Abstract
Introduction
Melanoma is a malignant neoplasm originating in melanocytes or cells derived from them. It presents as primary or metastatic, and the most common primary site is cutaneous. Melanomas express multiple peptides and antigens in their neoplastic cells making them excellent targets for the patient’s own immune system. Therefore, immunotherapy represents an efficient therapeutic strategy in this group of patients. Specifically, immune checkpoint inhibitors (ICPI) directed against cytotoxic T-lymphocyte associated antigen 4 (CTLA-4) or programmed cell death protein (PD-1) and its ligand PD-L1, have shown significant impact on survival and quality of life.1-4 However, these treatments can also over-enhance the immune responses with imbalance in the immune system, leading to immune-related adverse events (IRAEs), which can potentially affect any tissue or organ in the body with a broad clinical spectrum of presentations. Prior studies have reported an overall incidence of IRAEs associated with ICPI as high as 80% to 90%, 5 and a specific incidence of 55% for combined therapy (ipilimumab plus nivolumab), 20% to 30% for ipilimumab as a single agent, and 10% to 15% for nivolumab alone. 6 A meta-analysis by Wang et al, including 46 studies representing 12 808 oncologic patients treated with ICPI, showed the overall incidence of IRAEs was of 26.8% for any grade, and 6.1% for severe reactions.7,8
Dermatological IRAEs are the most common with an incidence of approximately 13% to 43%,9,10 followed by gastrointestinal manifestations with a prevalence of 22% to 40%.5,9,11 Biochemical abnormalities such as transaminitis and lipase elevation are encountered in 1% to 10% and 5% to 14%, respectively.3,12 Endocrinological IRAEs have a general incidence of 40%, 13 with thyroiditis being the most common (5%-23%).11,13,14 Pneumonitis is also frequent with an incidence of 3% to 10% and has been described predominantly in patients with underlying lung disease and/or lung cancer. 15 Finally, musculoskeletal disorders are also frequent, including arthralgia (9%-43%), myositis (21%), and synovitis (7%).5,9,16
Clinically IRAEs are graded according to the Common Terminology Criteria for Adverse Events (CTCAE). 17 Overall, grade 1 toxicities are mild not requiring suspension of therapy, grade 2 reactions require symptomatic treatment with resumed therapy once controlled, grade 3 toxicities considered moderate, lead to temporary suspension of immunotherapy and initiation of high-dose corticosteroids. Finally, grade 4 reactions are life-threatening requiring urgent care and complete suspension of therapy, and grade 5 reactions are death related.
The presentation of IRAEs on imaging is variable, as most cases are diagnosed clinically without requiring imaging confirmation. However, PET/CT findings related to IRAEs have been reported in up to 61% to 70% of patients during treatment, with approximately 15.2% classified as moderate to severe (grade 3-4).18-20 As a powerful tool for staging metastatic melanoma and monitoring therapy response, PET/CT also has high sensitivity for detecting inflammation, 9 making it valuable for identifying immune-related adverse events. Notably, in up to 33% of cases as reported by Tirumani et al in a cohort of 147 patients,8,16 inflammatory changes can be seen on imaging before the onset of symptoms, allowing for earlier intervention, which—through therapy discontinuation or immunosuppression—can significantly improve patient outcomes.21-24
The aim of this study was to determine the frequency and distribution of IRAE encountered on 18F-FDG PET/CT in a large academic cancer centre.
Materials and Methods
Study Design
We conducted a retrospective observational cohort study, including consecutive patients with metastatic melanoma receiving immunotherapy, referred for therapy response assessment with 18F-FDG PET/CT at our institution over 12 years (January 2010-August 2022). These were retrospectively identified using a radiology information system search engine. This study received approval of the Institutional Research Ethics Board and informed consent was waived due to the retrospective nature of the study.
Patients that fulfilled the inclusion criteria were included in the cohort. Each PET/CT performed for these patients during treatment was recorded as an independent time-point. PET/CT studies were prospectively interpreted by one of 6 radiologists/nuclear medicine physicians with at least 5 years’ experience reporting PET/CT (median 13.2 years, range 8-22 years). All studies with findings suspicious for an IRAE were documented and confirmed by 2 reviewers (VM and CO) in consensus. All potential IRAE identified on PET/CT (ie, thyroiditis, colitis, and myositis) were recorded.
The reference standard was determined for each IRAE and the clinical assessment included physical examination, confirmatory laboratory tests, correlative imaging, endoscopy, and/or biopsy, obtained in temporal proximity to the corresponding PET/CT. Diagnostic performance of PET/CT was analyzed at a timepoint level and individually for each IRAE, since at least 3 patients had more than one IRAE at the same timepoint.
Imaging Acquisition
18F-FDG PET/CT images were acquired according to the institutional protocol with the same parameters in 2 different scanners: a Siemens mCT40 PET/CT (2010-2020) and a Siemens Vision scanner (2020-present). Patients were positioned supine with arms down. Images were obtained from the top of the skull to the feet after the administration of 5 MBq/kg of FDG and Iodinated oral contrast material was given for bowel opacification; no intravenous contrast material was used. The average injected dose of FDG was 411 MBq (272-547) [11.1 mCi (7.35-14.7)] and the average uptake time was 62.6 minutes (49-94). Overall, the same parameters were used in both scanners: 5 to 9 bed positions were obtained, depending on patient height, with an acquisition time of 2 to 3 minutes per bed position. CT parameters were 120 kV; 3.0 mm slice width, 2.0 mm collimation; 0.8 seconds rotation time; 8.4 mm feed/rotation. A PET emission scan using time of flight with scatter correction was obtained covering the identical transverse field of view.
Statistical Analysis
Demographic data (age, gender), tumour characteristics at staging (primary tumour histology and location, TNM stage), immunotherapy treatment regimen, imaging characteristics (type of IRAE and location), clinical severity, clinical and imaging follow-up were tabulated. Descriptive statistics were reported through mean and standard deviation (SD) or median and interquartile range (IQR) in continuous variables depending on their distribution while proportion and 95% confidence intervals (95% CI) was used for the categorical variables. When comparing between IRAE and non-IRAE patients, for categorical variables Chi2 or Fisher exact were used depending on the expected cell values and for continuous variables unpaired t test or Mann-Whitney were performed depending on the sample distribution.
We evaluated the diagnostic performance of PET/CT at the timepoint level and at the IRAE level. True positives were defined as cases with PET/CT findings concordant with the reference standard. False positives those with PET/CT findings excluded or not confirmed by the reference standard. False negatives as those diagnosed by the reference standard but not identified on PET/CT and true negative cases with negative PET/CT, and negative reference standard.
Sensitivity, specificity, positive predictive value, negative predictive value, accuracy, and their 95% CI were calculated based on the construction of a contingency table. Diagnostic yield was calculated for each IRAE using the formula described by Park et al 25 where the sum of true positives for each IRAE, independent of their clinical impact, was divided by the cohort size.
Statistical analysis was performed using R and P < .05 were considered significant.
Results
We evaluated 147 patients at 201 timepoints (ie, follow-up, therapy response assessment) during their disease. A total of 35 patients had more than 1 scan, with a median time between scans of 4.78 months (range 1.01-17.2). Most patients, 87/147 (59.2%) were men, and the mean age (±SD) at the time of the study was 61.4 ± 14.7 years (range 26-88). Baseline characteristics at the time of diagnosis including type and subtype of melanoma, primary location, and metastasis at staging are described in Table 1.
Demographics and Baseline Tumour Characteristics in the Entire Cohort at the Time of Diagnosis.

Note. n = number; SD = standard deviation; NS = no specified.
Overall, there were 36/147 patients (24.5%) with IRAEs recorded according to the standard of reference, with 39 IRAEs in the entire cohort, given that 3 patients had more than one IRAE; A total of 204 events were considered at the IRAE level. From the 36 patients with recorded IRAE, 16/36 (44.4%) were women and 20/36 (55.6%) men. At the time of presentation 69.4% (25/36) were older than 60 years old, 6/36 (16.7%) were between 50 and 60 years, 1/36 (2.8%) between 40 and 50 years, 2/36 (5.5%) between 30 and 40, and 2/36 (5.5%) were less than 30 years old.
At a time point level, PET/CT identified 36/36 (100%) patients with IRAEs confirmed by the reference standard. Additionally, PET/CT was positive in 3 patients where the reference standard was negative (false positives). Meanwhile, clinical suspicion identified 26/36 (72%) cases confirmed by the reference standards, with 10 false negative and 8 false positive results.
At the IRAE level, PET/CT identified 36/39 (92%) of IRAEs confirmed by the reference standard, with 3 false positive and 3 false negative results, as shown in Table 2. False positive results corresponded to vasculitis (extracranial ICA), panniculitis, and adrenalitis; all re-evaluated and excluded by the reference standard. False negative results corresponded to hepatitis (2/3) and colitis (1/3), both diagnosed by the reference standard; of note, these 3 events were suspected clinically. Significant examples are shown in Figures 1 to 6.
Diagnostic Performance of PET/CT per Immune Related Adverse Event.
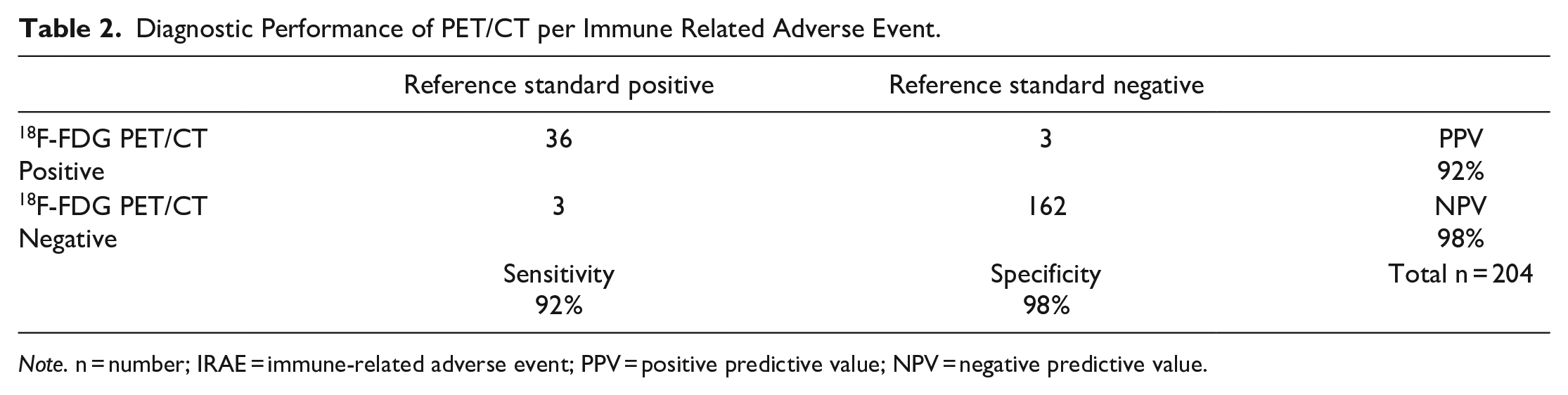
Note. n = number; IRAE = immune-related adverse event; PPV = positive predictive value; NPV = negative predictive value.
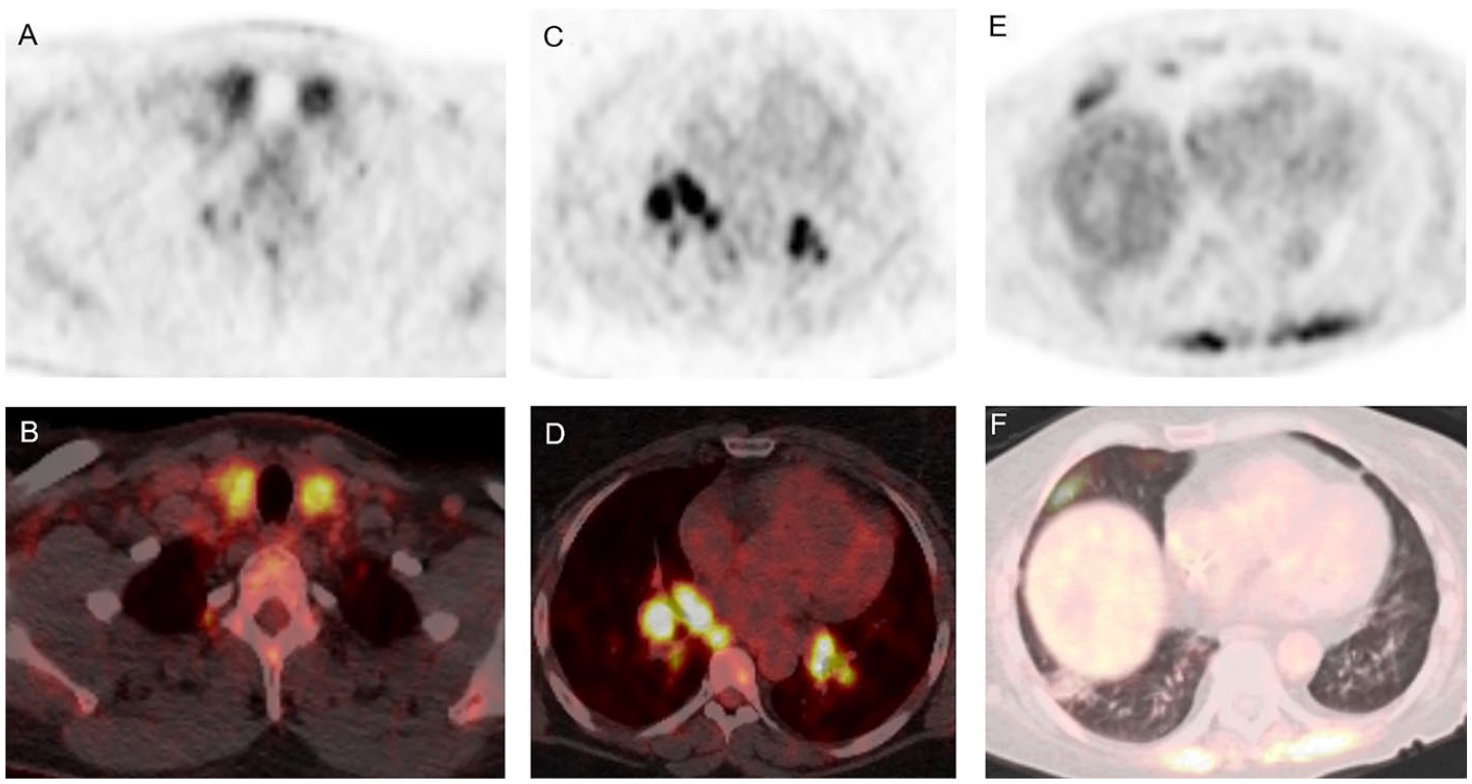
Common IRAE findings on 18F-FDG PET CT. (A, B) PET and fused PET CT axial images showing diffusely increased FDG uptake in a patient with clinically confirmed immune-related thyroiditis. (C, D) PET and fused PET CT axial images showing several metabolically active bilateral hilar lymph nodes with symmetric distribution, consistent with immune-related sarcoid-like reaction. (E, F) PET and fused PET CT axial images showing metabolically active patchy ground-glass opacities in the right lower lobe, in keeping with immune-related pneumonitis.
Sixty-six-year-old female patient with history of BRAF mutated cutaneous melanoma on immunotherapy with ipilimumab. Follow-up 18F-FDG PET CT (A) MIP, (B) axial PET, (C) axial fused PET CT, showed patchy FDG uptake in the right hepatic lobe, corresponding to patchy contrast enhancement on the arterial phase (D). This finding suggested immune-related hepatitis, which was later confirmed with the presence of elevated ASL, ALT, and GGT. The resolution of these laboratory abnormalities after therapy modification, further supported the diagnosis.
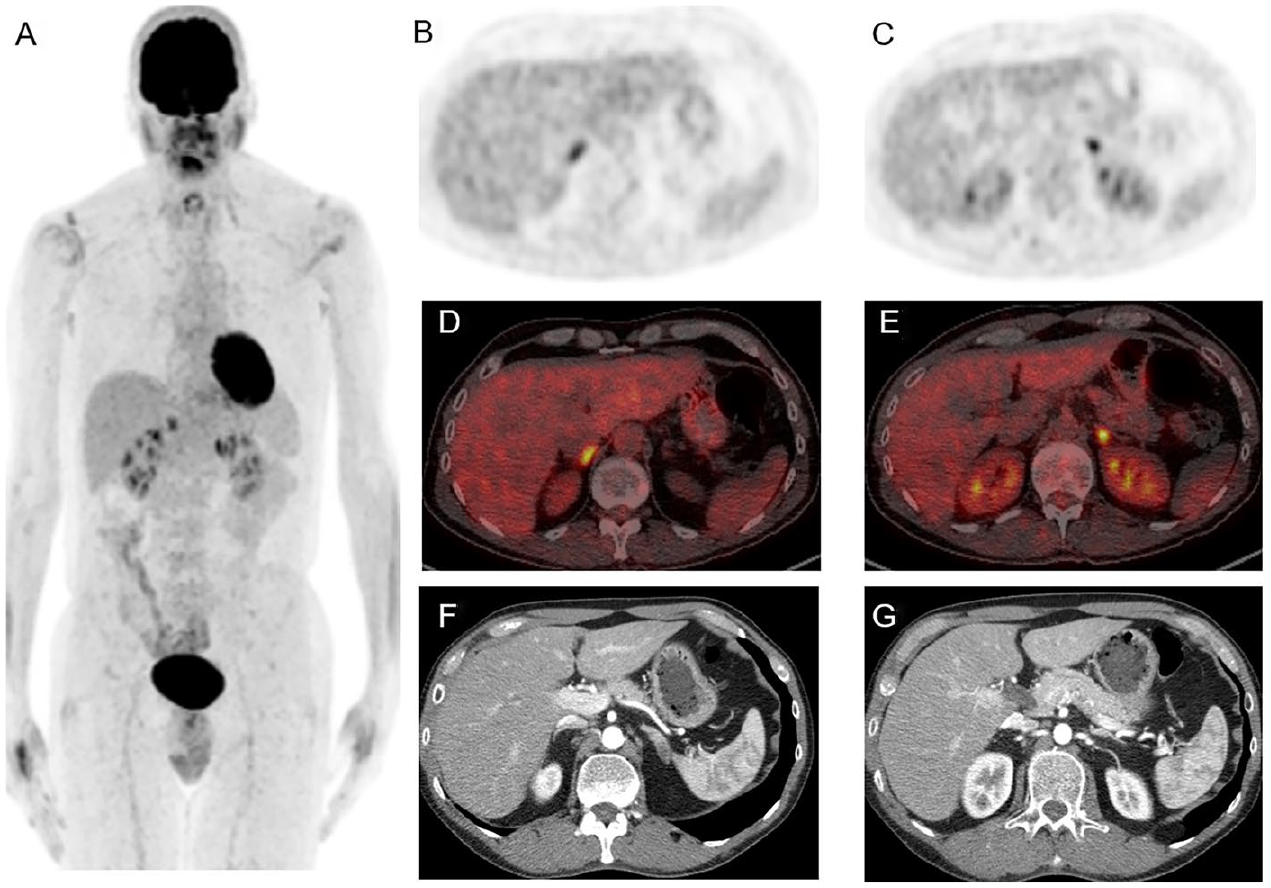
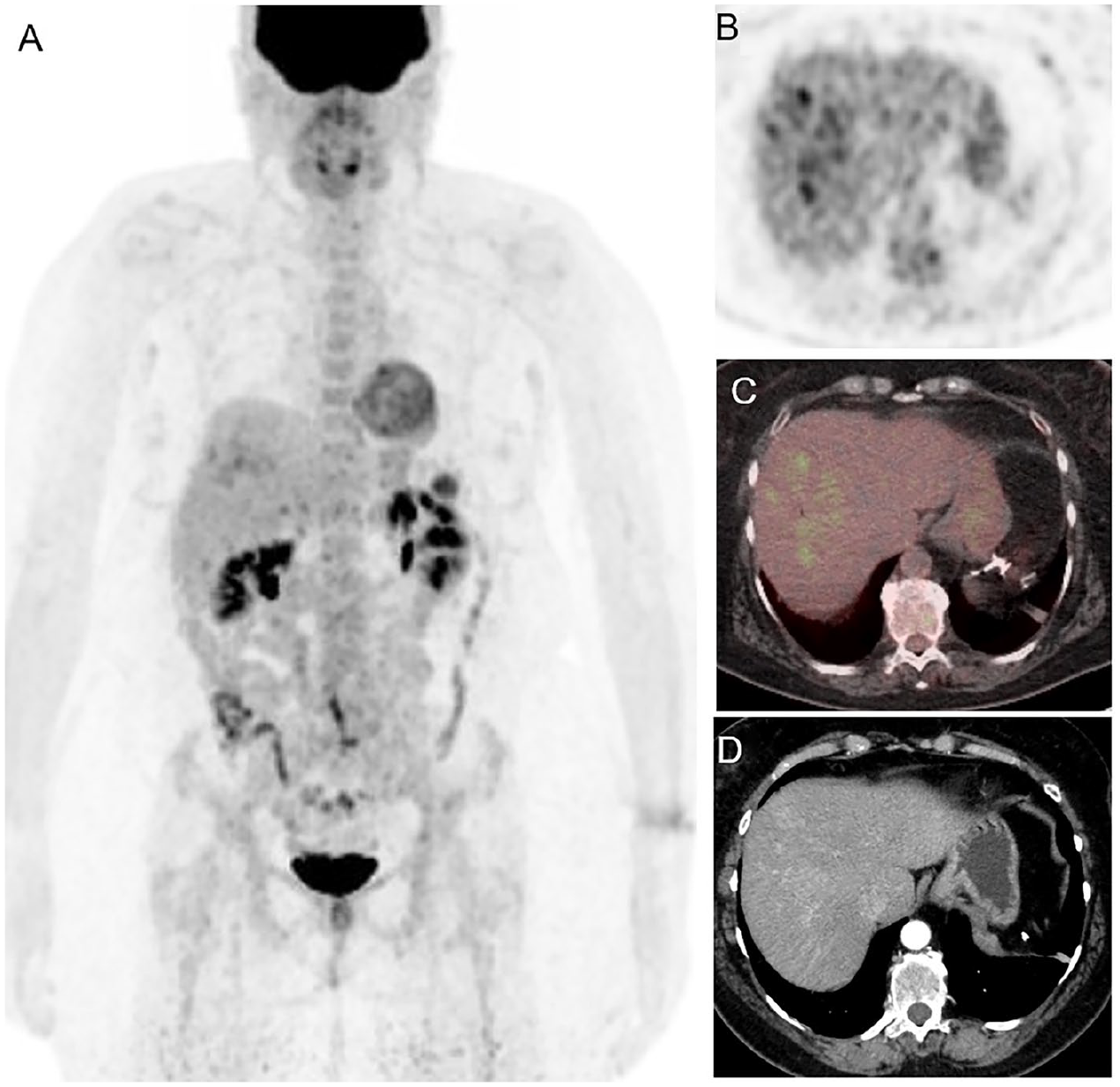
Sixty-five-year-old female patient with history of mucosal anorectal melanoma on combined immunotherapy. Follow-up 18F-FDG PET CT (A) MIP, (B, C) axial PET, (D, E) axial fused PET CT, (F, G) correlative contrast enhanced CT, showed diffusely increased uptake in both adrenal glands which appear enlarged without a definitive CT correlation. Findings were suggestive of immune-related adrenalitis, which was clinically confirmed after PET CT.
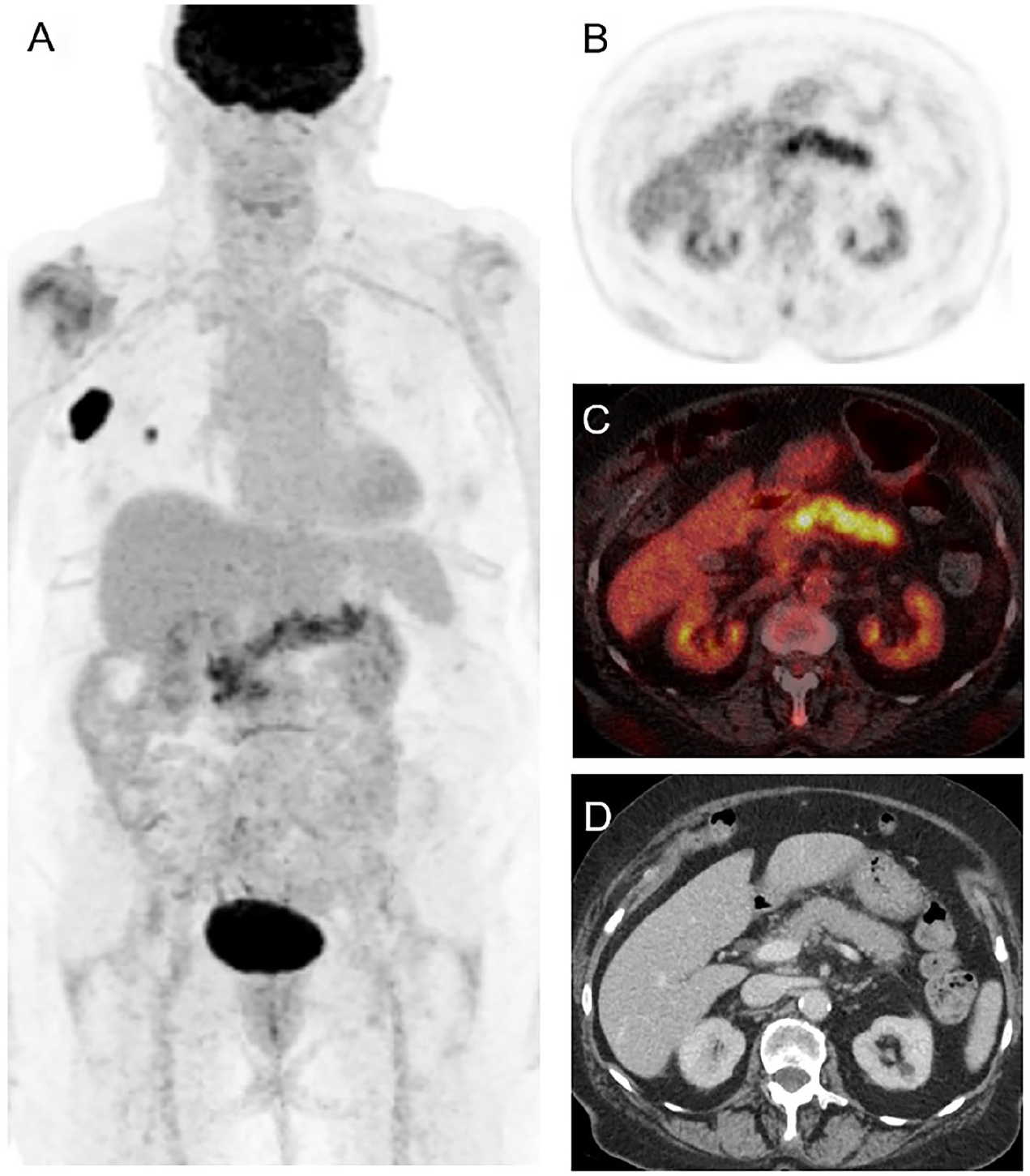
Seventy-six-year-old female patient with history of NRAS mutated melanoma on immunotherapy with nivolumab. Follow-up 18F-FDG PET CT (A) MIP image, showing persistent metabolically active disease in the right chest wall and axilla. (B) axial PET, (C) axial fused PET CT, (D) correlative contrast enhanced CT, showing diffusely increased FDG uptake within the pancreas with associated mild edematous changes, which was consistent with the clinical diagnosis of pancreatitis with elevated pancreatic enzymes.
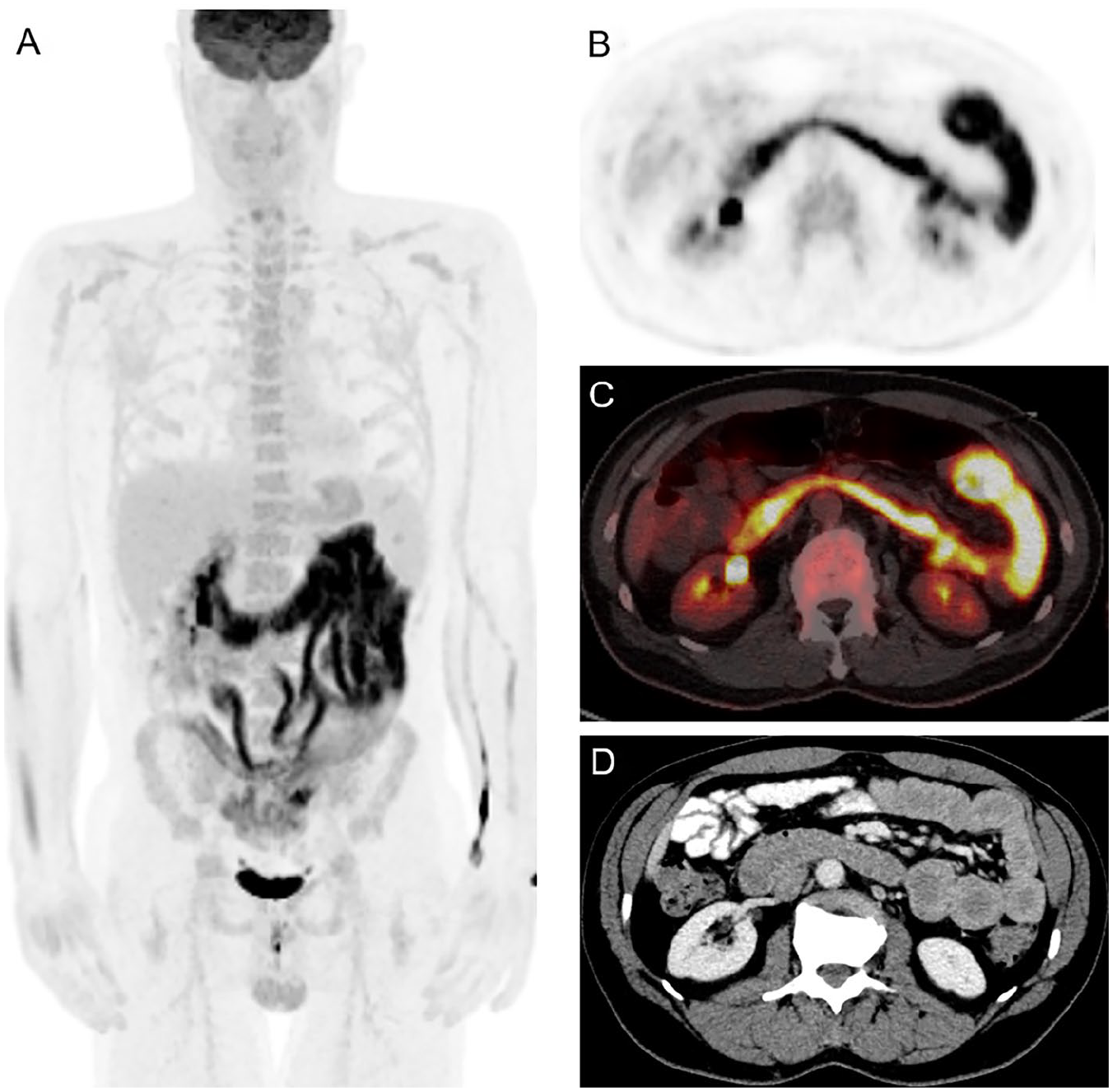
Forty-nine-year-old male patient with history of BRAF mutated melanoma on combination immunotherapy. Follow-up 18F-FDG PET CT (A) MIP, (B) axial PET, (C) axial fused PET CT, (D) correlative contrast enhanced CT, showed metabolically active circumferential wall thickening throughout the jejunum with mild dilatation, consistent with the clinical suspicion of immune-related enteritis.
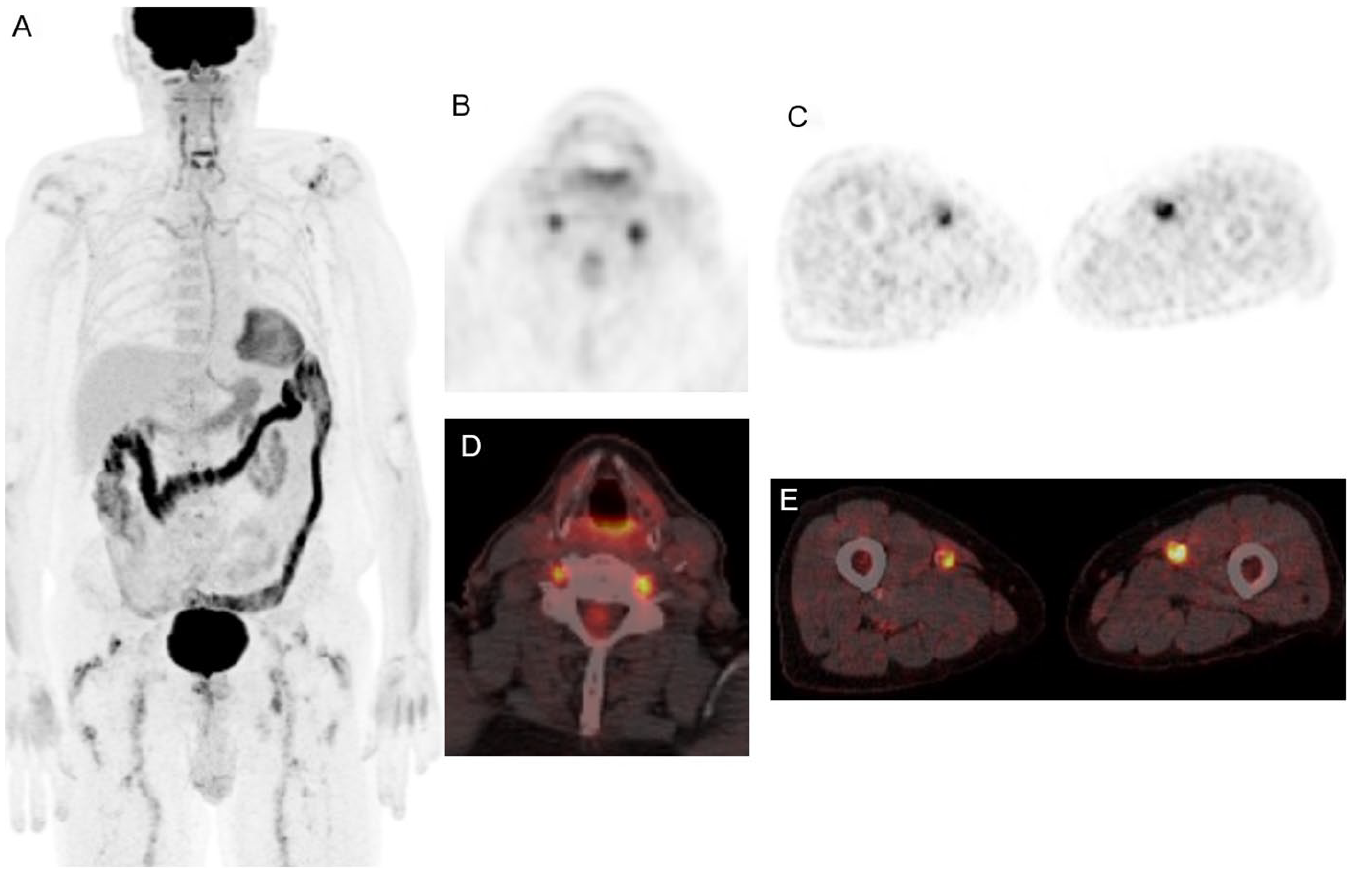
Eighty-year-old male patient with history of cutaneous melanoma on immunotherapy with nivolumab. Follow-up 18F-FDG PET CT (A) MIP, (B, C) axial PET, (D, E) axial fused PET CT showed diffusely increased FDG uptake in the bilateral vertebral and femoral arteries, which was consistent with posteriorly confirmed immune-related vasculitis.
Thirteen out of 39 (33.3%) cases identified on PET/CT were not suspected clinically but confirmed by the reference standard; these corresponded to sarcoid like reaction (4/13), pneumonitis (4/13), thyroiditis (2/13), colitis (1/13), hepatitis (1/13), myositis (1/13).
The most frequent IRAEs, both suspected clinically and on PET/CT, corresponded to thyroiditis and colitis (Table 3). Among the PET/CT positive cases, the majority corresponded to grade 2 severity according to CTCAE, as seen in Table 4. Grade 3 and 4 IRAEs, requiring temporary or definitive discontinuation of immunotherapy and treatment with steroids, corresponded to: colitis 2/10 (20%), pancreatitis 2/10 (20%), pneumonitis 2/10 (20%), sarcoid-like reaction 2/10 (20%), adrenalitis 1/10 (10%), and enteritis 1/10 (10%).
Distribution and Frequency of Immune Therapy Related Adverse Events.
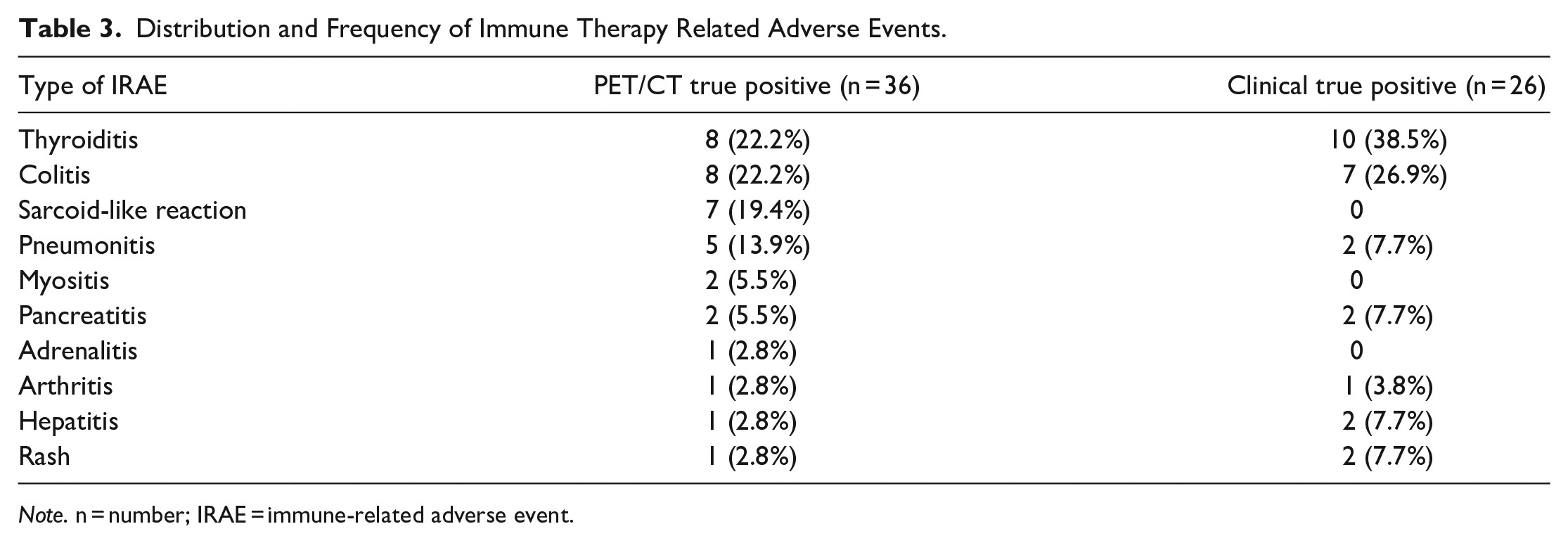
Note. n = number; IRAE = immune-related adverse event.
Severity of Positive PET/CT Immunotherapy Related Adverse Events According to the Common Terminology Criteria for Adverse Events.

Note. n = number; IRAE = immune-related adverse event; CTCAE = Common Terminology Criteria for Adverse Events.
Regarding immunotherapy regimen, 24/36 (66.7%) of the events presented while receiving single-agent therapy as follows: 12/24 (50%) with nivolumab, 11/24 (45.8%) with pembrolizumab, and 1/24 (4.2%) with ipilimumab. 12/36 (33.3%) occurred under combined therapy with ipilimumab/nivolumab.
PET/CT diagnostic yields for each IRAE are listed in Table 5, with the highest yields seen for thyroiditis, colitis, and sarcoid-like reaction.
PET/CT Diagnostic Yield for Each Immune Therapy Related Adverse Event With 95% CI.
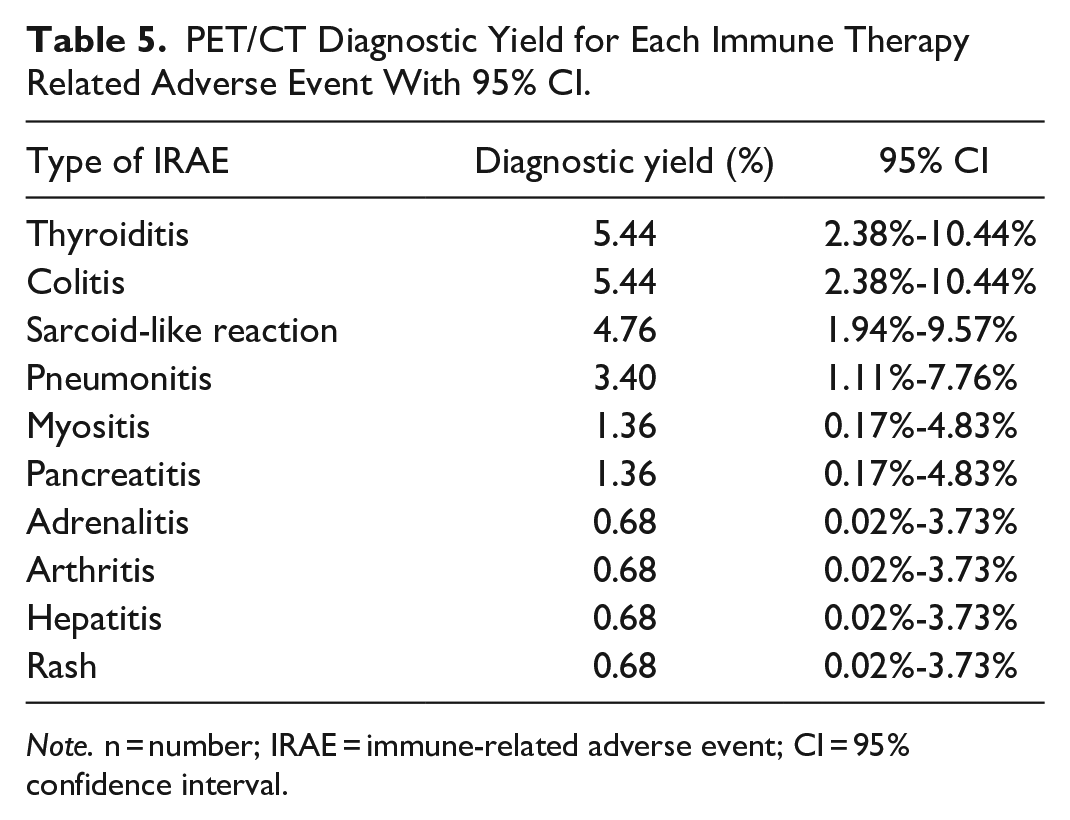
Note. n = number; IRAE = immune-related adverse event; CI = 95% confidence interval.
Discussion
The upregulation of immune pathways with monoclonal antibodies (mAbs) or ICPIs has altered treatment management and survival outcomes of patients with metastatic melanoma in the past decade. However, these therapeutic options are not exempt from a wide variety of adverse events which potentially can affect any tissue in the body, regardless of patients age, gender, and general status. The clinical incidence of IRAEs has been reported to be variable and there are still many unknowns of how, when, and why they occur. Some of these IRAEs have mild presentation allowing continuation of therapy, while others have a severe presentation; some IRAEs occur early during treatment, while others long time after completing immunotherapy.5,6,11
The clinically reported incidence of IRAEs is variable and has been described in up to 80% to 90% of patients treated with ICCIs. In a meta-analysis by Wang et al, 7 including 46 studies representing 12 808 oncologic patients treated with immunotherapy, the overall incidence of IRAEs was of 26.8% (7.8), which is in line with our results with an incidence of 24.5%. Another recent study 26 including 4489 patients with a mean age of 74.9 years, with metastatic melanoma under immunotherapy, reported a cumulative incidence of IRAEs of 46.8% at 6 months of therapy. In our cohort, the overall incidence of IRAEs on PET/CT was of 18%. The wide variability in the clinical incidence of IRAEs among different studies, may be influenced by the type of immunotherapy regimen. 5 A recent meta-analysis from 2022 27 including 2 main randomized trials with a total of 1853 patients (Keynote 006 trial-NCT01866319 and Keynote 054 trial-European Organization for Research and Treatment of Cancer [EORTC] 1325), found an incidence of IRAEs of 28% in patients under single therapy with pembrolizumab, 10% with ipilimumab single agent, and 37% in patients treated with pembrolizumab versus placebo (7%). In our cohort, the majority of PET/CT positive IRAEs presented while receiving single-agent therapy (66.7%), being nivolumab the most common associated agent.5,8 The higher incidence of adverse effects in the single-agent group, contrary to most literature, is likely due to the higher proportion of patients receiving single-agent therapy in our cohort (73.6%). Given that single-agent therapy was more common during the earlier years of our study period (2015-2017), this temporal trend may have influenced our findings. Further research should consider stratifying patients by treatment era to account for changes in clinical practice over time.
The most frequently encountered IRAEs were thyroiditis, colitis, and sarcoid-like reaction representing 22%, 22%, and 19%, respectively. This is in line with what is reported in the literature, although again, reports are mostly clinical, not imaging related.5-7,28 Note is made that not many rash cutaneous IRAEs were reported, maybe because of inherent difficulties of the PET detecting subtle cutaneous inflammation.
In recent years, PET/CT has become a central diagnostic tool for the staging and restaging of patients with melanoma, particularly for therapy response assessment. Also, given its inherited high sensitivity to detect inflammation, it can potentially identify IRAEs in up to 61% to 70% of cases as described in the literature.3,4 A recent study by Andersen et al determined moderate to high sensitivity of PET/CT for detection of IRAEs, others than skin and heart conditions, supporting that a systematic assessment can help in early detection. 29 Most importantly, other studies have shown that in up to 33% of cases, inflammatory changes can be detected on imaging, particularly PET/CT, before the patient develops any symptoms to raise clinical suspicion.8,16 In our cohort, among the positive PET/CT findings, 33.3% of the cases were subclinical but confirmed by the reference standard after PET/CT findings, similar to what has been described in the literature. It is important for the reporting radiologists/nuclear medicine physicians to be aware of the IRAEs in this patient population and not to misinterpret them as disease progression.
Regarding severity, the majority of IRAEs detected on PET/CT (58%) were grade 2, similar with has been described in the literature; however, the frequence of grade 3 and 4 IRAEs was 28%, compared to 15% in the literature.3,4 This may be due to the retrospective nature of the current study, with possible under documentation of mild/grade 1 IRAEs in clinical notes, including cutaneous cases.
Regarding false negative and false positive PET/CT findings, these may be expected given the non-specific nature of presentation and imaging findings which can also be easily mistaken for physiological or inflammatory changes secondary to other entities. These may include skin rash, mild myositis, cardiac inflammation, hematological conditions such as anemia, among others.
Furthermore, several publications have demonstrated a positive association between the development of IRAEs and outcomes in patients on ICIs, across different tumour types, including advance melanoma.30,31 These have shown a direct relation between the developed IRAEs and incremental gains in survival outcomes, which is explained by the hypothesis that both the anti-tumour response and the development of IRAEs are representative of a robust immune reaction, where self-reactive T cells infiltrate both the tumour and the organs that develop IRAEs. Differences have been described in the median progression free survival of 9.2 and 4.8 months for patients with or without IRAEs, respectively. 32 This hypothesis supports the importance of the radiologist being able to detect IRAEs early during treatment, since they could represent a potential predictor of therapy response and outcomes. However, large-scale prospective data is needed to prove this concept, where the high sensitivity of PET/CT to detect IRAEs in whole body images may play a crucial role. As a future project, we are considering long term follow up of this cohort to correlate presence or absence of IRAEs with survival outcomes.
There are several limitations in the study that must be acknowledged. First, there are inherent drawbacks, due to the retrospective nature of this study. The reference standard is reliant on what has been documented in clinical notes, rather than prospectively collected. Second, the reported incidence of IRAEs in most studies is mainly clinical and, although their imaging findings have been widely described, its specific incidence in each imaging modality, including PET/CT, has not been clearly assessed to date. Third, the clinical severity was not explicitly assigned in the clinical notes of this group of patients and was determined by consensus considering treatment and clinical follow-up, which can lead to poor differentiation between grades 1 and 2, specifically, which can explain the unexpected low incidence of reported mild rash/cutaneous reactions in our cohort.
Conclusion
18F-FDG PET/CT has a high sensitivity and specificity for the detection of IRAEs in patients with metastatic melanoma under immunotherapy and can detect clinically unsuspected IRAE in up to 33% of cases, with significant implications for patient management.
Footnotes
Acknowledgements
Natalia Rodriguez Moreno, epidemiologist.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
