Abstract
Background
Antibiotics are fundamental to the management of critically ill adults with severe infection. 1 Adults critically ill from infection frequently experience organ dysfunction, such as acute kidney injury (AKI)2,3 and acute brain dysfunction manifesting as delirium and coma. 4 Recent observational studies have suggested an association between the concurrent administration of vancomycin and piperacillin-tazobactam and development of AKI,5–7 although results among critically ill adults have been conflicting.8–17 These prior studies have not addressed the concern that the most common alternative to piperacillin-tazobactam, antipseudomonal cephalosporins, may cause neurotoxicity which manifests on a spectrum of findings including coma, delirium, myoclonus, and seizures.18–27 Neurotoxicity from cephalosporins has been reported to develop even with appropriate dosing and in patients with normal renal function.28–32
Since the organism causing an infection is rarely known at the time of initial presentation, empiric broad-spectrum antibiotics are commonly prescribed for patients with risk factors for drug-resistant organisms. In clinical practice, this often involves administration of an antibiotic covering resistant gram-positive organisms (eg, vancomycin) and an antibiotic targeting Pseudomonas and other resistant gram-negative organisms (eg, piperacillin-tazobactam or an antipseudomonal cephalosporin such as cefepime or ceftazidime). Given the safety concerns with these antibiotics, additional research is urgently needed to inform the optimal choice of empiric, anti-pseudomonal antibiotic among critically ill adults being treated for infection.
Methods
Patient Population and Antibiotic Receipt
We conducted a secondary analysis of data from the Isotonic Solutions and Major Adverse Renal Events Trial (SMART). SMART was a prospective, pragmatic, unblinded, cluster-randomized, multiple-crossover trial comparing balanced crystalloids versus saline for intravenous fluid administration among critically ill adults admitted to 5 intensive care units (ICUs) at Vanderbilt University Medical Center (VUMC) between June 1, 2015, and April 30, 2017. 33 The administration of antimicrobials during the SMART trial was determined by treating clinicians without guidance from the institution.
This secondary analysis was approved by the Vanderbilt Institutional Review Board (IRB) with waiver of informed consent (IRB number, 201690). This analysis included patients from the SMART trial dataset who had received either piperacillin-tazobactam or an anti-pseudomonal cephalosporin within 24 h of admission to the ICU. Our institution uses an extended infusion of piperacillin-tazobactam and our ICU multidisciplinary rounds include a clinical pharmacist who assists in the dosing of all medications including antibiotics. Though our institution does not offer therapeutic drug monitoring for penicillins or cephalosporins, all vancomycin orders are accompanied by a mandatory pharmacy consultation. The 2 anti-pseudomonal cephalosporins on the health system formulary during the trial were cefepime and ceftazidime/ceftazidime-avibactam. This analysis compared patients who received piperacillin-tazobactam to patients who received anti-pseudomonal cephalosporins. Patients who received both piperacillin-tazobactam and an anti-pseudomonal cephalosporin during their ICU admission were analyzed according to the antibiotic they received first.
Medications With Renal and Neurologic Impact
To assess for the effect of the coadministration of medications that may impact renal and neurologic function, the electronic health record was queried for the presence of the following medications within 24 h of receipt of qualifying anti-pseudomonal antibiotics. Medications that may impact renal function were defined as intravenous (IV) contrast, angiotensin converting enzyme inhibitor or angiotensin receptor blocker, oral or IV nonsteroidal anti-inflammatory drugs (not including aspirin), trimethoprim-sulfamethoxazole, IV acyclovir, IV Amphotericin B, loop and thiazide diuretics, IV aminoglycosides, sodium-glucose CoTransporter-2 inhibitors. Medications that may impact neurologic function included benzodiazepines, barbiturates, opiate pain medications, non-opiate pain medications (ie, gabapentin, pregabalin, cyclobenzaprine), anti-psychotics, propofol, dexmedetomidine, ketamine, and oral or IV corticosteroids.
Outcomes
We aimed to assess both potential adverse renal and neurologic effects of piperacillin-tazobactam and anti-pseudomonal cephalosporins. Our renal outcome was Major Adverse Kidney Events within 30 days (MAKE30). MAKE30 is a composite measure of death, new receipt of renal replacement therapy (RRT), and persistent renal dysfunction, defined as final serum creatinine value before hospital discharge (censored at 30 days after enrollment) that was ≥200% of the baseline creatinine value. Assessment of all components of this outcome was censored at hospital discharge. MAKE30 was the primary outcome for the original SMART trial and is (1) validated for electronic assessment, 34 (2) patient centered, 35 and (3) designed to capture clinically relevant renal dysfunction rather than short-term alterations in urine output or plasma creatine. Patients who had received RRT prior to ICU admission were only eligible for the death component of MAKE30.
Our neurologic outcome was the number of days alive and free of delirium and coma between ICU admission and 28 days following ICU admission. This outcome was defined as the number of days from the last day on which the patient experienced delirium, as assessed by a positive Confusion Assessment Method for the ICU 36 (CAM-ICU), or coma, as assessed by a score of −4 or −5 on Richmond Agitation and Sedation Scale 37 (RASS) until 28 days, censored at hospital discharge. Patients who died prior to 28 days received a value of 0. This outcome (1) is validated for electronic assessment using data routinely collected and charted in the electronic medical record in the trial setting, 38 (2) addresses the competing risk of death, and (3) has been used in prior trials of ICU interventions targeting the treatment or prevention of delirium as a clinically-relevant measure of neurologic function during critical illness.39,40 CAM-ICU and RASS were not assessed in the original SMART study and were extracted from the electronic health record for this study. At our institution, these assessments are collected once per 12 h nursing shift on all ICU patients.
Secondary outcomes included the development of new receipt of renal-replacement therapy, persistent renal dysfunction, the occurrence of death or RRT, and days alive and free of RRT, days alive and free of vasopressors, days alive and free of mechanical ventilation, and days alive and free of ICU admission at 28 days.
Statistical Considerations
To examine the relationship between initial antibiotic received and MAKE30, we fit multivariable logistic regression with pre-specified baseline co-variates of age, sex, probability of in-hospital death as calculated by University HealthSystem Consortium (UHC) expected mortality, 41 baseline creatinine, type of ICU, source of admission to the ICU, receipt of mechanical ventilation within 24 h of admission, receipt of vasopressors within 24 h of admission, receipt of RRT prior to admission, presence of AKI on ICU admission, presence of chronic kidney disease, and presence of sepsis. Presence of AKI on ICU admission was defined by the Kidney Disease: Improving Global Outcomes (KDIGO) creatinine criteria. 42
To examine the relationship between initial antibiotic received and the number of days alive and free of delirium and coma at 28 days, which is a non-parametrically distributed continuous variable, we fit a multivariable proportional odds regression model with pre-specified baseline co-variates of age, sex, UHC expected mortality, 43 type of ICU, source of admission to the ICU, receipt of mechanical ventilation within 24 h of admission, receipt of vasopressors within 24 h of admission, presence of coma on admission, receipt of RRT prior to admission, presence of AKI on ICU admission, presence of chronic kidney disease, and presence of sepsis. For both models, variables were selected using clinical knowledge and results from prior studies.
To understand whether the effect of antibiotic choice differed based on patients’ baseline characteristics, we assessed for effect modification by repeating the above multivariable analyses and including an interaction term between antibiotic group and the following pre-specified potential effect modifiers: prior receipt of RRT; presence of AKI at ICU admission; chronic kidney disease; coma at ICU admission; age; sex; body mass index (BMI); source of admission; and antibiotic duration.
To account for the possibility of indication bias in the choice between piperacillin-tazobactam and anti-pseudomonal cephalosporins, we performed a sensitivity analysis adjusting for the propensity to receive piperacillin-tazobactam. A propensity score for the outcome of receipt of piperacillin-tazobactam was calculated using a logistic regression model including all available baseline variables. The propensity score for each patient was then included as a covariate in multivariable modeling for the outcomes of MAKE30 and the number of days alive and free of delirium and coma. Additional sensitivity analyses were performed adding the baseline receipt of vancomycin and medications that may impact renal and neurologic function to their respective models.
Additional analyses included comparison of secondary outcomes between groups using multivariable modeling with the same baseline covariates used in the primary analyses.
As a secondary analysis of an existing dataset from a completed clinical trial, the available sample size was fixed. To avoid overfitting in multivariable modeling, simulation studies suggest at least 10 to 15 outcome events per degree of freedom. 44 With a fixed sample size of 3199 patients, and a fixed number of MAKE30 events of 933, we limited the number of covariates, interactions, and nonlinear terms in our regression modeling to fewer than 60 degrees of freedom. Two-sided P-values <.05 were considered significant. As a secondary analysis, all findings were considered hypothesis generating and corrections for multiple testing were not performed. All analyses were performed using R version 3.4.1 (2017, Vienna, Austria).
Results
Patients and Antibiotic Receipt
Among the 15 802 patients in the SMART dataset, 3199 (20.2%) received either piperacillin-tazobactam or an antipseudomonal cephalosporin within 24 h of ICU admission and were included in this analysis. Of these, 2375 patients (74.2%) were in the piperacillin-tazobactam group and 824 patients (25.8%) were in the anti-pseudomonal cephalosporin group. Among patients in the antipseudomonal cephalosporin group, 63% received cefepime and 33% received ceftazidime with 4% receiving both. The number of patients who ever received both piperacillin-tazobactam and an anti-pseudomonal cephalosporin during the hospitalization was 127 (4%). Baseline characteristics are displayed in Table 1.
Baseline Characteristics by Initial Antibiotic Received.
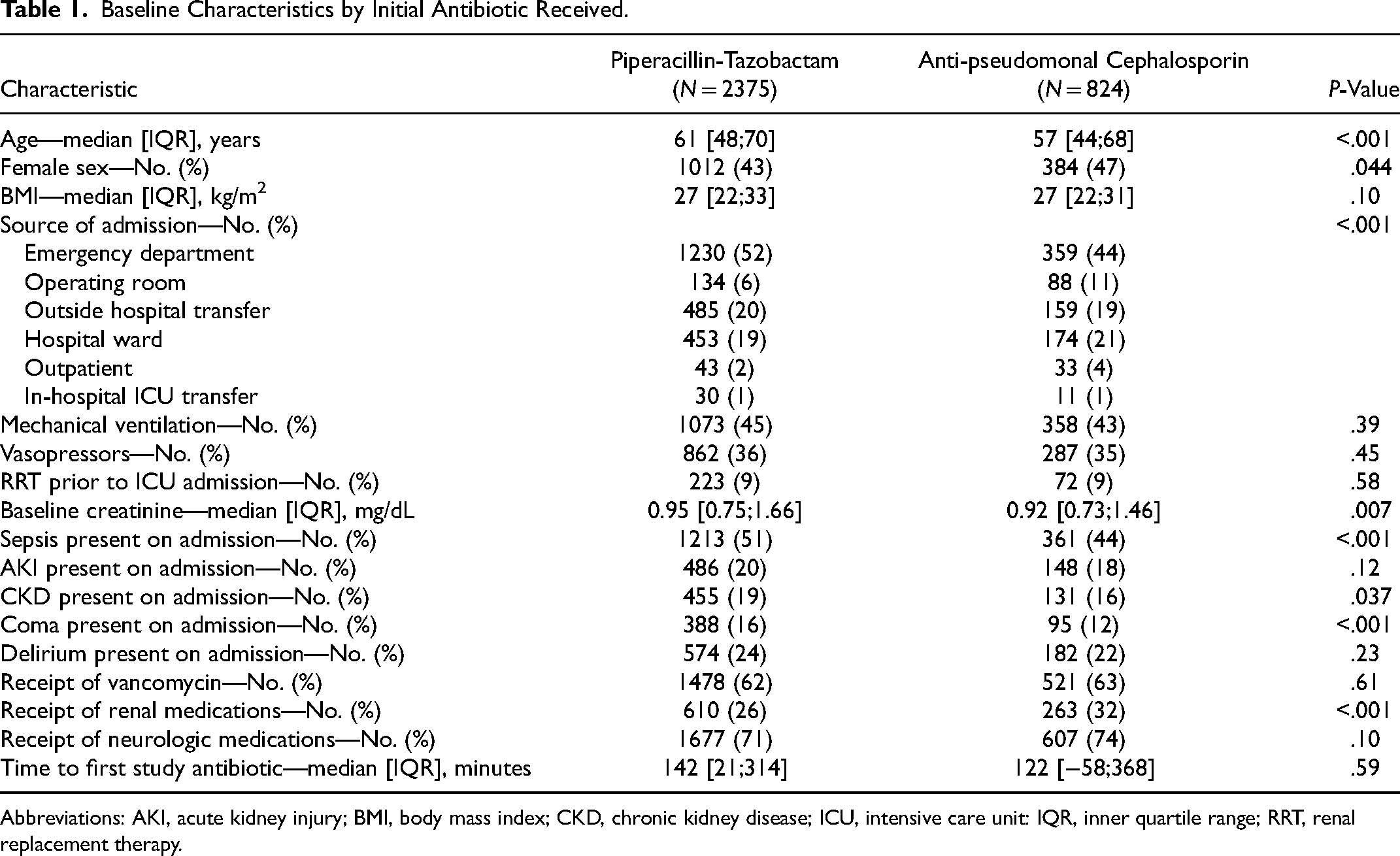
Abbreviations: AKI, acute kidney injury; BMI, body mass index; CKD, chronic kidney disease; ICU, intensive care unit: IQR, inner quartile range; RRT, renal replacement therapy.
Overall, the median duration of anti-pseudomonal antibiotics was 2.6 days (IQR, 1.1 to 5.6). The median duration of piperacillin-tazobactam was 2.3 days (IQR, 1.0-4.7), the median duration of cefepime was 2.5 days (IQR, 0.7 to 5.2), and the median duration of ceftazidime was 2.2 days (IQR, 0.9 to 4.7). A total of 2768 patients (86.5%) received vancomycin, and the median duration of vancomycin receipt was 2.1 days (IQR, 0.8 to 4.9).
Primary Analyses
In multivariable logistic regression adjusting for pre-specified baseline covariates, the odds of MAKE30 did not differ between the groups (odds ratio (OR), 1.03; 95% CI, 0.83-1.27; P = .80; C-statistic for model 0.82) (Tables 2 and 3). Compared with receipt of an anti-pseudomonal cephalosporin, receipt of piperacillin-tazobactam was not associated with a higher risk of MAKE30 in any of the pre-specified subgroups (Figure 1, Figure S1).
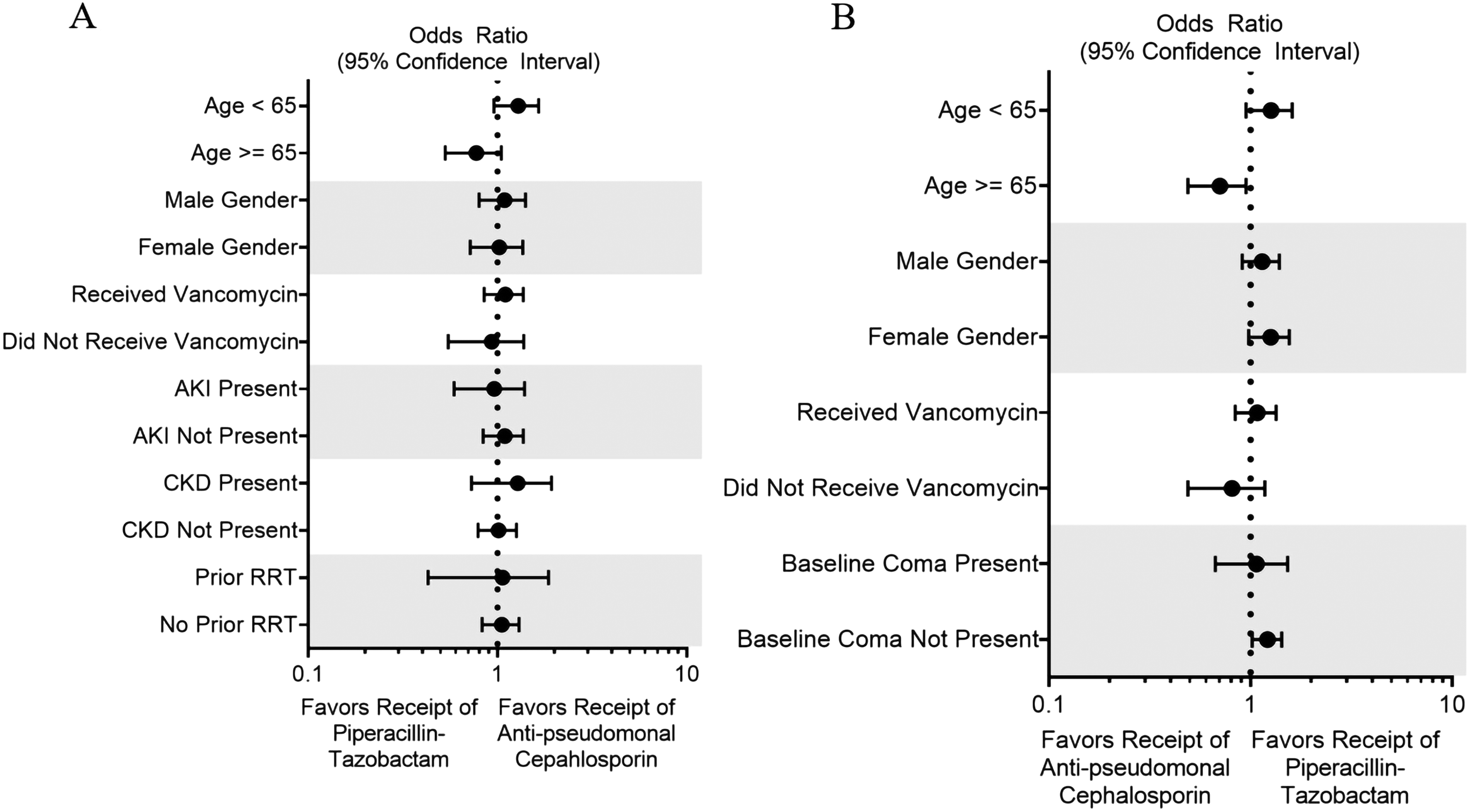
Subgroup analyses. Displayed are the odds of major adverse kidney events within 30 days (Panel A) and days alive and free of delirium and come by 28 days (Panel B) for patients in the piperacillin-tazobactam group compared with patients in the anti-pseudomonal cephalosporin group. The odds ratio and 95% confidence intervals are after adjustment for pre-specified baseline covariates.
Outcomes by Initial Antibiotic Received.
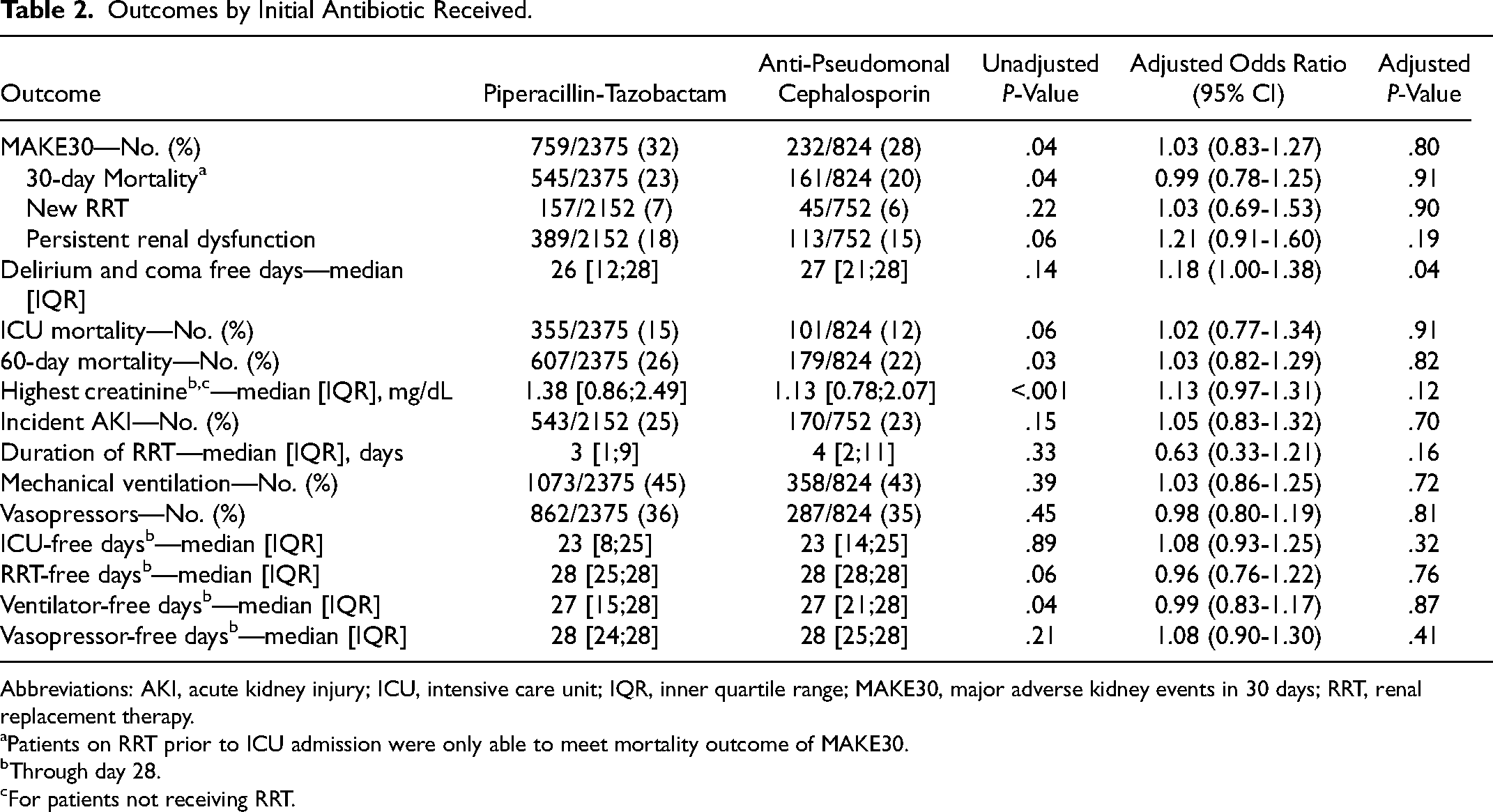
Abbreviations: AKI, acute kidney injury; ICU, intensive care unit; IQR, inner quartile range; MAKE30, major adverse kidney events in 30 days; RRT, renal replacement therapy.
Patients on RRT prior to ICU admission were only able to meet mortality outcome of MAKE30.
Through day 28.
For patients not receiving RRT.
Adjusted Models for MAKE30 and Delirium and Coma-Free Days.
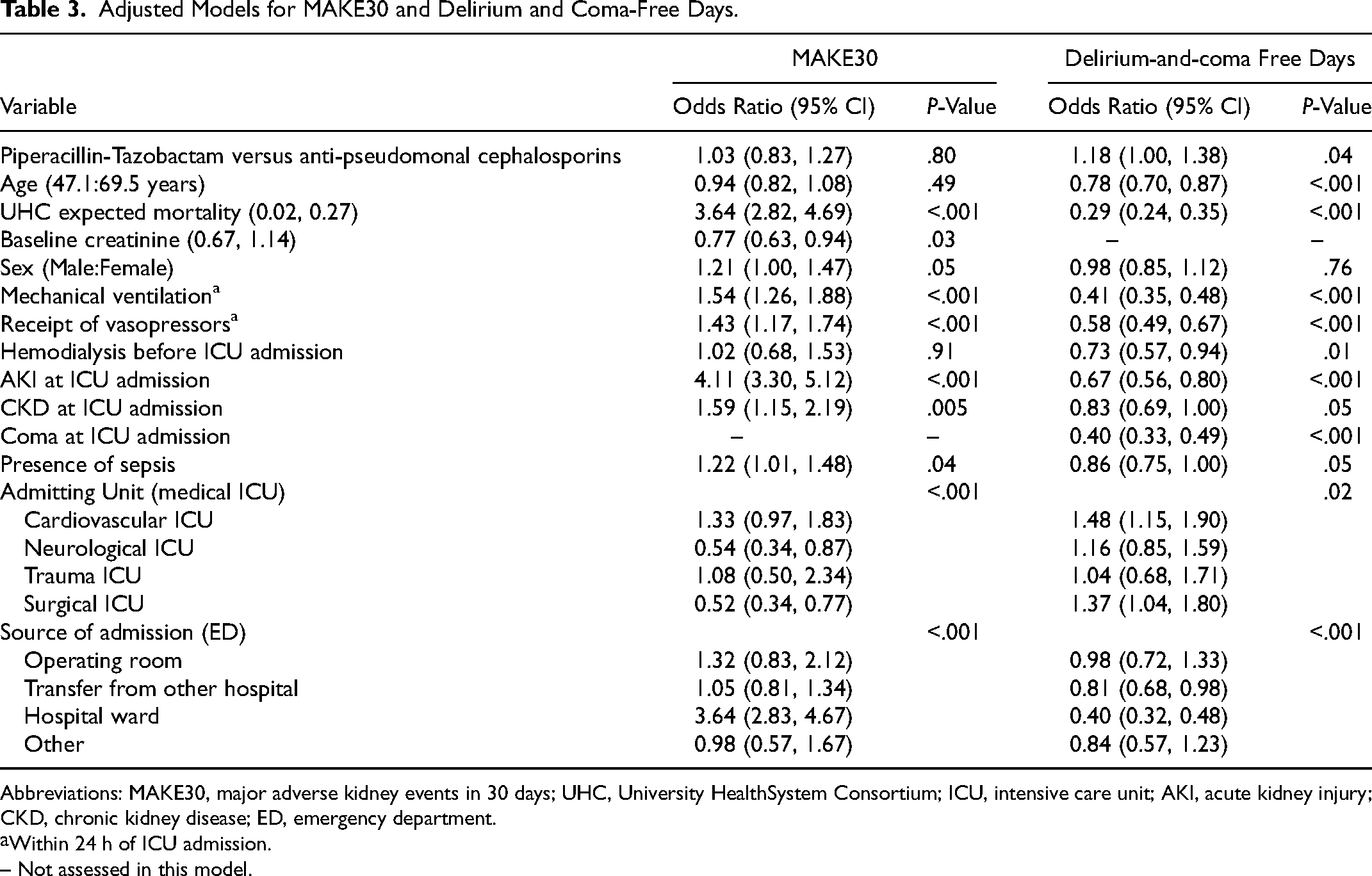
Abbreviations: MAKE30, major adverse kidney events in 30 days; UHC, University HealthSystem Consortium; ICU, intensive care unit; AKI, acute kidney injury; CKD, chronic kidney disease; ED, emergency department.
Within 24 h of ICU admission.
– Not assessed in this model.
In the multivariable proportional odds model adjusting for pre-specified baseline covariates, the piperacillin-tazobactam group experienced a greater number of days alive and free from delirium and coma (OR 1.18; 95% CI, 1.00 to 1.38; P = .04; C-statistic for model 0.77) (Tables 2 and 3). Compared with receipt of an anti-pseudomonal cephalosporin, receipt of piperacillin-tazobactam appeared to be associated with a greater number of days alive and free of delirium and coma among the subgroups of patients with no baseline coma (Figure 1).
Propensity Score Analysis
A propensity score analysis to account for indication bias demonstrated a similar distribution of between patients receiving piperacillin-tazobactam and receiving an anti-pseudomonal cephalosporin (Figure S2). Receipt of piperacillin-tazobactam rather than an antipseudomonal cephalosporin was associated with older age, male sex, admission ICU (medical compared to cardiovascular, neurologic, trauma, or surgical), admission location (emergency department compared to operating room, outside facility, hospital ward, or other), and presence of coma at enrollment, but was not associated with BMI, receipt of mechanical ventilation or vasopressors, baseline CKD, prior receipt of RRT, or presence of sepsis (Table S1).
Sensitivity Analyses
In a sensitivity analysis adjusting for the propensity score, the odds of experiencing MAKE30 did not differ between the piperacillin-tazobactam group and the anti-pseudomonal cephalosporin group (OR 1.01; 95% CI, 0.81 to 1.25) but the number of days alive and free of delirium and coma was greater in the piperacillin-tazobactam group than in the anti-pseudomonal cephalosporin group (OR 1.19; 95% CI, 1.02 to 1.40) (Table S2).
Because cefepime has been more frequently reported with neurotoxicity, a sensitivity analysis excluding patients with ceftazidime was performed which also demonstrated more days alive and free of delirium and coma among patients who received piperacillin-tazobactam (OR 1.26; 95% CI 1.04 to 1.52) (Table S3).
Because of concern of receipt of other medications which impact renal or neurologic function, a sensitivity analysis adjusting for baseline receipt of these variables were performed. There remained no impact on antibiotic choice on MAKE30 (OR 1.03, 95% CI 0.83 to 1.28). In this analysis there was no difference in days alive and free of delirium and coma (OR 1.07, 95% CI 0.83 to 1.37) (Table S4).
Additional Clinical Outcomes
In multivariable analysis adjusting for pre-specified baseline covariates, the piperacillin-tazobactam group did not differ significantly from the anti-pseudomonal cephalosporin group with regard to the number of RRT-free days (OR 0.96; 95% CI, 0.76 to 1.22), vasopressor-free days (OR 1.08; 95% CI, 0.90 to 1.30), ventilator-free days (OR 0.99; 95% CI, 0.83 to 1.17), or ICU-free days (OR 1.08; 95% CI, 0.93 to 1.25) (Table 2).
Discussion
In this secondary analysis of data from a large clinical trial, the initial receipt of piperacillin-tazobactam compared to anti-pseudomonal cephalosporins was not associated with an increased risk of major adverse kidney events or a greater number of days alive and free from delirium and coma.
Prior observational studies evaluating the association between receipt of piperacillin-tazobactam and AKI have provided conflicting results. Many of these studies have not accounted for confounding by indication and severity of illness or have limited their analyses to laboratory measurements (eg, highest creatinine) without account for the competing risks of new RRT and death. While several studies have suggested that the combination of vancomycin and piperacillin-tazobactam may be associated with AKI, these analyses may be particularly susceptible to confounding by indication bias.5–7 Further, studies including critically ill populations have provided mixed results with some showing no increased risk of AKI compared to other beta lactams.9,11 In contrast to some prior studies, we defined the exposure based on the receipt of piperacillin-tazobactam, regardless of receipt of vancomycin, as patients who receive both antibiotics are likely to be different than patients receiving monotherapy with an alternative antibiotic. However, 87% of patients in the piperacillin-tazobactam group received vancomycin, which was similar to the 86% of patients in the anti-pseudomonal cephalosporins group who received vancomycin. The differences between our results and those of prior studies may be explained by the ability to robustly adjust for confounding by indication bias using the granular data collected as part of a prospective randomized trial, examination of a longer-term more clinically-focused outcomes compared with changes in creatinine, or other differences in patient population.
If piperacillin-tazobactam were to cause AKI in some patients, that risk would have to be weighed against the potential adverse effects of alternative therapies, a factor not examined in prior studies. Anti-pseudomonal cephalosporins are the most commonly used alternative to piperacillin-tazobactam and have long been hypothesized to cause neurotoxicity in certain contexts. The mechanism by which cefepime and ceftazidime cause direct neurotoxicity is unclear and evaluation of cefepime neurotoxicity is difficult given the absence of a definitive diagnostic test. Given the absence of a gold-standard diagnostic test and poorly understood pathophysiology of the syndrome, we chose to use a measure that captures a broad range of patient-oriented neurologic outcomes, the number of days alive and free from delirium and coma. Our observational analysis remains susceptible to residual confounding, as evidenced by lack of congruence between our primary model and sensitivity analysis accounting for the receipt of medications that may impact neurologic function. This highlights the need for further research.
Our study had several strengths. First, as a secondary analysis of a clinical trial focused on the development of AKI among critically ill adults, we were able to use granular, prospectively collected data to assess the occurrence of the patient-centered MAKE30 outcome, accounting for AKI and the competing risks of new RRT and death. Second, we evaluated both AKI and neurological dysfunction, which represent potential competing considerations for clinicians choosing between piperacillin-tazobactam and the most common alternative, the antipseudomonal cephalosporins. Third, we used multivariable modeling and propensity score analysis to attempt to account for measured confounders.
Our study also has a number of limitations. The single-center nature of this cohort limits its generalizability. We combined cefepime and ceftazidime into a single group for analysis, whereas the risk for neurotoxicity may differ between the two. Second, we also did not assess for efficacy of antibiotics but we noticed no difference in our clinical outcomes of mortality or support-free days. Third, seizure and myoclonus, known sequalae of cephalosporin-induced neurotoxicity, were not assessed because of lack of reliable evaluation methods. Since there is no gold-standard measurement of neurotoxicity from cefepime and ceftazidime, we elected to use delirium and coma free days as a validated assessment of neurologic dysfunction in critically ill patients. However, we recognize that the sensitivity and specificity of delirium and coma free days for this outcome is unknown. Fourth, in this study the median duration of antibiotics was 2.2-2.6 days which is more clinically consistent with shorter-term empiric broad spectrum antibiotic use compared with definitive, long-term therapeutic courses. There are studies which suggest that shorter durations of antibiotics are not associated with the same risk of adverse renal and neurologic outcomes.16,27 However, shorter durations of broad-spectrum antibiotics are common when starting empiric antibiotics relevant to critically ill adults with infection. Fifth, because our outcomes examined the number of days alive and free of delirium and coma from enrollment until 28 days, delirium and coma that occurred prior to receipt of the antibiotic could contribute to the outcome; the short overall duration of antibiotic exposure may increase the risk of the observed findings being due to residual confounding. Lastly, our results cannot rule out the possibility of residual confounding, even after attempting to account for severity of illness and indication bias in propensity score analysis and multivariable modeling.
Conclusions
In conclusion, this secondary analysis of a large clinical trial found that, among critically ill adults, receipt of piperacillin-tazobactam was not associated with a greater risk of major adverse kidney events or a greater number of days alive and free of delirium and coma. Prospective, randomized trials are urgently needed to rigorously define the effectiveness and safety of these commonly used antimicrobials and to inform the optimal choice of empiric anti-pseudomonal antibiotics in clinical care.
Supplemental Material
sj-docx-1-jic-10.1177_08850666231184177 - Supplemental material for Piperacillin-Tazobactam Versus Anti-Pseudomonal Cephalosporins and Renal and Neurologic Outcomes in Critically Ill Adults: A Secondary Analysis of the SMART Trial
Supplemental material, sj-docx-1-jic-10.1177_08850666231184177 for Piperacillin-Tazobactam Versus Anti-Pseudomonal Cephalosporins and Renal and Neurologic Outcomes in Critically Ill Adults: A Secondary Analysis of the SMART Trial by Edward T. Qian, Li Wang, Joanna L. Stollings, Jonathan D. Casey, Todd W. Rice and Matthew W. Semler in Journal of Intensive Care Medicine
Footnotes
Author Contributions
Study concept and design: ETQ, JLS, JDC, TWR, and MWS. Acquisition of data: EQ, LW, MWS. Analysis and interpretation of data: LW, ETQ, JLS, JDC, MWS, and TWR. Drafting of the manuscript: ETQ. Critical revision of the manuscript for important intellectual content: JLS, JDC, TWR, and MWS. Statistical analysis: LW. Responsibility for integrity of work as a whole: ETQ, MWS.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: E.T.Q. was supported in part by the NIH (T32HL087738). J.D.C. was supported in part by the NIH (K12HL133117 and K23HL153584). T.W.R. was supported in part by the NIH. M.W.S. was supported in part by the NHLBI (K23HL143053). The funding institutions had no role in (1) conception, design, or conduct of the study, (2) collection, management, analysis, interpretation, or presentation of the data, or (3) preparation, review, or approval of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
