Abstract
Introduction
Mammographic density is one of the strongest risk factors for breast cancer: Women with extremely dense breasts have a 4 to 6× higher risk of developing breast cancer compared to those with fatty breasts.1,2 Women with heterogeneously dense breasts and no other risk factors are at least 1.5× higher risk for developing cancer than those with scattered density and women with extremely dense breasts are at twice the risk. 3
Risk for these women is even higher if they have other risk factors, such as family history. High breast density is one of the most prevalent risk factors for both premenopausal and postmenopausal women and has the largest effect on the Population-Attributable Risk Proportion. 4 It is estimated that density accounts for 39% of premenopausal and 26% of postmenopausal breast cancer. 4 Breast density is now incorporated in some risk models and should be used to optimize risk assessment. 5 Risk assessment should be performed to identify women who should be offered supplemental screening (especially MRI).
Cancers are masked in dense tissue on mammograms. Forty percent of cancers are missed in women with extremely dense breasts, and 30% are missed in heterogeneously dense breasts. 6 Those undetected cancers continue to grow and potentially spread, and are more likely to present as a palpable lump, referred to as an interval cancer, in women with dense breasts than in women with non-dense breasts.7,8 Interval cancers tend to be larger 9 and are more often metastatic to lymph nodes at the time of diagnosis than cancers found on screening mammograms, are more likely to require more intensive and harsh treatments, and have poorer outcomes with decreased survival.10-17 Women with the densest breasts are 13 to 18× more likely to have an interval cancer than women with fatty breasts, with worse outcomes.7,8
Randomized trials showed breast cancer mortality reduction of 20% overall.18-21 In observational studies, breast cancer mortality reduction is 25% to 31% for women invited for screening, versus 38% to 48% for women actually screened.22,23 Women do not all benefit equally from screening mammography. In the Dutch National screening program, a case-control study showed 41% mortality reduction in women with non-dense breasts, but only by 13% in women with dense breasts. 24
The goal of supplemental screening is to give women with dense breasts equity in the opportunity for early detection. By doing so, interval cancers are reduced, 25 deaths from breast cancer are reduced,26,27 and women can be successfully treated with less aggressive therapies: fewer mastectomies, fewer axillary dissections and lymphoedema, and less chemotherapy.28,29
Women in British Columbia (BC) have qualified for supplemental screening breast ultrasound (US) based on density classifications c or d since 2019. A study found an ICDR of 7 cancers per 1000 screening examinations in the first year in 695 patients all having a first/prevalence screen. 30 Given the high incremental cancer detection rate, we wondered whether it might have been a statical fluke given the relatively low number of screens performed, and whether it would decrease once women were attending for subsequent screens. The purpose of this study was to determine the cancer detection rate with inclusion of a larger number of examinations performed, and since some of the women were having incidence screens.
Methods
This study was approved by our institutional review board which granted a waiver of consent. A retrospective review was performed for all US-guided biopsies, prompted by screening US examinations performed in a community imaging clinic in Vancouver from August 1, 2021, to December 31, 2022. The biopsies were performed between August 25, 2021, and July 27, 2023. Our study population included women who were referred for US screening because of dense breasts (ACR BI-RADS categories c or d) classified based on previous negative screening or diagnostic mammograms, regardless of whether they had other risk factors such as family or previous personal history of breast cancer. All US examinations were performed by experienced technologists. Our screening breast US technologists are required to have completed the American Registry for Diagnostic Medical Sonography (ARDMS) Examination in Sonography Principles and Instrumentation and that for ARDMS Breast Sonography. Each breast sonography technologist performs 15 to 20 diagnostic breast US a day. There were 8 breast sonographers in 2022, with an average of 10 years of breast US experience. Four of them also conduct mammography, with an average mammography experience of 18 years. There were 5 breast imaging radiologists including one with recent fellowship training in breast imaging and others with 26, 40, 44, and 49 years of experience in breast imaging, respectively. Breast Imaging Radiologists determined, based upon their longitudinal experience with technologists, which women were selected to undertake screening breast sonography. High resolution linear array transducers were used: 5 to 15 MHz (GE Logic S8, Gyeonggi-do, Korea), and 5 to 14 MHz (Ultrasonix Touch, Richmond, BC, Canada).
Images were recorded by the sonographer for the interpreting radiologist to review only in the case of detection of an abnormality, including simple and complicated cysts, and non-cystic masses. All lesions were imaged in at least 2 orthogonal planes, and Doppler was used for masses that were not simple cysts. Cine loops were recorded at the discretion of the technologist, especially for subtle findings that were of low conspicuity on static images. The axilla was routinely scanned, and lymph nodes were documented. Our protocol did not require representative images of all quadrants and the subareolar region as described by Berg and Mendelson, 31 but technologists had to document all areas including the axilla included in the scan using a standard diagram.
Although inclusion of the axilla is not considered necessary when there is no suspicious finding on mammography or screening US,32,33 we routinely do scan the axillae to ensure any potential heterotopic parenchyma extending into the axillary tail has been scanned, so that parenchymal masses located there can be detected.
In BC, women may self-refer for screening mammograms starting at age 40. They must be asymptomatic, with no previous breast cancer, no breast implants, and not be pregnant. In 2022, 3 other jurisdictions in Canada did the same, 2 started at 45, but most started at 50. Women with a first-degree family history may attend annually in all jurisdictions. All others may attend only biennially. Although 7 Canadian provinces allow annual mammograms for women with Category D density, and this has been shown to decrease interval cancers, 34 that is not the case in BC.
In BC, women with a personal history of breast cancer or atypical ductal hyperplasia (ADH) may not attend the organized screening program, but rather must be referred for annual diagnostic mammography. There is no organized high-risk screening program in our province and supplemental MRI screening is not routinely offered to women with a personal history of breast cancer. All screening mammograms are done only with 2D mammography. In cases where a suspicious finding was seen at screening US, it was at the discretion of the interpreting radiologist to repeat the mammogram, depending on how much time had passed since her last mammogram. Because these are done as diagnostic studies, most were performed with digital breast tomosynthesis (DBT). Information was collected to determine whether the cancer was seen on the repeat mammogram, with DBT only, or with 2D + DBT. Follow-up data were collected for any woman whose biopsy showed atypia, papillary lesion ± atypia, pseudoangiomatous stromal hyperplasia (PASH), radial sclerosing lesion or malignancy.
All biopsies were performed by one of the authors (PBG). Most biopsies were performed with a 10 cm 14-gauge needle using a spring activated device (Bard Magnum, Becton, Dickinson and Company, Franklin Lakes, NJ) with 3 to 4 cores obtained (range 1-6), at the discretion of the radiologist, based on confidence of accurate targeting. All solid masses had at least 3 cores. Some masses disappeared after 1 or 2 cores, indicating that they were complicated cysts, subsequently confirmed on histology. Rarely, fine-needle aspiration biopsy was performed rather than core. All specimens were processed and interpreted by specialized breast pathologists at our provincial cancer agency. It is our practice to always place a clip after the biopsy, unless the lesion disappears during the procedure. When we anticipate that the lesion might be a complicated cyst (and disappear) we place a clip posterior to the lesion before any samples are taken. We use a variety of clips, including surgical vascular clips and commercially-available post-breast-biopsy clips. In all cases where a clip is deployed, post procedure mammograms are performed.
The histology/cytology reports for all biopsies were reviewed. The following information was collected from our clinic’s radiology information system: patient age, breast density, whether the screening US was her first (prevalent) or subsequent (incident) study, whether there was a personal or first-degree family history of breast cancer, and the time interval between the screening study and the biopsy. For cancers, histology included ER, PR and Her2 receptors, grade, and axillary node histology if core biopsy was performed. Ki67 was not reported. Surgical pathology reports were requested from the patient’s family doctor or surgeon, to determine node status on sentinel node biopsy or axillary dissection. Tumour size was noted and included the measurement from the screening US and from the pathology report for the cancers excised. AJCC 8th edition pathologic stage was recorded for each cancer.
Radiologic-pathologic review was performed for all biopsies. At the discretion of the radiologist, benign concordant cases could be returned to routine screening, with the recommendation that screening US continue to be performed halfway between planned mammograms. Alternatively, follow-up targeted US could be recommended. This was typically at 1 year, but in some cases could be at 6 and 12 months. None of the lesions followed this way showed any adverse change prompting repeat biopsy, and none were subsequently diagnosed as malignant.
For patients who returned to the same clinic for follow-up imaging, lesion size was assessed to determine any change. For cases where histology showed a papillary lesion, radial sclerosing lesion, PASH, or atypia, inquiries regarding recommended additional investigations done elsewhere were sent to the referring physicians for information about outcomes. There was no access to tumour registry for outcomes.
Results
From August 1, 2021, to December 31, 2022, screening US was performed for 5257 women. Core needle biopsy was performed on 247 women, with some of the biopsies performed in 2023.
Most had a single mass biopsied (Figure 1), but some had as many as 4 (2 suspicious masses in each breast). In the case of a suspicious breast mass and an abnormal lymph node, the node was biopsied during the same appointment. If there were multiple masses and only one was suspicious (BI-RADS 4 or 5), a second non-suspicious (BI-RADS 3) might also be biopsied at the discretion of the radiologist. In the 247 women, 279 breast masses were biopsied.
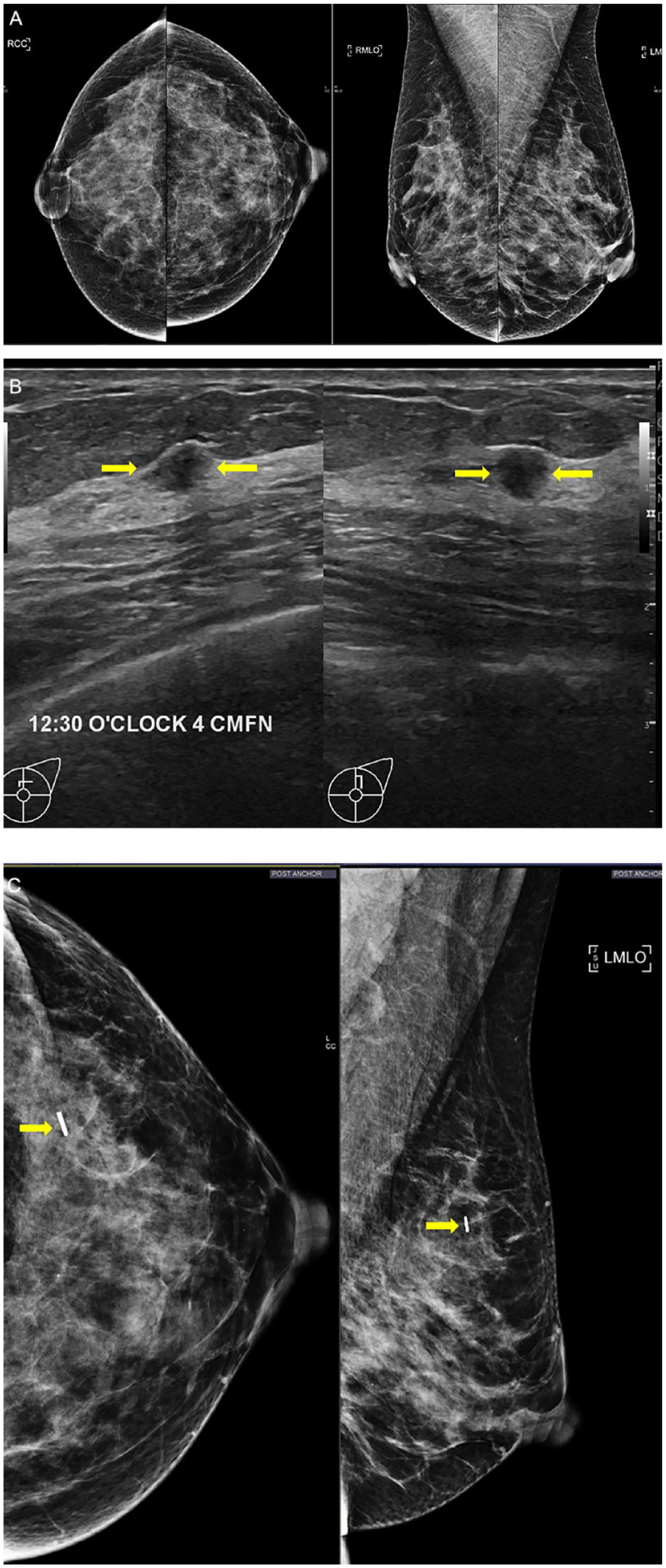
Age 51 category d, no personal or family history. (A) Screening mammogram. (B) Screening ultrasound, transverse (L) and sagittal (R) scans of the left breast, 13 months after screening mammogram. Suspicious mass at the 12:30 o’clock position, 4 cm from the nipple (arrows) measured 7 mm maximum dimension at US, and there were no suspicious axillary nodes. Assessed as BI-RADS 4A. No additional mammogram was performed before biopsy. Core biopsy showed IDC, ER+, PR+, Her2−, and Grade 3. At excision, size was 4.5 mm and Grade 1. Four sentinel nodes excised, all negative. Final stage 1A. (C) Post procedure mammogram shows no mass at the site of the vascular surgical clip (arrows).
Eighteen women had 2 sites biopsied. Of these, 14 had no cancer. Of the 4 that had cancer, one had cancer at both sites, and the others had cancer at one of the sites and the others were benign. Four women had 3 sites biopsied. In one, all 3 sites were benign. In another, 1 site was ILC, one was a papilloma with atypia, and one was nonspecific benign. In 2 others, one site was cancer and the other 2 were benign. Two women had 4 sites biopsied. Neither had cancer: in one of the women, all 4 were papillomas and in the other, all 4 were PASH.
BI-RADS final assessment categories were as follows for the 247 women: Cat 3—3, Cat 4 (otherwise not specified)—52, Cat 4A—114, Cat 4B—47, Cat 4C—21, Cat 5—10. For the women diagnosed with cancers, 6 were category 4 (otherwise not specified), 5 were category 4A, 3 were category 4B, 12 were category 4C, and 6 were category 5.
Of the 3 women who had biopsies for a BI-RADS 3 mass, one was age 69 and had 2 sisters with breast cancer, one was age 55 and her mother had breast cancer, and one was age 48 with no personal or family history of breast cancer. The number of BI-RADS 3 cases for the remaining screening US examinations was not recorded.
94/247 (38.1%) women had a diagnostic mammogram after the screening breast US detected an abnormality for which biopsy would be recommended. In 20/94 (20.6%) an abnormality was seen on the repeat mammogram, and that finding was visible only on DBT in 12 (Figure 2). The mean wait time between the screening US and biopsy was 11.95 weeks (range 1-52 weeks).
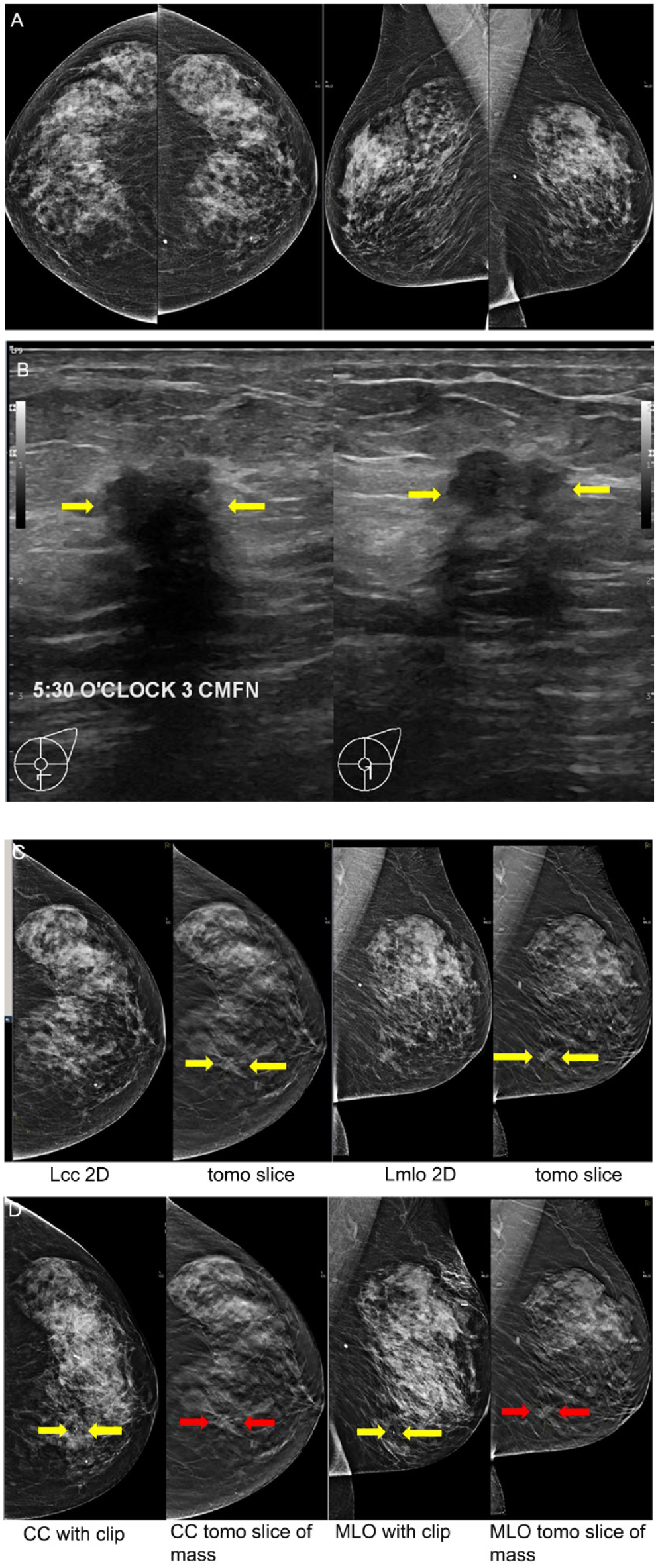
Age 61 Category c, no other risk factors. (A) Screening mammogram. (B) Screening ultrasound 1 year after the screening mammogram shows a suspicious 11 mm mass at the 5:30 o’clock position, 3 cm from the nipple, with no suspicious axillary nodes. Assessed as BI-RADS 4C. (C) The mammogram performed with tomosynthesis 6 weeks after the screening US, showed the cancer (arrows) on the cc tomosynthesis image only, less conspicuously in retrospect on the MLO 2D and tomosynthesis slice. Core biopsy was performed 16.5 weeks after the screening US study, and showed invasive apocrine adenocarcinoma, triple negative, and grade was not stated. (D) Post clip images confirm the coil shaped clip marker (yellow arrows) corresponding to the site of the suspicious mass seen on the tomosynthesis (red arrows).
Core histology showed cyst/fibrocystic change in 56, fibroadenoma/fibroadenomatous change in 86, malignancy in 31, radial sclerosing lesion in 5, PASH in 11, papilloma without atypia in 18, and papillomas with ADH in 3 (Table 1). Histology was benign not specified in 60. One papilloma with atypia was upgraded to DCIS at surgery. A total 32 cancers were found, for ICDR of 6.1/1000 screens and PPV3 (32/247) 13.0%.
Breast Cancers Diagnosed With Supplemental Screening Ultrasound, Tumour Histology, Grade, and AJCC 8th ed Stage, and Baseline Characteristics.
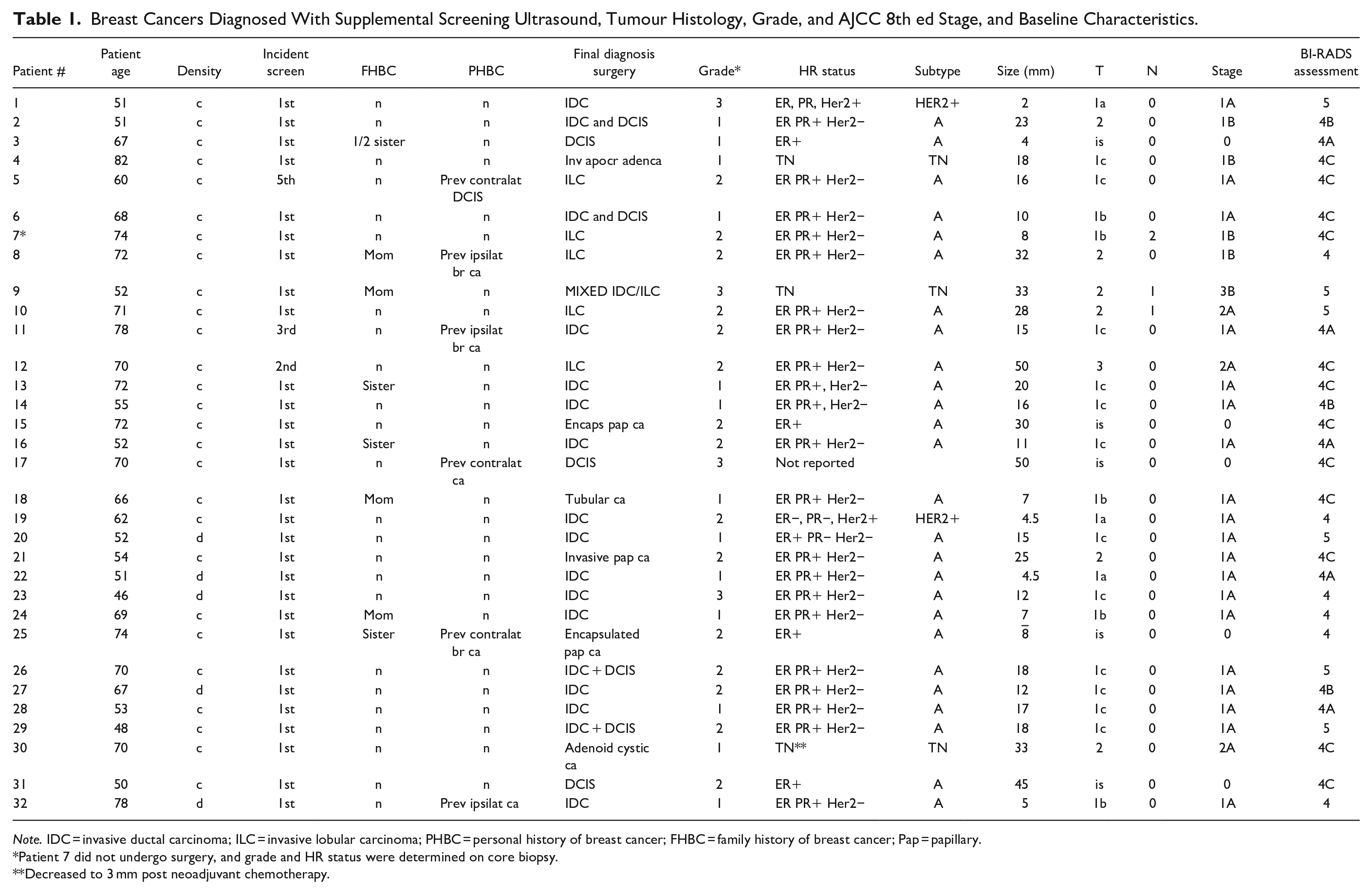
Note. IDC = invasive ductal carcinoma; ILC = invasive lobular carcinoma; PHBC = personal history of breast cancer; FHBC = family history of breast cancer; Pap = papillary.
Patient 7 did not undergo surgery, and grade and HR status were determined on core biopsy.
Decreased to 3 mm post neoadjuvant chemotherapy.
The average age of women diagnosed with cancer was 63.3 (range 46-82 years). Of 32 cancers, 26 (81.3%) were invasive including 5 (15.6%) invasive lobular carcinomas, and 6 (18.8%) were non-invasive. Average size was 13.4 mm (range 5->40 mm) on US and final pathologic stage for ductal cancers was stage 0 in 5/32 (15.6%) and 1A in 18/32 (56.3%), only 2 were HER2+, and 1 TN, 1 mixed ductal/lobular carcinoma (TN) was 3B (3.1%), while the 5 lobular cancers were stage 1A (1), 1B (2), and 2A (2), and 1 adenoid cystic carcinoma (TN) was stage 2A. One patient declined surgery and received endocrine therapy, with no change in size at 20 months. One patient with triple negative cancer had neoadjuvant chemotherapy, and her cancer decreased from 33 mm on US to 3 mm at surgery. Twelve cancers were unchanged or smaller on the surgical report, and 18 measured larger on surgical excision.
Twenty-nine cancers were found on prevalent screens and 3 on incident screens. Of the 3 cancers seen on a subsequent scan, 2 were ILC, and one was IDC. 27/32 cancers (84.4%) were in Category c density, and 5 (15.6%) were category d, 20 (62.5%) occurring in women with no personal and/or first-degree family history of breast cancer.
Of the entire cohort of 247 patients who had biopsies, 91 had a mother or sister (or both) with breast cancer. Twenty-four had a previous personal history, 8 of whom also had a first-degree family history. Two had ADH. Of the cancers: 4 were in women with a previous cancer, 4 were in women with a first-degree family history, and 2 were in women with both a personal and family history.
Eight of the 32 cancers (25%) were in women with a mother or sister with breast cancer and 24 (75%) were in women with no family history. Six of the cancers were in women with a previous personal history, 3 of unknown pathology, diagnosed 10 to 24 years ago and 3 had different pathologies from the new cancer. If we deduct them from the total, then we found 26 cancers in 5250 screens, or 4.95 cancers per thousand screens.
Of the 32 cancers, 27 were in women in Category c, 16 of them had no other risk factors: 6 had a first-degree family history; 4 had a personal history and one had both a first degree and personal history of breast cancer. Five of the cancers were in women with Category d density; 4 of them had no other risk factors; one had a personal history of breast cancer and none had a family history.
One woman whose cancer was triple negative had a suspicious node on the screening study, that showed metastasis on core biopsy. She received neoadjuvant chemotherapy and had no positive sentinel nodes on her subsequent surgery, which included preoperative localization of the node that had been biopsied. One woman had ILC, and no abnormal nodes were seen at the time of her screening breast US, but 2 of 4 sentinel nodes were positive for metastases. All the other women who had no abnormal axillary nodes at the time of the US examination had no positive sentinel node at surgery. One woman had no suspicious breast mass but had abnormal axillary nodes. Core biopsy led to a diagnosis of B-cell lymphoma. She was not included in the analysis.
No diagnostic imaging follow-up was recommended when histology was definitive for cyst, fibroadenoma or fat necrosis, and considered to be concordant with the imaging appearance of the lesion. These women returned to routine screening or routine annual surveillance mammography in women with previous cancer.
For the 60 masses where the histology was benign, but non-specific, it was at the discretion of the radiologist who did the biopsy and performed radiologic/pathologic review, to recommend imaging surveillance (or not). Most were recommended to resume routine screening. If surveillance was recommended, it was for 6 or 12 months.
Discussion
This is a large single centre study of supplemental screening US performed for women with dense breasts (categories c and d) after a normal screening or asymptomatic diagnostic mammogram, and found a high ICDR of 6.1/1000, 84% invasive cancers and 87.5% at early stage (0 or 1) with a mean imaging size of cancers of 13.6 mm. To our knowledge, this is the largest report on supplemental US screening in Canada.
US is the most economical and widely available supplemental screening modality. But it is less sensitive than the functional modalities and has lower specificity. It has been proven to increase detection of small invasive, node-negative cancers, and reduce interval cancers. 25 Breast US is widely available as a targeted examination for diagnosis but has not been widely available for screening in Canada. Our study found a slightly lower ICDR for supplemental screening US (6.1/1000) to that previously reported (7/1000), but higher than reported elsewhere. This is due, in part, to the inclusion of women in Categories c and d with previous cancer. The ICDR for screening US performed either automated (ABUS) or hand-held (HHUS) in women with dense breasts, but without previous cancer is 2.0 to 2.7 cancers per 1000 screening examinations, with a mean size range of invasive cancers of 7 to 14 mm, and 88% of cancers found to be invasive. 35 Weigert studied ICDR over 4 years of screening US, finding 4 cancers/1000 in the first year, 3.3 the second, 3.1 the third, and 3.3 the fourth. 36 Hooley reported 3.2 cancers/1000 screens in their first year. 37 These 2 studies were both after annual 2D mammography. Our study used HHUS performed by highly trained technologists and should be comparable to ABUS. The higher ICDR in our study is likely explained by the fact that in our province, mammography screening in most women without previous cancer and without a first-degree family history is offered only biennially instead of annually and with 2D mammography instead of DBT.
In the U.S.A, screening mammography is performed annually, and increasingly performed with DBT. DBT increases cancer detection compared to 2D mammography, while also reducing recalls. But it is not as effective as other modalities in detecting cancers in dense breasts.38,39 As of 1 May 2024, 90% of facilities had DBT units and 48% of all accredited units were DBT in the United States. 40 This may explain the relatively low ICDRs seen in the DBTUST trial: 1.3/1000 in the first year and 1.0/1000 in years 2 and 3. 41 In most of the world, screening mammography is performed biennially with only 2D mammography. DBT is not used currently in any organized screening program in Canada. It is not surprising that more cancers were found with supplemental US in an environment with less frequent mammography screening with 2D. Another reason for the higher ICDR is that we included patients who had previous breast cancer, who are known to be at least intermediate risk. If these cases are eliminated, the incremental cancer detection rate is 26 cancers in 5250 screens, or 4.95 cancers per thousand.
To understand the context of our high cancer detection rate, there are other considerations. The first is the screening mammography eligibility in our province, and the rest of Canada. In our province, women may self-refer for screening mammograms starting at age 40. In 2022, 3 other jurisdictions did the same, 2 started at 45, but most started at 50. Women with a first-degree family history could attend annually in all jurisdictions, but even then, could not begin screening until age 50. All others may attend only biennially, and all screening programs in Canada but one, use only full-field digital mammography. In only one province is digital breast tomosynthesis now used for screening. Although 7 Canadian provinces allow annual mammograms for women with Category D density, and this has been shown to decrease interval cancers, 42 that is not the case in our province.
Another consideration is that the ACR guidelines for breast MRI after a breast cancer diagnosis are not followed in Canada. The ACR recommends annual MRI for women with previous cancer who have dense breasts, and for women of any breast density who were diagnosed younger than age 50. 43 The supplemental screening modalities for these women vary by province, and sometimes by hospital, but in our province, only breast US is available, and even then is not offered in most locations.
Contrast-enhanced MRI using intravenous gadolinium is the most sensitive modality for detection of cancer, but also the most expensive. It has been shown to detect primarily invasive cancers and reduce interval cancers and late-stage disease.44,45 Screening breast MRI has also been shown to increase detection of biologically significant cancers in women at average-risk including those with non-dense breasts. 46
In Canada, MRI for screening is most often limited to women with genetic mutations, those who had chest radiation younger than age 30, or those at >25% lifetime risk by risk models.47,48 In the USA, it is recommended for women at greater than 20% lifetime risk, including women who’ve had cancer who have dense breasts, and women of any breast density who were diagnosed with breast cancer younger than age 50. 43 In Europe, MRI is recommended for women aged 50 to 69 with breast density category d, ideally every 2 to 3 years but no less often than every 4 years. 49 MRI is less tolerable than other modalities because of claustrophobia, and in 2 large studies, over 40% of women declined MRI even though it was free.50,51 Use of abbreviated MRI will decrease cost, increase access, and increase tolerance for some women with claustrophobia. It has not been widely implemented yet.
Contrast-enhanced mammography has similar sensitivity to MRI, but with higher specificity. 52 It is not yet widely available in Canada but has been purchased by many institutions. Molecular breast imaging detects cancer because of the preferential uptake of intravenously injected 99mTc-sestamibi in mitotically active breast tissue. 53 Molecular breast imaging is very poorly available in Canada.
To maximize detection of as many low-stage cancers as possible, we recommend that women have their screening US examination halfway between planned mammograms. For women attending biennially, we see it as an advantage to alternate the examinations. Rather than have a screening US at the same time as the mammogram only every 2 years, they are being screened annually, albeit with either modality. We also recommend alternating modalities for women attending for annual mammograms, to potentially detect rapidly growing cancers.
Of the 3 cancers found on incident screens, 2 were in women with previous ipsilateral breast cancer and were not seen on their most recent prior mammograms (less than 1 year before the US), and no repeat mammogram was done after the finding on US. The third patient had no personal or family history of breast cancer. Her most recent prior mammogram was 1 year before the US and was repeated. The cancer was visible on both 2D and DBT.
Seven jurisdictions in Canada currently offer annual screening mammograms to women in category d, and some plan to offer supplemental screening only to women in density Category d, ± women with a first-degree family history. Although those women are at highest risk, the majority of cancers (84%) in this study were in women in Category c and 62% of the cancers occurred in women with no personal or family history of breast cancer. This is understandable, given that there are 4 to 5 times more women in category c as in category d,54,55 and 85% of breast cancers occur in women with no family history. 56 This underscores that supplemental screening should be offered to all women in both density categories c and d to bring equity to women with dense breasts in the detection of early cancers to both reduce mortality and provide better quality-of-life for women with cancer.
Cost effectiveness is a consideration in our publicly funded health care system. But although not paid for by the patients personally, the reimbursement from the Ministry of Health in our province is relatively low: a core biopsy of 1 to 5 samples is reimbursed $94.43, and a post-biopsy marker is reimbursed $155.85. 57 Moreover, it has now been shown that because of the high cost of novel therapies used to treat advanced cancers, detecting cancers at earlier stages and reducing advanced cancers is cost-saving. 58
This study has several limitations. The screening US examinations were performed in a single centre, where both the radiologists and technologists have extensive experience in diagnostic and screening US. Because a referral was required for patients to attend a community imaging clinic, it is possible that some of the recommended follow up examinations were not performed. This could theoretically miss a false negative biopsy and underestimate the sensitivity of screening US. This is very unlikely in this clinic, given that it has been shown that when radiologic-pathologic review is performed, even false negative histology does not result in delay of diagnosis. 59
Our PPV3 was relatively low at 13.0%. Most studies were prevalence screens, and this should improve as more women attend for subsequent screening examinations. Weigert showed that PPV3 increased as radiologists gained experience, follow-up their biopsy recommendations, and when prior studies are available. 36 PPV might also be improved by using elastography, opto-acoustic or other non-invasive vascular imaging, or artificial Intelligence (DL/radiomics), none of which are available at our clinic.
In conclusion, these data illustrate the important role of screening US in the Canadian context, and in many other jurisdictions where screening mammography, is performed most often with FFDM, and biennially. This essential life-saving examination should be available for women with dense breasts to give them equity in the early detection of breast cancer, and fee codes should be created to incentivize radiologists to offer the service.
Footnotes
Acknowledgements
The authors would like to thank Abigail Doherty for her help managing the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
