Abstract
Objectives:
The purpose of this article is to provide a detailed and updated review of the physics, techniques, indications, limitations, reporting, implementation and management of contrast enhanced mammography.
Background:
Contrast enhanced mammography (CEM), is an emerging iodine-based modified dual energy mammography technique. In addition to having the same advantages as standard full-field digital mammography (FFDM), CEM provides information regarding tumor enhancement, relying on tumor angiogenesis, similar to dynamic contrast enhanced magnetic resonance imaging (DCE-MRI). This article reviews current literature on CEM and highlights considerations that are critical to the successful use of this modality.
Conclusion:
Multiple studies point to the advantage of using CEM in the diagnostic setting of breast imaging, which approaches that of DCE-MRI.
Introduction
While screening mammography has been shown to reduce mortality from breast cancer, its sensitivity can often be limited. 1 Dynamic contrast enhanced magnetic resonance imaging (DCE-MRI) highlights tumors based on angiogenesis (i.e. the rapid formation of permeable blood vessels around the tumor) and is known to be the most sensitive modality in detecting breast cancer. 2,3
The introduction of full-field digital mammography (FFDM) enabled the use of contrast enhanced mammography (CEM), an iodine-based modified 2-dimensional (2D) digital mammography (DM) exam. CEM, also known as contrast enhanced spectral mammography (CESM) or contrast enhanced digital mammography (CEDM), utilizes dual energy technique which also relies on tumor angiogenesis. 4 -6
Physics
A straightforward approach for using contrast enhanced imaging is with temporal subtraction, where images are acquired before and after intravenous (IV) administration of the contrast agent. 7 However, because reduced breast compression between the two acquisitions is required to ensure adequate contrast medium delivery, this may result in motion artifacts. 8(p5),9
As an alternative, dual-energy subtraction is now employed. Paired low-energy (LE at 23-32 kVp) and high-energy (HE at 45-49 kVp) images are obtained 4,8(p6),9 -12 after the injection. Since the low-energy views are below the K-shell absorption edge (33.2 keV), they are equivalent to FFDM. 4,8(p6),9 -14 With increasing energy, iodine displays an abrupt increase in the photoelectric absorption of x-ray at 33.2 keV, as opposed to breast tissue. 4,7,8(p6) Recombined views are formed by the subtraction of the low-energy images from the high-energy images, and highlight areas of iodine uptake only 8(p6),9-10 (Figure 1A-C).
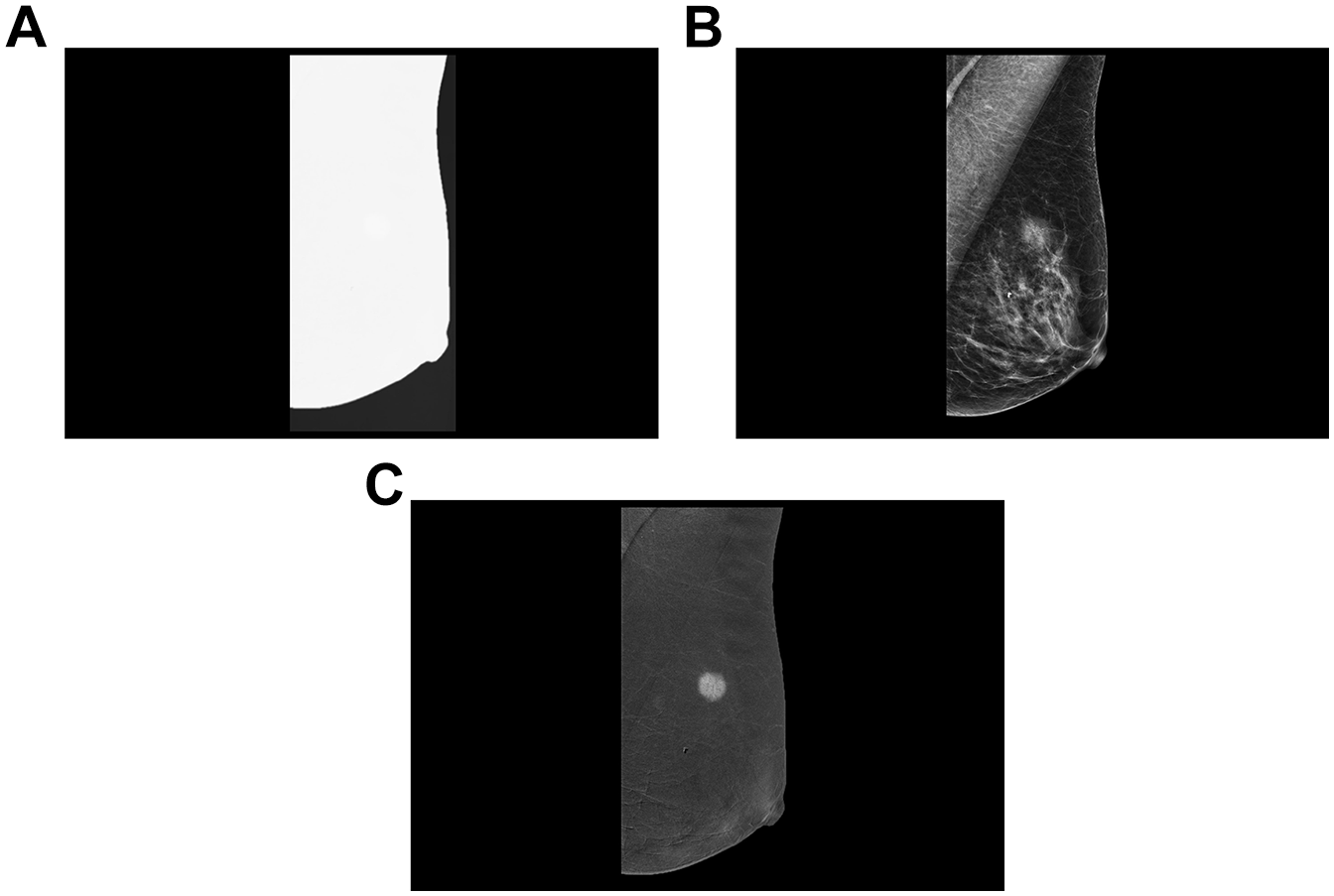
From left to right: Medioaleral oblique projection of the left breast showing non-informative high-energy image (A), low-energy image (B) equivalent to standard mammogram and recombined image (C), which shows iodine uptake of an invasive breast malignancy.
The increase in radiation dose of CEM compared to a FFDM ranges from 106% to 180% with one study reported it to be significantly lower than digital breast tomosynthesis (DBT). 12,15 -17 This reported variability is likely a function of equipment age and vendor variability.
Technique
The contrast enhanced mammography technique begins by using a power injector to perform an IV injection of non-ionic low-osmolar iodinated contrast material with concentration of 300-350 mg/mL at a standard dose of 1.5 mL/kg and a rate of 3 mL/s, before any compression is applied. 9 -12,18,19 Two minutes after the start of the injection, the breast is placed into compression and paired low-energy and high-energy images are obtained for each craniocaudal (CC) and mediolateral oblique (MLO) views. 9 -12,18,19 The image acquisition recommended to be completed within 7 minutes. 9 -12 While some publications state that the order of image acquisition does not affect image quality, it is generally accepted that if there is a side of concern, it should be included in the first or second image and at least one view of the contralateral breast be included within the first 3 images. 11,12,18 If required, additional views can be obtained after 7 minutes (Figure 2). From the author’s experience, some cancers may show enhancement even later than 20 minutes after injection and therefore, additional views may be obtained even after reviewing the images. If the imaging acquisition cannot start at 2 minutes, the study may continue regardless (Figure 3A, 3B). In such a case, or if it is expected that a lesion will not be seen on standard mammography views, one may want to alter the order of imaging acquisition to allow better visualization of the lesion. Reinjection of contrast should be avoided since the drug manufacturer recommends a maximum contrast volume of 120-150 ml.
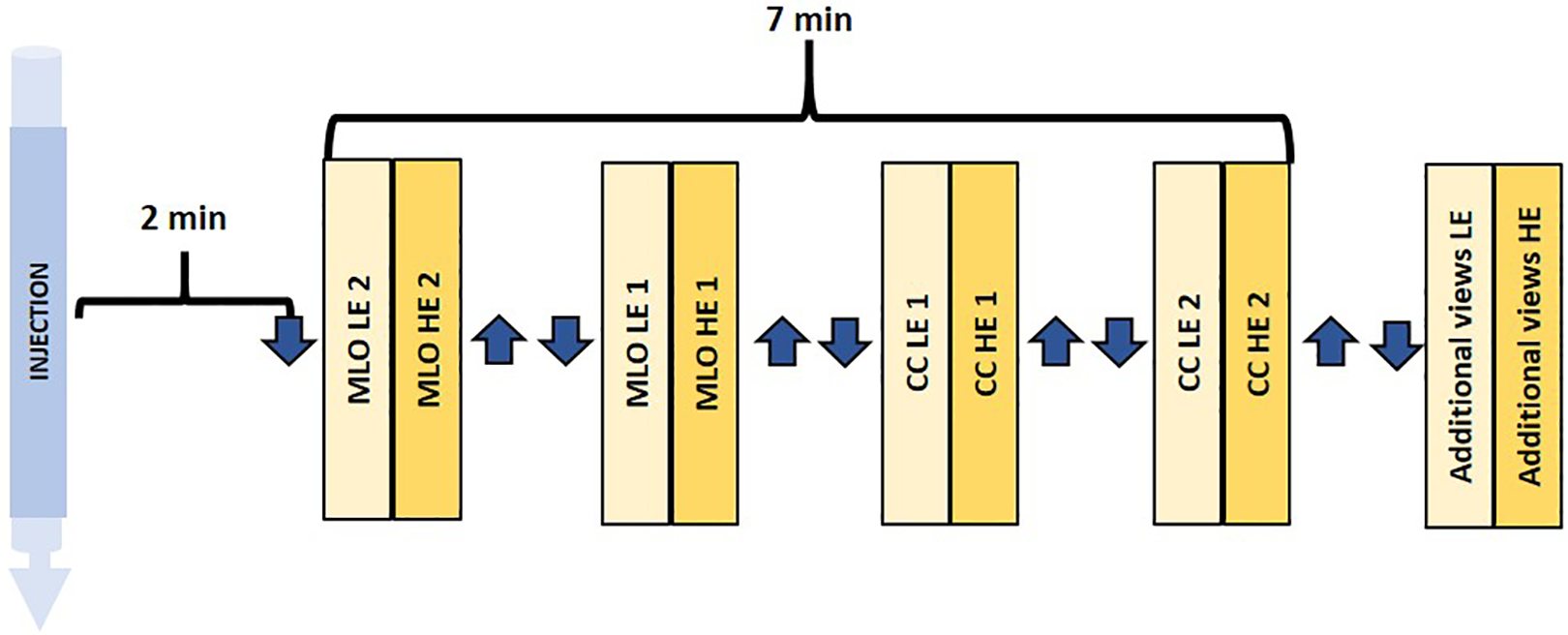
Schematic workflow of contact-enhanced mammography exam. Abbreviations: craniocaudal = CC, mediolateral oblique =MLO, HE = high-energy, LE = low-energy. Symbols: 1 = breast of interest, 2 = contralateral breast, arrow down = compression, arrow up = release compression. * Protocol used by the authors. Note that some authors start with the breast of interest and some start with CC view.
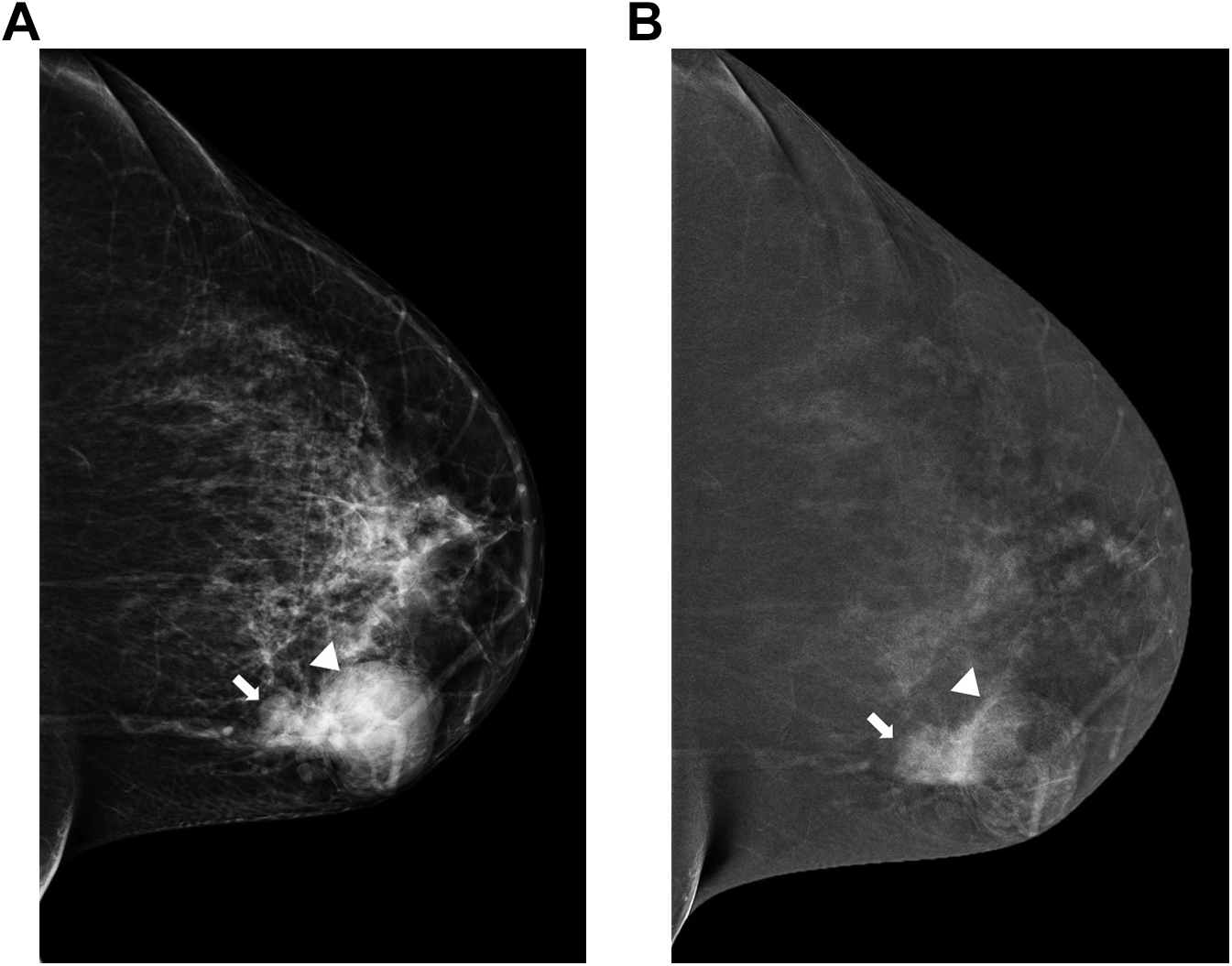
Craniocaudal projections of the right breast in a 46-year-old female who presented for left breast lump. The patient developed an allergic reaction to contrast iodine and therefore images acquisition started only 16 minutes after intravenous contrast venous injection. Lobulated mass confined by well circumscribed margin seen on low-energy image (A). Recombined image (B) still shows marked degree of enhancement within the smaller component of the lesion (B) whereas milder enhancement seen within the larger component (arrow head), likely due to washout. The lesion was biopsied under ultrasound guidance (not shown), proved to be high grade invasive mammary carcinoma.
Image Interpretation and Reporting
Vendor guidelines recommend that the images be reviewed on a mammography approved workstation or Picture Archiving and Communication System (PACS).
According to one reader study that compared the LE images to the complete CEM examination among radiologists and residents, CEM required only a small learning curve. 20 Nonetheless, performing 1-year follow-up examinations may be considered until gaining more experience. 18
While a previous review recommended that the LE images should be interpreted first, 12 a recent study reported that reviewing the recombined images first has similar diagnostic performance, while reading time is one-third faster. 21 Regardless of the order of image review, the interpretation of CEM exams must rely on reviewing both the LE views, which provide morphological information similar to conventional mammography, and the recombined images, which provide information about the vascularization of the lesion. 8(p12),9 -12,18 Currently, there is no dedicated Breast Imaging Reporting and Data System (BI-RADS) lexicon for CEM, but the mammographic and DCE-MRI descriptors of the BI-RADS lexicon can be easily adopted for use. 8(p12),12,22 -25
The reporting of the LE images is identical to standard mammography, starting with a description of the breast density. Similar to DCE-MRI, the recombined views report first describes the background parenchymal enhancement (BPE), defined by the degree of enhancement (i.e., minimal, mild, moderate and marked, or <25% of the breast, 25-50%, 50-75% and over 75%, respectively); (Figure 4A-D). In cases of moderate or marked BPE, enhancing lesions may be missed. 8(p15),12,23 The degree of BPE present on CEM appears to be minimally affected by timing during the menstrual cycle, contrasting that on DCE-MRI. 12,19
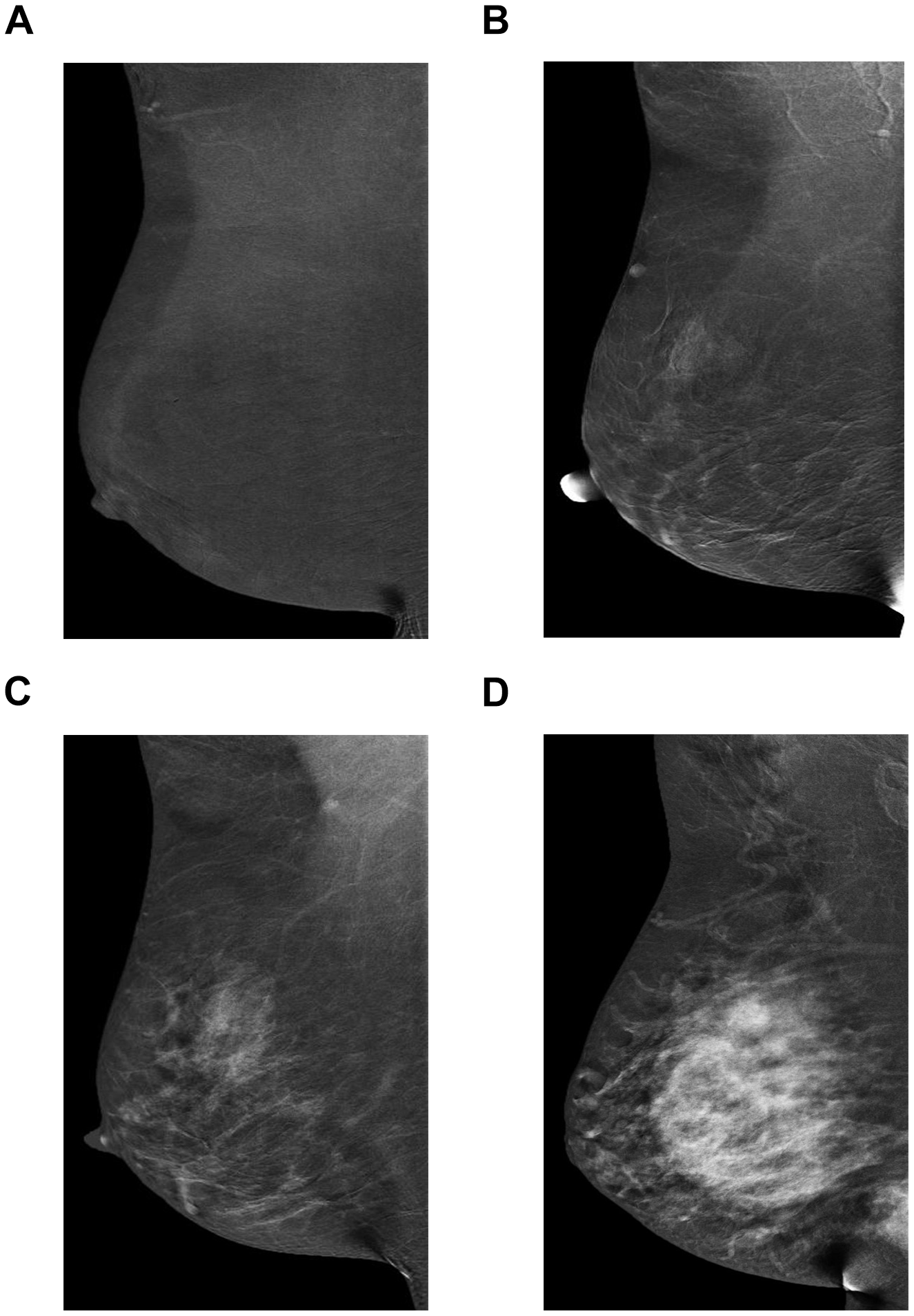
Mediolateral oblique projections of the right breast of 4 different women from left to right show minimal (A), mild (B), moderate (C), and marked (D) degree of background parenchymal enhancement.
Like DCE-MRI, enhancing lesions detected by CEM may be categorized as a focus, mass, or non-mass enhancement.(8(p15),23,25 It should be recognized that both malignant and benign lesions can present with or without contrast enhancement 26,27 and that malignancy can present as an area of asymmetric enhancement with no correlation abnormality on the LE views. 9 -12,24 One must avoid dismissing a non-enhancing suspicious lesion seen on LE images as benign; like DCE-MRI, some cancers may present little or no enhancement on CEM. 8(p15),9 -12,24 -27 Furthermore, enhancing lesion on recombined images can lead to a retrospective detection of an abnormal area on the LE images. 8(p15),12
Masses
Masses can present with or without enhancement on contrast enhanced mammography. 8(p15),12 Enhancing masses should be evaluated further with ultrasound (US) for better characterization and possible targeting for biopsy. 12 While non-enhancing masses most likely represent cysts or fibroadenomas (Figure 5), 8(p15,16),12 the LE images must be carefully reviewed to exclude suspicious characteristics. A mass with rim enhancement might be associated with “eclipse sign” which refers to a thin, well demarcated rim of enhancement and no central enhancement, compatible with a simple cyst. 8(p17),12 However, if the enhancing wall is thick or nodular, US must be performed to exclude the possibility of a malignant complex cystic or necrotic mass 8(p17),12 (Figure 6A, B).
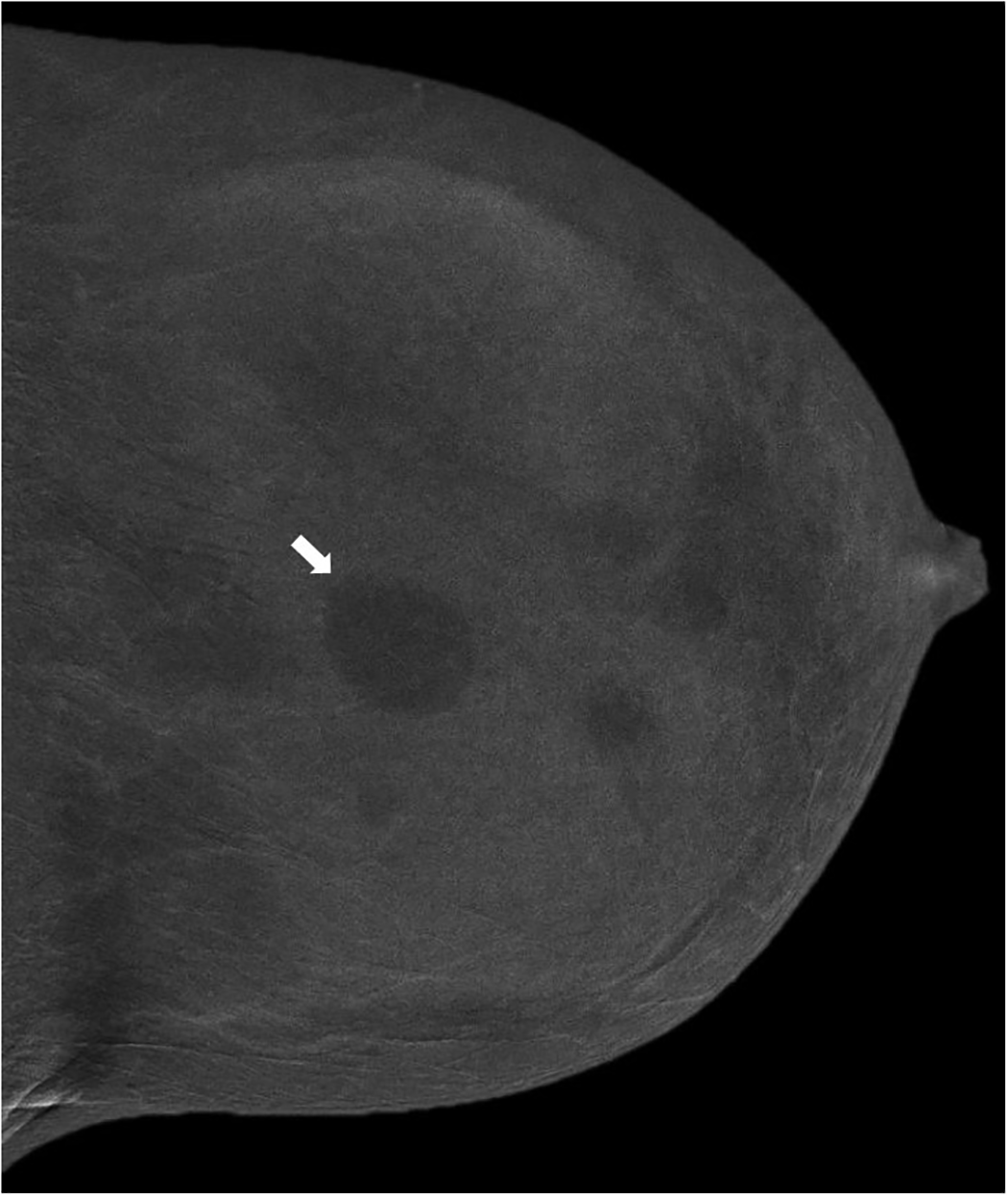
Recombined images in a craniocaudal projection of the left breast in a 41-year-old patient show multiple rounded non-enhancing masses shown to be cysts on ultrasound. Arrow points to the largest mass.
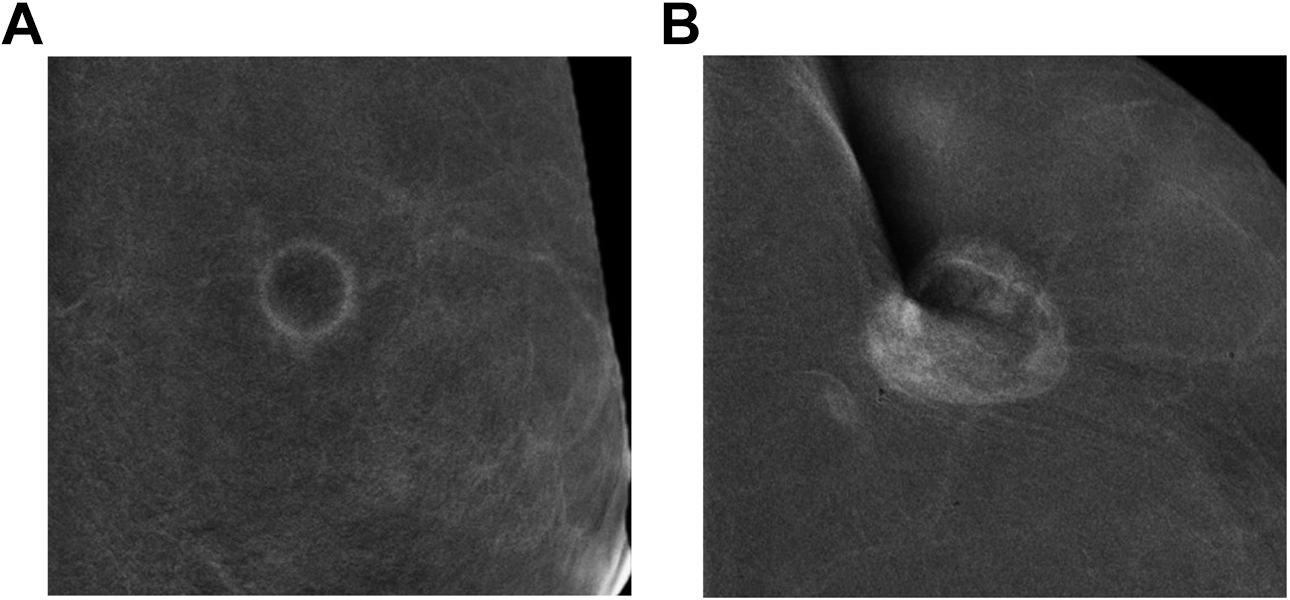
Two rim enhancing masses on recombined views. On the left, cyst showing eclipse sign (A). On the right, triple negative invasive mammary carcinoma demonstrates thick and irregular rim enhancement (B).
In general, masses with irregular shape, non-circumscribed margins (irregular and spiculated), and heterogeneous internal enhancement have the highest prediction of malignancy with positive predictive value (PPV) of 92.4%. Round and oval-shaped masses have a negative predictive value (NPV) of 44.9%, 25 indicating that malignancy cannot be excluded based on shape alone. Although the internal enhancement pattern of the mass lesions has a very low specificity (58.0%) and NPV (40.0%), dark internal septations are most suggestive of fibroadenomas 22,23,25 with phyllodes tumors being another consideration (Figure 7A, B).
An example of invasive mammary carcinoma. Recombined view in a craniocaudal projection of the right breast in a 42-year-old patient shows irregular mass with irregular and spiculated margin and heterogeneous enhancement (A). (B) An example of fibroadenoma. Recombined view in a craniocaudal projection of the left breast in a 37-year-old female shows an ovoid mass with dark non-enhancing septations.
Architectural Distortion
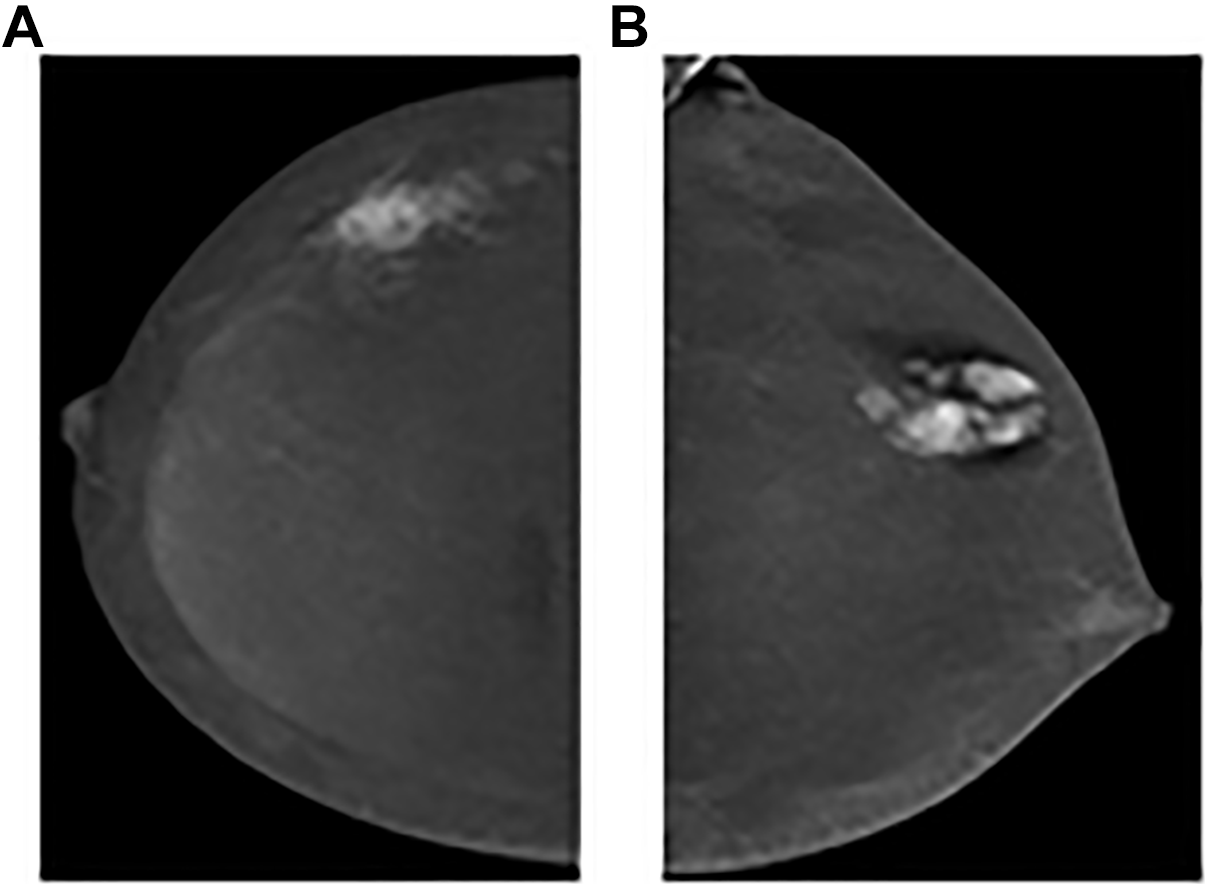
Architectural distortion (AD) can be seen with benign or malignant entities. 28 While DBT has shown high PPV (74.5%) associated with AD, especially when there is an associated abnormality detected by US, 29,30 CEM may assist in differentiating benign from malignant AD by showing a high degree of enhancement. 31 However, if AD is confirmed on LE image or DBT, biopsy must be performed regardless of degree of enhancement as some low-grade malignant lesions may not enhance 12,18,28 -31 (Figure 8A, B).
A and B, Craniocaudal projection of the right breast in a 68-year-old female who was called back from screening. Tomosynthesis view (A) shows a large area of architectural distortion (arrow). Recombined view in the same projection shows no enhancement. The lesion was biopsied under ultrasound guidance (not shown), proved to be grade 1 invasive lobular carcinoma.
Microcalcifications
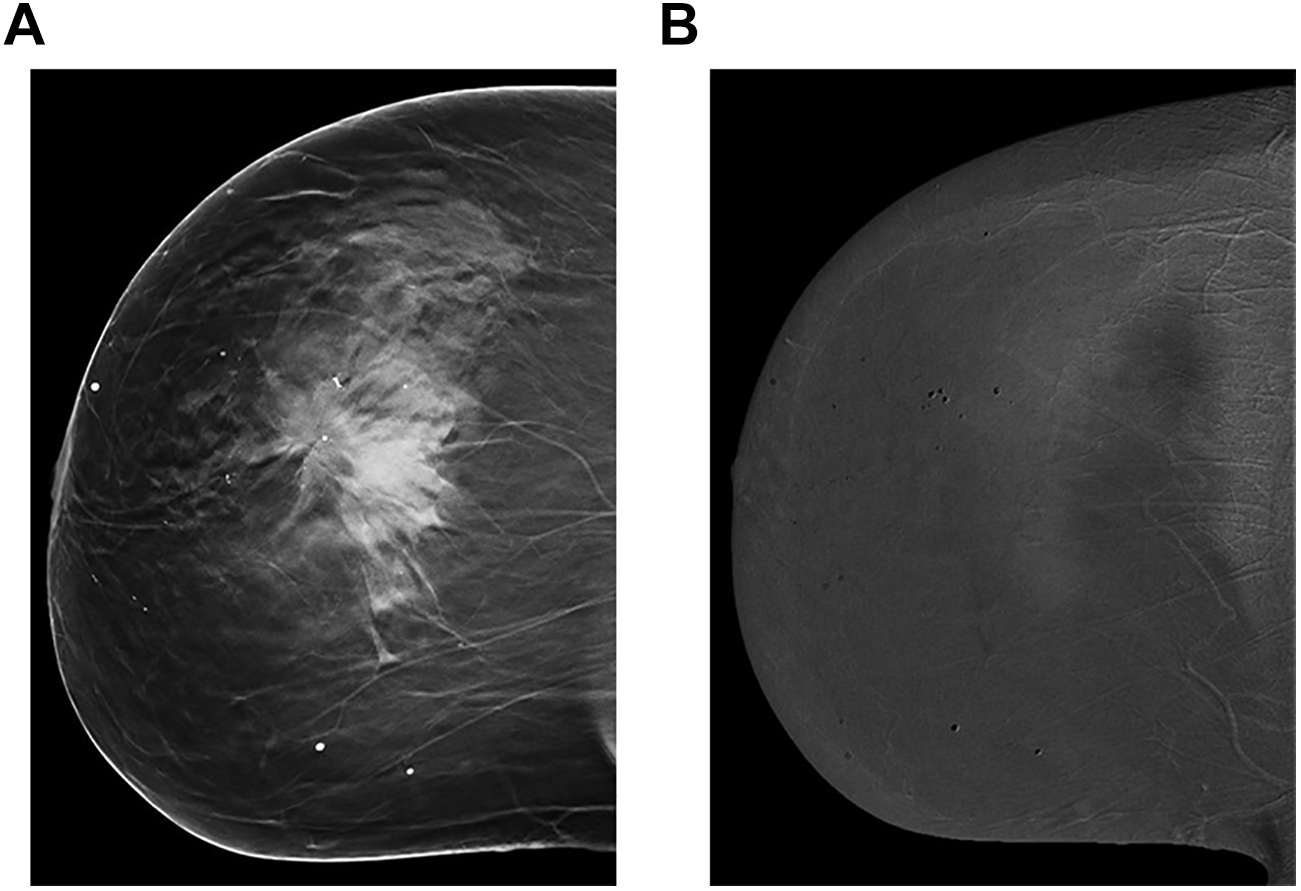
Microcalcifications can be characterized based on the LE and magnification views. 8,12,32 Although more common with malignant entities, one must be cognizant that associated enhancement can also be seen within benign areas of microcalcifications. 8,12,18,33 Houben et al. 34 reported only minimal improvement in sensitivity and NPV in the assessment of microcalcifications when using CEM compared to FFDM. 34 From the author’s experience (AK), CEM might show an occult mass or may help to increase biopsy target accuracy. Regardless of the presence of enhancement, microcalcifications with malignant or indeterminate morphologic features require tissue sampling 8,12,18,32 -34 (Figure 9A, B).
A and B, An example of high grade ductal carcinoma in situ. 62-year-old female who presented from screening for further assessment of left breast microcalcifications. Magnification view in craniocaudal projection (A) shows pleomorphic branching microcalcifications in a segmental distribution. Recombine view in a craniocaudal projection shows associated mild non-mass enhancement.
Asymmetries
Asymmetries (one view, focal, or global) 35 require multimodal evaluation. The absence of associated enhancement might help the radiologist determine that an asymmetry is benign and prevent unnecessary short-term follow-up imaging or biopsy, but if the asymmetry presents as a new mammographic finding without a benign correlate, it should be biopsied regardless of enhancement. 36
Other Interpretation Considerations
Some CEM users have suggested the use of a score to quantify lesion enhancement relative to the BPE. 8,37,38 Although this may assist in distinguishing malignant from benign lesions and could support the decision to biopsy, it should be kept in mind that some malignant lesions may show minimal or no enhancement at all. 8(p15),9 -12,24 -27
Another descriptor that can be utilized is the timing of lesion enhancement in early and delayed images. Although CEM is not a dynamic scan, some authors have observed that availability of images obtained more than 7 minutes after injection can provide similar information to that of DCE-MRI. This is due to the fact that malignant lesions tend to washout on the delayed images, whereas benign lesions tend to demonstrate a progressive enhancement pattern. 8(p15),39
Ultimately, the degree of suspicion of morphologic findings on both the LE and the HE images should determine the appropriate final BI-RADS category, using the standard categories that range from 0 to 6. 22,23 If CEM exams are performed in conjunction with additional mammographic views or US, the final BI-RADS category should consider all exams.
A suggested structured report of CEM exam is provided in Table 1.
Contrast Enhanced Mammography Report.

Limitations
Though beneficial in numerous circumstances, CEM is not immune to a variety of risks and limitations. Therefore, CEM procedures must be supervised by a physician and staff must be trained on the administration of iodine contrast agents and management of complications. 12 Adverse reactions to low-osmolality iodine agents can occur in 1-3% of patients and are often mild and self-limiting while the risk of death is reported to be 2.1 per 1 million. 9 The use of CEM in patients with known renal failure is contraindicated and for patients who are at risk for renal failure, evaluation of renal function is required, as is the standard practice for Contrast Enhanced Computed Tomography (CE-CT). It is generally recommended not to premedicate patients with a history of allergic reaction and to offer alternatives such as DCE-MRI if possible. 12
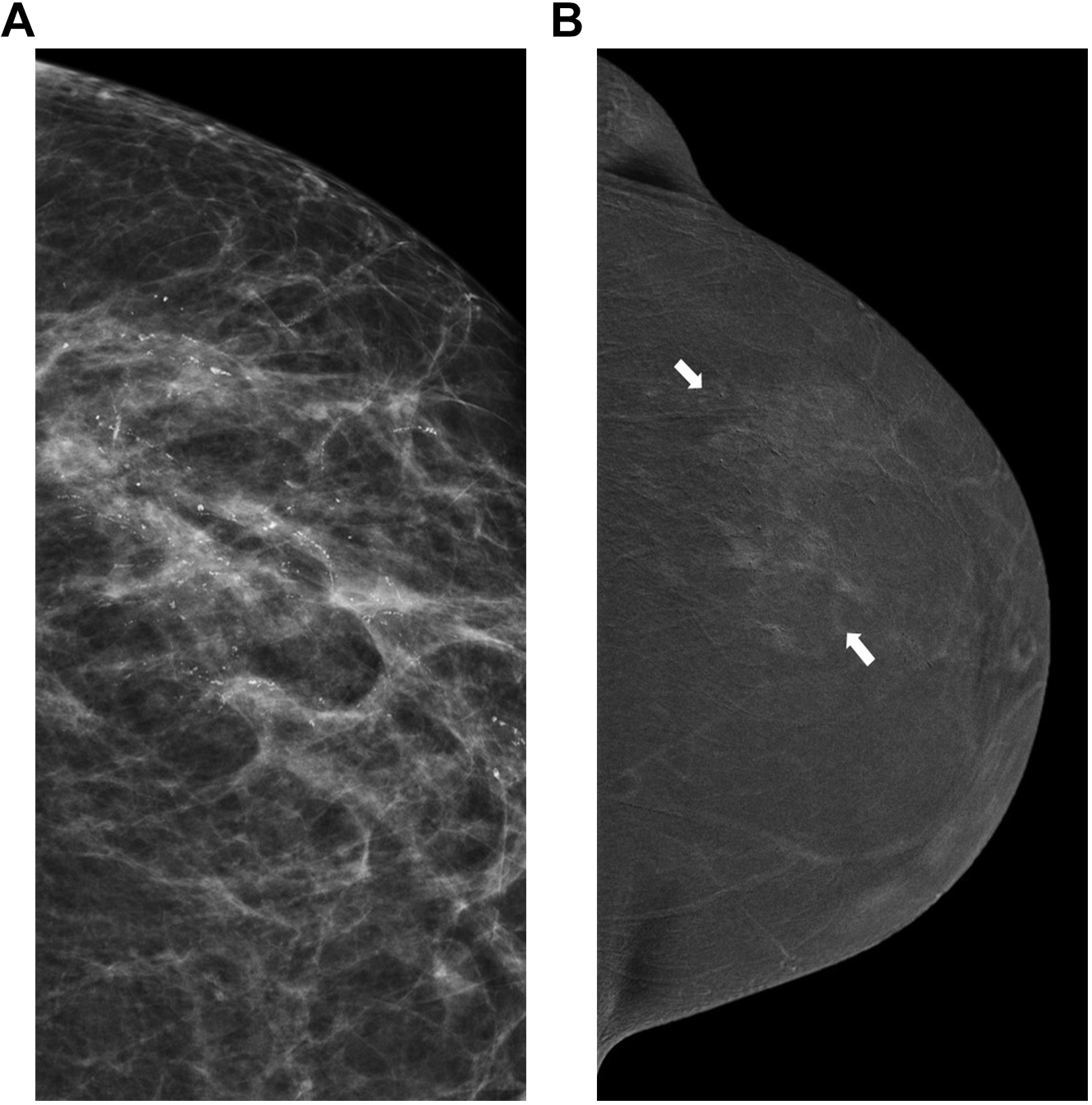
Another limitation of CEM is its propensity to be subject to a multitude of artifacts. Some artifacts, such as inadequate positioning, abnormal timing to contrast bolus, contrast spill on the compression plate or detector, or trapped air within skin folds, might be technical in nature. 40 Other artifacts that are more unique to CEM may include: negative contrast enhancement (the absence of enhancement in cysts or coarse calcifications) 8,40 ; rim artifact (also called “scattered radiation,” “halo,” or “breast-within-a-breast” caused by non-uniformity of breast thickness resulting in a halo appearance within the breast) 40 ; ripple artifacts (motion or cardiac pulsations between the LE and HE views) 41 ; and transient retention of contrast in vein (likely related to stasis of contrast caused by compression). 40 CEM images on patients with implants may be suboptimal. 12 However, if performed correctly, CEM can be used with implant displaced views (Figure 10A-F).
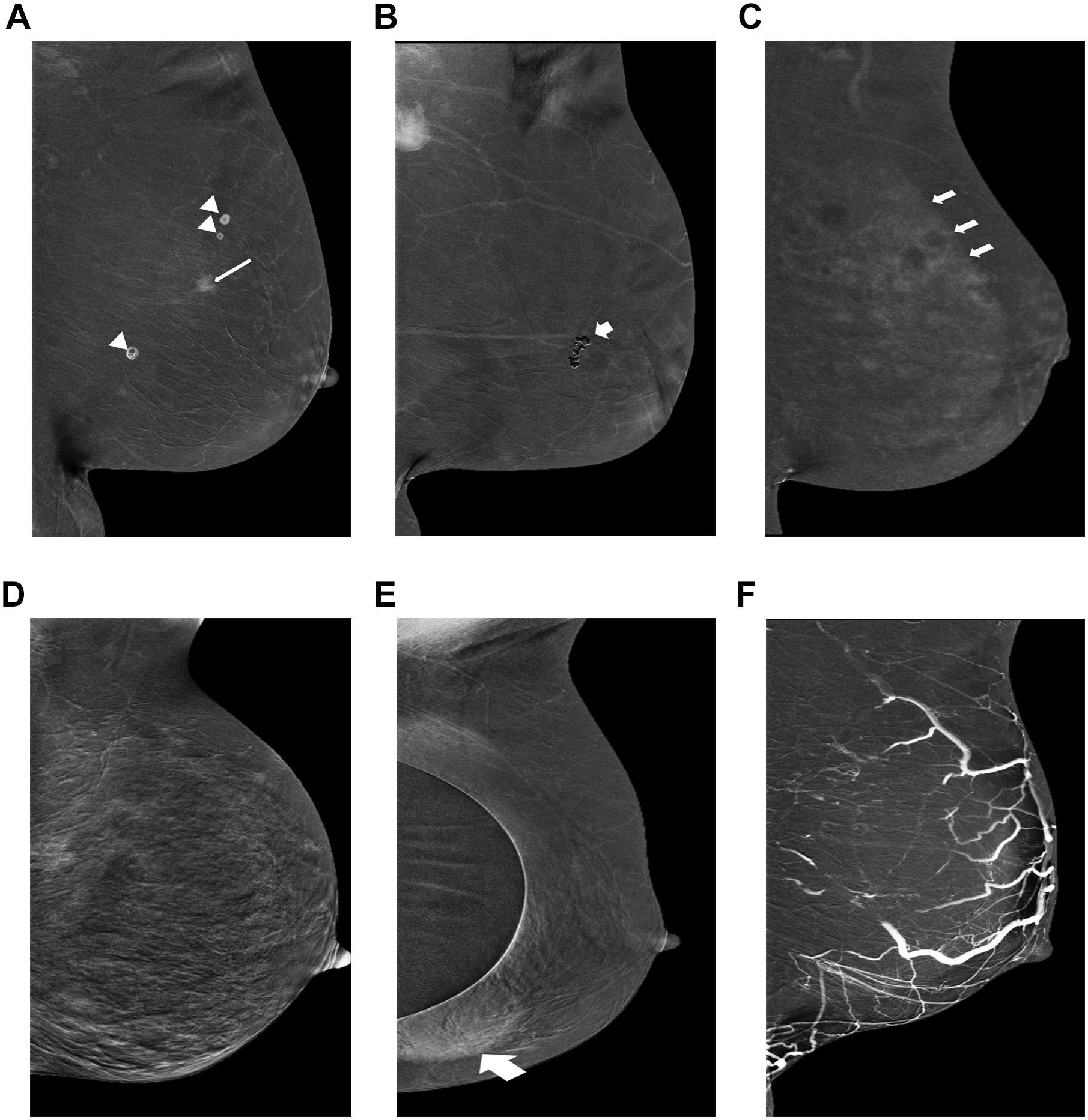
Recombined views in mediolateral oblique projections of the left breast of 6 different patients. From left to right: (A) Contrast spill on the compression plate (arrow heads) should be distinguished from enhancing invasive mammary carcinoma (long arrow); (B) “Negative” artifact caused by regional coarse calcifications (short arrow); (C) Rim artifact (“scattered radiation” “halo” or “breast-within-a-breast”), caused by non-uniform breast thickness resulting in a “halo” appearance within the breast (arrows); (D) “Ripple artifacts” motion or cardiac pulsations between the low-energy and high-energy views, result in coarse and diffusely distorted appearing image; (E) Transient retention of contrast in veins likely related to stasis of contrast caused by compression; (F) Implant displaced view image is hampered by the presence of implants result in obscuring of underlying breast lesion (thick arrow).
Similar to other imaging modalities, false positive and false negative results may occur with CEM. As previously discussed, enhancing lesions may be missed when there is increased BPE, or may show little enhancement, such as with invasive lobular carcinoma (ILC) or ductal carcinoma in situ (DCIS). 8(p342),9 -12,18,26,32 -34,42 On the other hand, benign lesions such as fibroadenomas, inflamed cysts, abscesses, and papillomas can be associated with contrast enhancement, resulting in unnecessary imaging and biopsies. 9 -12,18
Tagliafico et al. 27 performed a meta-analysis study and found that, on the basis of 994 lesions evaluated in 920 patients, the sensitivity of CEM was very high (95% confidence interval 0.96-1.00). Despite this, the specificity was relatively low with large confidence intervals (95% confidence interval 0.38-0.77), but with great variability across studies. Suter et al., 41 on the other hand, performed a meta-analysis of prospective studies and found that CEM had a lower sensitivity but a higher specificity than Tagliafico’s findings (85% and 77%, respectively). This higher specificity may be attributed to greater experience with CEM and improved pre-specified evaluation criteria. In any case, correlation with previous exams such as FFDM, US, DCE-MRI or even chest CE-CT, can reduce false positive rate and improve accuracy. 43
Finally, the smaller field of view available in CEM results in inferior detection of chest wall invasion, internal mammary metastasis, and axillary node disease as compared to DCE-MRI. 12
Clinical Use
CEM is primarily used in the diagnostic setting and has shown high sensitivity which approaches that of DCE-MRI, especially with dense breasts, with higher rate of PPV and specificity 9 -12,18,44 -52 (Table 2). The easy access to CEM allows same day assessment with a focussed second look US and reduced diagnostic time while freeing up MRI resources. Therefore, it is prudent to implement CEM as an alternative to DCE-MRI when enhancement information would be helpful, or even in other settings where DCE-MRI would not be considered due to access or patient’s limitations. 9 -12,18
Comparison Between MRI and CEM in the Diagnosis Setting in Selected Clinical Studies.
The following outlines several common clinical scenarios where CEM may be of benefit.
Staging of Breast Cancer
Multiple studies appear to agree that the use of CEM is more sensitive than DM or US in the evaluation of extent of disease in breast cancer, especially in cases of dense breasts, 23,44 -49 representing an acceptable substitute for DCE-MRI 50 -57 (Figure 11A, B; Figure 12A, B). MRI is required for lesions that are not included in the mammography field of view, unless staging CT can provide the required supplementary information.
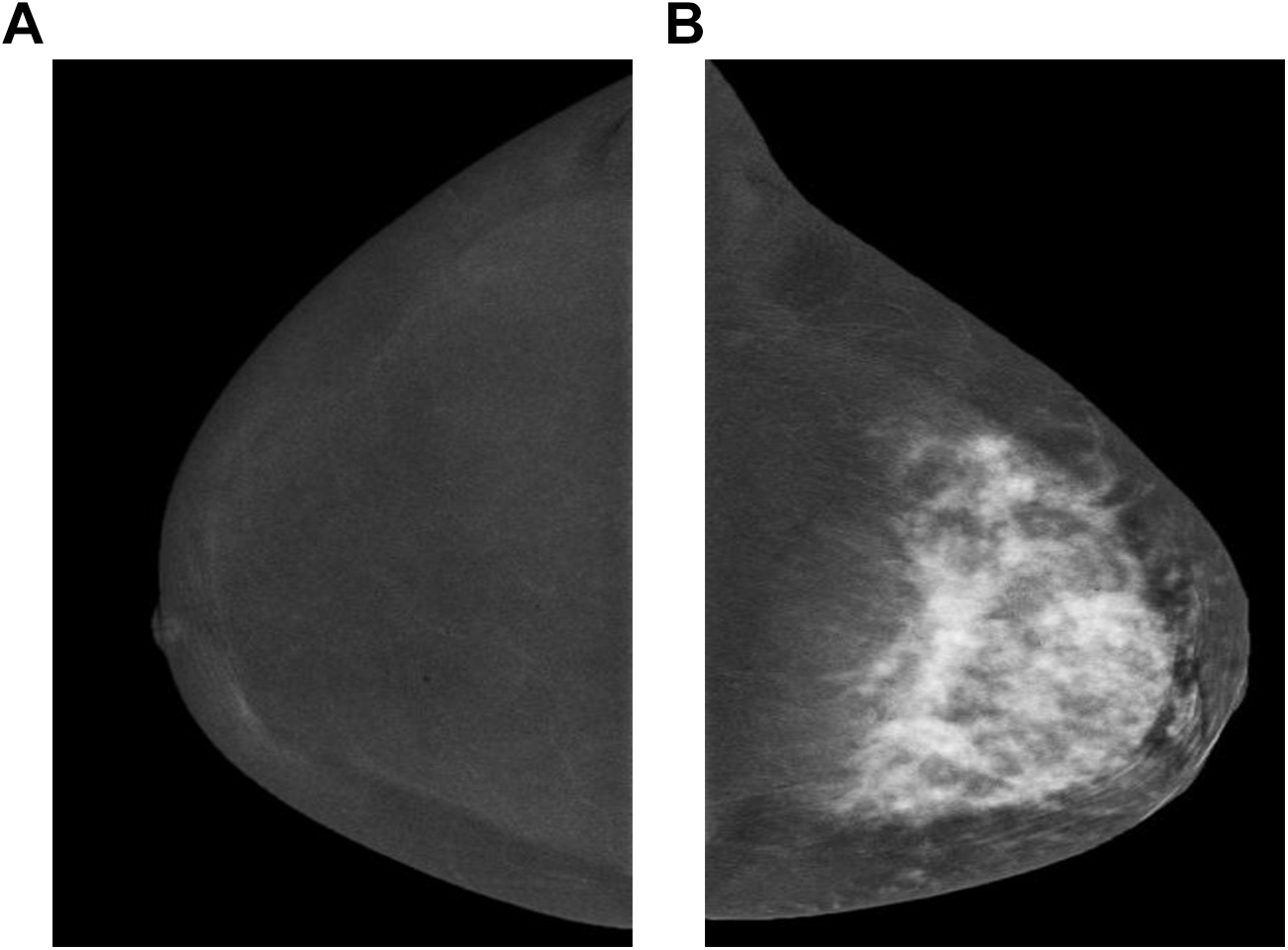
Bilateral recombined views in craniocaudal projections of a 48-year-old female who presented with a left breast lump. Normal appearance right breast (A) in comparison to extensive enhancement of the left breast (B), in keeping with locally advanced breast malignancy. Histopathology confirmed invasive lobular type.
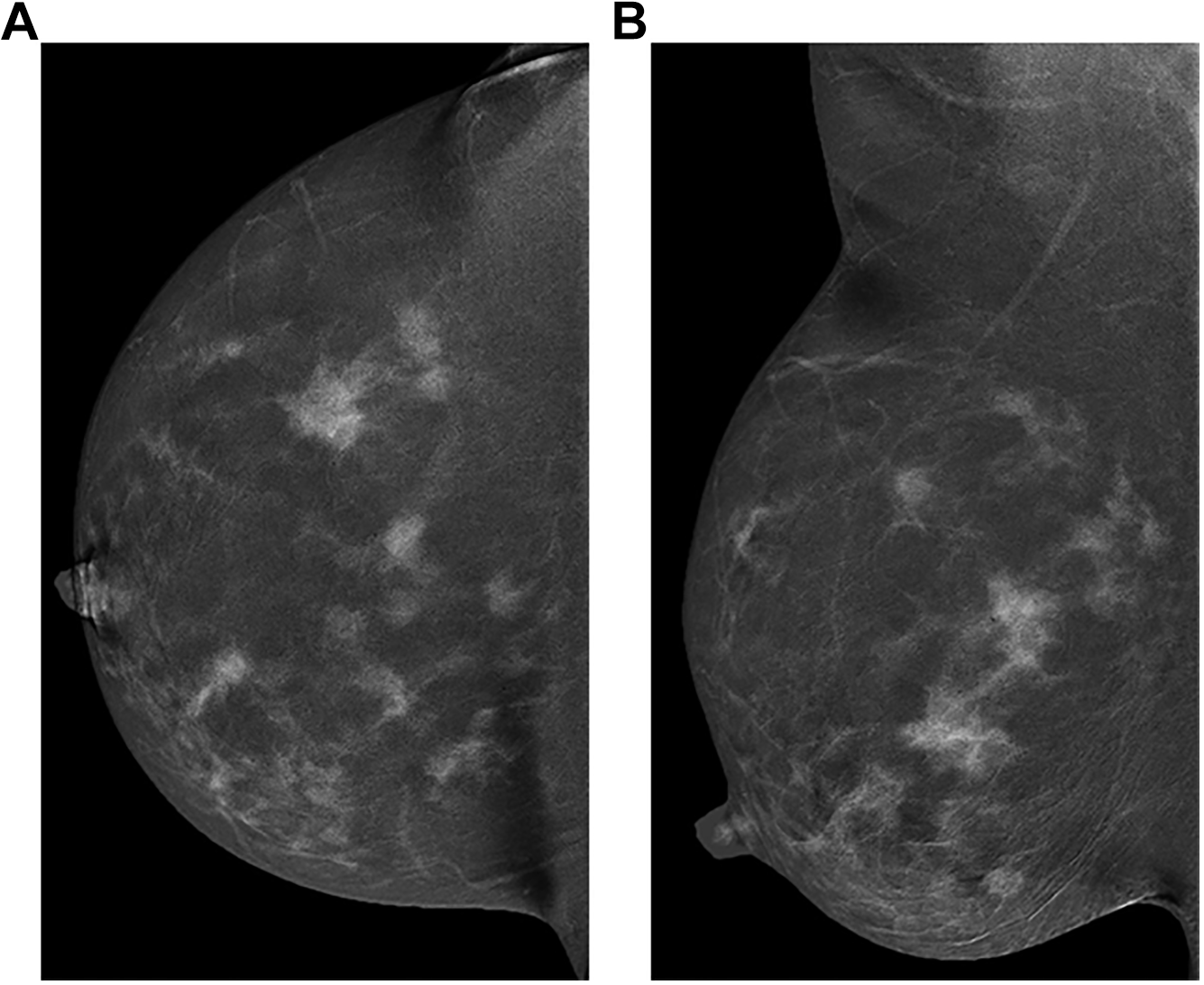
An example of multicentric disease. Recombined view in craniocaudal (A) and mediolateral oblique (B) projections of the right breast in a 29-year-old female who presented with right breast lump.
A good correlation between lesion size on CEM and final histopathological size was described by several authors. 52,53 A study by Lobbes et al. found no change in surgical plans when performing DCE-MRI after CEM, indicating lack of benefit to adding DCE-MRI after CEM. 54 When compared with FFDM, CEM has been found to be superior at delineating malignant lesions and can reduce US scanning time. 53,54 Patel et al. 55 performed a retrospective review of the performance of CEM in the assessment of 30 women who were diagnosed with ILC, showing that only 2 out of 30 (6.7%) cases required surgical re-excision.
When compared with DCE-MRI, CEM might identify fewer incidental contralateral breast malignancies but has been shown to have fewer false positive results and significantly higher PPV. 50,56 The detection of secondary cancers with CEM was also similar to that with DCE-MRI.
Evaluation of Recalls From Abnormal Mammogram and Problem Solving
Another interesting value provided by CEM is in the assessment of patients who are called back after an abnormal mammogram, either as an adjunct to or in place of traditional diagnostic mammography. 18 Studies report high sensitivity for CEM in the assessment of patients who have been recalled after screening, particularly in those with increased breast density. 7,9,33,34,45 Lobbes et al. 57 showed that CEM is an excellent problem-solving tool to address inconclusive findings on screening mammography, especially in reducing the number of false positive recalls. In addition, CEM is associated with high NPV, suggesting that a negative CEM in these women can be used to exclude malignancy and the need for short-term follow-up. 57 In their reader study, Lalji et al. 20 included 76% of patients recalled from screening and found that CEM had a sensitivity of 97% and specificity of 70%. Tardivel et al. 46 showed that CEM drove 21% of changes in surgical or diagnostic strategies in breast cancer staging, in problem-solving issues after conventional breast imaging, or in recurrence detection.
For a solitary mass detected by standard mammography, the main role of CEM is to help identify any additional abnormalities, but the evaluation must include assessment with US. 18 As for asymmetries, Wessam et al. 58 showed that focal asymmetry with associated enhancement on CEM was highly correlated with malignancy, while non-enhancing focal asymmetry was correlated with benign pathology (P ≤ 0.001).
As previously discussed, the morphology of a lesion is very important, regardless of the degree or pattern of enhancement. Therefore, one should not dismiss a suspicious finding on LE image that does not enhance on CEM. 8(p.15),12
Breast Symptoms
CEM is reported to be a promising tool in the setting of symptomatic breast complaints (e.g., palpable mass, localized breast pain, and nipple discharge). Jong et al. 7 showed statistically significant improvement of overall CEM performance compared to LE images alone in the assessment of breast symptoms. Tennant et al. 59 reported a sensitivity of 94.5% in the CEM exams of symptomatic patients and Luczynska et al. 49 showed significantly higher sensitivity and accuracy for CEM than FFDM or US in symptomatic breasts. The area under the curve (AUC) for CEM in the latter study was similar to US but significantly larger than that of MG. Lu et al., 60 on the other hand, showed that in symptomatic patients, the differences between CEM and US were not statistically significant.
To the author’s knowledge, there is no published study comparing CEM to DBT with US in symptomatic patients, which is the more common practice for evaluating patients with breast symptoms.
Some breast malignancies, such as invasive lobular carcinoma (ILC), may not be detected by either LE or recombined views and may only be detected by US. In fact, a finding detected by US may direct the reader to identify a subtle correlate on LE and recombined images. Therefore, the standard of using US in the assessment of palpable lumps should be maintained while CEM can be used as an adjunct tool for reassurance. 12
Response to Treatment
An additional indication for CEM is in the evaluation of response to treatment. Studies have found that the performance of CEM is comparable with that of DCE-MRI in the ability to determine residual breast cancer after treatment. 9,11 Iotti et al. 61 performed a prospective review of the use of CEM and DCE-MRI before, during, and after neoadjuvant therapy, showing that CEM is as reliable as DCE-MRI in assessing the response to treatment and may be considered as a suitable alternative. 61 A subsequent retrospective study has shown that CEM and DCE-MRI had comparable PPVs and sensitivity for identifying residual disease. 62 Similar results were described by Marinovich et al. and Patel et al. 63,64 If being used for this indication, a baseline CEM should be conducted to evaluate the disease extent before treatment begins (Figure 13A, B).
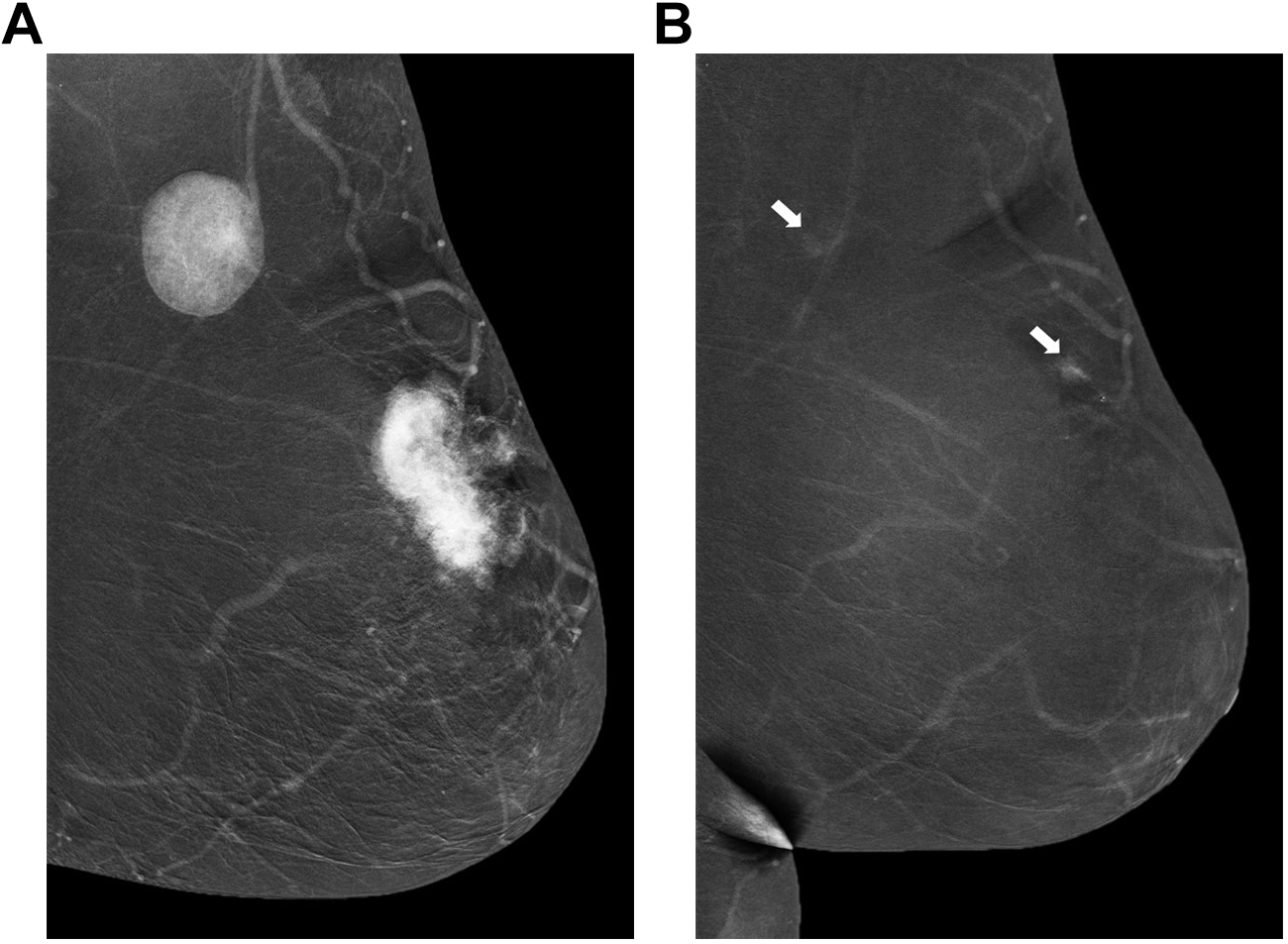
Recombined views in mediolateral oblique projections from a different exam date, in a 72-year-old female. First exam (A), obtained for further assessment of metastatic lymphadenopathy, shows irregular enhancing mass with lobulated margin at the superior aspect of the left breast as well as a markedly abnormal appearing axillary node. Following neoadjuvant therapy (B), there is significant reduction in the size of the primary lesion and lymph node (arrows) indicates near complete response. Minimal residual invasive mammary carcinoma and no pathologic lymph nodes were confirmed on final histopathology.
To the author’s knowledge, no studies have been published investigating the use of CEM to monitor response to hormonal treatment.
Screening and Surveillance
The increasing use of supplemental screening with US and DCE-MRI for women with dense breasts or at intermediate risk for breast cancer raises questions regarding the role of CEM in this setting. Initial results in high-risk screening as reported by Jochelson 65 are promising as CEM sensitivity, specificity and PPV were found to be comparable to those of breast DCE-MRI in detecting mammographically occult invasive lobular cancer in 2 out of 309 patients. A third patient with a focus of enhancement on MRI that was occult at CEM was upgraded from atypical ductal hyperplasia to DCIS at surgery. 65 Sorin et al. 66 have shown a high overall incremental cancer detection rate (ICDR) (13.1 cancers per 1000 women) when CEM was used in place of FFDM in patients with a personal history of breast cancer or with an intermediate lifetime risk. Furthermore, when no cancer was detected by CEM, there was no added benefit in the performance of US as an adjunct tool. A prevalence study reported by Sung et al. used CEM for screening 904 patients with either dense breasts, personal history of breast cancer, or an intermediate to high lifetime risk of developing breast cancer. The study found a high ICDR (15.5 per 1000), with 7 out of 15 cancers detected only by the recombined views. 67 These results are promising considering the use of supplementary screening with DCE-MRI in patients with dense breasts has an ICDR of 16.5 per 1000. 68
The ACR recently announced the launch of Contrast Enhanced Mammography Imaging Screening Trial (CMIST) to determine if CEM screening provides more accurate detection than current screening methods (DBT) in women with average to intermediate risk and dense breasts. 69 Until further data is available, CEM cannot be offered as a replacement for DCE-MRI in the intermediate to high risk screening setting.
CEM vs DBT and DCE-MRI
Depending on the vendor, DBT can be obtained either as a separate exam or in conjunction with CEM under the same compression, resulting in higher radiation exposures although still below the recommended maximum dose. 15
Studies have shown that conventional DBT is more accurate in detecting breast cancers and AD than FFDM, in both clinical and screening populations, especially in dense breast. 70 -72 Patel et al. 31 showed high sensitivity and NPV when using CEM in the assessment of architectural distortion originally detected by DBT.
In a study by Petrillo et al. 73 where 53 benign and 81 malignant lesions in women who underwent DBT, CEM and DCE-MRI, a sensitivity of 91% was demonstrated when CEM was combined with DBT, similar to the sensitivity of MRI time-intensity curve, but less than 96% for DCE-MRI. The combination of CEM with DBT was shown to be of particular value in the detection and assessment of the extent of breast cancer, differentiation of benign from malignant disease, and assessment of multifocal disease. 73
To date, there is no commercially available contrast enhanced tomosynthesis (CET) that allows dual-energy technique to be applied to DBT without increased radiation exposure. Few studies have investigated the use of CET prototypes, 74 -76 showing that CET is able to show lesion morphology more accurately.
Management and Biopsy
In the instance that negative or benign findings (BI-RADS category 1 or 2) cannot be determined based on CEM alone, a second-look US is usually needed. If a lesion was categorized as probably benign (BI-RADS category 3) based on CEM, additional CEM is necessary at the time of follow-up. Suspicious findings (BI-RADS category 4 or 5) should be sampled by either percutaneous or surgical biopsy. Though timing during the menstrual cycle appears to have minimal effect on the amount of BPE at CEM, 12,19 one may want to consider performing the follow-up exam at the second week of the menstrual cycle, if applicable. 12
US-guided biopsy is the ideal technique for tissue sampling by providing real-time imaging. If there is no US correlate to an area of abnormal enhancement, even subtle LE findings can be targeted for stereotactic-guided biopsy. If the suspicious abnormality is visible on DBT, it could be used to guide tissue sampling.
A dedicated CEM-guided biopsy method, has recently been approved by the American FDA and Health Canada. 77,78 This technique enables the use of dual-energy exposure with an add-on stereotactic guided biopsy in either a vertical or horizontal approach (Figure 14A, B). Alternatively, a biopsy may be attempted using a technique similar to that used for mammographic wire localizations, except that it would be performed after the administration of IV contrast agent using the dual-energy technique. 79 Otherwise, MRI-guided biopsy is the next option to consider.
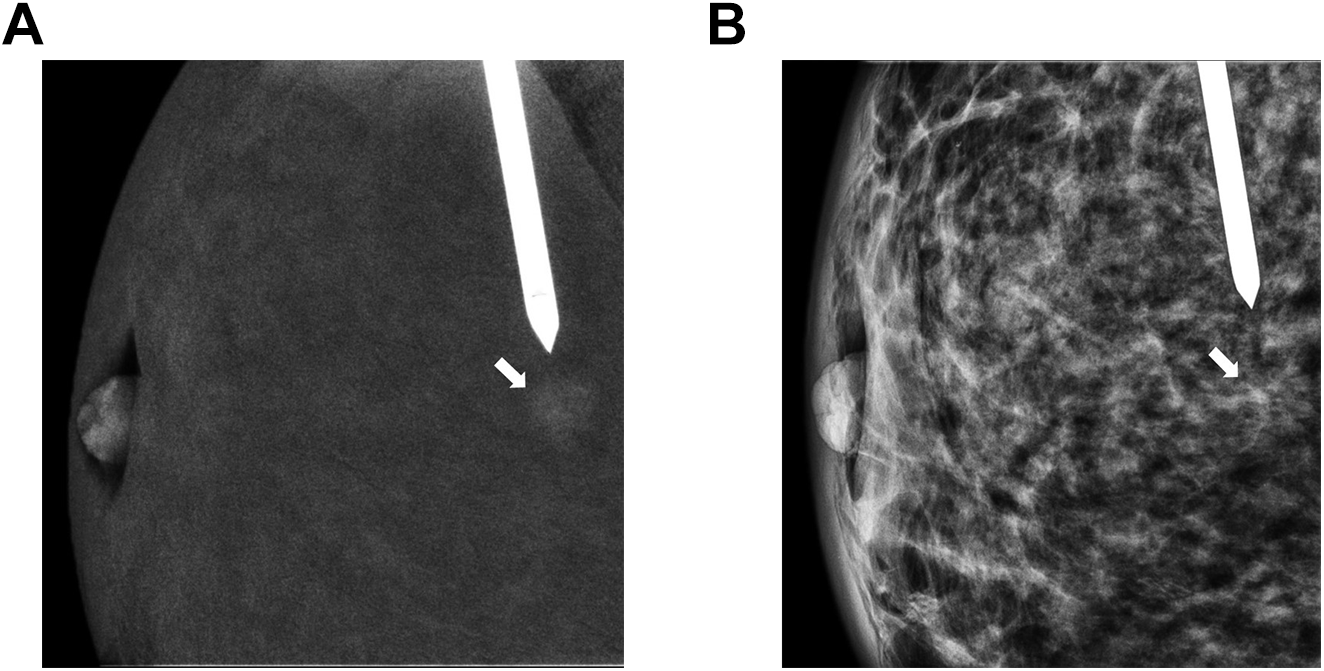
Recombined view (A) in a pre-firing position taken during CEM-guided biopsy in a horizontal approach, targeting an irregular small enhancing mass (arrow). Note mild degree of associated distortion appreciated on the low-energy view from the same projection (B) which can be used as a landmark if the lesion enhancement starts to washout.
Regardless of the modality used for biopsy, a tissue marker should be placed in the area biopsied.
Concordant benign biopsy results and lesions detected only by recombined views are 2 instances in which a 6-month follow-up with repeat CEM should be conducted. 12 This is consistent with the current practice with MRI findings.
Conclusion
CEM is an emerging technique that can be used for multiple diagnostic breast imaging indications with a sensitivity and specificity approaching that of DCE-MRI, particularly in dense breast. The main advantages of CEM are providing rapid streaming to treatment and allowing diverse diagnostic breast MRI resources to be available for other purposes such as supplementary screening. Future perspective might be the development of contrast enhanced tomosynthesis (CET), at the same radiation dose, which will add the benefit of 3-dimensional information to that of the contrast enhancement.
The role of CEM in supplementary screening is promising based on preliminary data but future research is needed to determine what patients may benefit most. The forthcoming Contrast-Enhanced Mammography Imaging Screening Trial (CMIST), will help to determine if CEM can improve breast cancer detection compared with DBT in patients with dense breasts.
Footnotes
Acknowledgments
Kalan Lynn, Natalia Mlynski, Western University. Magda Spasic, London X-Ray Associates.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The author is the PI of a funded study by GE assessing the feasibility of contrast enhanced mammography software to added to standard stereotactic guided biopsy.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
