Abstract
There are few recommendations in Canada to assist clinicians in selecting appropriate biopsy techniques (fine-needle aspiration, core-needle biopsy, vacuum-assisted biopsy, vacuum-assisted excision) and imaging technologies (mammography, ultrasound, magnetic resonance imaging, contrast-enhanced mammography) for biopsy guidance. Limited existing recommendations from other countries do not consider the unique aspects of the Canadian healthcare system. To address this gap, 17 experts participated in a modified Delphi panel to reach consensus on biopsy-related topics and provide recommendations. The panel was comprised of 12 radiologists, 2 pathologists, and 3 surgeons from 6 provinces across Canada. Panelists engaged in two rounds of anonymized voting, with an in-person discussion held between the rounds. The modified Delphi panel adhered to best practices, including establishing consensus definitions prior to voting, utilizing anonymized voting, and abstaining from communication among panelists before the in-person meeting. A rigorous statistical approach was utilized to analyze the points of agreement and disagreement. Consensus findings covered a wide range of topics, including recommendations for initial biopsy technique based on lesion type and imaging modality, patient management or rebiopsy considerations after the initial biopsy, procedural recommendations (i.e., gauge size, number of samples), patient considerations (i.e., drug allergies, pregnancy). Overall, 347 individual items were included in the final analysis, 286 (82%) of which achieved consensus. These consensus recommendations intend to offer general recommendations to help standardize and improve practices across Canada and were endorsed by the Canadian Society of Breast Imaging. However, they should be evaluated in the context of each individual case and emerging evidence.
Introduction
Breast cancer is the most prevalent cancer among women in Canada.1,2 Given the high incidence, accurate diagnosis is important to enable timely treatment. One such method is the image-guided needle biopsy, a well-established, minimally invasive diagnostic technique for suspicious breast lesions. 3 It is a less expensive alternative to diagnostic surgical excisions, which are known to have higher rates of adverse events and less favourable cosmetic outcomes. 3 Fine-needle aspiration (FNA), core-needle biopsy (CNB), and vacuum-assisted biopsy (VAB) are 3 common biopsy techniques. Vacuum assisted excision (VAE) is an emerging option, mainly recognized for its role as an alternative to surgical excision in select cases. 4 These interventional techniques are conducted with the assistance of imaging technologies such as mammography, ultrasound (US), magnetic resonance imaging (MRI), or contrast-enhanced mammography (CEM). The choice of sampling technique and imaging technology is driven by the location, type, and size of the lesion, as well as the availability and cost of biopsy equipment. Current literature reports a wide range of sensitivity (0.72-1.00) and specificity (0.76-1.00) for these approaches, depending on the specific biopsy technique and lesion characteristics.3,5,6
Although most clinicians are aware of the advantages and disadvantages of biopsy techniques, formal recommendations are important to standardize practice. Modified Delphi panels are commonly used to develop clinical practice guidelines where clear guidance is missing or not sufficient. This is achieved through anonymous voting rounds, open discussions, and subsequent re-voting among a panel of experts. 7
To date, there are limited Canadian recommendations to aid clinicians in deciding which techniques are appropriate for use based on varied clinical scenarios, and existing recommendations from other countries do not account for the nuances of the Canadian healthcare system. Recommendations tailored to the unique aspects of the Canadian healthcare system are needed to ensure every patient receives optimal treatment. This paper reports the consensus recommendations from an expert panel of Canadian radiologists, pathologists, and surgeons regarding optimal practices for breast biopsy using a modified Delphi approach.
Methods
This paper adhered to the ACCORD guidelines for transparent reporting of methods. The checklist can be found in Supplemental Materials.
Participants
The panel comprised 12 radiologists, 2 pathologists, and 3 surgeons. Panelists were invited to participate by email after being selected based on their clinical and research experience. The aim was to create a diverse panel based on discipline, geography, and gender to capture distinct perspectives. All panelists completed fellowships specializing in breast within their respective fields—radiology, pathology, or surgery—except for one, who received specialized clinical training during residency due to the unavailability of fellowships at the time. Their experience ranges from 4 to 23 years, with an average of 11.75 years. Most panelists (n = 13) work in academic centres, 2 work exclusively in community settings, and 2 work in both academic and community settings. Two panelists were designated as the panel leads (ZSK, CW), and their responsibilities included developing the consensus questionnaire and facilitating the discussion. A modified Delphi expert and one medical writer supported the consensus panel but did not participate in voting. Measures were taken to ensure the study sponsor did not impact the content and findings. Specifically, the study sponsor supported meeting logistics but did not influence the content of the survey questions, resource material, or references, and was not present in the room during consensus discussions. Panelists were reimbursed for travel costs but were not compensated for their time or study involvement.
Study Design and Preparation
A two-round modified Delphi process was used (Figure 1). An initial survey was administered to panelists to assess their familiarity with and access to breast biopsy techniques. Concurrently, a targeted literature review was conducted to inform the questionnaire development for voting. The review focused on current guidelines and best practices from other geographies, along with recent comparative evidence of breast biopsy approaches. Articles were identified using multiple databases (PubMed, Embase, and Google Scholar). Priority was given to recent papers published in English. The findings of the review presented an overview of the current breast biopsy landscape and identified key issues and controversies within the field. Panelists were provided with the literature review findings, including links to relevant articles.
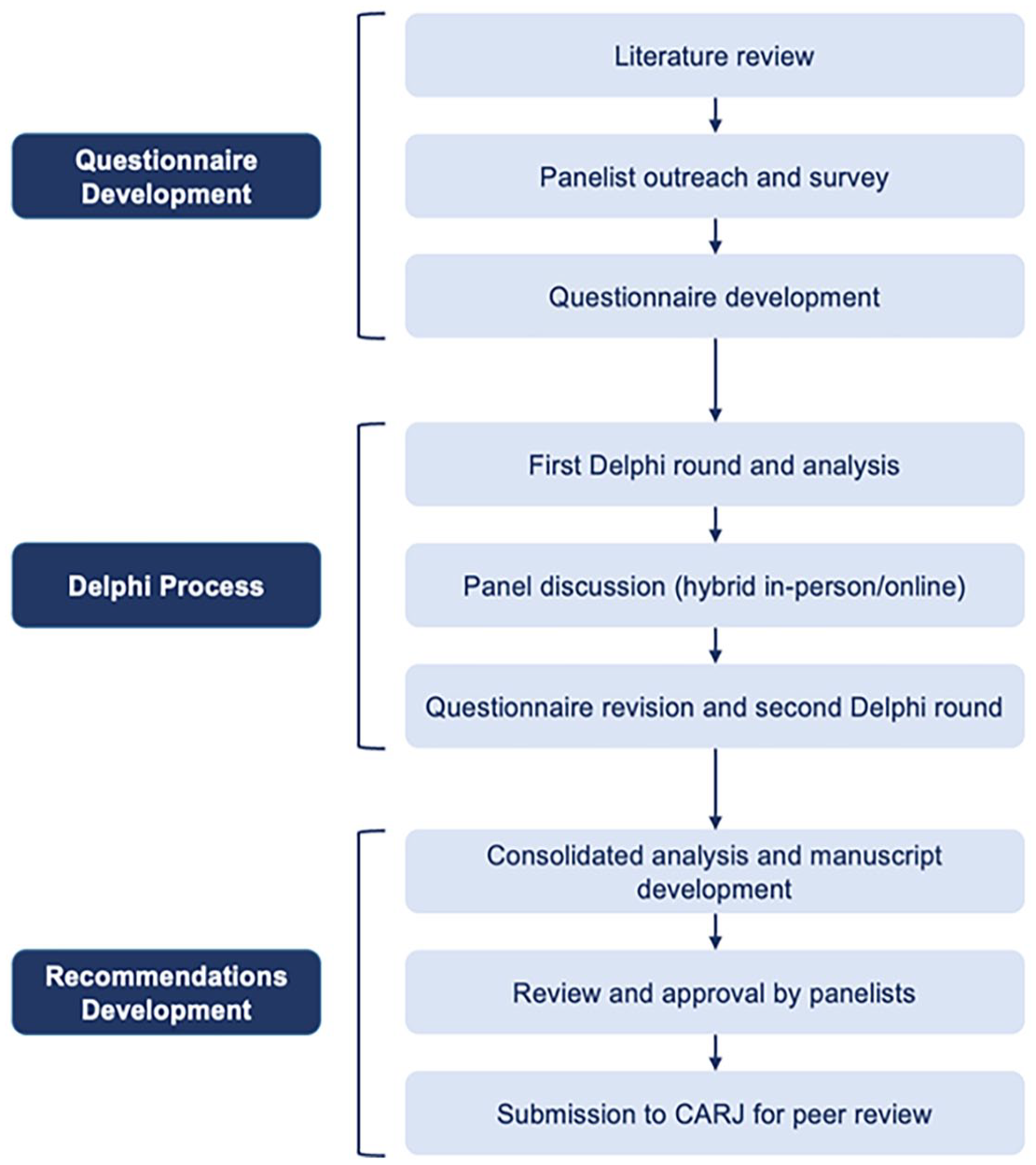
Study flowchart.
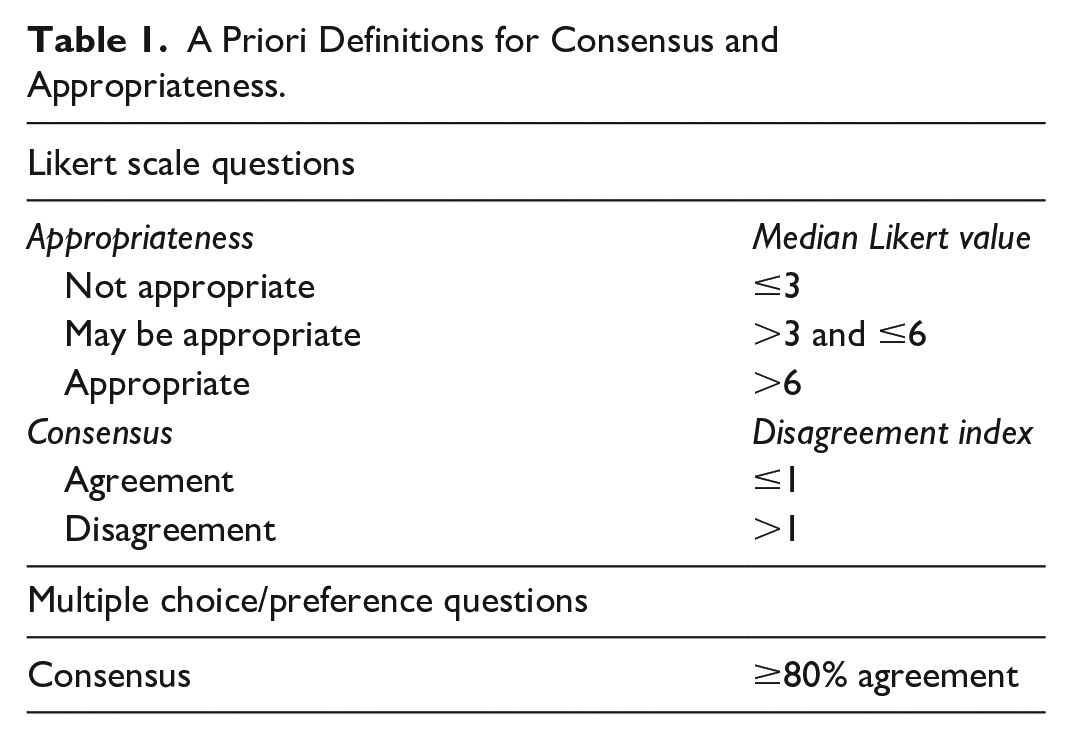
Informed by the literature review and initial participant survey, a questionnaire was developed for first-round voting. The questionnaire included Likert-scale, multiple-choice, and ranking questions. For questions that were not relevant to all specialties (radiology, pathology, surgery), an extra answer option, “not relevant to my practice,” was provided. Prior to voting, consensus and appropriateness criteria were clearly defined for each question type (Table 1). For multiple-choice and ranking questions, the agreement threshold of 80% was chosen based on relevant literature.8,9 For Likert-scale questions, appropriateness was assessed using pre-determined median ranges. Items with a median score of 3 or below were deemed not appropriate, while scores between 3 and 6 fell into a middle range, neither appropriate nor not appropriate, and were considered may be appropriate. Scores above 6 were considered appropriate. Consensus was determined using the Inter-percentile Range Adjusted for Symmetry approach and disagreement index based on previous literature. 7 The questionnaires for both voting rounds were developed and administered through a secure, web-based survey platform (SurveyMonkey). The results were collected anonymously, exported and analyzed by the non-voting observers using Microsoft Excel (Version 2405 Build 16.0.17628.2006). The questionnaire is provided in Supplemental Materials.
A Priori Definitions for Consensus and Appropriateness.
Voting Rounds and Panel Discussion
First-round voting was held remotely before any interaction among panelists and following dissemination of the literature review. This initial round focused on the panelists’ present clinical practice patterns and yielded baseline data on items for which consensus was already present, and items that required further discussion. Following first-round voting, an in-person meeting was held in Montreal, Canada, in April 2024, with 6 panelists participating virtually. The panelists were provided a summary of the anonymized first-round voting results to review prior to discussion, which included vote counts and whether consensus was achieved. Panel discussions were deliberately centred on topics where consensus was not reached in the first-round, and every panelist was given an opportunity to provide insights. Based on the discussion, the questionnaire was refined as needed for second-round voting, which only included items that had not achieved consensus. At the end of the in-person meeting, panelists were invited to participate in the second and final voting round to minimize the risk of attrition.
Results
All participants (N = 17) completed both surveys, except one participant who did not respond to 3 questions in the second round. Panelist specialties and practice regions are provided in Table 2. Overall, 347 individual items were included in the final analysis, 286 (82%) of which achieved consensus based on a priori criteria detailed in Table 1. The findings are presented in 3 sections: (1) Recommendations for initial biopsy based on lesion type and imaging modality; (2) Patient management or re-biopsy considerations after the initial biopsy; and (3) Procedural recommendations (ie, gauge size, number of samples), patient considerations (ie, drug allergies, pregnancy), and other specific recommendations to guide decision-making. The key consensus findings and takeaways are summarized in Table 3.
Panelist Characteristics.
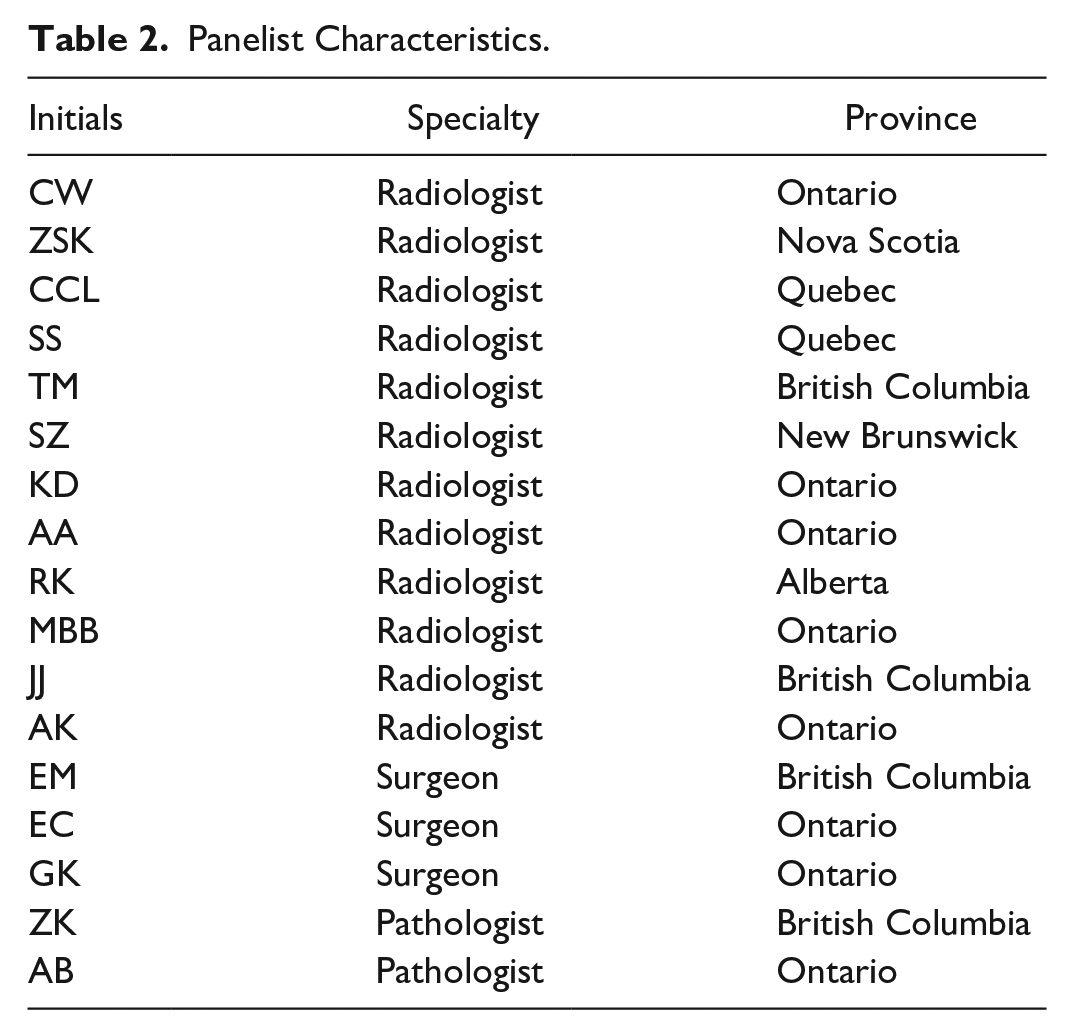
Consensus Summary.
Green: agreement (disagreement index ≤1) and appropriate (median score >6); yellow: agreement (disagreement index ≤1) and may be appropriate (median score >3 and ≤6); red: agreement (disagreement index ≤1) and not appropriate (median score ≤3); blue: agreement not reached (disagreement index >1).
Definitions
For this panel, needle biopsy techniques are defined based on previous literature and current clinical practices.4,10 Briefly, FNA involves inserting a small needle (typically 18G or smaller) into the area of concern to collect a small sample of fluid or tissue for cytology analysis, while CNB uses a larger hollow needle to extract small cylindrical tissue samples (i.e., cores) from the lesion for histologic analysis. VAB uses a larger, lower-gauge needle inserted through a hollow probe; suction is then used to draw the breast tissue into the probe, where a rotating internal blade cuts and collects a large sample. Although not its primary purpose, VAB may occasionally remove the entire lesion, particularly when small. Surgical excision is the most invasive approach, including the removal of part or the entirety of a lesion in the surgery setting. VAE is similar to VAB, with a clear aim to remove the lesion, offering a less invasive alternative to surgical excision. Typically, the VAE approach uses an 8- or 9-gauge needle to obtain a sample of around 4 g, aiming for a comparable sample size to surgical excision. 11 The number of samples collected varies based on the device and needle size used. 4 This novel approach is being integrated into practice in various regions, including Europe. 12 Notably, panelists were also given “surgical referral” as a voting option on various items, which was considered distinct from “surgical excision.” This option recommends surgeon input for the diagnosis or management of the lesion. Similarly, in some cases, panelists were provided with “VAE with surgical consult” as a voting option, meaning VAE could only be used after consulting with surgeons. Worth noting, in certain centres, surgical referrals can also involve multidisciplinary teams, including breast specialists or dedicated breast centres, depending on local practices and care pathways.
Recommendations for Initial Biopsy
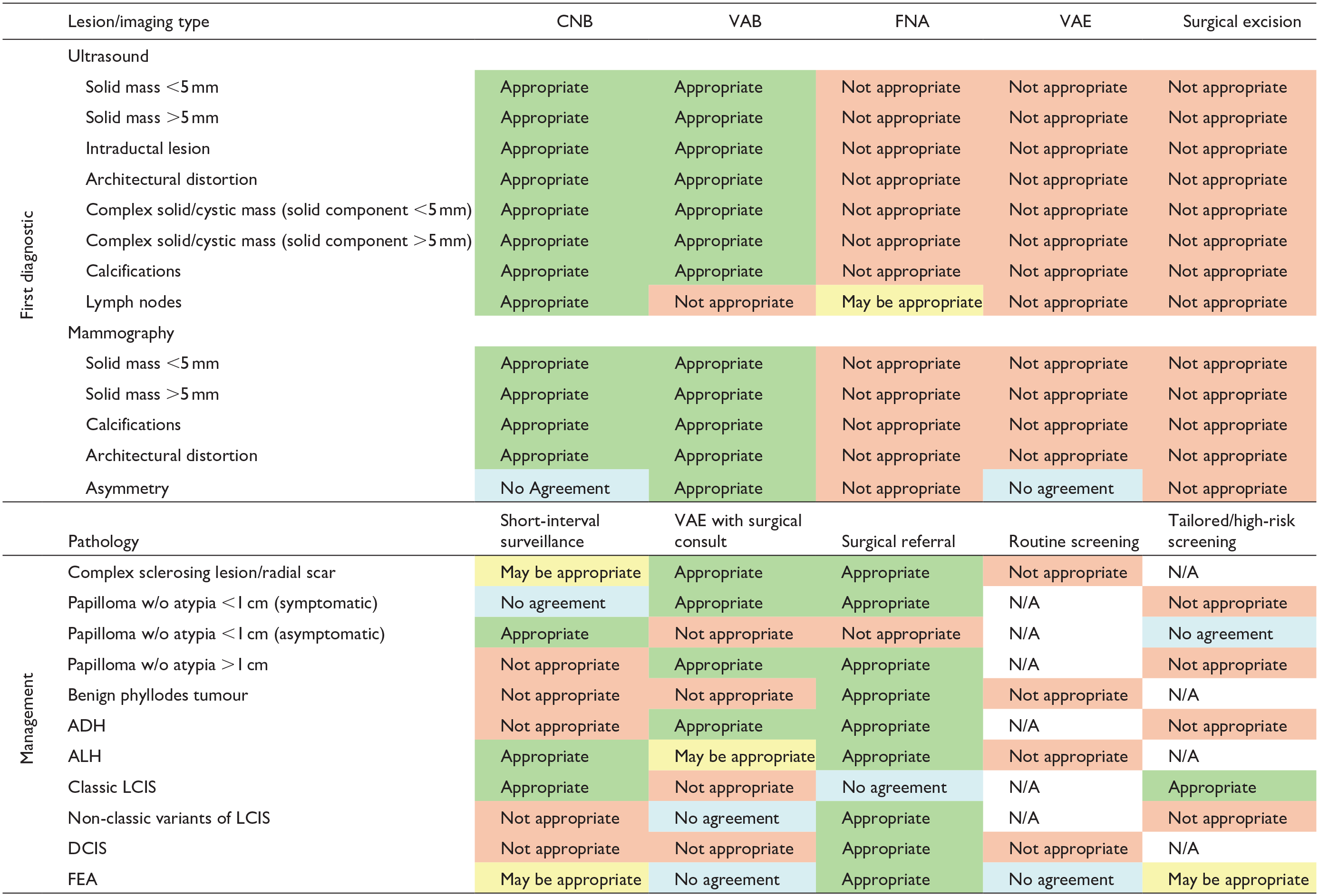
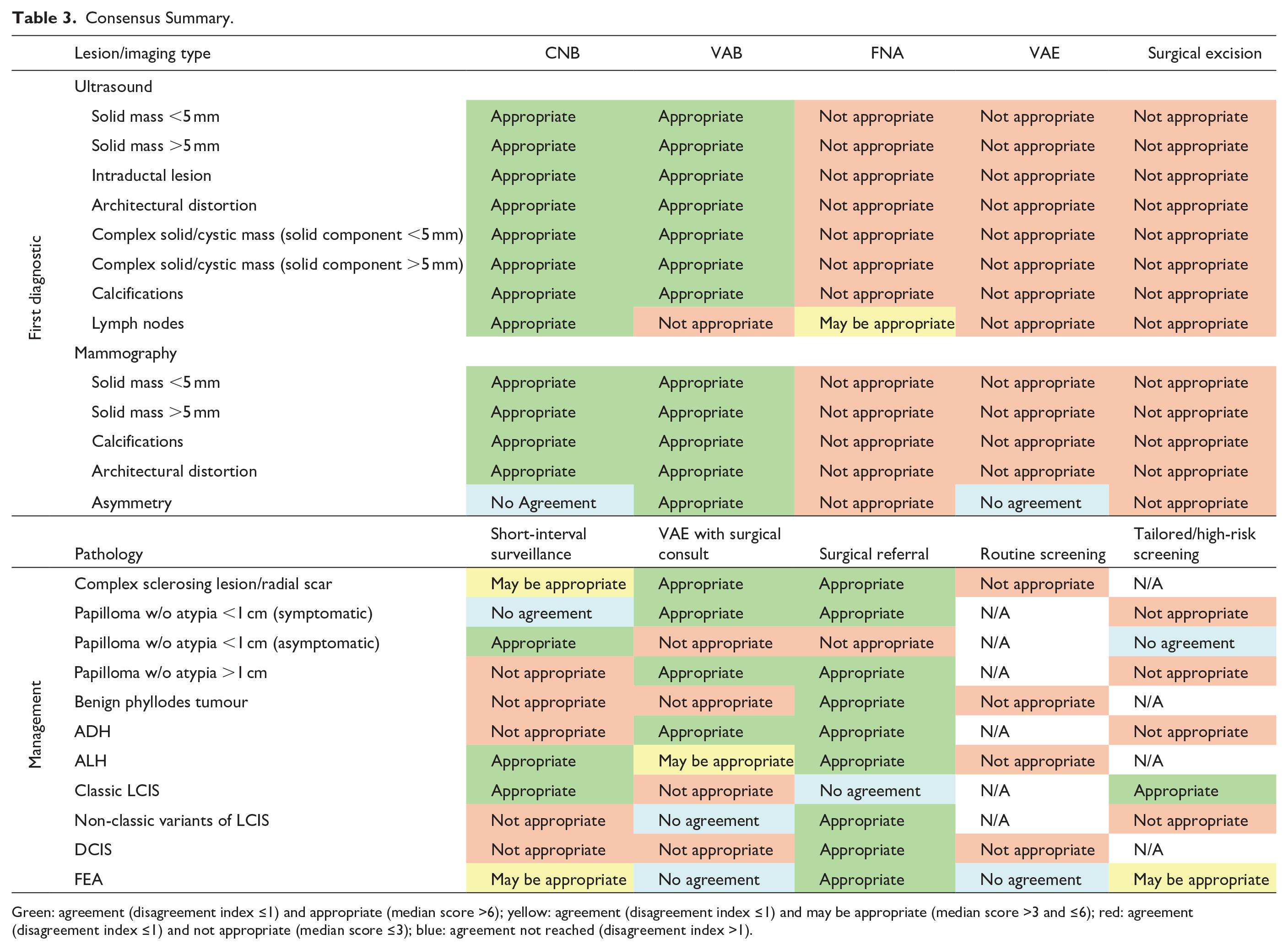
The panelists provided insights on selecting the best approach for initial biopsy based on imaging modality. These recommendations are summarized in Figure 2 for lesions visible under US, Figure 3 for mammography, and Supplemental Table 1 for MRI/CEM.

Appropriateness of US-guided biopsy techniques by lesion type.
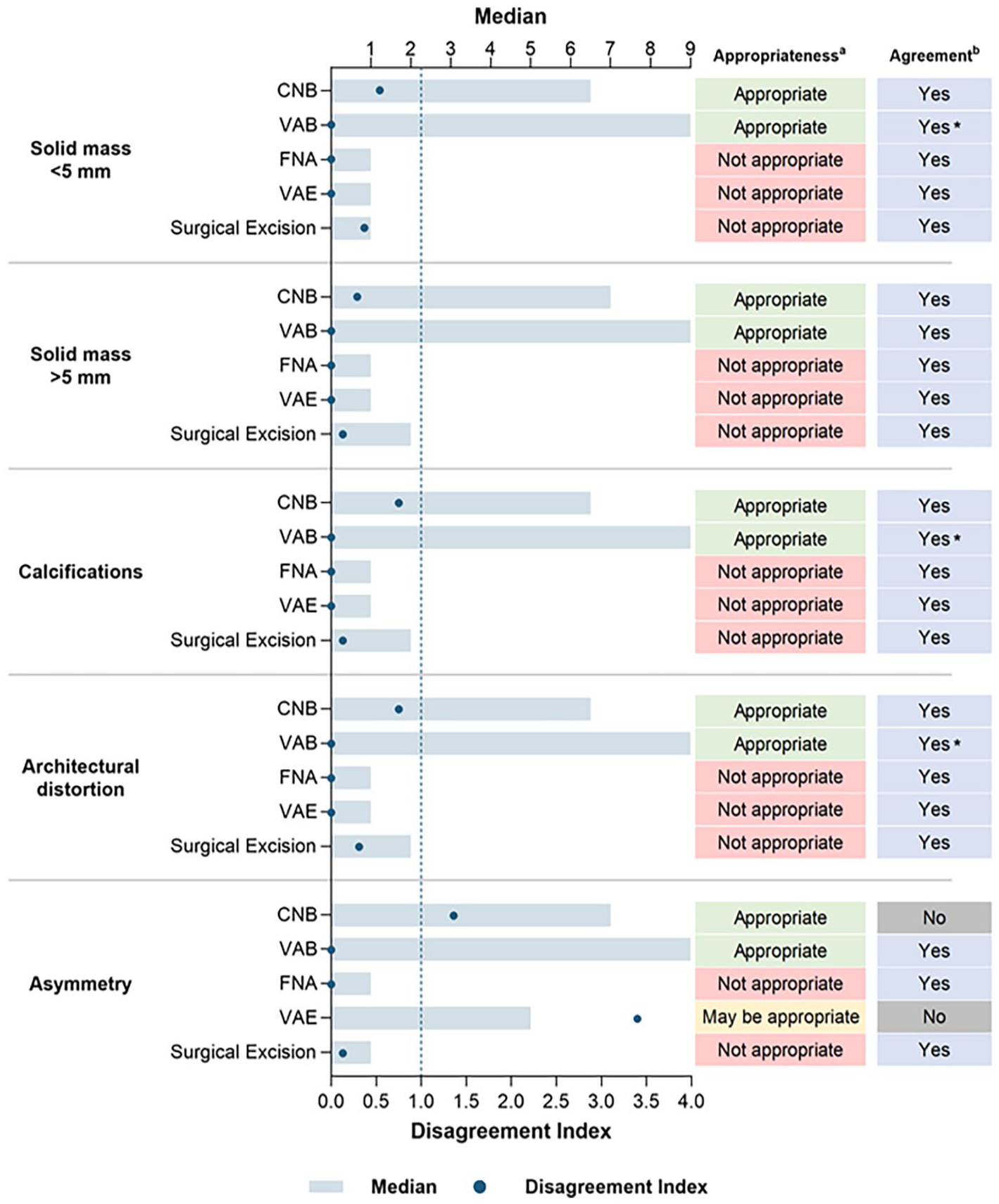
Appropriateness of mammography-guided biopsy techniques by lesion type.
Ultrasound
There was unanimous panelist agreement that a biopsy should be conducted preferentially under US guidance when a lesion is visible on US. Both CNB and VAB were considered appropriate across all lesion types in the breast under US except for lymph nodes, for which only CNB was considered appropriate, and FNA was considered may be appropriate.
Mammography
For lesions detected on mammography, VAB and CNB were deemed appropriate for all types except asymmetry. In cases of asymmetry, the panelists agreed on only VAB as the appropriate choice. When both techniques are available to the provider, the panel unanimously recommended VAB over CNB. A biopsy of axillary lymph nodes under mammographic guidance was not considered appropriate and, therefore, not included for voting.
MRI/CEM
MRI/CEM were grouped together for voting as they both utilize intravenous contrast uptake for lesion identification and targeting. For lesions observed under MRI/CEM where no subsequent correlate was identified on mammographic or sonographic assessment, VAB and VAE were considered appropriate techniques, while CNB was identified as “may be appropriate,” especially when there is limited access to VAB.
Recommendations for Post-Initial Biopsy
Panelists provided recommendations regarding post-initial biopsy considerations for high-risk/noninvasive concordant pathology, that is, lesions that may have a high upgrade rate at surgical excision or association with malignancy. These include complex sclerosing lesion (CSL)/radial scar (RS), symptomatic papilloma without atypia <1 cm, asymptomatic papilloma without atypia <1 cm, papilloma without atypia >1 cm, benign phyllodes tumour, atypical ductal hyperplasia (ADH), atypical lobular hyperplasia (ALH), classic lobular carcinoma in situ (LCIS), non-classic variants of LCIS, ductal carcinoma in situ (DCIS), and flat epithelial atypia (FEA). The results for lesion management are detailed in Figure 4, and re-biopsy considerations are provided in Supplemental Table 2. Various other high-risk lesions were not included in the scope of this consensus due to their rarity and evolving upgrade rate data that emerge as more lesions are encountered in clinical practice. 13

Appropriateness of management methods by pathology.
For lesion management, surgical referral, not considered equivalent to or necessitating surgical excision, was appropriate for all pathologies except: (i) papilloma without atypia <1 cm (asymptomatic), where referral was deemed not appropriate as it was not felt to be necessary, and (ii) classic LCIS, where referral may not be necessary. Nevertheless, breast surgeons were considered the most appropriate specialists to help guide management and follow-up decisions as well as provide an overall risk assessment. VAE following surgical consult was considered appropriate for managing CSL/RS, symptomatic papilloma without atypia <1 cm, papilloma without atypia >1 cm, and ADH. Panelists also agreed surveillance was an appropriate approach for managing asymptomatic papilloma without atypia <1 cm, classic LCIS, and ALH without the need for surgical referral. In cases of discordant pathology following CNB, panelists suggested re-biopsy using VAB, VAE, or CNB with a lower gauge needle (i.e., larger sample). For discordant pathology following VAB, re-biopsy should be performed using VAE, surgical excision, or VAB with the same or lower gauge (i.e., larger sample size).
Procedural Recommendations and Patient-Specific Considerations
Panelists opined on specific biopsy approaches, including gauge size, number of samples, clip placement, local anaesthetic use, and considerations for specific patient groups. Figure 5 details gauge size and sample considerations, and Supplemental Table 3 provides other procedural and patient-specific recommendations.
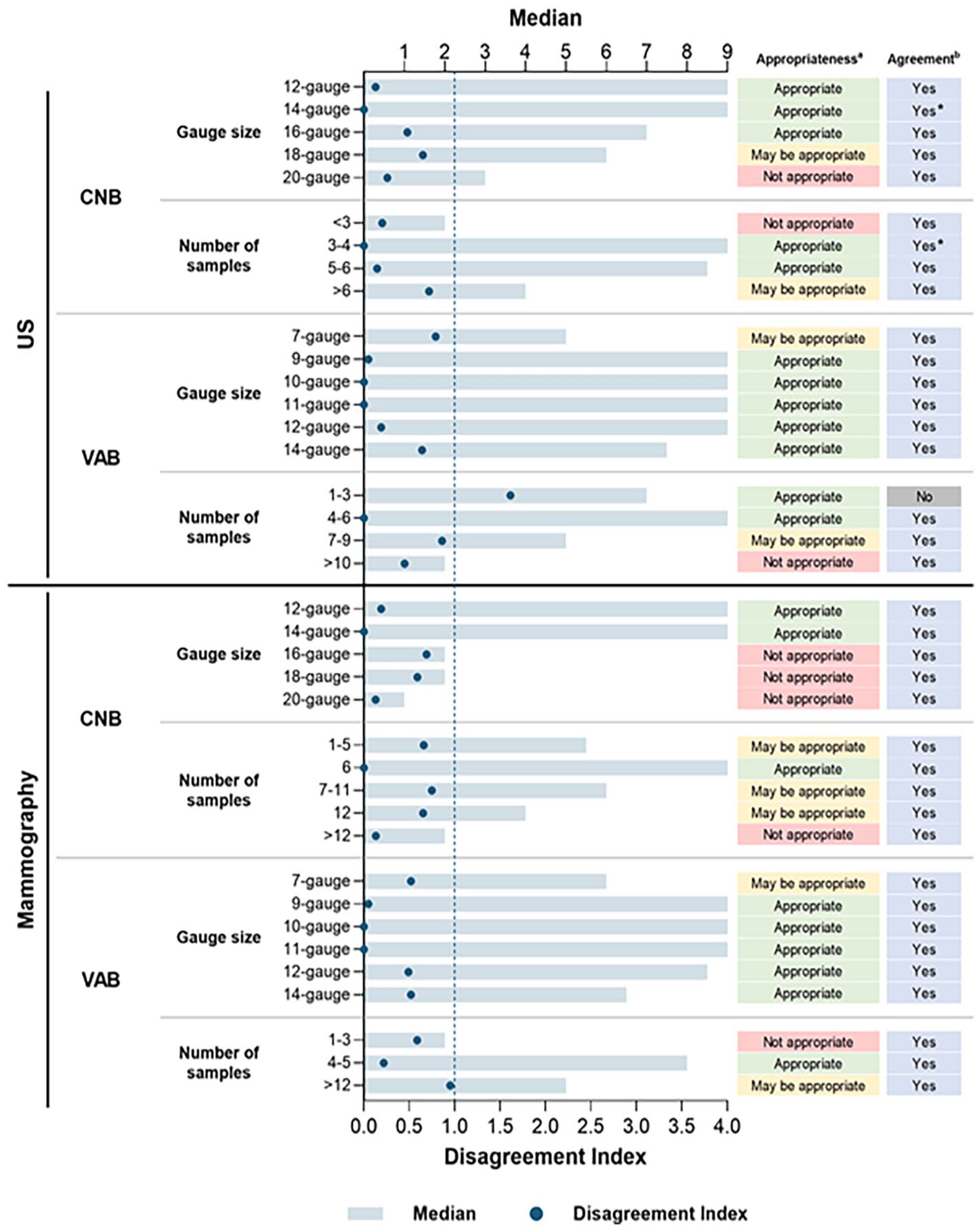
Appropriateness of gauge size and sample quantity for CNB and VAB.
Biopsy Sample Size and Number
In general, panelists agreed a lower needle gauge provides samples with better diagnostic quality, and more core samples increase the likelihood of diagnostic success for both CNB and VAB. The recommended number of samples for VAB and CNB on either US or mammography ranged from 3 to 6. Panelists agreed no further sampling is warranted if the lesion is no longer visible after the initial passes. To note, the number of samples was voted on independently of needle size, resulting in general recommendations rather than specific sample numbers for each needle size.
Clip/Marker Utilization
Clip placement was considered appropriate across all visual modalities and lesion types. The only exception was for biopsies of low-suspicion lesions with US-guidance, for which clip placement was considered may be appropriate, that is, if malignancy risk is low and the lesion is easy to relocate, clip placement may not be needed. However, it is recommended for small (eg, <1 cm) lesions, which may be harder to relocalize, regardless of suspicion level. Clip placement is mandatory prior to neoadjuvant chemotherapy and would be necessary following cyst aspiration if the contents are bloody and being sent to cytology for analysis.
Anaesthesia
Local anaesthetic use with epinephrine was considered not necessary for routine CNB under US or mammography, however, using lidocaine with epinephrine was recommended for VAB. If epinephrine is contraindicated, local anaesthetic without epinephrine can be utilized, and VAB does not need to be avoided.
Pregnancy and Lactation
For pregnant/lactating patients, the panel unanimously advised against altering Breast Imaging-Reporting and Data System (BI-RADS) 3 categorization and agreed biopsies do not need to be avoided if otherwise indicated. CNB under US was identified as the preferred approach for pregnant/lactating patients.
Anticoagulation
For patients on blood thinners, panelists recommended against discontinuing blood thinners before CNB or VAB as per the Society of Interventional Radiology (SIR) guidelines, 14 and agreed VAB should not be avoided in these patients. In summary, anticoagulation should not be discontinued prior to image guided breast biopsy, for all modalities and lesion types. For all patients, every effort to decrease the risk of bleeding, including avoiding obvious vessels seen on tomography or utilizing Doppler prior to sampling under US guidance is recommended.
Specific Cases
The panelists also provided recommendations for specific cases, including MRI/CEM, inflammation/infection, calcifications, and retroareolar lesions (Supplemental Table 1). VAB was the most appropriate approach for lesions visible on MRI/CEM (with no mammographic or sonographic correlate). Moreover, the panelists advised retroareolar lesions visible on US should be biopsied with the same level of suspicion as parenchymal breast lesions, and should not be avoided or downgraded to BI-RADS 3 due to location.
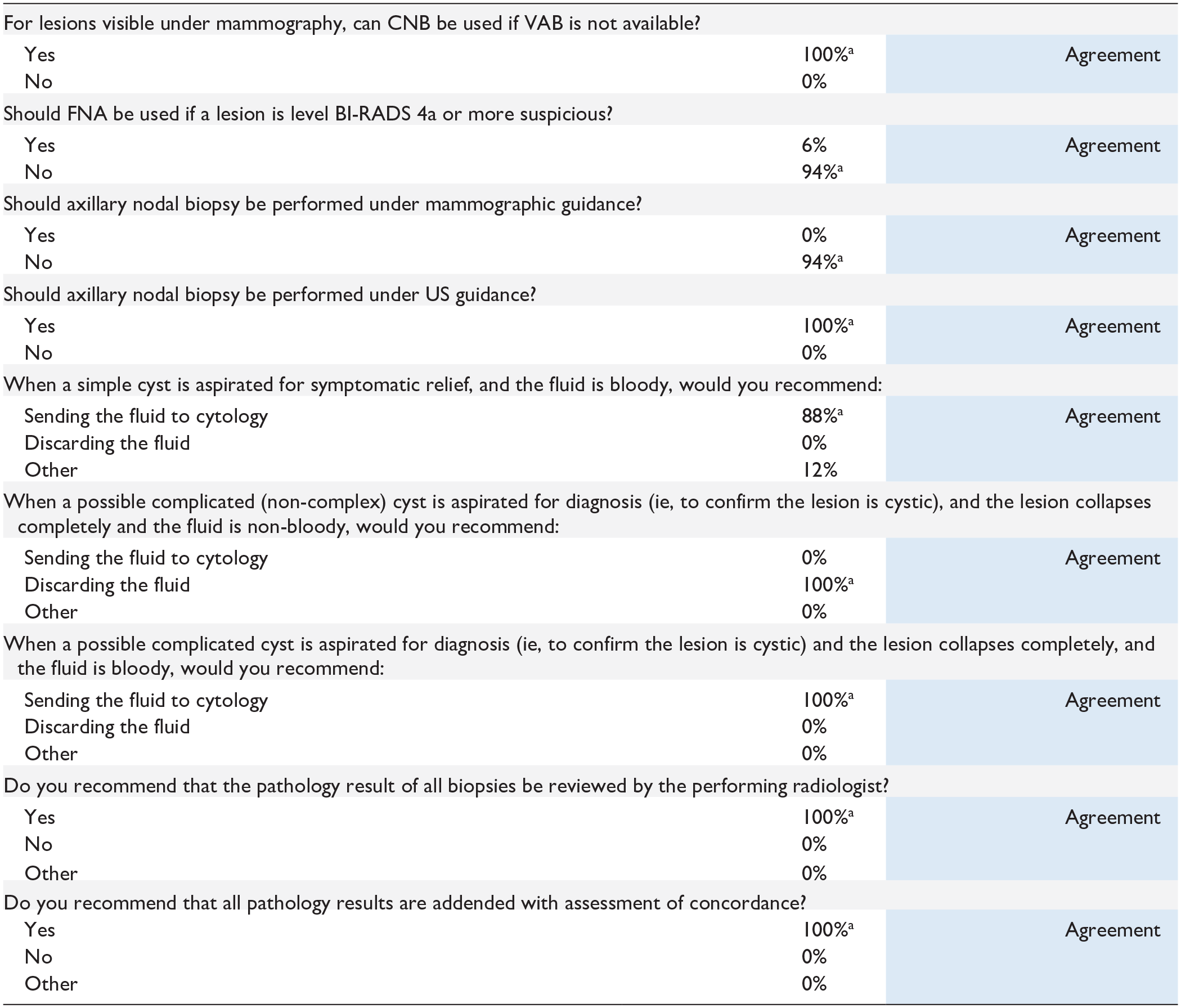
Recommendations were made to guide decision-making across a range of topics, including biopsy approach based on specific considerations (e.g., the availability of various modalities/techniques and the level of suspicion), utility of aspiration in the symptomatic relief and confirmatory diagnosis of cysts, and review and reporting considerations for the pathologists. Consensus was reached in all questions, and the findings are in Table 4.
Additional Recommendations Based on Multiple Choice Questions.
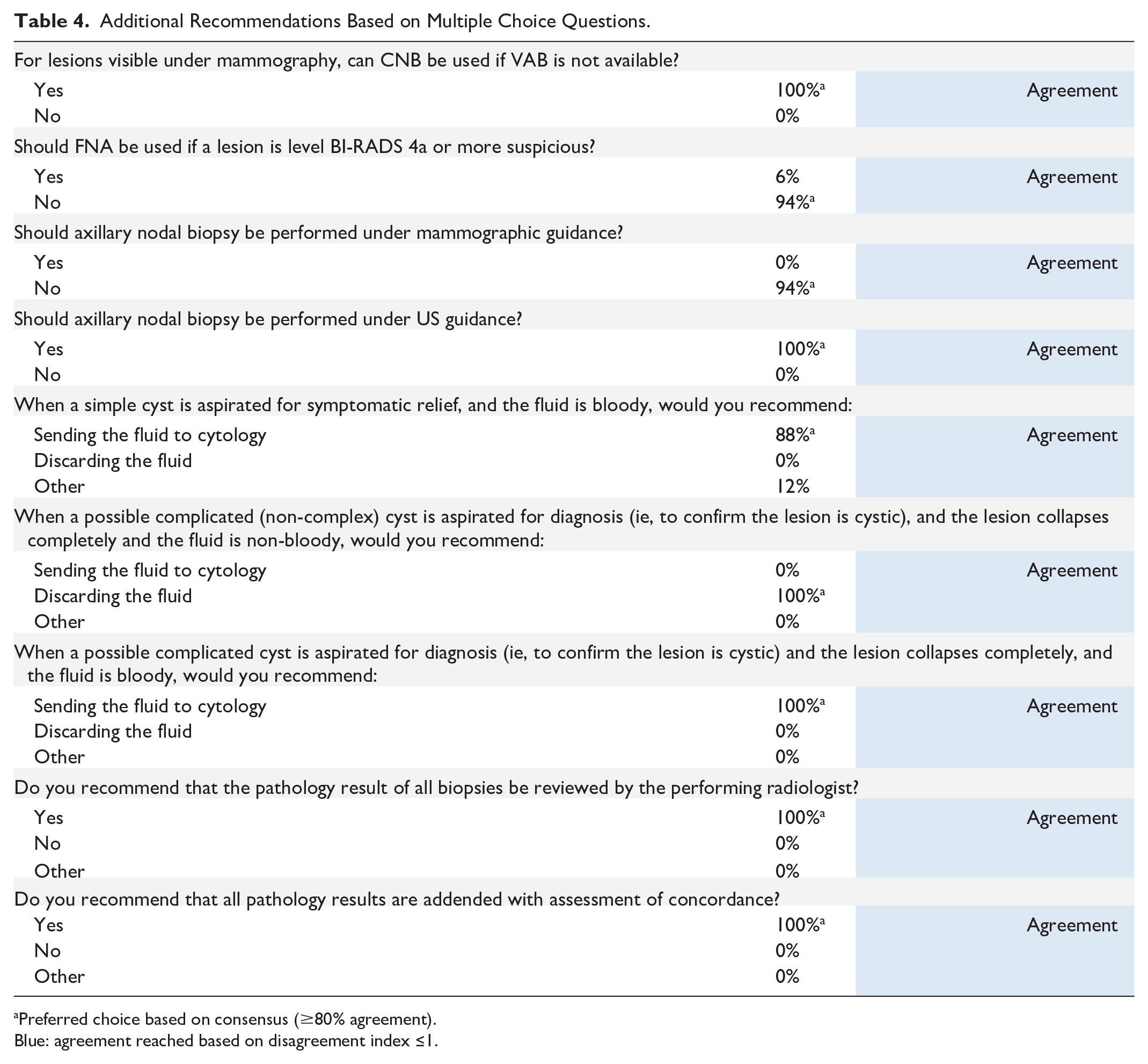
Preferred choice based on consensus (≥80% agreement).
Blue: agreement reached based on disagreement index ≤1.
Discussion
This manuscript provides consensus recommendations on breast biopsy practices for Canadian providers. Recent global and regional (i.e., Europe) efforts have been made to provide recommendations on best practices due to the variability and complexities of breast biopsy approaches. However, there are limited Canadian recommendations to assist in selecting the most appropriate breast biopsy techniques. Existing recommendations from other countries overlook the nuances of Canada’s publicly funded and provincially managed healthcare system. Furthermore, Canada’s geographic diversity leads to disparities in access to services and specialists, emphasizing the need for tailored guidance in clinical decision-making.15,16 Therefore, this work is timely and essential to address these gaps.
Overall, 17 specialists engaged in 2 voting rounds, adhering to best practices for the modified Delphi method.7,8 Aside from the agreements/disagreements detailed in results, the panel engaged in important discussions during the in-person meeting. Key points from these discussions are highlighted in this section and should be considered together with consensus findings.
General Approach to Diagnostic Biopsy
A key recommendation was that if a lesion can be detected on US, the initial biopsy should be conducted with US guidance, preferably using CNB. In contrast, VAB is recommended for lesions detected under mammography and MRI/CEM.
Vacuum-Assisted Excision
Panelists discussed VAE should not be used for initial diagnostic biopsy. This recommendation is consistent with European guidelines and other previous reports, 13 advising all high-risk lesions have an initial biopsy with CNB or VAB. Then, depending on lesion type, VAE or surgical excision may be considered after the initial biopsy.17,18 While VAE has gained popularity and acceptance in other regions (i.e., Europe), its adoption and availability in Canadian healthcare settings remain limited. 12 Panelists see an increasing role for VAE in Canada because of its advantages over surgical excision, including its minimally invasive nature, use of local anaesthesia, shorter recovery, and reduced costs. 19 Their recommendations on when VAE is appropriate largely align with relevant guidelines. 17 As demonstrated in panel discussions, effective integration of VAE into Canadian practice will require coordinated discussions between surgeons and radiologists to establish clinical pathways. Notably, panelists with a surgery background supported VAE use by radiologists as it would enable the use of surgical resources for more complex/urgent cases. Radiologists, however, preferred their surgical colleagues initiate requests for this newer technique, as this would make them more comfortable implementing a process change. These discussions demonstrate a clear need for pathways to be developed to facilitate VAE’s emergence and acceptance in the Canadian landscape.
Fine Needle Aspiration
Panelists agreed FNA should be avoided for sampling of primary lesions of the breast. FNA may be considered for lymph node biopsies visible on US in certain situations. The group highlighted that when FNA is used, it is usually in the setting of a prominent, easy-to-access lymph node. However, opinions among panelists were divided; while some avoid FNA due to lower accuracy, others see it as a quick and easy option when there is known cancer or a highly suspicious lesion. Panelists emphasized the decision to use FNA is site-specific, largely due to concerns about sample size and whether cytology services and interpretation expertise are available. When real-time interpretation is possible, FNA can be a suitable option, providing good sensitivity and specificity for cancer detection. However, several panelists cautioned against FNA as the primary method for biopsy due to challenges in achieving precise pathological diagnoses and receptor marker characterization. As such, when panelists voted on their preference between CNB and FNA for lymph node biopsies, CNB emerged as the favoured approach. Nevertheless, the panel highlighted the utility of FNA in the confirmatory diagnosis of possibly complicated cysts, as well as symptomatic relief for simple cysts. For possibly complicated cysts, distinct post-sampling approaches were recommended based on the presence of blood in the fluid following lesion collapse. Specifically, if no blood is present, the fluid can be discarded following aspiration. However, in the case of a bloody fluid following aspiration, referral to cytology was recommended for both apparently simple and possibly complicated cysts.
Core Needle Biopsy
While panelists considered CNB appropriate for almost all lesion types, VAB was the favoured choice for mammography-detected lesions due to the higher likelihood of an accurate diagnosis. During the discussion, some panelists reported VAB was not available at their facility. With this context, consensus was reached that CNB is acceptable as an alternative only when VAB is unavailable. Importantly, several panelists advocated for VAB as the standard of care for lesions biopsied under mammography, highlighting considerations such as speed, accuracy, patient comfort, and upgrade rates.20-23
The panel agreed CNB is an appropriate and recommended biopsy method for all lesion types visible on US. When a lesion is identified on MRI/CEM, and if there is no correlate on subsequent mammographic or sonographic diagnostic assessment, the panel recommended against CNB for sampling under MRI/CEM guidance. This is primarily due to the limited ability to confirm adequate targeting in real time. As a result, a larger sample size obtained through VAB is considered more likely to ensure adequate targeting and sampling, increasing the likelihood of accurate diagnosis.
Vacuum-Assisted Biopsy
VAB was considered appropriate for all lesion types under US, mammography, and MRI/CEM except for sampling of lymph nodes. The panelists discussed the benefits of VAB, including ease of use, sample size, patient comfort as well as a decreased likelihood of discordant results, and upgrade and rebiopsy rates—benefits which are widely reported in the existing literature.24-30 Several radiologists expressed a preference for VAB (when available) under mammography because it provides them their greatest chance of success at obtaining a good diagnostic sample. However, during discussion, panelists with a pathology background raised concerns regarding the performance of VAB on suspicious papillary lesions as in cases of malignancy, it can be challenging to evaluate for invasion on the biopsy sample if VAB is utilized.
Management Methods
Several panelists noted during discussion that 6-month imaging follow-up periods for benign-concordant lesions (e.g., fibroadenomas) may not be required. The panel agreed that if follow-up imaging was felt necessary, imaging in 12 months is generally more appropriate for most benign-concordant lesions, as supported by the literature. 31
Panelists agreed asymptomatic papillomas <1 cm without atypia can be managed conservatively through short-interval surveillance. However, all other papillomas (i.e., >1 cm or symptomatic) require surgical referral for possible surgical excision or VAE at the request of the surgeon. These recommendations are supported by the literature, which demonstrates upgrade rates for benign papillomas are highly influenced by lesion size or the presence of symptoms. 32 While the analysis considered surgical referral “not appropriate” for an asymptomatic papilloma <1 cm without atypia, panelists’ vote likely indicated that it would be unnecessary, taking into account the optimal utilization of resources. Worth noting, while the Delphi method can produce actionable recommendations, it has limitations in fully capturing nuanced considerations. 33 As such, the panel recommendations should be interpreted within the context and likely intent of the experts. While a consensus of “not appropriate” may reflect a lack of clinical justification, in scenarios such as the one mentioned above, it simply indicates the availability of a more efficient or appropriate option. While the underlying rationale for a “not appropriate” consensus may vary across different cases as discussed here, the real-world implications are similar: the panel advised providers against using management methods voted as “not appropriate.”
In LCIS discussions, panelists highlighted the management approach should vary based on type. While surveillance and tailored screening are acceptable management methods for classic LCIS, surgical referral was considered appropriate for the non-classic variants, including pleomorphic and florid types. The panelists referenced guidelines established by the Canadian Association of Radiologists during the discussion, emphasizing their importance in informing decision-making. 34 The management of flat epithelial atypia (FEA) sparked debate due to its diagnostic complexity, and the panelists expressed concerns about the concordance of an FEA diagnosis. Participants pointed to a wide range of upgrade rates (1%-8%) as well as variance across institutions for managing FEA (i.e., surgical excision and surveillance). As a result, achieving consensus on FEA management proved challenging, and varied approaches were appropriate (i.e., surgical referral) or may be appropriate (i.e., surveillance/screening). Ultimately, clinical considerations, including patient needs and local practice (e.g., upgrade rates), need to determine the choice of FEA management method. In fact, panelists considered multiple management strategies as appropriate (or may be appropriate) for several high-risk pathologies, highlighting the importance of specific pathology results and practical considerations for clinical decision-making. For example, surgical referral and VAE with surgical consult were both voted as appropriate for CSL/RS, while short-interval surveillance was considered “may be appropriate.” The ultimate management decision will depend on various factors, such as CSL/RS atypia status. The literature supports surgical intervention in cases of atypia, depending on risk factors. Conversely, in the absence of atypia and when histological findings are concordant, clinical and imaging follow-up (i.e., surveillance) can be a reasonable approach.13,35
Pregnancy/Lactating Population
Panelists agreed pregnancy/lactational status should not delay biopsy or affect BI-RADS categorization. During discussion, panelists addressed the difficulty of monitoring breast lesions once a woman undergoes lactational changes since the visibility of lesions can be difficult when breasts engorge. The panel believes additional data is necessary to develop more specific recommendations regarding biopsy during pregnancy, but emphasizes that clinicians should not delay biopsy until after childbirth. While the panel recommended both CNB and VAB as potential options, CNB was identified as the preferred approach to minimize complications such as milk fistula.
Clip Placement Considerations
Clip placement was considered appropriate for all lesions and should be standard practice in an ideal setting. However, there are constraints to practice, including budgetary and resource limitations. As a result, when resource management is a priority, clip placement was not considered necessary for lower suspicion lesions biopsied under US guidance, especially for larger lesions since these are likely easy to relocate.
Medication Considerations
Panelists advised against discontinuing blood thinners prior to VAB and CNB based on SIR guidance, 14 and agreed using local anaesthetic with epinephrine was not necessary for CNB but recommended for VAB. Some radiologists reported lidocaine with epinephrine is easily obtainable, leading them to utilize it for nearly every case. Others noted it was more difficult to obtain, resulting in less frequent use. There was a discussion regarding the potential risk for skin necrosis. Studies have debunked this once held belief; the panel’s surgeons referred to data from the surgical space, demonstrating its safe use in the skin. 36
Limitations
This work includes several limitations. First, while over 80% of items achieved consensus, the panel did not agree on certain topics. This is not unexpected given the complexity of the topic, and differences in access (i.e., VAB) and policies (i.e., screening) within and across provinces. Notably, other consensus efforts using a modified Delphi approach also did not achieve consensus on every topic.33,37,38 Second, while modified Delphi approaches are commonly used for clinical guideline development, there are intrinsic limitations, including a lack of ability to capture nuances and intent behind expert recommendations. We attempted to address this limitation by providing detailed insights regarding panel discussions. Third, access and availability limitations may limit the recommendation of certain devices and techniques. Despite capturing a diverse set of experts, it is possible that not all viewpoints across Canada were represented. Fourth, the recommended number of samples for CNB/VAB was determined independently of needle size. Clinicians should avoid assuming any needle size can be paired with any sample number from the recommended range, as these factors are interdependent in clinical practice. Finally, the recommendations from the panel cannot account for every clinical scenario and are limited to the currently available evidence. Alongside these consensus recommendations, physicians should carefully evaluate the existing literature and/or guidelines and the unique circumstances of each case when determining the most appropriate biopsy technique and management strategy. These recommendations are not intended to supplant professional judgment. Physicians may deviate from these recommendations as necessitated by the individual patient, practice setting or available resources. Breast radiologists are encouraged to engage with one another and their multidisciplinary colleagues (e.g., breast pathologists, breast surgeons, medical oncologists) to develop clinical management protocols suited to their institution. Furthermore, providers should integrate panel recommendations with emerging evidence, especially for high-risk lesions where upgrade rates vary across practices.13,39 As our knowledge of high-risk lesions advances, management recommendations may also evolve.40,41
Conclusion
To address the lack of recommendations for breast biopsy practices in Canada, a modified Delphi panel was conducted with 17 Canadian physicians. The panel provided recommendations, including guidance for initial biopsy technique based on lesion type and imaging modality, patient management or re-biopsy considerations after the initial biopsy, and procedural recommendations and patient considerations. These recommendations are designed to provide general guidance and standardize practices across Canada and were endorsed by the Canadian Society of Breast Imaging. They should be evaluated considering each individual patient’s unique circumstances and continually emerging evidence.
Supplemental Material
sj-docx-1-caj-10.1177_08465371251325500 – Supplemental material for Canadian Recommendations on Optimal Breast Biopsy Practices Developed Using a Modified Delphi Panel
Supplemental material, sj-docx-1-caj-10.1177_08465371251325500 for Canadian Recommendations on Optimal Breast Biopsy Practices Developed Using a Modified Delphi Panel by Zina S. Kellow, Afsaneh Alikhassi, Anita Bane, Mary Beth Bissell, Erin Cordeiro, Kavita Dhamanaskar, Jenny Jessup, Ryan Conor Kirwan, Gary Ko, Zuzana Kos, Ameya Kulkarni, Christophe Cloutier Lambert, Tetyana Martin, Elaine McKevitt, Silma Solorzano, Saly Zahra and Caitlin Ward in Canadian Association of Radiologists Journal
Footnotes
Acknowledgements
We acknowledge the Canadian Society of Breast Imaging (CSBI) who reviewed and endorse the recommendations. We also acknowledge Dr. Halit O. Yapici, Brendan Schuler, and Sarah Colton of Boston Strategic Partners, Inc. for their support with study design, data analysis, and manuscript development.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ZSK gave an educational talk for GE. CW has received gratuities from GE, BD, and Mammography Educators for speaking engagements and has served as the lead radiologist for a GE show site. GK has received speaker honorariums from Oncology Education and Merck. ZK has been compensated for participation in advisory committees by Eli Lilly and AstraZeneca, and has also received travel expense reimbursement from AstraZeneca and Daiichi Sankyo.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by BD (Becton, Dickinson and Company).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.