Abstract
Screening mammography has been shown to reduce breast cancer mortality by 41% in screened women ages 40–69 years. There is misinformation about breast screening and the Canadian breast screening guidelines. This can decrease confidence in screening mammography and can lead to suboptimal recommendations. We review some of this misinformation to help radiologists and referring physicians navigate the varied international and provincial guidelines. We address the ages to start and stop breast screening. We explore how these recommendations may vary for specific populations such as patients who are at increased risk, transgender patients and minorities. We identify who would benefit from supplemental screening and review the available supplemental screening modalities including ultrasound, MRI, contrast-enhanced mammography and others. We describe emerging technologies including the potential use of artificial intelligence for breast screening. We provide background on why screening policies vary across the country between provinces and territories. This review is intended to help radiologists and referring physicians understand and navigate the varied international and provincial recommendations and guidelines and make the best recommendations for their patients.
Introduction
The benefits of screening for breast cancer are seen when it detects breast cancer at an early stage, before it spreads to lymph nodes and elsewhere in the body. Screening mammography has been shown to reduce the rate of advanced stage breast cancers by 25% and therefore reduce breast cancer mortality by 41% in screened women ages 40–69 years. 1 Randomized controlled trials have shown an average of 25% reduction in breast cancer mortality after 10–20 years of screening in women who have been invited to participate.2-5 Women who participate in screening mammography have a larger benefit, in the order of 40% reduction.
The Misinformation
Canadian family physicians and nurse practitioners receive screening information from the Canadian Task Force on Preventive Health Care (CTFPHC), a government-funded panel that contains no breast cancer expert input or oversight.
6
Their most recent breast cancer screening guidelines in 2018 were unchanged from the 2011 guidelines, with the exception that women should participate in shared decision-making about whether they choose to participate.
6
The 2018 guidelines did not consider any newer research regarding screening and benefits of early detection. They relied only on randomized trials done from the 1960s to the 1980s, one of which has now been discredited.
7
As a result, the CTFPHC stated that ‘harms’ of screening outweigh benefits for women 40–49, and that women of average risk in this age group should not be routinely screened. To aid health practitioners in the shared decision-making process, CTFPHC provided a ‘tool,’ in which the data was based on the now-discredited Canadian study that exaggerated the harms and understated the benefits
6
. The CSBI has re-created the 1000 women decision tool with accurate data, shown in Figure 1.The CTFPHC has not revised the guideline or the decision tool despite this new evidence. Canadian society of breast imaging figure demonstrating the Canadian task force on preventive health care decision tool compared to accurate data.
In addition the CTFPHC recommends that women aged 50–74 be screened only every 2–3 years and states that women with dense breasts are average risk and do not need supplemental screening.
Age and Interval for Breast Screening
All women, especially Asian, Hispanic and Black women and those of Ashkenazi Jewish descent, should be evaluated for breast cancer risk no later than age 30.
8
For average risk women no screening is recommended until age 40. Figure 2 illustrates the breast screening referral pathway. Breast screening referral pathway. All eligible patients should receive mammography. The decision whether to add supplemental screening ultrasound, contrast enhanced mammography, or MRI is based on lifetime risk and breast density. There is variable access to MRI across Canada and not all patients may be MRI candidates.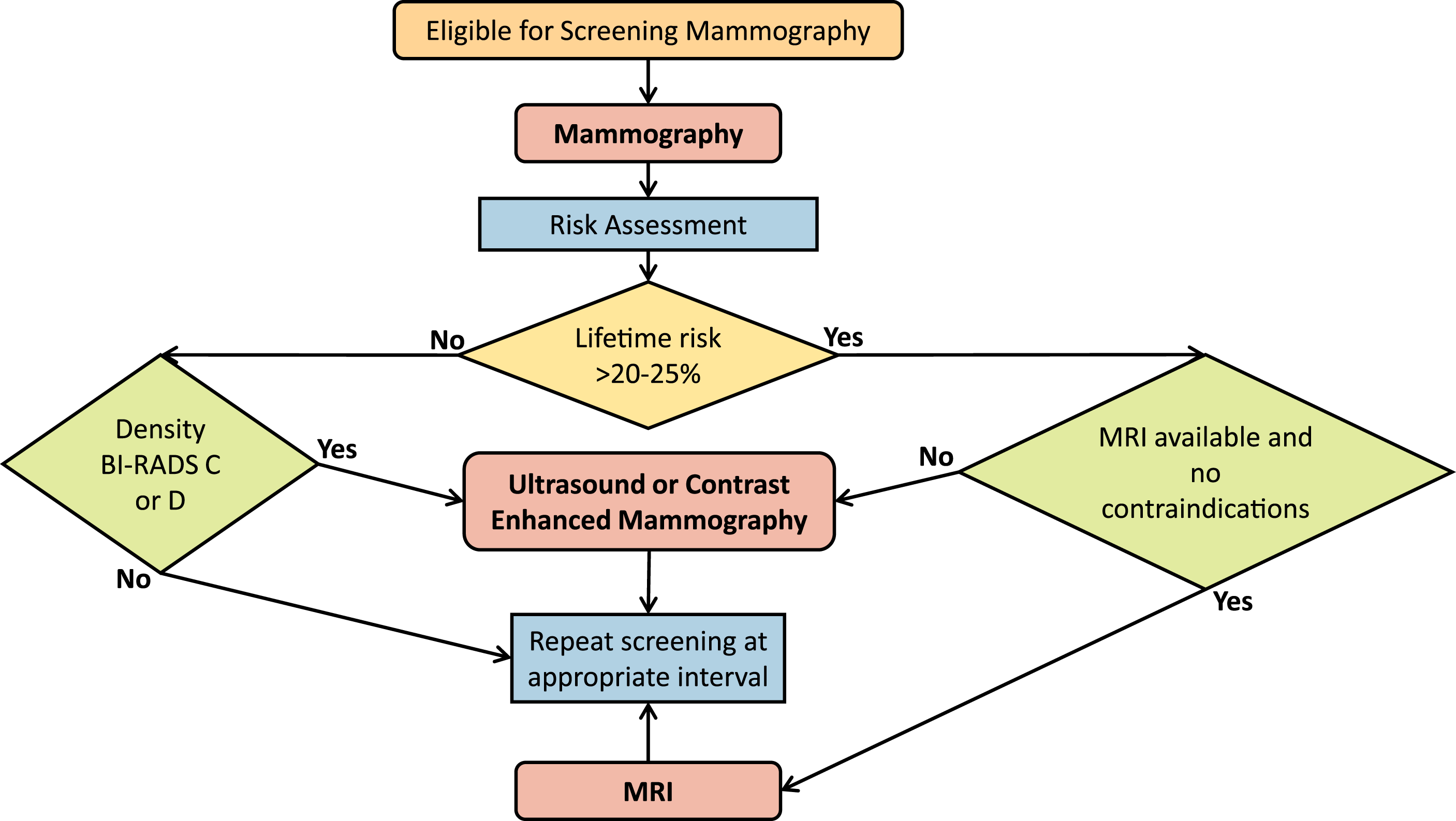
The ACR Commission on Breast Imaging recommendation for women of average risk 40–49 years of age is for screening mammography every 12 months.9,10 Because breast cancer in premenopausal patients grows at a more rapid rate than in postmenopausal women, and because most women under 50 are premenopausal, a shorter screening interval is strongly recommended. Annual screening mammography for all women saves the most lives. 10 The benefit of annual screening is more significant in premenopausal women, although still important in postmenopausal women, especially given that many women older than 50 are premenopausal, and many are on menopausal hormone therapy. For women 40 years and older with the densest breasts, Seely et al. 12 showed a benefit in reducing interval cancers (cancers detected in between regular screening) using annual screening mammography in Canada.
The risk of breast cancer continues to increase with age, so it is very important to continue screening older women if they are in good health, have a life expectancy of at least 10 years, and are able to have treatment (even if only minimal) if cancer is found.13,14 The risk of breast cancer for a woman in her 60s is 1 in 28 (3.5%) and the risk of dying from breast cancer is 0.5%, while for a woman in her 70s it is 1 in 25 (4.1%) and of dying of it is 0.8%. 15 Incidence of cancer increases with age and early detection allows less aggressive medical and surgical treatments. This is particularly important in the older, frail population. Risks of screening include ‘false positives’ or recalls, which may be a more significant consideration for women who need a driver or caregiver to attend. Additionally, overdiagnosis is a consideration, particularly in patients with significant comorbidities. In general, overdiagnosis estimates range from 1% to 10%. 16 A study of benefits and risks of screening mammography in older women with a focus on overdiagnosis found benefits outweigh risks until age 90. 17
The Canadian Association of Radiologists (CAR) recommends the following for average risk women, endorsed by the Canadian Society of Breast Imaging (CSBI): 18
Asymptomatic women 40–49 years should undergo screening mammography every year. Asymptomatic women aged 50–74 should undergo screening mammography every 1–2 years. Women over the age of 74 should have screening mammography at 1 to 2-year intervals if they are in good general health.
First Line Breast Cancer Screening
Some patients may request ultrasound instead of mammography due to discomfort or concerns about low-dose radiation associated with mammography. It is important to emphasize the reasons that screening mammography is recommended. Mammography has been shown in 6 randomized controlled trials and numerous meta-analyses and observational studies to reduce mortality from breast cancer by 25%–40%.3,5,11 Mammographic sensitivity is limited by dense breast tissue, and supplemental screening with breast ultrasound detects additional small cancers missed on mammograms. Breast ultrasound on its own, however, misses many cancers that are diagnosed on mammography: Ultrasound is not reliable at detecting breast calcifications that are found in 45% of breast cancers.19,20 For this reason, it is strongly recommended that patients start with a mammogram.
In many practices the patient’s wishes may be respected and they are provided ultrasound screening. At these visits, the patient should be advised that screening without mammography is against current medical advice. They should be reassured that the risk of radiation used in modern mammography is negligible after age 40. 21 The patient should be encouraged to express concerns and ask questions. Many patients refuse mammography due to a lack of information about risks and benefits and these can be addressed at the time of their exam. Some patients will accept mammography after their concerns are addressed, while others do not. Careful documentation that the patient declined mammography against medical advice is recommended. 22
Supplemental Breast Screening
Breast tissue density refers to the proportion of fibroglandular tissue relative to fat in the breast and is divided from least to most dense using the ACR BI-RADS categories A, B, C, and D. 23 Categories C and D are regarded as dense. Breast density assessment is subjective. Some provincial screening programmes do not use the ACR BI-RADS categories. There is great potential for improved breast density standardization with artificial intelligence (AI).
Ideally, women with dense breasts should be offered supplemental screening, even in the absence of additional risk factors such as a family history. Supplemental screening with ultrasound at one Canadian site found 7 cancers per 1000 screens. Of those, 40% had no family history and 60% were in BI-RADS category C. 24
Adding breast ultrasound to mammography routinely is not required for all women. About 43% of women aged 40 and older have dense breasts. 25 Mammography is highly sensitive for patients with non-dense breast tissue. Mammography sensitivity is 81%–93% for Category A, 84%–90% for Category B, 69%–81% for Category C, and 57%–71% for Category D. 25 Patients with dense breasts have high rates of interval cancers. 26 In high-risk patients, supplemental screening with MRI is recommended.
Ultrasound for supplemental breast screening. Ultrasound detection of cancers missed on mammograms was first reported in 1995 27 and many single centre studies have since confirmed the potential of ultrasound as a supplemental screen for women with dense breasts.27-32 The Avon-ACRIN (American College of Radiology) 6666 multicentre study demonstrated ultrasound and MRI detected cancers missed on mammography in 2809 women at increased risk with dense breast tissue in at least one quadrant. 28 The incremental cancer detection rate (ICDR) and positive predictive value for biopsy (PPV3) for ultrasound were 5.3/1000 and 11% in the first year, and 3.7/1000 and 16% in the second and third years, averaging 4.3/1000 and 14% overall. Of the cancers seen only on US, 94% were small (median size 10 mm), invasive and 96% of those staged were node negative. MRI detected an additional 14.7/1000 after negative ultrasound and mammography.
Researchers in Conneticut, the first state in the USA to enact breast density notification legislation and to perform high volume supplemental US screening, reported ICDR and PPV1 of 3.2–3.25 cancers/1000 and 6.5%–6.7%.30,31 In one centre, PPV3 increased to 20.1% by the fourth year of screening. 32
The cancer incidence in probably benign (BI-RADS 3) findings at screening ultrasound was .8% in the Avon ACRIN 6666 trial and only one of 745 BI-RADS 3 masses demonstrated suspicious changes at the 6 months follow-up. 29 The initial 6 months follow-up ultrasound is therefore no longer required, and a 1-year diagnostic follow-up is now recommended for BI-RADS 3 lesions detected at screening ultrasound.
Jurisdictional Screening Access. Alberta (AB), British Columbia (BC), Manitoba (MB), New Brunswick (NB), Newfoundland and Labrador (NL), Northwest Territories (NT), Nova Scotia (NS), Nunavut (NU), Ontario (ON), Prince Edward Island (PE), Quebec (QC), Saskatchewan (SK), Yukon (YT). *Women can self refer after the first referral is made by the family physician.

The CSBI states that supplemental screening breast ultrasound may be considered for patients with BI-RADS density categories C and D but acknowledges that there are challenges with providing supplemental screening for this subpopulation. 35
The optimal timing of supplemental ultrasound has not been established. If performed at the same time as screening mammography, only one clinic visit is required. A mass seen on screening mammography may be diagnosed as a cyst on a concurrent ultrasound, precluding recall, thereby improving specificity. When screening mammography is performed biennially, alternating with ultrasound allows annual screening, and opportunity for earlier detection and decreased interval cancers.
Preliminary publication of the ongoing Japan Strategic Anti-cancer Randomized Trial (J-START) demonstrated significantly higher sensitivity in the group having mammography plus ultrasound than in the group having mammography alone (91.1% vs 77.0%; P = 0.0004). 36 Specificity was significantly lower (87.7% vs 91.4%; P < 0.0001). More cancers were detected in the intervention group (184 vs 117; P = 0.0003) and were more frequently stage 0 and 1 (144 vs 79; P = 0.0194). Fewer interval cancers were detected in the intervention group 18 vs 35; (P = 0.034). This is important because reduction of interval cancers is a surrogate for mortality reduction.
Hand held or automated breast screening ultrasound (ABUS) can be performed. ICDR in hand held and ABUS is similar, averaging 2.1-2.7/1000 for physician- and technologist-performed examinations, but the ranges are wide. 37 A recent audit of hand held screening ultrasound from Canada showed an ICDR of 7/1000 on the prevalence scan. 24
Technologists who perform diagnostic breast ultrasound can perform supplemental screening ultrasound. The entire breast should be scanned, from clavicle to inframammary crease and from sternum to anterior axillary line. The axilla does not need to be included. Routine axillary scanning during screening breast US does not improve cancer detection, but increases false-positives. However, it is important to scan the axillary tail because many women have axillary tail parenchyma. 38
MRI for supplemental breast screening. Breast MRI finds 16 cancers per thousand in women in Category D and reduces interval cancers. 39 The American College of Radiology and the CSBI recommend MRI screening for women with ≥20%–25% lifetime risk, including women with genetics-based increased risk (and their untested first-degree relatives), a history of chest or mantle radiation therapy at a young age, women with personal histories of breast cancer and dense tissue or those of any diagnosed by age 50. Others with histories of breast cancer and those with atypia at biopsy should consider additional surveillance with MRI, especially if other risk factors are present. 40 The DENSE trial from the Netherlands showed an incremental cancer detection rate of 16.5 per thousand, and a 6-fold reduction in the interval cancer rate in average risk women with dense breasts who underwent MRI. 39 The second round of screening showed a sustained CDR of 6/1000 and all were early stage 0,1 breast cancers, with fewer false positives (2.7% vs 8% in the first round). 41
The European Society of Breast Imaging recommends screening breast MRI be offered every 2–4 years for women aged 50–70 years with extremely dense breasts. 42 They state that there is currently insufficient evidence to recommend MRI for women with heterogeneously dense breasts but urge the medical community to investigate the value of MRI screening for women with less dense breast tissue in high-quality trials. Where MRI is unavailable ultrasound in combination with mammography may be used as an alternative. They added the proviso that women should be adequately informed ‘about the different performance levels of different non-mammographic screening methods’.
Breast MRI requires intravenous gadolinium. It is associated with low rates of adverse events including nephrogenic systemic fibrosis. There can be gadolinium deposition in various organs including bone, skin, solid organs and brain but with no known long-term effects 43 unless in very high doses, as seen rarely in people with poor renal function, and less with macrocyclic agents. Information provided by radiologists in shared decision-making with the patient should include having an IV placed, the side effects of contrast, false alarms, the need for biopsy to resolve findings, and overdiagnosis, which they estimate could be as high as 22%, related to in situ and indolent invasive cancers. 42
Contrast-enhanced mammography. Contrast-enhanced mammography (CEM) is almost as sensitive as MRI and is much less expensive but has been far less evaluated for screening and is not yet Health Canada approved for screening.44-46 CEM in addition to mammography can increase sensitivity from 71.5% to 92.7% and specificity from 51.8% to 67.9%. 47 Another study showed 13 of 14 cancers (93%) occult on mammography but seen on CEM were in women with dense breasts. 48 The ICDR can be improved by 13 cases per 1000 patients in the intermediate risk category.
Diagnostic CEM is in use in Canada for problem-solving, pre-operative staging, surgical planning and assessment of treatment response during neoadjuvant chemotherapy. The radiation is higher than that of digital mammography or DBT but is within the allowable range.49,50 CEM requires patient consent to contrast and an IV. IV setup and injection are the same as for MRI, but CEM imaging and interpretation are faster than MRI. As in CT, most contrast reactions are mild and resolve spontaneously. Moderate reactions occur in less than 1% and personnel must be appropriately trained to manage these. Fatal outcomes are extremely rare with low-osmolality contrast agents.51,52
Other modalities for supplemental breast screening. Molecular Breast Imaging using technetium 99 m sestamibi is not yet available in Canada. It was developed at the Mayo Clinic and is in use in some centres for supplemental screening. The effective dose of radiation is approximately 4 times that of digital mammography or digital breast tomosynthesis (DBT) but is still within safe limits. 53 It takes 40 min to perform.
Adding MBI to 2D mammography in women with dense breasts has been shown to increase cancer detection by 7.7–8.8 invasive cancers per 1000 screened with a PPV3 of 19.4%–25%. 54
A multicenter trial comparing DBT to MBI in women aged 40–75 years with dense breasts is underway. The preliminary report showed cancer detection rates per thousand of 1.9 for DBT vs 11.2 for MBI (ICDR of 9.3 for MBI). PPV was 8% for DBT, 26% for MBI and 21% for the combination of DBT and MBI. 55
Digital breast tomosynthesis. DBT can reduce the rate of false positives and increases the cancer detection rate by up to 40%.56,57 However, it does not significantly increase cancer detection in Category D breasts. In Canada, there are higher recall rates than in the United Kingdom or in Europe. The CSBI strongly supports screening with DBT to reduce the rate of these false positives. 58 Reduced false positives also decrease the need for diagnostic work ups. Kerlikowski et al. 59 found that compared to digital mammography, DBT significantly reduced advanced cancer rates per 1000 examinations in the 3.6% of women with extremely dense breasts and at high risk of breast cancer but not for women in other density categories regardless of risk. Interval cancer rates were not decreased by DBT in any density category or risk level.
While DBT may be considered a supplemental screening tool to digital mammography, it does not replace supplemental screening with other imaging modalities in patients with dense breast tissue.60,61 Other supplemental screening modalities such as breast MRI, CEM or breast ultrasound should be used in these patients.
Thermography. Health Canada 62 and the U.S. Food and Drug Administration 63 strongly recommend against use of thermography for breast cancer screening. Thermography relies on breast cancer increasing the surface temperature of the breast and does not accurately detect breast cancers. Thermography is associated with low sensitivity of 25% and high rates of false positives that are 2 times higher than mammography. 64
Supplemental breast screening interval. The optimal screening frequency (e.g. annual and biennial) of supplemental screening has not been established, but it may depend to some extent, on how often a given woman is having screening mammograms. In one study supplemental MRI in average risk women of all breast densities found 16 per 1000 cancers in the first year, with no interval cancers over the next 3 years. 65 The DENSE trial in the Netherlands showed that it was cost-effective to offer screening breast MRI every 3 or 4 years, followed by alternating MRI with screening mammography every 2 years. 66 When women are having mammograms only every 2 years, the advantage of alternative year supplemental screening ultrasound allows them to ‘be screened’ (with one or the other modality) annually, allowing earlier detection of cancer.
What to Recommend for Specific Populations
Increased risk. There are multiple online risk calculation tools, and the choice of which to use depends on the purpose of the assessment.67,68 Women with increased risk for breast cancer need earlier and more intensive screening. For women with genetics-based increased risk (and their untested first-degree relatives) or with a calculated lifetime risk of 20%–25% or more, high-risk screening (mammography plus MRI) should be performed annually beginning at age 30 until age 69. 8 Screening mammography should continue thereafter, for as long as a woman is in excellent general health, with a life expectancy of at least 10 years, and as long as she is well enough to be treated, should a cancer be discovered.
For women with histories of chest radiation therapy (10 Gy or greater cumulative dose between 10 and 30 years of age), annual mammographic and MRI screening should begin at age 25 or 8 years after radiation therapy, whichever is later. 8
Some women with a personal history of breast cancer and breast conservation therapy may also be at high risk for new or recurrent breast cancer (≥20%) and may be eligible for high-risk screening. 8 Patients with biopsy diagnosis of atypia or lobular neoplasia (atypical ductal hyperplasia, atypical lobular hyperplasia, and lobular carcinoma in situ) may benefit from high-risk screening protocol, although this is not as well documented.
Transgender patients. Transgender, gender diverse and non-binary patients are at risk of under-screening. They may face barriers to screening related to sexual orientation, gender identity and gender dysphoria.69,70
Screening strategies can be approached based on anatomy present. Transgender patients with breast tissue and no history of chest construction surgery, or top surgery (bilateral subcutaneous mastectomy) should undergo screening as per the routine screening protocol. Screening mammography is not feasible in transgender patients who have had chest construction (top) surgery. Follow-up clinical breast examination with a primary care provider is advised for these patients. If there is clinical concern or the patient is high risk, physical exam and consideration of diagnostic ultrasound or other modalities may be considered.
Transgender patients with breast tissue secondary to gender affirming hormone therapy should undergo screening mammography. However, there are no evidence-based guidelines for screening in transgender women relative to hormone usage.71,72 A 2019 Position Statement from the Society of Breast Imaging (SBI) indicates most experts advise annual screening mammography starting at 40 for transgender women who have received hormone therapy for 5 or more years. 73 Transgender women are thought to be at lower risk than cisgender women. 73 There is limited evidence regarding how to incorporate risk factors including duration of hormone use, family history and BMI.71,72
Minorities. Black, Asian and Hispanic women are diagnosed with breast cancer younger than white women, with a peak incidence in the mid-40s. 74 Black and Asian women also have a higher risk profile and are more likely to develop more aggressive tumours, for example, triple negative.74-76 All women should be able to self-refer for screening mammography starting at age 40, but those minorities are especially encouraged and should be able to do so. Those with dense breasts should also have access to supplemental screening.74-76
Artificial Intelligence in Breast Screening
There is great potential for artificial intelligence (AI) to improve breast imaging. AI methods excel at automatically recognizing complex patterns in imaging data and providing quantitative, rather than qualitative, assessments of radiographic characteristics. 77 There are many emerging AI technologies to improve breast density standardization and automated breast cancer detection. AI in breast cancer screening is new, but promising. Some practices are already incorporating AI into practice; however, a systematic review of studies demonstrated mixed results. It is uncertain whether the quality of research was equivalent in the studies pooled for analysis. The conclusion was that more research is required.
Discrepancies in Screening Policies
Because health care is regulated in each province or territory, each jurisdiction has different recommendations and screening access. A summary of the recommendations is found in Table 1. 34
The CSBI is working with the CAR to advocate for changes to the federally funded Canadian Task Force and to unify the provincial and territorial screening program recommendations.
The Canadian Task Force on Preventive Health Care (CTFPHC) 6 and the US Preventive Services Task Force (USPSTF) 78 provide federal government funded guidelines and make similar recommendations to start screening at age 50. Neither panel included content experts. The panel that issued breast cancer screening guidelines had no breast surgeon, oncologist, radiologist or patient. In the US, the 2009 and 2016 breast screening recommendations were suspended by Congress. 79 During this ongoing moratorium, the 2002 USPSTF guideline, recommending screening starting at age 40, remains in place. The USPSTF is now reviewing their guidelines and taking racial disparities into consideration. Canada should do the same, given that Black and Asian women have a younger age of breast cancer onset and peak incidence and should not wait till age 50 to begin screening. There has been no effort in Canada by the federal government to apply a similar moratorium on, or review of the CTFPHC guidelines, which is why physician education is so important.
Canadian physicians need to be aware that the decision tool supplied to them by the CTFPHC for use in shared decision-making is largely influenced by the Canadian National Breast Screening Study-1, which has now been shown to be invalid because of proven compromise of the randomization process. 80
The CSBI has re-created the 1000 women decision tool with accurate data, shown in Figure 2.
Conclusion
We have addressed common misinformation about breast screening and the Canadian breast screening guidelines. The ages to screen and optimal screening intervals for average and high-risk patients have been reviewed. We describe the different imaging modalities used in screening and supplemental screening and that more research is required to explore the potential benefit of AI in breast screening. The current provincial screening recommendations have been provided, and ideally should change to allow optimal screening uniformly across Canada, including self-referral for mammography starting at age 40. We hope this review will help radiologists and referring physicians understand and navigate the varied international and provincial recommendations and guidelines and make the best recommendations for their patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors C.Y.-H. and J.S. sit on the Canadian Society of Breast Imaging Board of Directors. Author P.G. is the Medical Director of Dense Breast Canada.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
