Abstract
The Canadian Association of Radiologists Incidental Findings Working Group (CAR IFWG) has developed new recommendations for the management of incidental findings of the spleen, lymph nodes, peritoneum, and mesentery, tailored to the Canadian healthcare context. This guidance addresses splenomegaly, focal splenic lesions, splenic artery aneurysms, lymphadenopathy, mesenteric panniculitis, and peritoneal nodules. Building on prior American College of Radiology (ACR) guidance and integrating recent evidence, the CAR IFWG offers a pragmatic approach emphasizing radiologic features, clinical context, and patient risk factors to minimize unnecessary follow-up. The recommendations aim to streamline care, reduce patient anxiety, and support radiologists in distinguishing benign from potentially malignant findings in asymptomatic individuals.
Introduction
The Canadian Association of Radiologists (CAR) Incidental Findings Working Group (IFWG) consists of both academic subspecialty and general radiologists tasked with authoring guidelines specific to the Canadian health care system. Guidelines on how to manage incidental renal, 1 hepatobiliary, 2 pancreatic, 3 and musculoskeletal imaging findings 4 are frequently downloaded resources for both academic and community radiologists. Based on CAR member feedback, the IFWG elected to develop guidelines on splenic and nodal incidental findings. Specific questions posed to the working group were how to screen for splenomegaly, deal with focal splenic lesions, and manage splenic artery aneurysms. Along with guidance on assessing lymph nodes, the IFWG included the assessment of mesenteric panniculitis/lipodystrophy and peritoneal nodules into this manuscript as well.
Following a comprehensive evaluation of the available literature, all incidental findings and management recommendations were discussed as a group for consensus. For a summary of recommendations, please see Table 1.
Summary Statements/Summary of Recommendations.

While the information and tables presented below are based on the best available scientific evidence, significant gaps in the literature remain. Ultimately, these management recommendations reflect consensus recommendations rather than a fully evidence-based standard of care.
Splenomegaly
Apparent enlargement of the spleen, or splenomegaly, is a common incidental finding in diagnostic imaging. The incidence of splenomegaly is likely inflated by a lack of agreed-upon standard measurements for a normal sized spleen. While pathologic literature suggests an average spleen weight of approximately 150 g for adults, 5 this measurement is quite dependent on patient body habitus. The radiologic measurement of the spleen similarly is highly variable based upon patient height and weight. 6 Commonly accepted measurements of the upper limits of normal splenic length range from 12 to 13 cm.7,8 In one large study, 26% of normal Caucasian male patients exceeded a 12 cm single measurement, with 9% measuring >13 cm and 2% measuring >14 cm. In the same study, 6% of normal Caucasian female patients exceeded a single measurement of 12 cm, with 2% measuring >13 cm and none measuring >14 cm. 6 Although medical calculators can diagnose splenomegaly by correcting for body size, they are cumbersome, requiring knowledge of the patient’s height, weight, and gender. 6 Although the literature suggests that splenic volume calculation may represent the future of spleen measurement, 7 other studies showing a close correlation between a single largest measurement and total spleen volume6,8 favour continuing with the current status quo of providing a single value to represent spleen size. Volume calculations can be referenced to body size when more accuracy is required, particularly to avoid overdiagnosing splenomegaly in larger patients.
Focal Splenic Lesions
Background
Splenic incidental findings are defined as lesions detected on imaging in the spleen not related to the clinical history. Incidental splenic lesions are less common than in other organs such as liver or kidneys, but increased demand for imaging means that their frequency is rising. 9 Incidental focal splenic lesions have a wide range of etiologies, ranging from common benign diagnoses (cysts, granulomas, and hemangiomas) to lymphoma or metastases to exceedingly rare primary malignancies such as angiosarcoma. In one study, 1.5% of trauma patients with CT had an incidental splenic lesion 10 and the vast majority are benign. 11 Benign lesions are almost always asymptomatic, whereas malignant lesions are very rarely entirely incidental or a solitary isolated finding. 12
Siewert et al conducted an observational study of 379 patients with splenic masses, finding that 205 (54.1%) had no history of malignancy or symptoms, classifying them as truly incidental. Only 1.0% of these patients (2/205) had malignant masses (ovarian cancer metastasis and lymphoma), and 1 patient had a rapidly growing mass diagnosed as sclerosing angiomatoid nodular transformation of the spleen (SANT). 13 This raises questions about the necessity of further workup for incidental indeterminate splenic lesions in asymptomatic patients. Malignant masses were found in only 0.6% of patients (2/337) with isolated splenic masses, all being symptomatic, indicating that truly incidental splenic masses are generally not clinically significant. However, 27.6% of patients with constitutional symptoms and no malignancy history had lymphoma, with 25% presenting as isolated splenic involvement. Thus, for patients with constitutional symptoms and an indeterminate splenic mass, further evaluation is warranted. Factors like calcifications, lesion count, and splenic size were not useful in distinguishing benign from malignant lesions. 14
Lymphoma is the most common malignancy of the spleen, either primary or part of diffuse systemic disease.12,15 Splenic involvement occurs in approximately 33% of patients with Hodgkins and 30% to 40% in patients with non-Hodgkins Lymphoma. 16 Lymphoma can present in many forms including splenomegaly, diffuse nodules (either in a miliary pattern or larger nodules), or a solitary mass. 12 Primary splenic lymphoma confined only to the spleen ± perisplenic nodes is very rare, comprising less than 1% of cases, and most patients will present with constitutional symptoms. 12
In patients with a history of malignancy, isolated splenic metastases are rare, occurring in only 1% of patients in an observational oncologic study 17 or reported in a few case reports.18,19 Splenic metastases typically occur due to hematogenous spread and most frequently seen in patients with known or widespread cancer. 12 The most common cancers to metastasize to the spleen are breast, lung, ovarian, stomach, melanoma, 9 and prostate. 20 In a recent series, only 33.8% of splenic masses were found to be malignant even in patients with a history of malignancy. Furthermore, every patient with splenic metastases had multiple areas of disease involvement, 13 highlighting that isolated splenic metastases are quite rare.
When a new indeterminate mass is detected in the spleen on ultrasound, there is always the possibility that other metastatic lesions may not be visualized on the scan. 9 Single or multiple homogeneous hyperechoic splenic masses are statistically likely to be benign hemangiomas, whereas lymphoma is almost always hypoechoic.12,21 Unfortunately, splenic metastases have a varied appearance on ultrasound, with approximately 50% appearing hypoechoic but others appearing hyperechoic (especially from colon cancer or melanoma), heterogeneous (either targetoid or mixed hypo- and hyperechoic), or cystic. 22 Thus, a hyperechoic splenic lesion on ultrasound cannot necessarily be considered benign and the patient still needs to be managed based on their clinical history or presence of symptoms.
Recommendations
The 2013 American College of Radiology (ACR) recommendations on managing incidental abdominal and pelvic findings provides an algorithm to manage incidental splenic lesions based on broad categories that combined the use of prior imaging and clinical history/risk factors to aid the reporting radiologist. 23 The CAR IFWG built on this algorithm by reviewing subsequently published literature and including lesions detected incidentally on ultrasound (Figure 1).
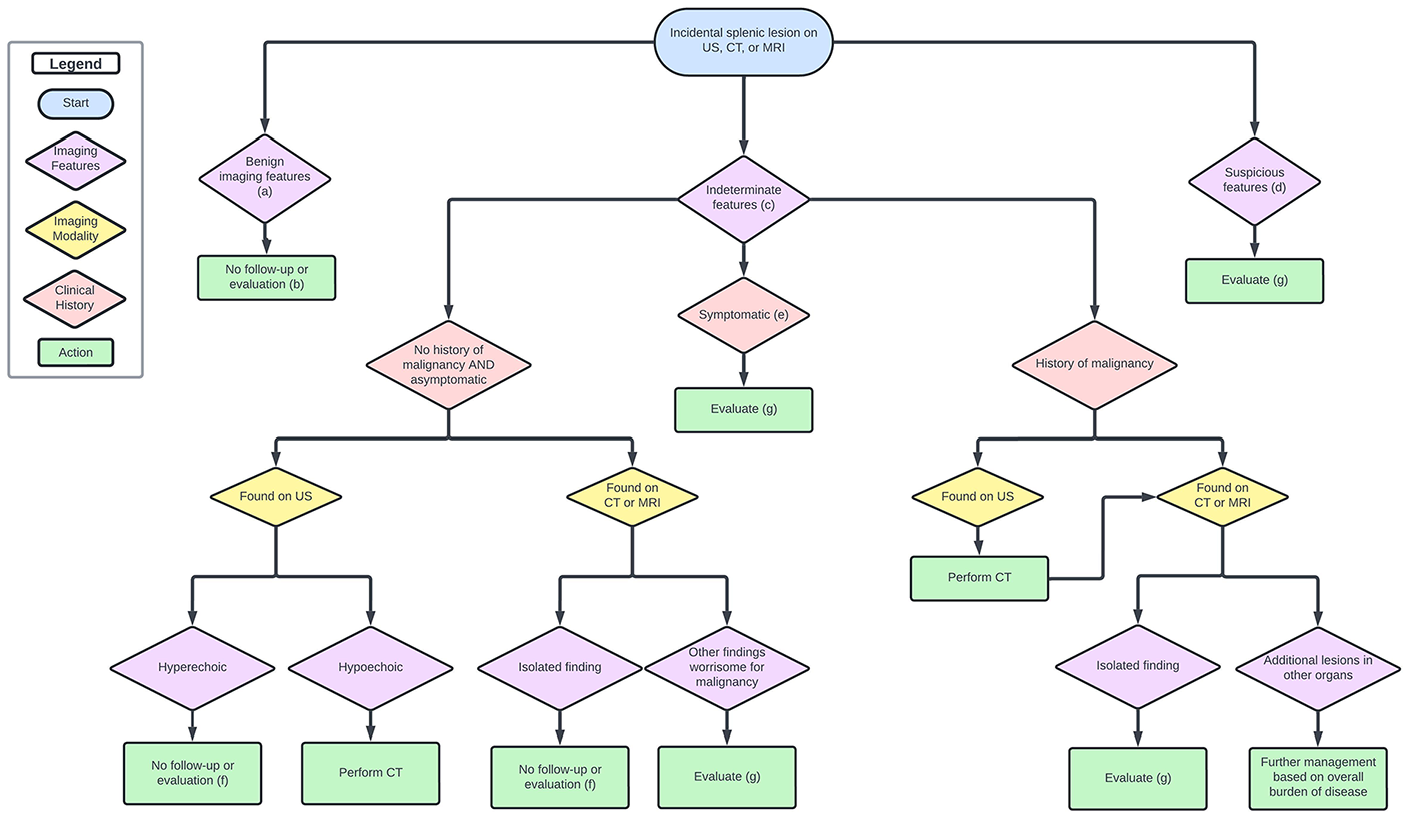
Decision-making algorithm for incidental findings on spleen and nodes.
Clearly benign features of an incidental splenic mass include being completely anechoic on ultrasound 24 or homogenously low attenuation (<20 HU) on CT with no enhancement and smooth margins. A solid lesion can be confidently diagnosed as a hemangioma there is discontinuous, peripheral, centripetal enhancement on CT or MRI, although this is less commonly seen compared to hepatic hemangiomas. These were all considered clearly benign features on the ACR white paper in 2013, and subsequent studies have confirmed that benign lesions are more likely to be cystic, homogenous, and have well-defined boarders, and no restricted diffusion on MRI.
In contrast, clearly suspicious features of a splenic mass include heterogeneous enhancement, irregular margins, internal necrosis, evidence of splenic parenchymal or vascular invasion, findings in other organs suspicious for malignancy, associated lymphadenopathy, or a mass that is growing. The current literature is sparse in advising what growth rate is suspicious, but it is the view of the IFWG that if a solid lesion grows by more than 5 mm a year, further evaluation is warranted. Mild interval growth in an otherwise benign appearing mass (such as a simple cyst or hemangioma) is not worrisome. For example, hepatic hemangiomas have been shown to normally grow up to 2 mm annually, and this likely occurs in splenic hemangiomas as well. Radiologists should still be aware that if a patient has a growing or new splenic cyst compared to prior scans, it could represent echinococcal disease in endemic areas.25,26
If the imaging features of the splenic lesion are not clearly benign or are suspicious as defined above, comparison with prior imaging and the clinical context is essential.
Indeterminate splenic lesions are defined as heterogenous, intermediate attenuation >20 HU on CT, or enhancing but with smooth margins. On ultrasound, single or multiple splenic masses that are hypo- or hyperechoic (but not simple cystic) are also considered indeterminate as they have a broad differential including hemangiomas, lymphangiomas, SANTs, pseudotumors, infection, sarcoidosis, lymphoma, multiple myeloma, sarcoma, or metastasis.15,22,27 A hyperechoic incidental lesion on ultrasound requires no further follow-up, but as the likelihood of malignancy is higher with a hypoechoic lesion, the IFWG recommends CT assessment for an indeterminate hypoechoic mass in order to first exclude any other findings worrisome for malignancy.
If the new splenic lesion(s) are associated with other sites of metastatic involvement, however, they can simply be monitored along with the rest of the metastatic disease or lymphoma on follow-up imaging. 26
Further Investigation
The choice between MRI, PET/CT, or biopsy to further evaluate a splenic lesion depends on the clinical context. PET/CT is the most helpful if the primary concern is systemic lymphoma or metastatic disease from a prior malignancy known to be FDG-avid. Otherwise, MRI is helpful in evaluating multiple splenic masses, as lymphangiomas are often mistaken for metastatic disease on other modalities. On MRI, multiple hypointense lesions on all sequences can be seen in granulomatous disease (eg, occurring in 24%-59% of sarcoid patients). Siderotic nodules in cirrhotic patients are also T2-dark and show susceptibility (Gamnma-Gandy bodies). 28
Most common reasons for splenic biopsy include an indeterminate lesion despite further imaging with MRI, suspected lymphoma and no other accessible sites for biopsy, or a new splenic mass in a patient with a known history of malignancy and no other accessible sites. 29 There is a common misconception that splenic biopsy poses an unacceptable risk to the patient. This issue has been well studied and splenic biopsy is an effective and safe procedure for histologic diagnosis of indeterminate splenic lesions with a recent study showing a 91.1% accuracy rate (ie, sufficient material for histologic assessment) a 6.7% risk of minor complications and no reported major complications. 30 A meta-analysis and systematic review by McInnes et al in 2011 demonstrated a pooled major complication rate of 2.2% but if the biopsy gauge is kept to under 18G, the major complication rate fell to 1.3%, comparable to the complication rate for liver and renal biopsies. 31 These findings have been confirmed on subsequent studies, showing a major complication rate of 1% and minor complication rate of 7.2%. 32 It is of the opinion of the IFWG that percutaneous splenic biopsy is safe and effective for working up indeterminate splenic lesions.
Splenic Artery Aneurysms
The estimated incidence of splenic artery aneurysms is 0.8% 33 but the frequency will likely increase with the expanding use of diagnostic imaging in the aging population. Approximately 80% of splenic artery aneurysms are found incidentally 34 and are the most common visceral artery aneurysm. 33 Chronic liver disease leading to portal hypertension and pregnancy are the most significant risk factors for developing a splenic artery aneurysm and also the leading risk factor for rupture 35 with a mortality rate between 25% and 70%. 34 In light of this, the Society of Vascular Surgery has produced clinical practice guidelines for managing visceral artery aneurysms. 36 These guidelines recommend that patients with an incidental splenic artery aneurysm ≥3 cm and any pregnant patient or patient with increased risk of rupture (patients with portal hypertension, those requiring liver transplant, or patients with a splenic artery aneurysm felt to be non-degenerative/atherosclerotic, such as a mycotic aneurysm or pancreatitis-related pseudoaneurysm) with an incidental splenic artery aneurysm of any size should be referred to vascular surgery for management. By contrast, the Cardiovascular and Interventional Radiological Society of Europe (CIRSE) recommends that splenic artery aneurysms ≥2 cm, aneurysms showing growth of >0.5 cm/year, or aneurysms felt to be non-degenerative (saccular morphology or any other suggestion of a mycotic or pancreatitis-related etiology) should be referred to an endovascular specialist for consideration of treatment. 37 The Working Group endorses the 2024 CIRSE recommendations, which are closely aligned with current Canadian interventional radiology practice patterns. A patient with a <2 cm splenic artery aneurysm or with significant comorbidities to treatment can be followed with CT or MR angiography every 12 months. Ultrasound can be considered for follow-up if the aneurysm is well seen, and the radiologist is confident that any change in the aneurysm will be detected. There is no published literature addressing when it may be safe to discontinue follow-up for a splenic artery aneurysm, but the IFWG recommends that radiologists discuss this with their local vascular specialist when an aneurysm <2 cm demonstrates ongoing imaging stability.
Nodes
When assessing a lymph node, it is important to assess the size as well as other features such as shape, presence of a fatty hilum, and calcification. There is considerable overlap in size and morphologic features of benign and malignant lymph nodes. Benign lymph nodes usually have a smooth, circumscribed margin, and homogeneous signal intensity and enhancement with a fatty hilum. Calcification is most commonly a result of a benign process such as granulomatous infection or sarcoidosis but can be seen with treated lymphoma or certain malignancies such as mucinous tumours, papillary thyroid cancer, osteosarcoma, or chondrosarcoma. In addition, it is important to assess the remainder of the scan to determine if there is other evidence of malignancy or a cause for reactive lymph nodes.
The following recommendations are specifically for patients with no known or suspected malignancy or lymphoproliferative disorder. All size measurements should be based on shortest axis in any plane.
Mediastinal and hilar lymph nodes are frequently enlarged and reactive in the setting of various diseases including pneumonia, emphysema, interstitial lung disease, congestive heart failure, and sarcoidosis. One study demonstrated that isolated lymph nodes less than 15 mm in short axis were always reactive. 38 This 15 mm cutoff was also adopted by the ACR Incidental Findings Committee.39,40 The CAR IFWG generally cosigns this recommendation, but asserts that internal mammary and para-esophageal nodes are seen sufficiently infrequently as benign incidental findings that these should be excluded from the statement. Radiologists should consider the possibility of incidental breast pathology when internal mammary nodes >5 mm in the long axis are seen.41,42 Paraesophageal nodes >5 mm in the short axis or clusters of ≥3 paraesophageal nodes should raise the possibility of underlying esophageal pathology.
With respect to intra-abdominal lymph nodes, there is poor evidence and a lack of consensus in the literature as to a specific cut-off that warrants further evaluation.
For larger lymph nodes, it is crucial to evaluate the clinical history and imaging for other causes of potential reactive lymph nodes, such as infectious or inflammatory bowel disease, liver disease, or autoimmune conditions.
For incidentally detected enlarged lymph nodes with concerning features and no clear cause, compare with prior imaging if available. Nodes stable for at least 1 year can be deemed benign, with no further workup needed. If nodes have increased in size, further evaluation with biopsy or imaging (PET/CT or CT to check for other nodes or primary malignancy elsewhere) is advised. Without prior imaging, correlate with clinical or laboratory signs of potential malignancy, particularly lymphoproliferative disorders.
For enlarged gastrohepatic nodes (≥8 mm) and retrocrural nodes (≥6 mm), consider upper endoscopy to rule out esophageal or gastric cancer. For enlarged mediastinal or hilar nodes (≥15 mm) and retroperitoneal, mesenteric, or pelvic nodes (≥10 mm), a follow-up CT in 3 to 6 months may be useful if there are no clinical concerns. If stable, a follow-up CT at 12 months is recommended. In cases of significant concern for metastatic or lymphoproliferative disease, consider a full-body CT to rule out additional disease.
Further evaluation with PET/CT is not universally recommended in cases of incidentally detected lymphadenopathy, because reactive lymph nodes may also demonstrate increased FDG uptake and may result in further diagnostic confusion. 45 PET may be advised for suspected lymphoproliferative disorders or to identify a nonvisualized primary malignancy; it can also help determine the optimal biopsy site.
The use of MR diffusion weighted imaging (DWI) has been shown to be a useful tool in some settings in evaluating for metastatic lymph nodes, particularly in the setting of certain known malignancies such as in prostate, cervical, and colorectal cancer. While few studies have assessed its utility in incidental lymphadenopathy, DWI may assist in differentiating between benign and malignant lymph nodes. 46 Thus, if lymph nodes are incidentally discovered on MRI and DWI was performed, the ADC map should be subjectively evaluated and nodes showing restricted diffusion should be treated with a higher index of suspicion.
Mesenteric Lipodystrophy/Panniculitis
Mesenteric panniculitis (also known as “sclerosing mesenteritis,” “retractile mesenteritis,” “mesenteric lipodystrophy,” or “mesenteric fibrosis”) is a chronic idiopathic inflammation of the mesenteric fat leading to varying degrees of inflammation and fibrosis.47-51 Some potential etiologies include trauma, previous surgery, autoimmune disorders, cancer, and ischemia of the mesentery.47-51 There are conflicting reports regarding its prevalence, ranging from 0.16% to 7.8%.47,48,52 Most studies show a male predominance with a ratio of 2:150,51 and it is most commonly seen in fifth to seventh decade of life.47,48,50,51
While the majority of patients are asymptomatic, some patients will complain of abdominal and flank pain (30%-70%), systemic symptoms such as fever and malaise, weight loss (20%-23%), nausea, vomiting, change in bowel habits (25%), and a palpable mass.47,48,51 The duration of symptoms in one study population ranged from 3 weeks to 2 years. 52 A very small subset of patients may have a chronic debilitating course or bowel obstruction requiring surgical and medical intervention. 51
CT findings of mesenteric panniculitis include mass-like stranding of the mesenteric fat typically at the root of the small bowel mesentery (“misty mesentery sign”), often encapsulated by a pseudocapsule and with associated mesenteric soft tissue nodules within the encapsulated area of fat stranding. Additional characteristic features include “fat ring” or “fat halo” sign (ring of clear fat without stranding surrounding the mesenteric soft tissue nodules), and intact vessels crossing the fatty mass.47-49,53 In the fibrotic-predominant type, fibrosis and retraction of the bowel and mesenteric vasculature, known as “retractile mesenteritis,” can lead to bowel obstruction and submucosal edema due to vascular encasement or thrombosis.48,49,51,54 On MRI, the fat stranding is typically hypointense to mesenteric fat on T1 and T2-weighted imaging, slightly hyperintense on T2 fat-saturated images and it does not demonstrate diffusion restriction. In the fibrotic-predominant type, the fibrosis is T1 and T2 hypointense.48,49
Although mesenteric panniculitis itself is a non-malignant disease, its imaging appearance may mimic lymphoproliferative disease or metastatic lymphadenopathy.47,54 The most important differential diagnosis is lymphoma which may have the same appearance as mesenteric panniculitis. The “fat ring sign” (preservation of a ring of normal fat around vessels and nodes in the affected area) was historically described as a specific feature, but this has subsequently been seen in lymphoproliferative disease.49,53,55 The fibrosis-predominant type can present as a mesenteric retractile mass with calcifications, indistinguishable from carcinoid and desmoid tumours, and biopsy may be required in these cases.48,49,53
A 2015 systemic review of 675 articles, of which only 14 were eligible, failed to find a single study showing an association between mesenteric panniculitis and subsequent malignancy with certainty due to selection bias, incomplete follow-up and lack of methodological consistency. 54
Two large retrospective studies involving over 3000 consecutive CT examinations at academic centres yielded opposite results on the relationship between mesenteric panniculitis and malignancy. In both study populations, 48% to 60% of the abdominal CTs were performed for malignancy staging or had known cancer at the time of imaging. One found a strong association between mesenteric panniculitis and current or future malignancy, and the other found no association.47,56 In a systematic review, 38% of reported patients had known underlying malignancy at the time of the diagnosis of mesenteric panniculitis. 54 The prevalence of pre-existing cancer in these studies creates a significant confounder in determining the risk of malignancy and their results cannot necessarily be applied to patients without such a history, which is the clinical scenario most often encountered by radiologists.
A retrospective study of 444 patients with imaging features of mesenteric panniculitis identified 2 CT features predictive of malignancy: (1) large soft tissue nodules with a short axis diameter greater than 10 mm within the mesenteric panniculitis, and (2) lymphadenopathy in another abdominopelvic region. Using both criteria yielded a sensitivity of 100% and specificity of 99% for identifying underlying malignancy. Notably, only 1% (5 patients) were diagnosed with new malignancies during follow-up, all of which were low-grade B-cell lymphomas. However, the follow-up period was relatively short. 55
F-18 FDG PET/CT can be helpful in distinguishing mesenteric panniculitis from neoplastic disease mimicking mesenteric panniculitis. Lack of uptake on PET has been found to have a high diagnostic accuracy in excluding malignancy, although increased uptake on PET does not reliably distinguish malignancy from mesenteric panniculitis as both may show activity. 57
Peritoneum
Peritoneal nodules frequently raise concern for metastatic disease (carcinomatosis) but a variety of other conditions may present as mimics. Concerning features for an incidental peritoneal nodule seen on CT or MRI include irregular margins, multiple nodules, a background of hazy peritoneal fat, or a history of malignancy; the presence of any of these necessitates immediate management.
The possibility of a primary tumour elsewhere must be considered. If there is no evidence of a primary and the size is larger than 1 cm, consider PET/biopsy of the nodule if this changes clinical management. If size is less than 1 cm, either short-term (≤3 months) follow-up or surgical consultation and laparoscopy should be considered for diagnosis depending on the morphology and index of suspicion. Especially important primary cancers to consider and to assess for peritoneal spread include appendiceal/gastrointestinal, pancreatic, or gynecologic cancers.
Small solid soft tissue nodules without otherwise concerning morphology and no history of malignancy may represent benign conditions or small reactive lymph nodes, and imaging stability is a key feature to assess. Guidance on how to approach a solitary peritoneal nodule is minimal, but the IFWG feels that imaging stability of over 12 months is consistent with a benign etiology. Some possibilities include tumour-like conditions such as fibrosis/fibromatosis, endometriosis, leiomyomatosis, extramedullary hematopoiesis, or splenosis. 58 Other conditions to consider include dropped gallstones or retained surgical material.
The peritoneal cavity usually contains 50 to 75 mL of clear free fluid which serves as a lubricant to the tissues. 59 Free fluid is a common imaging finding, with multiple studies demonstrating the presence of small amounts of free fluid to be physiologic in both men and women, even in setting of trauma. In a large retrospective study, a small amount of isolated pelvic free fluid was found in 4.9% (49 of 1000) of male patients with blunt trauma who did not have an undiagnosed bowel and/or mesenteric injury, with “small” defined as fluid seen on 5 or fewer contiguous 5-mm-thick sections or a mean volume of 2.3 mL. 60 Although it is commonly accepted that premenopausal female patients will often have a small amount of physiologic free fluid (up to 38% of premenopausal females), 61 various studies have now documented physiologic fluid in male and post-menopausal female patients. A small ultrasound study targeting healthy male volunteers found free fluid in 4 of the 10 volunteers. 62 Another study employing pelvic MRI concluded that peritoneal fluid of less than 10 mL is not clinically significant in men and postmenopausal women, with peritoneal fluid identified in 3.8% (39 of 1017) of healthy men and 16.8% (52 of 310) of healthy post-menopausal women. 63
Abnormal free fluid has multiple causes including infection, inflammation, and malignancy. The clinical context is important to consider, such as any history of cirrhosis, trauma, hemorrhage, infection, and malignancy. In the setting of malignancy with new ascites, the finding would be concerning for malignant peritoneal involvement and a careful examination for associated peritoneal nodules would be important to perform.
Another common condition involving the peritoneum is fat necrosis, resulting from adipose tissue infarction due to traumatic or ischemic injury. This leads to organized fat necrosis surrounded by a fibrous capsule, isolating it from surrounding tissue. Epiploic appendagitis and omental infarcts are common examples that can cause abdominal pain. Additionally, pancreatitis may release lipolytic enzymes, leading to nodular saponified fat. On imaging, fat necrosis appears as a central fatty core with possible inflammation and calcifications. Encapsulated fat rarely shows mild mass effect without invading or displacing adjacent organs. 64 Clinical findings that may indicate fat necrosis instead of malignancy include focal tenderness at palpation of the area and a history of surgery or trauma. Fat infarction can also be infected, which may demonstrate stranding surrounding the area of fat or presentation with infectious symptoms. The diagnosis of fat necrosis may be influenced by the clinical history, specifically evidence of pain, malignancy, surgery, or trauma. Imaging features suggesting liposarcoma would include invasion of adjacent organs, mass effect, and an increase in size over time. The presence of thick moderately or markedly enhancing septa (>2 mm) within a fatty mass would also raise concern for liposarcoma. 65 Fat necrosis should not invade or displace other structures, and if anything only decreases in size with time. Nodular appearing non-fatty areas may appear in both fat necrosis and liposarcoma. In the context of encapsulated fat necrosis mimicking a malignant entity such as liposarcoma, short term imaging follow-up in 3 to 6 months may be required for differentiation.
Footnotes
Acknowledgements
The authors would like to acknowledge and thank the CAR members who provided their feedback and peer review during the preparation of these guidelines.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
