Abstract
Purpose:
To correlate parathyroid scintigraphy results with patient biochemistry, surgery, and pathology to inform appropriateness criteria and assess biochemical metrics in the imaging workup of hyperparathyroidism.
Methods:
This retrospective study included 421 patients who underwent parathyroid scintigraphy. Patients were grouped based on primary versus secondary hyperparathyroidism, and clinical profiles were reviewed for scan result, blood work, surgical results, and pathology. Performance metrics of scintigraphy were analyzed. Demographics, bloodwork, and location were compared between positive and negative scans. Predictors of positive scans were identified by multivariate logistic regression analysis. The performance of biochemistry to predict scan results was evaluated by ROC analyses.
Results:
Positive tests—occurring in 52% of patients—were associated with higher parathyroid hormone (PTH) and corrected calcium. However, PTH was only predictive of a positive test in patients with secondary hyperparathyroidism. On multivariate analysis, male sex, corrected calcium, and younger age were predictors of a positive scan. Corrected calcium was the most predictive with an OR of 1.28 for every 0.1 mmol/L increase. Based on ROC analysis, corrected calcium had an AUC of 0.628 and a cutoff of 2.65 mmol/L maximized sensitivity (88%) and specificity (35%) for a positive test.
Conclusion:
In this large retrospective cohort, several biochemical metrics, including corrected calcium levels, were predictive of a positive scintigraphy study. Furthermore, biochemistry, including PTH levels, significantly differed between primary and secondary hyperparathyroidism suggesting that tailored biochemical metrics are required. This work sets a foundation for the development of a robust biochemical scoring system to optimize patient selection for parathyroid imaging.
Introduction
The parathyroid glands, typically 4 in number, are embedded in the posterior quadrants of the thyroid gland. Occasionally, ectopic parathyroid glands can be present elsewhere in the neck and chest. These glands produce and release parathyroid hormone (PTH) into the bloodstream, which plays an important role in regulating calcium levels and mineral content in the skeleton. Increased PTH, seen with hyperparathyroidism, can cause electrolyte disturbances—primarily hypercalcemia and relatively low phosphorus—and associated symptoms/morbidity. Classically, patients present with fatigue, constipation, kidney stones, bone pain, and other nonspecific symptoms. In the modern day of frequent and accessible blood work, asymptomatic and normocalcemic patients with hyperparathyroidism may be identified incidentally. For clinically complex cases, relatively new clinical metrics such as a calcium/phosphorus ratio1,2 or “parathyroid function index” (Ca/P × PTH) 3 have been described to accurately diagnose patients with subclinical disease.
In nearly all cases of primary hyperparathyroidism, the cause is a parathyroid adenoma, which is a hyperfunctioning benign tumor of the parathyroid gland that autonomously secretes PTH. Secondary hyperparathyroidism occurs when excess PTH is released in the setting of hyperplasia of one or more parathyroid glands in response to an independent stimulus. In most cases, secondary hyperparathyroidism occurs in the setting of kidney failure due to the associated impaired absorption of calcium from the intestinal tract. Tertiary hyperparathyroidism occurs in the setting of longstanding secondary hyperparathyroidism when hypertrophied glands function autonomously despite treatment or removal of the original stimulus. Although hyperparathyroidism can be treated expectantly and medically, definitive curative treatment is surgical. Consensus guidelines for surgical patient selection were most recently updated in 2016 and, among numerous indications, include any patient who is symptomatic or under the age of 50. 4 In cases considered for surgery, pre-operative localization of a culprit abnormal parathyroid gland can allow for a minimally invasive parathyroidectomy (ie, local neck dissection) which limits the extent of surgery, thereby reducing potential complications while maintaining similar cure rates to bilateral neck exploration. 5 Accurate pre-operative localization of diseased parathyroid glands is crucial for this approach.
Nuclear medicine parathyroid scintigraphy is a common and well-established method for identifying and localizing parathyroid adenomas; a 2012 meta-analysis demonstrated an overall sensitivity of 78.9% and a positive predictive value of 90.7%. 6 In recent years, fused SPECT/CT imaging has become a mainstay, and this technique further improves localization with some groups reporting >90% sensitivity. 7 Other imaging techniques such as 4D-CT, requiring multiphase iodine contrast-enhanced series, and targeted neck ultrasound are also available for the localization of parathyroid adenomas. In practice, multiple tests are often employed. Reports vary on which is the most sensitive modality, with scintigraphy or CT typically demonstrating high accuracy.7-9 Scintigraphy has the unique benefit of highlighting ectopic parathyroid glands that may be outside the field of view with anatomic imaging techniques focused on the neck. 7 Additionally, there is minimal concern for renal failure and contrast allergies which may preclude some patients from contrast-enhanced CT. Nevertheless, parathyroid scintigraphy requires intravenous access, several hours of patient and hospital resources, and ionizing radiation to the patient. Thus, it is important to optimize patient selection for those who may benefit the most from this imaging study. There is a general paucity of guidelines for the application of parathyroid scintigraphy.
Several groups have studied the relationship between patient factors (such as limited biochemistry) and parathyroid scintigraphy performance, but sample sizes have often been low and results have varied. In general, positive scans are correlated with high PTH,10-17 high total corrected calcium,12,15,17,18 as well as increased adenoma size16,18,19 and weight.16,20 Decreased sensitivity has been found to correlate with low PTH,7,21,22 low total corrected calcium, 9 small adenoma size,7,8,23 and multi-gland disease.8,9,23 A single group correlated high ionized calcium and low phosphorus with positive scans. 13 On the contrary, numerous studies have found no correlation between scan performance and total corrected calcium7,10,11,13,22,23 or phosphorus.7,10,13,17,23 Several other parathyroid-related biochemical variables have also been studied and revealed no correlation to scan performance including Vitamin D,7,10,13,16,17 alkaline phosphatase,7,13,16,17,23 creatinine,7,16,23 and 24-hour urine calcium.7,13,16 The correlation of scan performance with advanced diagnostic metrics for hyperparathyroidism (ie, Ca/P ratio or parathyroid function index) has not been investigated. Thus, in routine clinical practice, it is unclear how the biochemical workup of hyperparathyroidism relates to parathyroid scintigraphy performance metrics.
In this study, parathyroid scintigraphy performance was assessed in relation to patient biochemistry, surgical findings, and pathology to further elucidate metrics predictive of positive imaging and help inform improved appropriateness criteria for the imaging workup of hyperparathyroidism.
Methods
Study Design and Patient Selection
A single-center retrospective review was performed on sequential patients who underwent parathyroid scintigraphy at our tertiary care academic institution between 2016 and 2020. This retrospective timeframe was chosen to allow a sufficient timeframe for any potential surgical intervention and pathological results to be available for the assessed cohort. An exemption from research ethics board review was granted due to the quality improvement nature of this study. Patients were identified through our local PACS which would retrieve all parathyroid scintigraphy studies performed at our center over the study period. Main inclusion criteria were the availability of the imaging requisition and finalized report. For patients with multiple studies, only the most recent scan was included in the main analysis, as this would be the best correlate for associated biochemistry and subsequent surgical/pathology results.
Parathyroid Scintigraphy
Parathyroid studies were all performed on a BrightView SPECT-CT system (Phillips, Amsterdam). Briefly, approximately 1000 MBq of 99mTc-Sestamibi was administered intravenously with SPECT-CT imaging at immediate and delayed (3-hour) time points. All studies were clinically interpreted and reported by specialty-trained nuclear medicine physicians. Original reports were reviewed to determine the result and location of imaging abnormalities. Consensus review of the original report (2 authors—one with 3 years experience and one nuclear medicine fellow) was performed of any cases that had indefinite wording of conclusions to classify them as either negative or positive. This review was blinded to the associated biochemical and pathology results. Original images were not reinterpreted as part of this study. With this approach, only one indeterminately worded study was identified and excluded.
Data Collection
The nuclear medicine study report and patient electronic medical records were reviewed to collect demographic information (age and sex), type of hyperparathyroidism (primary or secondary, based on diagnosis by referring physician), bloodwork, operative notes, and pathology reports. The laboratory values collected were serum parathyroid hormone (PTH), calcium, albumin, phosphorus, and vitamin D. Vitamin D levels were ultimately not included as there were too few observations (n = 232, 52% of patients). Missing data for the other biochemical markers were not imputed; statistical analyses were performed based on available data, and missing data is reflected in provided sample size for each analysis. The most recent laboratory values up to 6 months before parathyroid scintigraphy were used; if not available, then bloodwork from up to 2 months after the scan was used, provided there was no interval parathyroidectomy. Corrected calcium was calculated using the formula: corrected calcium = serum calcium + (0.028 × (40 − serum albumin)). The calcium to phosphorus ratio (Ca/P)1,2 and parathyroid function index (Ca/P × PTH) 3 were also calculated.
Among the patients who had parathyroidectomies, surgical and pathology reports were reviewed for histopathological diagnosis, location of abnormal parathyroid glands, and (if applicable) adenoma weight. True positives were defined as either an adenoma or parathyroid hyperplasia on the pathology report. Cases of parathyroid malignancy were included in the overall cohort but not for diagnostic performance analyses. Pathology results that were indeterminate/inconclusive were categorized as normal. Location of abnormal parathyroid glands was compared between scintigraphy and surgical/pathology reports, in terms of laterality (right vs left) and position (superior, inferior, or ectopic). Concordance of both laterality and position, and laterality only was determined. For multifocal disease, a scan was considered concordant if at least 2 sites of involvement were concordant.
Statistical Analyses
Data analysis was performed using R (version 4.4.2, R Core Team 2021) and GraphPad Prism 6 (GraphPad Software, Boston, Massachusetts USA). Graphs were generated using GraphPad Prism and CorelDraw X6 (Alludo, Ottawa, Canada).
Patient demographics and laboratory values were compared between positive and negative scans using univariate analyses: either unpaired t-tests or Mann Whitney U tests for continuous data depending on data normality and either Fisher’s exact tests or Chi Square tests for categorical data depending on the size of the matrix. Univariate analyses were also used to test for heterogeneity between patients with primary and secondary hyperparathyroidism. Potential interactions between type of hyperparathyroidism, sex, age, and laboratory data were assessed by Two-way ANOVA. Two multivariate logistic regression analyses were performed using the lme4 package 24 to determine what factors were associated with a positive scan and with proceeding to surgery. The units of calcium and phosphorus used in regression models were increments of 0.1 mmol/L and PTH units were increments of 10 pmol/L, since the scale of these units have more physiological relevance.
ROC curves were plotted for calcium, phosphorus, PTH, Ca/P, and Ca/P × PTH, and the outcome of the parathyroid exam. Maximum sensitivity and specificity of calcium to classify positive and negative tests was determined using the R package Cutpointr 25 to determine optimal calcium level cutoff.
Data are presented as mean and standard deviation unless otherwise indicated. A P-value of <.05 was considered significant.
Results
In the 5-year study period, 422 patients underwent parathyroid scintigraphy at our institution; one patient was excluded due to an indeterminate scan result (Figure 1). Characteristics of the overall cohort are presented in Table 1. Of the 421 included studies, 52% were positive and patients with positive tests had significantly higher PTH (P = .032), corrected serum calcium (P < .001), calcium/phosphorus ratio (P = .002), and parathyroid function index (P < .001). Serum phosphorus was not significantly different between the groups. There were fewer tests performed in males (32.5%), but the proportion of males with positive tests was greater than females (59.1% vs 47.9%; P = .037). Patients with positive scans were more likely to undergo parathyroidectomy (47.0% with positive scan, 19.1% with negative scan; P < .001).
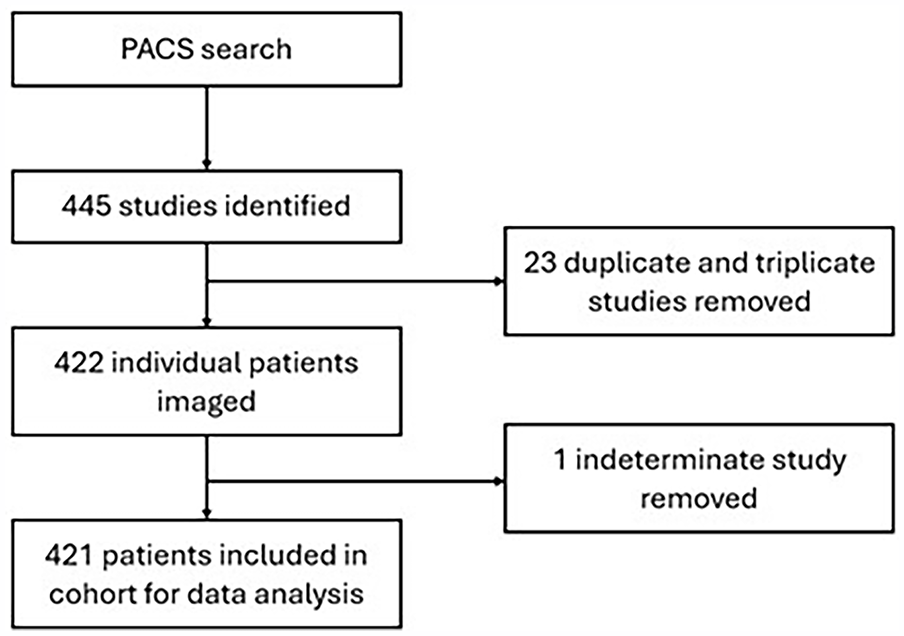
Flow chart depicting patient identification for inclusion in the study cohort.
Descriptive Characteristics of the Study Cohort Compared by Parathyroid Scintigraphy Result. Means Are Provided With Standard Deviation.
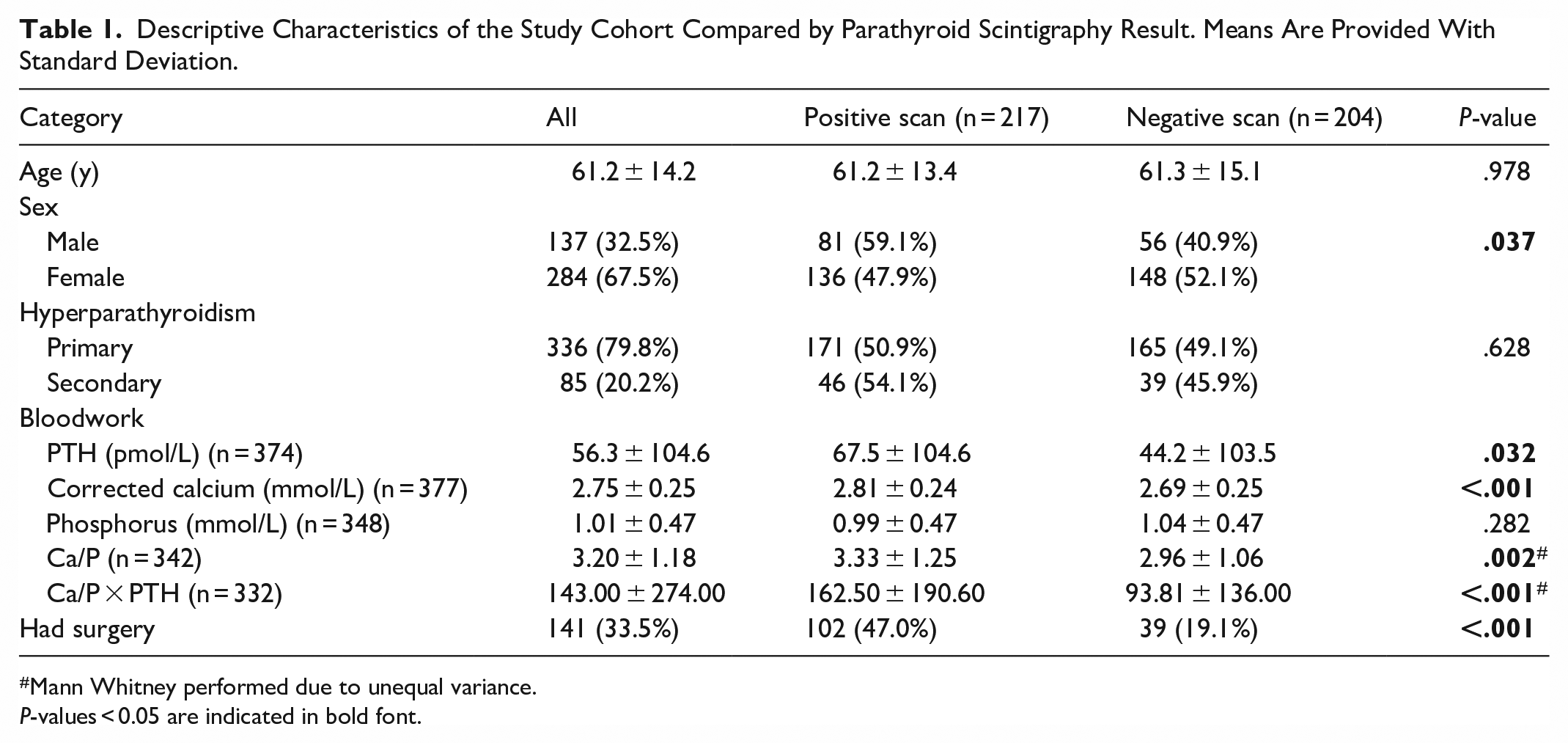
Mann Whitney performed due to unequal variance.
P-values < 0.05 are indicated in bold font.
Over the 5-year study period, 22 patients had multiple scans, including one patient who underwent 3 studies. The average interval between scans was 747 days ± 396 days. Fourteen patients had unchanged results, 5 positive and 9 negative. Five patients (22.7%) initially had negative scans before a subsequent positive result; 4 of these patients had surgery, and all of them had pathology-proven adenomas. Three patients (13.6%) with positive studies had subsequent negative scans and none of these patients went on to have surgery.
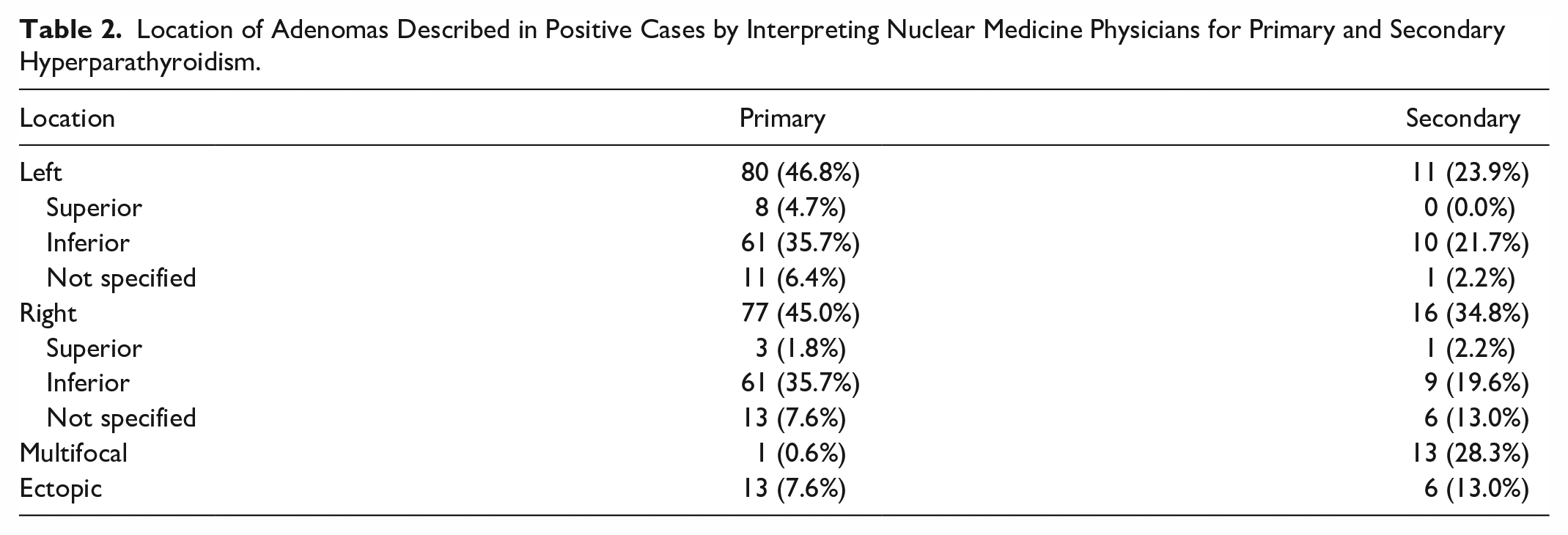
Of the 171 positive scans in patients with primary hyperparathyroidism, 46.8% and 45.0% of adenomas were identified in the left and right neck, respectively; 7.6% of abnormal glands were ectopically located; and one case demonstrated multifocal disease (Table 2). When the precise location of a non-ectopically located single adenoma was specified by the interpreting nuclear medicine physician (n = 135), 91.9% of adenomas were at the inferior aspect of the thyroid gland compared to 8.1% located superiorly. For positive scans in secondary hyperparathyroidism (n = 46), 23.9% and 34.8% of adenomas were in the left and right neck, respectively. Multifocal abnormalities were present in 28.3%, and 13.0% demonstrated an ectopic location.
Location of Adenomas Described in Positive Cases by Interpreting Nuclear Medicine Physicians for Primary and Secondary Hyperparathyroidism.
While there was no significant difference in the proportion of positive scans between patients with primary and secondary hyperparathyroidism, given the different underlying pathologies, a series of univariate analyses was performed to test for heterogeneity between these groups (Table 3) prior to building a predictive model. Those with suspected primary hyperparathyroidism (n = 336) were older, more likely to be female, were less likely to have subsequent surgery, had lower serum PTH and phosphorus, and had higher corrected serum calcium (all P-values <.001). Additionally, there were several interactions between these individual variables, type of hyperparathyroidism, and a positive test result (Figure 2). For patients with primary hyperparathyroidism, only increased corrected serum calcium was associated with a positive scan (P < .0001), while age and serum PTH were not. In contrast, for patients with secondary hyperparathyroidism, older age (P = .0242), higher serum PTH (P = .0012), and higher corrected serum calcium (P = .0226) were associated with a positive scan. Patients with secondary hyperparathyroidism were further complicated by an interaction between age, sex, and a positive test result as age was only predictive of a positive test result in men (Negative test: 45.8 ± 12.8 years old vs Positive test: 58.4 ± 12.8 years old, P = .0085). A specific age cut-off for this predictive effect could not be calculated based on this sample.
Descriptive Characteristics of the Study Cohort Compared by the Etiology of Hyperparathyroidism. Means Are Provided With Standard Deviation.
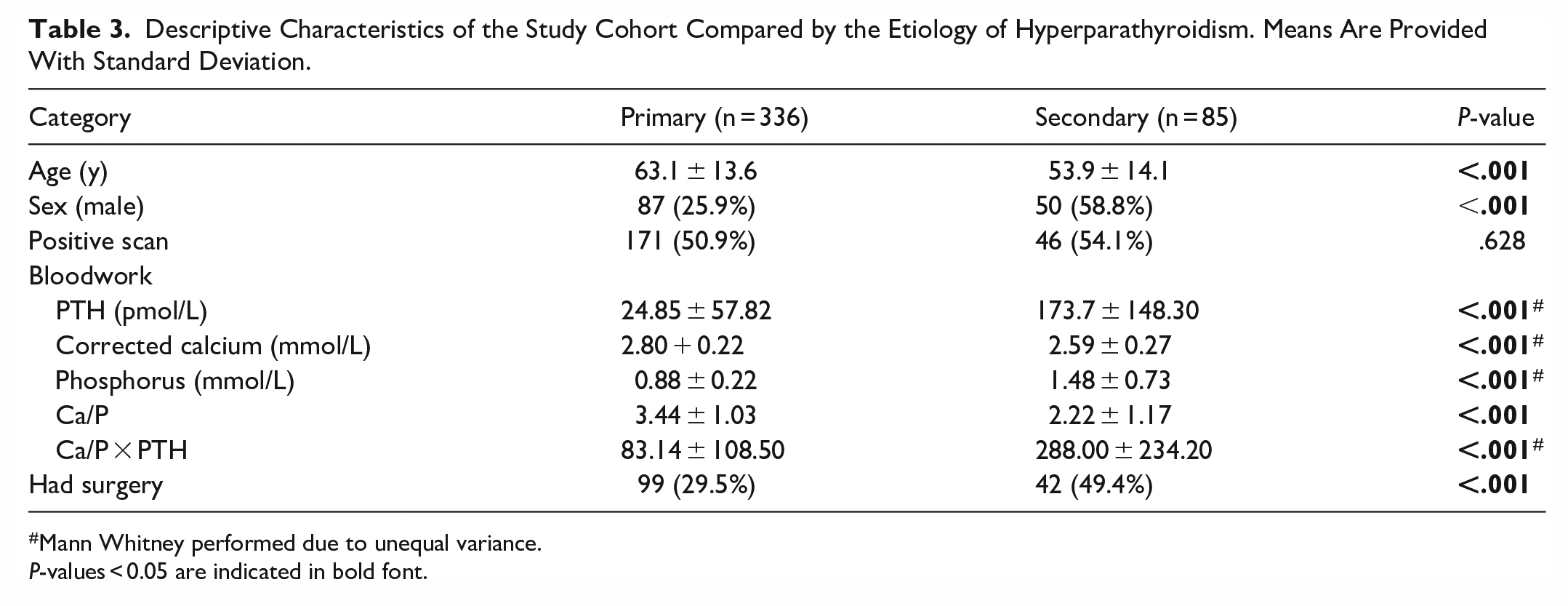
Mann Whitney performed due to unequal variance.
P-values < 0.05 are indicated in bold font.
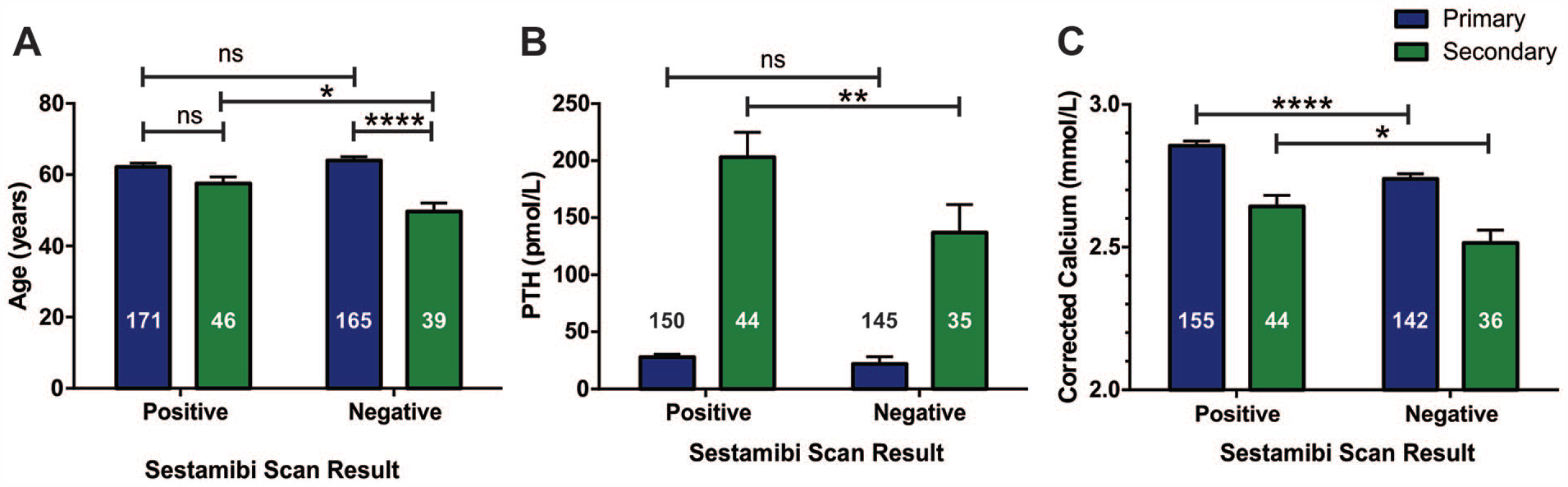
Association of age (A), serum PTH (B), and corrected serum calcium (C) on sestamibi scan result depending on type of hyperparathyroidism.
Given the complexity of the interactions of biochemical data in patients with secondary hyperparathyroidism and the lower relative sample size (n = 85, 20% of total patients), the model to predict a positive parathyroid scan only used patients with primary hyperparathyroidism. Controlling for covariates with a multivariate logistic regression model, increased corrected serum calcium (P < .001), younger age (P = .023), and male sex (P = .031) were significant predictors of a positive scan (Figure 3). Calcium was the most predictive independent variable with an OR of 1.31 for every 0.1 mmol/L increase in value.
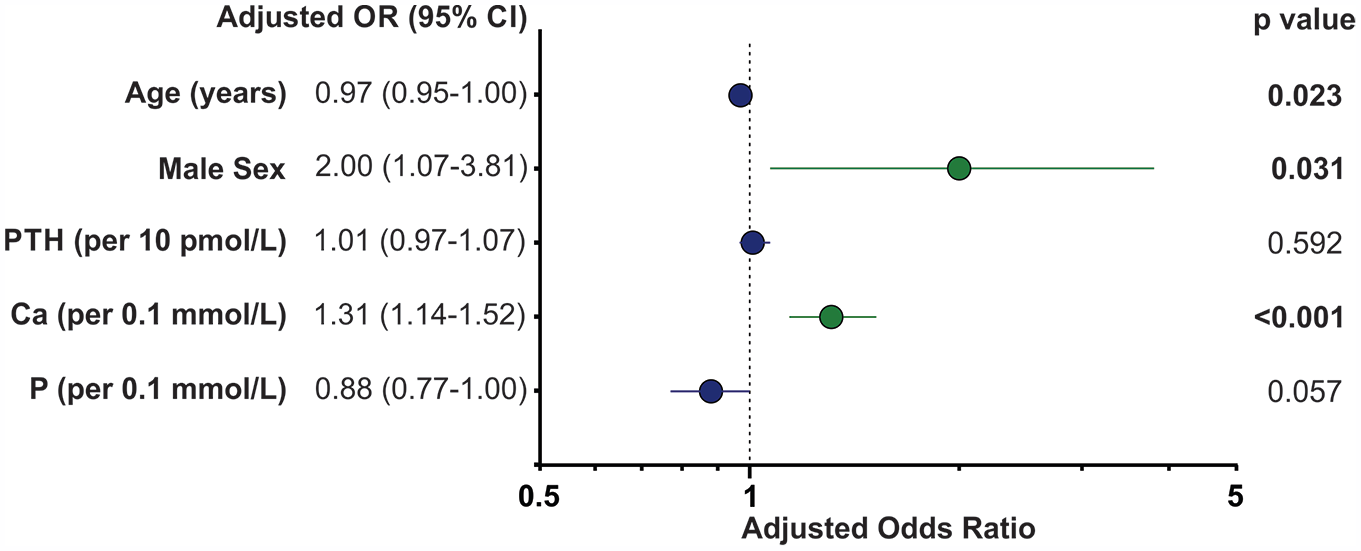
Multivariate logistic regression of predictors of positive scans in patients with primary hyperparathyroidism.
Given that corrected serum calcium was the most significant variable in the regression, an ROC was determined for discrimination between positive and negative tests based on corrected serum calcium in patients with primary hyperparathyroidism (AUC 0.644, 95% CI 0.581-0.706, P < .0001; Figure 4A). The maximal summed sensitivity and specificity for the prediction of a positive test in patients with primary hyperparathyroidism was at a cutoff of 2.65 mmol/L corresponding to a sensitivity of 88.4% and a specificity of 35.2%. Additional ROCs were plotted for all biochemical metrics (Figure 4B). The AUCs for each metric were: PTH 0.705 (95% CI 0.646-0.764), corrected serum calcium 0.644 (95% CI 0.581-0.706), serum phosphorus 0.629 (95% CI 0.562-0.695), calcium/phosphorus ratio 0.649 (95% CI 0.583-0.715), and parathyroid function index 0.723 (95% CI 0.661-0.785). There was no statistically significant difference between these curves.
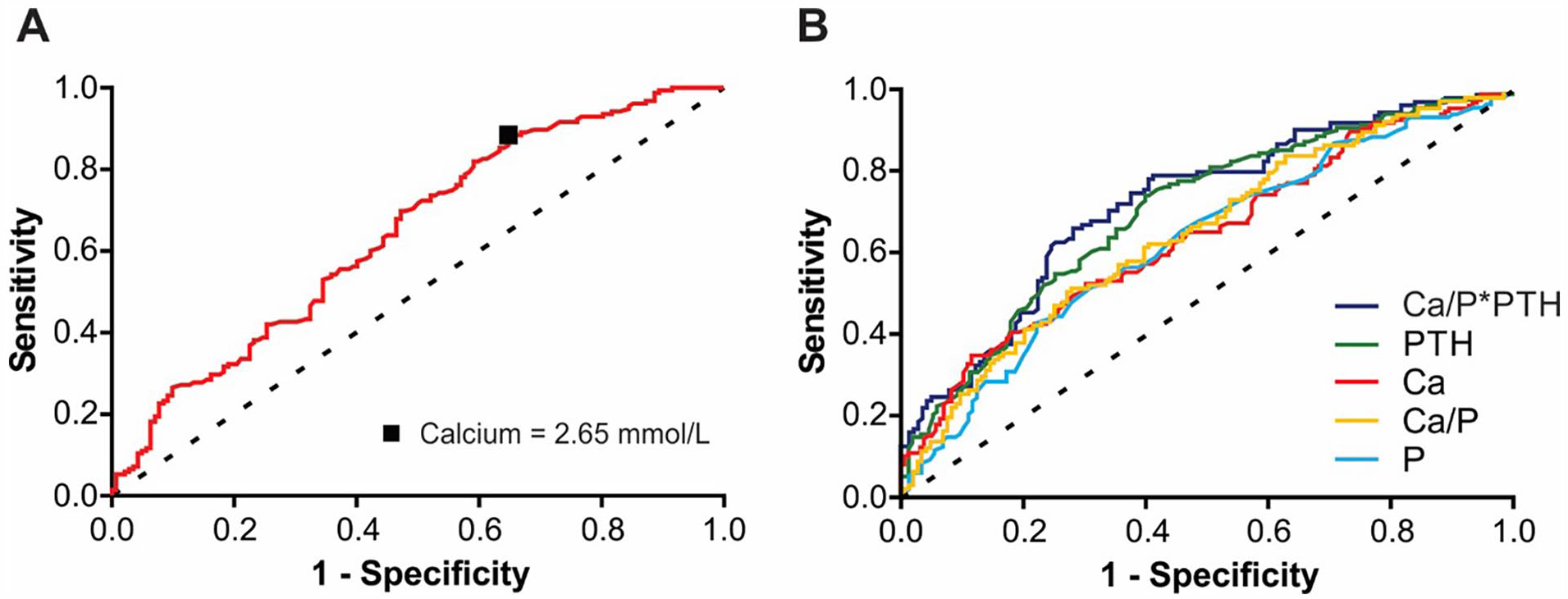
Diagnostic performance of biochemical metrics to classify positive and negative scans in patients with primary hyperparathyroidism. (A) Corrected serum calcium ROC with a plotted maximal sum of sensitivity and specificity cut-off. (B) All biochemical metrics with the legend displaying metrics in order of decreasing AUC.
Logistic regression was performed to determine factors associated with patients proceeding to surgery. Parathyroidectomy was most strongly predicted by having a positive parathyroid scan (OR 3.55) and also by having secondary hyperparathyroidism, increased corrected calcium, and younger age (Figure 5).
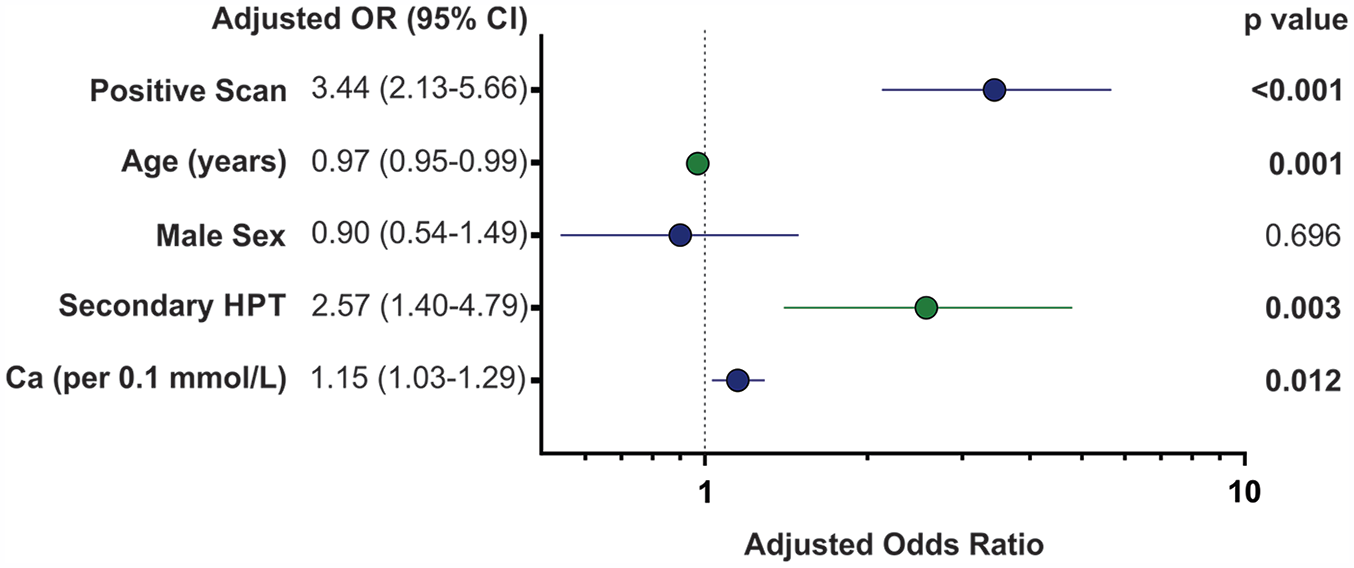
Multivariate logistic regression of predictors of patients who underwent parathyroidectomies.
Evaluating the concordance between scan result and pathology in the patients that had surgery (Table 4), there were no false positive scans; specificity and positive predictive value were 100%. Sensitivity was 58.8% when comparing the radiologically described location of an atypical gland and the surgical note described location. This improved to 65.6% if scoring only the reported laterality (ie, ignoring superior or inferior descriptors) and finally increased to 77.1% for any location. The overall negative predictive value for scintigraphy was 21.1%. Subdividing cases with positive pathology by their final diagnosis (adenoma or hyperplasia; Table 4), the rate of positive scans was similar, but adenomas were correctly localized at a significantly higher rate than hyperplasia (66.7% vs 37.8%; P = .003). This improved to 76.3% versus 37.8% when scoring only for described laterality. In patients with pathology-proven adenomas, adenoma weight was not significantly different between those with true positive and false negative scans (Positive test: 1.534 ± 2.363 vs Negative test: 1.845 ± 2.259, P = .6681).
Concordance of Scan and Pathology Results With Subdivisions by Pathology Diagnosis. Scan Result Was Considered Concordant for Patients With Multifocal Disease if ≥50% of Abnormal Glands Were Identified.
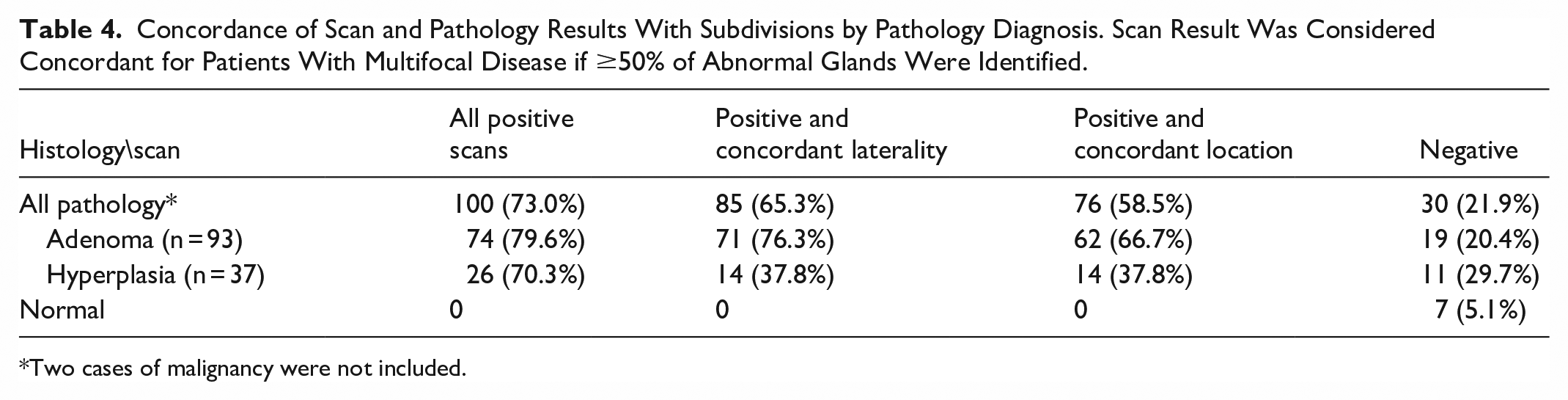
Two cases of malignancy were not included.
Discussion
Diagnostic Performance
Our overall scintigraphic sensitivity for the detection of pathology-proven abnormal parathyroid glands of 77.1% is similar to a 2012 meta-analysis. 6 Scintigraphy demonstrated a high positive predictive value (100%) and therefore positive studies can confidently advance to surgical management. However, as 48% of studies were negative with low negative predictive value (21.1%), a negative scan does not exclude a parathyroid adenoma or hyperplasia. In such cases, other imaging modalities such as 4D-CT and neck ultrasound may have additive value for localization.
Throughout our study period, 22 patients underwent repeat imaging with scintigraphy, and 36% of patients with an initial negative study subsequently had a positive result, with the average interval between scans being greater than 2 years. Other groups have demonstrated comparable conversion rates to positive scans of 28% (n = 39) to 41% (n = 49).26,27 This supports repeat scintigraphy in patients with failed localization who have had a delay in undergoing surgery and are unsuitable for conservative management. Unfortunately, given the small patient numbers in this cohort, dedicated analyses of biochemical differences between time points could not be performed. This indicates that there is likely a progressive sensitivity of parathyroid adenoma scintigraphic visualization and that the timing of a scan is important for diagnostic performance.
In a minority of cases (3.3%), the laterality of a positive scan did not match the laterally of a pathology-proven adenoma, which supports the continued practice of intra-operative PTH monitoring and bilateral neck exploration as needed.
Biochemical Metrics for Patient Selection for Scintigraphy
Corrected calcium was the most predictive independent marker for a positive scan in patients with primary hyperparathyroidism and yielded a maximal summed sensitivity and specificity of 88.4% and 35.2% at a cutoff of 2.65 mmol/L. If sensitivity alone was prioritized to avoid low-yield exams, a cutoff of 2.1 mmol/L would capture 97% to 100% of positive scans, but this is below the normal range for calcium (2.20-2.60 mmol/L) and thus not clinically applicable. A cutoff of 2.4 mmol/L corresponds to a sensitivity of 92% to 98% which could be helpful for decision-making in equivocal cases in this patient population.
Although increased PTH was associated with a positive scan in the overall cohort, this metric was not predictive when accounting for patients who had primary versus secondary hyperparathyroidism. This aligns with a recent Canadian publication noting abnormal parathyroid scans in patients with normal and minimally elevated PTH. 28 A prospective study 15 correlated high corrected calcium and PTH with positive studies and recommended against parathyroid scintigraphy if the serum calcium was below 2.51 mmol/L but could not identify a lower limit of PTH that would predict a negative scan. A recent retrospective study 17 suggested cutoff values of 2.80 mmol/L for calcium and 125 pg/mL for PTH (equivalent to 13.3 pmol/L) above which patients may be more likely to have positive imaging, but this calcium cutoff was not statistically significant and >60% of patients below either value still had positive scintigraphic localization. It is clear the reliance solely on PTH levels is inadequate for appropriate referral for parathyroid scintigraphy.
Importantly, since our study cohort included only those with known hyperparathyroidism and typically abnormal biochemistry, extrapolation of these results and cutoffs to patients who do not have a clinical diagnosis of hyperparathyroidism is not valid.
To the best of our knowledge, this is the first study to correlate parathyroid scintigraphy results with several advanced biochemical metrics implemented in clinical workup to aid in the diagnosis of hyperparathyroidism. Parathyroid function index and calcium/phosphorus ratio were higher in patients with positive scans, but neither yielded a superior clinical cutoff for patient selection for scintigraphy in this cohort. Thus, a single biochemical metric is not optimal for the selection of patients for parathyroid imaging.
Demographic and Clinical Considerations
Patients with both primary and secondary hyperparathyroidism experience high circulating PTH, however the underlying disease process and pathophysiology are unique. Parathyroid scintigraphy is typically discussed in the context of identifying adenomas, but guidelines do include its use in secondary hyperparathyroidism—especially for the identification of ectopic parathyroid glands. 29 Within this context, hyperplastic glands are typically smaller than adenomas and sensitivity is relatively decreased. 30 Parathyroid hyperplasia does not preclude patients from having superimposed adenomas, but most patients in our cohort with secondary hyperparathyroidism had 4-gland hyperplasia (necessitating bilateral neck dissections). Scintigraphy had a lower performance in this group with a sensitivity of just 37.8% in identifying ≥50% of pathology-proven hyperplastic parathyroid glands. Thus, there is decreased utility of parathyroid scintigraphy to identify all sites of pathology in secondary hyperparathyroidism.
In patients with suspected secondary hyperparathyroidism, those with negative scans were significantly younger than those with positive scans; this trend was not seen in the primary hyperparathyroidism group. This raises the possibility of a fundamental difference in hyperplastic parathyroid glands as patients age. Alternatively, this could reflect patient selection with younger patients receiving more timely and intensive care earlier in the disease process when abnormal glands are below the resolution of scintigraphy.
Male sex was predictive of positive scans in our cohort and this is of unknown significance. Again, there may be a fundamental sex difference in parathyroid physiology and/or disease processes that impacts radiopharmaceutical uptake. Alternatively, this could also simply relate to sample selection in our cohort of those being considered for surgery. It is possible that females are more likely to present early (or in general) with symptoms, biochemical disturbance, or surgical criteria leading to imaging earlier in the disease process.
Surgical Considerations and Location of Adenomas
In the workup of hyperparathyroidism, the role of imaging, including scintigraphy, is to direct surgery for patients who have met the criteria for parathyroidectomy. Consensus guidelines from 2016 arrive at this decision based on age, symptoms, biochemistry, bone mineral density, history of vertebral body fractures, and the presence of kidney stones. 4 Notably, positive scintigraphy or other parathyroid imaging modalities are not a criterion to identify surgical candidates and rather exclusively aid in localization. However, having a positive scan was the most predictive variable for patients having surgery in our cohort, suggesting that it may play a role in the decision to pursue surgery or that patients are being inappropriately referred for scintigraphy. A future avenue of research would be to correlate scan results with a wider array of clinical factors included in the criteria for surgical candidacy.
The pre-operative localization of parathyroid adenomas is necessary to allow a minimally invasive parathyroidectomy (unilateral neck dissection) which limits patient morbidity. For patients in this cohort, scintigraphy identified the location of culprit adenomas 66.7% of the time and successfully directed the laterality of the surgery in 76.3% of cases. The discrepancy between these values may relate to the different approach in which imaging and surgical descriptions localize superior and inferior glands. Whereas imaging assessment often describes the location of an abnormal parathyroid gland in relation to the adjacent thyroid gland, a surgical description could include relation of the parathyroid glands based on their embryological descent and subsequent position relative to the juxtaposition of the inferior thyroid artery and the recurrent laryngeal nerve—structures below the resolution of typical anatomic imaging assessment. It has previously been reported that this difference can result in embryologically “superior” glands being mislabeled scintigraphically as “inferior.” 31 In this study, when specifying adenomas as “superior” or “inferior,” 92% of glands were described as inferior. A predilection for adenomas favoring the inferior glands is described, 32 but not to this extent. Superior and inferior resections require different surgical approaches and may impact patient positioning and initial incisions. More accurate methods to differentiate superior and inferior glands on imaging beyond the relationship to the thyroid gland have been validated, 33 which could lead to optimized surgical approaches.
Limitations
There are several limitations to the current study. Only hyperparathyroid patients referred for scintigraphy were included in our analysis, which potentially introduces selection bias for biochemical metrics related to hyperparathyroidism. Therefore, our predictive model only applies to patient with suspected or known hyperparathyroidism. Additionally, this population may represent more symptomatic presentations being considered for surgery.
Our sample size and power were reduced as there was a lack of consistent biochemical workup in patients and limited which biochemical markers could be assess—less than 50% of our study patients had serum Vitamin D measurements. Factors that could influence biochemistry were not assessed, such as Vitamin D supplements or calcimimetics.
As this was a retrospective study, prospective validation studies will be required to better implement biochemical metrics into the routine clinical selection of patients for parathyroid imaging. Finally, a clinical scoring system with certain weightings on biochemical metrics (such as Ca and PTH) and demographics (age, sex) predictive of positive parathyroid imaging workup would be incredibly impactful. Future work involving a large cohort initiative, preferably multi-institutional, is needed to develop such a robust scoring system.
Conclusions
In this large retrospective cohort, several biochemical metrics, including corrected calcium levels, were predictive of a positive imaging study. However, other metrics, such as PTH levels, were not predictive and significantly differed between primary and secondary hyperparathyroidism, suggesting that a tailored biochemical metric is required for selecting hyperparathyroidism patients for imaging workup. This study highlights the deficiency and provides a foundation for the development of predictive demographic and biochemical metrics to optimize patient selection for advanced imaging in the setting of hyperparathyroidism.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
