Abstract
Thrombosis and bleeding after implementation of an intermediate-dose prophylactic anticoagulation protocol in intensive care unit (ICU) patients with coronavirus disease 2019 (COVID-19): a multicenter screening study
Introduction
Coronavirus disease 2019 (COVID-19) pneumonia causes hypoxemic respiratory failure and multiorgan dysfunction and is associated with high mortality. In addition, hallmarks of severe COVID-19 pneumonia in patients admitted to the intensive care unit (ICU) include the development of a prothrombotic state 1 with pulmonary vascular thrombosis and deep vein thrombosis (DVT) despite standard-dose thromboprophylaxis.2–8 Several studies have noted a discrepancy between rates of pulmonary vascular thrombosis and DVT4,9,10 with only 42% of patients with pulmonary vascular thrombosis having concurrent DVT, 11 suggesting macrothrombi formation in situ, 12 in addition to thrombo-embolic events. A recent meta-analysis 13 suggested a rate of venous thromboembolism (VTE) of 27.9% in ICU patients with COVID-19 pneumonia. Thrombotic complications in the arterial circulation and clotting of renal replacement circuits 3 as well as intravascular access devices 14 have also been reported.
In response to the elevated risk of VTE in critically ill patients with COVID-19, current guidelines15,16 propose the use of enhanced prophylactic anticoagulation. These recommendations are not supported by data from the most recent randomized controlled trial showing no effect of intermediate-dose compared with standard-dose thromboprophylaxis on the incidence of VTE. 17 However, the lack of systematic screening for thrombotic events in that study, which carries a risk that the true VTE incidence was underestimated, is an obvious limitation. In fact, observational data from small screening studies6,8,18,19 suggest that a significant proportion of patients develop asymptomatic DVT, which appears associated with mortality. 20
The Surviving Sepsis Campaign guidelines 21 currently recommends against the use of therapeutic dose anticoagulation and emphasize the lack of evidence in using intermediate-dosing in critically ill COVID-19 patients. Given the disparate rates of VTE reported in the literature, different anticoagulation strategies between centers, and inconsistent use of systematic screening, the true rate of VTE and bleeding complications in patients receiving intermediate-dose thromboprophylaxis remains uncertain. Accordingly, using prospective Point-of-Care Ultrasound (POCUS) screening in addition to clinician-guided VTE diagnostics, we aimed to study the prevalence of VTE and bleeding in patients with critical COVID-19 exposed to intermediate-dose thromboprophylaxis.
Materials and Methods
The study conformed to the Declaration of Helsinki and was approved by the Swedish Ethical Review Authority (approval number 2020-02550). Informed consent was obtained from the patient or next of kin prior to enrollment.
Patient Selection
We conducted a prospective observational cohort study in patients admitted to 6 ICUs at the 2 Karolinska University Hospitals (Solna site and Huddinge site) in Stockholm, Sweden. We included adult (18 years of age or older) ICU patients with confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by polymerase chain reaction. We excluded patients who were discharged from the ICU before screening ultrasound assessment was possible. We also excluded patients who declined participation and patients from whom informed consent was not possible to obtain (from patient or next-of-kin). Finally, we excluded patients exposed to frequent prone positioning preventing screening ultrasound. Patients were screened for eligibility from June 21, 2020, through February 18, 2021, at the Solna site and from December 6, 2020, through February 18, 2021, at the Huddinge site. Eligibility was continuously assessed and examinations were performed when 1 of the 2 ultrasound examiners was available. As Karolinska University Hospital is a bi-center tertiary care facility composed of multiple critical care units, the ability to include patients at both sites simultaneously was limited. Patients were followed until April 12, 2021.
Local ICU Treatment Protocol
According to the local treatment protocol at the Karolinska University Hospital (implemented mid-April 2020), the following initial intermediate thromboprophylaxis doses were recommended for all COVID-19 patients treated in the ICU: 2500 IU of dalteparin twice daily (or 20 mg enoxaparin twice daily) for patients with a body weight less than 50 kg; 5000 IU of dalteparin twice daily (or 40 mg enoxaparin twice daily) for patients with a bodyweight of 50 to 90 kg; and 7500 IU of dalteparin twice daily (or 60 mg enoxaparin twice daily) for patients with body weight above 90 kg. In patients with suspected or confirmed VTE, the recommended dalteparin dose was 200 IU/kg per day (or 1 mg/kg enoxaparin twice daily). Alternatively, continuous heparin infusion was used. Prophylactic and therapeutic dalteparin doses were adjusted based on frequent analysis of anti-factor Xa trough levels (target 0.2-0.3 IU/mL for prophylactic dosing and 0.4-0.6 IU/mL for therapeutic dosing). When the anti-factor Xa concentration was outside the target range, the daily dalteparin dose was typically adjusted by 2500 to 5000 IU or more depending on the magnitude of deviation from the target. After dose-adjustment, anti-factor Xa was reassessed before administration of the fourth dose. Patients with kidney dysfunction, especially those treated with renal replacement therapy, were often exposed to more frequent anti-factor Xa monitoring. From June 2020, treatment with dexamethasone (6 mg/day) or betamethasone (6-8 mg/day) for 10 days was recommended for all ICU patients.
Local Medical Ward Protocol
For adult patients hospitalized with COVID-19 pneumonia outside the ICU the following standard low-dose thromboprophylaxis doses were recommended: dalteparin 2500 IU (or enoxaparin 20 mg) once daily for patients with a body weight of below 50 kg; dalteparin 5000 IU (or enoxaparin 40 mg) once daily for patients with a body weight of 50 to 90 kg and 7500 IU (or enoxaparin 60 mg) of dalteparin once daily for patients with a body weight above 90 kg. For patients who progressed to severe COVID-19 requiring intermediate care and for patients with any of the following risk factors, intermediate-dose prophylaxis was recommended: D-dimer >3 mg/l, fibrinogen >8 g/l, cancer, previous VTE, or known thrombophilia.
Screening Ultrasonography
All study subjects underwent 2-region compression ultrasound of the lower extremities including the common femoral (CFV), femoral (FV), and popliteal veins (PV) with the patient in the supine position. 22 A high-frequency (5-12 MHz) linear transducer was used with two-dimensional gray-scale venous visualization. Color Doppler was added to visualize blood flow. Compression ultrasonography was performed at the following sites bilaterally: the CFV cranial to the sapheno-femoral junction, the sapheno-femoral junction, the bifurcation of the CFV into the FV, the PV cranial to the trifurcation, and the PV at the level of trifurcation. Examinations were performed by 2 final-year medical students with POCUS training using Vivid S70 and Venue Go (GE Ultrasound, Horten, Norway) and lower extremity vein preset. Study images were acquired and archived for review. All ultrasound images were analyzed offline on a dedicated post-processing software (EchoPAC, GE Ultrasound) and reviewed by an experienced physician specialized in diagnostic ultrasound.
Data Collection
Medical records were reviewed for patient characteristics including pre-existing medical conditions, home medication, ICU treatments, radiological, physiological, and laboratory exam results, and outcome data. Computed tomography pulmonary angiography (CTPA) and laboratory exams were performed at the discretion of the treating physician.
Operational Definitions
Major bleeding was defined using the criteria guidelines from the International Society on Thrombosis and Haemostasis (ISTH) which includes fatal bleeding and/or symptomatic intracranial, retroperitoneal, intraspinal, intraocular, pericardial, intramuscular bleeding, and/or bleeding causing a drop in hemoglobin level of 20 g/L or more or leading to transfusion of 2 or more units of red blood cells. 23 Non-major bleeding was based on ISTH Scientific and Standardization Committee recommendation 24 of any bleeding that does not meet the ISTH criteria for major bleeding and requires medical intervention by a healthcare professional.
Outcomes
The primary outcome was VTE, defined as proximal lower-extremity DVT (detected on formal or screening ultrasound), pulmonary vascular thrombosis (detected by CTPA), and/or venous thrombosis detected at other sites. Secondary outcomes included arterial thromboembolism (including valvular thrombosis) and bleeding. The exploratory clinical outcomes included death until April 12, 2021.
Statistical Analysis
To provide prevalence data of thrombotic and hemorrhagic events after implementation of intermediate-dose VTE prophylaxis we chose to include a convenience sample of 100 patients. Categorical variables are presented as numbers and percentages. Continuous variables are presented as median with interquartile range (IQR). Predictive values for VTE were assessed by calculation of the area under the receiver operating characteristics (ROC) curve and presented with 95% confidence intervals. In exploratory analyses, we used crude logistic regression analyses to assess the association between baseline characteristics with a prevalence ≥10% and VTE. Odds ratios were considered statistically significant if the 95% confidence intervals did not contain 1.0. All statistical analyses were performed using Stata version 15.1 (StataCorp, College Station, USA).
Results
Patients
A total of 169 COVID-19 patients treated in the study ICUs were eligible for study inclusion. Overall, 54 of these patients were discharged to the medical ward, transferred to another hospital, or died before inclusion was possible. Of the remaining 115 patients, 6 patients were excluded due to declined participation by patients or relatives. Communication to gather consent from relatives was not possible in 3 patients and 6 patients were not eligible for inclusion due to frequent prone positioning. Therefore, we included 59% (100 of 169) of all eligible patients (Figure 1). Median time between ICU admission and inclusion was 5 (IQR, 3-12) days.
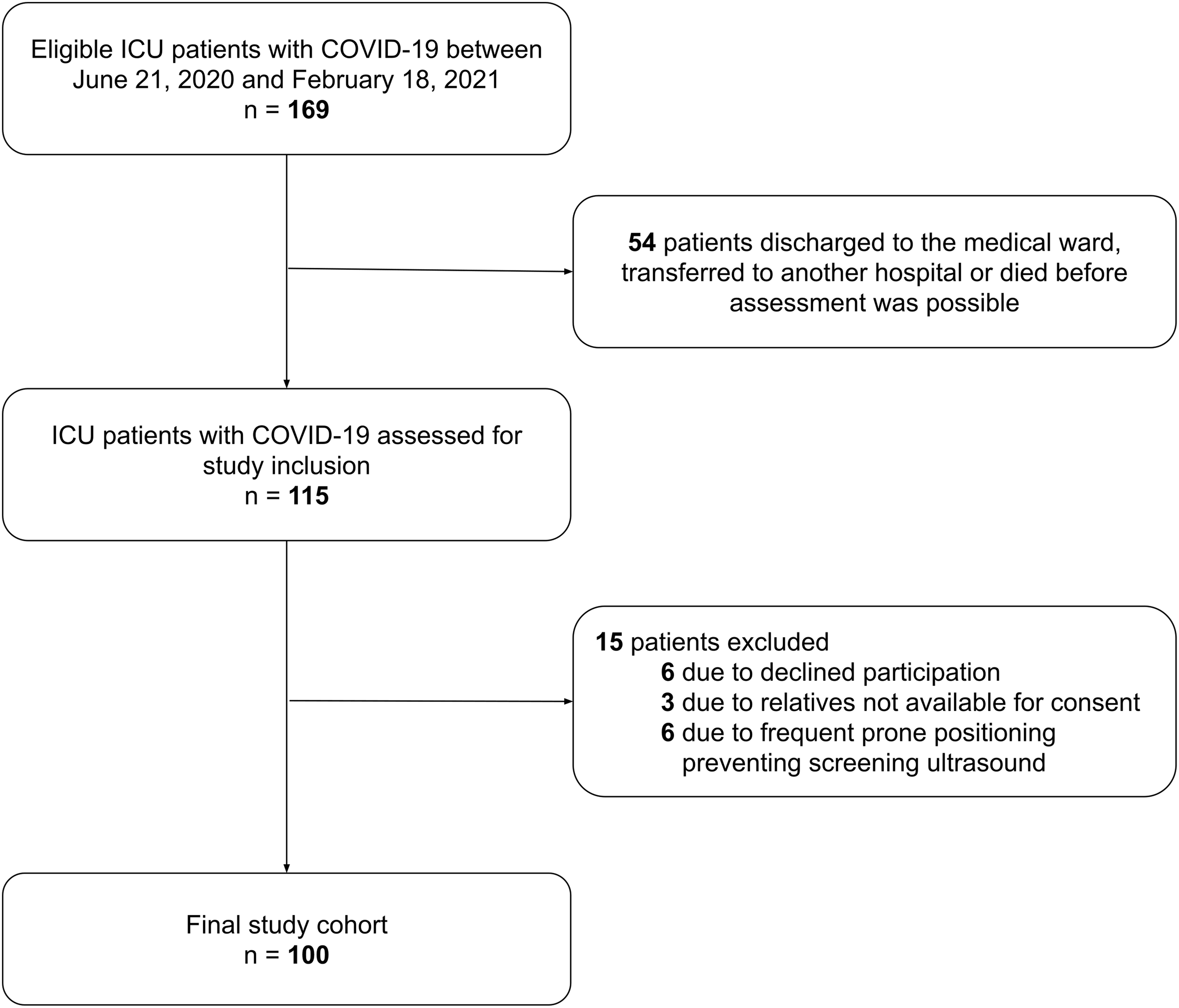
Patient selection.
Baseline characteristics of included patients are shown in Table 1. Median age was 68 (IQR, 57-73) years; 71% were men; and 46% had a BMI of 30 kg/m2 or above. Overall 88% had one or more co-existing conditions, with hypertension (58%), type 2 diabetes (34%), and immunodeficiency (22%) being the most common. Pre-existing thrombophilia was present in 3% of study patients, and 7% had a prior history of VTE. Pre-admission antithrombotic drug use was observed in 37% (26% were antiplatelet drug users and 12% were anticoagulant drug users).
Baseline Characteristics.

Abbreviations: BMI, body mass index; HFrEF, heart failure with reduced ejection fraction; HFpEF, heart failure with preserved ejection fraction; ICU, intensive care unit.
Data are n (%) or median (IQR).
Process of care
Process of care characteristics is summarized in Table 2 and Appendix A. At study inclusion (day of ultrasound screening), 95% received low molecular weight heparin (LMWH) with the majority (82.1%) receiving intermediate-dose prophylaxis. In addition, 97% received systemic corticosteroid therapy; 30% received remdesivir; and 23% received immunomodulator therapy with either tocilizumab, anakinra, or convalescent plasma. During ICU stay, 79% received mechanical ventilation for a median duration of 18 (IQR, 10-27) days; 16% received renal replacement therapy; and 2% received extracorporeal membrane oxygenation. The majority (86%) were exposed to intermittent prone positioning. Median ICU length of stay was 19 (IQR, 10-31) days.
Process of Care Characteristics.
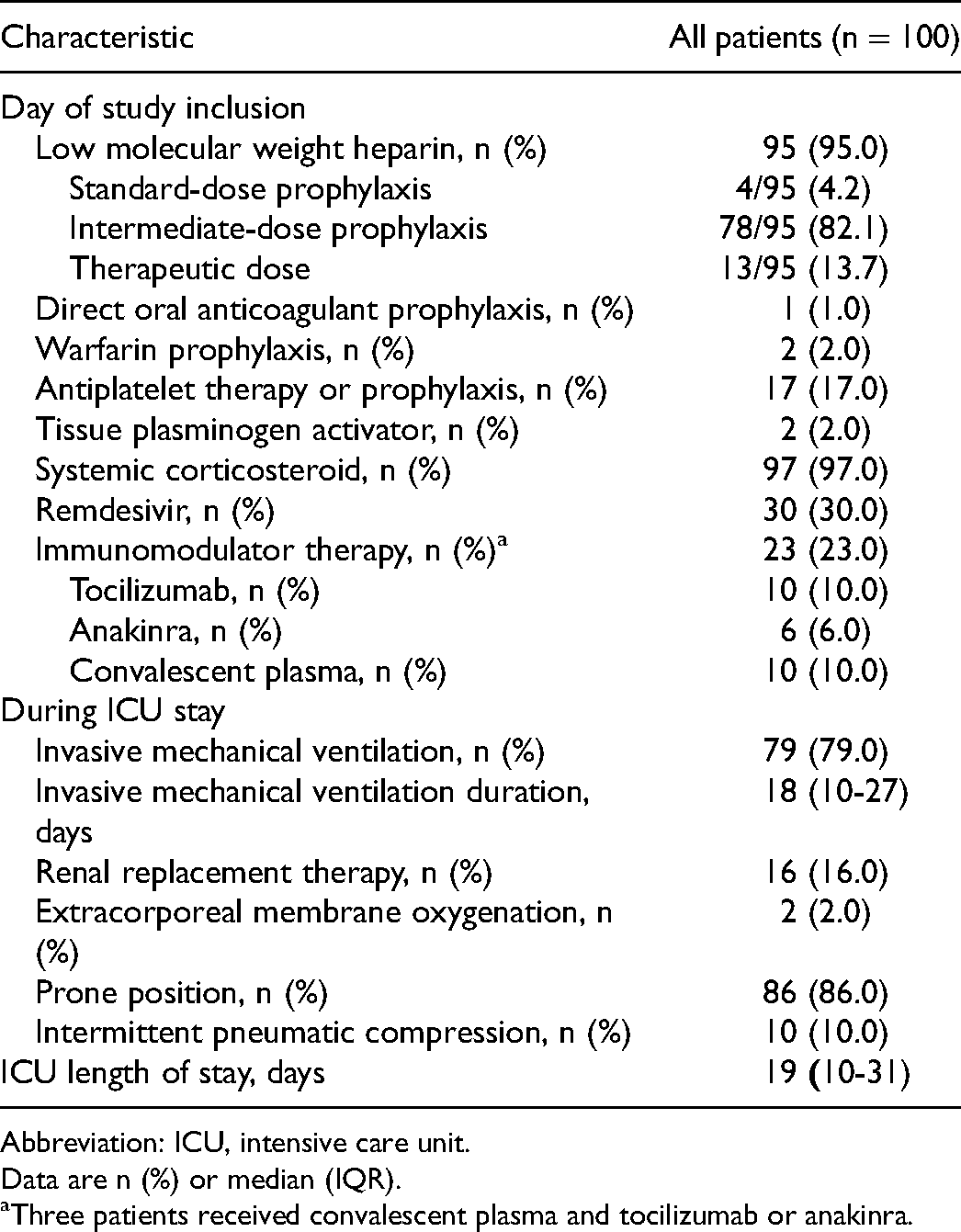
Abbreviation: ICU, intensive care unit.
Data are n (%) or median (IQR).
Three patients received convalescent plasma and tocilizumab or anakinra.
Outcomes
Outcome data is presented in Table 3. During a median follow-up of 120 (IQR, 89-134) days, 37% were diagnosed with VTE with the majority (78.4%) being diagnosed after arrival to the ICU. Overall, 20% had proximal lower-extremity DVT (19 of 20 were asymptomatic and detected on screening); 22% had pulmonary vascular thrombosis; and 4% had venous thrombosis at other sites (inferior vena cava thrombus, right atrial thrombus, or central venous catheter thrombus). Arterial thromboembolism occurred in 4% of patients. A total of 6% had both proximal lower-extremity DVT and pulmonary vascular thrombosis. One patient had a thrombus in the inferior vena cava and a right atrial thrombus. The same patient had no imaging evidence of lower extremity DVT or pulmonary vascular thrombosis and therefore only contributed to the total VTE rate. Among the 19 patients with asymptomatic DVT, 13 (68.4%) patients were treated with intermediate-dose LMWH, 1 (5.3%) patient was treated with standard prophylactic dose and 5 (26.3%) patients received therapeutic anticoagulation prior to ultrasound screening. Among the 37 patients that developed VTE, 62.2% received intermediate-dose prophylaxis prior to VTE diagnosis (see Appendix C). Details regarding characteristics of anti-factor Xa monitoring in no VTE and VTE patients are presented in Appendix D. We observed a high proportion of anti-factor Xa levels outside the prophylactic target range (0.2-0.3 IU/ml). For example, the median (IQR) proportion of values below the target range was 29% (0%-63%) in no VTE patients and 14% (0$-33%) before VTE diagnosis in VTE patients. In contrast, 33% (0%-67%) of values were within the therapeutic target range (0.4-0.6 IU/mL) before VTE diagnosis. Major and non-major bleeding occurred in 10% and 18% of patients, respectively. Detailed information regarding antithrombotic therapy prior to bleeding events is presented in Appendix E. As of April 12, 2021, 39 patients had died and 61 patients were discharged alive from ICU.
Outcomes.
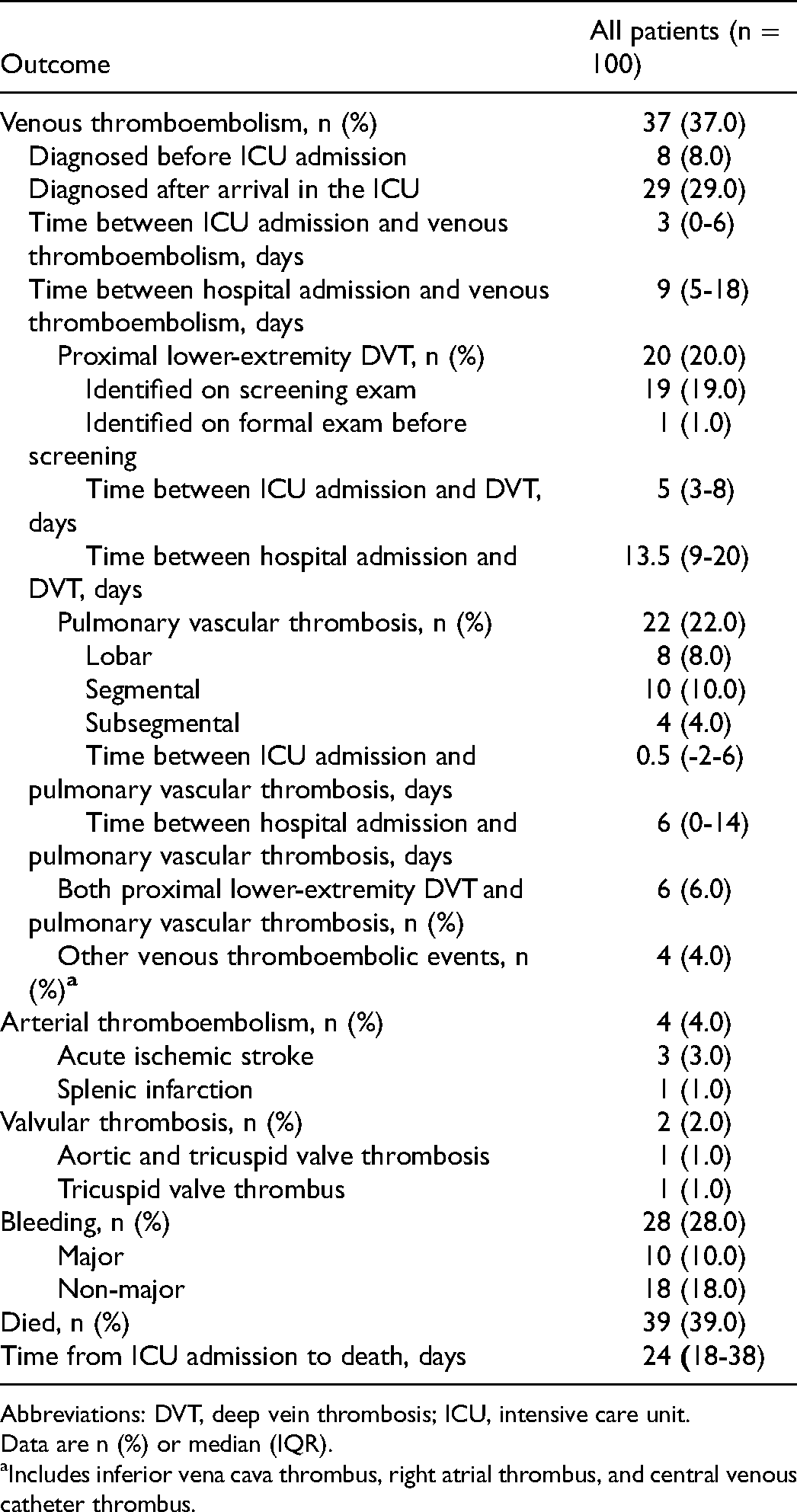
Abbreviations: DVT, deep vein thrombosis; ICU, intensive care unit.
Data are n (%) or median (IQR).
Includes inferior vena cava thrombus, right atrial thrombus, and central venous catheter thrombus.
The distribution of D-dimer values over time in patients with VTE and in those without VTE is shown in Figure 2. D-dimer on hospital and ICU admission predicted VTE with a ROC area of 0.64 (95% CI 0.52-0.76) and 0.65 (95% CI 0.53-0.78), respectively. Supplementary biochemical data is presented in Appendix B.
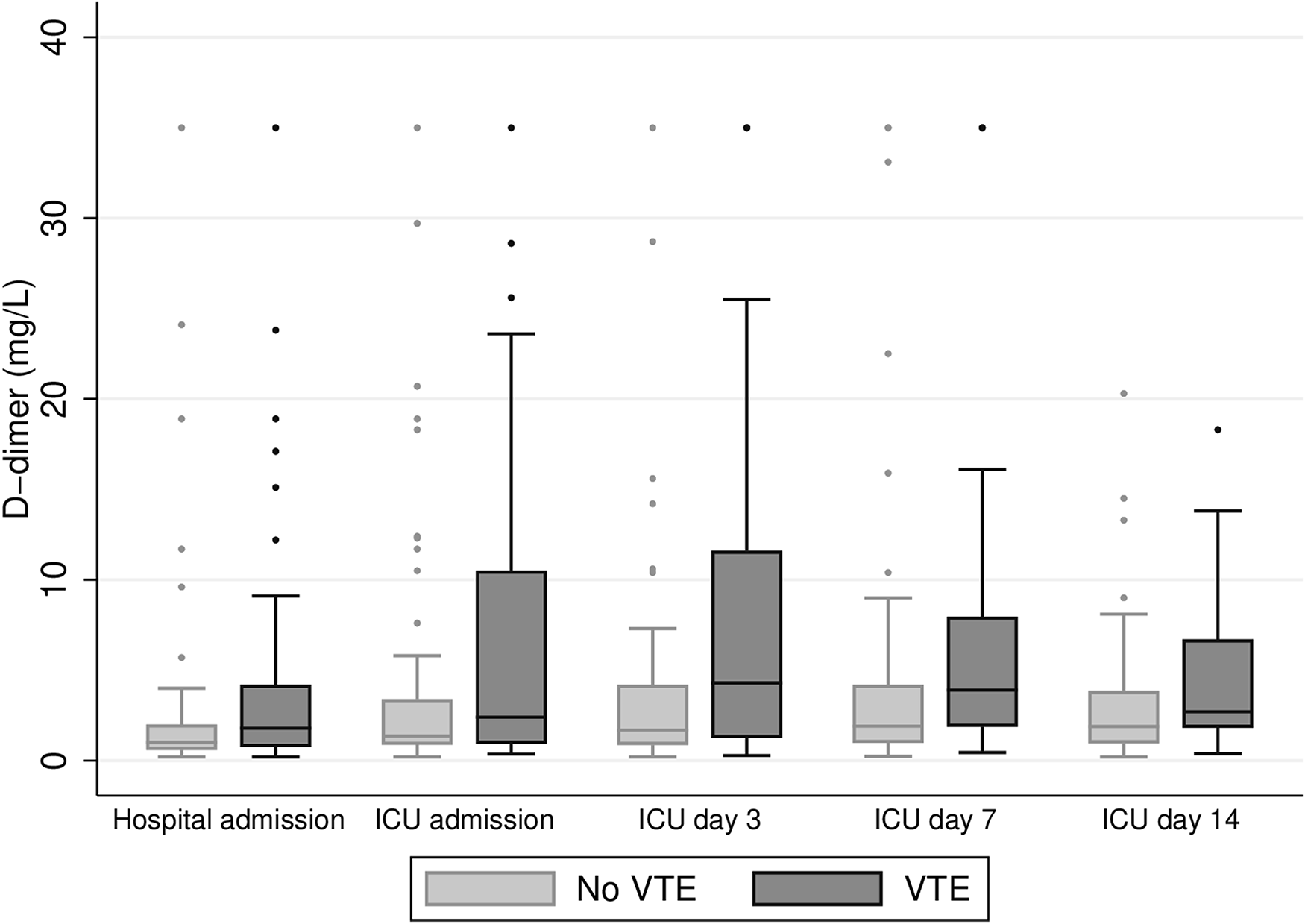
D-dimer levels in patients without VTE and in those with venous tromboembolism.
On univariate logistic regression analysis of 14 baseline characteristics, only pre-existing heart failure was significantly associated with VTE (unadjusted OR 4.67, 95% CI 1.13-19.34) (Figure 3). We observed no significant association between VTE and death (unadjusted OR 0.92, 95% CI 0.40-2.13).
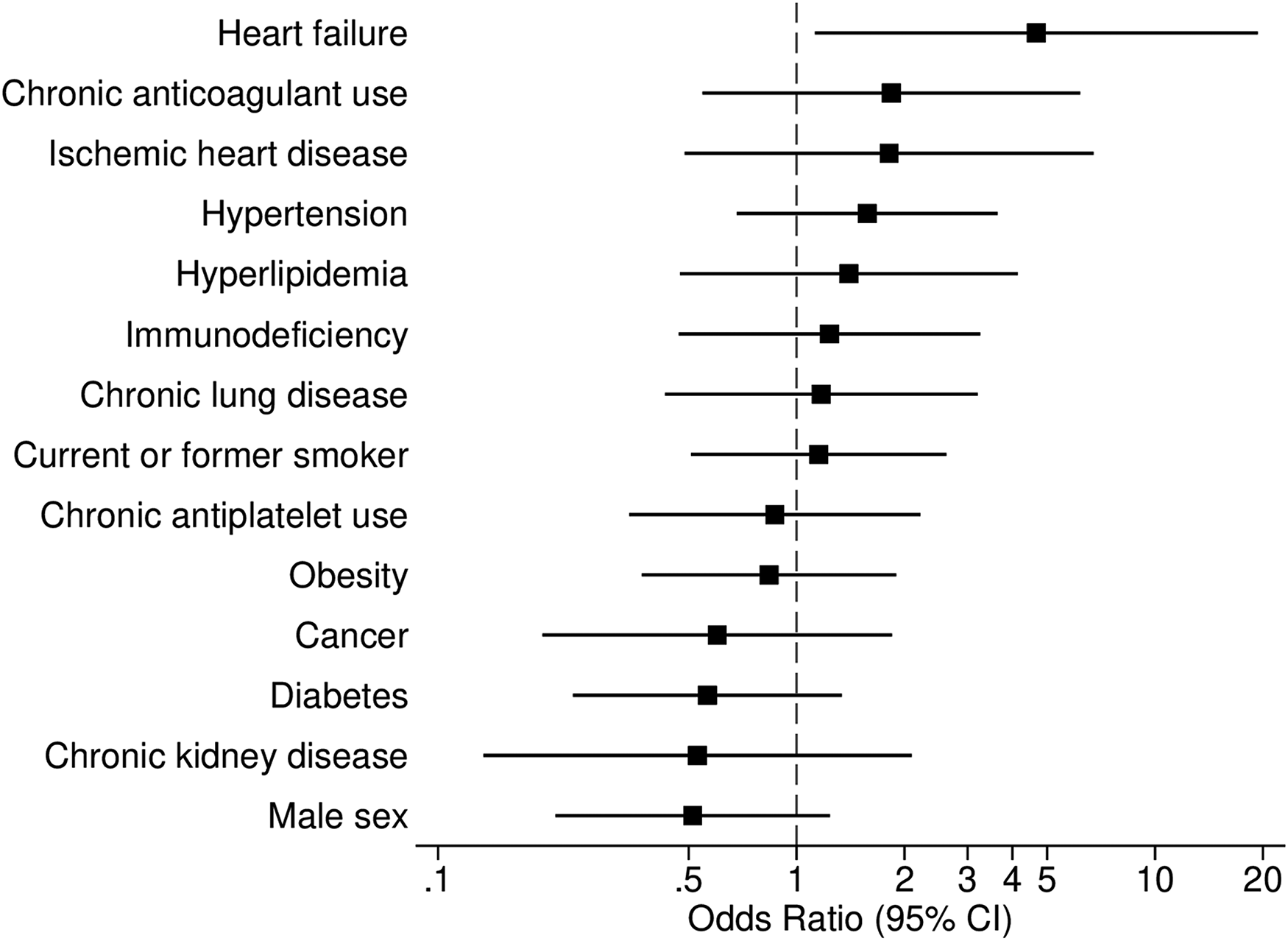
Univariate logistic regression analyses showing the associations between selected baseline characteristics and venous thromboembolism.
Discussion
Key Findings
We conducted a prospective multicenter screening study to assess the prevalence of VTE in 100 patients with critical COVID-19 after implementation of intermediate-dose prophylactic anticoagulation. We found VTE in 37% of our patients. Proximal lower-extremity DVT was diagnosed in 20% of patients, with the majority (19 of 20 patients) found on screening ultrasonography despite ongoing intermediate-dose or therapeutic-dose LMWH. Pulmonary vascular thrombosis and other venous thromboembolic events were found in 22% and 4% of patients, respectively. Finally, bleeding complications was observed in almost one-third of patients.
Comparison with Previous Studies
To the best of our knowledge, our study is the first screening assessment of VTE prevalence in critically ill COVID-19 patients after implementation of an intermediate-dose VTE prophylaxis protocol guided by frequent anti-factor Xa measurements. In a recent systematic review, Jiménez et al 13 report a lower VTE rate of 27.9% in ICU patients compared to our study. However, that analysis included studies without a systematic screening approach. Among the included screening studies, the reported VTE prevalence was between 32% 18 and 100% 25 in cohorts receiving standard-dose thromboprophylaxis. In the few studies reporting VTE prophylaxis use and thromboprophylaxis regime8,18,19,26–28 patient cohorts were relatively small (n=20-88) and only one study (n=25) assessed both DVT and pulmonary vascular thrombosis prevalence with a rate of 32% while receiving mainly standard-dose. 18 Data regarding the use of intermediate-dosing is also limited to one study of 30 patients in which a DVT rate of 13% was found through ultrasound screening. 26 However, the overall VTE rate was not established as CTPA was not performed. More importantly, a critical difference of several of these reports compared to our study is the inclusion of distal and upper extremity DVT of limited clinical significance.
The prevalence of VTE in critical COVID-19 has also been reported in several retrospective studies2–4,14,29,30 relying on diagnostics performed at the discretion of treating clinicians. However, the lack of any screening method (ultrasonography or CTPA) carries a nonnegligible risk of missing many thrombotic events. Indeed, in a systematic review, the overall VTE prevalence among nonscreening studies was less than half of the prevalence observed when screening methods were used. 31 Our data, showing that approximately half of all VTE events were identified on screening ultrasound, support these previous observations.
Despite the implementation of an intermediate-dose prophylactic anticoagulation protocol, we observed a high prevalence of VTE. As hospitalized patients may progress to severe COVID-19 necessitating ICU admission, it is highly plausible that low-dose prophylactic anticoagulation in noncritically ill patients may impact VTE rates in the ICU. This is also supported by the most recent and largest randomized controlled trials from the REMAP-CAP, ACTIV-4a, and ATTACC investigators 32 showing benefit in clinical outcomes including increased probability of survival and reduction in major thrombotic events with the use of therapeutic anticoagulation in noncritically ill patients. The opposite was observed by the same investigators reporting potential harm from the use of therapeutic anticoagulation in critically ill COVID-19 patients in the ICU without image-proven thrombosis. 33 As the usual care arm in that study consisted of patients receiving mostly intermediate-dose anticoagulation, the optimal dosing strategy, standard low-dose or intermediate-dose, remains to be investigated in a future trial from the REMAP-CAP platform. The INSPIRATION randomized controlled trial of intermediate-dose versus standard-dose found no beneficial effect of enhanced anticoagulation on mortality or VTE occurrence. 17 However, the lack of screening methods and a very low rate of VTE among ICU patients in both the intermediate-dose group (3.3%) and standard-dose group (3.5%) suggest that the occurrence of VTE was underestimated. In contrast, we observed a considerably higher VTE rate (24%) even if all asymptomatic DVTs were excluded. Additionally, Moll et al compared an intermediate-dose strategy to standard-dose in a propensity-matched analysis and found a VTE rate of 23.4% in 47 patients without imaging surveillance. 32 In accordance with the INSPIRATION trial, they found similar VTE rates in both groups.
Major bleeding occurred in 10% of our cohort, similar to the occurrence rate observed by Moll et al 34 and higher than observed in studies using standard-dose prophylactic anticoagulation. For example, one systematic review 13 reported a prevalence of major bleeding of 3.9% overall, with a prevalence of 21.4% in cohorts receiving intermediate-dose or therapeutic anticoagulation. In our cohort, the use of acetylsalicylic acid was more common among patients who developed major and nonmajor bleeding (40% and 22.2%, respectively) compared to patients without bleeding events (12.5%).
No patient in our cohort had central pulmonary artery thrombosis. Helms et al 3 reported a notably higher prevalence of central pulmonary artery thrombosis among patients receiving standard-dose anticoagulation. Our findings of mostly segmental and subsegmental macrothrombosis together accounted for 64% (14 of 22), supported by previous studies.9,11,35 Similarly to prior studies4,10,11 we also observed a high prevalence of DVT with concurrent pulmonary vascular thrombosis in only 6%. These observations of predominantly distal pulmonary thrombi together with low rates of concurrent DVT suggest that local inflammation-causing immunothrombosis may be a prominent feature of severe COVID-19 pneumonia, 12 in addition to thrombo-embolic events from the deep venous system. Immunothrombosis is a recognized feature of acute respiratory distress syndrome (ARDS) pathogenesis. 36 The formation of pulmonary immunothrombosis may be triggered by several factors in COVID-19 critical illness including a dysregulated immune response due to inflammatory pulmonary infiltrates, hypoxia, prolonged immobilization causing venous stasis, proinflammatory comorbidities, and viral-mediated endothelial dysfunction resulting in coagulation activation.12,37,38 Pathologic findings frequently include thrombotic macro- and microangiopathy in COVID-19 ARDS39,40 which often overlap radiographically identified areas of ground glass opacities. 35
Study Implications
Our findings imply that a screening approach to detect thrombotic events is highly warranted in ICU patients with COVID-19 even with the use of enhanced anticoagulation. Surveillance of coagulopathy using intensivist-performed DVT ultrasonography has a high agreement with comprehensive exams 41 and may also limit hospital staff exposure during a pandemic. Ultrasound also avoids radiation exposure and is often readily available in the ICU to allow for early detection of subclinical thrombosis and prevent early stages of VTE progression by using adequate thromboprophylaxis in critically ill patients. This is important since the identification of asymptomatic proximal DVT has been independently associated with higher mortality in acutely ill patients. 20
Frequent surveillance using ultrasonography in COVID-19 patients has been recommended by an international consensus. 42 According to a pre-pandemic study, 43 detection of DVT leads to an earlier diagnosis of pulmonary embolism as well as lower mortality in critically ill patients. Additionally, the increased prevalence of pulmonary thrombi and immunothrombosis in patients with COVID-19 may advise caution to the treating clinicians in interpreting negative lower extremity ultrasound exams in order to eliminate the likelihood of pulmonary vascular thrombosis. Furthermore, a screening approach may improve the individualized risk-benefit assessment of an intensified thromboprophylaxis regime by identifying patients requiring prolonged duration as well as allowing for de-escalation of anticoagulation dosing in patients with high bleeding risk. Moreover, as most proximal DVTs were identified in the first week of ICU admission similar to prior screening studies,19, 44 this may also prompt clinicians to increased vigilance in performing imaging studies early during the ICU course. Finally, our study implies that the ability to maintain anti-factor Xa levels within the desired target range is challenging in critical COVID-19. We also acknowledge that the optimal target range for thromboprophylaxis in such patients remains uncertain and should be further assessed in clinical studies.
Strengths and Limitations
Our study has several strengths. To the best of our knowledge, it is the first systematic assessment of the prevalence of VTE in ICU patients with COVID-19 receiving intermediate-dose thromboprophylaxis. Data was prospectively collected from 6 different ICUs adhering to the same treatment protocols including thromboprophylaxis strategy. Our study patients share many characteristics with other published COVID-19 cohorts, which provides a degree of external validity for generalizing our results to similar cohorts.
Our study has limitations. Screening ultrasound was only performed once during ICU stay, excluding the possibility to identify later asymptomatic DVTs. Furthermore, the screening ultrasound was performed on patients in different stages of the disease and at different lengths of stay in the ICU. In addition, CTPA was not part of the screening protocol. However, at least one CTPA was performed in 93% of study participants and was repeated in more than half of these participants, thus providing robust information about the prevalence of pulmonary vascular thrombosis. Moreover, we were unable to assess 54 out of 169 (32%) eligible patients. However, pre-assessment discharge occurred randomly throughout the study period. It is therefore unlikely that the inclusion of these patients would have altered our findings dramatically. Finally, we lack data on VTE prevalence before the implementation of the enhanced thromboprophylaxis protocol. Therefore, we are unable to assess the prophylactic effect of this change in routine practice.
Conclusions
In our cohort of critically ill patients with COVID-19 exposed to intermediate-dose thromboprophylaxis, we observed a high prevalence of thrombotic and hemorrhagic complications. Imaging studies to identify thrombotic events is warranted in ICU patients to provide optimal care. Ongoing trials and future investigations are awaited to determine the optimal dose and duration of anticoagulation therapy to prevent VTE, immunothrombosis, and attenuate the risk of bleeding.
Footnotes
Abbreviations
Acknowledgments
The authors extend our gratitude to all patients participating in the study. They also thank all ICU staff members at Karolinska University Hospital for their support in the midst of the pandemic.
Authors’ Contributions
KA, NK, and JM designed the study, collected data, performed data analysis, and wrote the article. EM collected data, performed data analysis, and reviewed the article. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JM was supported by Region Stockholm (clinical research appointment and ALF project grants).
Appendices
Antithrombotic Medications Before Nonmajor and Major Bleeding was Discovered.
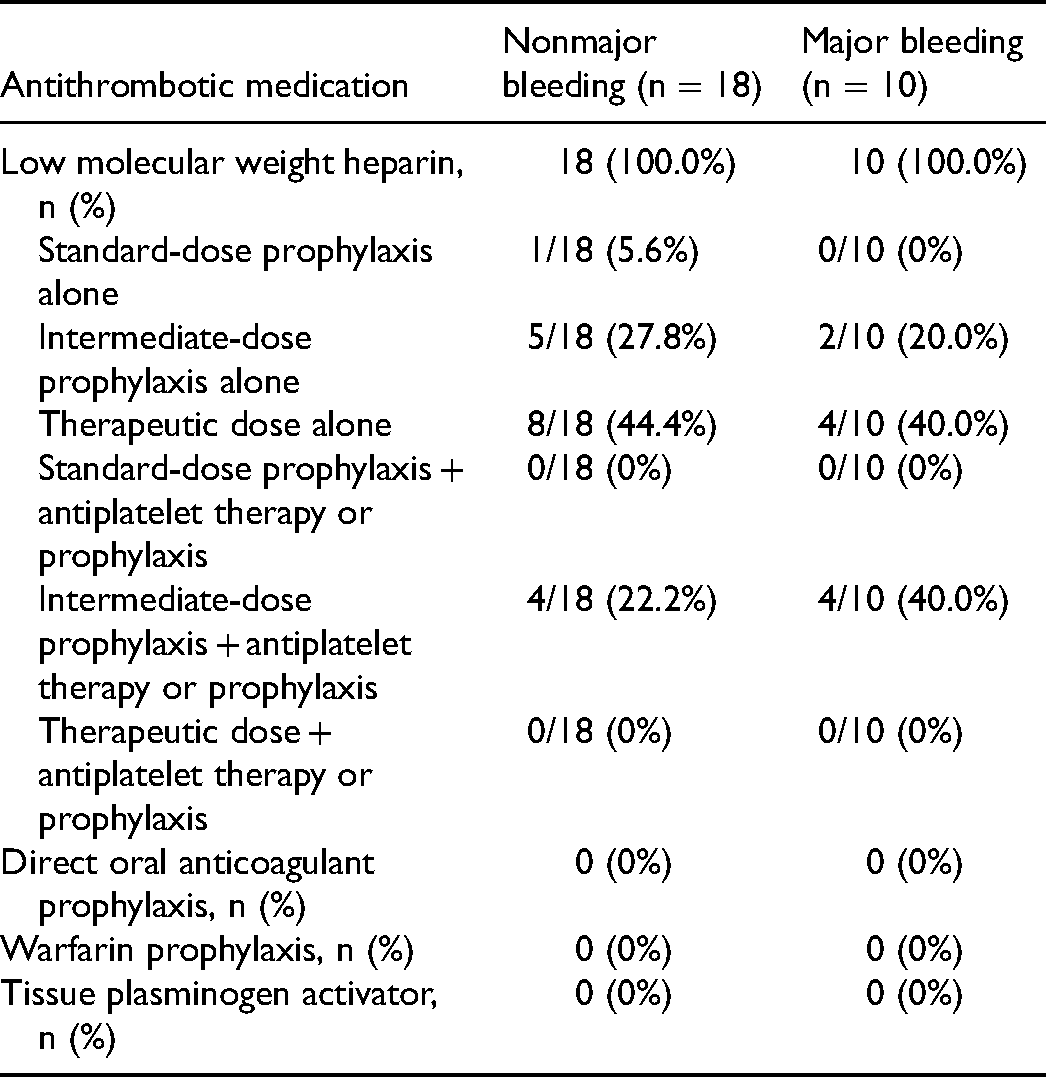
| Antithrombotic medication | Nonmajor bleeding (n = 18) | Major bleeding (n = 10) |
|---|---|---|
| Low molecular weight heparin, n (%) | 18 (100.0%) | 10 (100.0%) |
| Standard-dose prophylaxis alone | 1/18 (5.6%) | 0/10 (0%) |
| Intermediate-dose prophylaxis alone | 5/18 (27.8%) | 2/10 (20.0%) |
| Therapeutic dose alone | 8/18 (44.4%) | 4/10 (40.0%) |
| Standard-dose prophylaxis + antiplatelet therapy or prophylaxis | 0/18 (0%) | 0/10 (0%) |
| Intermediate-dose prophylaxis + antiplatelet therapy or prophylaxis | 4/18 (22.2%) | 4/10 (40.0%) |
| Therapeutic dose + antiplatelet therapy or prophylaxis | 0/18 (0%) | 0/10 (0%) |
| Direct oral anticoagulant prophylaxis, n (%) | 0 (0%) | 0 (0%) |
| Warfarin prophylaxis, n (%) | 0 (0%) | 0 (0%) |
| Tissue plasminogen activator, n (%) | 0 (0%) | 0 (0%) |
