Abstract
Varicella-zoster virus vaccination is recommended for virtually all young children in the United States, Canada, and several other countries. Varicella vaccine is a live attenuated virus that retains some of its neurotropic properties. Herpes zoster caused by vaccine virus still occurs in immunized children, although the rate is much lower than in children who had wild-type varicella. It was commonly thought that 2 varicella vaccinations would protect children against the most serious complication of meningitis following herpes zoster; however, 2 meningitis cases have already been published. We now report a third case of varicella vaccine meningitis and define risk factors shared by all 3 immunized adolescents. The diagnosis in cerebrospinal fluid in this third case was verified by amplifying and sequencing portions of the viral genome, to document fixed alleles found only in the vaccine strain. Viral antibody was also detected in the cerebrospinal fluid by confocal microscopy. When compared with the other 2 cases, remarkably all 3 were 14 years old when meningitis occurred. All 3 were treated with intravenous acyclovir, with complete recovery. The adolescent in our case report also had recurrent asthma, which was treated with both prednisone tablets and beclomethasone inhaler before onset of meningitis. When the 3 cases were considered together, they suggested that immunity to varicella-zoster virus may be waning sufficiently in some twice-immunized adolescents to make them vulnerable to varicella vaccine virus reactivation and subsequent meningitis. This complication rarely happens in children after wild-type varicella.
Meningitis as a complication of herpes zoster is extraordinarily unusual in immunocompetent children and adolescents who have had wild-type varicella after their first birthday. 1 No cases of associated meningitis were documented in a 22-year retrospective survey of herpes zoster cases in otherwise healthy children (through age 19 years) seen by pediatricians at the Mayo Clinic before approval of varicella vaccination. 2 In a prior report, we described 3 children with severe cutaneous herpes zoster caused by the varicella vaccine virus. 3 In this report, we extend that observation to include an even more serious and unusual complication of varicella vaccination and herpes zoster, namely, varicella vaccine meningitis. The live attenuated varicella vaccine (strain Oka; Varivax) was approved for administration to children in the United States by the Food and Drug Administration in 1995. 4 Extensive clinical trials had been carried out in both Japan and the United States. 5 The first trials were carried out in Japan because the vaccine was developed by the Takahashi virology laboratory in Osaka. 6 The vaccine effectiveness is high, and the disease varicella has nearly disappeared from the United States because of high immunization rates. 7,8 The safety profile has also been excellent. 9
The varicella vaccine virus is a live attenuated virus. Therefore, like its wild-type counterpart, the vaccine virus can enter neurons after local replication in the skin. The vaccine virus can also establish latency in the dorsal root ganglia and occasionally reactivate to cause herpes zoster. 10 Herpes zoster after varicella vaccination is generally less common and less severe than herpes zoster after wild-type varicella disease. 11 Vaccine-related herpes zoster can also lead to meningitis. 12,13 Because these earlier cases of varicella vaccine meningitis have occurred following herpes zoster in children who have received only a single varicella vaccination, it was thought that 2 varicella vaccinations would effectively protect against subsequent varicella vaccine meningitis. 14 However, we now present the third case of viral meningitis caused by varicella vaccine virus in a twice-immunized and otherwise healthy adolescent. We also review the 2 prior varicella vaccine meningitis cases in twice-immunocompetent adolescents, both of which are very similar to our case. 15,16 Altogether, these 3 cases elucidate risk factors for varicella vaccine meningitis in older children who have received 2 varicella vaccinations.
Methods
We measured levels of varicella antibody in cerebrospinal fluid by an adaption of the varicella fluorescent antibody to membrane antigen test, in which we substituted confocal microscopy for conventional fluorescence microscopy. 17,18 Confocal microscopy is several-fold more sensitive than conventional fluorescence microscopy for detection of antibody. The 3-D confocal images were prepared with the Imaris software program. Likewise, methods for varicella-specific sequencing have been described in earlier publications from this virology laboratory. 3,19,20 Our methods follow guidelines among varicella laboratories for identifying fixed alleles specific to the genome of the varicella vaccine virus (strain Oka). 21 -24 Briefly, the cerebrospinal fluid sample was stored frozen after aliquots were removed for the initial screening tests (cell count, glucose, protein, bacterial culture, screening PCR). Subsequently, the sample was thawed for the first time in order to perform partial sequencing of the viral genome known to be present in cerebrospinal fluid after the varicella-positive screening PCR test.
Because the varicella open reading frame (ORF) 62 contains the most single nucleotide polymorphisms specific to the vaccine strain, regions of ORF62 were PCR-amplified from the sample of cerebrospinal fluid using previously described primers and the Expand Hi Fidelity DNA polymerase (Roche Diagnostics, 11732641001). 25 The PCR amplicons were then sequenced by Sanger methodology at the University of Iowa Core Facility. The generated sequences were then aligned to either the varicella wild-type reference sequence (Dumas, GenBank X04370.1) or the varicella vaccine virus sequence (vOka, Genbank AB097932) to provide a consensus sequence using a locally written computer program.
Results
Illustrative Case Report
The patient was a 14-year-old female adolescent admitted to hospital in 2019 because of increasingly severe headaches. The headaches were accompanied by dizziness, nausea, vomiting, and confusion. When she first presented in the emergency department late in the evening, she was noted to be febrile. Her weight was 72 kg, with a body mass index of 23. A complete blood cell count and metabolic panel were unremarkable. A head computed tomogram was performed and read as normal. Thereafter, a lumbar puncture was performed, with the following results on cerebrospinal fluid: colorless; protein, 132 mg/dL; glucose, 45 mg/dL; red blood cells, 5/mm3; and nucleated cell count, 775/mm3, with 82% lymphocytes. A Biofire Filmarray encephalitis/meningitis multiplex panel test on the cerebrospinal fluid was positive for varicella-zoster virus. Within 12 hours after admission, therapy with intravenous acyclovir (30 mg/kg/d) was initiated. Later in the same day, a small cluster of vesicles was observed over her right knee (see Discussion and Figure 2, panel C). Fluid from one vesicle was tested by Focus Integrated Cycler for both herpes simplex virus and varicella virus DNA; only varicella was positive. Head magnetic resonance imaging was performed and was normal except for increased enhancement along the leptomeningeal surface, consistent with meningitis.
On further questioning after admission, the patient revealed that she had been having intermittent but less severe headaches as well as abdominal pain and back pain for 2 weeks. She had gone to a different medical clinic, where a computed tomogram of the abdomen/pelvis with contrast was performed 10 days before the current admission. That scan did not show any abnormalities. During her hospitalization, she completed a 7-day course of intravenous acyclovir, followed by 2 weeks of valacyclovir tablets (1 g thrice daily), with complete resolution of symptoms.
Her past medical history was also notable for recurrent bouts of asthma since age 3 years; the asthma attacks were induced by viral respiratory infections. Because of continued exacerbations up to the present time, she was intermittently using an oral metered-dose inhaler of beclomethasone (80 µg twice daily). Four weeks before her current admission, her asthma worsened and she had sought treatment at a third medical facility. At that clinic, prednisone tablets 20 mg daily for 5 days were prescribed. She had never taken prednisone tablets in the past. Four days later, she refilled her prescription for the beclomethasone inhaler, which she had continued to use until admission to the hospital.
She had received all recommended childhood immunizations, including a live attenuated varicella vaccine at ages 1 and 5 years. She had received a measles-mumps-rubella vaccination on the same 2 days. Neither she nor her mother recalled any family member with varicella or herpes zoster rashes in the month preceding onset of meningitis. Therefore, there was no possibility of a recent transmission of varicella vaccine virus to the adolescent. Also see further information in the Discussion.
One month after discharge, she returned for an immunology evaluation. Serum levels of immunoglobulins IgM, IgA, and IgG were within normal limits, as were levels of the 4 IgG subclasses. Similarly, the levels of IgG antibodies to both tetanus toxoid and diphtheria toxoid were within normal ranges for a child who had previously received 4 tetanus-diphtheria-pertussis vaccinations. The total serum complement level was normal. The B-cell phenotyping profile for immunodeficiency and immune competence assessment was normal. The percentages of CD3, CD4, CD8, and CD16 + CD56 lymphocytes were normal by flow cytometry analysis. Likewise, a CD8 immune competence assessment was normal as measured by gamma interferon level and CD107a/CD107b markers. All immunology testing was performed by Mayo Medical Laboratories, Rochester, MN.
Varicella Antibody in Cerebrospinal Fluid
In prior studies, the presence of varicella antibody in cerebrospinal fluid has been a sensitive indicator of central nervous system infection, sometimes more sensitive than measuring varicella DNA. 26 -28 To confirm the result of the multiplex assay, we tested for varicella antibody in the cerebrospinal fluid of our patient and the test was positive (Figure 1). Four representative images are shown in panels A to D; arrows designate the presence of fluorescent-labeled varicella antibody in the cerebrospinal fluid reacting with virus-infected cells. Panels A and B include 2-D images and panels C and D include renderings of 3-D images; panels E and F are positive and negative control images, respectively.
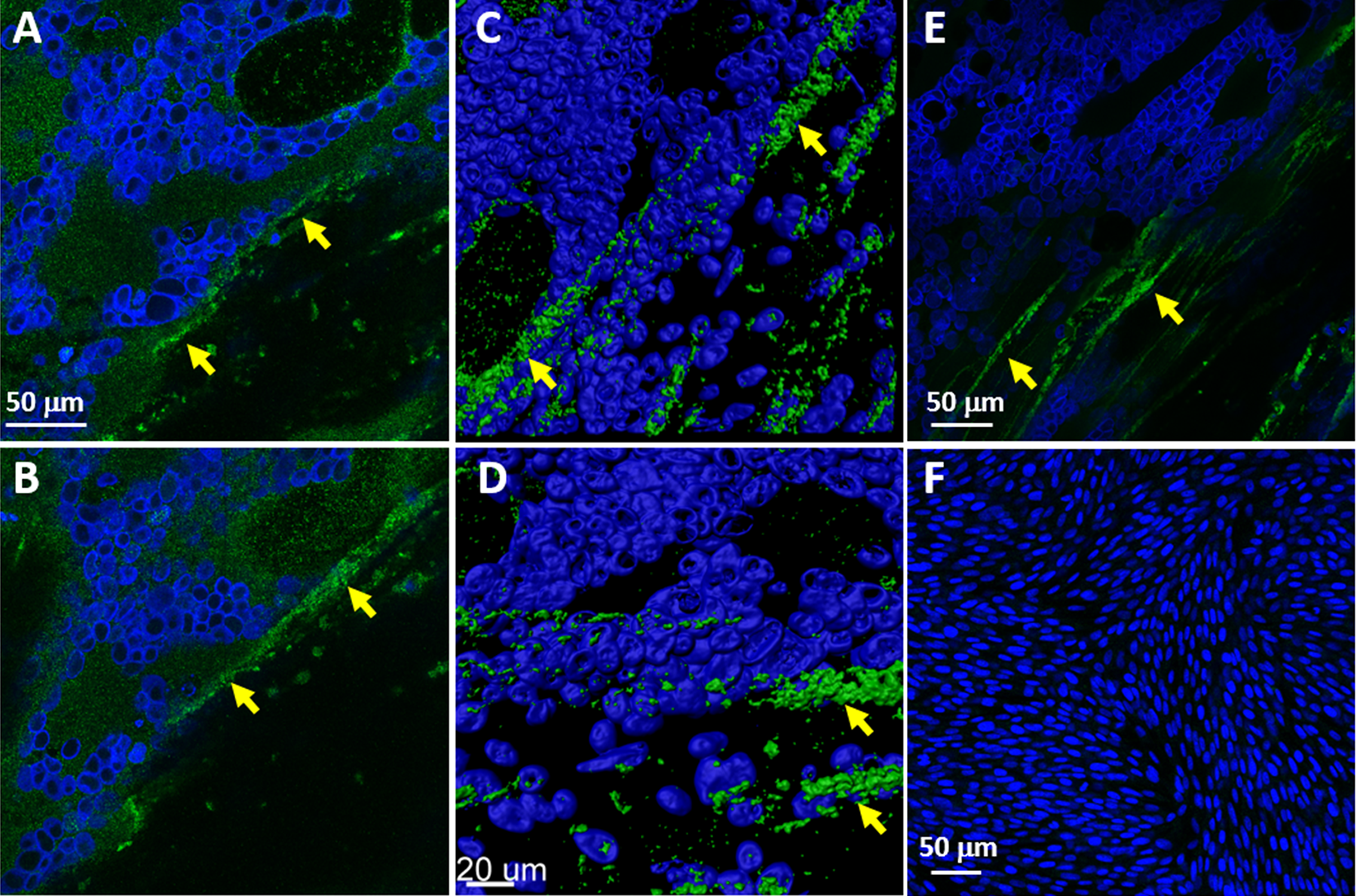
Detection of antibody to varicella virus in the cerebrospinal fluid of meningitis case. The patient’s antibody that attaches to virus-infected cells is stained fluorescent green; these positive areas are designated by yellow arrows (A-D). Positive and negative controls (E, F). Nuclei within the cells are stained dark blue (For interpretation of the references to colours in this figure legend, refer to the online version of this article).
Viral Sequencing Data
The commercial Biofire Filmarray screening test of cerebrospinal fluid is both sensitive and specific. 29 The test was positive for varicella DNA. However, the test kit does not differentiate wild-type from vaccine-type varicella strains. Therefore, further sequencing was necessary to amplify specific varicella genes in the cerebrospinal fluid sample. The viral genome contains around 70 individual genes. A very informative sequence is ORF62, because there are several single nucleotide polymorphisms in ORF62 that are always found in vaccine-type ORF62 genes but are never found in wild-type varicella ORF62 genes. 30,31 The viral sequencing data demonstrated that the patient’s isolate was vaccine-type and not wild-type. For example, the polymorphism at nucleotide position 107252, which is always found in vaccine-type virus but never in wild-type virus, was present in the viral DNA of our patient. 21 Thus, our varicella sequencing results confirmed that the vaccine strain was responsible for the meningitis. These results were very similar to those previously reported in cases from 2017 and 2019 (Table 1).
Prominent Features Among 3 Immunized and Immunocompetent Adolescents With Varicella Vaccine Meningitis and Herpes Zoster.
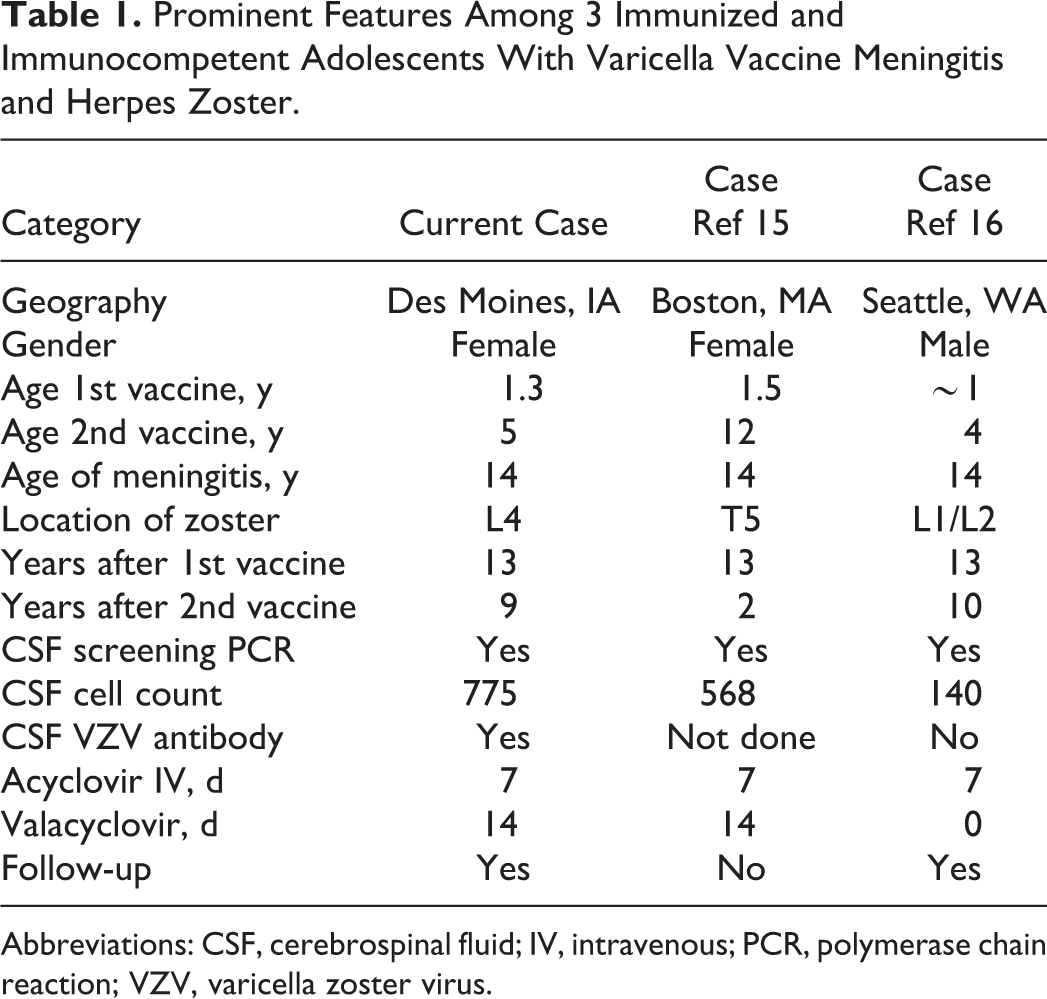
Abbreviations: CSF, cerebrospinal fluid; IV, intravenous; PCR, polymerase chain reaction; VZV, varicella zoster virus.
Comparison With 2 Prior Cases
Based on our review of the literature, we have diagnosed the third case of varicella vaccine meningitis in a twice-immunized immunocompetent adolescent. There are strikingly similarities among the 3 cases, as shown in Table 1. First, all 3 patients were aged 14 years. 15,16 Second, the 3 patients have likely had latent vaccine virus in their dorsal root ganglia for 13 years, since their first varicella vaccination. Third, 2 of the 3 patients had herpes zoster in their lower extremities. Note that we did not include another patient described in reference 16, because that patient was immunocompromised when he developed herpes zoster. As mentioned in the Introduction, we also did not include any meningitis cases that occurred in children who had received only 1 varicella vaccination.
Most varicella vaccinations in children under the age of 2 years are administered by injection into the thigh. Thus, the location of herpes zoster in our patient’s lower extremity was most likely secondary to local viral replication in the thigh following her initial varicella vaccination and subsequent orthograde transport of vaccine virus via sensory nerves to lumbar dorsal root ganglia innervating the L2-L4 dermatomes, followed by re-emergence of the virus in the lower extremity years later. The location of thoracic herpes zoster following immunization is easily explained, based on early data from Japan that showed about a 50% likelihood of viremia after the first varicella vaccination. 32 During the viremia, the virus either directly entered thoracic dorsal root ganglia after exiting their segmental artery blood supply or the virus exited capillaries beneath the truncal skin and caused a few tiny unrecognized vesicles with sequent spread of virus to thoracic dorsal root ganglia.
Discussion
In a Medline literature search between 1960 and 2004, we found 1 case report of herpes zoster meningitis in an otherwise healthy 12-year-old boy who had had wild-type varicella and no varicella vaccination. 1,2 We now present data from 3 patients described in Table 1, suggesting that some vaccine recipients who have received 2 varicella vaccinations beginning after their first birthday already are exhibiting waning immunity by age 14 years, sufficient to allow reactivation with varicella vaccine meningitis as a complication of herpes zoster. The phenomenon that twice-immunized children could lose immunity within 9-10 years after their second immunization had not been anticipated. But there are similarities to laboratory findings in older adults who have herpes zoster. Clinical studies performed decades ago in the United States and Finland revealed that 38% to 46% of adult herpes zoster patients will have a pleocytosis in the cerebrospinal fluid, an inflammatory response highly suggestive of viral meningitis. 33,34 In the 3 cases of vaccinated adolescents (Table 1), all 3 patients had headache and all 3 had pleocytosis with detectable varicella DNA in the cerebrospinal fluid. We know that vaccine virus reactivates less frequently than wild-type virus, 11,35 but we may never know the percentage of children with abnormal cerebrospinal fluid values after vaccine-related herpes zoster without clinical meningitis.
In our patient, one prior risk factor came to particular attention. The patient has had a diagnosis of asthma since 2007 and frequently used bronchodilating inhalers, but never systemic corticosteroids until 2019. She exhibited the most common clinical asthma phenotype with bronchial hyperresponsiveness following viral respiratory infection. 36 We have shown an increased risk of herpes zoster in children with asthma, irrespective of inhaled corticosteroid usage 37,38 ; most of these children would have had wild-type varicella. In our 2019 report of 3 cases of severe herpes zoster caused by varicella vaccine virus, one of the 3 children developed asthma after he recovered from the shingles. 3 Other groups have shown an increased risk of herpes zoster in both children and adults with asthma. 39 -42 In our current case, a precipitating risk factor for herpes zoster may have been the addition of prednisone tablets to her standard inhaled beclomethasone regimen for treatment of recurrent asthma. There was no information about an asthma history in the other 2 case reports (Table 1).
Systemic corticosteroid regimens have been known to precipitate herpes zoster in children with cancer and other chronic diseases since the 1950s. 43,44 Based on studies examining the risk of herpes zoster in patients being treated for various rheumatologic diseases, dosages of prednisone of over 7.5 mg per day were sufficient to increase the likelihood of developing herpes zoster. 45 -47 Many of these children with chronic rheumatologic diseases were taking prednisone for long periods. However, some of the cases of herpes zoster occurred within weeks of starting prednisone therapy. 48 Of further interest, data from the older literature suggests that children with asthma who were recently treated with corticosteroids were particularly vulnerable to adverse events following primary varicella infection. 49 -51 In spite of the concerns listed in the preceding sentences, we note that the dosage of prednisone received by our patient was not high when her weight was taken into consideration.
Further, we propose a pathogenesis model for varicella vaccine meningitis, based not only on a varicella animal model 52 but also on data from a closely related herpesvirus of swine, namely, pseudorabies virus (Figure 2). Zosteriform rashes during pseudorabies disease are secondary to virus traveling in itch and pain sensory fibers. 53 In our adolescent patient, vaccine virus entered the dorsal root ganglia after the first varicella vaccination at age 1 year (A). At age 14 years, vaccine virus acquired at age 1 year reactivated from latency (B) and traveled in sensory fibers to both the skin over the right leg (C) and also to the gray matter of the adjacent spinal cord. 54 Viral replication in the spinal cord led to meningitis (D). We postulate that rapid initiation of intravenous acyclovir therapy may have prevented a progression from meningitis to meningoencephalitis (E). Finally, although this report is meant to raise awareness of varicella vaccine meningitis, we note the rarity of this neurologic infectious disease in the United States.
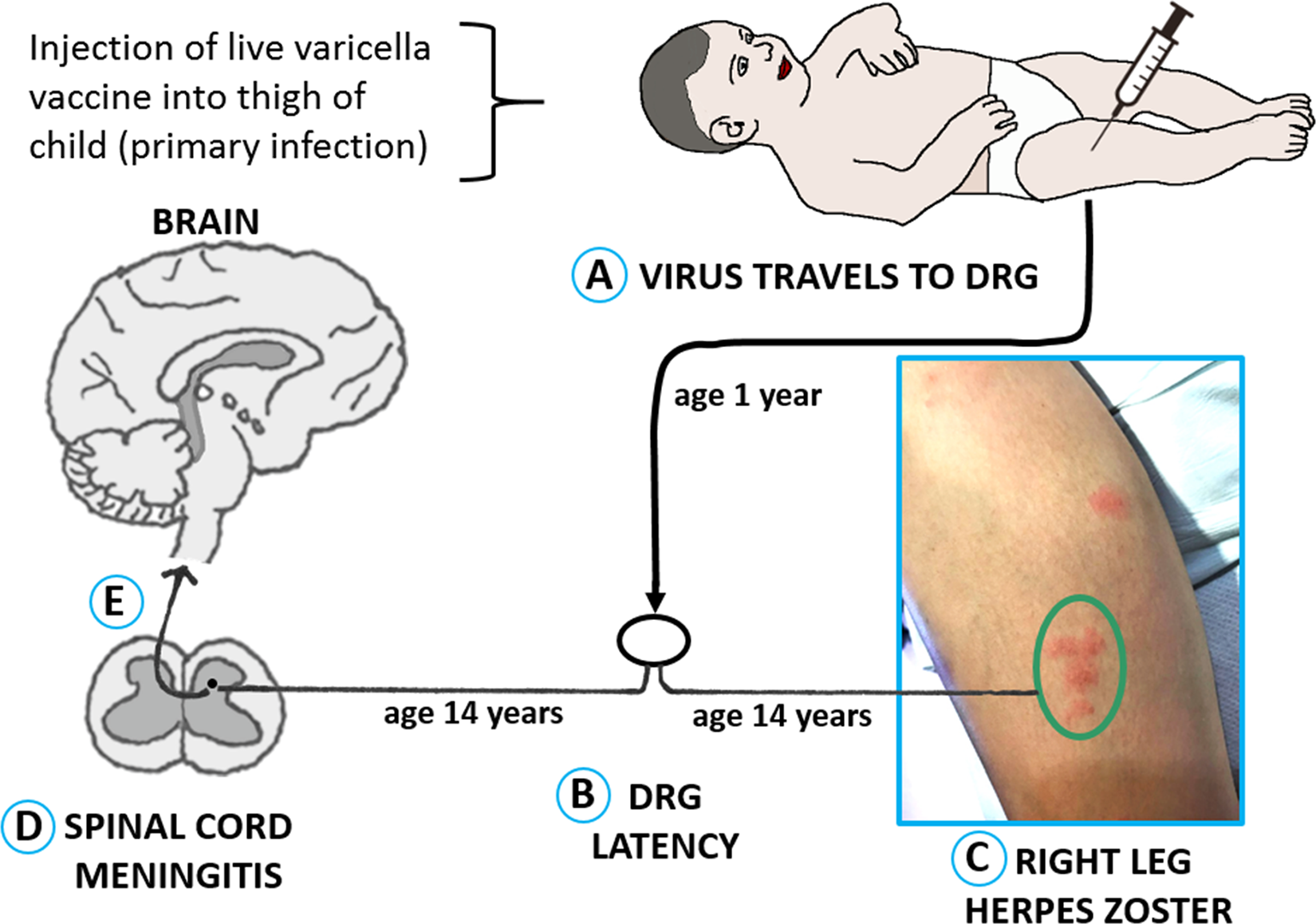
Model for pathogenesis of varicella vaccine meningitis following herpes zoster in an immunized adolescent. This figure includes a photo of the herpes zoster rash on the right leg of our case of meningitis (panel C). DRG, dorsal root ganglia.
Footnotes
Author Contributions
VR, SCE, KLR, and MW provided clinical management; WJ, JEC, and CG carried out the virology assays and prepared the figures; RJC reviewed the methodology from the virology assays; VR, CG, and DJB prepared the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research by Dr Grose is funded in part by NIH grant AI153817, which supports research to advance vaccine safety.
Ethical Approval
This case study was approved by the University of Iowa Institutional Review Board, Iowa City, IA, USA.
