Abstract
Abdominal adhesions are still among the most common postsurgical peritoneal inflammation-related complications. Adhesion-related disorders are still highly costly and prevalent due to advances in surgical techniques, treatment methods, and various drugs. The present study aimed to investigate the effects of Poly (D,L-lactide-co-glycolide) (PLGA)- polyethylene glycol (PEG6000) Nanofiber + Melatonin on the abdominal adhesion model in rats. For this purpose, PLGA-PEG6000 Nanofiber + Melatonin matrix was fabricated and implanted in an experimental abdominal adhesion model in rats. Our study consisted of an in vitro and an in vivo part. The degradation and release profile of the matrix and Melatonin (Mel) embedded matrix was performed in vitro. In vivo, the procedure was carried out with 18 Wistar male rats. Rats were divided into three groups as follows: Sham, Matrix, and Mel + Matrix, respectively. Consequent to degradation and release profiling in vitro, an experimental adhesion model was created and fabricated pure matrix (2 × 2 cm2), and matrix (2 × 2 cm2, 0.25 mg melatonin/per matrix embedded) was applied to injury area in related groups. Intra-abdominal adhesion scores were determined on post-op 21st day, under general anesthesia. Following, cecum, peritoneal tissue, and adhesive bands were harvested. Macroscopic analysis (severity of adhesion formation), Hematoxlyn&Eosin and Masson’s Trichrome staining (for the examination of the levels of infiltration of inflammatory cells, fibrosis, and neovascularization) were performed for the evaluation of the effects of Mel embedded and pure matrix Our results indicated that PLGA-PEG6000 Nanofiber + Melatonin matrix was degraded completely in rats abdominal cavity and significantly reduced adhesion formation compared to other groups macroscopically (p < 0.05). On the other hand, the histopathological analysis indicated that the fabricated matrix reduced inflammatory cell infiltration, fibrosis, and neovascularization levels.
Introduction
Abdominal adhesions are bands of scar-like tissue that form inside the abdomen. The bands arise between two or more organs or organs and the abdominal wall. Adhesion may occur following chronic peritoneal inflammation, peritoneal dialysis, and endometriosis. 1 Adhesions are a common complication of surgery, occurring in up to 50%–95% of people who undergo abdominal or pelvic surgery, and place a tremendous clinical and financial burden on public health. 2 Adhesion formation causes a range of symptoms like chronic pain, bowel obstruction, urinary dysfunction, and female infertility.3,4 Despite the deleterious effects, novel therapies, drugs, or barriers have limited effects on the prevention of intraabdominal adhesion. Moreover, an inadequate dose of drugs reaching the injured area, a higher absorption rate peritoneum, and a lack of adhesion-specific drugs are also other factors that disable the prevention of adhesion formation. 5 In addition, the drugs such as steroids, Trasilol, Heparin, dicumarol calcium channel blockers, and nonsteroid antiinflammatory agents 6 currently used for adhesion prevention have several side effects,7 –10 Due to the unavoidable disadvantages of drugs, the adhesion barriers became popular but have limited effect and are high cost.11,12 Whilst many methods have been advanced in an attempt to alleviate the formation of adhesions, no single approach has been wholly satisfactory.13 –15
Melatonin (Mel), chemical formula is N-acetyl-5-methoxy-triptamine, is a pleiotropic hormone synthesized from the pineal gland and has numerous effects. Melatonin has strong antioxidant, antiinflammatory, antithrombotic, and anti-fibrotic properties.16,17 Mel has charmed the interest of researchers due to its potential benefit for alleviating adhesion formation. Previous studies indicated the beneficial effects of Mel on adhesion formation.13,18 Novel studies reported different administration ways for Mel. Oral administration of Mel exhibits low biocompatibility, is poorly soluble, highly permeable, and shows a short half-life within <45 min.19,20 Instead of oral treatment, transdermal, transepidermal, and intranasal drug carrier systems providing long-term drug delivery draw attention. 19 While polymeric films are courageous options for drug carrier systems, there are various other carrier systems with more advanced technology such as lipids, 21 micelles, 22 nanoparticles, 23 and nanofibers. 24 Nanofibers have many advantages as drug carrier systems. For instance, materials developed by an electrospun technique such as biodegradable polymers are non-toxic, have great mechanical strength, and have high porosity providing a wide surface area. 25 Zhang et al., 26 reported that electrospun Poly (D,L-lactide-co-glycolide)(PLGA) and polyethylene glycol (PEG) polymers in different concentrations have gained hydrophilicity properties and can be used as carriers for hydrophilic drugs. Furthermore, both PLGA and PEG polymers are approved by the US FDA (United States Food and Drug Administration). 27
In this study, we aimed to develop a melatonin-embedded PLGA/PEG matrix, to profile the release and morphological characteristics of the matrix in vivo, and to investigate the effectiveness of the matrix using an in vivo experimental adhesion model.
Materials and methods
In vitro experiment
Preparation of PLGA-PEG6000 matrix
PLGA-PEG6000 nanofiber was prepared by a previously described electrospinning method. 28 Briefly, a 15% PLGA-PEG6000 (70:30) polymer solution was prepared in the DCM-DMF (70:30) solvent system. Mel was prepared freshly (dissolved in 1% ethanol and saturated with physiological serum). 29 Then, Mel (40 mg) was added to 0.5% of the polymer with a final dose of 0.25 mg Mel per/matrix (2 × 2 cm2). The polymer matrix was mixed until complete homogeneity. Based on the literature, appropriate electrospinning parameters were obtained with a flow rate of 0.8 mL/h, distance of 20 cm, and voltage of 20 kV. Mel treatment was performed under red light to minimize Mel degradation. 28 All treatments with Mel were performed under red light to minimize the degradation of Mel.
Biodegradability study
Specimens of 1 × 3 cm2 PLGA-PEG6000 matrix were used for the biodegradability study. Specimens were immersed in 10 mL of phosphate buffered saline (PBS, 0.1 M, pH 7.4). The samples in PBS were kept in an incubator at 37°C and were taken from the PBS at 24-h intervals for 15 days.
Afterward, the specimens were dried in a vacuum oven for 24 h, weighed, and % mass loss was calculated against initial weight.
In vitro drug release
Mel-embedded nanofibers weighing about 50 mg, were placed in 50 ml pH 7.4 PBS. Mel release behavior tests were carried out three times with a UV-Spectrophotometer (Shimadzu, JAPAN) at a wavelength of 220 nm at 37°C and standard solutions were prepared at 0.5, 1, 2, 5, and 10 mg/L, respectively. 30 To maintain the sink conditions, PBS was replaced with the same volume.
In vivo experiment
Animals
In this study, 18 Wistar Albino male rats (250–300 g; 4–6 weeks) were obtained from the Çanakkale Onsekiz Mart University Experimental Research Centre. Rats were housed at 22 ± 2°C temperature and 12/12 h light/dark cycle. During the experiment, rats were fed with standard food pellets and water ad libitum. The experimental protocol of this study was approved by the Çanakkale Onsekiz Mart University Animal Experiments Local Ethics Committee (Protocol number: 2022/01-04). The whole protocol was proper with the 1964 Helsinki Declaration.
Study Design
Animals were divided into three groups (n = 6) randomly as follows:
Group 1 (Sham, n = 6): The peritoneal adhesion model was established, and no procedure was applied.
Group 2 (Matrix, n = 6): The peritoneal adhesion model was established and a 2 × 2 cm2 pure matrix (without Mel) was applied between the abraded cecum and peritoneum.
Group 3 (Mel + Matrix, n = 6): The peritoneal adhesion model was established and 0.25 mg Mel (2 × 2 cm2 per/matrix) embedded matrix was applied between the cecum and peritoneum.
Adhesion model
The adhesion model was created as previously described.31,32 Under general anesthesia, the abdomen was shaved and prepared with a topical povidone-iodine solution, then a laparotomy was performed through a 5-cm midline incision. The cecum was exteriorized and abraded with a No:15 scalpel until punctuated bleeding occurred in an area of 2 cm2, the cecum was not perforated. Then, an area of 2 cm2 of the parietal peritoneum directly anterior to the cecum was excised from the abdominal wall. Then 2 × 2 cm2 matrix barrier was placed between the abraded surfaces in Matrix and Mel + Matrix groups. Finally, the peritoneal cavity and abdominal wall were closed with a continuous suture of 3/0 absorbable suture (Polyglactin-910).
Euthanasia and tissue harvesting
The experimental study was concluded on the 21st day. Animals were anesthetized with ketamine-xylazine (70/7 mg/kg). The abdominal cavity was reopened, and a “L” shaped incision was made from the left iliac spine to the right iliac spine, and the right subcostal margin, to avoid the initial incision and adhesion areas. Macroscopic adhesion scoring was performed before sample harvesting for histopathologic evaluation. Then the rats were euthanized by cervical dislocation. The injured cecum area, opposite peritoneal area, and adhesion bands were harvested and stored in 10% formaldehyde for histopathological evaluation.
Macroscopic score
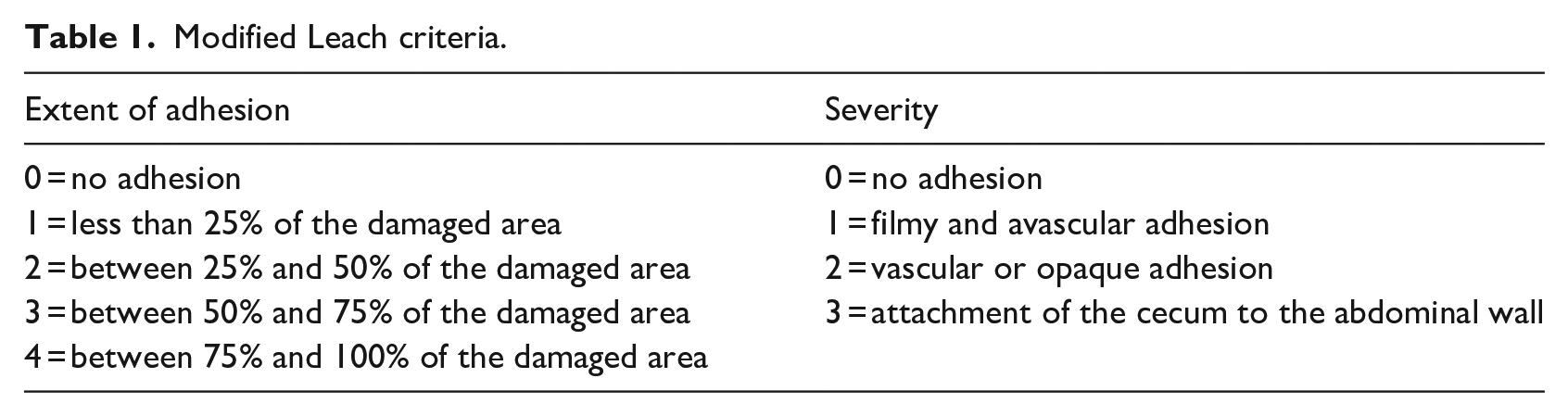
Macroscopic scoring was performed via modified Leach et al. model 33 and represented in Table 1.
Modified Leach criteria.
Histopathologic evaluation
Tissue specimens were stained with Hematoxylin&Eosin (H&E) and Masson trichrome (MT) for histopathological evaluation of the cecum and anterior abdominal wall adhesion. Specimens were fixed in 10% formaldehyde for 48 h at room temperature. After the fixation, the tissues were washed overnight under tap water then passed through the rising alcohol series, and were cleared in xylol. Tissue blocks were obtained by embedding them in paraffin. Tissue sections of 4–5 µm thickness were prepared using a microtome (Slee Cut, 5062, Mainz, Germany).
The tissue sections were deparaffinized in xylol, passed through decreasing degrees of alcohol, and washed into water. The sections were stained with Hematoxylin (Merck, 105174) for nucleus and ground staining and after washing, stained with Eosin (Merck, 102439). The sections were then passed through a rising alcohol series, cleared in toluol, and sections were covered with entellan.
MT stains were performed according to the manufacturer’s instructions. Tissue sections were deparaffinized and rehydrated by the routine histological method. Bouin’s solution, Weigert’s iron hematoxylin working solution, and aniline blue solution were used according to the kit instructions (Sigma-Aldrich, USA).
Adhesive lesions associated with the caecal region and anterior abdominal wall were examined under a light microscope (Olympus CX40, Olympus, Japan) and image analysis software (Kameram, Argenit, İstanbul). Adhesion area was estimated via the following parameters; inflammatory cell infiltrate (0: absent or normal in number, 1: slightly increased, 2: moderate infiltration, 3: massive infiltration), fibrosis (0: absent, 1: mild, 2: moderate, 3: dense), and neovascularization (0: absent, 1: one to two vessels, 2: three to nine vessels, 3: ten or more vessels). 34
Statistical analyses
Macroscopic adhesion scores determined by modified Leach scores and histopathological evaluation results were represented as mean ± Standard Error (SE). Statistically significant data were analyzed by “SPSS for Windows version 24” (Chicago, IL, USA). Multiple group comparison was performed by One-Way ANOVA, and the comparison of two groups was performed by Tukey posthoc test. The significance was inferred at p < 0.05.
Results
In vitro results
Time of degradation
The degradation time and related % mass loss of the PLGA-PEG6000 matrix were represented in Figure 1. PLGA-PEG6000 matrix was mostly degraded within the first 24 h and reached 42%. Afterward, approximately 2%–3% degradation was observed per day and 83.42% of the polymer was decomposed at the end of 15 days.
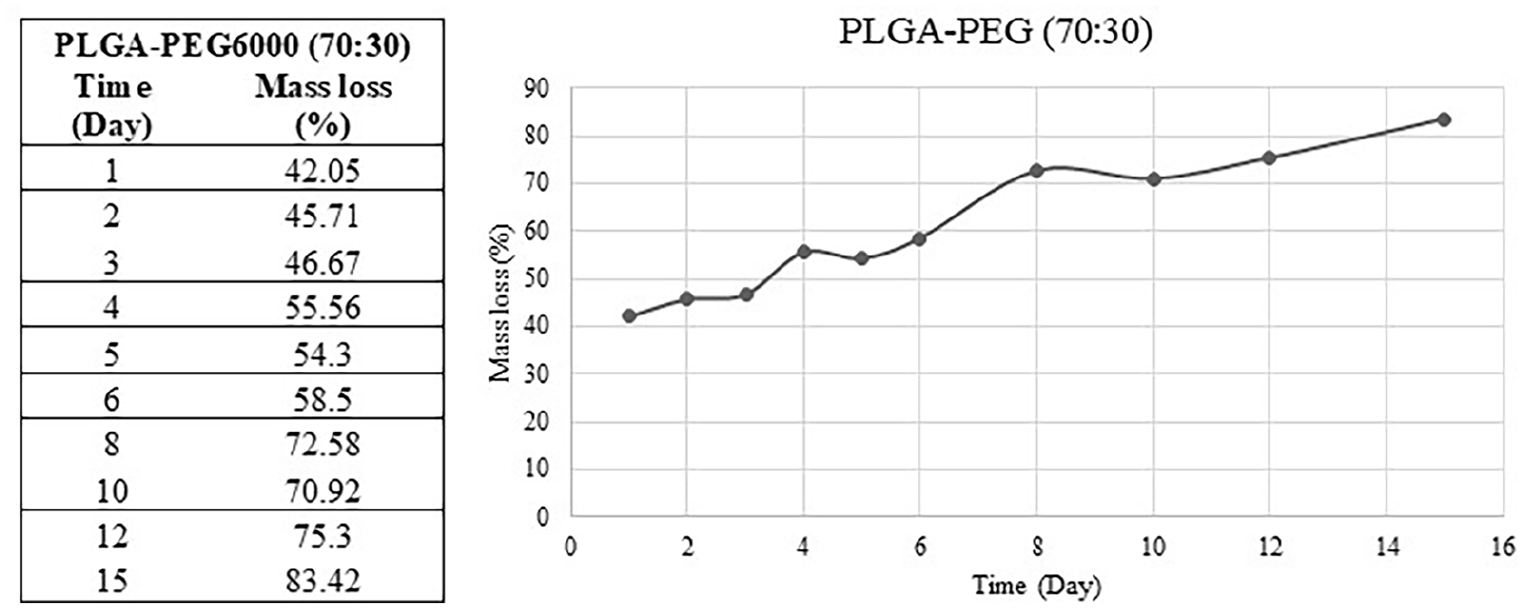
Degradation and % mass loss of PLGA-PEG6000 matrix.
In vitro drug release
The release profile of Mel from the matrix is represented in Figure 2. It is clear from Figure 2 that the release rate of Mel from the matrix continued for about 100 h and approximately at 8 h, Mel indicated a burst release of about 7%, then followed a slow release. Even if the release solution was refreshed with the same volume PBS 100 h later. Mel release reached about 15% which might be due to the hydrophilicity of the polymer. 30
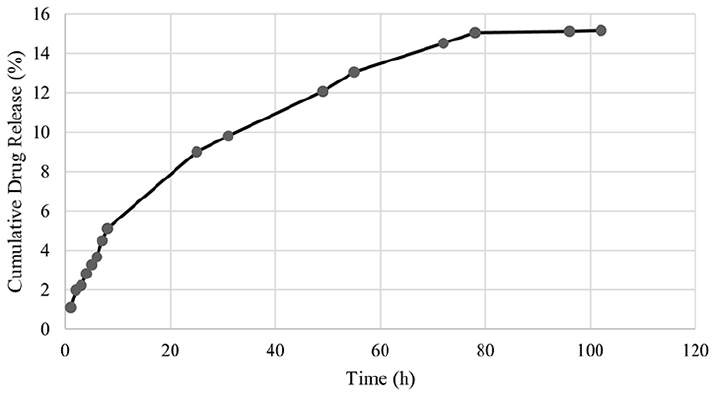
Cumulative release profile of melatonin from the PLGA-PEG6000 matrix.
In vivo results
Macroscopic scores
In our study macroscopic adhesion scoring was performed according to the extent of adhesion formation and severity of adhesion. Macroscopic scores are represented in Table 2 and Figure 3. Our results indicated that there was not a statistically significant difference between the Sham (3.33 ± 0.49) and Matrix (3.66 ± 0.33) groups for the extent of adhesion (p > 0.05). Further, the extent of adhesion formation significantly decreased in the Mel + Matrix group (1.50 ± 0.42) compared to the Sham and Matrix groups (p = 0.02 and 0.007, respectively). Similarly, the severity of adhesion formation reduced in the Mel + Matrix group (1.66 ± 0.49) compared to Sham (2.83 ± 0.16) and Matriks (3.00 ± 0.00) groups (p = 0.038 and 0.018, respectively). There was no statistically significant difference between the Sham and Matrix groups (p > 0.05).
Extent and severity scores of adhesion formation.
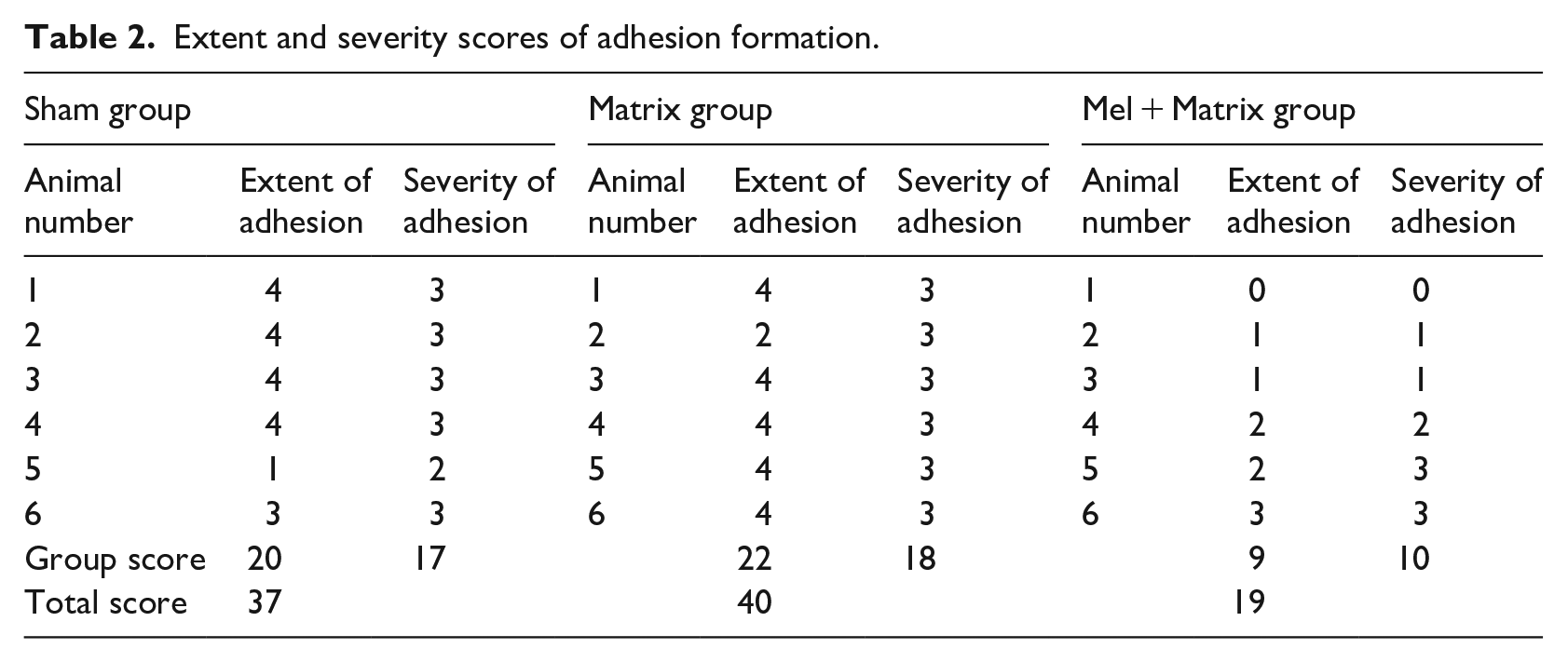
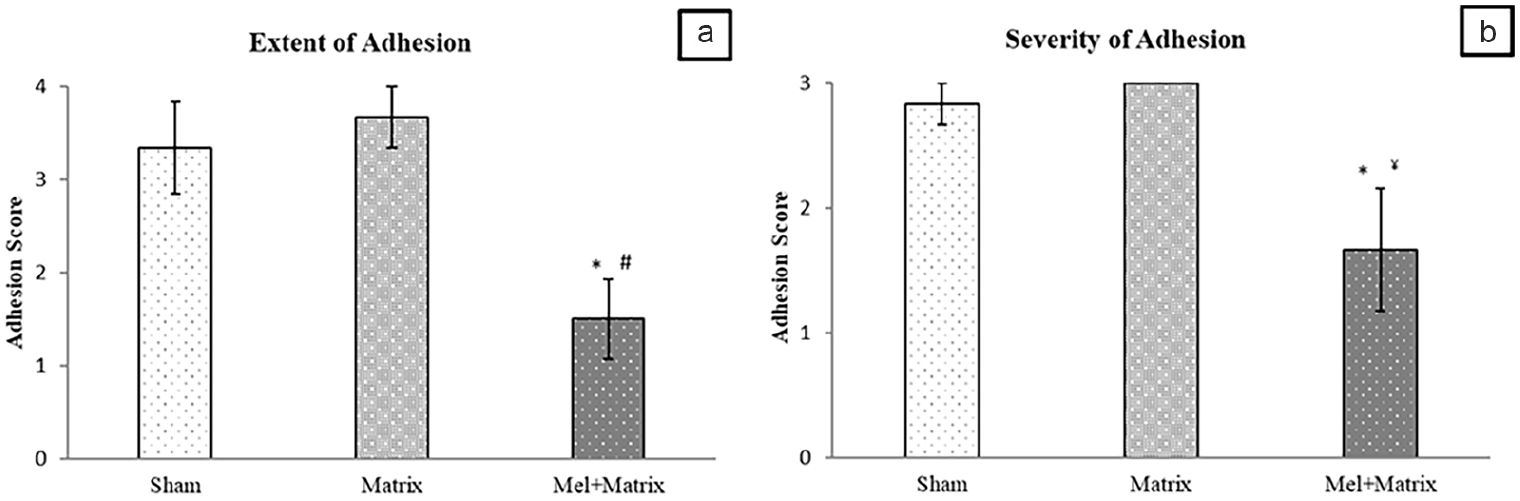
(a) Extent of adhesion scores and (b) severity of adhesion scores of the experimental groups.
Histopathological evaluation results
Our histopathologic findings showed fibrous bands extending between the abdominal wall and cecum in the Sham and Matrix groups. Infiltration of inflammatory cells including neutrophils, plasma cells, and eosinophils was observed in the adhesion areas in the Sham and Matrix groups. Fusion of the muscular layer of the abdominal wall and cecum was observed in the adhesion areas in the Sham and Matrix groups. In the Mel + Matrix group, in addition to inhibition of adhesion, suppression of inflammation in the cecum and abdominal wall was observed.
In our study fibrosis, neovascularization, and inflammation levels were determined histopathologically and represented in Figures 4 and 5. Fibrosis levels of the Mel + Matrix group (0.50 ± 0.34) decreased compared to the Sham (2.00 ± 0.36) and Matrix (2.50 ± 0.22) groups (p = 0.001 and 0.011, respectively). There was not a significant difference between the Sham and Matrix groups (p > 0.05).
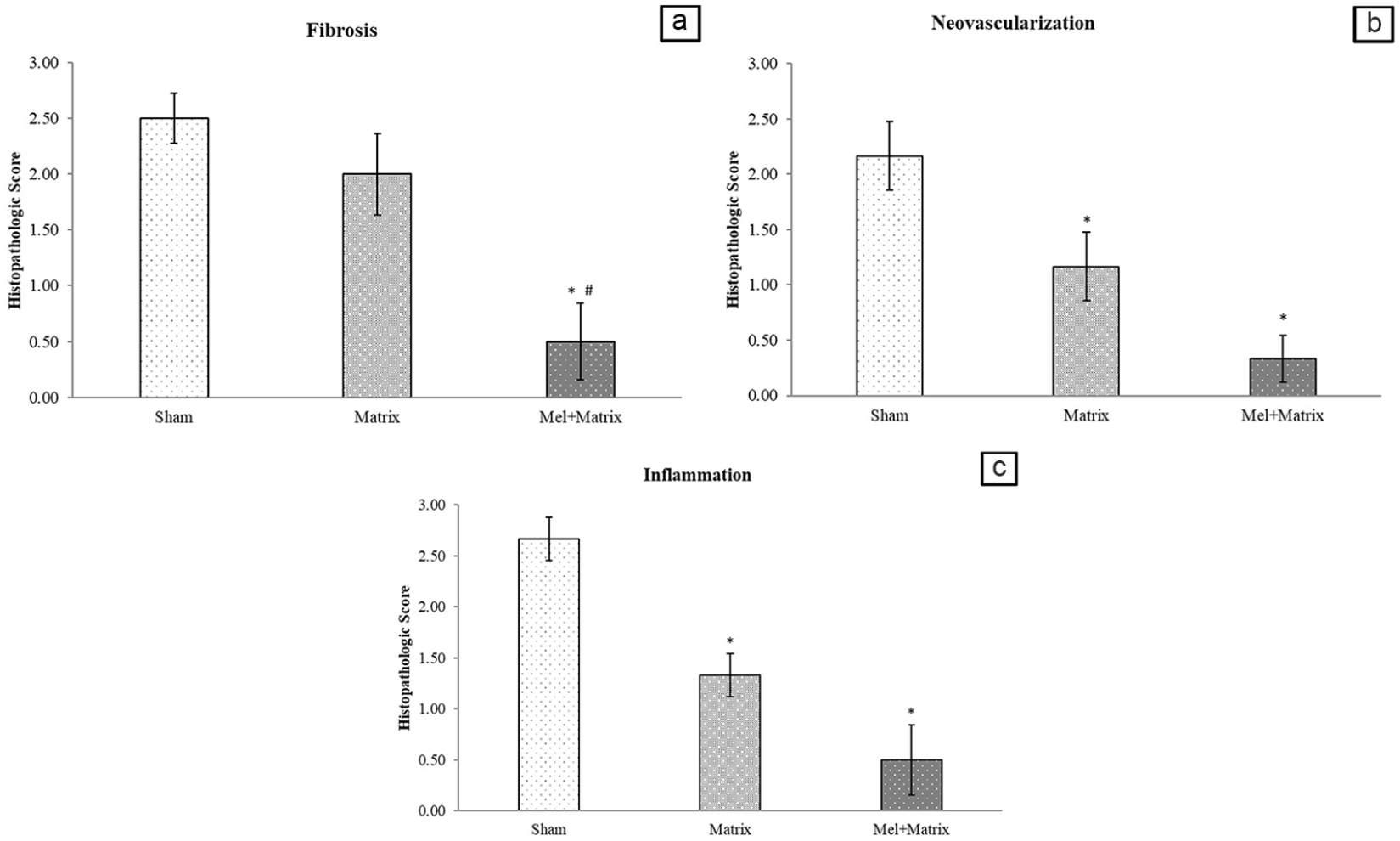
(a) Fibrosis (b) neovascularisation and (c) inflammation scores of adhesion formation.
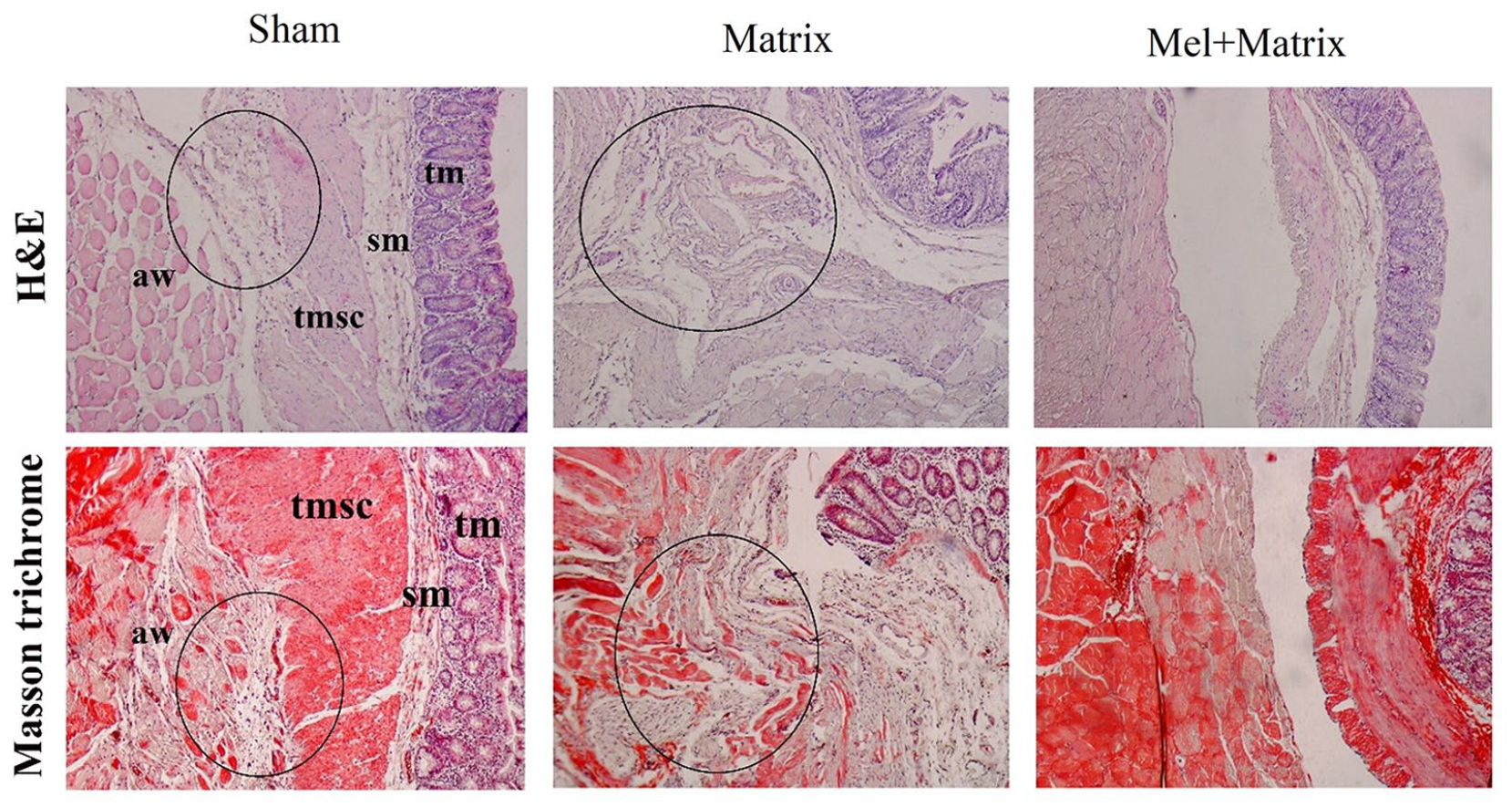
Representative images of H&E and MT staining.
Neovascularization is another parameter that was evaluated in this study. According to our results, neovascularization levels were significantly decreased in the Mel + Matriks group (0.33 ± 0.21) compared to the Sham (2.16 ± 0.30; p = 0.001). Similarly, neovascularization levels of the Matrix group were also decreased compared to Sham (p = 0.049). There was not a significant difference between the Matrix and Mel + Matrix groups (p = 0.121).
Inflammation was also determined histopathologically in this study. The inflammation levels of Matrix (1.33 ± 0.21) and Mel + Matrix (0.50 ± 0.34) groups were decreased compared to the Sham (2.66 ± 0.21) (p = 0.007 and 0.001, respectively). In addition, there was a decrease in inflammation levels in the Mel + Matrix group compared to the Matrix group, but the result is statistically insignificant (p = 0.095).
Discussion
In this study, we investigated the effects of the Mel-embedded PLGA-PEG6000 nanofiber matrix on experimental peritoneal adhesion in rats. PLGA-PEG6000 nanofiber matrix was fabricated by electrospun method and Mel was embedded with a final dose of 0.25 mg Mel per/matrix (2 × 2 cm2). Following fabrication, first matrix degradation profile and then the Mel release profile from the matrix were performed. After optimization of Mel release from the matrix, a 2 × 2 cm2 piece of the divided matrix was applied to an experimental adhesion model in rats. Our results indicated that Mel-embedded PLGA-PEG6000 nanofibers reduced adhesion formation. To the best of our knowledge, this is the first study in which Mel and PLGA-PEG6000 matrix combination was applied in an experimental adhesion model and alleviated adhesion formation at macroscopic and histopathology levels.
Abdominal adhesions are one of the most common postoperative surgical complications leading to functional disorders and need advanced surgical techniques or treatment methods for healing. Current approaches like various surgical techniques, therapeutic agents, antifibrinolytic agents, antibiotics, and mechanical barriers were reported as insufficient for the prevention of adhesions. Novel methods containing tissue engineering technologies have promising effects on the prevention of adhesion formation. In recent years biocompatibility and biodegradability of tissue-engineered physical barriers led them to be one of the rising trends in abdominal adhesion prevention. 35 However, adhesion formation is still a challenging complication of abdominal surgeries due to all the above-mentioned methods. In addition, newly developed biomaterials for the prevention of adhesions can not be fit into clinical use immediately.
Over the past 20 years, several biomaterials were developed for preventing adhesion formation.36 –38 Novel biomaterials present a convertible structure that is appropriate to the specificity of the pathology. Studies have shown that coating, seeding, or embedding various therapeutic agents makes the biomaterial more effective for the targeted treatment.39,40 Common features of these biomaterials are being biodegradable, biocompatible, bioresorbable, having non-toxic metabolic compounds, and providing controlled release. Controllable release from the biomaterial maintains an optional dose of the contents. Drugs, therapeutic agents, anti-inflammatory, antifibrinolytic, or antiadhesive agents could be embedded into biomaterials and the release of the above-mentioned agent could be controlled optionally.41,42 Based on that, we developed a Mel-embedded PLGA-PEG6000 biomaterial of which the controlled melatonin-releasing profile was examined for 15 days in the in vitro part of our study, which is in line with previous studies.12,43 The antiadhesive, antiinflammatory, and antifibrinolytic effects of Mel on adhesion formation are well known. So, in our study, Mel was preferred as an embedding and controlled release therapeutic agent into the PLGA-PEG600 matrix. Further, abdominal adhesions generally begin to form between 7 and 15 days following surgeries. 44 Studies pointed out that PLGA-PEG structured biomaterials were effective in the prevention of adhesion formation. However, it was also suggested that coating the matrix with. Thus, this study focused on the development of a biodegradable and bioresorbable nanofiber matrix with controlled release of Mel to ensure reduced tissue adhesion. In this study, the Mel release profile was examined for 100 h, and we observed that 7% of Mel was released at the first 8 h, and continued slightly increasing until the 18% release peak at the 100th h.
Biomaterials for the prevention of adhesions are still being developed, but present studies are commonly in vitro and there are limited studies that were applied in vivo.45,46 In our study after the fabrication, degradation, and drug release analysis in vivo experimental process was performed. Following the establishment of the adhesion model and application of the matrix, macroscopic degradation, and histopathological analysis were performed from the adhesion formation specimen which was harvested from the matrix-applied abraded area. Before in vivo studies precut matrix was sterilized using gamma sterilization (gamma rays), to prevent any infection-related interference/complications. Gamma sterilization was preferred as it was suggested previously that this method was more appropriate for the sterilization of such materials (PLGA, PLC, and PLA). 47 After the 21st day of the experiment degradation of the matrix and adhesion formation scores were evaluated. As it is clear from the previous studies that the matrix in the abraded area must be wholly degraded,48,49 our results indicated that the whole matrix of all groups was degraded in the abraded area at the end of 21 days. In vivo, part of the study was completed without any fatality, or infection, none of the rats exhibited fever or severe inflammatory reactions. Pure PLGA behaves as a physical barrier against adhesion formation.12,50 It has been demonstrated that PLA/PEG barriers are successful and safe in a human, but there is no consensus about their ease of application. Moreover, the matrix could be considered a solid material that isolates the injured area from the surrounding tissues. Novel studies indicated that the PEG-PLGA membranes restrict the fibrous connection between the injured abdomen and nearby tissues. 51 Recently, it was investigated that the composite PLGA-PEG biomaterials displayed good mechanical properties, thermostability, and hydrophilicity. Additionally, the PLGA-PEG membrane facilitated the exchange of nutrients and wastes through its microporous structure, accelerating the healing of the peritoneum. 45 Abdominal defects with cecum adhesion in rats demonstrated that the chitosan-PLGA matrix was effective in preventing tissue adhesion as a barrier and insufficient wound sites at the beginning of the wound healing process. 52 Previous studies suggested that PLGA application with an antiinflammatory agent might be more effective for the prevention of adhesion formation. The results of the previous animal adhesion model demonstrated that electrospun nanofibrous PLGA had anti-adhesive properties and suggested that these properties significantly improved after the incorporation of antiinflammatory agents in PLGA. 53 PLGA membrane was used to cover the wounds of rabbits. The results demonstrated that the PLGA membrane could be effective in preventing abdominal adhesions in rabbits, mostly involved in the reduction of fibrinogen efflux and inhibition of collagen fiber proliferation at the end of 2 weeks. 54 In a study using a rat peritoneal adhesion model macroscopic observation after 1 week has demonstrated that PLGA nanofiber membranes have a slight antiadhesion efficacy. 43 An adhesion induction model used in mice suggested that in considering the FDA-approved polymers, nanofibrous membranes prepared from PLGA represented the highest efficacy for the prevention of abdominal adhesion and held promising potential as a novel anti-adhesive agent. 55 According to the literature, in our study PLGA-PEG matrix itself slightly reduced the severity of adhesion macroscopically. Also, the matrix group indicated a reduction in neovascularization and inflammation levels histopathologically. Thus, we suggested that the PLGA-PEG matrix acted as a physical barrier and was slightly effective in adhesion formation.
In this study, we examined the intensity and severity of adhesion formation macroscopically by Leach scoring. According to our results, there was a decrease in the intensity of adhesion formation in the Mel + Matrix group compared to Sham. Similarly, the severity of adhesion was also decreased in the Mel + Matrix group. Novel studies indicated that PLGA nanofibers act like physical barriers and prevent adhesion formation. In a study, it was suggested that the PLGA membrane has good biocompatibility and alleviates tendon adhesion after injury in rats. 56 One of the studies indicated that PLGA nanofibers prevented adhesion formation and severity of adhesion in an abdominal adhesion model in rats. 53 Moreover, Mel is known as a scavenger for reactive oxygen species, an inhibitor of NF-κB and cytokine release, and a strong anti-inflammatory agent. 57 Previous studies reported that Mel is also effective in reducing adhesion formation and edema. 38
Inflammation and fibrin formation immediately emerge following peritoneal injury due to surgery, infection, or irritation. Activation of the coagulation cascade in the peritoneal cavity triggers the transition of fibrinogen into fibrin. 58 Further, the detrimental membrane phospholipids, the physical barriers of cell membranes, contribute to adhesion formation. 59 Angiogenesis is one of the other crucial actions in the development of adhesions because the extent of early neovascularization correlates with the severity of adhesion formation. 60 In our study to evaluate inflammation, fibrosis, and neovascularization, H&E and Masson’s Trichome stainings were performed. The Mel + Matrix group indicated a significant decrease in inflammation, fibrosis, and neovascularization levels compared to the SHAM and Matrix groups. Further neovascularization and fibrosis levels of H&E-stained specimens of the Matrix group indicated a significant decrease compared to Sham. Thus, it could be suggested that a physical barrier between the cecum and peritoneum might reduce adhesion formation. With this, we suggested that a matrix with/without Mel would be able to prevent adhesion formation as a physical barrier. All our microscopic and macroscopic outcomes correlate suggesting that the controlled release of Mel was effective in the prevention of adhesion formation. Moreover, our study was carried out for 21 days and fabricated matrix and Mel + Matrix biomaterial took action and were wholly effective during the hemostasis, inflammatory, and proliferation phases of wound healing. Our results indicated that Mel-embedded PLGA-PEG600 nanofiber alleviated the abdominal adhesion at macroscopic and microscopic levels.
In conclusion, this study provides a new idea for the use of PLGA/PEG absorbable electrospun polymer membranes in the clinical prevention of postoperative peritoneal adhesions, but further research on its cell adhesion, toxicity, and molecular mechanism is needed before clinical application. Our study investigated that Mel embedded matrix indicated lower clinical adhesion scores and less inflammation. Mel-embedded PLGA-PEG matrix facilitated diffusion of Mel into traumatized peritoneal tissues and contributed to the slower proliferation of fibroblasts to the injured area promoting the synthesis of less collagen and reducing adhesions.
Limitations and future directions
The priority of this study was to develop a novel matrix and investigate the preventive effects on adhesion formation. The effects of the matrix were evaluated by only histopathological and macroscopical levels. Our team further aimed to investigate the effects of the Mel-embedded PLGA-PEG6000 matrix with the additive of genetic analysis and cell experiment. Depending on our results, we suggested that the Mel-embedded PLGA-PEG6000 matrix produced promising effects on adhesion formation. Future studies might be established on the enhancement of antifibrotic and antiadhesive effects of our present matrix through embedding novel therapeutic agents.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Çanakkale Onsekiz Mart University Project Coordination Unit under Grant [number THD-2022-3995].
Availability of data
The data that support the findings of this study are available from the corresponding author.
