Abstract
Introduction
The aim of this study was to evaluate genetic risk factors in term-born children with antenatal periventricular hemorrhagic infarction (PVHI), presumed antenatal periventricular venous infarction and periventricular hemorrhagic infarction in preterm neonates.
Methods
Genetic analysis and magnetic resonance imaging were performed in 85 children: term-born children (≥36 gestational weeks) with antenatal periventricular hemorrhagic infarction (n = 6) or presumed antenatal (n = 40) periventricular venous infarction and preterm children (<36 gestational weeks) with periventricular hemorrhagic infarction (n = 39). Genetic testing was performed using exome or large gene panel (n = 6700 genes) sequencing.
Results
Pathogenic variants associated with stroke were found in 11 of 85 (12.9%) children with periventricular hemorrhagic infarction/periventricular venous infarction. Among the pathogenic variants,
Conclusions
Children with periventricular hemorrhagic infarction/periventricular venous infarction have high prevalence of pathogenic variants in collagene genes (
Periventricular hemorrhagic infarction (PVHI) is a well-described pathology among extremely premature infants.1–3 The mechanism behind periventricular hemorrhagic infarction in prematurely born infants is germinal matrix and intraventricular hemorrhage leading to periventricular venous obstruction and congestion and ending in periventricular infarction.3–7 On cerebral ultrasonography, periventricular hemorrhagic infarction is usually visible as porencephalic cavitation of various severity within the periventricular white matter. 3 On magnetic resonance imaging (MRI), periventricular hemorrhagic infarction is evaluated as prevalently unilateral medullary venous territory infarction in the periventricular area during the acute stage and as a porencephalic enlargement of the lateral ventricle of various severity, and/or periventricular gliosis and/or cystic changes with a spared cortex during the chronic stage.8–12 Germinal matrix hemorrhage is especially common in premature infants born below 32 gestational weeks because of the suggested fragile state of the vessel wall in the germinal matrix.3,8,13,14
Antenatal periventricular hemorrhagic infarction may be detected by fetal ultrasonography incidentally15,16 and probably reflect the same pathologic mechanism as is seen in preterm-born children.10,17–19 If the child manages to be born at term, there may be no neurologic symptoms after birth, as the stroke is already in the chronic phase. 16
Some children with undetected antenatal stroke survive and stabilize. After an unremarkable term birth and neonatal period, chronic signs of stroke are found on MRI later during infancy if spastic hemiparesis, delayed milestone, or seizures develop.8–10,12,14,17,20–23 Stroke in these children is presumed to have occurred in the antenatal period and MRI suggests periventricular venous infarction (PVI) as the causative pathology in two-thirds of those cases, although the hemorrhage is not always found on MRI.9,24
The injury visualized on MRI during childhood both in term-born children with presumed antenatal periventricular venous infarction and in preterm-born children with periventricular hemorrhagic infarction is similar. 10 MRI findings suggest that presumed antenatal periventricular venous infarction occurs at a certain stage of vascular development of the brain at gestational weeks 24-34 with germinal matrix hemorrhage as the source.3,8,11,13 Vascular disruption and development of periventricular hemorrhagic infarction before gestational week 24 can lead to schizencephaly, and after gestational week 24 can lead to porencephaly19,25–29 the severity of which depends on the size of initial stroke. Clinically, periventricular hemorrhagic infarction/periventricular venous infarction leads to congenital hemi- or tetraparesis of various severity, epilepsy, and cognitive disability.22,29–34 The outcome depends on the initial size of the periventricular hemorrhagic infarction/periventricular venous infarction.3,34–37
Research of the risk factors for presumed antenatal periventricular venous infarction has been inadequate and evaluation of the causes of germinal matrix hemorrhage in fetuses is still insufficient.8,9,14,17,20 Recent research shows that prenatal infection and chorioamnionitis may play a critical role in some term-born children with presumed antenatal periventricular venous infarction.11,38 Previously suspected thrombophilia risk is not increased in children with neonatal or presumed arterial ischemic stroke, or with presumed antenatal periventricular venous infarction. 39 A recent meta-analysis of antenatal periventricular hemorrhagic infarction studies showed typical pregnancy without co-occurring conditions in 39% of cases. 40
In children with antenatal intracerebral hemorrhage, genetic profiling has suggested presence of impaired hemostatic, prothrombotic, collagen, and X-linked
The aim of this study was to evaluate the variants causing genetic collagenopathies and coagulopathies in term-born children with antenatal periventricular hemorrhagic infarction and presumed antenatal periventricular venous infarction, as well as in preterm-born children with periventricular hemorrhagic infarction.
We hypothesize that children with periventricular hemorrhagic infarction/periventricular venous infarction show high prevalence of collagenopathies, such as has been found earlier in fetuses and in children with porencephaly.
Methods
Patient Selection
The data of term or near term-born children (≥36 gestational weeks) with antenatal periventricular hemorrhagic infarction (n = 6) diagnosed during the fetal period, or with presumed antenatal periventricular venous infarction (n = 43) diagnosed during infancy were drawn from the Estonian Pediatric Stroke Database for 1994-2019. The Estonian Pediatric Stroke Database has been created retrospectively since 1994 and prospectively with continuously added cases since 2003.9,11,21,22 The vascular classification of patients in the Estonian Pediatric Stroke Database is based on MRI according to Kirton and coauthors.9,12,14 Chronic changes after periventricular hemorrhagic infarction/periventricular venous infarction are described on MRI, based on this classification, 12 as a medullary venous territory infarction in the periventricular area, with a porencephalic enlargement of the lateral ventricle, and/or periventricular gliosis and/or cystic changes, spared cortex, and presence of hemosiderin in the area of the germinal matrix and in the ventricles and/or in periventricular lesion.8–12 The radiologic images for the present study were reviewed by radiology resident and 2 neuroradiologists to confirm the vascular genesis of periventricular venous infarction. Evaluation of the MRI findings, including the size of the basal ganglia and thalamus (normal/mild or moderate/ severe decrease in the size) and enlargement of the lateral ventricles (normal/mild or moderate/severe enlargement) was performed.
Preterm-born patients (<36 gestational weeks) with periventricular hemorrhagic infarction (n = 74) in the Estonian Pediatric Stroke Database were identified from the clinical database of a third-level regional university hospital, accounting for about one-third of the neonatal cases in the country. The medical records of neonatal intensive care unit of the hospital were searched for children with the diagnosis of severe grade of intraventricular hemorrhage (grade III or IV) or periventricular hemorrhagic infarction/periventricular venous infarction1,3,50 treated at the hospital between 2002 and 2019. Also the hospital's electronic database between 2002-2019 was searched for
Children were excluded from the study if there occurred at least 1 of the following: (1) acute arterial ischemic stroke, (2) sinovenous thrombosis, (3) hemorrhage in the brain parenchyma without association with germinal matrix hemorrhage or intraventricular hemorrhage, or (4) other documented diseases or congenital malformations involving the central nervous system.
The flowchart of the inclusion criteria for the final study group of periventricular hemorrhagic infarction/periventricular venous infarction children (n = 85) is shown in Figure 1. All participants were Caucasian.
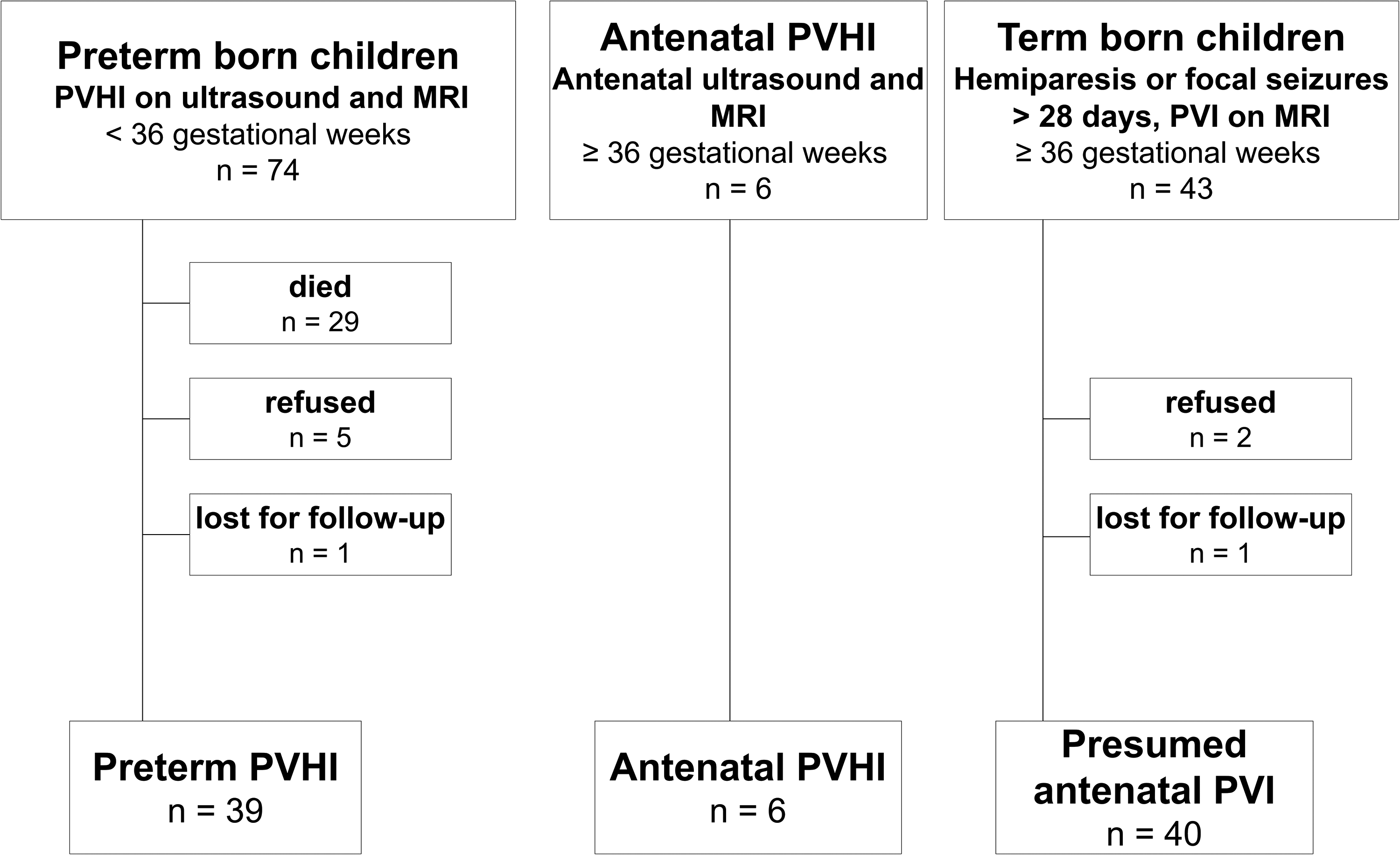
The flowchart of the inclusion criteria for periventricular venous infarction children for genetic testing.
Methods
The clinical data of pregnancy and delivery were extracted from medical records. For term-born children with antenatal periventricular hemorrhagic infarction or presumed antenatal periventricular venous infarction from 1994 to 2019 and for preterm children with PHVI since 2002, genetic testing was suggested as part of a larger follow-up study of perinatal stroke patients. The neurologic motor deficit was evaluated and defined according to the Surveillance of Cerebral Palsy in Europe criteria 52 : children with persisting increased tone and pathologic reflexes, resulting in an abnormal pattern of movement and posture, were considered to have unilateral or bilateral spastic cerebral palsy. Severity of the motor deficit was evaluated according to the Pediatric Stroke Outcome Measure (PSOM) 53 by the child neurologist (R.L.): mild, no impact on function; moderate, some functional limitations; severe, missing function.
Epilepsy was diagnosed by the treating pediatric neurologist according to the following definition: at least 2 unprovoked seizures occurring >24 hours apart or 1 unprovoked seizure with high recurrence risk, or diagnosis of the epilepsy syndrome. 36
Genetic Testing
Molecular screening was carried out on DNA extracted from the leukocytes of blood samples, with next-generation sequencing methods. In 60 individuals, whole exome sequencing was performed with Agilent SureSelectXT Human All Exon V7 library capture followed by sequencing on the NovaSeq 6000 platform (Illumina Inc.). The TruSight One Expanded panel covering the 6700 Mendelian disease-associated genes was sequenced on the Illumina NextSeq platform in a clinical setting in 23 individuals. In addition, 2 patients had received genetic diagnoses through single gene clinical testing before next-generation sequencing became available. Raw sequencing reads were aligned to the hg19 reference genome using the BWA MEM 54 algorithm. Variants were first called for individual samples using GATK HaplotypeCaller, 55 before multisample joint aggregation and reannotation using GATK GenotypeGVCFs. Read alignment and variant calling was carried out in the High Performance Computing Center of the University of Tartu “UT Rocket.” 56 The variant call set was annotated using hail 0.1 (Hail Team. Hail 0.1. https://github.com/hail-is/hail) and then uploaded to the Seqr platform (https://github.com/broadinstitute/seqr) for collaborative variant analysis. Variant filtration focused on rare variants in coding regions and on splice-sites predicted to affect protein function following the known inheritance pattern in 2 gene lists: (1) genes associated with stroke or associated with stroke through vascular stability, (2) genes associated with coagulopathies (Supplementary Tables 1 and 2). Familial segregation of the variants was assessed using Sanger sequencing. Also, if the variants did not meet the in-house criteria for quality control, the variants were Sanger sequenced for validation. Shortly, the variants were sent for Sanger validation if the sequencing depth was below 20, the variant allele was present in less than 30% of the reads, or the variant quality was flagged by the GATK VQSR algorithm.
The variants were classified according to the criteria published by the American College of Medical Genetics (ACMG).
57
The variants published in the known databases ClinVar and HGMDPro as disease-causing were considered pathogenic. The novel variants detected first by our study group were classified as pathogenic or likely pathogenic, if the variant fulfilled the following criteria: (1) a missense variant that replaces glycine by another amino acid in the triple helix domain, for
The whole genetic testing was performed at the Medical Laboratory of Tartu University Hospital, which is accredited under ISO 15189:2012.
The studies were approved by the Ethics Review Committee on Human Research of the University of Tartu by protocol no 294/M-18. Written informed consent was provided by all individual participants older than 7 years and by their parents in all cases.
Statistical Analysis
Prior to further analysis, normality of the data was evaluated using the Shapiro-Wilk test. Continuous data were summarized as medians with the interquartile range (IQR) and range (minimum, maximum), and categorical data, as absolute counts and percentages. Differences between the 2 groups were analyzed by the nonparametric Mann-Whitney
Results
Clinical Characteristics of the Study Group
The representative demographic data of the final study group of 85 periventricular hemorrhagic infarction/periventricular venous infarction patients are shown in Table 1. The study group included 39 preterm-born children with periventricular hemorrhagic infarction, 6 children with antenatal periventricular hemorrhagic infarction, and 40 children with presumed antenatal periventricular venous infarction. The study groups were well comparable in terms of maternal age, previous pregnancy loss, number of pregnancies, deliveries and mode of conception, gender of patients, number of patients small for gestational age, and number of patients with severe motor deficit or epilepsy (Table 1). Compared to the preterm periventricular hemorrhagic infarction group, the presumed antenatal periventricular venous infarction group showed significantly lower incidence of emergency caesarean section and 5-minute Apgar score <7 (Table 1).
Demographics and Distribution of Pathogenic/Likely Pathogenic Variants and Variants of Unknown Significance Associated With Stroke in Different PVHI/PVI Subgroups.
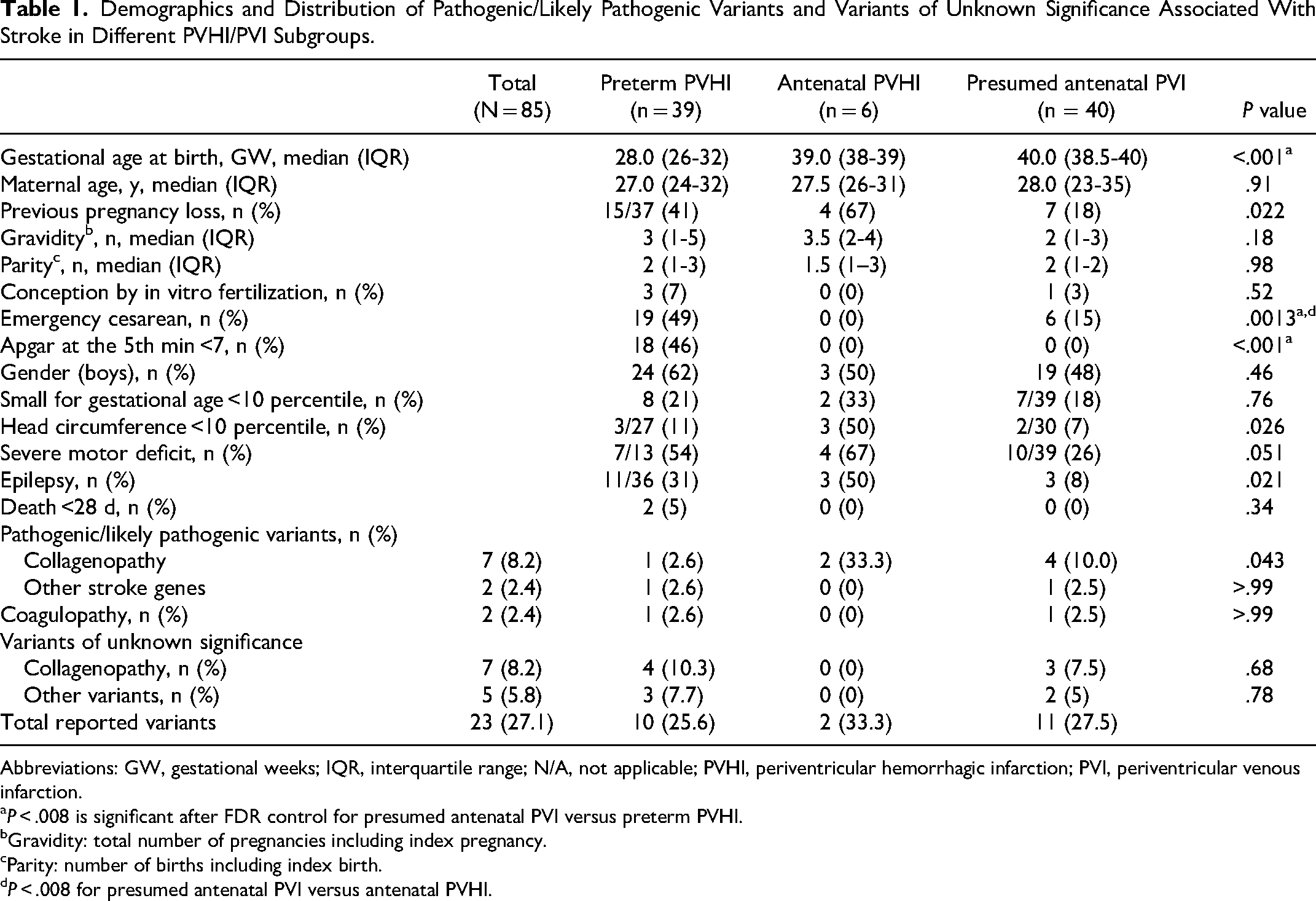
Abbreviations: GW, gestational weeks; IQR, interquartile range; N/A, not applicable; PVHI, periventricular hemorrhagic infarction; PVI, periventricular venous infarction.
Gravidity: total number of pregnancies including index pregnancy.
Parity: number of births including index birth.
Altogether, we identified 11 of 85 patients (12.9%) with periventricular hemorrhagic infarction/periventricular venous infarction carrying pathogenic and likely pathogenic variants in genes associated with collagenopathy and coagulopathy, or in other genes associated with stroke. All pathogenic or likely pathogenic variants are listed in Table 2. Among the pathogenic variants,
Description of the Stroke Patients With Pathogenic or Likely Pathogenic Gene Variants, Their MRI Findings, and Clinical Outcome.
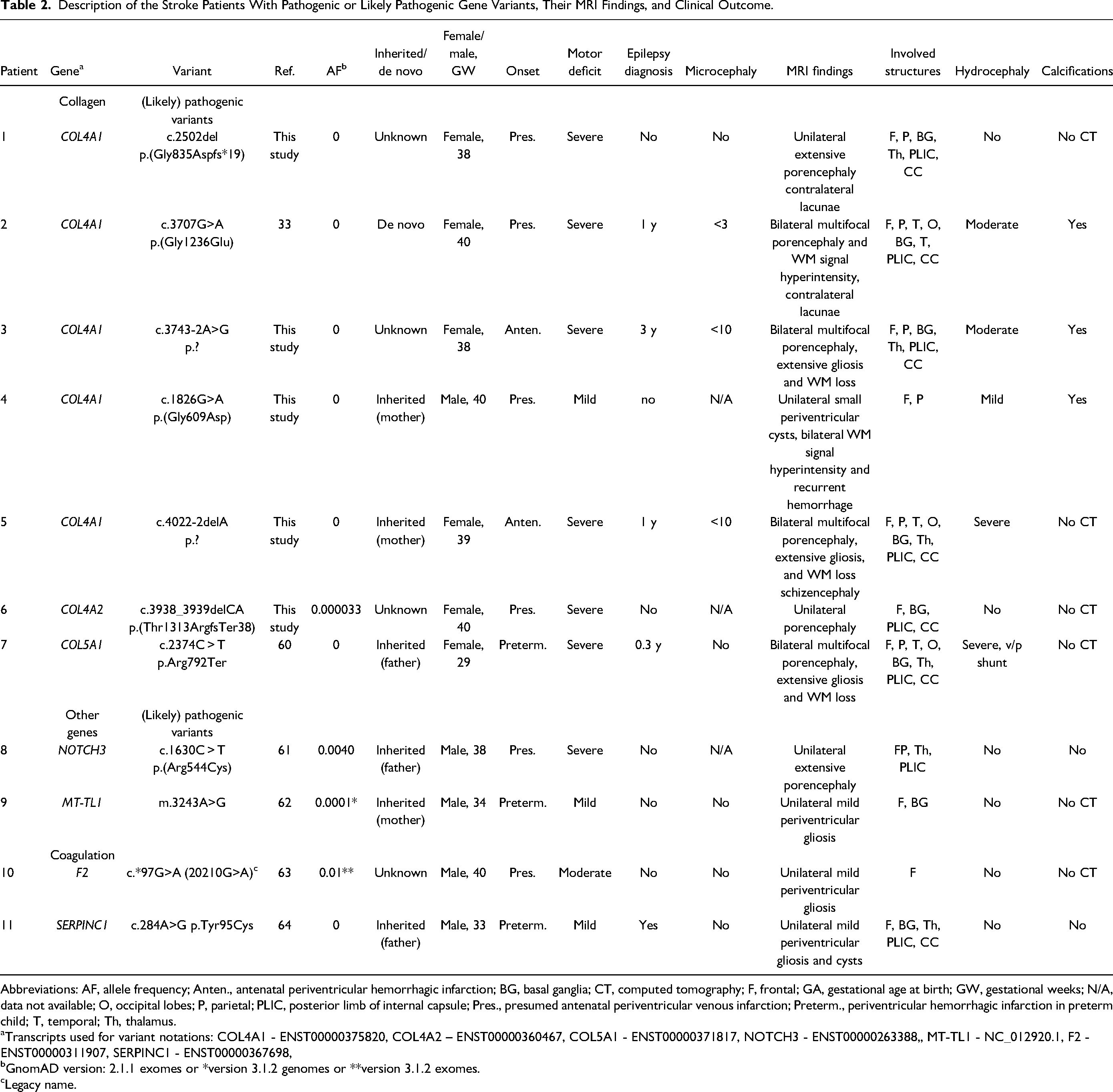
Abbreviations: AF, allele frequency; Anten., antenatal periventricular hemorrhagic infarction; BG, basal ganglia; CT, computed tomography; F, frontal; GA, gestational age at birth; GW, gestational weeks; N/A, data not available; O, occipital lobes; P, parietal; PLIC, posterior limb of internal capsule; Pres., presumed antenatal periventricular venous infarction; Preterm., periventricular hemorrhagic infarction in preterm child; T, temporal; Th, thalamus.
Transcripts used for variant notations: COL4A1 - ENST00000375820, COL4A2 – ENST00000360467, COL5A1 - ENST00000371817, NOTCH3 - ENST00000263388,, MT-TL1 - NC_012920.1, F2 - ENST00000311907, SERPINC1 - ENST00000367698,
GnomAD version: 2.1.1 exomes or *version 3.1.2 genomes or **version 3.1.2 exomes.
Legacy name.
Although the most severe cases were diagnosed already clinically, 5 of 11 cases with pathogenic or likely pathogenic variants had not been tested earlier, among them 3 cases with collagenopathy. These had been considered as too mild cases, and genetic disease had not been suspected earlier. All sequence changes detected in this group were unique; however, it was found that
Only 2 of 85 children (2.4%) had variants connected with coagulopathy: 1 child with presumed antenatal periventricular venous infarction had the common prothrombin 20210G>A (rs1799963) risk variant and 1 preterm-born patient with periventricular hemorrhagic infarction and antithrombin 3 deficiency had
There were 2 of 85 (2.4%) pathogenic findings in other genes that have earlier been associated with stroke:
In addition, variants of unknown significance in collagen (
There were 7 patients with variants of unknown significance in collagen genes: 2 patients with periventricular hemorrhagic infarction among preterm-born children and 1 with presumed antenatal periventricular venous infarction had a change in
Risk Factors in the Group of Collagenopathies and in the Group of Children Without Detected Gene Variants (Figure 2)
Children with collagenopathies (
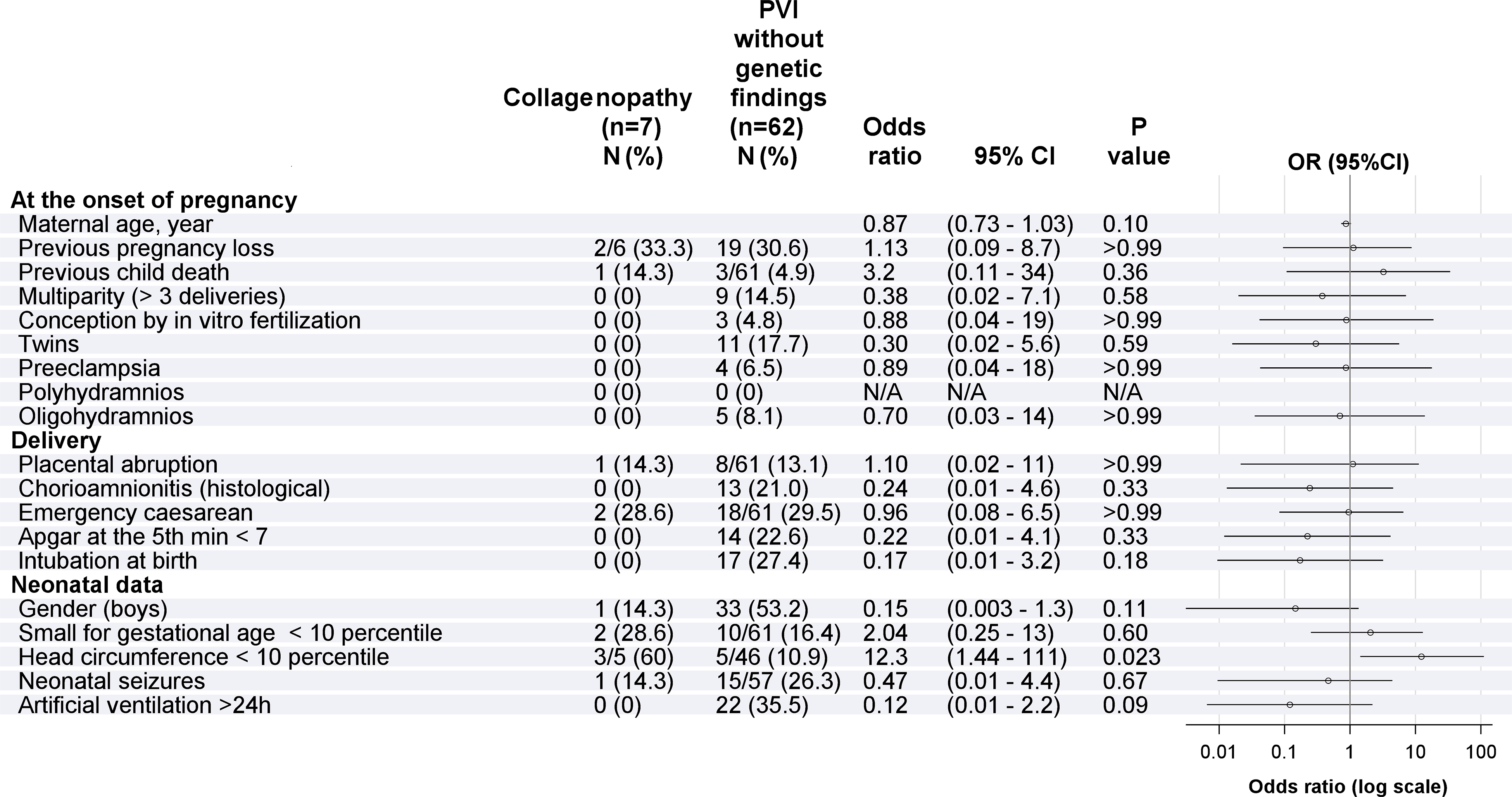
Maternal-, pregnancy-, and delivery-related risk factors in children with pathogenic/likely pathogenic variants in the collagen genes compared to children with periventricular hemorrhagic infarction/periventricular venous infarction without genetic variants associated with stroke. The adjusted false discovery rate significance threshold level was 0.0028, but no risk factors was revealed as significant (raw
MRI Characteristics of Children With Genetic Changes in Collagen Genes (Figure 3)
Children with collagenopathies had significantly more often bilateral multifocal stroke (
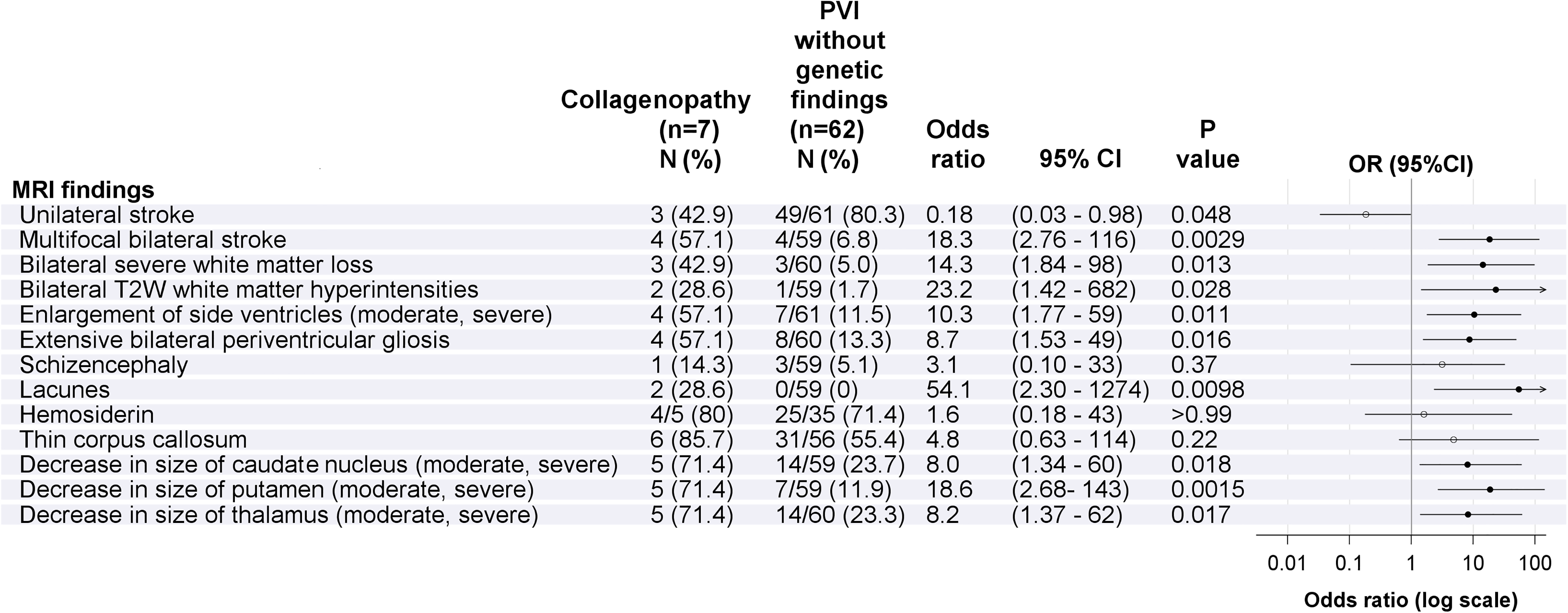
MRI findings for children with collagenopathies compared to children with periventricular hemorrhagic infarction/periventricular venous infarction without genetic changes. Only raw
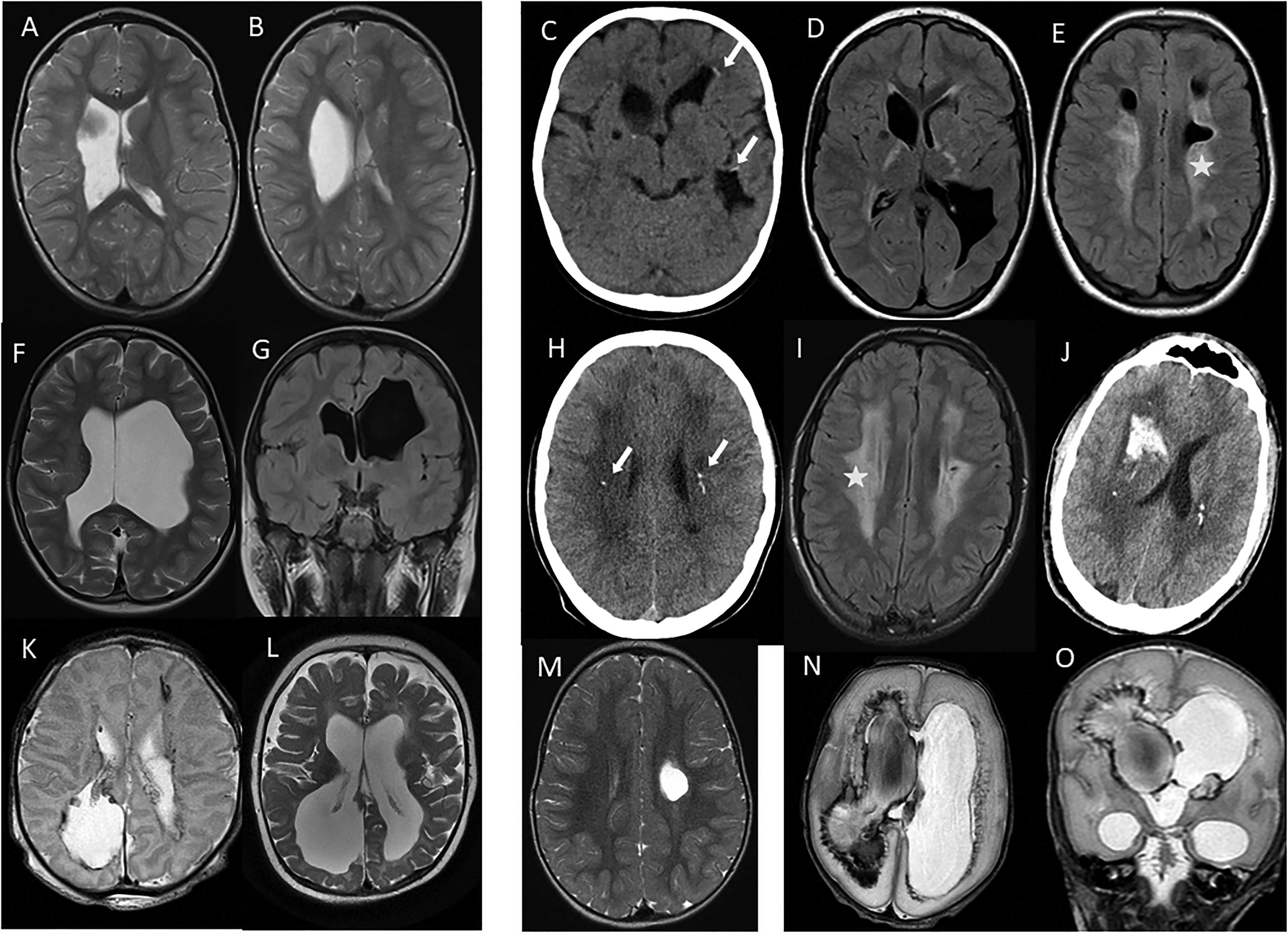
Individual MRI findings for children with collagenopathies. Patient 1: (A, B) axial T2-weighted MRI images of a girl with presumed antenatal periventricular venous infarction with unilateral focal porencephalic enlargement of the left ventricle at age 3 years. Patient 2: (C) axial computed tomography image with calcifications (arrows) of a girl with presumed antenatal periventricular venous infarction at age 13 months; and (D-E) axial MRI fluid-attenuated inversion recovery images with multiple bilateral porencephalic enlargement of the lateral ventricles and bilateral signal increase of the white matter (star) at age 12 years. Patient 3: (F) axial T2-weighted image of a girl with antenatal periventricular hemorrhagic infarction with severe porencephalic enlargement of the lateral ventricles and loss of white matter at age 7 years; and (G) coronal fluid-attenuated inversion recovery image with thin corpus callosum at age 19 years. Patient 4: (H) axial computed tomography image of a boy with presumed antenatal periventricular venous infarction with calcifications (arrows) at age 12 years; (I) axial fluid-attenuated inversion recovery image with bilateral signal increase of the white matter and small periventricular cysts on the left at age 14 years; and (J) at age 22 years, computed tomography image shows large intraparenchymal hemorrhages. Patient 5: (K) axial T2-weighted MRI image of a girl with antenatal periventricular hemorrhagic infarction with bilateral porencephalic enlargements of the ventricles and hemosiderin in side ventricles and parenchyma on the first day of life; and (L) axial T2-weighted MRI image at age 10 months with enlarged lateral ventricles and loss of white matter. Patient 6: (M) axial T2-weighted image of a girl with presumed antenatal periventricular venous infarction with unilateral focal porencephalic enlargement of the left lateral ventricle at age 2 years. Patient 7: (N) axial and (O) coronal T2-weighted images of a preterm-born girl at gestational week 29 with extensive bilateral intraventricular hemorrhage and right-side periventricular hemorrhagic infarction with ventriculomegaly at age 14 days.
Outcome Characteristics of Children With Possibly Stroke-Causing Genetic Variants
No deaths occurred among children with collagenopathies or other pathogenic variants during the study period (Table 3). Moderate-severe motor deficit developed more often in children with pathogenic variants in collagen genes (85.7%) compared to children without genetic variants (20.7%), respectively (
Outcome in Children With Periventricular Hemorrhagic Infarction / periventricular Venous Infarction With Pathogenic Variants and In Children Without Pathogenic Variants or Variants of Unknown Significance.

*
Discussion
Main Finding of the Study
We found high prevalence of collagenopathies, specifically pathogenic/likely pathogenic variants in
Emergence of Collagenopathies
Defects
Type IV Collagens
Type IV collagen is abundantly detected in different tissues, causing a wide spectrum of phenotypes involving different organ systems, with brain abnormalities and neurologic sequelae being more prevalent.29,32,33 Type IV collagens are expressed in the basal membrane of a wide range of tissues, including vessel walls. Most of the pathogenic variants in
Defective
MRI changes in children with
Individuals affected with collagenopathies have a wide spectrum of phenotypes causing congenital hemiparesis, epilepsy, intellectual disability, leukoencephalopathy, microbleeds, and lacunar stroke.32,33,43,47 Severe motor deficit and epilepsy developed more often in children with pathogenic variants in collagen genes (85.7%) compared to children without genetic variants (20.7%), respectively. Epilepsy has been found earlier in 55 of 123 patients with
Collagens V and I
Besides
The fact that
Familial Segregation of the Variants
Perinatal Risk Factors
We found no pregnancy, delivery, or neonatal symptoms that could distinguish periventricular hemorrhagic infarction/periventricular venous infarction children with collagenopathy from those without an identified genetic cause, as also suggested earlier. 33 If antenatal periventricular hemorrhagic infarction is not detected with prenatal ultrasonography, the birth is likely to be uneventful like in children with presumed antenatal stroke. However, the head circumference of 3 of 7 children in our group with collagenopathy was <10 percentiles, all with bilateral porencephaly and white matter loss, and poor outcome with severe motor deficit. This finding indicates that a small head circumference at birth can be a possible sign of congenital brain damage, including periventricular hemorrhagic infarction and collagenopathy. Collagenopathy and antenatal stroke should be one of the differential diagnoses in children with microcephaly at birth, as noted earlier.29,33
Coagulopathy
The role of inherited thrombophilia in the pathogenesis of perinatal stroke is still controversial. Some studies have suggested abnormalities in coagulation as a possible cause of perinatal stroke.23,41 Others have found that thrombophilia risk is not increased in perinatal stroke in term-born children, including presumed antenatal periventricular venous infarction and arterial ischemic stroke, compared to controls.39,63 Recent study highlight the importance of arterial endothelial dysfunction, rather than thrombocytopenia.
69
An earlier study of preterm infants with an atypical presentation of periventricular hemorrhagic infarction recommends testing of thrombophilia, especially for factor V Leiden and prothrombin gene mutation, which was found in 7 of 17 cases, although
Other Genes
Interestingly, we found 2 variants in other genes that can be connected with microangiopathy and stroke or strokelike episodes, both being familial cases. In the the group of periventricular hemorrhagic infarction among preterm-born children one child had the most common mitochondrial variant m.3243A>G in
A child with presumed antenatal periventricular hemorrhagic infarction and a pathogenic change in
Indications for Testing
Periventricular hemorrhagic infarction/periventricular venous infarction leads to lifelong impairments: motor deficit of various severity, cognitive impairment, behavioral problems, and epilepsy.13,17,22,36,72 Poor outcome has been found in survivors among preterm children with periventricular hemorrhagic infarction.
35
Systematic genetic testing has been suggested earlier in term-born children with intraparenchymal hemorrhages,
42
in children with antenatal intracranial hemorrhage, especially in severe cases,
41
and in children with porencephaly.
29
Systemic genetic evaluation in patients with periventricular hemorrhagic infarction/periventricular venous infarction is not a routine clinical practice. Although the most severe cases were diagnosed already clinically, 5 of 11 children with pathogenic/likely pathogenic variants had not been tested earlier as genetic disease was not suspected in mild cases. Awareness of the genetic causes of periventricular hemorrhagic infarction/periventricular venous infarction gives the children's parents an understanding that it is not only preterm birth, perinatal stress, postnatal environment, or possible treatment failure that can cause severe periventricular hemorrhagic infarction.48,49 Therefore, we suggest that in all children with periventricular hemorrhagic infarction/periventricular venous infarction, both term-born and preterm, genetic testing should be considered, especially in the case of bilateral and multifocal stroke with bilateral white matter alterations; however, even with unilateral periventricular hemorrhagic infarction/periventricular venous infarction. First of all, collagen genes (
Limitations
The most important limitation of the study is the small size of the groups of term-born children with antenatal and presumed antenatal periventricular hemorrhagic infarction/periventricular venous infarction and preterm-born children with periventricular hemorrhagic infarction. Yet it is the largest study of patients with presumed antenatal periventricular venous infarction who were evaluated genetically and radiologically as a cohort.
None of the preterm-born children with periventricular hemorrhagic infarction diagnosis who died had been genetically tested except for 2 cases among preterm children in whom the samples for genetic testing were taken prior to death. Therefore, there can be more children with genetic variants among the preterm population with severe intraventricular hemorrhage or periventricular hemorrhagic infarction, as children with the worst outcomes were not tested.
Conclusion
Our data suggest that children with periventricular hemorrhagic infarction/periventricular venous infarction have high prevalence of pathogenic variants in collagen genes (
Supplemental Material
sj-csv-1-jcn-10.1177_08830738231186233 - Supplemental material for High Prevalence of Collagenopathies in Preterm- and Term-Born Children With Periventricular Venous Hemorrhagic Infarction
Supplemental material, sj-csv-1-jcn-10.1177_08830738231186233 for High Prevalence of Collagenopathies in Preterm- and Term-Born Children With Periventricular Venous Hemorrhagic Infarction by Norman Ilves, Sander Pajusalu, Tiina Kahre, Rael Laugesaar, Ustina Šamarina, Dagmar Loorits, Pille Kool and Pilvi Ilves in Journal of Child Neurology
Supplemental Material
sj-csv-2-jcn-10.1177_08830738231186233 - Supplemental material for High Prevalence of Collagenopathies in Preterm- and Term-Born Children With Periventricular Venous Hemorrhagic Infarction
Supplemental material, sj-csv-2-jcn-10.1177_08830738231186233 for High Prevalence of Collagenopathies in Preterm- and Term-Born Children With Periventricular Venous Hemorrhagic Infarction by Norman Ilves, Sander Pajusalu, Tiina Kahre, Rael Laugesaar, Ustina Šamarina, Dagmar Loorits, Pille Kool and Pilvi Ilves in Journal of Child Neurology
Supplemental Material
sj-docx-3-jcn-10.1177_08830738231186233 - Supplemental material for High Prevalence of Collagenopathies in Preterm- and Term-Born Children With Periventricular Venous Hemorrhagic Infarction
Supplemental material, sj-docx-3-jcn-10.1177_08830738231186233 for High Prevalence of Collagenopathies in Preterm- and Term-Born Children With Periventricular Venous Hemorrhagic Infarction by Norman Ilves, Sander Pajusalu, Tiina Kahre, Rael Laugesaar, Ustina Šamarina, Dagmar Loorits, Pille Kool and Pilvi Ilves in Journal of Child Neurology
Footnotes
Acknowledgments
The authors thank all pregnant women, patients and their parents, as well as colleagues from the Department of Genetics of Tartu University Hospital for their contribution.
Author Contributions
Conceptualization: NI, RL PI; Data curation: NI, TK, RL PI; Formal Analysis: NI, SP, TK, US; Funding acquisition: RL, PI, SP; Supervision: PI, SP; Statistical analysis: PK, NI, PI; Writing – original draft: NI, PI, TK, RL, SP; Writing – review & editing: all authors. All authors contributed to manuscript revision, read, and approved the submitted version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Estonian Research Council grants (PUT148, PRG471, PSG774, PRG1912, MOBTP175) and basic financing from the University of Tartu.
Supplemental Material
Supplemental material for this article is available online.
Ethical Approval
The studies were approved by the Ethics Review Committee on Human Research of the University of Tartu by protocol no 294/M-18. Written informed consent was provided by all individual participants older than 7 years and by their parents in all cases.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
