Abstract
Sickle cell disease (SCD) is associated with substantial morbidity and early mortality in afflicted adults. Cardiopulmonary complications that occur at increased frequency in SCD such as pulmonary embolism, pulmonary arterial hypertension, and acute chest syndrome can acutely worsen right ventricular function and lead to cardiogenic shock. Mechanical circulatory support including venoarterial extracorporeal membrane oxygenation (VA ECMO) is being increasingly utilized to treat hemodynamic collapse in various patient populations. However, a paucity of literature exists to guide the use of mechanical circulatory support in adults with SCD where disease-related sequela and unique hematologic aspects of this disorder may complicate extracorporeal therapy and must be understood. Here, we review the literature and describe three cases of adult patients with SCD who developed cardiogenic shock from acute decompensated right heart failure and were treated clinically with VA ECMO. Using an in vitro ECMO system, we investigate a potential increased risk of systemic fat emboli in patients with SCD who may be experiencing vaso-occlusive events with bone marrow involvement given the high-volume shunting of blood from venous to arterial systems with VA ECMO. The purpose of this study is to describe available extracorporeal life support experiences, review potential complications, and discuss the special considerations needed to further our understanding of the utility of VA ECMO in those with SCD.
Keywords
Introduction
Sickle cell disease (SCD) arises from a point mutation in the ß-globin gene that leads to polymerization of the resultant mutant form of hemoglobin (Hb), Hb S, in red blood cells when oxygen tension is low. Hb S polymerization causes erythrocyte membrane distortion, red cell fragility, altered cellular rheology, hemolysis, and impairments in microcirculatory flow. This can lead to the entrapment of erythrocytes in the microvasculature, known as vaso-occlusion, and to the activation of the innate immune and complement systems inciting endothelial cell dysfunction.1,2 The sequela of SCD includes chronic hemolysis, anemia, vaso-occlusive episodes (VOEs), hypercoagulability, ongoing inflammation, and both pulmonary and systemic vasculopathy, which contribute to acute and chronic right ventricular dysfunction (RVD). 3 A major vasculopathic complication in SCD that is associated with increased mortality and RVD is pulmonary hypertension (PH). There are multiple factors that can drive PH in SCD and a range of hemodynamic findings on right heart catheterization. 4 Mediators of SCD-related PH include oxidative stress and nitric oxygen scavenging by cell-free Hb with hemolytic anemia, thromboembolic disease secondary to hypercoagulability, and diastolic left heart disease from high cardiac output state in the setting of chronic anemia.2,3,5 Furthermore, in SCD patients without PH, imaging studies have shown dilated right and left heart chambers in steady state, thought to relate to the severity of anemia with secondary increases in stroke volume, and often diastolic heart disease. While these individuals did not often have significant RVD, it is clear that many aspects of SCD pathophysiology set the stage for increased right heart stress.3,6–8
Frequent complications of SCD such as acute chest syndrome (ACS) and pulmonary embolism (PE) can acutely worsen right ventricular (RV) pressure load and function. ACS, a common cause of death in adults with SCD, arises from events inciting vaso-occlusion in pulmonary vasculature that include infection, thrombosis, hypoxemia, and notably, fat embolism, which is suspected to cause a significant portion of ACS in adults.9,10 Fat emboli result from bone marrow necrosis that occurs during VOE leading to the release of fat and bone marrow particulate into the venous circulation that travels to the lungs and incites local vaso-occlusion. The resultant ischemia and endothelial injury can further propagate additional vaso-occlusion in the pulmonary vasculature and may prompt ACS to progress rapidly. 10 The increase in pulmonary afterload that occurs in ACS can lead to cor pulmonale, which has been reported in 13% of patients in a series describing 84 consecutive ACS episodes. 11 Similar to ACS, PE can also lead to decompensated right heart failure and is associated with a higher in-patient mortality in those with SCD. 12 In addition to increased right-sided pressure from clot burden, pulmonary thromboembolism, itself, may trigger ACS and further worsen pulmonary vascular resistance.13,14
In patients with SCD, acute pressure overload on top of chronic pulmonary vasculopathy and elevated right-sided demand from high pulmonary flow can converge to cause decompensated right heart failure and cardiovascular collapse. 3 In this setting, mechanical circulatory support with venoarterial extracorporeal membrane oxygenation (VA ECMO) has been attempted as a rescue measure. VA ECMO diverts venous blood away from the right side of the heart, decompressing the RV, while at the same time providing perfusion by returning oxygenated blood to the arterial system after it passes through a membrane oxygenator. While numerous reports describe successful use of VA ECMO for decompensated right heart failure, particularly in the setting of PE, there is a paucity of literature detailing VA ECMO utilization in patients with SCD. 15 Here, we provide a literature review and a description of three cases of patients with SCD who were placed on VA ECMO with severe decompensated right heart failure to further our understanding of the use of this mechanical circulatory support modality, outcomes, and complications in this setting.
Methods
Clinical Data
The retrospective review of available clinical data from patients with SCD treated for clinical indications with ECMO at the University of Maryland Medical center was approved by the institutional review board at the University of Maryland, Baltimore and granted a waiver of written informed consent (HP-00078659).
Particle Membrane Oxygenator cut-off In vitro Study
An in vitro circulatory loop was constructed with a Rotaflow centrifugal pump (Maquet Getinge Group, Wayne, NJ) and a Quadrox-i® membrane oxygenator (Maquet Cardiopulmonary AG; Hirrlingen, Germany). Normal saline was used as the working fluid. A mixture of saline and red fluorescent particles (UVPMS-BR-0.995, Cospheric, Somis, CA) were injected upstream of the oxygenator. Particles of 100, 75-90, 53-63, and 30 microns were used. The largest particles were injected first until the specific size particles were visualized. An expanded argon laser was used to illuminate the flow in the tubing downstream of the oxygenator. Particle streak lines were recorded using a Nikon Z 6II digital camera (Nikon USA, Mellvile, NY).
Case Description
Case 1
A 38-year-old woman with SCD (Hb SS) and history of infrequent VOE presented with diffuse body pain and was admitted (hospital day 0) for treatment of VOE. On hospital day 1, her Hb dropped from 7.3 mg/dL on admission to 5.3 mg/dL and two units of crossmatched blood were administered. She developed worsening leukocytosis, fever, hypoxemia, hypotension and was placed on mechanical ventilation. Intubation was followed by further hemodynamic decline with bedside transthoracic echocardiogram (TTE) showing severe RV dilation and septal bowing. CTA imaging showed diffuse ground glass opacifications and bibasilar subsegmental acute pulmonary emboli whereas baseline CTA performed previously no abnormalities (Figure 1a). Increased RV: left ventricular (LV) ratio and flattening of the interventricular septum were noted (Figure 1b). Alteplase was administered given finding of PEs. In the setting of worsening shock, she was transferred to our center and placed on VA ECMO. At the time of cannulation, she had significant hemodynamic instability, acidosis, and vasoactive requirements. Notable labs include Hb 6.5 g/dL, arterial blood gas: pH 7.13/PaCO2 24/PaO2 191, lactate >17 mmol/L, bicarbonate 10 mmol/L, and creatinine elevated at 1.9 mg/dL from normal baseline. Two units of O- uncrossed blood were used to prime the ECMO circuit. After cannulation, HgB A + F by column was 70%. She was alert and followed commands before and several hours after ECMO initiation. Vasopressors were able to be weaned off with epinephrine remaining for inotropic support. TTE the day after cannulation, hospital day 2, with 3.2L/min of VA ECMO flow showed decreased LV size and filling with normal wall thickness. LV ejection fraction was 20% with good contractility of the inferolateral and lateral walls. RV size was normal with severely reduced systolic function. On hospital day 2, the patient stopped following commands. The neurologic examination only demonstrated withdrawal only in right upper extremity and the presence of cough, gag, and corneal reflexes. Pupils were pinpoint and non-reactive. CT imaging of head, chest, abdomen, and pelvis showed no acute intracranial abnormality, bilateral lower lung lobe consolidations, and new retroperitoneal and peritoneal hemorrhage. Coagulopathy was corrected with matched transfusion. Per institutional practice, patients with SCD are routinely matched for ABO, C, c, D, E, e, and Kell antigens. All anticoagulation remained held during the entirety of the ECMO course. Direct antiglobulin testing was negative and there was no concern for transfusion reaction based on hematology assessment. An exchange transfusion was performed. Post-exchange Hb was 8.5 g/dL and HbA + F was 85%. Renal function worsened requiring renal replacement therapy and metabolic acidosis subsequently resolved. On hospital day 3, cardiac function and pulse pressure began to improve. Inotropic epinephrine was weaned off to prevent emerging harlequin syndrome in the setting of declining right radial arterial PaO2 (67 mm Hg at the lowest) and worsening chest radiographic opacities. Mental status remained poor and repeat head CT obtained on hospital day 3 showed diffuse cerebral edema and tonsillar herniation, which progressed to brain death. Autopsy showed numerous microscopic pulmonary emboli and fat emboli in alveolar capillaries
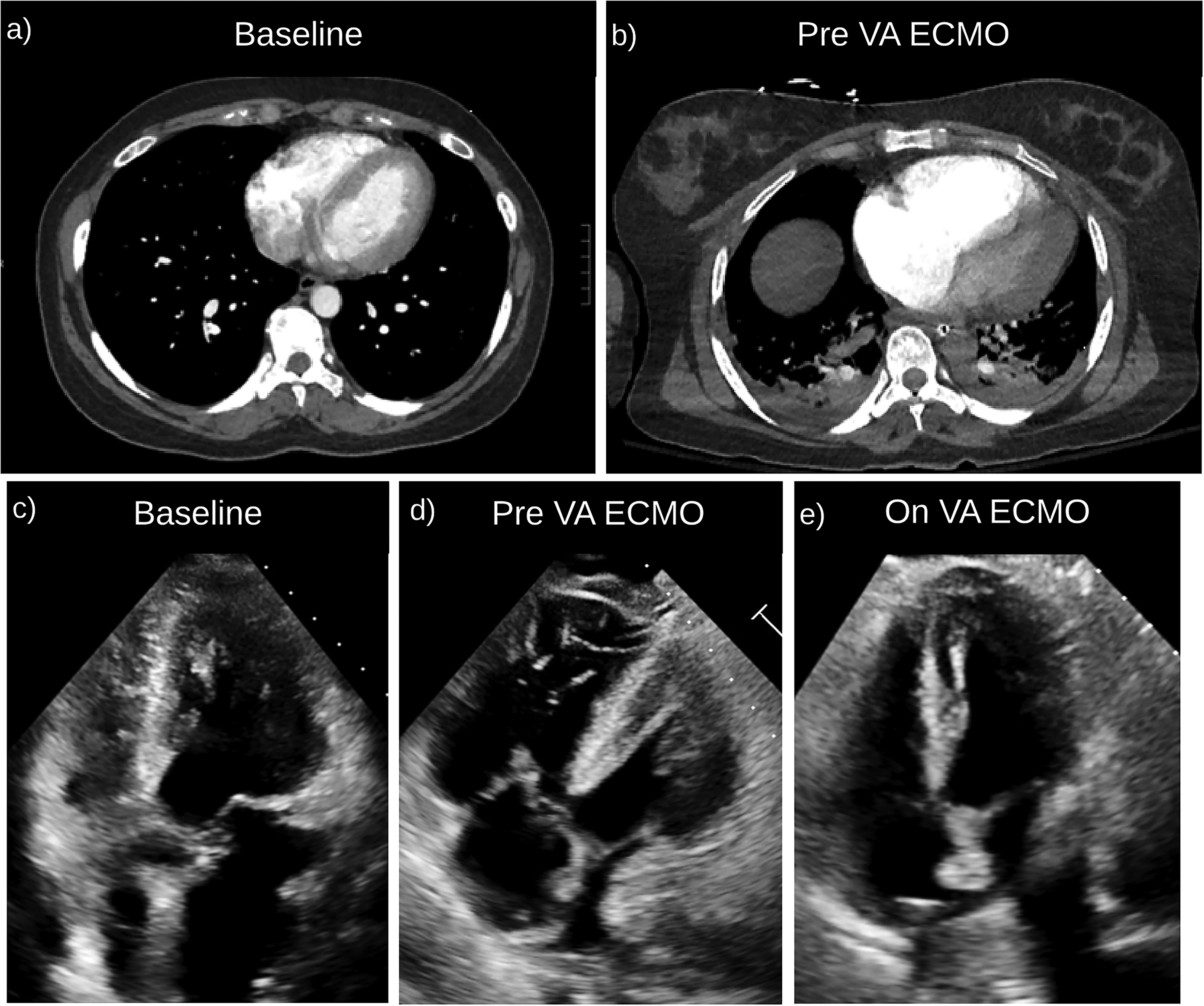
(a) Patient 1: baseline CTA chest with normal heart size and no PE. (b) CTA just prior to ECMO with subsegmental bibasilar PE and increased RV:LV ratio and flattening of the interventricular septum suggestive of right heart strain. (c) Patient 3: baseline TTE (9 months) prior to VA ECMO initiation: normal RV wall size and function. Trace tricuspid regurgitation (TR). Normal LV size, wall thickness and function (LVEF 60-65%)
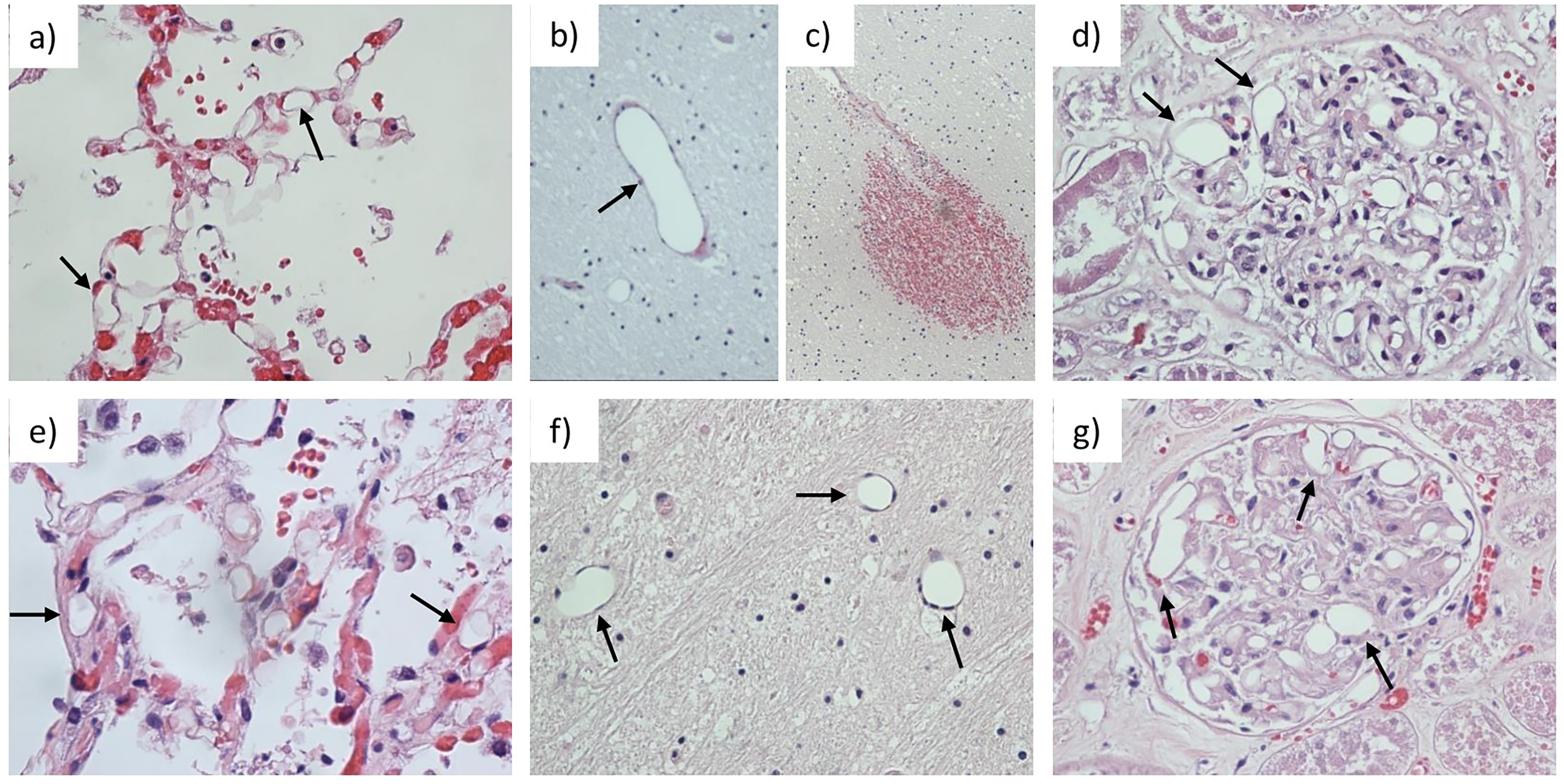
(a, e) Lung specimen showing vacuolization (arrows) in alveolar capillaries consistent with fat embolization in Cases 1 and 2, respectively. (b, f) Distorted vacuolated brain capillaries (arrows) found in Cases 1 and 2, respectively, and (c) brain petechial hemorrhages found in Case 1, consistent with cerebral fat emboli versus severe hypoxic-ischemic injury. (d, g) Vacuolization and dilated glomerylar capillary lumens (arrows) from Case 1 and 2, respectively, consistent with renal fat embolism.
Case 2
A 48-year-old man with history of SCD Hb S, beta zero thalassemia (HbS/ ß0-thal), and intermittent episodes of VOE presented with 3-4 days of subjective fever, cough and abdominal, back, and thigh pain typical of pain crisis leading to admission (hospital day 0). Hb was 7.2 mg/dL. Shortly after admission, he developed hypoxemic respiratory failure and hemodynamic instability requiring mechanical ventilation and vasopressors, respectively. He was subsequently transferred to our center for exchange transfusion consideration on hospital day 1. Upon arrival, hemodynamics worsened and TTE demonstrated severe RV dilation with akinesia of the mid-free wall and normal apical motion. The LV was underfilled with normal but hyperdynamic function. Chest radiograph showed diffuse widespread opacities. Hb dropped to 5.7 mg/dL and platelets were 31 K/mcL. He developed a short PEA arrest lasting 2 minutes with return of spontaneous circulation. Given concern for massive PE, VA ECMO was initiated given persistent hemodynamic instability. ECMO initiation was complicated by two additional short arrests secondary to ventricular fibrillation, each lasting under 3 minutes. After ECMO cannulation, vasopressors were initially weaned off and subsequent hypertension was treated with calcium channel blocker infusion. Heparin was initiated for anticoagulation. Lactic acidosis persisted and he developed worsening liver function and kidney injury requiring renal replacement therapy. Hemodynamic instability returned requiring vasopressor support. Mental status remained poor, and he developed signs of brain herniation with unequal pupils. The decision was made by the family to withdraw life-sustaining support on hospital day 3 given multiorgan failure and poor prognosis. Serum parvovirus IgM and IgG were both elevated at 2.7 and 12 index, respectively. Autopsy did not demonstrate PE. On histology, there were diffuse thrombi and fat emboli in lung capillaries (Figure 2e) consistent with ACS. There were diffuse fat emboli in the kidneys (Figure 2g
Case 3
A 60-year-old woman with HbS/ ß0-thal and end-stage renal disease presented with weakness, fatigue, diarrhea, and leukocytosis. Clostridium difficile infection was diagnosed with course complicated by fulminant colitis requiring colectomy. Chest CT imaging immediately prior to colectomy showed bilateral upper lobe subsegmental pulmonary emboli without evidence of right heart strain and bilateral lower lobe consolidation consistent with pneumonia. On post-operative day 1, she had a rapid decline in hemodynamic status requiring escalating doses of vasopressors. TTE showed severely dilated RV and severely reduced RV systolic function with relative apical sparing (new from a study 9 months prior noting normal RV function and RVSP). There was decreased LV size and filling with normal wall thickness and EF 65-70% (Figure 1 c, d). Given suspicion for worsening PE burden and severe hemodynamic decline, the patient was placed on VA ECMO. After cannulation, vasopressors were initially weaned off. However, progressive abdominal distension developed, and ECMO blood flow rates declined consistent with abdominal compartment syndrome requiring a decompressive laparotomy. The patient transiently improved but hemoptysis, bloody output from the abdominal wound vac requiring massive transfusion, and hemodynamic instability then occurred. Upon abdominal re-exploration, bleeding from the inferior border of the pancreas and diffuse raw surface bleeding consistent with coagulopathy was discovered. Repeat TTE showed RV decompression and improved function (Figure 1e). Transfusion requirement continued and on follow-up CT, spontaneous bleeding from inferior aspect of the splenic artery was identified and treated with embolization. All anticoagulation was held throughout the entirety of the ECMO course and coagulopathies were corrected. However, the patient remained critically ill with a continuous vasopressor requirement, lactic acidosis (5-7 mmol/L), progressive hyperbilirubinemia (8.3 mg/dL), and the development of tension pneumothorax requiring chest tube placement. On post-operative day 4 following colectomy, a decision was made by family to withdraw life-sustaining support given poor clinical status and multiorgan failure. No autopsy was performed though clinical course was consistent with a diagnosis of ACS and PE inciting cardiovascular collapse.
Results
With two of the above patients showing pulmonary and likely arterial cerebral and renal fat emboli on autopsy (Figure 2), a theoretical risk of increasing systemic fat embolism with the use of VA ECMO support in patients with SCD was identified. To further study this potential risk, the size of particulate matter that could be shunted from the venous to arterial limb of a VA ECMO circuit by traversing the oxygenator was investigated. Using an in vitro ECMO circuit and Quadrox-i® membrane oxygenator (MAQUET Cardiopulmonary AG; Hirrlingen, Germany), the standard oxygenator used in each of these cases, we found that fluorescent particles ranging from 30 to 100 microns were all able to traverse through the oxygenator when administered to an ECMO circuit flowing crystalloid at 4L/min (Supplemental Figure 1). Particles larger than 100 microns were not tested. Of note, whole blood, which may confer alterations in particle transmissibility, was not evaluated in this model.
Discussion
The utility of VA ECMO in adult patients with SCD remains poorly understood. Here, we describe three cases where VA ECMO was used in the setting of cardiogenic shock from acute decompensated right heart failure mediated by ACS and/or PE (fat or thromboembolic). The complications described during VA ECMO therapy and the poor outcomes experienced in each of these cases highlight many of the special considerations needed with ECMO utilization in this patient population.
Very few reports are available describing VA ECMO use in adults with SCD, and thus, little information exists to guide decision-making. In fact, this literature is limited, to our knowledge, to three isolated successful cases with ACS, PE, and post-cardiotomy shock as indications for VA ECMO, and one study describing the use of extracorporeal life support in a cohort of adult SCD patients with ACS.9,17–19 This report by Boissier et al includes 12 patients who received VA ECMO and 10 additional patients who were given pulmonary support via venovenous (VV) ECMO where oxygenated blood is returned to the right atrium after initial diversion to the extracorporeal membrane oxygenator via the vena cava. 9 In those managed with VA ECMO, mortality was very high (83.3%) as were hemorrhagic and ischemic complications, which occurred in 60% and 33.3%, respectively. Like our three cases described above, most patients treated with VA ECMO in this cohort had acute right heart failure, cor pulmonale, and multiorgan failure (83.3%). Interestingly, those treated with VV ECMO also had high incidence of cor pulmonale (90%) and multiorgan failure (70%). However, they had a somewhat lower, though not significantly different, mortality rate (60%). While the decision to utilize VA ECMO over VV support in this study was not reviewed in detail, it was noted that those placed on VA ECMO experienced increased cardiac arrest events and more severe lactic acidosis prior to ECMO initiation, signifying severe hemodynamic compromise. 9 According to recent Extracorporeal Life Support Organization database analyses that included all adult patients placed on ECMO from 2018 to 2022, the overall mortality of those on VV ECMO or VA ECMO support was 42% and 32%, respectively, which is significantly better than the ECMO survival reported by Boissier et al for SCD patients in their cohort.9,20
The selection of modality when considering mechanical circulatory support in patients with SCD is critical. Given that sudden increases in pressure load, often on top of chronic pulmonary vasculopathy, underlie right heart failure in SCD, identifying the etiology of decompensation will assist in ECMO modality selection. 3 For triggers of right heart failure like ACS, both VV and VA ECMO have been used. Given the pathophysiology of ACS, isolated pulmonary support with VV ECMO may be a favorable therapy because the correction of hypoxemia may help reverse the inciting cause of increased RV pressure load via relief of hypoxic vasoconstriction and prevention of continued pulmonary vaso-occlusion. 9 In addition to potential reductions in pulmonary vascular resistance, assistance with gas exchange can lead to further hemodynamic improvement as derangements like respiratory acidosis resolve. For those in clear need of hemodynamic support, such as those with left heart dysfunction, cardiac arrest or severe shock not thought to be reversible with isolated pulmonary support, VA ECMO may be more favorable. VA ECMO decompresses the RV by diverting blood to the ECMO circuit and assists with perfusion by directing the oxygenated blood to the arterial system. With this, there are decreases in RV end-diastolic volume and pressure (Figure 1c, d). This reduction in preload and often RV afterload secondary to the rerouting of blood improves the ratio of myocardial oxygen supply to demand, decreases RV strain, and can assist in RV recovery. 21 However, the complications specific to VA ECMO that may occur in patients with SCD, discussed below, need to be strongly weighed.
An additional mechanical circulatory support consideration is the use of VV ECMO in a configuration that offers right ventricular assist device (RVAD) support, such as with the use of a dual lumen cannula like the Protek Duo® (LinaNova, London, UK). With the Protek Duo, the proximal drainage port is stationed in the right atrium and the distal blood return lumen sits within and can deliver oxygenated blood directly to the pulmonary artery, off-loading the RV. RVAD support will decrease right atrial pressure and increase mean pulmonary artery pressure. The amount of blood flow achieved is dependent on preload and afterload, and, thus, will vary based on the patient's physiologic state and the etiology of PH.21,22 The decision to provide RVAD support relies heavily on the cause of RV decompensation and the overall clinical scenario. While RVAD use might be appropriate in SCD patients with ACS and acute respiratory distress syndrome where pulmonary vascular resistance may be, in part, reversible with improvement in gas exchange, RVAD support is considered contraindicated in severe precapillary PH due to risk of pulmonary hemorrhage. 23 In the case of decompensated right heart failure in the setting of PE, the 2019 European Society of Cardiology guidelines comment that VA ECMO may be helpful in high-risk PE and numerous reports are available describing the use successful of VA ECMO for mechanical support in this setting in non-SCD populations. 15 On the other hand, the use of RVAD support in PE is limited to case reports leaving the utility and drawbacks of RVAD therapy in the setting of acutely elevated pulmonary vascular resistance due to an obstructive etiology poorly described. 21 In patients with SCD including the cases detailed in this series, PE may trigger ACS and thus, the exact etiology of RV decompensation may be less clear and/or multifactorial, complicating mechanical circulatory support decision-making.13,14
If VA ECMO is deemed necessary in an adult patient with SCD, there must be high awareness of the potential complications. The majority of VA ECMO runs in the literature have occurred in SCD patients with ACS. Thus, compromise in pulmonary function is a predominant feature. As cardiac function starts to improve, unequal oxygenation, known as harlequin syndrome, may begin to develop as was the concern in the first Case of this series. Harlequin syndrome arises when native pulmonary gas exchange remains poor, yet cardiac function begins to recover on peripheral VA ECMO. The improving heart ejects increased volumes of poorly oxygenated blood antegrade, which preferentially perfuse the arteries most proximal to the aortic valve including the coronary arteries and the cerebral vessels. This can cause myocardial and cerebral hypoxemia. If harlequin physiology persists after clinical optimization of native lung function, increasing ECMO flow, reducing inotropic support and augmenting volume removal with diuresis or ultrafiltration may be attempted to decrease the ratio of native cardiac output relative to ECMO flow. The goal of these maneuvers is to improve unequal oxygenation by allowing more retrograde flow from the ECMO circuit to reach all organs. 24 However, refractory cases of harlequin syndrome may warrant conversion of the ECMO circuit to an alternative configuration.
A potentially unique consideration of VA ECMO in SCD is highlighted in Cases 1 and 2. Fat embolism is a known complication of SCD and is a common cause of ACS in adult patients. 10 When fat and bone marrow particles are released into the venous circulation during vaso-occlusion, particles larger than 20 microns cannot readily pass through pulmonary capillary network. 25 With the presence of an intracardiac shunt, such as a patent foramen ovale as in Case 2, and/or physiologic intrapulmonary shunts, fat emboli may pass into the arterial system causing systemic fat emboli syndrome and organ injury. 26 With blood diverted from the venous system to the arterial system in VA ECMO at a typical rate 3-4 L/min, there is a hypothetical risk of greatly exacerbating systemic fat embolism. Given the passage of particles up to at least 100 microns, attention should be paid to the possibility of systemic embolism in a patient with VOC when considering VA ECMO therapy. While not definitive, shunting of blood to the arterial system via VA ECMO could have contributed to cerebral and renal fat emboli causing ischemic injury in Cases 1 and 2. At this time, this risk remains theoretical and should not change clinical decision-making without further study, which is warranted.
Hematologic features of SCD pose significant additional challenges in ECMO management and deserve careful attention. An aggressive and early exchange transfusion approach may be essential to mitigate processes such as fat embolism, ACS and the hematologic complications of extracorporeal therapy that are likely increased in SCD. While SCD is associated with hypercoagulability and an increased risk of thrombosis, patients with SCD are also more prone to bleeding. 27 Coagulopathy can further be exacerbated by critical illness and extracorporeal therapy, itself, which can cause acquired vWF deficiency, thrombocytopenia and platelet dysfunction. 28 Accordingly, bleeding complications were noted in two of three cases (Cases 1 and 3) in this series and in 60% of those in the cohort of SCD patients treated with ECMO reported by Boissier et al. 9 In addition, erythrocytes may be more prone to hemolysis under the shear forces of the extracorporeal circuit in SCD. Free Hb released during hemolysis can deplete nitric oxide, further impair platelet activity, and contribute to bleeding risk. 28 Thus, increased transfusion requirements are likely to occur, which can be complicated in the SCD patient, particularly when alloimmunization is present and the availability of matched red blood cell units is limited. Once an admitted patient is identified as having SCD, it is standard at our institution for all transfusions to matched for ABO, Rh, and Kell antigens underscoring the importance of early communication with transfusion laboratories. Furthermore, it may be institutional practice to prime the ECMO circuit with uncrossmatched blood during emergent cannulation as was done in Case 1. This could result in hemolytic transfusion reactions, which can be life-threatening. Thus, all transfusion decisions should be guided by the expertise of hematology and transfusion medicine specialists at the onset of hospital course and careful monitoring should be implemented. Consideration of thrombotic risks in SCD patients and the need for anticoagulation, particularly in VA ECMO, is also warranted. Exposure of blood to large non-biologic surface areas in the ECMO circuit can be pro-thrombotic. While centers, including ours, successfully run ECMO circuits without anticoagulation, this is ideally restricted to VV modalities where thrombosis and embolization may not be as dire as when blood is returned to the arterial circulation. Thus, the need anticoagulation for or the inability to safely initiate it as was the case with two patients in this series (Cases 1 and 3) can contribute to additional risk.
The outcomes for SCD patients placed on VA ECMO in this case series and in the literature are strikingly poor. Each of the patients described here experienced a complicated ECMO course, multiorgan failure, and none of them survived. Given the paucity of available reports on VA ECMO in SCD, the exact role for VA ECMO in adult SCD patients remains unknown. Thus, further study is needed to define the utility of VA ECMO in this population, identify measures that can be taken to avoid therapy complications and improve outcomes. For example, if time allows careful exchange transfusion of allo-antigen-matched blood with inotropic support prior to initiation of mechanical cardiovascular support could be attempted to potentially increase the chance of an improved outcome with VA ECMO therapy.
Conclusions
An in-depth understanding of SCD pathophysiology, identification of etiologies driving cardiovascular decline, and careful contemplation of potential complications that may be experienced with VA ECMO are essential when considering this mechanical circulatory support modality. The outcomes of patients with SCD managed with VA ECMO appear poor based on scant available literature and further study is critical to enhance our understanding of this therapy in SCD.
Supplemental Material
sj-docx-1-jic-10.1177_08850666241260605 - Supplemental material for Venoarterial Extracorporeal Membrane Oxygenation Therapy in Patients with Sickle Cell Disease: Case Series and Review for Intensive Care Physicians
Supplemental material, sj-docx-1-jic-10.1177_08850666241260605 for Venoarterial Extracorporeal Membrane Oxygenation Therapy in Patients with Sickle Cell Disease: Case Series and Review for Intensive Care Physicians by Alison Grazioli, Joseph Rabin, Raymond P. Rector, Zhongjun J. Wu, Allen P. Burke, Nima Sharifai, Aakash Shah, Bradley S. Taylor and Mark T. Gladwin in Journal of Intensive Care Medicine
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Footnotes
Acknowledgements
Dr Mark Gladwin receives research support from NIH grants R01HL098032, R01HL125886, UH3HL143192, the Department of Defense, and Globin Solutions, Inc. He previously received research support for Baer Corp.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
