Abstract
Background:
Seizures are common neurological events in children, with neuroimaging playing a crucial role in evaluating new-onset seizures. While magnetic resonance imaging (MRI) is often preferred over computed tomography (CT) for pediatric seizure imaging due to higher sensitivity and lack of ionizing radiation, practices regarding imaging protocols and sedation use vary. Currently, there are no published Canada-wide guidelines describing the practices for workup of pediatric seizures.
Methods:
A cross-sectional survey was conducted among radiologists at 16 Canadian tertiary pediatric centers to assess neuroimaging practices for children with new-onset seizures. The survey explored the presence and content of dedicated seizure MRI protocols, sedation use, and strategies to facilitate non-sedated MRI.
Results:
Fifteen centers (94%) responded. Only 2 (13%) reported using dedicated new-onset seizure protocols, while 10 (67%) used epilepsy-specific MRI protocols, and the others used different approaches, including variations of routine brain MRI. MRI sequences varied across institutions. Sedation use also varied, with a median sedation age range of 3 months to 6 years. Non-sedated MRI techniques such as feed-and-swaddle (93%) and natural sleep (27%) were commonly used for infants. Video goggles (67%) and child life specialist support (53%) were often used for older children. Only 2 institutions (13%) had fast MRI protocols, and virtual reality preparation was uncommon (13%).
Conclusion:
Considerable variability exists in MRI protocols and sedation practices across Canadian pediatric centers evaluating new-onset seizures. Our findings emphasize the need for national consensus guidelines to standardize imaging protocols, reduce sedation use, and optimize care for pediatric seizure patients.
Introduction
A seizure is a paroxysmal change in neurological function secondary to abnormal, excessive, synchronous neuronal activity. 1 Approximately 8% to 10% of the population will experience at least one seizure in their lifetime. 2 The incidence of acute symptomatic seizures is highest in infancy, and seizures account for 1% of all pediatric emergency department visits.3,4
Depending on the clinical history, physical examination, laboratory investigations, and electroencephalography (EEG), neuroimaging may be used to investigate first-time seizures in children. While neuroimaging is not necessary when the clinical history and EEG suggest idiopathic epilepsy or simple febrile seizures, it is recommended for convulsive or focal seizures, seizure clusters, changes in seizure pattern, or seizures with post-ictal focal neurological deficits.5,6 Anatomical neuroimaging can identify structural lesions such as tuberous sclerosis complex (TSC) or brain tumors as potential causes of seizures. 7
Computed tomography (CT) and magnetic resonance imaging (MRI) are the 2 main methods of structural neuroimaging. CT is often useful in emergency situations due to its fast scanning speed and accessibility. 8 However, it has poor overall sensitivity (30%) and produces low resolution images, particularly in the temporal fossa, leading to a failure in detecting abnormalities in up to 50% of patients with epileptogenic structural lesions. 8 Moreover, CT exposes patients to ionizing radiation, to which children are particularly sensitive. 9 MRI is thus the imaging modality of choice: its high resolution offers great sensitivity without exposing patients to ionizing radiation. 8 However, MRI often requires longer acquisition times and is highly sensitive to motion, which can make imaging young children challenging. 8 High-quality MR images of the brain may require that the child lie still for up to an hour; therefore, younger children have traditionally undergone MRI under sedation or general anesthesia.10,11 Sedation and anesthesia carry potential neurodevelopmental risks, increase costs, and lengthen appointment and wait times.12-15 To minimize sedation with pediatric MRIs, several non-pharmaceutical strategies are employed, including feeding and swaddling infants to induce sleep, supporting patients with child life specialists, and using audiovisual distraction tools.16,17
Despite the high incidence of pediatric seizures, there are no published nationwide guidelines regarding imaging practices for children presenting with a first-time seizure. The imaging modality of choice, protocols, use of sedation and approaches to non-sedated scans for new-onset seizures at Canadian pediatric centers have yet to be described. To address this gap, we conducted an environmental scan of current imaging practices for the work up of pediatric seizures across Canada, as a first step toward establishing national consensus and best-practice recommendations.
Methods
Study Design and Survey Characteristics
This is a cross-sectional descriptive study developed to understand the neuroimaging methods for assessing children with new-onset seizures in Canada. The study was approved by the research ethics board of the Children’s Hospital of Eastern Ontario (CHEO; #21/120X). The authors of the manuscript designed a conditional branching survey to compare the neuroimaging practices from the 16 Canadian pediatric tertiary care centers across 5 domains: (1) the presence or absence of a dedicated protocol for new-onset seizures; (2) the sequences used in these MRI protocols; (3) the methods used to obtain the brain MRI according to patient’s age; and (4) the use of sedation for brain MRI. The survey consisted of multi-select, free-text, and yes/no questions. A copy of the survey can be found in the Supplemental Materials.
In this study, a dedicated new-onset seizure protocol refers to abbreviated MRI sequences designed for the initial evaluation of children presenting with a first seizure, aimed at rapidly identifying emergent intracranial pathology. Meanwhile, an epilepsy-specific protocol refers to comprehensive MRI sequences optimized for detecting subtle epileptogenic lesions and supporting pre-surgical planning.
Survey Recipients
One radiologist from each of the 15 Canadian pediatric tertiary care centers was approached to complete the survey. An additional radiologist from the Centre hospitalier universitaire (CHU) de Sherbrooke was included, as this institution has a pediatric radiologist on staff, bringing the total number of invited centers to 16. The email addresses of the radiologists were obtained from each hospital’s website. A standardized email containing the electronic survey link was sent to either: (1) a pediatric neuroradiologist at each Canadian pediatric hospital with expertise or clinical interest in seizures or (2) the division or section head of radiology. The survey was available from December 2022 to September 2023. Reminder emails were sent over the course of this period, ranging from 0 to 3 reminders in addition to the original recruitment email, depending on whether the radiologist had already completed the survey.
Survey Apparatus and Statistical Analysis
The survey was created using REDCap, an online survey tool. Survey results were summarized as counts and percentages. Descriptive statistical analyses were performed using Microsoft Excel.
Results
Imaging Protocols Used for New-Onset Seizures
Pediatric radiologists from 15 of 16 pediatric tertiary care hospitals (94%) responded to the survey. For the evaluation of new-onset seizures, 10 institutions (67%) reported using an epilepsy-specific MRI protocol, 5 (33%) used a routine brain MRI protocol, 2 (13%) used a dedicated new-onset seizure MRI protocol, and 4 (27%) reported using head CT. This was a multi-select question, so institutions could choose more than one imaging modality. These results are summarized in Figure 1.
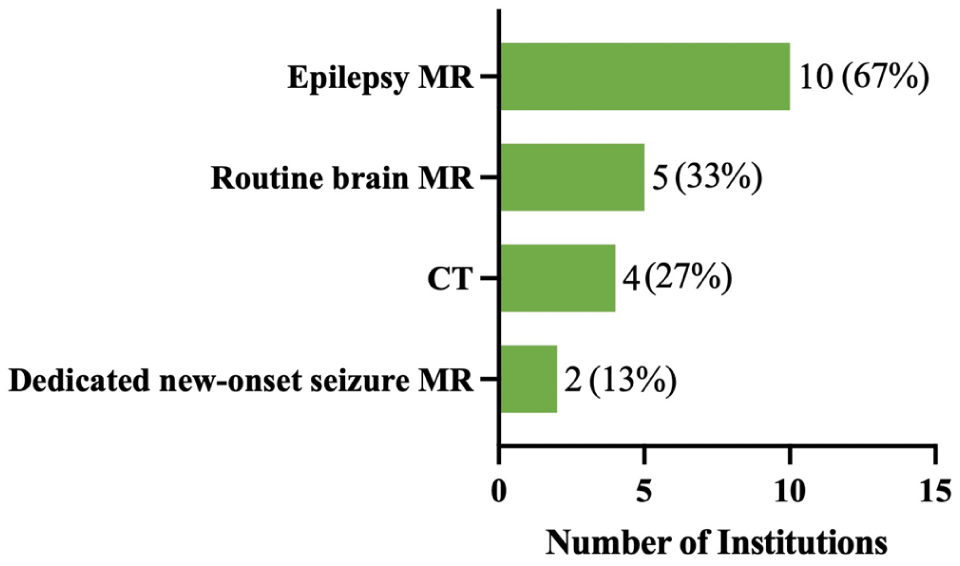
Surveyed site percentages of imaging protocols used for new onset seizures in response to “How does your institution assess new-onset seizures with imaging? Please select all that apply, regardless of whether the method is widely adopted and adhered to.”
One institution specified the use of a routine brain MRI protocol supplemented with an additional coronal inversion recovery (IR) sequence. Another institution mentioned that a non-contrast brain CT is often performed before MRI to rule out life-threatening intracranial pathologies, and that prior to deciding on specific MR sequences, the neuroradiologists will try to obtain further clinical information and review the CT images.
Sequences Used in the MRI Protocols
The common MRI sequences used by the surveyed institutions for imaging pediatric patients with new-onset seizures is summarized in Table 1. In addition to these sequences, several institutions reported supplementary techniques as part of their epilepsy MRI protocols. One institution used proton density (PD) imaging in axial and coronal planes when clinically indicated, another employed a 3D PD inversion recovery (PDIR) sequence, and a third acquired 3-mm T2 fast spin-echo (T2 FSE) slices perpendicular to the hippocampi along with 3D FLAIR reconstruction.
MR Sequences Used to Assess New-Onset Seizures.

Use of Sedation and Anesthesia for Brain MRI
Across the 15 institutions represented, sedation was used to obtain brain MRIs in children ranging in age from birth to 8 years (Figure 2). The lower age limit for sedation varied from newborn to 9 months, with a median of 3 months. The upper age limit ranged from 5 to 8 years, with a median of 6 years.
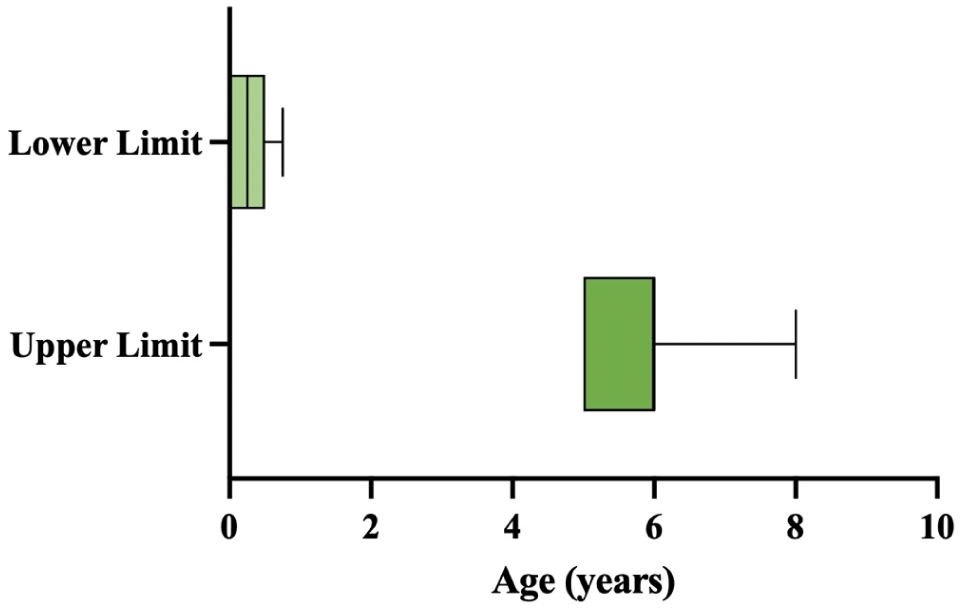
Boxplot illustrating the reported lower and upper age limits for sedated brain MRI across Canadian pediatric tertiary care hospitals (n = 15). Each box represents the interquartile range (IQR) of the reported ages, with the horizontal line inside the box indicating the median. The whiskers extend to the minimum and maximum reported age limits.
One institution clarified that the decision to use sedation to obtain brain MRI varied significantly depending on the type of exam and the findings from an assessment by a child life specialist. Similarly, another institution reported that the decision to proceed with sedation was determined on a case-by-case basis, depending on clinical judgment.
Approaches to Non-Sedated Brain MRI
Figure 3 summarizes the non-sedation techniques (and associated age ranges) used for pediatric brain MRIs. Fourteen out of 15 (93%) radiologists reported that their institution uses the feed and swaddle method to facilitate non-sedated brain MRIs for infants, ranging from 0 to 12 months of age with a median upper age limit of 5 months. Four radiologists (27%) indicated that their institution performs brain MRI under natural sleep for young children, with reported upper age limits ranging from 6 weeks to 3 years.
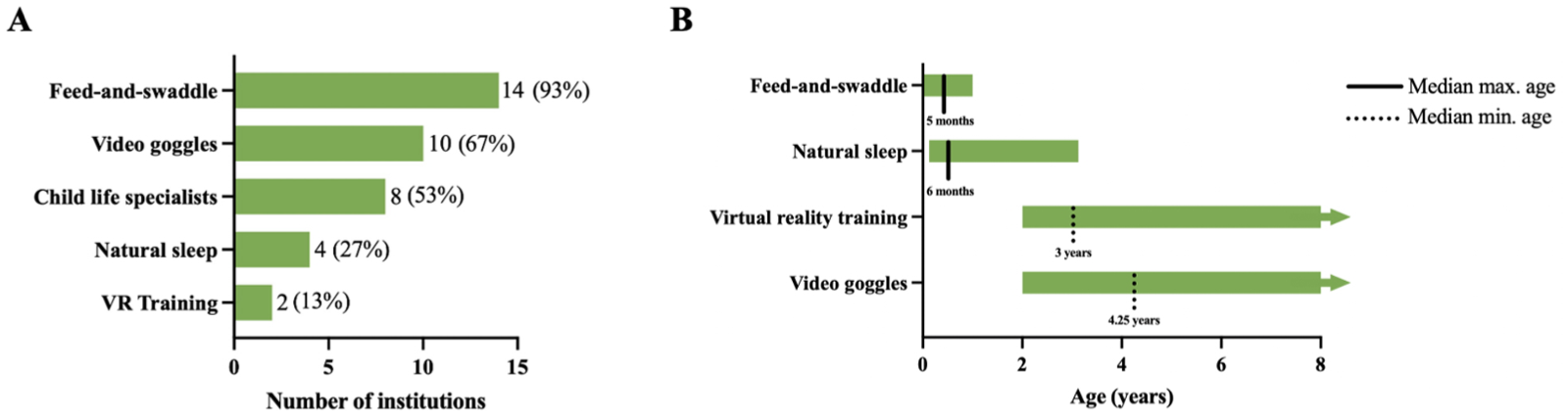
(A) Range of non-sedation techniques used for pediatric brain MRI scans across surveyed sites, based on responses to the question: “Which methods do you use to facilitate non-sedated brain MRIs? Please select all that apply.” (B) Reported age ranges for implementing non-sedation practices. Bars represent the full range of minimum and/or maximum ages across sites, while lines indicate the median reported minimum or maximum age.
Ten radiologists (67%) reported that their institution uses video goggles to facilitate non-sedated MRI in older children, with the minimum age ranging from 2 to 6 years. The median minimum age for starting video goggle use was 4.25 years.
Eight radiologists (53%) stated that their institution involves child life specialists to help prepare children for non-sedated brain MRI. One radiologist reported that children aged 4 years and older are guided through an MRI simulator by a child life specialist before undergoing the scan. Another radiologist described using a toy MRI with a technologist to familiarize the child with the procedure beforehand.
Two radiologists (13%) reported that their institution uses virtual reality (VR) training as part of MRI preparation. One institution used VR for children aged 2 years and older, while the other applied it to children between 4 and 7 years of age.
Dedicated Fast MRI Protocol for Non-Sedated Studies in Children With New-Onset Seizures
Two of 15 radiologists (13%) reported that their institution has a dedicated fast MRI protocol for non-sedated imaging in children with new-onset seizures.
At IWK Health Center (Halifax, NS), the fast MRI protocol is used selectively and has a total acquisition time of 5 to 7 minutes. The protocol includes single-shot fast spin-echo (SSFSE), 2D fast imaging employing steady-state acquisition (FIESTA), fast T1 gradient echo (GRE), and echo planar imaging diffusion-weighted imaging (EPI DWI) sequences.
At CHEO (Ottawa, ON), the fast MRI protocol for new-onset seizures has a total scan time of 5 minutes, including sagittal and axial T1, axial T2, FLAIR, DWI, GRE, and coronal T2 sequences. If the child remains cooperative, the MRI technologist may extend the scan by an additional 13 to 15 minutes to include 3D T1, high-resolution coronal T2, and magnetic resonance spectroscopy (MRS) sequences.
Discussion
The results of our national survey revealed that all Canadian pediatric radiology departments use MRI to assess new-onset seizures; however, only 2 of 15 institutions have a dedicated MRI protocol for new-onset seizures. Two-thirds of institutions employed epilepsy-specific protocols, while others relied on routine brain MRI protocols. While MRI plays an important role in evaluating pediatric seizures, its use should be guided by clinical context. The Choosing Wisely initiative recommends against neuroimaging for simple febrile seizures and for children older than 6 months with an unprovoked, generalized seizure who have returned to baseline. 18
All epilepsy-specific and dedicated new-onset seizure MRI protocols included 3D T1 MPRAGE sequences, often accompanied by high-resolution coronal imaging oriented to the temporal lobes, FLAIR, DWI, and T2 weighted sequences. These shared features align with current recommendations from the Neuroimaging Task Force and International League Against Epilepsy (ILAE), which recommend the harmonized neuroimaging of epilepsy structural sequences (HARNESS-MRI).19,20 The HARNESS-MRI protocol includes high-resolution 3D T1-weighted, high-resolution 3D FLAIR, and high in-plane 2D coronal T2-weighted MRI. 20 The European Society of Neuroradiology and American Academy of Neurology also recommend the HARNESS-MRI protocol, underscoring international alignment.18,21 The European Society of Neuroradiology further recommends the inclusion of susceptibility-weighted imaging (SWI) or GRE sequences to identify chronic hemosiderin depositions, small vascular malformations, or calcified granulomas. 21 Variability in additional sequences and the use of routine brain MRIs rather than epilepsy-specific protocols highlight the absence of national standardization. A prior European survey similarly found substantial heterogenicity across centers. 22
It is important to distinguish between MRI protocols aimed at excluding structural causes of seizures (eg, mass lesions or TSC), and those optimized for epilepsy surgical planning. While both rely on high-resolution neuroimaging, the latter often requires more advanced protocols to detect subtle cortical malformations and common causes of drug-resistant epilepsy, such as focal cortical dysplasia. 23
Age ranges for sedated MRI also varied widely, with a median lower age limit of 3 months and median upper age limit of 6 years. Such variation in sedation practices is well documented and likely reflects a complex interplay of institutional resources, provider expertise, technologist comfort, and patient-level factors.24,25 The Canadian Paediatric Society encourages standardized patient selection, provider training, monitoring, and institutional protocols for procedural sedation in children; however, national standardization remains lacking. 26
For non-sedated MRIs, most centers reported using feed-and-swaddle methods for infants, and some used natural sleep techniques for infants and young children. The feed-and-swaddle approach has previously demonstrated over 90% success in infants up to 6 months of age without complications and is therefore a suitable method to include in standardized non-sedated MRI protocols for this age group. 27
Non-pharmacologic distraction techniques such as child life support and video goggles were commonly used for older children, reflecting a shift toward minimizing sedation through behavioral strategies, which is well supported by previous studies.28,29 Additionally, virtual reality remains underutilized despite evidence supporting its role in reducing anxiety and improving cooperation during MRI.30,31
Even with increasing evidence supporting fast MRI protocols to shorten scan times and reduce sedation, only 2 centers reported their use.28,32,33 Adoption of fast MRI protocols is limited by scanner availability, resource constraints, and administrative barriers such as reimbursement and training requirements. 34 Although fast MRI protocols have demonstrated sufficient image quality, radiologists may also have concerns that abbreviated protocols could reduce diagnostic confidence for subtle epileptogenic lesions.33,35
A notable gap exists for children between approximately 5 months and 2 years of age, where non-sedation techniques are less effective. This finding aligns with previous literature, noting a lack of effective strategies for patients around the age of 1. 25 While the feed-and-swaddle method is widely used for infants up to a median age of 5 months, video goggles and virtual reality interventions predominantly start around 2 years of age or older, leaving this intermediate age group with limited non-sedated options. Fast MRI protocols offer a promising alternative by reducing scan times and improve the feasibility of non-sedated imaging in infants and toddlers. Additionally, broader implementation and evaluation of the natural sleep technique in younger age groups may further reduce sedation use, as prior studies have demonstrated a 62% success rate for natural sleep MRIs in children aged 1 to <2 years. 28
There are several limitations to our study. We did not collect data on diagnostic accuracy, image quality, patient-centered outcomes, or resource availability (eg, child life or VR access). Future studies should address these factors to optimize and standardize sedation-free imaging protocols for new-onset pediatric seizures. Such work will be essential to guide evidence-based guidelines, improve diagnostic consistency, and reduce sedation-related risks in children undergoing neuroimaging for new-onset seizures. Finally, as only one radiologist per institution completed the survey, responses may reflect individual rather than institutional practices. Although we cannot exclude heterogeneity within institutions, our survey questions were phrased to inquire about departmental and institutional protocols, and most respondents were senior radiologists familiar with departmental protocols including first-time seizure and epilepsy protocols.
Conclusion
In summary, our national survey of Canadian pediatric radiology departments revealed considerable variability in MRI protocols and sedation practices for children with new-onset seizures. While MRI is universally employed and many centers incorporate key HARNESS-MRI elements, only a minority utilize dedicated protocols. Sedation thresholds and non-pharmacologic strategies also varied, with limited adoption of fast MRIs. This heterogeneity likely reflects the lack of high-level evidence comparing the accuracy of different imaging protocols. A proposed national guideline would serve as a suggestion to guide practice, and could help improve diagnostic consistency, optimize sedation use, and support care for pediatric patients with new-onset seizures.
Supplemental Material
sj-pdf-1-caj-10.1177_08465371251398361 – Supplemental material for Imaging Methods for New-Onset Seizures at Canadian Pediatric Hospitals
Supplemental material, sj-pdf-1-caj-10.1177_08465371251398361 for Imaging Methods for New-Onset Seizures at Canadian Pediatric Hospitals by Kate Hurley, Ellen Song, Lucia Carpineta, Jean-Claude Décarie, Marc Elliott, Jonathan Grynspan, Naeem Khan, Pradeep Krishnan, Johanna Ortiz Jimenez, Yves Patenaude, Katya Rozovsky, Nina Stein, Makabongwe Tshuma, Xing-Chang Wei, Daniela Pohl and Elka Miller in Canadian Association of Radiologists Journal
Footnotes
Acknowledgements
Dr. Natasha Ferguson and Dr. Michael Jurkiewicz passed away during the preparation of this manuscript. We honor their invaluable contributions to this work and to the field.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to acknowledge the support of the Canada Summer Jobs Program, administered by Employment and Social Development Canada/Service Canada, which provided funding for a summer medical student to contribute to this project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
