Abstract
Introduction
Eslicarbazepine (ESL) is a once-daily, third-generation antiseizure medication for focal-onset seizures. The primary mechanism of action is enhancing the slow inactivation of voltage-gated sodium channels. The study objective was to review real-world experience regarding retention rate, efficacy, and tolerability of eslicarbazepine, soon after it became available for children in Canada.
Methods
A retrospective review was performed on all patients prescribed eslicarbazepine from September 2017 to June 2020, with at least 3 years of follow-up data, at 3 Canadian tertiary care pediatric centers.
Results
Fifty patients were identified, and the mean age of eslicarbazepine initiation was 12.4 years (range 3-19 years). Most patients had drug-resistant epilepsy, trying a mean of 5.04 (range 0-14) antiseizure medications before the initiation of eslicarbazepine. Twenty-four patients (48.0%) experienced adverse effects, including dizziness (n = 10), drowsiness (n = 6), dizziness and drowsiness (n = 1), nausea and abdominal pain (n = 4), transient unsteadiness and diplopia (n = 1), and negative mood changes (n = 2). None had serious adverse effects, including rash. The retention rate of eslicarbazepine at last follow-up was 70%. Fifteen (30%) had ≥50% seizure reduction, with 2 of these patients becoming seizure free. Ten (20%) had 25% to 50% reduction, 2 (4%) had worsening of seizures, and 17 (34%) had no change in seizure frequency.
Conclusion
The study results support the long-term effectiveness and tolerability of eslicarbazepine in a cohort of children with predominantly drug-resistant epilepsy in a real-life setting from 3 Canadian centers with initial use after approval. Adverse effects were nonserious, infrequently leading to eslicarbazepine discontinuation.
Epilepsy is common in childhood, with focal seizures being the most common seizure type. 1 Epilepsy is associated with worse quality of life 2 and increased morbidity and mortality than the general population, including the risk of sudden unexplained death in epilepsy. 3 These issues, among others, have led to the continued development of antiseizure medications. The number of available antiseizure medications, with novel mechanisms of action, has progressively increased over the past decades. Most of them are characterized by more favorable pharmacokinetics and drug interaction profiles than first-generation antiseizure medications, 4 but efficacy profiles do not seem to be improved, with little change in the incidence of drug-resistant epilepsy. 5
Eslicarbazepine acetate (ESL) is a once-daily, third-generation antiseizure medication for focal onset seizures. It is a member of the dibenzazepine family of antiseizure medications, which also includes carbamazepine and oxcarbazepine. 6 The primary mechanism of action is enhancing the slow inactivation of voltage-gated sodium channels,6–8 which may dampen neuronal excitability. 7 Eslicarbazepine differs from carbamazepine and oxcarbazepine, which act primarily on fast channel inactivation. 8 The differences in pharmacokinetics, pharmacodynamics, and metabolism1,2 may have an impact on the effectiveness and tolerability of these antiseizure medications. 5
Five phase III clinical trials found that eslicarbazepine was effective and well tolerated in adults (18 years) with focal seizures and supported the approval of eslicarbazepine in the United States.9,10 A subset of data pooled from 2 European double-blind clinical trials (with open-label extensions) of adjunctive eslicarbazepine in children aged 2-18 years with focal seizures were used to support the FDA approval of eslicarbazepine for the treatment of focal seizures in children. 11 A post hoc analysis of the pooled data from patients aged 4-17 years in study 208 (NCT01527513) and study 305 (NCT00988156) were analyzed.11,12 Both were randomized, double-blind, placebo-controlled studies of eslicarbazepine as adjunctive treatment in pediatric patients with refractory focal seizures receiving 1 or 2 antiseizure medications. Adjunctive eslicarbazepine was generally well tolerated in children aged 4-17 years, with good safety profile.
In 2017, eslicarbazepine could be prescribed and funded via Pharmacare and private insurance plans in Canada. In September 2018, Health Canada approved eslicarbazepine for adjunctive therapy in the treatment of focal-onset seizures in children and adolescents. Notably, a meta-analysis reviewing data from randomized controlled trials for adjunctive treatment with eslicarbazepine for focal seizures did not find that eslicarbazepine resulted in a significant seizure reduction in children. It was concluded that efficacy and tolerability of eslicarbazepine as adjunctive therapy in children with refractory focal-onset epilepsy should be further evaluated in future studies. 13 It is recognized that RTCs of antiseizure medications may not fully reflect clinical practice as they require strict dosing schedules and adjustment of concomitant antiseizure medications is discouraged. Thus, the purpose of our study is to review real-world data regarding retention rate, efficacy, and tolerability of eslicarbazepine, soon after it became available for children in Canada, in 3 tertiary care pediatric centers.
Methods
All patients prescribed eslicarbazepine from September 2017 to June 2020 were identified at 3 Canadian tertiary care pediatric centers: BC Children's Hospital, The Children's Hospital Health Sciences Centre in Winnipeg, and the Children's Hospital London Health Sciences Centre. Research ethics approval was obtained at each site. Included patients were 19 years or younger at the time of initiation of eslicarbazepine and had continued follow-up at their respective institution for at least 3 years. Patients were identified by investigators from epilepsy clinics at each center. Furthermore, at BC Children's Hospital, patients were also identified by the EEG database, where clinical data, including antiseizure medications, are systematically documented in all patients who obtain an EEG.
At each center, detailed chart reviews of the entire medical record were performed on all patients meeting inclusion criteria. The same data collection form was employed at each center, which included age, sex, seizure duration, epilepsy etiology, family history of seizures, epilepsy surgery, number of antiseizure medications previously tried, concomitant antiseizure medications, and adverse effects. Patient seizure diaries and medical records were reviewed to collect data on seizure type (2017 ILAE classification 14 ) and seizure frequency. The latter was defined as the mean monthly seizure frequency since at least the prior visit (either 3 or 6 months) before starting study antiseizure medications and classified as daily (≥1 seizure per day), weekly (≥1 seizure per week), or monthly (<1 seizure per week). As the majority of patients in the study previously tried carbamazepine and oxcarbazepine, the retention, response rate, adverse effects, and reason for discontinuation of these antiseizure medications were documented. The percentage of patients still on therapy (retention rate) over at least 3 years after starting eslicarbazepine time was established as primary endpoint, because retention data represents a global indicator of a drug's clinical effectiveness. Most patients previously tried carbamazepine or oxcarbazepine; therefore, response to these antiseizure medications was also documented, including patients with >50% reduction in seizure frequency, 25% to 50% reduction in seizure frequency, exacerbation of seizures, and seizure freedom. Occurrence of adverse effects of carbamazepine, oxcarbazepine, and eslicarbazepine therapy and reasons for discontinuation of these antiseizure medications were noted, including seizure exacerbation, adverse effects, inefficacy, and noncompliance.
Neurodevelopment and epilepsy comorbidities, such as psychiatric conditions and autism spectrum disorder, were also reviewed. Developmental delay was defined as delay in meeting at least 1 developmental milestone. Standardized neuropsychological evaluations were not available for most patients, and chart reviews for comorbidities were predominantly based on reports from parents and clinicians).
Carbamazepine, oxcarbazepine, and eslicarbazepine titration and dosing schedules were decided by the primary epileptologist, based on recommended pediatric dosing and response and tolerability to the antiseizure medications. Dosages ranged from 10 to 30 mg/kg, with maximum doses of 1700 mg. When patients were switching from carbamazepine to eslicarbazepine, standard titration and weaning schedules were used for each drug. When switching from OZC to eslicarbazepine, some patients did a direct switch, whereas others used titration and weaning schedules, as per physician preference. Screening for hyponatremia was performed on the primary physician's discretion. Data were summarized using descriptive statistics including means and ranges for continuous variables and frequencies and percentages for categorical variables.
Results
Between September 2017 to June 2020, 50 patients were initiated on eslicarbazepine at the 3 Canadian centers. Thirty-five patients were from BC Children's Hospital, 8 from Children's Hospital in Winnipeg, and 7 from the Children's Hospital London Health Sciences Centre. Refer to Table 1 for clinical features. All patients had at least 3 years of follow-up, with the mean age at follow-up being 14.5 years (range 3-22 years of age, CI 5.0-23). Twenty-five (50%) were female and 32 (64%) had a history of developmental delay ranging from mild to severe; 8 (16%) were diagnosed with autism spectrum disorder. Twenty-three (46%) had a previous history of neuropsychiatric comorbidities, which included anxiety and depression. The median age of seizure onset was 5.1 years (range 1 month to 16.1 years, CI 0.0-14). All patients had focal seizures, with 15 patients having focal to bilateral tonic-clonic seizures, 2 focal tonic, and 2 focal epileptic spasms.
Clinical Features of Patients on Eslicarbazepine.
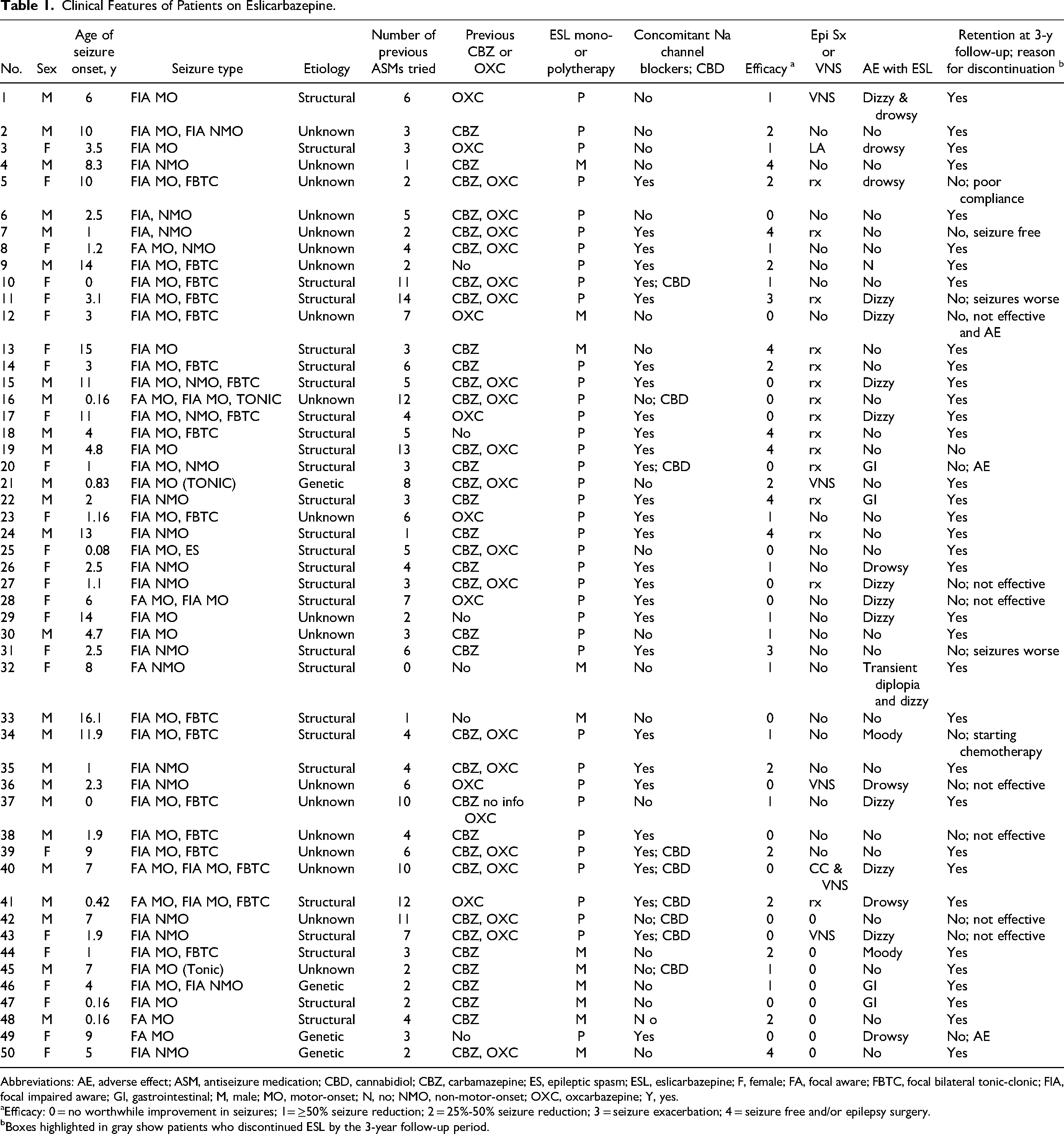
Abbreviations: AE, adverse effect; ASM, antiseizure medication; CBD, cannabidiol; CBZ, carbamazepine; ES, epileptic spasm; ESL, eslicarbazepine; F, female; FA, focal aware; FBTC, focal bilateral tonic-clonic; FIA, focal impaired aware; GI, gastrointestinal; M, male; MO, motor-onset; N, no; NMO, non-motor-onset; OXC, oxcarbazepine; Y, yes.
Efficacy: 0 = no worthwhile improvement in seizures; 1= ≥50% seizure reduction; 2 = 25%-50% seizure reduction; 3 = seizure exacerbation; 4 = seizure free and/or epilepsy surgery.
Boxes highlighted in gray show patients who discontinued ESL by the 3-year follow-up period.
With regard to seizure etiology, 27 (54%) had a structural etiology, such as malformation of cortical development on neuroimaging, 4 (8%) had an identified genetic etiology, and the remainder, 19 (38%), had unknown etiology. A family history of epilepsy was present in 16 (32%).
Most patients (n = 45, 90%) tried carbamazepine and/or oxcarbazepine before initiation of eslicarbazepine. Fifteen (40%) patients were treated with carbamazepine, 8 (16%) with oxcarbazepine, and 20 (40%) tried both carbamazepine and oxcarbazepine before starting eslicarbazepine. Of these patients, 19 (38%) were treated with carbamazepine, followed by oxcarbazepine. Only 5 of the patients (10%) did not try carbamazepine or oxcarbazepine, before initiating eslicarbazepine. Refer to Figure 1 for a summary of response and tolerability to carbamazepine, oxcarbazepine, and eslicarbazepine. Most patients had drug-resistant epilepsy, with a mean number of antiseizure medications before eslicarbazepine of 5 (range 0-14). Only 1 patient (2%) did not try other antiseizure medication before initiation of eslicarbazepine. Three patients (3 of 50, 6%) had tried the ketogenic diet before eslicarbazepine, 15 (30%) patients underwent resective epilepsy surgery, 1 had laser ablation of a nodular heterotopia, 4 patients had a vagal nerve stimulator, and 1 had a vagal nerve stimulator and a corpus callosotomy.
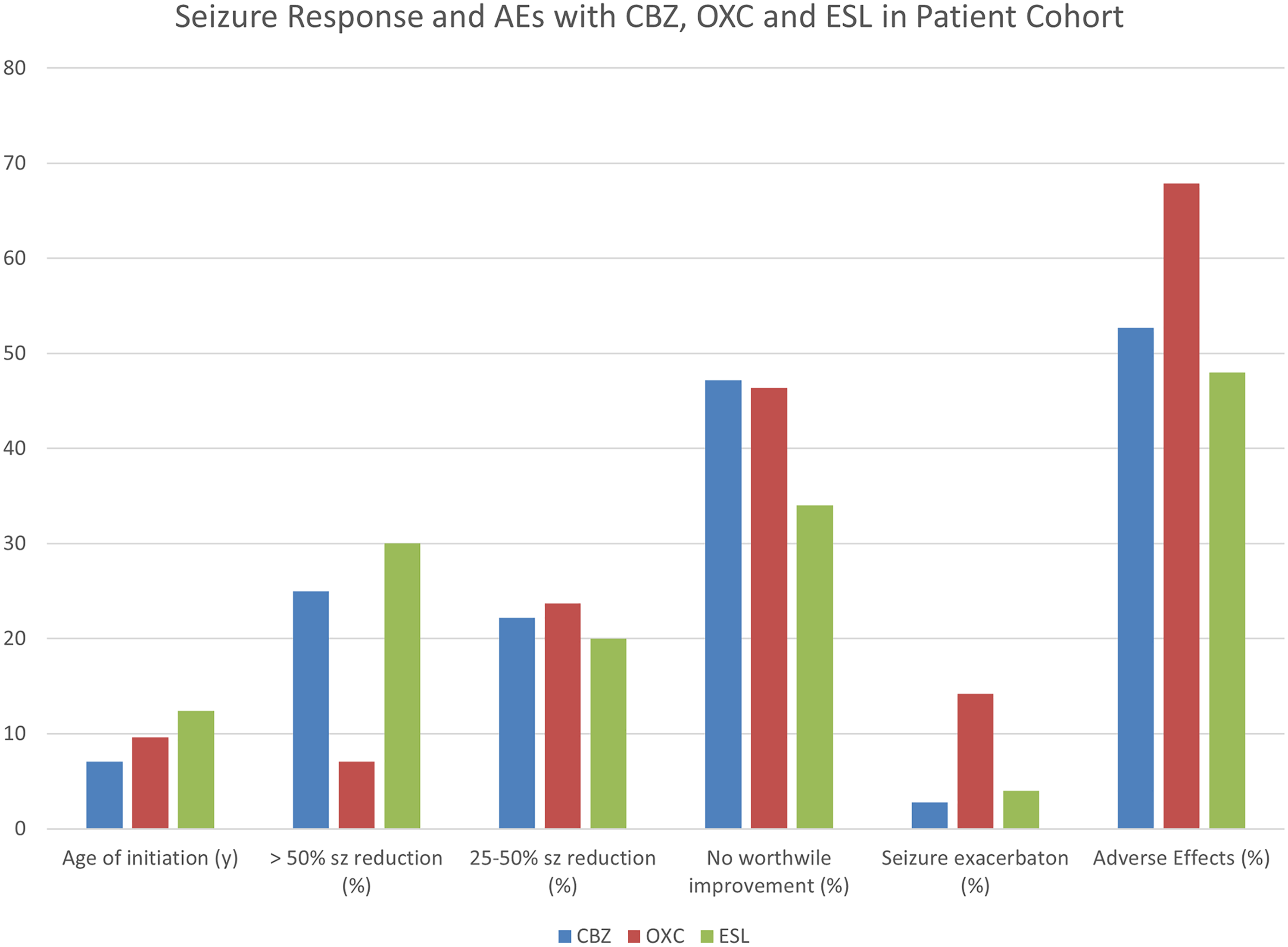
Seizure response and adverse effects with CBZ (carbamazepine), OXC (oxcarbazepine), and eslicarbazepine (ESL) in the patient cohort. Fifteen patients (40%) were treated with CBZ, 8 (16%) with OXC, and 20 (40%) tried both CBZ and OXC before starting eslicarbazepine. Only 5 of the patients (10%) did not try CBZ or OXC before initiating eslicarbazepine.
Carbamazepine
The mean age of carbamazepine initiation was 7.1 years (range: 0.83-16 years, CI 0-16.9). Most of the patients (n = 24, 66.7%) started carbamazepine within 1 year of seizure onset. Of the 36 patients treated with carbamazepine, 19 (52.7%) reported adverse effects. Twenty-four of the patients (67%) discontinued carbamazepine. Regarding the reason for discontinuation of carbamazepine, 14 (38.9%) had lack of response and 10 (27.8%) experienced adverse effects. On carbamazepine, 9 (25%) patients reported a >50% reduction of seizures, 8 (22.2%) reported a 25% to 50% seizure reduction, 1 (2.8%) experienced seizure exacerbation, 17 (47.2%) found it ineffective, and 1 (2.8%) became seizure free temporarily and weaned the medication.
Oxcarbazepine
The mean age of oxcarbazepine initiation was 9.6 years, and 5 (17.9%) started it within 1 year of seizure onset. Of 28 patients treated with oxcarbazepine, 19 (67.9%) reported adverse effects. Twenty-five patients discontinued oxcarbazepine for various reasons, including 10 (35.7%) with lack of response, 6 (21.4%) having adverse effects, 7 (25%) with both lack of response and adverse effects, and 2 (10%) noncompliant. Overall, 13 (46.4%) discontinued the oxcarbazepine in relation to adverse effects. Regarding seizure outcome on oxcarbazepine, 2 patients (7.1%) had ≥50% reduction, 9 (23.7%) had 25% to 50% seizure reduction, and 4 (14.2%) had seizure exacerbation. Thirteen (46.4%) found oxcarbazepine ineffective.
Eslicarbazepine
The mean age of eslicarbazepine initiation is 12.4 years of age (range 3-19 years). In the majority, eslicarbazepine was started late in the course of epilepsy, with only 5 (10%) having started eslicarbazepine within 1 year of seizure onset. Most patients had drug-resistant epilepsy, trying a mean of 5.04 (range 0-14) antiseizure medication before the initiation of eslicarbazepine, 39 (78%) on polytherapy while on eslicarbazepine, and 11 (22%) on eslicarbazepine monotherapy. All but 1 patient was treated with other antiseizure medications before eslicarbazepine. The most frequent reason for initiating eslicarbazepine was lack of effectiveness of previous treatment, 28 (56%); followed by poor tolerance to previous treatment, 11 (22%). In addition, 4 of the patients (8%) tried eslicarbazepine because of lack of effectiveness plus poor tolerance to previous treatment, and 7 of the patients (14%) initiated eslicarbazepine because of other reasons (most commonly, lack of compliance with previous treatment [
The most common concomitant antiseizure medications were lamotrigine (19), clobazam (13), and lacosamide (9). Of the 39 patients (78%) on polytherapy, 29 (58%) were on concomitant sodium channel–blocking antiseizure medications. Regarding concomitant antiseizure medications of the same family as eslicarbazepine, 1 patient was on carbamazepine and none were on oxcarbazepine.
On eslicarbazepine, 24 of the patients (48.0%) experienced adverse effects. Ten experienced dizziness, 6 had drowsiness, 1 had both dizziness and drowsiness, 4 had nausea or abdominal pain, 1 had acute unsteadiness and diplopia that resolved in a few days, and 2 had negative impacts on mood, including irritability and sadness. Adverse events were observed in 5 patients with monotherapy and 19 patients on polytherapy. Nine (18%) were on concomitant cannabidiol oil, of which 4 (44.4%) reported adverse effects. Only 1 patient on concomitant cannabidiol discontinued eslicarbazepine for nausea and abdominal pain. None of the patients developed a rash with eslicarbazepine. One patient had a rash with carbamazepine and 1 with oxcarbazepine, but these patients did not develop a rash with eslicarbazepine. None of the study patients developed hyponatremia on carbamazepine, oxcarbazepine, or eslicarbazepine.
The mean duration of eslicarbazepine was 3.15 years. The retention rate of eslicarbazepine was high, with 35 (70%) of patients still on eslicarbazepine at their last follow-up. Fifteen patients (30%) discontinued eslicarbazepine. Reasons for discontinuation include inefficacy in 7 (46.7%), adverse effects in 2 (13.3%), inefficacy and adverse effect in 1 (6.7%), poor compliance in 1 after successful resective surgery (6.7%), and seizure exacerbation in 2 (13.3%). One patient (6.7%) discontinued it because they were starting chemotherapy and wanted to avoid an enzyme-inducing agent. One patient achieved sustained seizure freedom with epilepsy surgery and discontinued it for that reason (6.7%). Adverse effects that led to discontinuation of eslicarbazepine in the 3 patients (20%) included dizziness, nausea and abdominal pain, and drowsiness, of whom 2 patients were on concomitant sodium channel blockers. Of all patients who discontinued eslicarbazepine for various reasons, all but 2 were on concomitant sodium channel–blocking agents. Those not on sodium channel blockers that discontinued eslicarbazepine did so because of inefficacy and dizziness in one individual and inefficacy in another.
Regarding seizure response with eslicarbazepine, 15 (30%) had ≥50% seizure reduction, with 2 of these patients becoming seizure free. Six patients had Engle I outcomes after epilepsy surgery, before starting eslicarbazepine, and continued on eslicarbazepine for ongoing seizure protection or treatment of focal wave seizures (auras). An additional patient started eslicarbazepine after a parietal cavernoma resection, became seizure free after surgery, and subsequently discontinued eslicarbazepine. Ten (20%) had 25% to 50% reduction, 2 (4%) had worsening of seizures, and 17 (34%) had no change in seizure frequency. Refer to Figure 2 for a summary of response, adverse effects reasons for discontinuation of eslicarbazepine.
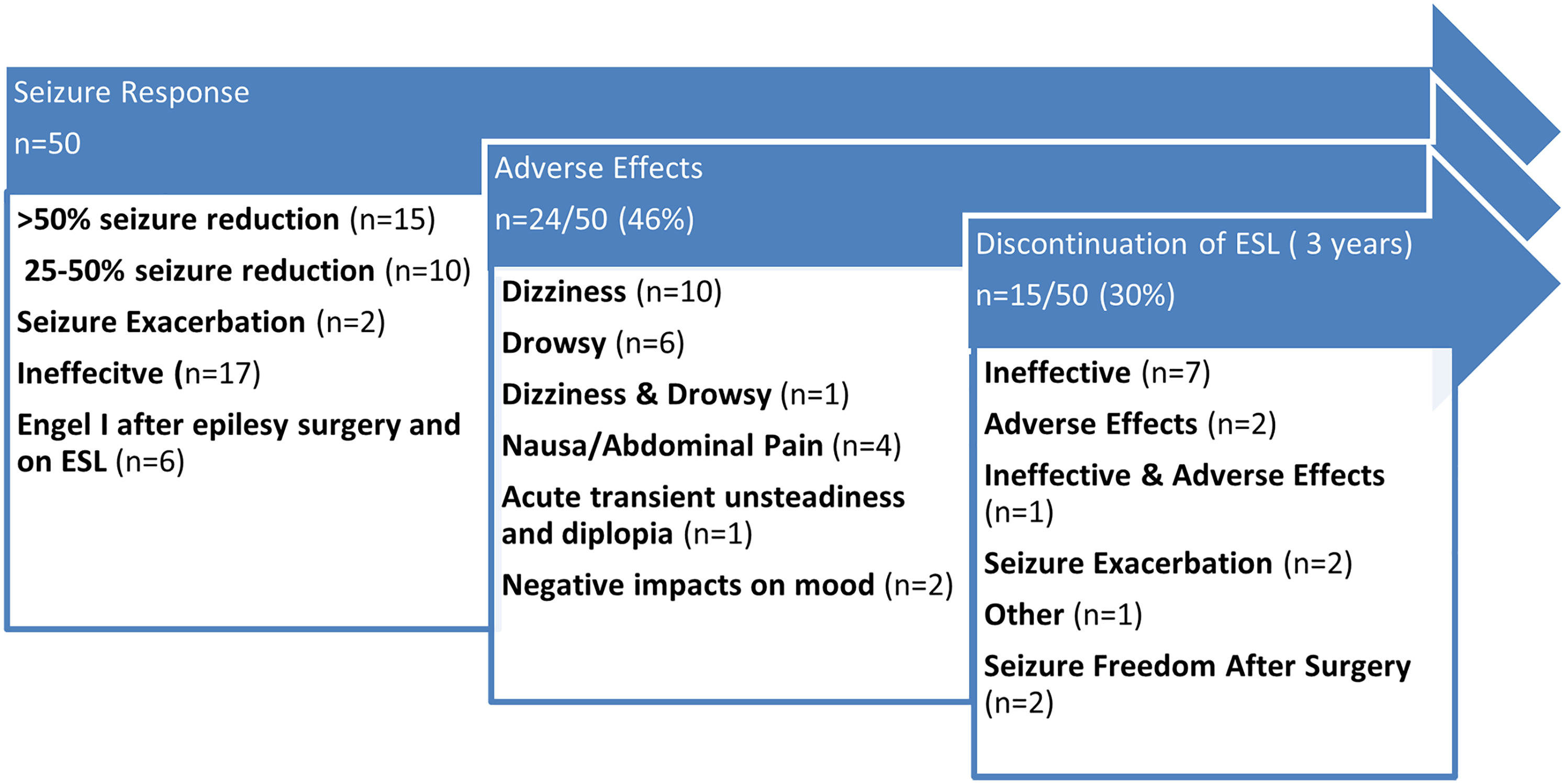
Seizure response, adverse effects, and reasons for discontinuation of eslicarbazepine.
Discussion
This multicenter, retrospective study provides real-world data on the effectiveness, safety, and tolerability of eslicarbazepine in children with epilepsy in the early days in Canada. Our cohort consisted of 50 pediatric patients from 3 Canadian centers, at the time eslicarbazepine first became available for children in Canada. Most patients (90%) also had previously tried carbamazepine and oxcarbazepine, which are drugs in the same family as eslicarbazepine. The patients predominantly had drug-resistant epilepsy, because often the most refractory and challenging patients are selected first to initiate antiseizure medications when newly available on the market. The results show that, in at least 3 years of follow-up, a third of children achieved a ≥50% reduction in seizures, and adverse effects were mild to moderate, infrequently leading to eslicarbazepine discontinuation. Efficacy and tolerability were demonstrated by the high retention rate of 70%.
Efficacy
Although eslicarbazepine was initiated late in the course of the epilepsy, with most patients having tried a mean of 5 antiseizure medications before eslicarbazepine, 30% had >50% reduction, and 20% had 25% to 50% reduction in seizures with eslicarbazepine. Effectiveness in the real-world setting was investigated via the Euro-Esli study, based on the largest database of patients treated with eslicarbazepine in everyday clinical practice across Europe (N = 2058), mostly confirming the results from clinical trials. 15 In this study, the responder rate was 75.6% at 12 months and 63.6% at the last visit, which ranged up to 3 years. The seizure freedom rate was 41.3% at 12 months and 32.6% at the last visit. 15 The response rate in this present study is lower for several reasons; previous predictors for response to eslicarbazepine include being on fewer than 2 concomitant antiseizure medications with eslicarbazepine. 15 In our study in contrast, 78% were already on more than 2 concomitant antiseizure medications. Although our cohort is too small to stratify response to treatment based on the number of concomitant antiseizure medications, the 2 patients that became seizure-free on eslicarbazepine, without epilepsy surgery, were on monotherapy. Another known favorable predictor of response to eslicarbazepine is absence of concomitant sodium channel blockers. 15 In our study, more than half, 58%, were on concomitant sodium channel blockers, which may have affected response rates.
In addition to concomitant antiseizure medications, better response to eslicarbazepine has been shown in patients treated relatively early in their disease course and in those who are less refractory to treatment, such as older age at onset of epilepsy and absence of seizures at baseline. 16 Our study patients predominantly consisted of children with drug-resistant epilepsy, and eslicarbazepine was started relatively late in their epilepsy course. The median age of seizure onset was 5.1 years, but the mean age of eslicarbazepine initiation was 12.4 years. Also, our study cohort included children who were refractory to treatment, with the mean number of previously tried antiseizure medications being 5. It is well established that the probability of seizure freedom diminishes progressively with successive antiseizure medication regimens, whether substitution or add-on therapy, particularly after failure of 2 regimens. 17 The fact that 30% had >50% seizure reduction with eslicarbazepine, with 2 seizure free without epilepsy surgery, demonstrates that eslicarbazepine should still be considered for improved seizure control in such patients. Another factor for a lower seizure response rate in this cohort compared with other real-world studies could be that one study observed that eslicarbazepine was more effective in patients without intellectual disability. 18 In our study, the majority, 64%, had a history of developmental delay, and 16% were diagnosed with autism spectrum disorder.
Furthermore, the overall response rate could be lower than other studies, as this is a pediatric cohort. Response to eslicarbazepine appears to be age dependent. Patients older than 65 years of age have a better response rate. 15 Also, the efficacy of eslicarbazepine in the pediatric population is not as robust as in adults. A meta-analysis of response to eslicarbazepine in adults and children, where children did not have a significant seizure reduction. 13 For children, the efficacy of eslicarbazepine as an adjunctive therapy for the treatment of focal-onset seizure in children was evaluated in a phase II trial (study 208), 11 which was primarily designed to evaluate the drug's effects on cognitive function, and a phase III trial (study 305). 12 Both studies were randomized, double-blind, and placebo-controlled.11,12 Enrolled children were aged 6-16 years 11 or 2-18 years, 12 had refractory focal-onset seizures and were receiving 1 to 2 antiseizure medications (except oxcarbazepine). In study 208, adjunctive eslicarbazepine was associated with significant benefits over placebo in terms of response rate. 11 However, in study 305, the differences in responder rate between the eslicarbazepine and the placebo groups were not statistically significant. 12 A post hoc subgroup analysis demonstrated that response to eslicarbazepine in children was age and dose dependent. 12 Patients 7-18 years of age, with higher dosing (30 mg/kg) had significant seizure reduction in comparison to placebo. In our cohort, dosing was based by guidelines at the discretion of the primary neurologist and not necessarily at 30 mg/kg. Seven of the children were <7 years of age at the time of eslicarbazepine initiation, which could have affected the response rates.
Adverse Effects
In this study, 48.0% of patients on eslicarbazepine experienced adverse effects, including dizziness, drowsiness, nausea or abdominal pain, and negative impacts on mood. Despite nearly half experiencing adverse effects, only 3 patients discontinued eslicarbazepine for adverse effects, specifically. There are marginal differences in adverse effects in those on monotherapy (36%) or polytherapy (56%). Therefore, consistent with other studies, most adverse effects were mild or moderate and based on the high retention rate, and more common early on. Adverse effects due to antiseizure medications mostly appear during initiation and early treatment, and some of them tend to regress spontaneously thereafter. In a study that reviewed eslicarbazepine, lacosamide, and Perampanel use, longer follow-up was associated with a reduced risk of adverse effect occurrence. All the adverse effects reported were mild or moderate in severity and did not differ from previously reported adverse effects. 19
In multinational phase III trials in children, the known safety profile of eslicarbazepine was confirmed, without any new findings of real concern.11,12 Long-term safety of adjunctive eslicarbazepine in children was examined by reviewing 2 long-term open-label extension studies of adjunctive eslicarbazepine in children with focal seizures. eslicarbazepine treatment was generally well tolerated in pediatric patients aged 4-17 years with focal seizures, and adverse effects were comparable to those observed in adults, with no new events of concern. 20 In the Euro-Esli study, 34.0% of patients reported adverse effects, and 13.6% discontinued treatment. 15 Similar to our cohort, most frequently reported adverse effects were dizziness (6.7%), fatigue (5.4%), and somnolence (5.1%).
Only 2 patients (4%) in this study experienced negative changes in mood, which is a favorable aspect of eslicarbazepine. 15 Severe described adverse effects with eslicarbazepine include hyponatremia and rash. In clinical trials, 1.2% experienced hyponatremia. The risk is thought to be higher in those older than 65 years of age. In the Euro-Esli study, hyponatremia was observed in 3.5%. 15 It is reassuring that none of the patients experienced significant hyponatremia. This could be related to the fact of a small cohort of children. No patient in the study developed a rash. Notably, developing a rash with other antiseizure medications can have cross-reactivity and increase the risk of rash with eslicarbazepine. One patient had a rash with carbamazepine and 1 with oxcarbazepine, but these patients did not develop a rash with eslicarbazepine. Two experienced seizure exacerbation and discontinued eslicarbazepine, lower than the Euro-Esli study of 7.5% at 12 months.
Nine of the study patients were on concomitant cannabidiol and eslicarbazepine. In adults, increases in serum level of eslicarbazepine can be associated with increasing cannabidiol doses. 21 We did not test cannabidiol serum levels or have standardized forms or doses of cannabidiol in our cohort. Only 1 patient on cannabidiol discontinued eslicarbazepine because of nausea and abdominal pain. Therefore, further information is needed in children of the interaction.
Two of our patients experienced seizure exacerbation and discontinued the drug, marginally lower than the Euro-Esli study of 7.5% at 12 months. 15
Retention
In the present study, the retention rate for eslicarbazepine was 70% over at least 3 years. In the majority of patients, eslicarbazepine was initiated because of lack of effectiveness of previous antiseizure medications, or poor tolerance to previous treatment. These patients predominantly had drug-resistant epilepsy and had tried a mean of 5 antiseizure medications, and 90% of them had previously tried carbamazepine or oxcarbazepine. Despite these characteristics of the cohort, the retention rate is similar to those previously reported with eslicarbazepine in adults and children. Our results are also similar to those of studies specifically in children. The study reviewing the long-term therapeutic effect of eslicarbazepine in children in an open-label extension of a cognition study in children aged 6-16 years found that the majority of patients remained on eslicarbazepine during the 2-year open label extension and treatment efficacy was maintained. 22 Adverse effects were consistent with the known safety profile of eslicarbazepine. This high retention rate confirms the tolerability and safety profile that we observed. A potential advantage of eslicarbazepine in the pediatric setting is that it is administered once daily, reducing medication burden and increasing likelihood of treatment adherence, in comparison with agents requiring multiple dosing per day. Fifteen of the study patients underwent resective epilepsy surgery, of which 6 had Engle 1 outcome. Eslicarbazepine was maintained for ongoing seizure protection, as it can be a gradual process to wean multiple antiseizure medications after surgery. Also, some patients continued to have FA seizures (auras), requiring antiseizure medication. Once-daily dosing likely may have increased compliance in these patients.
This study was too small to stratify for risk factors for retention. A recent study reviewing third-generation antiseizure medications found that females treated with eslicarbazepine had a 5 times greater dropout risk than men, and people of older age have higher retention rates.
19
An open-label pediatric study noted that retention at 12 months was associated with shorter duration of epilepsy, absence of any seizures at baseline, lower baseline seizure frequency (
Study limitations include retrospective design. Therefore, consistent baseline pre-treatment data and systematic seizure diaries to document seizure frequency were not possible. There were also variable differences in the dosing instructions, clinic follow-ups, titration schedules, response to previous antiseizure medications, and inconsistent adverse effect monitoring, which introduces various biases, namely, reporting bias that can limit the generalizability of the study. In this study, the early experience of eslicarbazepine is described and most of the initial patients had drug-resistant epilepsy and had tried carbamazepine and oxcarbazepine. Therefore, it is not reflective of all children who are currently prescribed eslicarbazepine. Also, some patients had epilepsy surgery at variable times during their epilepsy course, which could impact later responses to antiseizure medications.
Conclusion
At present data, current RTC data suggest that the efficacy of eslicarbazepine in children with focal epilepsy should be further evaluated. Our retrospective cohort supports the long-term effectiveness and tolerability of eslicarbazepine in a cohort of children with epilepsy in a real-life setting from 3 Canadian centers with initial use after approval. Despite initiation of eslicarbazepine late during the epilepsy course, the high number of concomitant antiseizure medications (more than half on sodium channel–blocking agents), inclusion of young children (<7 years old), and the high prevalence of drug-resistant epilepsy (most trialled > 5 antiseizure medications), 30% had a significant seizure reduction rate, with a 70% retention rate at 3 years. Adverse effects are nonserious, infrequently leading to eslicarbazepine discontinuation.
Footnotes
Acknowledgments
We thank Jeffrey Zhi for his help with identifying patients from the EEG database at BC Children's Hospital, and all neurologists at each institution who clinically managed these patients and helped to identify them for the study.
Author Contributions
YN did a literature review on the subject. AD developed the original concept and study design of the manuscript, prepared the figures, and drafted the manuscript. YN, QX, MN, and DH obtained clinical data by performing a detailed chart review. QX, MN, and AD identified eligible patients at their institute/center. AD and QX analyzed the data. All authors reviewed and edited it for important intellectual content. All authors gave approval to the final version of the manuscript to be submitted and all authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Approved by University of British Columbia Ethics Board H20-00629, University of Manitoba Research Ethics Board HS24660 (H2021:068), and Western University Research Ethics Board (116564) and Lawson Research Institute (R-20-543).
