Abstract
Introduction
Perinatal ischemic stroke has been defined as a group of heterogeneous conditions with brain injury, occurring between 20 weeks of fetal life and postnatal day 28. 1 Some term-born children with perinatal stroke are asymptomatic after birth. 2 If hemiparesis, delayed milestones, or seizures develop after the neonatal period and magnetic resonance imaging (MRI) shows signs of chronic brain damage characteristic of perinatal stroke, stroke is presumed to have occurred in the antenatal period.2–8 Among term-born children with presumed perinatal stroke, neuroimaging suggests periventricular venous infarction in up to two-thirds of cases.2,9
Periventricular venous infarction is common among extremely premature children especially in infants born before the 32nd full gestational week (GW) and in children with very low birthweight.10,11 Chronic brain damage visualized on MRI in term-born children with presumed periventricular venous infarction and in preterm infants with neonatal periventricular venous infarction is similar. 7 This finding suggests that presumed periventricular venous infarction may occur from gestational weeks 24 through 34 at a certain stage of vascular development of the brain, the germinal matrix being a likely source of intraventricular hemorrhage,4,12,13 leading to compression of medullary veins, resulting in periventricular infarction.14,15
Because of the lack of investigations and small study groups, the exact time window and the risk factors for development of presumed periventricular venous infarction remain largely unknown.2,5,16 Our previous study of 42 presumed perinatal stroke children included 29 periventricular venous infarction patients. The presence of prenatal infection and/or imminent miscarriage during pregnancy was significantly more frequent in presumed periventricular venous infarction than presumed arterial ischemic stroke patients (P = .04). 2 No earlier studies have comparatively assessed gestational risk factors in presumed periventricular venous infarction children and in an unaffected control group.
The aim of the present study was to compare term- or near-term-born children (≥36 gestational weeks) with presumed periventricular venous infarction, preterm-born children (<36 gestational weeks) with neonatal periventricular venous infarction, and consecutively born unaffected control infants (≥36 gestational weeks), to identify the prevalence of pregnancy- and delivery-associated risk factors, and gestational age at their occurrence.
Patients and Methods
Patients
Patients with presumed periventricular venous infarction were identified from the Pediatric Stroke Database according to the patient selection criteria as described earlier.2,3,6 The vascular classification of perinatal stroke patients in the Pediatric Stroke Database is based on MRI according to Kirton and coauthors. 17 Of the 43 cases with presumed periventricular venous infarction included in this study, 29 were previously reported in our seminal analysis of retrospective data from 1994 to 2014. 2 Between 2014 and 2019, a total of 14 additional presumed periventricular venous infarction cases were prospectively included in this study group.
Patients with neonatal periventricular venous infarction in the Pediatric Stroke Database (n = 86) were identified from the clinical database of a third-level regional university hospital, accounting for about one-third of the neonatal cases in the country in 1994-2019. The medical records of the neonatal intensive care unit of the hospital were searched for children with the diagnosis of severe grade of IVH (grade III or IV) or periventricular venous infarction10,11 treated at the hospital between 1994 and 2019. Also, the hospital's electronic database from 2008 was searched for the International Classification of Diseases, Tenth Revision (ICD-10), diagnosis codes P 52.2, P 52.3, P 52.4, P 52.6, and P 52.9. 18 Patients with grade III intraventricular hemorrhage (IVH) were included in the initial search to find all possible patients with periventricular venous infarction confirmed by MRI or autopsy.
Children with periventricular venous infarction were excluded if at least 1 of the following was present: (1) acute arterial ischemic stroke, sinovenous thrombosis, or hemorrhagic stroke; (2) other diseases, congenital anomalies, or genetic diseases involving the central nervous system; (3) absence of cerebral MRI or autopsy; (4) second child of a twin pregnancy when both had periventricular venous infarction (Figure 1).
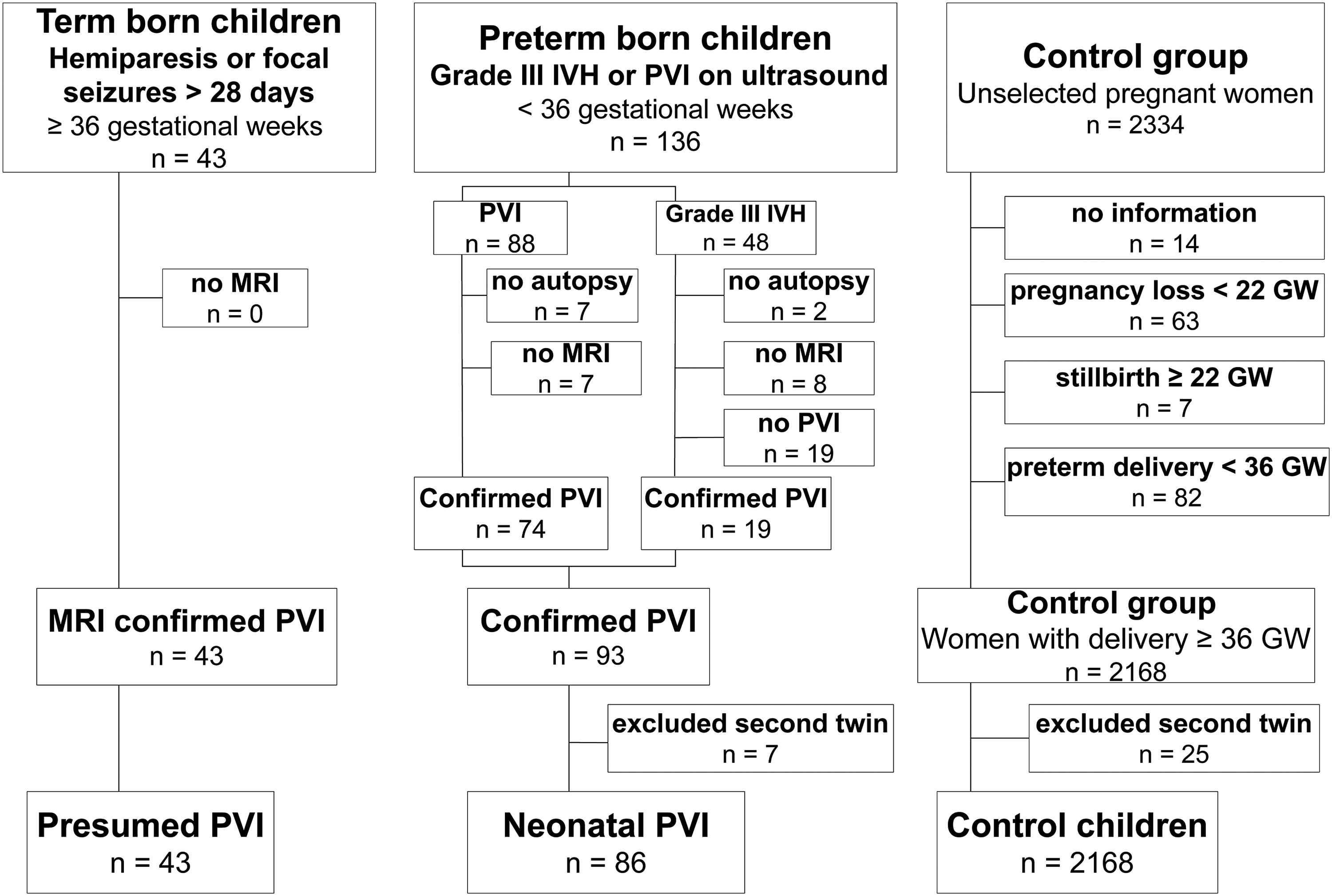
Formation of the study groups and the applied patient selection criteria.
The control group was formed of the cases in the observational pregnancy cohort of Happy Pregnancy Study (full name: Development of Novel Non-invasive Biomarkers for Fertility and Healthy Pregnancy, Principal Investigator M.L.), representing approximately two-thirds of all pregnant women receiving antenatal care at the Women's Clinic of a third-level regional university hospital in 2013-2015. This cohort comprised unselected pregnant women recruited prospectively during their first antenatal visit (n = 2334).
The flow chart of the inclusion criteria for periventricular venous infarction and control patients is shown in Figure 1. The final control group (n = 2168) comprised no cases of perinatal stroke. All participants were Caucasians.
Methods
Clinical data on pregnancy and delivery were extracted from medical records. Data about pregnancy and delivery of children with periventricular venous infarction, as well as autopsy and placental histology reports were requested from 14 hospitals, where antenatal care had been provided and/or childbirth had occurred, and were reviewed retrospectively. For the control group, epidemiologic data including reproductive history, parental lifestyle, and pregnancy risk factors were collected from 3 questionnaires completed at each trimester of pregnancy.
Risk Factor Data Extraction
Clinical information included the following:
risk factors at the onset of pregnancy: maternal age, gravidity, parity, elective termination of pregnancy or preterm delivery in reproductive history, threatened pregnancy loss <22 gestational weeks or imminent preterm delivery ≥22 gestational weeks during previous pregnancies, mode of conception: spontaneous or in vitro fertilization, pregestational hypertension and diabetes, multiple gestation; risk factors during index pregnancy: threatened pregnancy loss, imminent preterm delivery, bacterial infection treated with antibiotics, smoking during pregnancy, alcohol abuse (consumption at least once a week), polyhydramnios, oligohydramnios, hypertensive disorders or diabetes during pregnancy, anemia (hemoglobin <110 g/L at the first and third trimesters, <105 g/L at the second trimester of pregnancy), use of antenatal steroids and magnesium sulphate due to imminent preterm delivery; delivery data: gestational age and mode of delivery, level of maternity hospital, labor augmentation with oxytocin, placental abruption, histologically confirmed chorioamnionitis, shoulder dystocia, birth parameters: sex, length, weight, head circumference, Apgar scores at the first and fifth minute after birth, need for resuscitation, including mask ventilation, intubation, cardiac massage, or adrenaline. The neonate was classified as small for gestational age when gestational age and sex-adjusted weight centiles at birth were <10 percentiles, and restricted growth when these were <3 percentiles according to national normal values
19
; and death during first hospitalization.
Pyelonephritis was defined by presence of a positive urine culture as well as at least 1 of the following clinical findings: fever and flank or back pain. Appendicitis was confirmed when the patient needed surgical intervention. Diagnosis of urogenital tract infections was confirmed by chlamydiosis, gonorrhea, and trichomoniasis, as well as bacterial vaginosis based on positive nucleic acid amplification tests or bacterioscopy. Diagnosis of preeclampsia followed the international guidelines at the time of recruitment (ISSHP, 2014), co-occurrence of a new-onset hypertension (HTN; ≥140/ ≥90 mmHg) after 20 gestational weeks, and proteinuria or other signs of maternal organ dysfunction.
20
Gestational hypertension was diagnosed as new-onset hypertension after 20 gestational weeks without proteinuria or other maternal organ dysfunction.
20
Polyhydramnios was diagnosed when the deepest amniotic fluid pool was >8 cm and oligohydramnios was diagnosed when it remained <2 cm at the prenatal ultrasonographic scan.
Antibacterial treatment
Data on in- and out-patient antibacterial treatment during pregnancy were retrieved from hospital medical records (available for all study patients) and from the population-based Digital Prescription Centre (available only for the control group) along with the exact diagnosis of infection according to the ICD-10. 18 Only the infections treated with antibiotics were included.
Classification of infections treated with antibacterial therapy during pregnancy on the basis of ICD-10
18
was as follows:
Diseases of the respiratory system: codes J+O99.5 Diseases of the digestive system: codes: K+O99.6
K00-K14 Diseases of the oral cavity and salivary glands K35-K38 Appendicitis Diseases of the genitourinary tract: codes A+O+N except for O99.5 and O99.6)
A: Certain infectious and parasitic diseases including chlamydia (A 560) O: Pregnancy, childbirth, and puerperium) including chorioamnionitis including asymptomatic bacteriuria (O23.0-O23.9) N: diseases of the genitourinary system Other bacterial infection:
H: diseases of the eye and the ear I: diseases of the circulatory system L: diseases of the skin and subcutaneous system
The time of infection was identified on the basis of the first prescription redemption's time for the control group and on the basis of hospital admission or the first diagnosis for the periventricular venous infarction group.
Radiologic Evaluation
All radiologic images of patients with perinatal stroke, including periventricular venous infarction, were independently reviewed from the population-based Picture Archiving System by 2 neuroradiologists and a resident to confirm the vascular genesis of perinatal stroke.
All cranial ultrasonography examinations during the study period between 1994 and 2019 were performed by pediatric radiologists. The first ultrasonography was done on admission, after patient stabilization, followed by imaging on days 2-3 and on days 5-7 and further once a week until discharge. If the status of the patient deteriorated and severe IVH or periventricular venous infarction was clinically suspected, an additional ultrasonography was promptly performed by a pediatric radiologist on call. The diagnosis of periventricular venous infarction was confirmed by MRI examinations around a corrected age of 40 gestational weeks or later during infancy.
On cranial ultrasonography periventricular venous infarction is described as a parenchymal increase in echogenicity in the periventricular area, which can be fan- or globular-shaped and separate, or partially communicating with the ventricle.10,14,21
On MRI, periventricular venous infarction is described as a unilateral medullary venous territory infarction in the periventricular area, with a porencephalic enlargement of the lateral ventricle, periventricular gliosis, and/or cystic changes and the spared cortex. The residues of hemorrhage- hemosiderin can be visible in the area of the germinal matrix, ventricles, and/or in periventricular lesion.2,4,7,17
The individual data that support the findings of this study are available from the corresponding author on reasonable request.
Statistical Analysis
Prior to further analysis, normality of the data was evaluated using the Shapiro-Wilk test. Continuous data were summarized as medians with the interquartile range and range (minimum, maximum), and categorical data, as absolute counts and percentages. Differences between the 2 groups were analyzed by the nonparametric Mann-Whitney U test for continuous variables. Univariate comparison was analyzed using the χ2 test or Fisher exact test for dichotomous variables, as appropriate. Multinomial logistic regression was used to assess the relationship between the groups and infection subgroups (genitourinary tract infection, other infections, no bacterial infections). Odds ratio (OR) with the 95% confidence interval (CI) was estimated as the measure of association. All raw P values were 2-sided. Multiple testing was corrected using the false discovery rate linear step-up procedure. 22 The Benjamini-Hochberg critical values were calculated as (i/m)Q, where i is the rank in an ascending list of P values, m is the total number of tests, and Q is a false discovery rate of .05. The cutoff P value for significance of a single comparison for a set of risk factors was .012.
Finally, a multiple logistic regression model was constructed to determine independent predictors for presumed periventricular venous infarction. Variables with P <.2 in univariate comparison were entered in a stepwise multiple logistic regression model. Firth penalized likelihood approach was used for the models. 23 Model fits were examined by inspecting residual plots and calibration measures (Akaike information criterion). The output of the logistic regression model is presented as adjusted odds ratios (aORs) and 95% CI. Statistical evaluation was performed using the statistical package SAS, version 9.4 (SAS Institute, Cary, NC), and the R Statistical Software (version 4.0.2).
Results
Clinical Characteristics of the Study Groups
The representative demographic data of the study groups of 43 mothers and term or near-term (≥36 gestational weeks) born children with presumed antenatal periventricular venous infarction, 86 mothers and preterm (<36 gestational weeks) children with neonatal periventricular venous infarction, and of the control group of 2168 pregnant women and their unaffected term or near-term (≥36 gestational weeks) neonates are shown in Table 1. The study groups were well comparable in terms of maternal age, number of pregnancies, deliveries, and mode of conception. The children of the presumed periventricular venous infarction and control groups were similar regarding availability of prenatal care, Apgar scores, gestational age, birth weight, and head circumference. All pregnancies were monitored in the presumed periventricular venous infarction group, whereas 8 of 86 (9%) pregnancies were not monitored before their preterm birth in the neonatal periventricular venous infarction group (OR 9, 95% CI 0.5-167, P = .05). No deaths occurred among children with presumed periventricular venous infarction, but 37 of 86 (43%) neonates in the neonatal periventricular venous infarction group died during the first month (OR 66, 95% CI 14-1105, P < .001).
Maternal, Pregnancy-, and Delivery-Related Data.
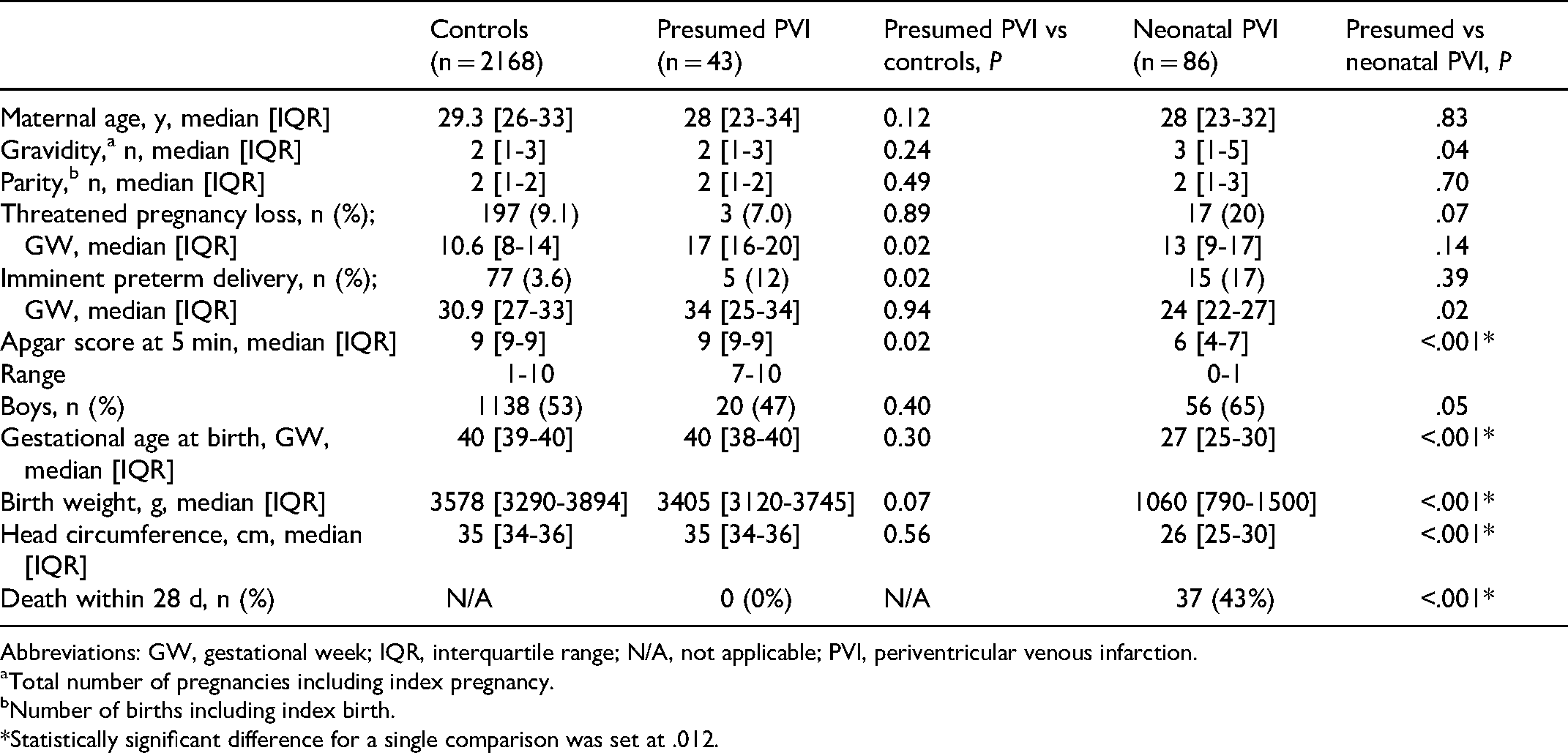
Abbreviations: GW, gestational week; IQR, interquartile range; N/A, not applicable; PVI, periventricular venous infarction.
Total number of pregnancies including index pregnancy.
Number of births including index birth.
*Statistically significant difference for a single comparison was set at .012.
Risk Factors in the Presumed Periventricular Venous Infarction and Control Groups
Pregnancies in the presumed periventricular venous infarction group were characterized by a significantly increased prevalence of maternal bacterial infections compared with control (P < .001; Figure 2, Table 2). Mothers of the presumed periventricular venous infarction group were more often diagnosed with genitourinary tract infections (P = .001) and, more specifically, with pyelonephritis (P < .001), compared to the control group. The incidence of maternal pyelonephritis among all patients in the presumed periventricular venous infarction group (10/43; 23%) compared with all control pregnancies (15/2168; 0.7%) differed more than 40 times (OR 43, 95% CI 16-112, P < .001).
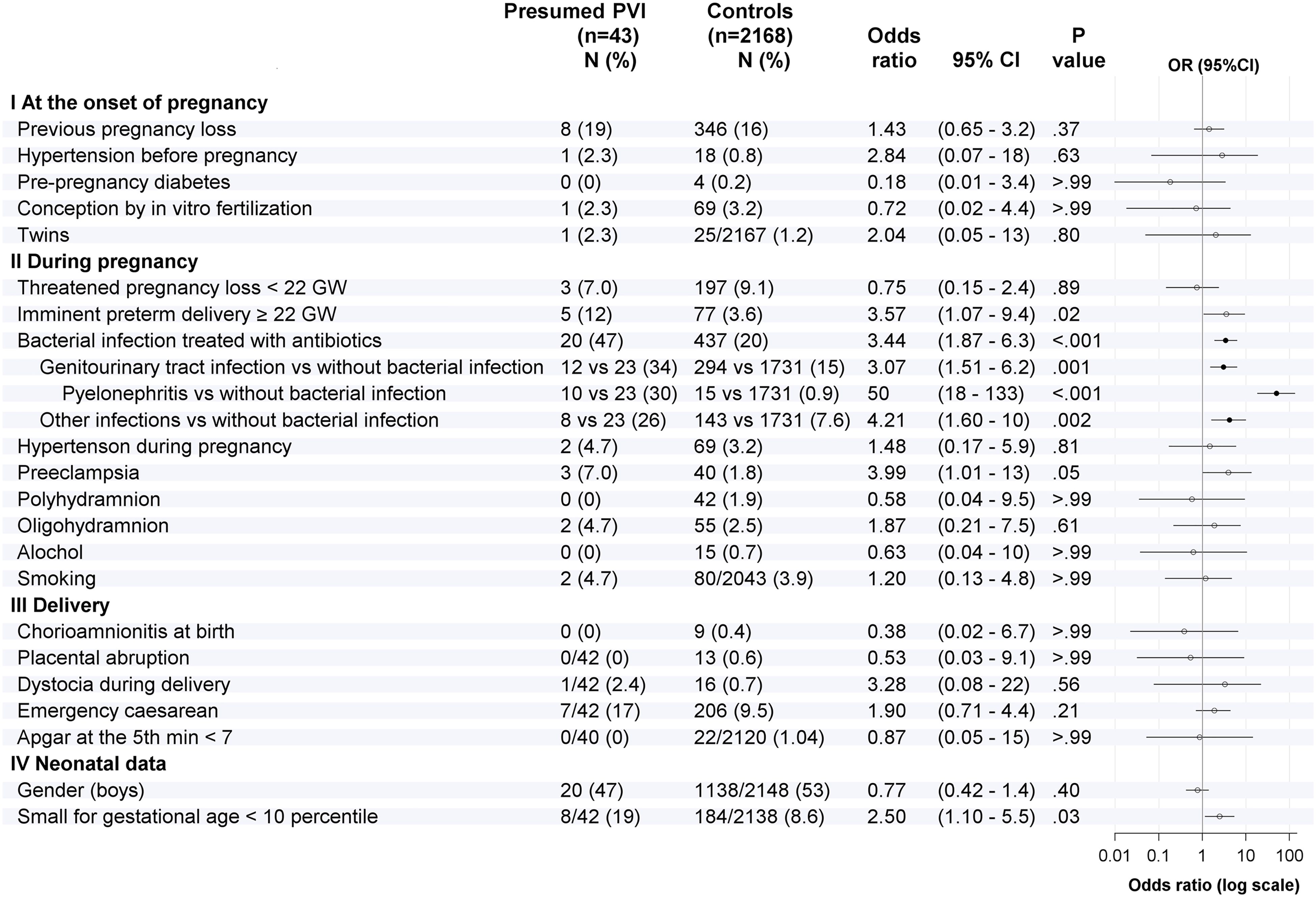
Comparison of risk factors between the presumed periventricular venous infarction and control groups.
Detailed Data on Bacterial Infections in the Study Groups.a
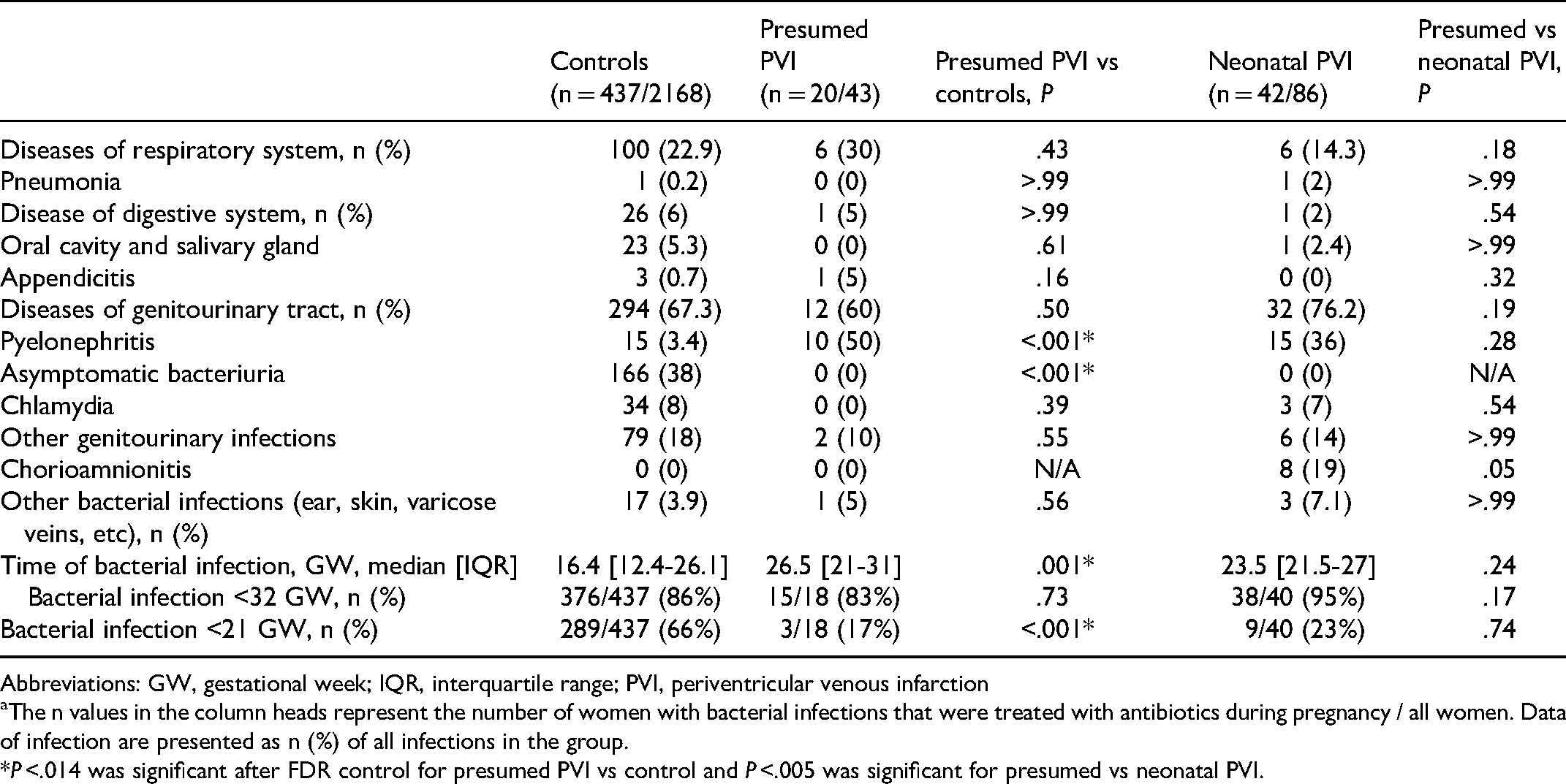
Abbreviations: GW, gestational week; IQR, interquartile range; PVI, periventricular venous infarction
The n values in the column heads represent the number of women with bacterial infections that were treated with antibiotics during pregnancy / all women. Data of infection are presented as n (%) of all infections in the group.
*P <.014 was significant after FDR control for presumed PVI vs control and P <.005 was significant for presumed vs neonatal PVI.
Mothers of children with presumed periventricular venous infarction had also significantly more often (P < .05) imminent preterm delivery at ≥22 gestational weeks and preeclampsia and delivered small-for-gestational-age neonates compared with control mothers (Figure 2). However, after controlling for FDR, these findings were not significantly different. Only 1 mother (1/43; 2.3%) in the presumed periventricular venous infarction group and 10 mothers (10/2168; 0.5%) in the control group had both preeclampsia and infection (P = .19).
Cases of presumed periventricular venous infarction did not differ from control regarding the incidence of delivery-related acute stress risk factors, such as placental abruption, chorioamnionitis, fetal dystocia, and mode of delivery (Figure 2).
Risk Factors in the Presumed and Neonatal Periventricular Venous Infarction Groups
The prevalence of pregnancies with any maternal bacterial infection in the presumed (20/43; 47%) and neonatal periventricular venous infarction (42/86; 49%) groups was similar (P = .72) (Figure 3, Table 2). The prevalence of genitourinary tract infection and, more specifically, maternal pyelonephritis in presumed (10/43; 23%) and neonatal periventricular venous infarction (15/86; 17%) did not differ (OR 1.4, 95% CI 0.6-3.5, P = .43).
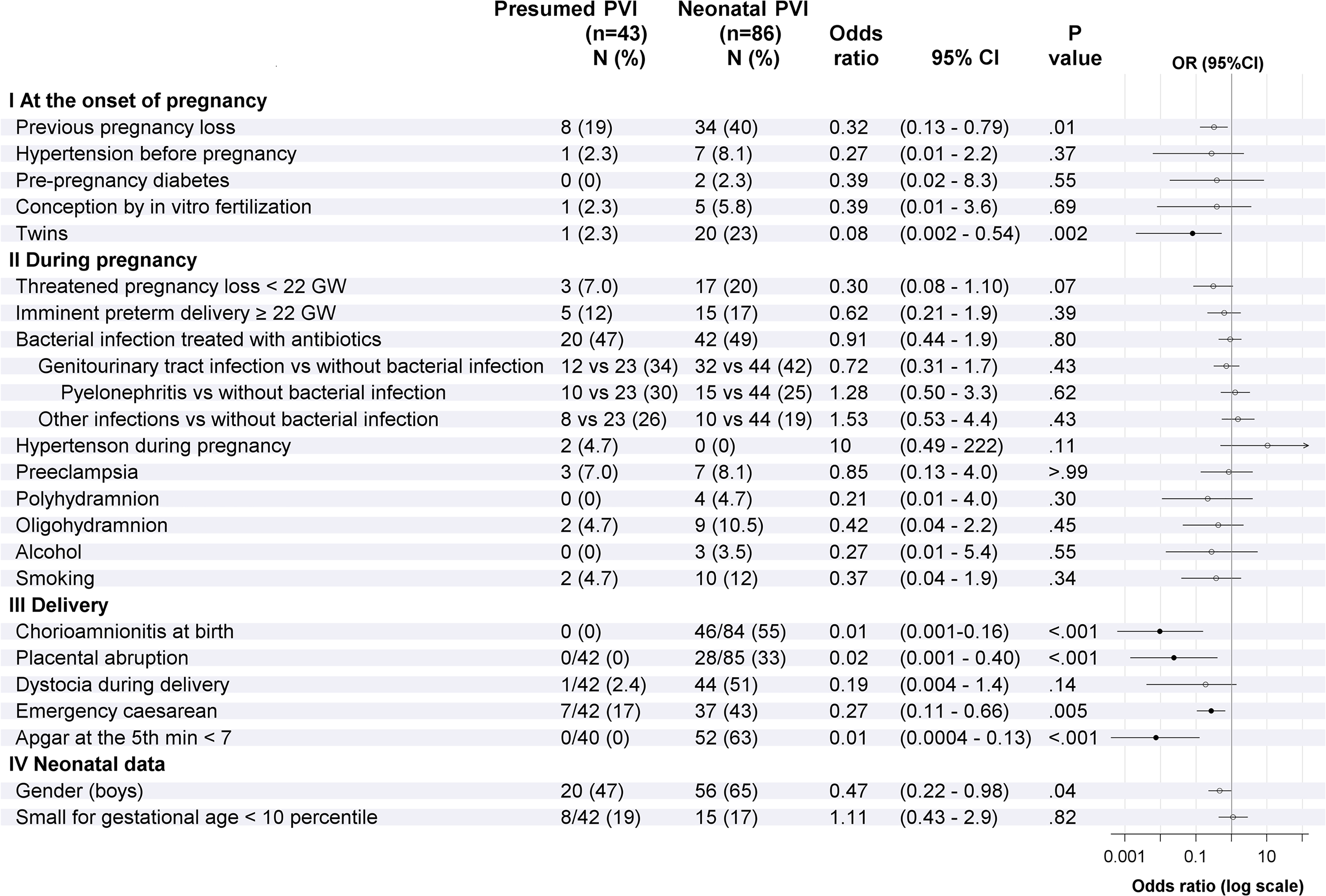
Comparison of risk factors between the presumed and neonatal periventricular venous infarction groups.
Compared to the neonatal periventricular venous infarction group, the presumed periventricular venous infarction group showed significantly lower incidence of twin pregnancies, male newborns, and birth distress related factors, such as chorioamnionitis, placental abruption, emergency cesarean section, and 5-minute Apgar score <7 (Figure 3). After FDR control, the difference in the gender proportions between the 2 periventricular venous infarction groups was not significant.
Bacterial Infection During Pregnancy
The dominating bacterial infection was genitourinary tract infection, including asymptomatic bacteriuria, accounting for at least 60% of all infections during pregnancy in all study groups (Table 2). We found no difference in the proportions of genitourinary tract infection and infections in other organ systems between the 3 study groups.
Notably, 166 of 437 (38%) mothers with any bacterial infection requiring antibacterial treatment in the control group had asymptomatic bacteriuria compared with absence of such cases in the neonatal and presumed periventricular venous infarction groups (OR 25, 95% CI 1.5-418, P < .001). However, only 15 of 437 (3.4%) of the control mothers with infection had pyelonephritis compared with 10 of 20 (50%) mothers in the presumed periventricular venous infarction group (OR 28, 95% CI 10-78, P < .001). We found no difference in the proportions of pyelonephritis among all infections in the presumed periventricular venous infarction versus neonatal periventricular venous infarction group, 10 of 20 (50%) and 15 of 42 (36%), respectively (OR 1.8, 95% CI 0.6-5.3, P = .28).
There was no difference in the median (interquartile range) gestational age of the first maternal bacterial infection among presumed versus neonatal periventricular venous infarction (26.5 [21-31] vs 23.5 [21.5-27] gestational weeks, respectively; P = .24), whereas infections were diagnosed earlier in pregnancy in the control group (16.4 [12.4-26.1], P = .001) than in the presumed periventricular venous infarction group. Bacterial infections before gestational week 21 were significantly more prevalent among controls (249/428; 58%) compared to cases with presumed periventricular venous infarction (3/18; 17%) (OR 7, 95% CI 2-24; P < .001). Individual data of gestational age at the time of bacterial infection are shown in Figure 4.
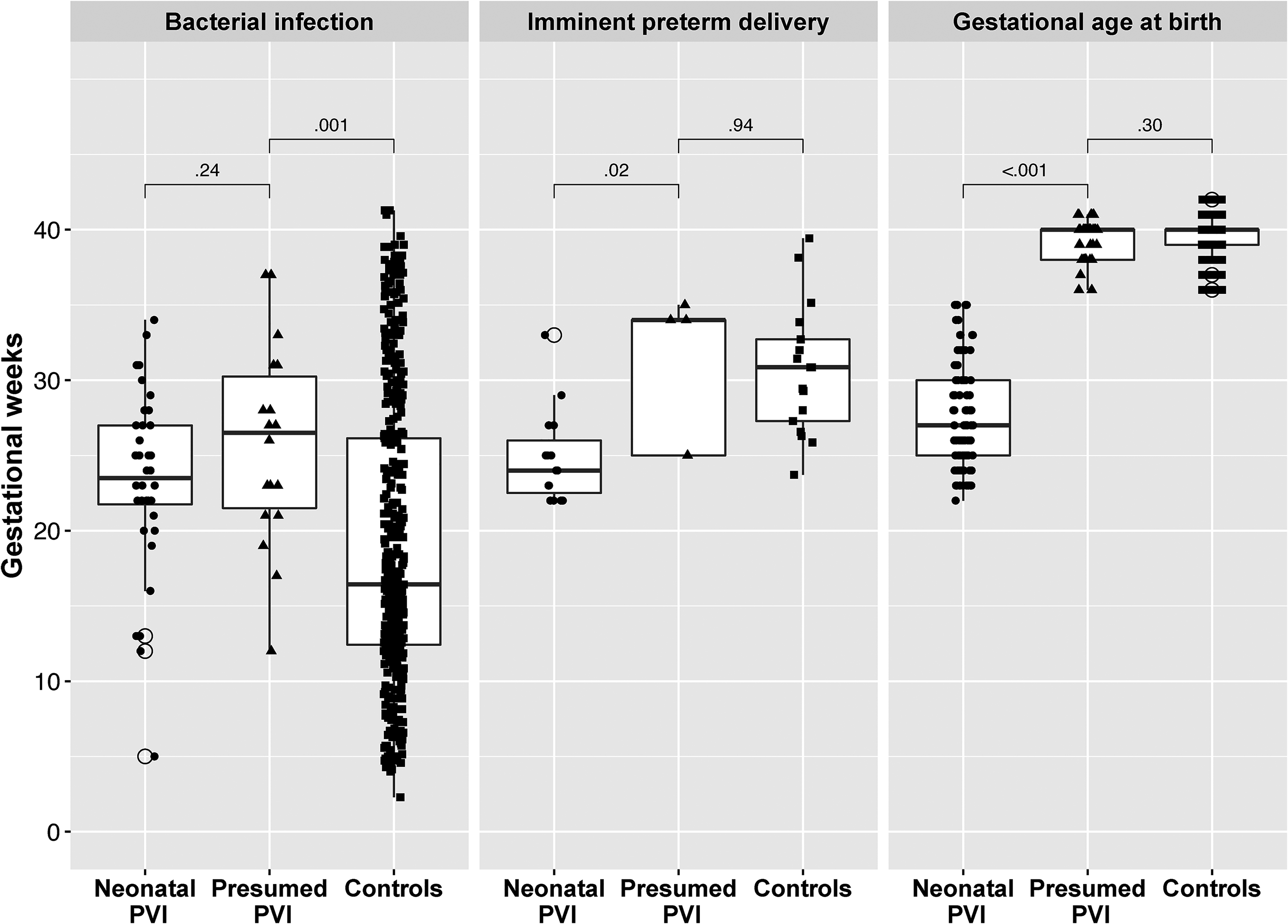
Individual data of gestational age at the time of bacterial infection, imminent preterm delivery, and birth.
Multiple Logistic Regression Analysis
Multiple logistic regression analysis revealed genitourinary tract infection (aOR 3.1, 95% CI 1.5-6.2; P = .002), other bacterial infection but genitourinary tract infection (aOR 4.4, 95% CI 2.0-9.8; P < .001), and preeclampsia (aOR 4.3, 95% CI 1.3-14; P = .02) as independent predictors for presumed periventricular venous infarction compared to control.
Twin pregnancy (aOR 8.3, 95% CI 1.4-48; P = .02) and at least 1 previous spontaneous pregnancy loss (aOR 3.0, 95% CI 1.2-7.6; P = .02) were independent predictors for neonatal versus presumed periventricular venous infarction in multiple logistic regression analysis.
Illustrative Cases of Presumed and Neonatal Periventricular Venous Infarction
Illustrative ultrasonography and MRI findings of similarities between presumed and neonatal periventricular venous infarction children are presented in Figure 5. Fetal brain damage at a certain stage of vascular development between gestational weeks 21 and 31 accounts for similar MRI findings, although the stroke is diagnosed at different ages.
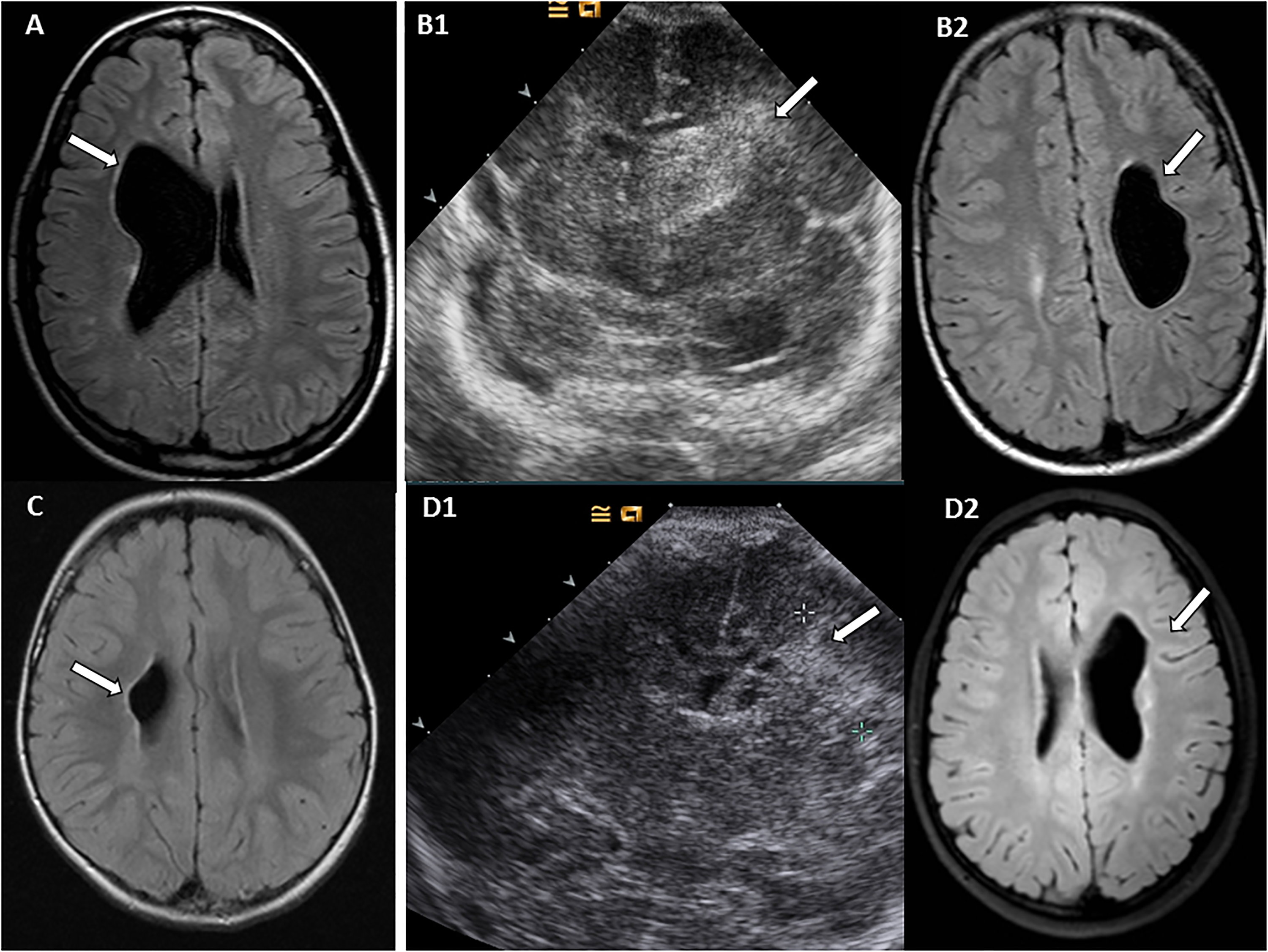
Illustrative cases of presumed and neonatal periventricular venous infarction with similar magnetic resonance imaging (MRI) findings. (A) A girl with presumed periventricular venous infarction was born at gestational week 40. Her mother had Escherichia coli pyelonephritis at gestational weeks 25 and at 27. At age 17 years, the girl has moderate developmental delay, mild left-side hemiparesis, and epilepsy. Axial T1-weighted (W) MRI view shows right-side periventricular venous infarction. (B) A boy with neonatal periventricular venous infarction was born at gestational week 27 and was treated for Escherichia coli congenital sepsis. His mother had E coli pyelonephritis at gestational week 20. (B1) At age 35 hours, cerebral ultrasonography shows left-side periventricular venous infarction in coronal view. (B2) At age 13 years, the boy cannot walk independently because of tetraparesis, he has mild cognitive delay, language problems, and epilepsy. Axial T1-weighted MRI view shows left-side periventricular venous infarction. (C) A girl with presumed periventricular venous infarction was born at gestational week 41. Her mother was hospitalized at gestational week 21 on day 4 of pyelonephritis. At age 8 years, she has severe hemiparesis and normal cognitive development. T1-weighted axial view shows right-side periventricular venous infarction. (D) A boy with neonatal periventricular venous infarction was born at gestational week 27 and was treated for congenital culture negative sepsis. Her mother was treated for pyelonephritis for 4 days prior to delivery. (D1) Cerebral ultrasonography on day 3 shows periventricular venous infarction in coronal view with cystic degeneration, suggesting antenatal development of periventricular venous infarction. (D2) At age 10 years, he has hemiparesis, mild cognitive delay, and epilepsy. Axial T1-weighted view shows left-side periventricular venous infarction.
Discussion
Our study provides evidence of possible association between periventricular venous infarction and maternal bacterial infection during pregnancy. Maternal bacterial infection occurred 3.4 times more often during the pregnancies of presumed periventricular venous infarction compared to the prospectively closely monitored consecutive control cohort of women with term or near-term deliveries. More precisely, the odds to have maternal pyelonephritis versus no bacterial infection were 50 times higher in presumed periventricular venous infarction versus control. In both the periventricular venous infarction and control groups, genitourinary tract infection was the most prevalent infection affecting at least two-thirds of the mothers receiving antibacterial treatment. Asymptomatic bacteriuria was predominating in the control group, whereas only 3% of the mothers with infection developed pyelonephritis. Thus, as suggested earlier, 24 development of pyelonephritis in pregnant women is not common. In contrast, in the presumed periventricular venous infarction group, pyelonephritis accounted for 50% and in the neonatal periventricular venous infarction group, for 36% of bacterial infection cases, whereas there were no cases of asymptomatic bacteriuria in either group. Maternal genitourinary tract infection was an independent risk factor for development of presumed periventricular venous infarction compared to control. These findings are in line with an earlier study describing association between hospital-reported maternal genitourinary tract infection and cerebral palsy in term-born children (hazard ratio 2.5, 95% CI 1.6-4.1). 25
Another important finding of our study was the similar incidence and timing of severe bacterial infection, including genitourinary tract infection and pyelonephritis, in mothers of children with presumed and neonatal periventricular venous infarction. Most of the infections in both periventricular venous infarction groups occurred between gestational weeks 21 and 31. This suggests that the effect of genitourinary tract infection occurs at the same gestational age and stage of vascular development of the fetal brain. In a previous study, 61% of antenatal IVH and periventricular venous infarction cases were diagnosed before gestational week 28. 21 In another study, the median age of antenatal stroke diagnosed by ultrasonography was gestational week 31.5 (range: gestational weeks 22-34). 26 The suggested time of presumed periventricular venous infarction from gestational weeks 24 through 34 4 or prior to gestational week 32 12 has been based merely on the known pathophysiology of germinal matrix hemorrhage. An earlier study provides evidence of possible association between maternal infection, especially pyelonephritis, from gestational weeks 21 to 31 and presumed periventricular venous infarction.
Although every fifth pregnant woman in our control group received antibacterial treatment, the median time of infection during pregnancy was significantly earlier compared to the presumed periventricular venous infarction group. This can be explained by the high rate of asymptomatic bacteriuria or chlamydia diagnosed and treated on the basis of routine screening during the first half of pregnancy, in the control group.
Intrauterine infection is considered to be one of the major maternal insults during pregnancy. Maternal infection, including chorioamnionitis, can initiate antenatal inflammatory response leading to injury of the developing brain and other fetal organs 27 and can cause preterm birth. 24 Several authors have noted an increased risk for severe germinal matrix hemorrhage and poor neurodevelopmental outcome in preterm infants exposed to antenatal infection.27,28 Studies on long-term outcome in term-born children after the mothers’ bacterial infection have been scarce. In a population-based cohort study, the third-trimester diagnosis of any hospital-reported maternal infection was associated with cerebral palsy in term-born children (hazard ratio 1.8, 95% CI 1.2-2.8). 25 Another meta-analysis found significant association of clinical and histologic chorioamnionitis with cerebral palsy. 29
Other Pregnancy Risk Factors
Recurrent miscarriage and antepartum bleeding can be risk factors for presumed periventricular venous infarction.2,5 In the present cohort, the number of mothers with imminent preterm delivery at gestational week ≥22 was higher among cases of presumed periventricular venous infarction compared to control; however, this difference was not significant after FDR correction. Preeclampsia and neonate being small for gestational age have been associated with presumed periventricular venous infarction in some earlier studies but have not been confirmed in a case-control study.5,16 In our study, the prevalence of preeclampsia and neonate being small for gestational age <10 percentiles was higher in the presumed periventricular venous infarction group compared to control, but after FDR correction the difference was not significant. However, in multiple regression analysis, preeclampsia was identified as an independent risk factor for presumed periventricular venous infarction.
Obstetric Risk Factors
We found no evidence of the role of obstetric risk factors like dystocia at birth, emergency caesarean section, placental abruption, chorioamnionitis at term delivery, and low Apgar scores in development of presumed periventricular venous infarction. This suggests that fetuses with presumed periventricular venous infarction have stabilized by birth and will likely undergo an uneventful delivery. Still, we found significantly lower Apgar scores, higher rate of placental abruption, and need for emergency caesarean section, indicating acute distress, in the neonatal periventricular venous infarction group compared with the presumed periventricular venous infarction group.
In multifactorial analysis, twin pregnancy and previous pregnancy loss were associated with neonatal periventricular venous infarction. Previous pregnancy loss may indicate that not only the prenatal environment but possibly also genetic factors are involved in development of periventricular venous infarction in children with neonatal periventricular venous infarction. 30
In Utero Occurrence of Periventricular Venous Infarction
As suggested previously, 12 presumed periventricular venous infarction represents in utero occurrence of a well-known acute version of periventricular venous infarction in preterm children. Antenatal stroke, often subclinical during pregnancy, may result in antenatal or neonatal death in up to 51% of cases.21,26 Although most preterm children develop periventricular venous infarction after birth, in a prospective study of infants born before gestational week 34, periventricular venous infarction was considered to be of antenatal origin in 27% of those who died. 15 Some fetuses with severe IVH or periventricular venous infarction are discovered during prenatal ultrasonography.21,26,31 Possibly, some fetuses with antenatal periventricular venous infarction are not detected in the fetal period, and survive acute insult and stabilize. These children with presumed periventricular venous infarction show no distress during term delivery, have no neurologic symptoms after birth, and periventricular venous infarction is detected only when hemiparesis or seizures will develop later during infancy. 2 After typical chronic periventricular damage has been found on MRI, presumed periventricular venous infarction is diagnosed.2,5,7,16
Limitations
The current study is subject to several limitations, including the retrospective collection of data about pregnancy risk factors for children with periventricular venous infarction. The cases of presumed and neonatal periventricular venous infarction were collected retrospectively from 1994 through 2014, and between 2014 and 2019, a total of 14 additional presumed periventricular venous infarction cases were included prospectively. However, the data of pregnancy risk factors for presumed periventricular venous infarction was still collected retrospectively because of the nature of this condition. Regrettable, a suitable control group was only available for the later years (2013-2015).
To minimize any possible bias, a well-controllable approach with standardized definitions of risk factors, available from routine medical records, was employed. If anything, we may have underestimated the true prevalence of infections in the periventricular venous infarction groups, as no data on outpatient antibiotic treatment or bacteriuria screening were available for earlier cases.
The study period was long including the time when presumed perinatal stroke was not a widely recognized entity; thus, some term-born children with mild hemiparesis as presumed periventricular venous infarction cases may have been missed. However, the problem of underdiagnosing presumed periventricular venous infarction is universal. 26
Changes in treatment strategies for preterm delivery at higher-level hospitals, including prenatal steroid administration and advances in postnatal care, can play a role in development of severe IVH or periventricular venous infarction in preterm infants. 11 Still, pregnancy-related risk factors have most likely remained unchanged. All participants of this study, like the general population of the study country, were Caucasians.
As suggested earlier, the etiology of neonatal periventricular venous infarction is probably multifactorial with contributions from both the environment during pregnancy and the genetic factors of the child. 30 In contrast to earlier findings, 16 a recent report did not confirm the previously stated higher risk of thrombophilia in children with perinatal stroke, including presumed periventricular venous infarction, compared to control. 32 In some periventricular venous infarction cases, connective tissue disorders may be a predisposing factor for germinal matrix hemorrhage. 30 Although further studies are urgently needed, clarification of possible genetic predisposition remains beyond the scope of the current analysis.
The results of this study suggest that active measures should be taken to prevent and immediately treat all maternal bacterial infections including asymptomatic bacteriuria, especially pyelonephritis, to avoid the inflammatory response of the fetus. The awareness of potential related risks should be raised among obstetricians and other specialists who regularly treat bacterial infections in pregnant women.
Our study demonstrates that children from pregnancies complicated with pyelonephritis may require neurologic follow-up and early MRI in case hemiparesis is detected. Early diagnosis facilitates timely rehabilitation of perinatal stroke patients.
Conclusion
Our data show an increased risk for the development of periventricular venous infarction in the case of maternal bacterial infections, especially between gestational weeks 21 and 31.
Footnotes
Acknowledgments
The authors thank all pregnant women, patients, and their parents, as well as the staff of the archives of the hospitals, for contribution.
Author Contributions
Conceptualization: NI, RL, PI; Data curation: NI, RL, KR, TM, MLa, MLi, DL PI; Formal Analysis: NI, PK,PI; Funding acquisition: RL, PI, MLa; Supervision: MLa, KR, PI; Statistical analysis: PK, NI, PI; Writing – original draft: NI, PI, RL, KR, TM, MLa; Writing – review &editing: all authors. All authors contributed to manuscript revision, read, and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Eesti Teadusagentuur (grant number PRG1021, PUT 148).
Ethical Approval
The studies were approved by the Ethics Review Committee on Human Research of the University. Happy Pregnancy study for the control group was approved by protocols no. 221/T-6, 17.12.2012, and no. 286/M-18, 15.10.2018, and all recruited participants signed a written informed consent. The periventricular venous infarction study was approved by protocol no. 294/M-18, 17.06.2019. The data of the risk factors for children with neonatal or presumed periventricular venous infarction were collected retrospectively, and the committee waived the need for patient consent.
