Abstract
Introduction/Background:
Patients with rheumatoid arthritis (RA) have a high risk of infections that may require intensive care unit (ICU) admission in case of resulting sepsis. Data regarding the mortality of these patients are very limited. This study investigated clinical characteristics and outcomes of patients with RA admitted to the ICU for sepsis and compared the results to a control cohort without RA.
Methods:
All patients with RA as well as sex-, age-, and admission year-matched controls admitted to the ICU of a university hospital for sepsis between 2006 and 2019 were retrospectively analyzed. Mortality was calculated for both the groups, and multivariate logistic regression was used to determine independent risk factors for sepsis mortality. The positive predictive value of common ICU scores was also investigated.
Results:
The study included 49 patients with RA (mean age 67.2 ± 9.0 years, 63.3% females) and 51 matched controls (mean age 67.4 ± 9.5 years, 64.7% females). Among the patients with RA, 42.9% (n = 21) were treated with conventional synthetic (cs) disease-modifying antirheumatic drugs (DMARDs) and 30.6% (n = 15) received glucocorticoids only. Seven (14.3%) patients received biologic (b) DMARDs. The hospital mortality was higher among patients with RA (42.9% vs 15.7%,
Conclusions:
Hospital sepsis mortality was higher in patients with RA than in controls. Rheumatoid arthritis itself is independently associated with an increased sepsis mortality. Renal replacement therapy, renal failure, and diabetes were associated with an increased mortality. Common ICU scores were less well predictors of sepsis mortality in patients with RA compared to non-RA controls.
Introduction
Rheumatic diseases are associated with an increased risk of infectious complications. Patients with rheumatoid arthritis (RA) are particularly susceptible to serious infectious diseases such as septic arthritis, osteomyelitis, and infections of the skin but also pneumonia and genitourinary infections.
1
Reasons for a higher susceptibility to infections might be a high disease activity,
2
–4
a prematurely aged immune system (immunosenescence),
5
–9
a potentially impaired humoral immunity to some pathogens,
10
or the often required immunosuppressive medication including corticosteroids.
1,2,11,12
Compared to the general population, mortality risk of infections is increased up to 2- to 6-fold.
13
–16
Nevertheless, despite national and international recommendations,
17,18
vaccination rates for common preventable pathogens such as
We therefore aimed to retrospectively investigate the in-hospital mortality of sepsis requiring ICU treatment in patients with RA in comparison to an age-, sex-, and admission year-matched control group in a single university center in Germany.
Methods and Study Design
We retrospectively analyzed all cases of patients with RA (≥ 18 years) admitted to the ICU of our University Hospital for sepsis between February 2006 and January 2019. To acquire a realistic sample of patients, no further restrictions to comorbidities, gender, or age have been applied. All data were extracted from the patient’s electronic record. We searched the hospital records using the
Sepsis severity is assessed and electronically documented generally at admission to our medical ICU using Sequential Organ Failure Assessment (SOFA) and the Acute Physiology and Chronic Health Evaluation II (APACHE II) and Simplified Acute Physiology Score II (SAPS II). These scores are helpful to determine the degree of organ dysfunction, disease severity, and therefore estimation of sepsis mortality. Data regarding patient demographics (eg, gender and ongoing medication) and clinical as well as laboratory parameters were obtained from the electronic patient charts and laboratory records at the time of ICU admission. The ethics committee of the Medical Faculty of the University of Leipzig has approved the design of the study (Reg-No. 352/19-ek).
Biostatistical Analysis
Continuous data were described using either mean and standard deviation or median and interquartile range. Categorical data were described using absolute or relative frequencies. Fisher exact test was performed to compare frequencies of categorical variables. To compare continuous data, student
Results
A total of 49 patients with RA admitted to the ICU for sepsis (mean age at admission 67.2 ± 9.0 years, 63.3% female) were enrolled. Prior to ICU admission, 42.9% (n = 21) were treated with conventional synthetic (cs) disease-modifying antirheumatic drugs (DMARDs, mostly methotrexate [MTX], n = 17) and 30.6% (n = 15) received glucocorticoids only. Biologic (b) DMARDs were used in 14.3% (n = 7) of the patients with 28.6% (n = 2) of them being tumor necrosis factor (TNF) inhibiting agents and 42.9% (n = 3) rituximab (Table 1). The control group included 51 patients (age 67.4 ± 9.5 years, 64.7% females). Median ICU length of stay was 6.0 (2.0-10.0) days for patients with RA and 4.0 (2.0-8.0) days for the controls (
Characteristics and Antirheumatic Medication Among Patients With Sepsis Having RA.
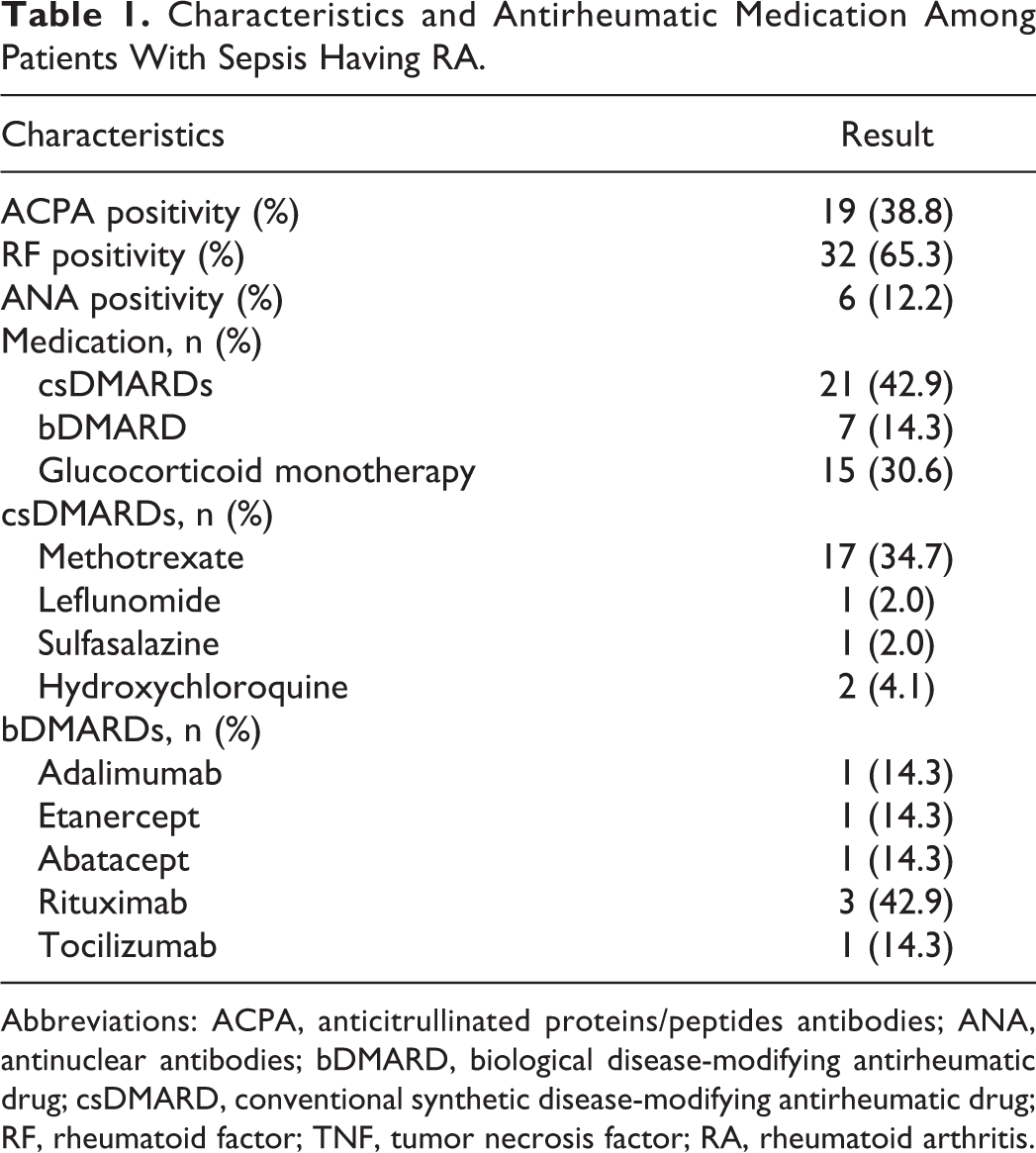
Abbreviations: ACPA, anticitrullinated proteins/peptides antibodies; ANA, antinuclear antibodies; bDMARD, biological disease-modifying antirheumatic drug; csDMARD, conventional synthetic disease-modifying antirheumatic drug; RF, rheumatoid factor; TNF, tumor necrosis factor; RA, rheumatoid arthritis.
During ICU treatment, patients with RA required mechanic ventilation in 69.4% (n = 34), vasopressor support in 77.6% (n = 38), and renal replacement therapy (RRT) in 24.5% (n = 12) cases. Among controls, mechanic ventilation was considered necessary in 62.8% (n = 32), vasopressor support in 51.0% (n = 26), and RRT in 35.3% (n = 18). While there was no difference regarding mechanic ventilation and RRT, vasopressor support was significantly more often required in patients with RA (
Clinical and Laboratory Data of Patients Having Sepsis With Versus Without Rheumatoid Arthritis.a
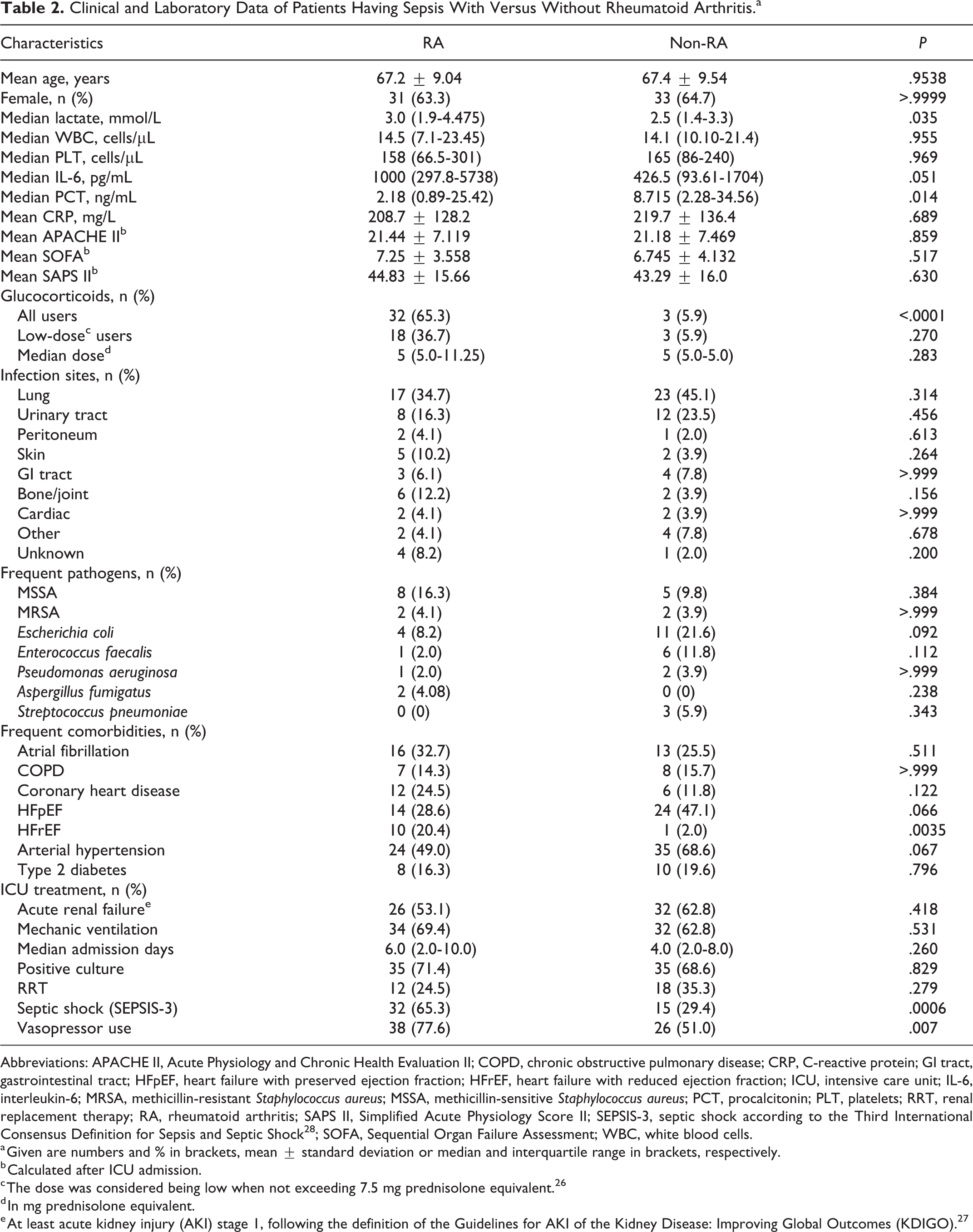
Abbreviations: APACHE II, Acute Physiology and Chronic Health Evaluation II; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; GI tract, gastrointestinal tract; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; ICU, intensive care unit; IL-6, interleukin-6; MRSA, methicillin-resistant
a Given are numbers and % in brackets, mean ± standard deviation or median and interquartile range in brackets, respectively.
b Calculated after ICU admission.
c The dose was considered being low when not exceeding 7.5 mg prednisolone equivalent. 26
d In mg prednisolone equivalent.
e At least acute kidney injury (AKI) stage 1, following the definition of the Guidelines for AKI of the Kidney Disease: Improving Global Outcomes (KDIGO). 27
Septic shock was more often diagnosed among patients with RA, 65.3% versus 29.4%,
Bivariate Analysis of Potential Risk Factors for Mortality in Patients With RA.
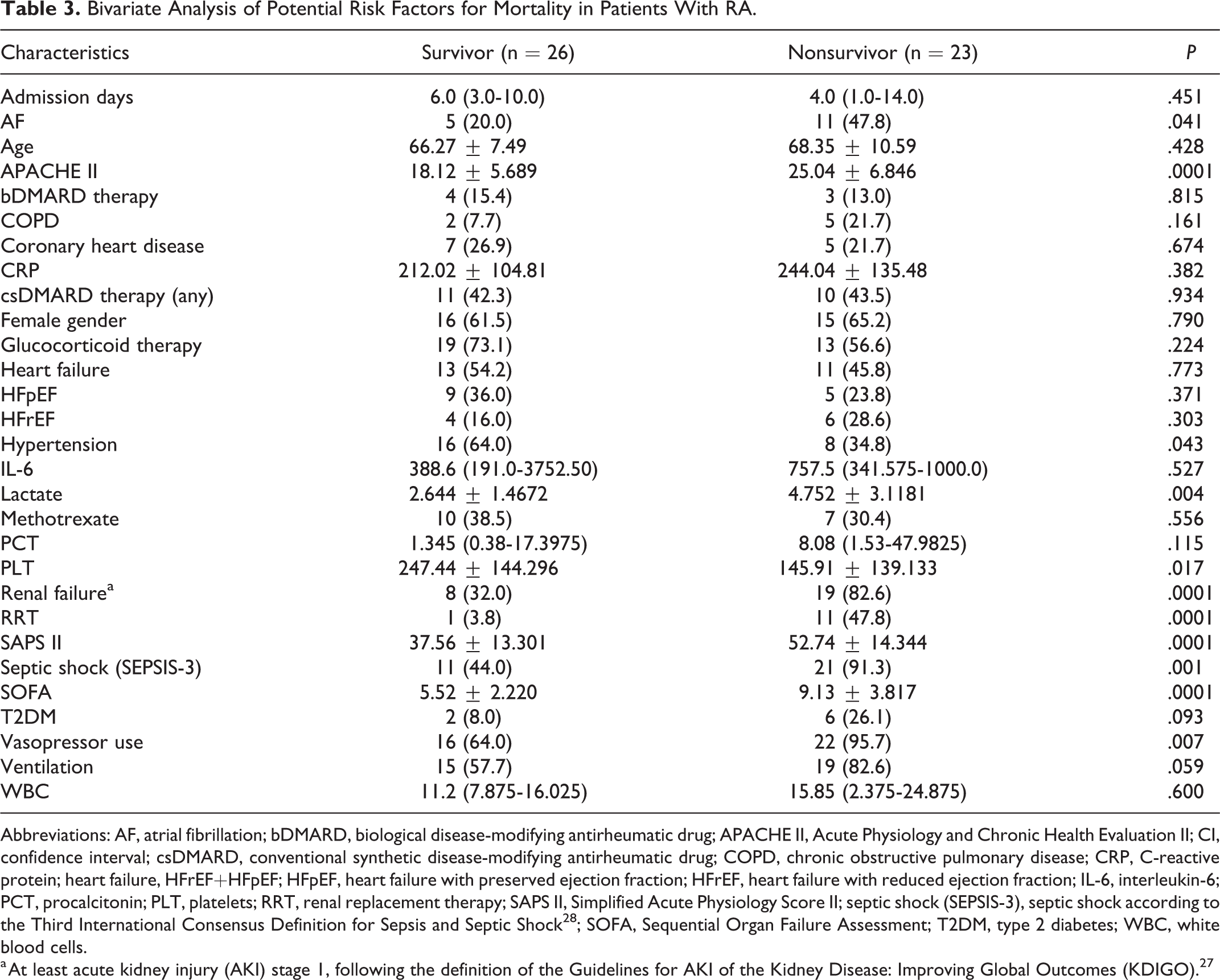
Abbreviations: AF, atrial fibrillation; bDMARD, biological disease-modifying antirheumatic drug; APACHE II, Acute Physiology and Chronic Health Evaluation II; CI, confidence interval; csDMARD, conventional synthetic disease-modifying antirheumatic drug; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; heart failure, HFrEF+HFpEF; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; IL-6, interleukin-6; PCT, procalcitonin; PLT, platelets; RRT, renal replacement therapy; SAPS II, Simplified Acute Physiology Score II; septic shock (SEPSIS-3), septic shock according to the Third International Consensus Definition for Sepsis and Septic Shock 28 ; SOFA, Sequential Organ Failure Assessment; T2DM, type 2 diabetes; WBC, white blood cells.
a At least acute kidney injury (AKI) stage 1, following the definition of the Guidelines for AKI of the Kidney Disease: Improving Global Outcomes (KDIGO). 27
The in-hospital mortality was significantly higher in patients with RA than among controls (42.9% vs 15.7%,
The performed ROC analysis (Table 4 and Figure 1) revealed that the area under the curve (AUC) for SOFA, SAPS II, and APACHE II was lower among patients with RA, reflecting a less well-predictive power of the scores for mortality in this group (Figure 1). A subset analysis of the RA cohort revealed all scores being significantly higher in nonsurvivors (Table 3); 70% of RA nonsurvivors had an SOFA and SAPS II above median, and 65% had an APACHE II above median (data not shown).
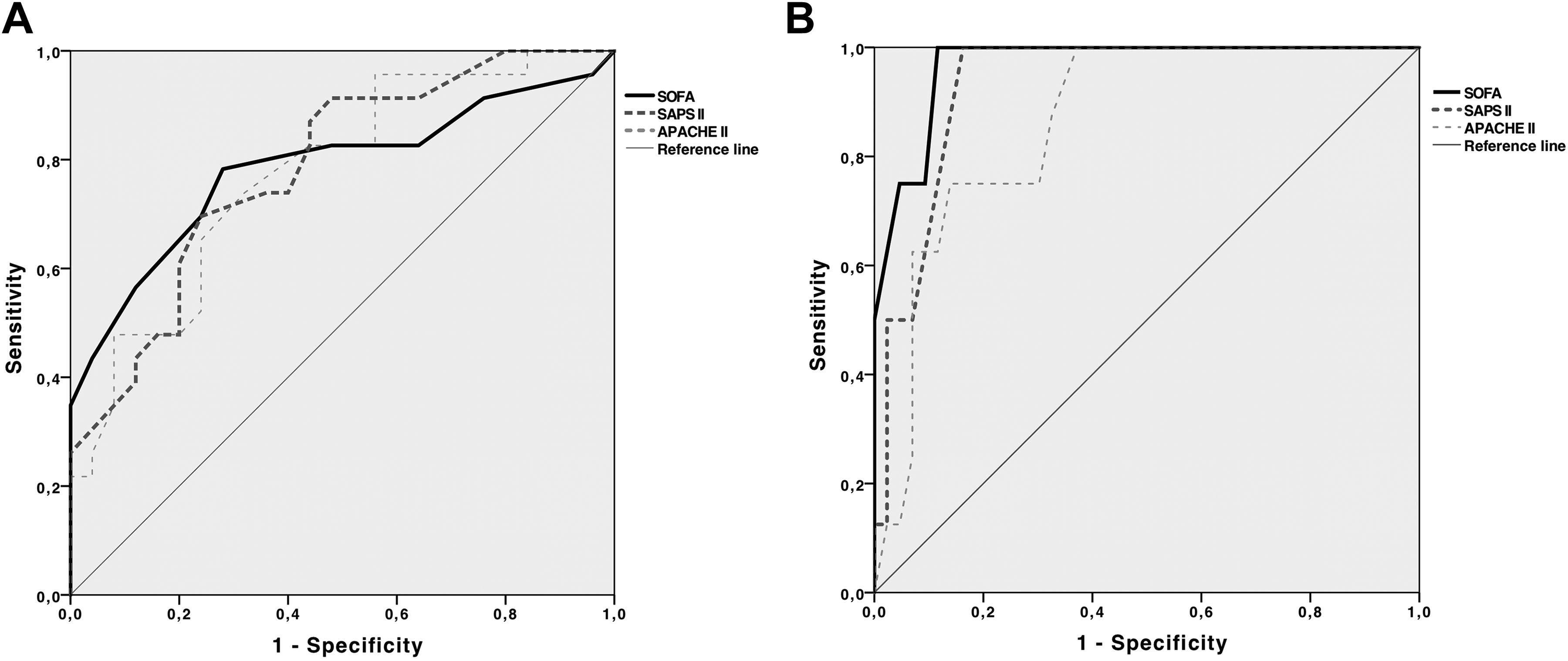
Receiver–operating characteristic (ROC) analysis of the value of the SOFA, SAPS II, and APACHE II score in predicting in-hospital mortality among RA (A) and non-RA (B) patients. See Table 4 for numbers. APACHE II indicates Acute Physiology and Chronic Health Evaluation II; RA, rheumatoid arthritis; SAPS II, Simplified Acute Physiology Score II; SOFA, Sequential Organ Failure Assessment.
Receiver–Operating Characteristic (ROC) Analysis of the Value of the SOFA, SAPS II, and APACHE II Score in Predicting Mortality.a
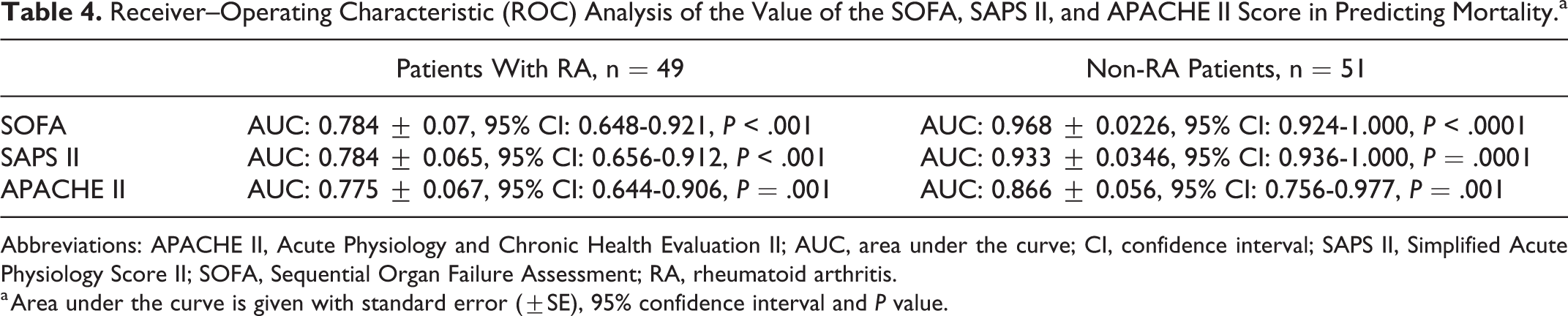
Abbreviations: APACHE II, Acute Physiology and Chronic Health Evaluation II; AUC, area under the curve; CI, confidence interval; SAPS II, Simplified Acute Physiology Score II; SOFA, Sequential Organ Failure Assessment; RA, rheumatoid arthritis.
a Area under the curve is given with standard error (±SE), 95% confidence interval and
In a multivariate analysis, RRT (odds ratio [OR]: 24.202, 95% CI: 1.528-383.219,
Multivariate Analysis Using Logistic Regression, Adjusted for Age and Gender, of the Relationship Between Mortality of Patients With RA and the Given Parameters.
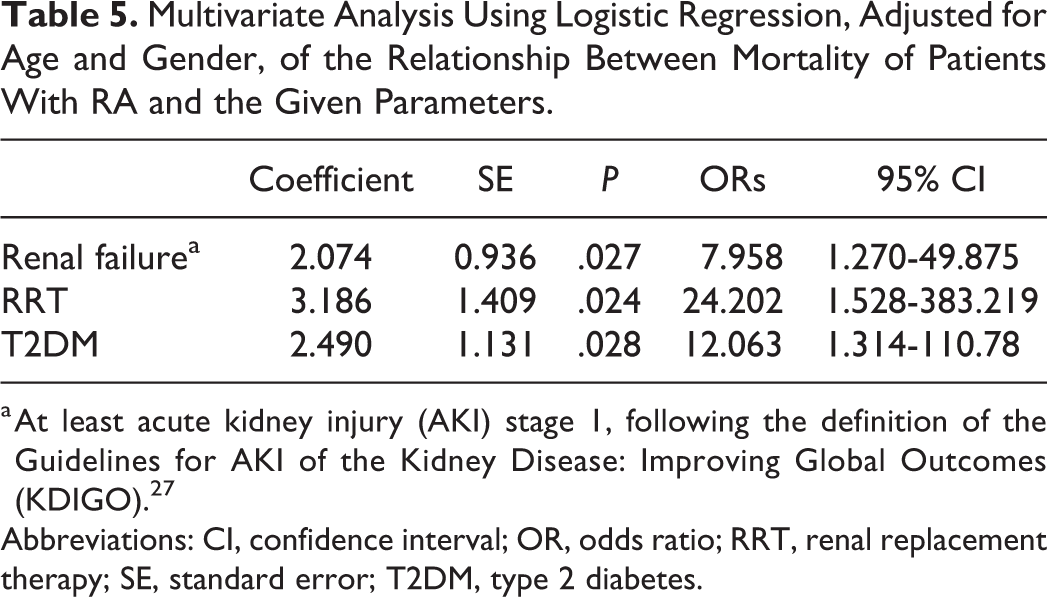
a At least acute kidney injury (AKI) stage 1, following the definition of the Guidelines for AKI of the Kidney Disease: Improving Global Outcomes (KDIGO). 27
Abbreviations: CI, confidence interval; OR, odds ratio; RRT, renal replacement therapy; SE, standard error; T2DM, type 2 diabetes.
Discussion
In this retrospective analysis, in-hospital mortality was found to be significantly higher among patients with RA compared to controls. One reason most likely is the higher incidence of septic shock in the RA group. Although multivariate analysis did not reveal septic shock to be independently associated with nonsurvival in our logistic regression model (maybe caused by the strong association of septic shock with other variables, eg, SOFA and vasopressor use), septic shock is surely highly associated with mortality. 29 Another reason for the high mortality might be the tertiary character of our hospital predominantly admitting severely ill, immunocompromised patients with RA. Furthermore, HFrEF was more prevalent the RA cohort than among non-RA patients. This finding was also reported by others 30 and could contribute to an increased mortality.
Interpreting our results in the light of the existing literature, we found one study that included 50 patients with various inflammatory rheumatic diseases, showing a mortality rate among the RA subgroup similar to that of our cohort.
25
Colleagues from Israel also reported a rather comparable mortality of 34.9% in 43 patients with RA under ICU treatment. Almost three-fourth of these patients were admitted to ICU for infection, 55.8% for septic shock.
24
In contrast, an investigation from Austria found a very low mortality rate of 10.8% in 74 patients with RA who have been admitted to an ICU for infection.
31
However, that study only included patients that needed parenteral antibiotics for infection and were admitted to an ICU, and sepsis was not specifically discussed. Another investigation from the United States exclusively focusing on
An interesting cohort study from the United States demonstrated a lower sepsis mortality among patients with autoimmune diseases compared to nonautoimmune controls (26.6% vs 34.6%,
Of particular interest of our study is the finding that no kind of disease-modifying or immunosuppressive medication (including bDMARDs, csDMARDs and glucocorticoids) had any influence on sepsis mortality in the patients with RA. This is partly in line with the findings of Sams et al who showed that
Multivariate analysis revealed RA itself being the most important independent risk factor for sepsis mortality. Other risk factors for nonsurvival were the need for RRT, renal failure, and type 2 diabetes. Diabetes mellitus was earlier identified as a strong predictor of infection in patients with RA
40
and has also been associated with sepsis mortality in patients with RA before (HR: 1.78, 95% CI: 1.19-3.52,
As demonstrated using ROC analysis, APACHE II 44 , SAPS II, 45 and SOFA 46 show a good power in predicting sepsis mortality, but the association clearly seems to be stronger in the control group than among the patients with RA. This finding is remarkable and raises the question for the underlying cause(s). The literature on the association of ICU scores and mortality in patients with rheumatic diseases is scarce and mostly focuses on vasculitis and SLE. 47,48 While SOFA was associated with mortality and showed a good predictive power in vasculitis patients, APACHE II was not associated with mortality. 48 In patients with SLE, APACHE II was unable to accurately predict mortality. 47 The results of our RA cohort are in line with the findings of Haviv-Yadid et al who showed an association between mortality and APACHE II as well as SOFA. 24 None of these studies compared the score’s power with a control group though. Different reasons could be causative for our finding of an impaired power in predicting mortality of patients with sepsis having RA. Since RA is not seldom associated with thrombocytosis, 49,50 platelet count could have been overestimated. This would particularly effect SOFA score, which shows the largest AUC deviation between patients with RA and non-RA controls in our study. Since overestimated platelets have no impact on APACHE II or SAPS II, there also have to be other reasons for the observed AUC deviation. Although both APACHE II and SAPS II account for chronic disease, they might not be ideal for RA: APACHE II only considers “chronic health problems” including a variety of diagnoses and therapies (liver cirrhosis, New York Hearth Association Class 4 heart failure, severe COPD, pulmonary hypertension, regular dialysis or immunocompromising conditions such as immunosuppression, chemotherapy, radiation, long-term or high-dose steroids, leukemia, lymphoma or AIDS) as 1 single parameter. If a patient with RA has no immunosuppression, then the score would not rise, despite the fact that RA itself impairs the immune system 5 –9 and increases the risk of infections. 1,3,4 The SAPS II also only accounts for metastatic cancer, hematologic malignancy, and AIDS under the term “chronic disease.” Consequently, both scores could underestimate sepsis severity which might explain the observed AUC deviation. We therefore speculate that the RA-related impairment of the immune system (“immunosenescence”), a potentially reduced response to some pathogens, thrombocytosis, and disease activity itself lead to a reduced reliability of ICU scores in terms of mortality prediction.
Our study has several limitations. The sample size is relatively small. Nevertheless, the data using a comparable non-RA septic cohort allow a good understanding of the scenario. Since the study is retrospective, there may be the risk of misclassification due to incorrect coding (ICD-10). However, we have reduced this risk by double-checking the diagnoses using the medical records. As a single-center study, there may also be a bias in disease severity. However, we believe that the results may contribute to further our understanding of patients with rheumatoid arthritis suffering from sepsis.
Conclusions
Mortality in patients with RA has been demonstrated to be substantially higher than in controls. Independent risk factors for sepsis mortality in patients with RA were RRT, renal failure, and diabetes. Rheumatoid arthritis itself was identified as being strongly related to sepsis mortality. The commonly used APACHE II, SAPS II, and SOFA scores demonstrated reduced power in predicting sepsis mortality in patients with RA compared to non-RA patients.
Footnotes
Authors’ Note
All procedures performed in this survey were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Data obtained in this study did not interfere with the course of treatment for patients included.
Marco Krasselt conceived the project, collected and interpreted the data, performed the statistical analysis, and drafted the manuscript. Christoph Baerwald contributed to data collection, interpretation, and was involved in manuscript drafting. Sirak Petros was responsible for ICU patient care and contributed important intellectual content to the manuscript. Olga Seifert was involved in the statistical analysis as well as manuscript preparation. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Baerwald received lecture fees from Merck, MSD, Mundipharma, and Pfizer. Doctores Krasselt, Petros and Seifert have nothing to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
