limitation. Since the majority of our patients are surgical or trauma, the results should be validated in medical ICU patients as well. Lastly, follow-up of the survivors was not obtained, which weakens the validity of the data. Further prospective studies with specified follow-up will be needed to address the change in muscle mass during critical-illness and explore molecular and pathophysiological pathways to evaluate the causes for atrophy and swelling as indicated by ultrasound.
Conclusion
In conclusion, different approaches to ultrasound measurement during critical-illness can equally detect muscle changes in the quadriceps muscles. While some patients show a decrease in muscle mass during the first week of critical illness, others show enlargement or swelling. This may result in a reduced probability of surviving the hospital stay. Causative reasons for these results still remain unclear. It can only be assumed, that swelling of the muscle with consecutive edema, necrosis, and muscle breakdown might be one contributing factor for the observed difference in mortality between the two groups.
Supplemental Material
Supplemental Material
sj-docx-1-jic-10.1177_08850666221132246 - Supplemental material for Comparison of Different Ultrasound Methods to Assess Changes in Muscle Mass in Critically ill Patients
Supplemental material, sj-docx-1-jic-10.1177_08850666221132246 for Comparison of Different Ultrasound Methods to Assess Changes in Muscle Mass in Critically ill Patients by Kristina E. Fuest, Hugo Lanz, Jana Schulz, Bernhard Ulm, Victoria A. Bennett, Julius J. Grunow, Björn Weiss, Manfred Blobner and Stefan J. Schaller in Journal of Intensive Care Medicine
Footnotes
Authorship Statement Section
SJS is the principal investigator and developed the protocol, BU is the study statistician. KEF, SJS and MB were involved in the ethical approval, KEF, BU, HL, MB und SJS were involved in the analysis and interpretation of the data. KEF, HL, ML and SJS were involved in data acquisition and quality assurance. All authors critically revised the manuscript and approved its final version.
Ethics Approval and Consent to Participate
The study was approved by the ethics committee of the School of Medicine of the Technical University of Munich (Grillparzer Str. 16, 81675 Munich, Chairperson Prof. Dr G. Schmidt, approval number 150/22S). The study was conducted in agreement with the principles of the Declaration of Helsinki. All participants were informed about the purpose of the study, the risks, and the potential benefits. Written informed consent was obtained by the local study physician from each participant for the original studies. The Department of Anaesthesiology and Intensive Care of the School of Medicine, Technical University of Munich coordinated the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BW reports personal fees from Orion Pharma Ltd and national (DAAD) and international grants (ESICM) outside the submitted work.
MB received research support from MSD (Haar, Germany) not related to this manuscript, received honoraria for giving lectures from GE Healthcare (Helsinki, Finland) and Grünenthal (Aachen, Germany). Dr. Schaller reports grants from Reactive Robotics GmbH (Munich, Germany), grants and non-financial support from STIMIT AG (Biel, Switzerland), Liberate Medical LLC (Crestwood USA), ESICM (Geneva, Switzerland), grants, personal fees and non-financial support from Fresenius Kabi Deutschland GmbH (Bad Homburg, Germany), personal fees from Springer Verlag GmbH (Vienna Germany) for educational purposes, non-financial support from Technical University of Munich (Munich, Germany) and from National and international societies (and their congress organizers) in the field of anesthesiology and intensive care medicine, outside the submitted work. Dr. Schaller held stocks in small amounts from Rhön-Klinikum AG and holds stocks in small amounts from Alphabeth Inc., Bayer AG and Siemens AG; these holdings have not affected any decisions regarding his research or this study. The other authors declare that they have no competing interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
