Abstract
Invasive lobular carcinoma (ILC) poses distinct diagnostic challenges due to its infiltrative single-file growth pattern, which often renders it mammographically occult, particularly in dense breast tissue. Contrast-enhanced mammography (CEM) combines the anatomical detail of conventional mammography with functional information from contrast uptake, likely improving the detection, staging, and assessment of ILC compared to conventional imaging techniques. CEM shows value in evaluating ILC tumor size and disease extent, especially in multifocal and multicentric disease, although MRI remains the gold standard. This review outlines the spectrum of ILC imaging features on CEM, including findings on both low-energy and recombined images. While CEM can provide ILC size and extent estimates comparable to MRI, its accuracy may be reduced in cases of non-mass enhancement or tumors larger than 3 cm. Additionally, ILC may demonstrate lower conspicuity enhancement than invasive ductal carcinoma (IDC), necessitating careful image interpretation. As clinical adoption of CEM increases, radiologists must become familiar with the variable imaging characteristics of ILC, to facilitate more accurate interpretation. Improved recognition of these features has the potential to support more precise treatment planning and better patient outcomes.
Introduction
Overview of Invasive Lobular Carcinoma
Invasive lobular carcinoma (ILC) is the second most common histologic subtype of breast cancer after invasive ductal carcinoma (IDC), accounting for approximately 5% to 15% of all invasive breast cancers.1,2 Compared to IDC, ILC typically presents in older patients and is more frequently associated with advanced stage at diagnosis, larger tumor size, and lymph node involvement.3,4 Due to its characteristic non–mass-forming and infiltrative growth pattern, ILC can be difficult to detect using conventional imaging modalities such as mammography and ultrasound. 5 Pathologically, ILC is characterized by a linear, single-file infiltrative growth pattern, often accompanied by a relative absence of desmoplastic stromal reaction, hemorrhage, necrosis, or calcification.1,6 Approximately 90% of ILCs exhibit loss of E-cadherin expression, along with cytoplasmic accumulation of p120 catenin, consistent with impaired cell adhesion and lobular differentiation.7,8 ILC tumors are most frequently estrogen receptor (ER)–positive and human epidermal growth factor receptor 2 (HER2)–negative.9,10 Given the subtle and infiltrative nature of ILC, which often leads to underestimation or missed detection on conventional imaging, there is increasing interest in alternative modalities that may improve diagnostic accuracy. In particular, functional imaging techniques that evaluate tumor vascularity, such as contrast-enhanced mammography (CEM), offer a promising approach to overcoming the limitations of structural imaging alone.
Overview of Contrast Enhanced Mammography
Contrast-enhanced mammography (CEM) is a dual-energy imaging technique that creates recombined (RC) images from the logarithmic subtraction of low-energy (LE) images from non-diagnostic high-energy images following the administration of iodinated contrast material. The LE images are comparable to standard mammograms, while RC images provide functional information on tumor vascularity; analogous to subtraction images in MRI. 11 This functional imaging enhances the detection of malignancy, particularly in dense breast tissue.
This review focuses on the imaging features of ILC on CEM; a relatively new modality for which there is currently limited published data on ILC-specific appearances. Improved recognition and understanding of these imaging characteristics promise to support radiologists in the accurate detection and assessment of ILC.
ILC Appearances on CEM
Low Energy (LE) Imaging Findings
The LE images obtained during CEM are most commonly obtained as 2D images which are analogous to standard full-field digital mammography (FFDM), and interpreted using the American College of Radiology (ACR) Breast Imaging Reporting and Data System (BI-RADS) mammography lexicon.12,13 Due to its subtle growth pattern and typical lack of calcifications, ILC may be mammographically occult in up to 30% of cases.1,14 Additionally, ILC may be visualized on only a single projection in roughly 35% of cases, most frequently on the craniocaudal (CC) view.15,16 ILC can be difficult to detect on FFDM due to a lack of central opacity or lack of conspicuous difference in density from the surrounding parenchyma (Figure 1).15,17 This challenge is further compounded in individuals with heterogeneously dense or extremely dense breasts (BI-RADS density categories c and d).15,18
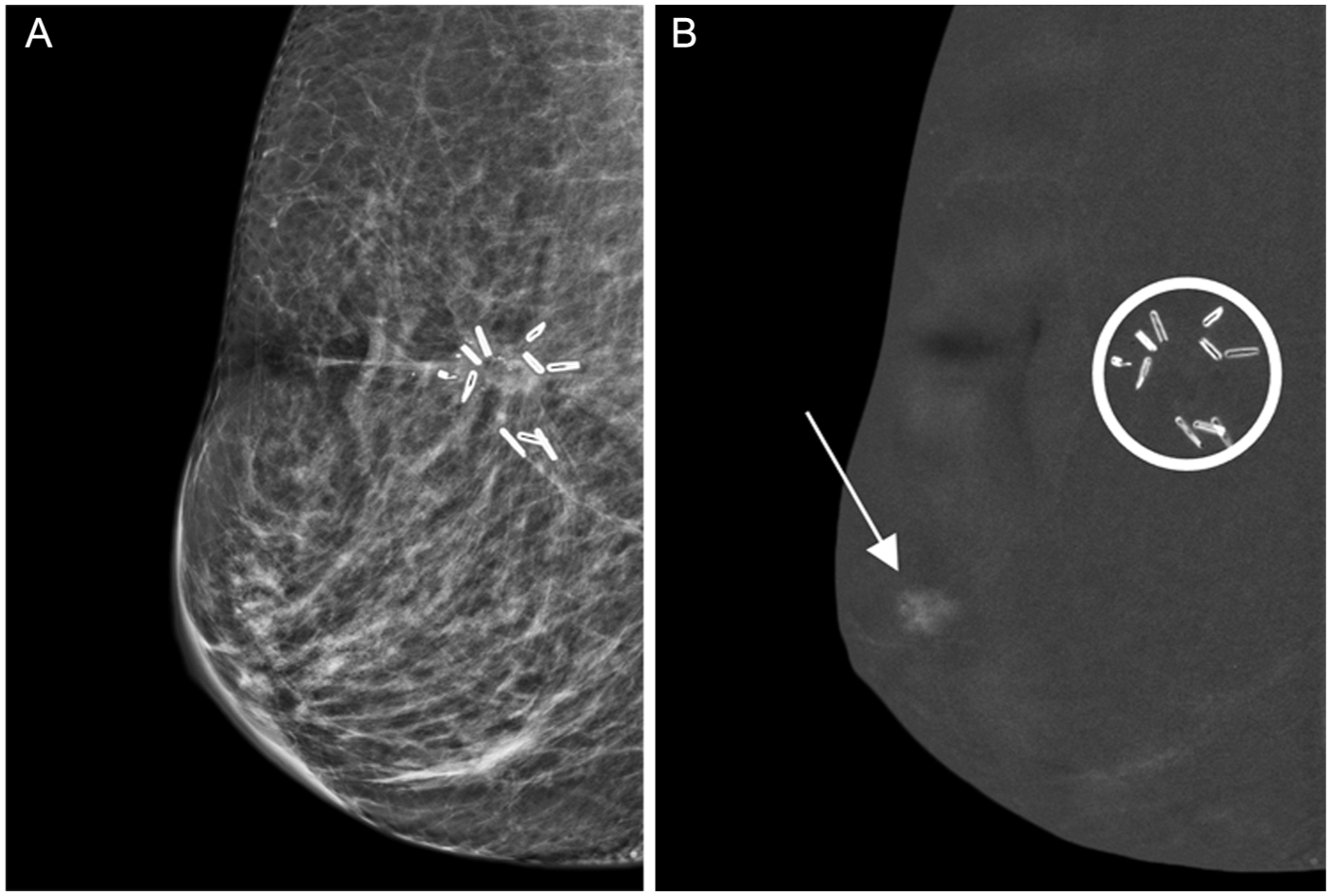
(A, B) ILC on surveillance CEM; inconspicuous on low energy view. 68-year-old patient with a history of right breast invasive lobular carcinoma (ILC), previously treated with breast-conservation surgery. Surveillance contrast-enhanced mammography (CEM) in the right mediolateral oblique (MLO) view demonstrates a 1 cm peri-areolar focal non-mass enhancement (NME) of moderate conspicuity on the recombined image (B—arrow), without a distinct corresponding abnormality on the low-energy image (A). Ultrasound-guided biopsy of a sonographic correlate (not shown) revealed grade 3 ILC, pleomorphic type. Immunohistochemistry showed ER positive, PR negative, and HER2 negative status. Surgical clips from prior breast conservation surgery as well as coil with hook shaped biopsy marker in the anterior surgical bed; shown within the circle (B), with no associated enhancement. The patient subsequently underwent right mastectomy with axillary dissection, with final pathology demonstrating unifocal T1N0 ILC. This surveillance CEM enabled early detection of node-negative recurrence, not visible on low energy images.
The most common mammographic presentation of ILC is a mass with spiculated or indistinct margins, typically iso- or hypodense relative to the surrounding parenchyma, observed in approximately 44% to 65% of cases (Figure 2).14,15 In contrast, round or well-circumscribed masses are uncommon, accounting for only 1% to 3% of ILC lesions. 19 The second most frequent mammographic finding is architectural distortion, present in approximately 10% to 34% of cases. 20 LE images may also be obtained with digital breast tomosynthesis (DBT), which has been shown to improve the detection rate of ILC, increasing it from 0.27 to 0.55 per 1000 screened patients, compared to conventional 2D mammography, primarily by improving the visualization of architectural distortion and margin spiculation, especially in dense breasts (Figure 3).21-23 Consequently, DBT tends to improve lesion detection, size estimation, and the identification of multifocal or bilateral disease relative to 2D mammography. 24
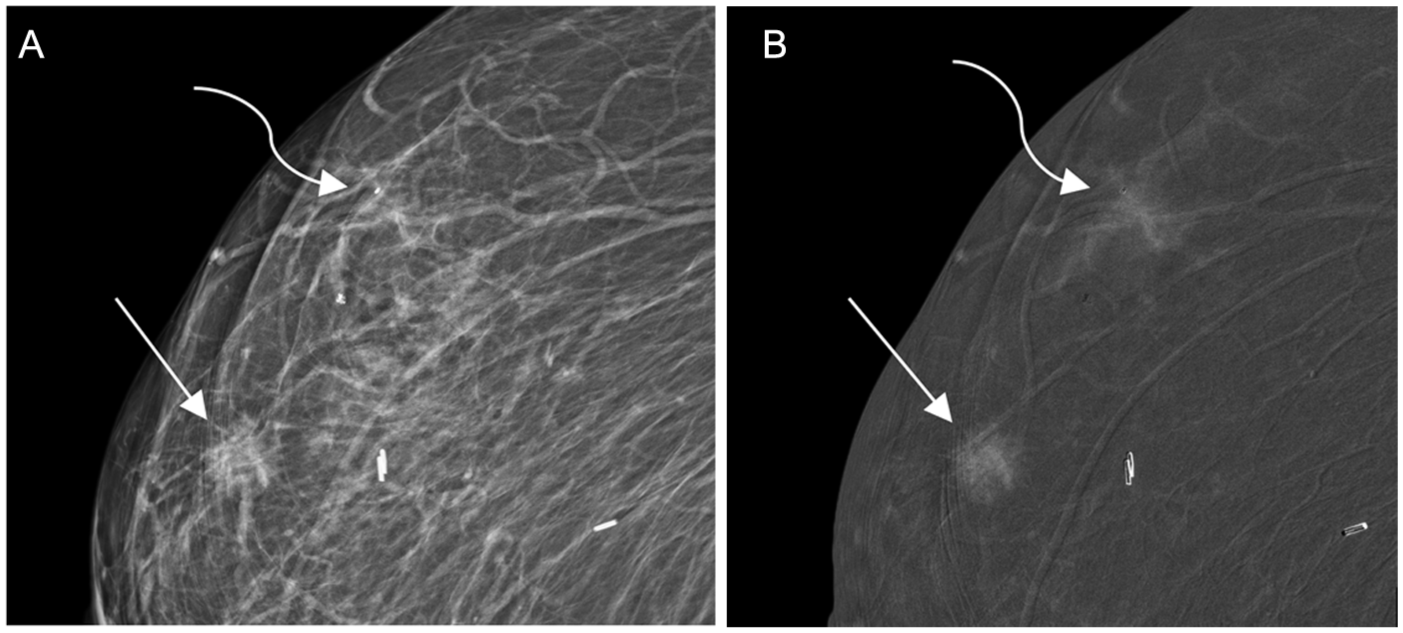
(A, B) Multifocal ILC. 92-year-old patient with a history of right breast invasive ductal carcinoma, previously treated with breast-conservation surgery, presented with 2 new palpable right breast masses. Further evaluation with CEM shows a spiculated mass (straight arrow) and focal asymmetry (curved arrow) on the right mediolateral oblique (MLO) low-energy image (A); both findings were confirmed on craniocaudal (CC) views (not shown). The focal asymmetry lacks central opacity and is difficult to differentiate from the surrounding normal parenchyma. Both lesions demonstrate moderate to high conspicuity enhancement on the corresponding MLO view recombined image (B). Ultrasound-guided biopsy of sonographic correlates (not shown) and subsequent surgical excision revealed 2 foci of grade 2 invasive lobular carcinoma; ER positive, PR negative, and HER2 negative. The patient underwent breast conservation surgery with negative margins. Mastectomy was not pursued due to comorbidities. CEM was instrumental in excluding multicentric and contralateral disease, thereby supporting the decision for limited surgery.
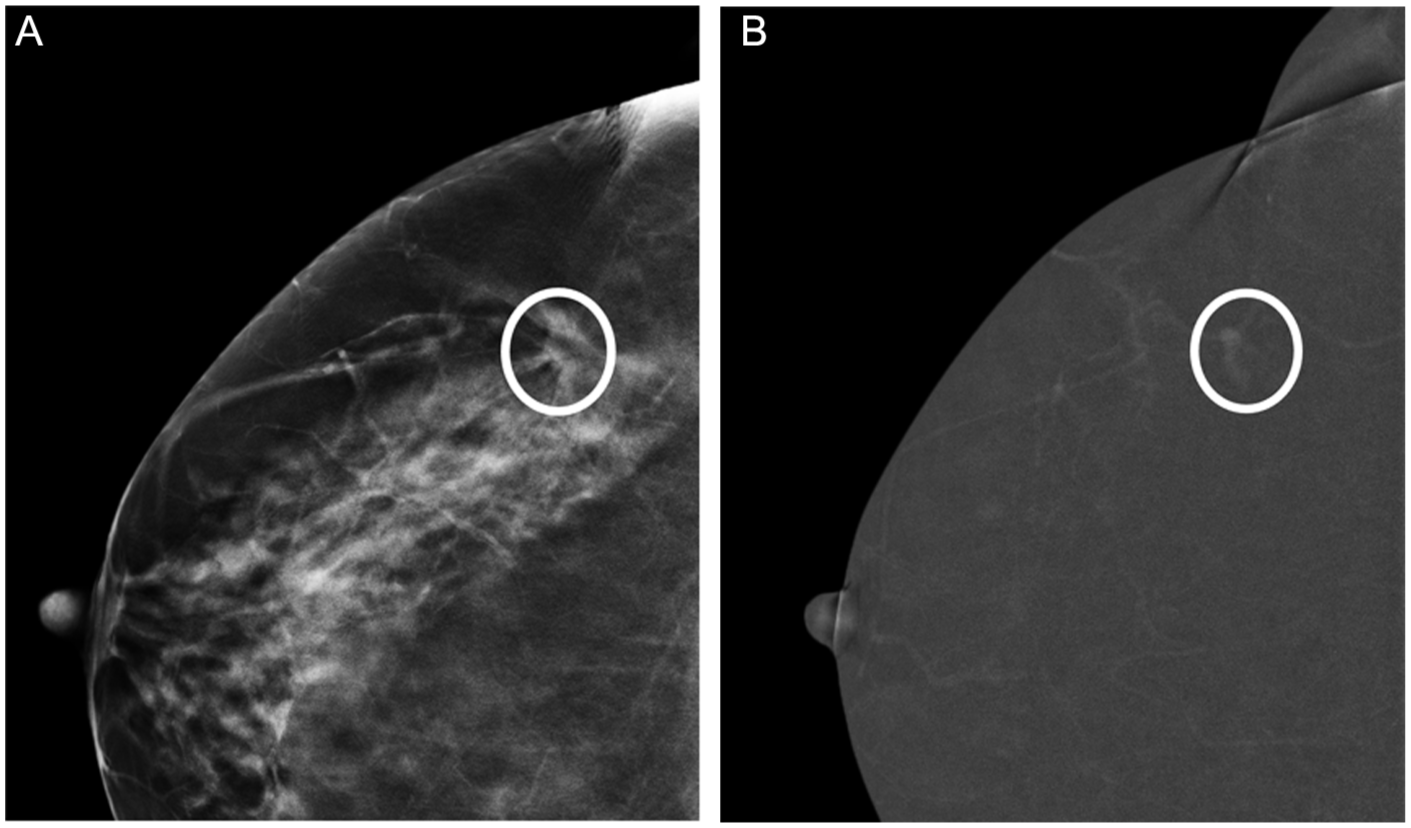
(A, B) ILC on tomosynthesis. 63-year-old patient recalled for workup of a right lateral breast asymmetry, identified on screening mammography (not shown). Right tomosynthesis in the CC view (A) shows an irregular mass with architectural distortion in the posterolateral right breast (circle), which was confirmed on the MLO view (not shown). Associated low to moderate conspicuity enhancement identified on the CC view recombined image (B—circle). Ultrasound guided biopsy of a sonographic correlate (not shown) revealed grade 2 invasive lobular carcinoma; ER positive, PR negative, HER2 negative.
ILC may also present as an asymmetry on mammography. These asymmetries occur in approximately 1% to 14% of cases, classically visible only on the CC projection (Figure 4).14,25 Additionally, ILC may present as focal asymmetries on mammography, which are visible on 2 views but lack the convex outward margins characteristic of a discrete mass.24,26 The presence of microcalcifications in pure ILC is notably rare and is more frequently observed in mixed invasive ductal carcinoma (IDC)/ILC cases.27,28 Another recognized mammographic feature of ILC is the “shrinking breast” phenomenon, in which the affected breast appears asymmetrically smaller over time, attributed to reduced tissue compressibility.29,30 At CEM, this may be accompanied by regional or diffuse non-mass enhancement on recombined images, a finding analogous to the pattern seen on MRI.31,32
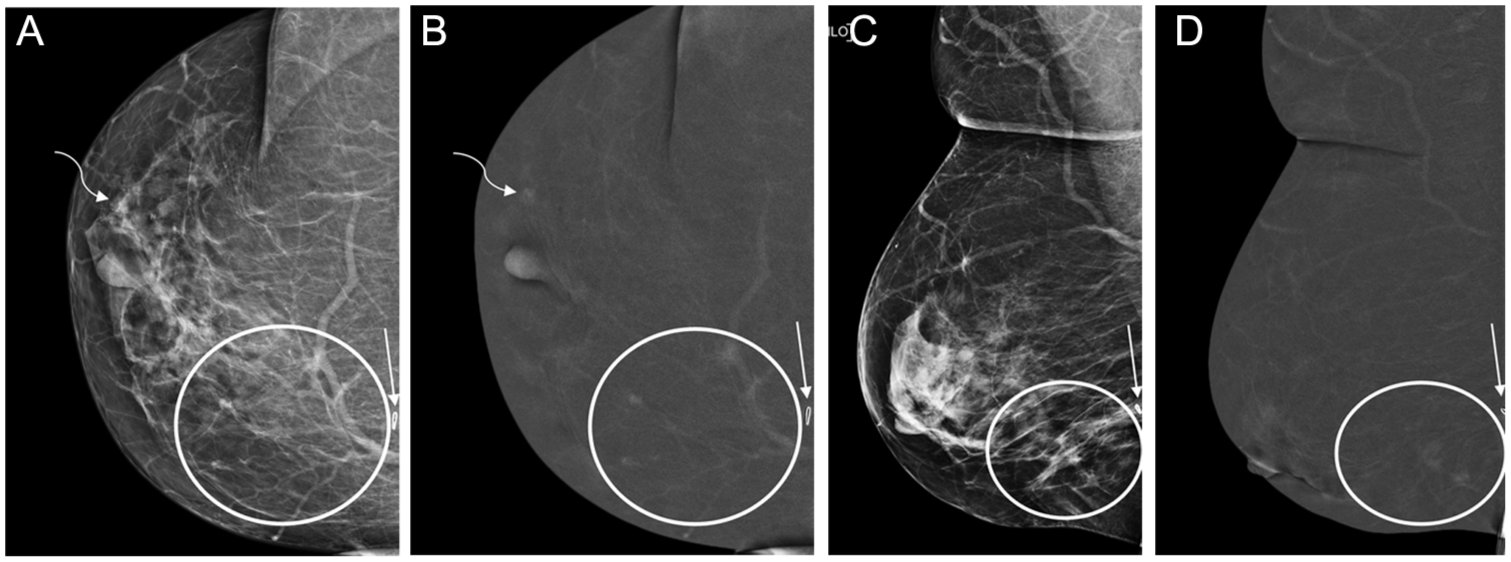
(A-D) Multifocal, multicentric ILC. 76-year-old patient recently diagnosed with grade 2 invasive lobular carcinoma (ILC) of the right breast; ER positive, PR negative, HER2 negative, following ultrasound guided biopsy of a palpable posteromedial mass. Due to its far posterior location, the mass was outside the field of view on CEM, which is a recognized limitation of CEM compared to MRI. Ultrasound (not shown) confirmed the post-biopsy marker (straight arrows) was located at the anterior aspect of the palpable mass. CEM, performed to aid in surgical planning, revealed a low-conspicuity enhancing asymmetry in the right lateral peri-areolar breast, seen on CC low-energy (A) and recombined (B) views (curved arrows). No definite correlate was identified on the MLO low-energy (C) or recombined (D) views. Tomosynthesis-guided biopsy of the right lateral peri-areolar asymmetry confirmed a second site of ILC, consistent with multicentric disease. Additional areas of non-mass enhancement in the lower inner quadrant on recombined views (B and D—circle), with corresponding asymmetries and focal asymmetries on low energy views, are more distinct on CC compared to MLO views (A and C respectively—circle). Surgical pathology following right mastectomy confirmed multifocal and multicentric ILC.
Recombined (RC) Imaging Findings
The lexicon for the RC image component of CEM is adapted from the BI-RADS MRI lexicon, with modifications to address imaging features unique to CEM.33,34 Current literature on the appearance of ILC on RC images is limited; however, it is likely to mirror enhancement patterns observed on MRI. ILC typically manifests as mass or non-mass enhancement (NME; Figure 5), with NME exhibiting focal, linear, segmental, regional, or diffuse distribution patterns.1,35,36 Evaluation of the internal enhancement patterns of NME on RC images—categorized as homogeneous, heterogeneous, or clumped—may not be clearly discernable due to resolution limitations of CEM compared to breast MRI.33,37 Unique to CEM, ILC may also present as an enhancing asymmetry, characterized by abnormal enhancement visible on only a single projection (Figure 6). 38 It is important to consider that differences in the timing of acquisition between the CC and MLO RC views may contribute to the appearance of enhancing asymmetries.
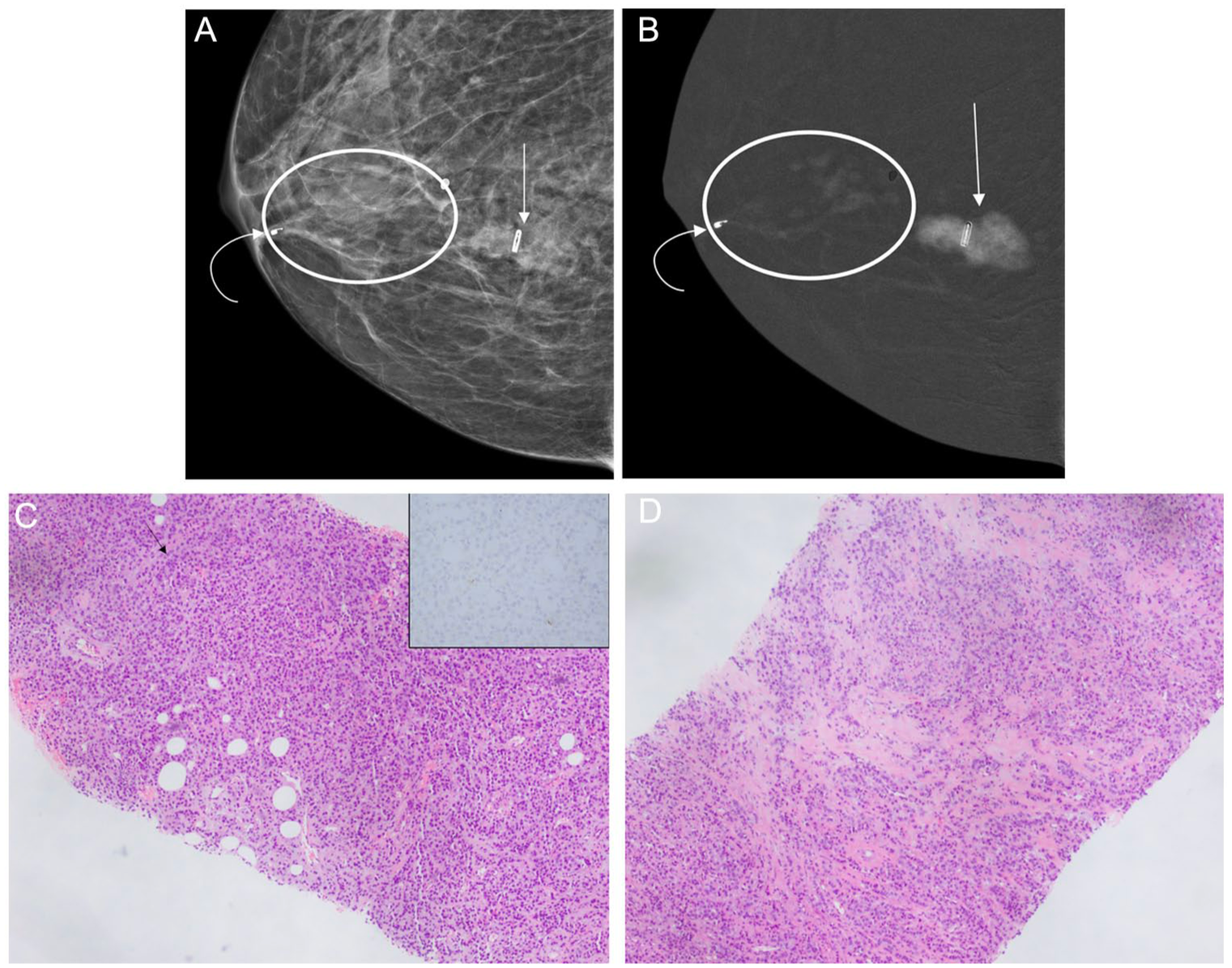
(A-D) Mass and non-mass enhancement of ILC. 76-year-old patient, recently diagnosed with grade 2 invasive lobular carcinoma (ILC) of the right breast; ER negative, PR negative, HER2 negative, following ultrasound guided biopsy of a palpable mass in the posteromedial right breast. CEM in the right CC view demonstrates a corresponding mass with partly circumscribed and partly indistinct margins on the low energy image (A—straight arrow); confirmed on the MLO view (not shown). Associated high-conspicuity enhancement on the recombined image (B—straight arrow). A post-biopsy marker is seen within the mass. (C) Photomicrograph of biopsy specimen of the index palpable mass shows an invasive lobular carcinoma, classic pattern (Nottingham grade 2). The invasive carcinoma shows a single-cell and linear pattern of invasion (black arrow). E-cadherin immunohistochemical stain was negative in the invasive carcinoma (inset), confirming lobular histology. There is little intervening breast tissue, accounting for the mass like appearance on imaging. An additional subcentimeter periareolar mass was identified on right breast ultrasound (not shown), with pathology from ultrasound guided biopsy revealing a second focus of ILC. On CEM, the coil with hook shaped marker from the periareolar breast biopsy (A, B—curved arrow) is located at the anterior aspect of moderate-conspicuity segmental, clumped, non-mass enhancement (B—oval), anterolateral to the index palpable mass. This area was confirmed as extension of ILC on surgical pathology following mastectomy, and had no distinct correlate on 2D mammography (A). (D) Photomicrograph of biopsy specimen in the region of non-mass enhancement, shows invasive lobular carcinoma, classic pattern (Nottingham grade 2). Cells appear more discohesive and infiltrative through the breast tissue, accounting for the non-mass like appearance on imaging.
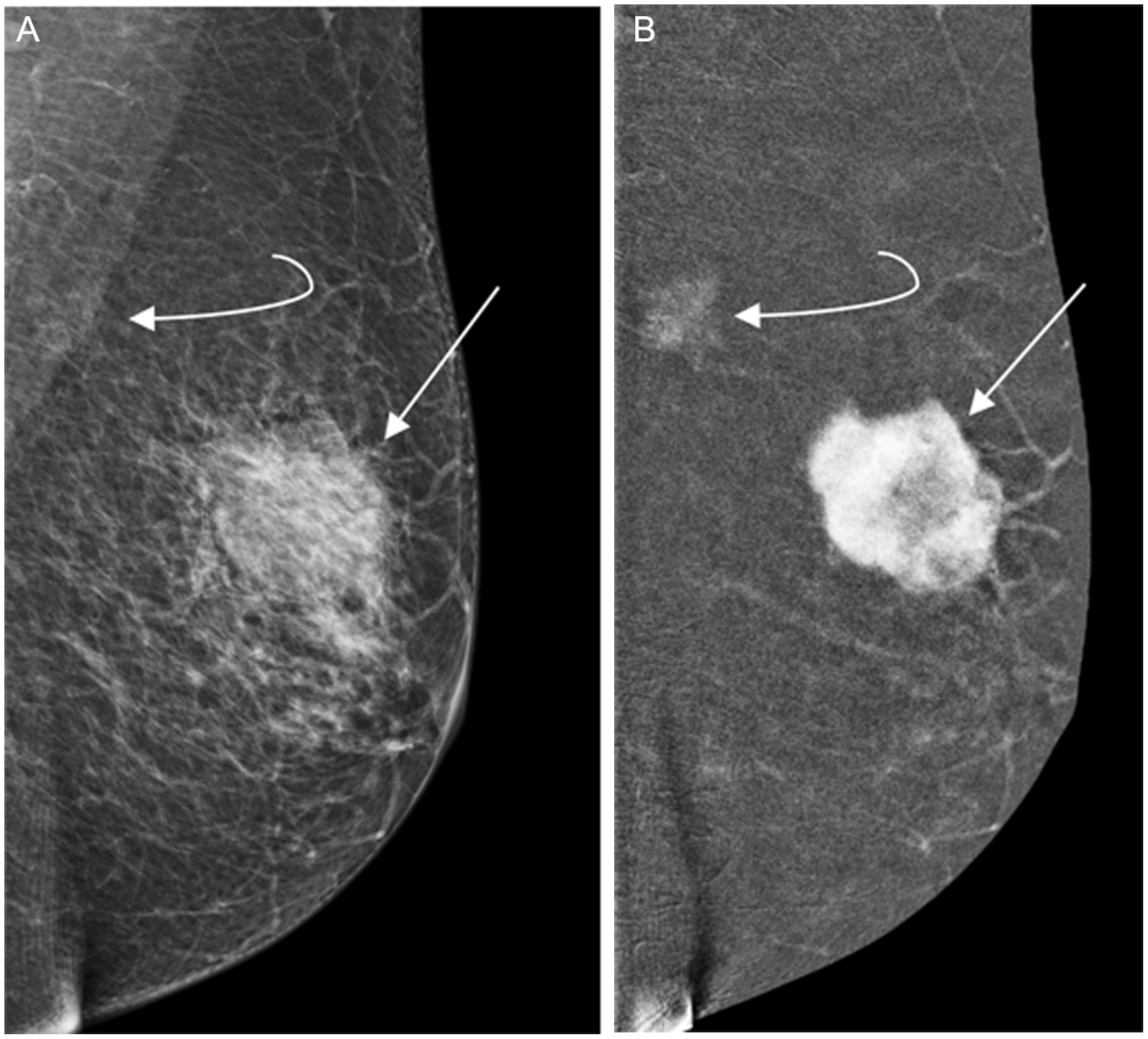
(A, B) Palpable invasive ductal carcinoma (IDC), with CEM detected synchronous ILC. 79-year-old patient presented with a palpable mass in the left breast, which was evaluated with CEM. Corresponding with the palpable abnormality is a 4 cm irregular mass on the MLO view low energy image (A—straight arrow), with associated heterogenous high conspicuity enhancement on the recombined image (B—straight arrow), in the mid-depth of the left supra-areolar breast. Ultrasound-guided biopsy revealed invasive ductal carcinoma (IDC); ER negative, PR negative, HER2 negative. Additionally, on the MLO recombined image (B) there is a 1.8 cm low-conspicuity enhancing asymmetry in the posterior breast, superimposed over the pectoralis muscle (curved arrow). There was no correlate on the CC view (not shown) likely due to the far posterior location; no sonographic correlate was identified. Although there is a correlate on the MLO low energy image (A—curved arrow), this may not have been identified without the associated enhancement, due to low density and overlap with the pectoralis muscle. Tomosynthesis-guided biopsy of this asymmetry revealed invasive lobular carcinoma (ILC); ER positive, PR positive, HER2 negative.
The degree of enhancement of ILC on RC images varies widely, ranging from low to moderate or high conspicuity relative to background parenchymal enhancement (BPE). Lesion enhancement in ILC generally exhibits lower conspicuity compared to that observed in IDC (Figure 6).38-40 Similar to MRI, elevated BPE may reduce the sensitivity of CEM for detecting ILC (Figure 7).41,42
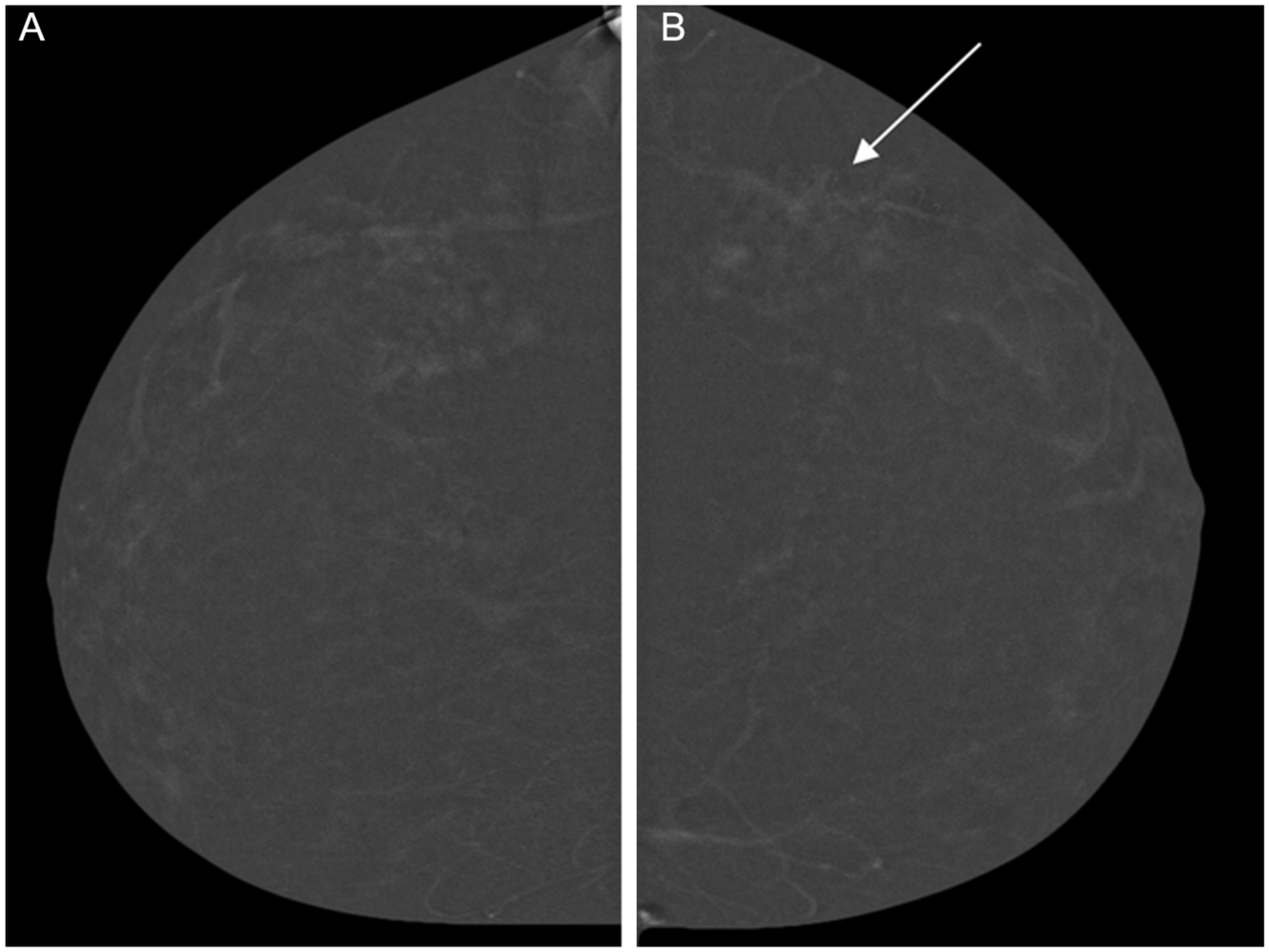
(A, B) ILC on CEM with moderate BPE. 50-year-old patient recalled from screening mammography for workup of an asymmetry identified in the lateral left breast (not shown). Bilateral recombined CEM images in the craniocaudal (CC) projections are shown (A—right, B—left).There is moderate, symmetric background parenchymal enhancement (BPE) bilaterally, limiting the overall sensitivity of the examination. A biopsy marker in the left breast (arrow), corresponds to the location of ultrasound guided biopsy of subtle architectural distortion (not shown), which revealed invasive lobular carcinoma (ILC). Pathology on subsequent surgical excision demonstrated left breast grade 2 ILC; ER positive, PR negative, HER2 negative, with a tumor size of 4.5 cm. Prophylactic contralateral mastectomy showed no evidence of in situ or invasive malignancy in the right breast.
An enhancing finding on RC images may have a correlate on LE views, but this is not invariably the case. When enhancement is observed on RC images without a definite correlate on LE views, CEM potentially plays a crucial role in detection by revealing abnormalities occult on conventional mammography, analogous to MRI.1,40 Furthermore, as LE and RC images are interpreted concurrently, enhancement seen on RC images can draw attention to subtle findings on LE images, which may otherwise be overlooked in the absence of contrast enhancement (Figure 6). The key imaging features of ILC on CEM are summarized in Table 1.
Key Imaging Features of Invasive Lobular Carcinoma on Contrast Enhanced Mammography.
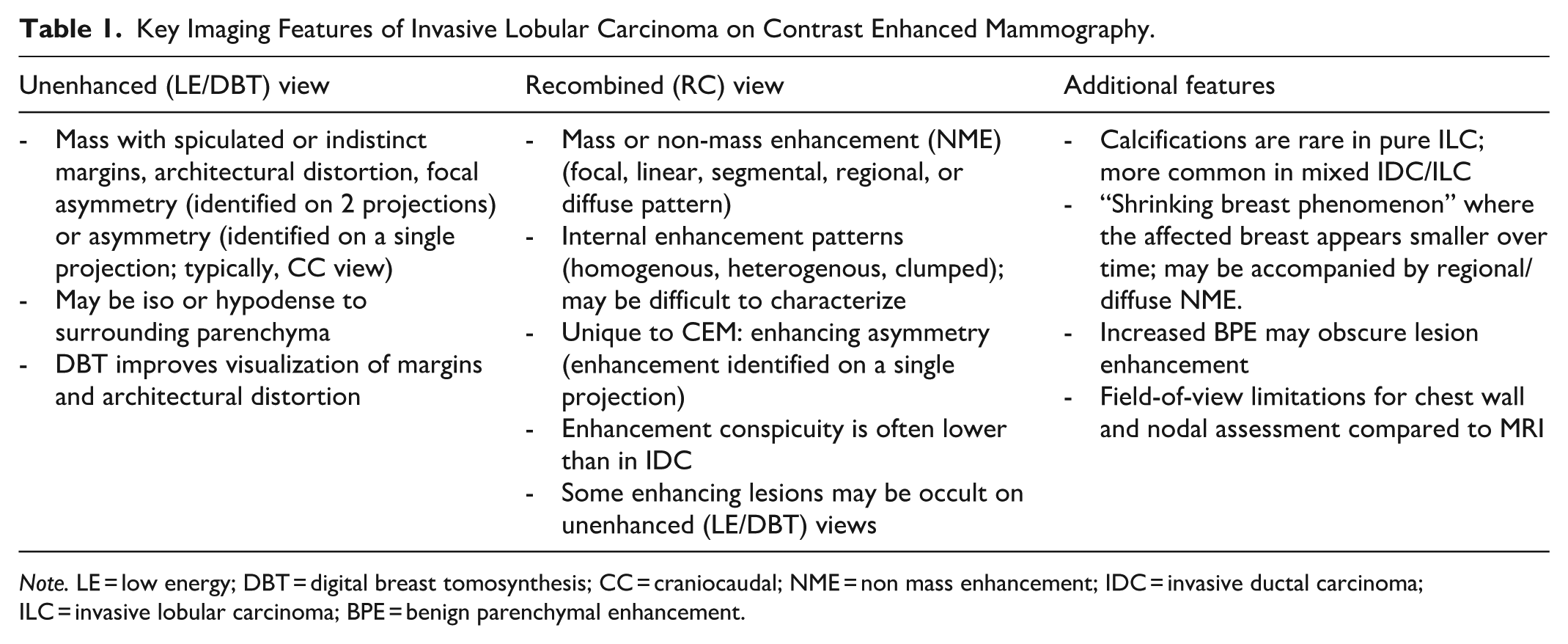
Note. LE = low energy; DBT = digital breast tomosynthesis; CC = craniocaudal; NME = non mass enhancement; IDC = invasive ductal carcinoma; ILC = invasive lobular carcinoma; BPE = benign parenchymal enhancement.
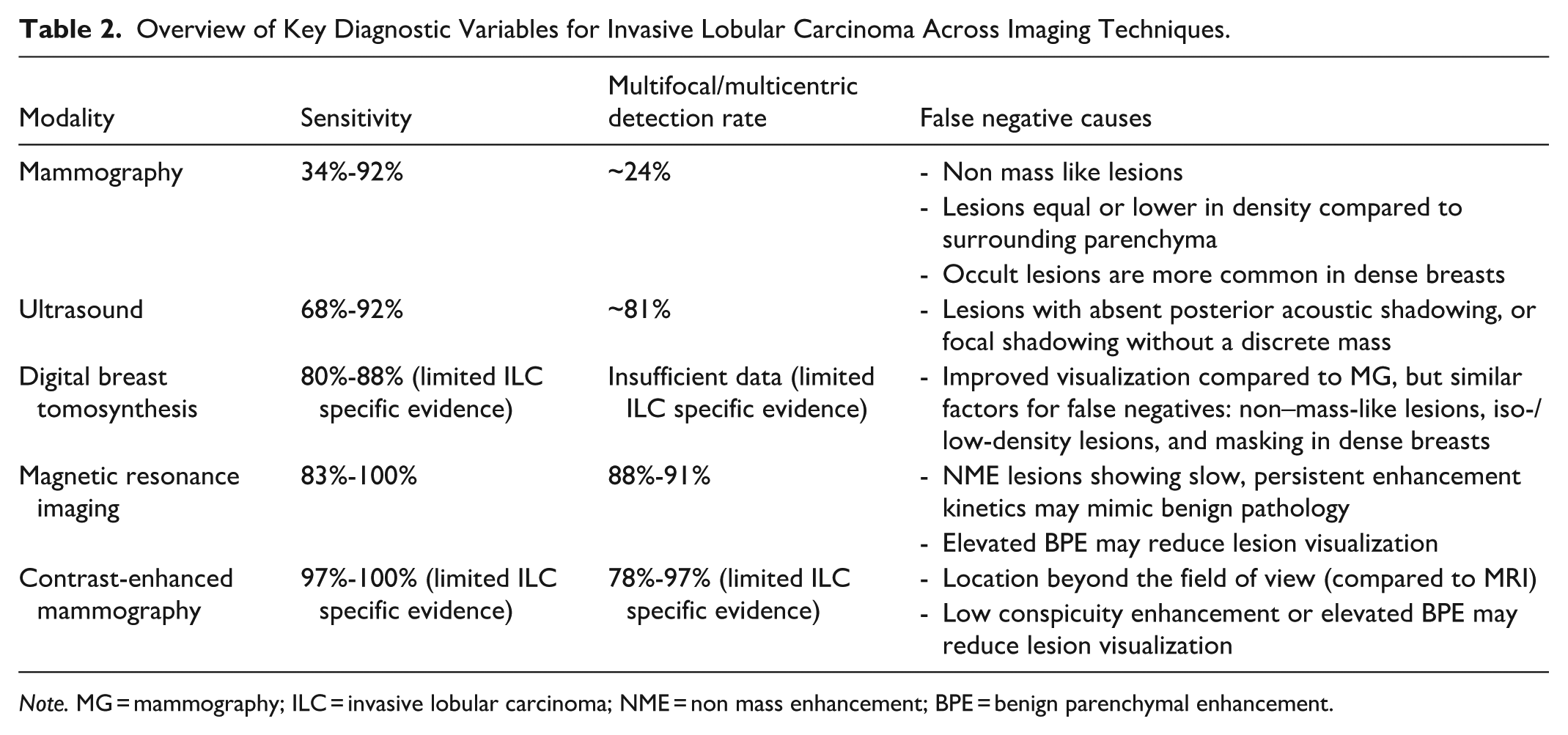
Evaluation of Invasive Lobular Carcinoma Across Imaging Modalities
Mammography remains the most widely available imaging modality for breast cancer detection; however, its sensitivity for ILC is highly variable, ranging from 34% to 92%, with the lower end of the range observed particularly in individuals with dense breast tissue.1,15,24 False negatives may occur, in particular, when ILC presents with subtle imaging features such as architectural distortion, asymmetry, or equal to low density lesions. Additionally, mammography frequently underestimates the true extent of disease, including tumor size and the presence of multifocal or multicentric lesions, with a multifocal/multicentric detection rate of approximately 24%. 1
Ultrasound demonstrates improved sensitivity compared to mammography, typically ranging between 68% and 92% for ILC detection.1,43 It also offers a more accurate estimation of tumor size and better depicts multifocal and multicentric disease, with detection rates of approximately 81%. 1 ILC on ultrasound most commonly presents as an irregular mass with angular margins and posterior acoustic shadowing; however, it may also appear more subtly, for example manifesting as focal shadowing without a discrete mass or lacking posterior shadowing entirely, making detection more challenging. 31
Reported sensitivity for detecting ILC with DBT ranges from 80% to 88%,44,45 though data specific to ILC remain somewhat limited. DBT offers improved tumor size estimation compared to 2D mammography and likely improves detection of multifocal and multicentric disease, although robust ILC specific data are limited.45,46 Despite these advantages, the causes of false-negative or occult disease on DBT remain similar to those seen with conventional mammography, including low-density or non-mass lesions, subtle architectural distortion, and lesion obscuration in dense parenchyma. 31
MRI is widely regarded as the most sensitive modality for detecting ILC, with reported sensitivities ranging from 83% to 100%.1,43,47 It is particularly effective in identifying multifocal and multicentric disease, with detection rates between 88% and 91%, and is superior to mammography and ultrasound for tumor size estimation.1,48 False negatives may occur when ILC manifests as non-mass enhancement (NME) with slow, progressive contrast kinetics that can mimic benign entities such as fibrocystic change. Additionally, elevated BPE may obscure lesion visibility and reduce diagnostic confidence, particularly in premenopausal patients or those on hormonal therapy. 49
CEM has demonstrated promising sensitivity for detecting ILC, with reported values ranging from 97% to 100%.1,38 Although ILC-specific data remain limited, current evidence suggests CEM performs well in estimating tumor size and identifying multifocal and multicentric disease, with detection rates between 78% and 97%.1,9 However, like other modalities, CEM has diagnostic limitations. False-negative findings may occur, particularly in ILC or other malignancies such as mucinous carcinoma, necrotic tumors, and Paget’s disease. These may exhibit absent or low conspicuity enhancement due to their histopathologic characteristics.37,50 The field of view of CEM is that of conventional mammography and DBT, with the same limitations in comparison to MRI. These include the breast periphery, axilla, and chest wall. Similar to MRI, elevated BPE can obscure lesion detection. 49 While these limitations must be considered, CEM remains a diagnostically comparable, and potentially more accessible and cost-effective alternative to MRI in the evaluation of ILC. These implications are discussed further in the next section. Table 2 summarizes key diagnostic variables for ILC across imaging techniques.
Overview of Key Diagnostic Variables for Invasive Lobular Carcinoma Across Imaging Techniques.
Note. MG = mammography; ILC = invasive lobular carcinoma; NME = non mass enhancement; BPE = benign parenchymal enhancement.
Clinical Implications
CEM is emerging as a valuable imaging modality in the management of ILC, with meaningful implications for surgical planning, patient outcomes, and healthcare accessibility.
Assessment of Disease Extent and Impact on Surgical Planning
Accurate preoperative assessment of tumor extent is critical in ILC, which has a propensity for multifocal, multicentric, and bilateral disease (Figure 4).1,51,52 Conventional imaging frequently underestimates disease burden, contributing to incomplete excision, positive margins, and increased re-excision rates.53,54
Preoperative MRI is often recommended to identify additional tumor sites in ILC, thereby facilitating accurate staging and optimal treatment planning. Use of preoperative breast MRI has been shown to reduce the risk of positive surgical margins and decrease the need for completion mastectomy. CEM has demonstrated superior performance compared to conventional mammography in delineating disease extent in biopsy-proven ILC, with diagnostic accuracy comparable to MRI.55,56 Although MRI demonstrated slightly higher sensitivity for detecting multifocal and contralateral disease, these differences were not statistically significant. 55 CEM showed higher specificity and diagnostic odds ratios compared to MRI, in some settings, supporting its reliability and suggesting a lower false-positive rate. 55 This may reduce overtreatment and unnecessary extensive surgery, without compromising diagnostic accuracy. 55
Both CEM and MRI outperform mammography and ultrasound in estimating ILC tumor size relative to histopathologic measurements. In a multicenter retrospective study, Lobbes et al reported that CEM overestimated tumor size by a mean of 2.1 mm and MRI by 1.5 mm compared with surgical pathology. 57 However, tumor size estimation by both CEM and MRI becomes less reliable for tumors larger than 3 cm or those presenting as non-mass enhancement, where variability in size assessment increases.57,58 Additionally, ILC frequently coexists with lobular carcinoma in situ (LCIS) which may exhibit enhancement, particularly in pleomorphic and florid LCIS subtypes. 59 This may confound invasive tumor size assessment, but still contributes to surgical planning (Figure 8).
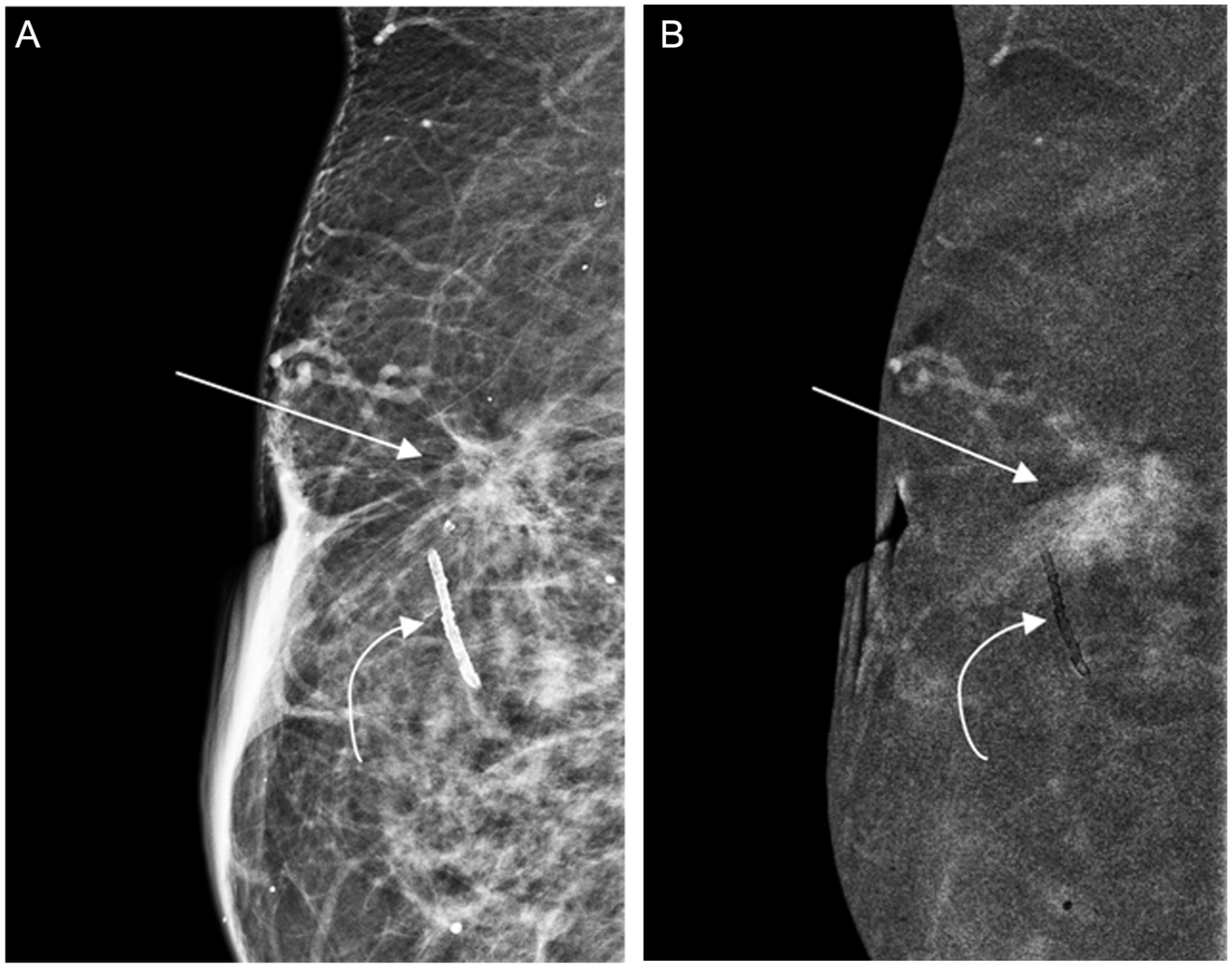
(A, B) ILC with LCIS. 76-year-old patient with a history of bilateral ductal carcinoma in situ (DCIS) treated with breast-conservation surgery 5 years earlier, presented for surveillance CEM. MLO view recombined image (B) demonstrates 3 cm moderate-conspicuity non-mass enhancement in the posteriorsuperior right breast (straight arrow), corresponding to postsurgical architectural distortion on the low energy MLO image (A—arrow). Magnification views (not shown) revealed an associated group of microcalcifications, which included fine and amorphous forms. Tomosynthesis-guided biopsy targeting the calcifications and architectural distortion revealed florid and pleomorphic lobular carcinoma in situ (LCIS). Pathology on subsequent surgical excision demonstrated grade 2 invasive lobular carcinoma (ILC) measuring 0.4 cm; ER positive, PR positive, and HER2 negative, in a background of florid grade 2 LCIS involving an estimated area of 2.0 cm. The extent of enhancement on recombined images exceeded the size of the invasive focus, reflecting the combined extent of the invasive and in situ components. Incidental vascular calcification noted (curved arrow). The contribution of CEM in this case was prompting workup and biopsy based on moderate-conspicuity enhancement on RC images, in a region of otherwise stable postsurgical change on LE images, leading to the diagnosis of ILC with LCIS.
While neoadjuvant chemotherapy and endocrine therapy have limited use in ILC, a small subgroup of patients receive these treatments to enable breast-conserving surgery. MRI and CEM offer comparable accuracy in assessing residual tumor size and extent after therapy, although ILC-specific data are limited for CEM. 1
CEM’s reasonable correlation with histopathology in estimating tumor size, combined with its effectiveness in detecting multifocal or contralateral disease, supports its utility in surgical decision-making. While CEM remains limited in evaluating chest wall invasion and regional lymph node involvement, including axillary and internal mammary chains, 60 it offers a viable alternative for patients who cannot undergo MRI, and may help streamline surgical planning in resource-limited or high-volume settings. 55
Impact on Patient Outcomes
Although long-term outcome data specific to CEM influencing ILC outcomes are still limited, its diagnostic performance indicates a promising role in optimizing treatment pathways. By improving detection of subtle features characteristic of ILC, CEM facilitates earlier and more accurate diagnosis compared to conventional imaging such as mammography and ultrasound. Improved delineation of disease extent enables more precise surgical planning, which may reduce rates of positive margins, re-excisions, and conversions from breast-conserving surgery to mastectomy. These improvements may contribute to lower patient morbidity, fewer delays in adjuvant therapy, improved cosmetic outcomes, and more streamlined, personalized care.55,61
Accessibility and Cost-Effectiveness
While MRI remains the reference standard for ILC management, its higher cost, longer acquisition time, and contraindications in certain patients—such as those with claustrophobia or metallic implants—can limit its utility. CEM offers several practical advantages—it is faster, less costly, and can be performed on existing digital mammography systems with minimal infrastructure changes.62,63 This makes it more easily integrated into routine breast imaging workflows, expanding access to functional imaging for a broader patient population. CEM, however, involves exposure to ionizing radiation and iodinated contrast agents, which may be contraindicated or less suitable for certain patients.62,63 Despite these limitations, the potential broader availability and scalability of CEM could support more inclusive research and equitable treatment planning for patients with ILC, particularly in resource-limited and rural settings where MRI access is limited. These attributes position CEM as a practical alternative to MRI, capable of reducing diagnostic disparities and improving care delivery across diverse healthcare environments.
Future Imaging Directions
While CEM and MRI are currently the most established functional imaging modalities for ILC evaluation, novel techniques are under investigation to address its unique diagnostic challenges. Molecular imaging, in particular, hold potential for enhancing sensitivity, lesion characterization, and informing treatment strategies.
Positron Emission Tomography (PET)
The effectiveness of 18F-Fluorodeoxyglucose (FDG)-PET/CT in ILC is limited due to typically low FDG uptake, especially in subtypes like Luminal A. This often hinders detection of both primary tumors and metastases, prompting interest in alternative PET tracers. While FDG-PET detects hypermetabolic activity by reflecting increased glucose uptake, 18F-Fluoroestradiol (FES) specifically targets estrogen receptors.1,44,64 As approximately 95% of ILCs are ER-positive, this tracer has shown better lesion detection than FDG-PET in ILC, and may assist in assessing receptor heterogeneity and guiding endocrine therapy. 64 Another tracer, 68Ga-Fibroblast activation protein inhibitor (FAPI), targets fibroblast activation protein in the tumor microenvironment, and shows higher uptake and tumor-to-background ratios in ILC compared to FDG. While early data suggest that these tracers may improve imaging accuracy, larger prospective studies are needed to establish the clinical utility of these emerging tracers in ILC imaging.1,44,64,65
Positron Emission Mammography (PEM)
Positron emission mammography (PEM) is a dedicated breast nuclear imaging technique that offers higher spatial resolution than whole-body PET/CT. PEM has been proposed as an alternative to MRI for evaluating disease extent in newly diagnosed breast cancer patients, and has demonstrated higher sensitivity and diagnostic accuracy than whole-body PET/CT for tumor detection. 1 However, its use is limited by the need for specialized equipment, restricting widespread availability.
Studies specifically assessing PEM in ILC are limited. However, a small retrospective study by Hussein et al found that adding PEM to mammography and ultrasound increased the sensitivity for ILC detection from 66% to 90.6% and specificity from 72.7% to 81.8%. 66 While these findings are promising, larger prospective studies are necessary to validate PEM’s role in imaging this histologic subtype and to clarify its clinical utility in treatment planning.
Artificial Intelligence and Radiomics
Artificial intelligence (AI) is playing an increasingly prominent role in breast imaging, with applications in lesion detection, risk prediction, and diagnostic workflow optimization. In mammography, DBT, and ultrasound, AI algorithms have improved diagnostic accuracy by identifying subtle imaging features that are particularly difficult to detect in ILC. 44 AI models can also integrate clinical risk factors, such as genetic mutations, with imaging and molecular data to generate detailed risk profiles, potentially improving predictions of disease progression and treatment response. 67
Radiomics extracts quantitative data from standard imaging studies, providing insight into tumor biology to enhance non-invasive tumor profiling and risk stratification, and thereby enabling more personalized treatment planning.67,68 Notably, radiomic analysis of CEM images has demonstrated potential in predicting molecular subtypes prior to biopsy. 69 When combined with AI, radiomics may support the development of integrated prognostic models that improve personalized risk assessment and long-term decision-making in ILC. This approach may in turn facilitate individualized treatment planning and disease monitoring, with the potential to improve outcomes for affected individuals. While early findings are encouraging, further prospective studies are needed to validate their clinical utility in routine practice.
Conclusion
ILC poses distinct diagnostic challenges due to its infiltrative single-file growth pattern and subtle imaging features. Conventional mammography and ultrasound frequently underestimate both its presence and extent, underscoring the need for functional imaging. CEM improves lesion conspicuity and demonstrates sensitivity comparable to MRI in detecting multifocal and multicentric disease, while also providing reliable tumor size estimation, particularly in lesions smaller than 3 cm. Although MRI remains the gold standard—especially for assessing chest wall invasion and lymph node involvement—CEM offers a more accessible and cost-effective alternative for preoperative evaluation. Radiologist familiarity with the variable imaging appearances of ILC on CEM promises to facilitate more accurate interpretation. As clinical experience with CEM grows, and more data specific to ILC become available, its role in staging and surgical planning is likely to expand, with the potential to positively impact patient outcomes. Ongoing advances in artificial intelligence, radiomics, and molecular imaging may complement CEM, potentially improving lesion characterization and supporting more individualized management of ILC.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
