Abstract
Introduction
Breast cancer is one of the most common cancers in the world and is a leading cause of cancer death in women worldwide. 1 In 2020, 2.3 million women were diagnosed with breast cancer and 685 000 deaths were caused by breast cancer globally. 1 A similar level of burden can be found in Canada, where breast cancer is the most common non-cutaneous cancer among Canadian women and the second leading cause of death from cancer among Canadian women. 2 According to the Canadian Cancer Society, it is estimated that about one in 8 Canadian women will develop breast cancer in their lifetime and that one in 34 will die from breast cancer. 2
Screening mammography plays an important role as it can help identify breast cancer when it is too small to be detected with physical examination, which then allows for earlier diagnosis, more treatment options, and better chances of survival. 3 For women aged 40 to 69 years old, screening mammography has been shown to reduce mortality from breast cancers by 41% in those screened. 4 In British Columbia (BC), there has been an organized breast cancer screening program across the province since 1988, overseen by BC Cancer. 3
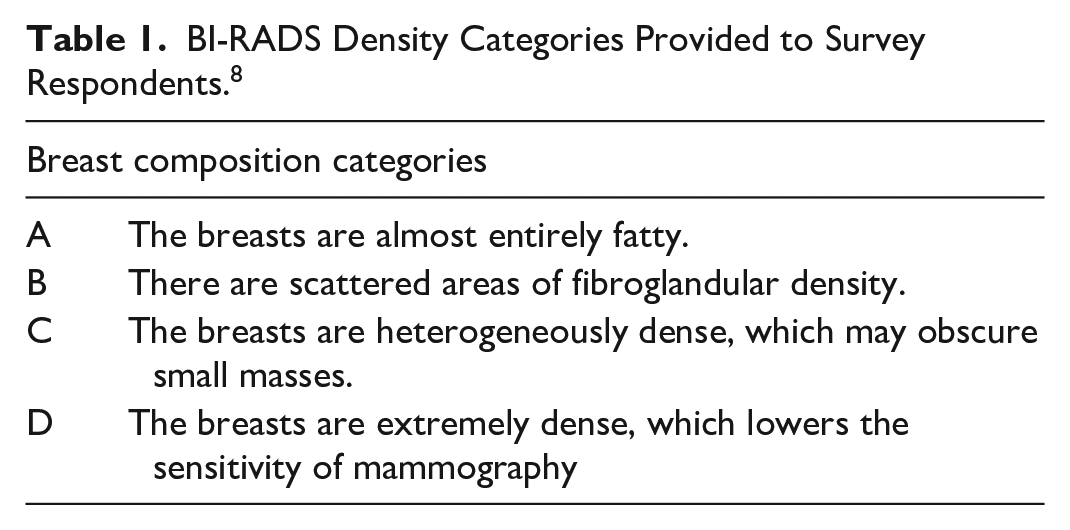
Breast density refers to the proportion of fibroglandular tissue to fat in the breast.5-8 Breasts with more fibroglandular tissue are denser.5-8 Increased density limits the sensitivity of mammography because cancers are harder to see on the background of more fibroglandular tissue.5-7 In addition, patients with dense breasts are at increased risk of breast cancer compared to patients with less dense breasts.5-7 Breast density reporting on screening mammography has been mandated in BC since October 2018. These scores are based on the Breast Imaging Reporting and Data System (BI-RADS) scoring system, where breast density is assigned a score of A, B, C, or D. Breasts with BI-RADS C and D scores are considered dense breasts, with BI-RADS D indicating the highest density.
Currently, there are no clear guidelines or consensus from radiology societies and organizations on a recommended supplemental imaging modality or protocol for average risk patients who have dense breasts. 7 The reporting of BI-RADS density scores is merely intended to facilitate discussions between providers and patients on breast cancer risk, as well as on the limitations of mammography. 8 The BC Cancer Breast Screening Program does not recommend ordering supplemental testing with breast ultrasound, magnetic resonance imaging (MRI), or contrast enhanced mammography (CEM) for patients based solely on higher BI-RADS density scores due to a current lack of evidence on the benefits and limitations of supplemental testing. 8 However, these investigations remain available to order at the discretion of primary care providers, who are traditionally responsible for assessing patients’ breast cancer risk, discussing benefits and limitations of supplemental testing, and arranging additional investigations as warranted. Supplemental screening with breast ultrasound has been a Medical Services Plan (MSP) covered service since 2019; however, supplemental screening with MRI and CEM are not.9,10 What remains to be answered is how the inclusion of BI-RADS density scores in screening mammography reports has influenced the current practice of primary care providers in BC when screening for breast cancer in patients.
The primary objective of this study was to evaluate how primary care providers in Canada utilize BI-RADS density scores reported on normal screening mammograms of average risk, asymptomatic patients in their clinical practice. The secondary objective of this study was to determine if there are any patterns related to primary care provider demographics and practice settings in BC that could be linked to differences in screening practices for patients based on BI-RADS density scores.
Methodology
Design
A prospective cross-sectional survey study was conducted with primary care providers practicing in the province of BC. For the purposes of this study, primary care providers were defined as family physicians (FPs) and nurse practitioners (NPs).
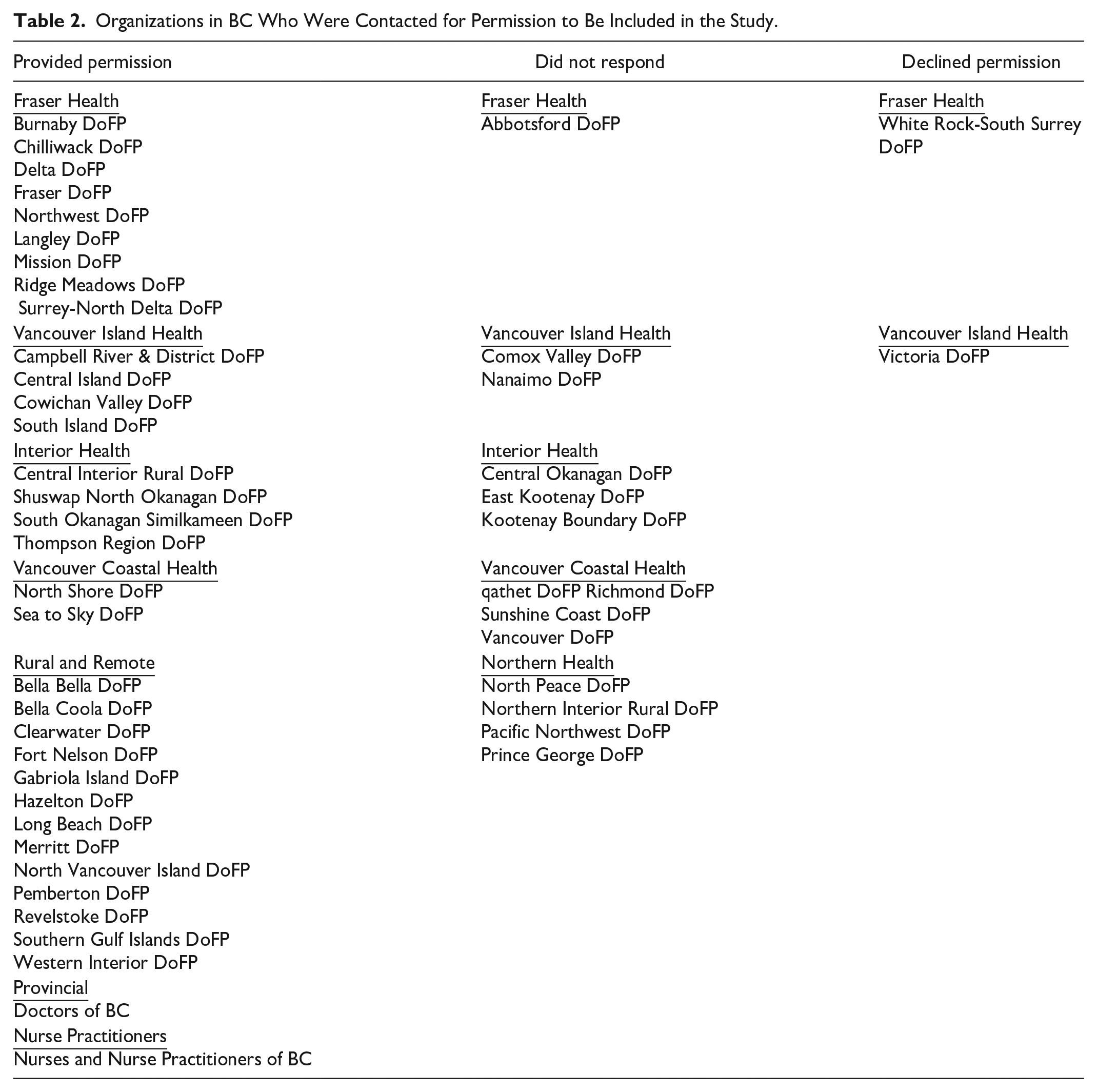
Registrant members of various Divisions of Family Practices (DoFP) located across BC and the Nurse and Nurse Practitioners of BC were invited to participate. These organizations were contacted to request permission for an invitation to be sent to their members via email, newsletters, or website bulletins. Only those organizations who provided permission were included in the study. Participants were invited to answer questions through the virtual Survey Tool administered using Qualtrics software and accessed the survey directly from a link in the invitation. All participants were asked to review a consent and privacy statement that detailed the protection of their data before proceeding to access the survey. Participants were included if they were actively practicing in BC at the time of the survey, conducted breast cancer screening for women 40 years of age and older, and were able to read and complete the survey in English. Those excluded were providers who were not actively practicing in BC or could not complete the survey in English.
A description of the BI-RADS density categories (detailed in Table 1) was provided to participants in the survey. 8 The survey consisted of 4 question sections: (1) participant demographics, (2) knowledge of breast cancer risk in relation to breast density, (3) attitudes toward breast density in breast cancer screening, and (4) practice patterns based on reported BI-RADS density scores. Questions were primarily closed-ended using a predefined set of answer options, with fields to enter open-ended responses as applicable, or questions with choices ranging from “always” to “never.”
BI-RADS Density Categories Provided to Survey Respondents. 8
Data Analysis
Survey responses were analyzed on a per-question basis, meaning that for each question, only the participants who entered a response were included in the respective analysis. Descriptive statistics were calculated using percentages and further stratified by participant demographics. Supplemental testing based on BI-RADS density scores alone were adjusted for by gender, profession, years in practice, and community practice size. P values were derived from Fisher’s exact test and results were regarded as statistically significant at P < .05. Open-ended responses were analyzed by 2 independent reviewers and categorized into prevalent themes using an inductive thematic analysis approach.
Results
A total of 50 organizations in BC were contacted and 34 provided permission for invitations to be sent to their registrant members (Table 2).
Organizations in BC Who Were Contacted for Permission to Be Included in the Study.
Participant Demographics
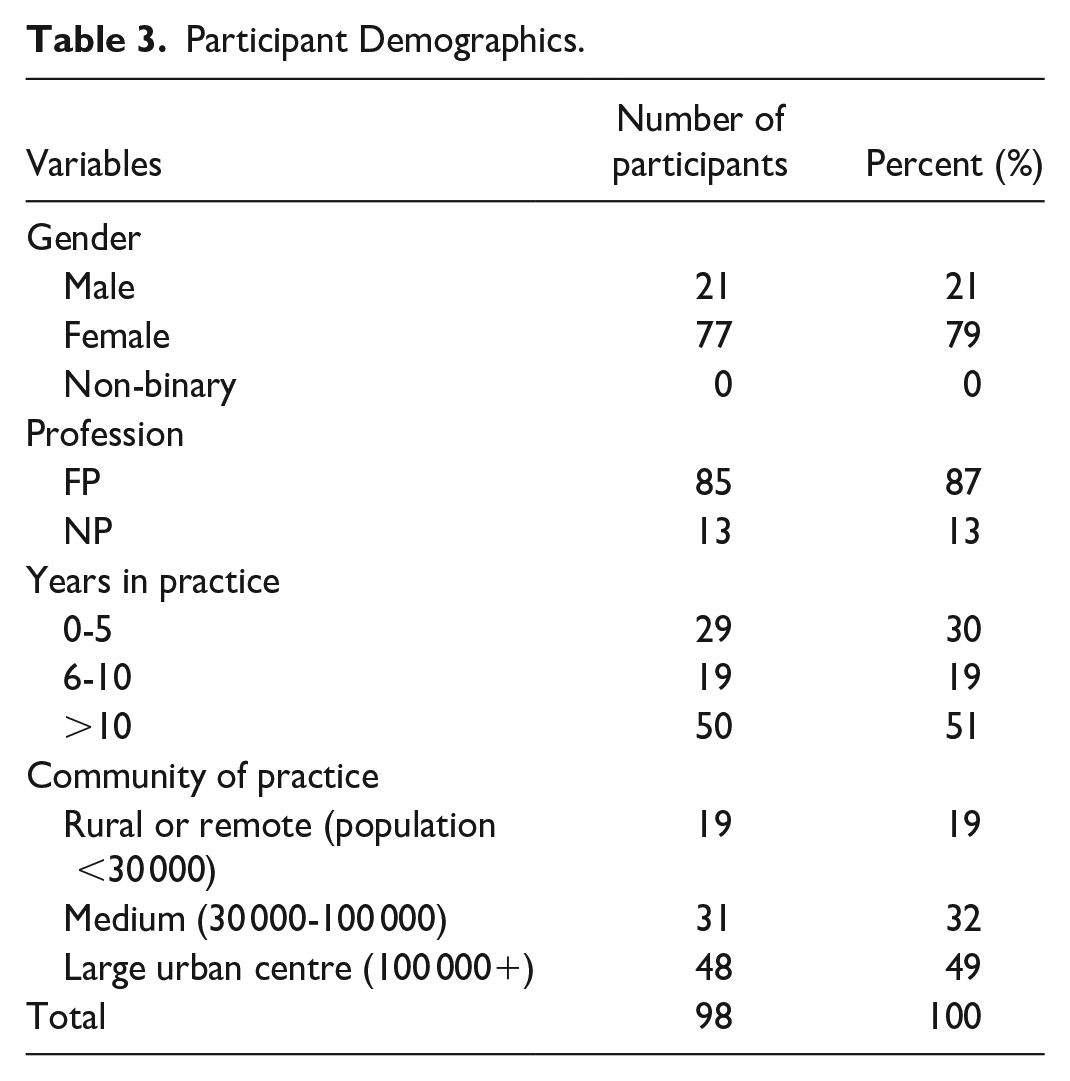
In all, 98 primary care providers responded to the survey between November 5, 2023 and January 14, 2024. Eighty-seven percent were FPs (n = 85) and 13% were NPs (n = 13). Of these participants, 21% (n = 21) were male and 79% (n = 77) were female. Fifty-one percent had been in practice for more than 10 years (n = 50) and 49% practiced in large urban centres with populations over 100 000 (n = 48). The full participant demographics are detailed in Table 3.
Participant Demographics.
Supplemental Testing
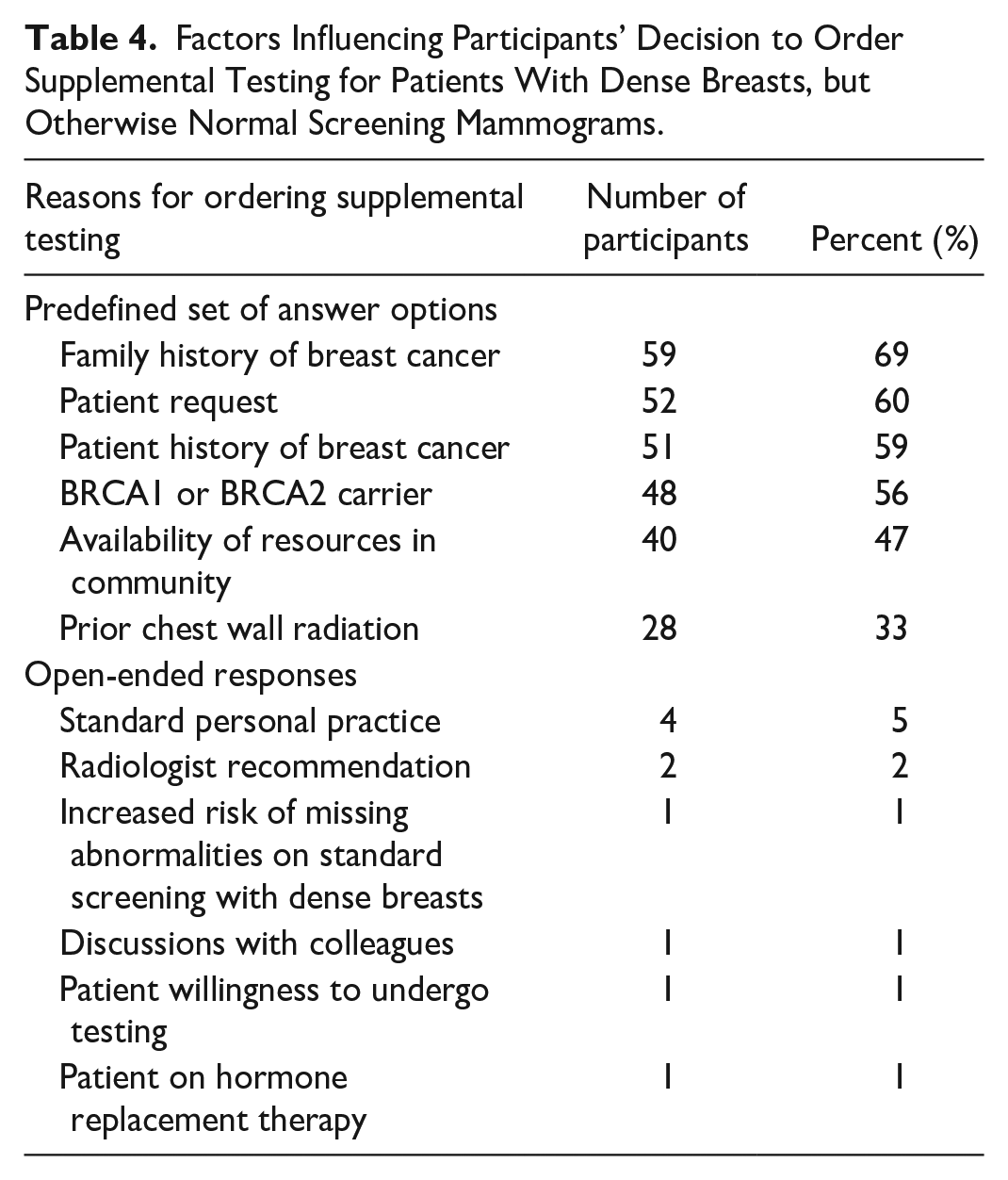
The percentage of participants who order supplemental testing based on BI-RADS density scores alone were: 8% (n = 8) if patients have BI-RADS D; 37% (n = 35) if patients have BI-RADS C or D; and 2% (n = 2) if patients have BI-RADS B, C, or D. Overall, 53% (n = 50) of participants do not order supplemental testing based on BI-RADS density scores alone. Of the participants that order supplemental testing, 73% (n = 44) would order a breast ultrasound only, 5% (n = 3) would order a breast MRI only, 7% (n = 4) would order a breast ultrasound and MRI, 13% (n = 8) would order a breast ultrasound and mammogram, and 2% (n = 1) would order a breast ultrasound, MRI, and mammogram. Factors influencing participants’ decision to order supplemental testing are listed in Tables 4 and 5.
Factors Influencing Participants’ Decision to Order Supplemental Testing for Patients With Dense Breasts, but Otherwise Normal Screening Mammograms.
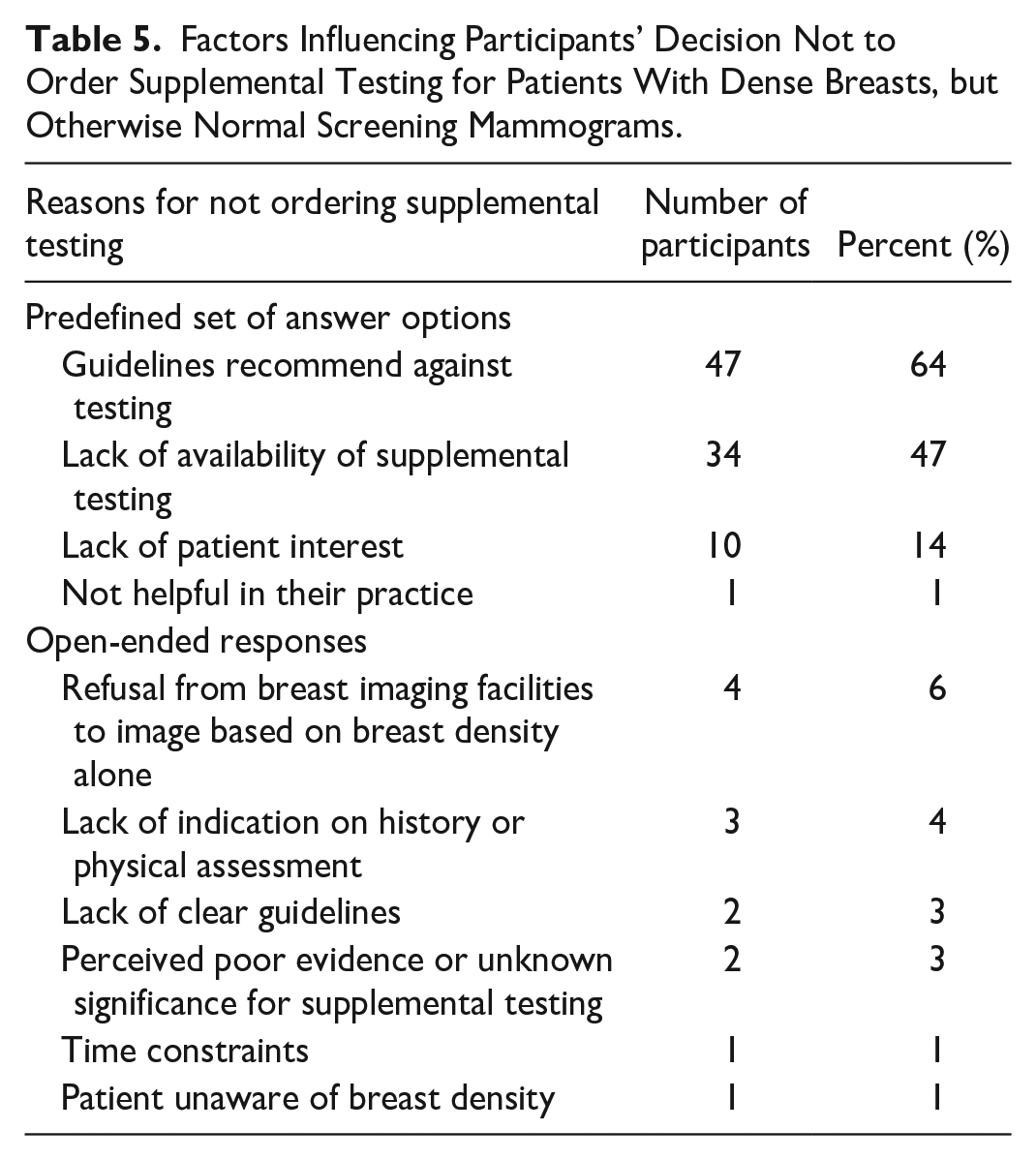
Factors Influencing Participants’ Decision Not to Order Supplemental Testing for Patients With Dense Breasts, but Otherwise Normal Screening Mammograms.
When stratified by participant demographics (Figure 1), 48% (n = 36) of female participants and 45% (n = 9) of male participants would order supplemental testing based on BI-RADS density scores alone (P = 1). Forty-nine percent (n = 40) of FPs and 39% (n = 5) of NPs would order supplemental testing based on BI-RADS density scores alone (P = .56). Fifty-three percent (n = 26) of participants who had been in practice for more than 10 years, 50% (n = 9) of those who had been in practice for 6 to 10 years, and 36% (n = 10) of those in practice for 5 years or less would order supplemental testing based on BI-RADS density scores alone (P = .34). Fifty-seven percent (n = 26) of those practicing in large urban centres, 43% (n = 13) of those practicing in medium-sized communities, and 32% (n = 6) of those in rural or remote communities would order supplemental testing based on BI-RADS density scores alone (P = .17).
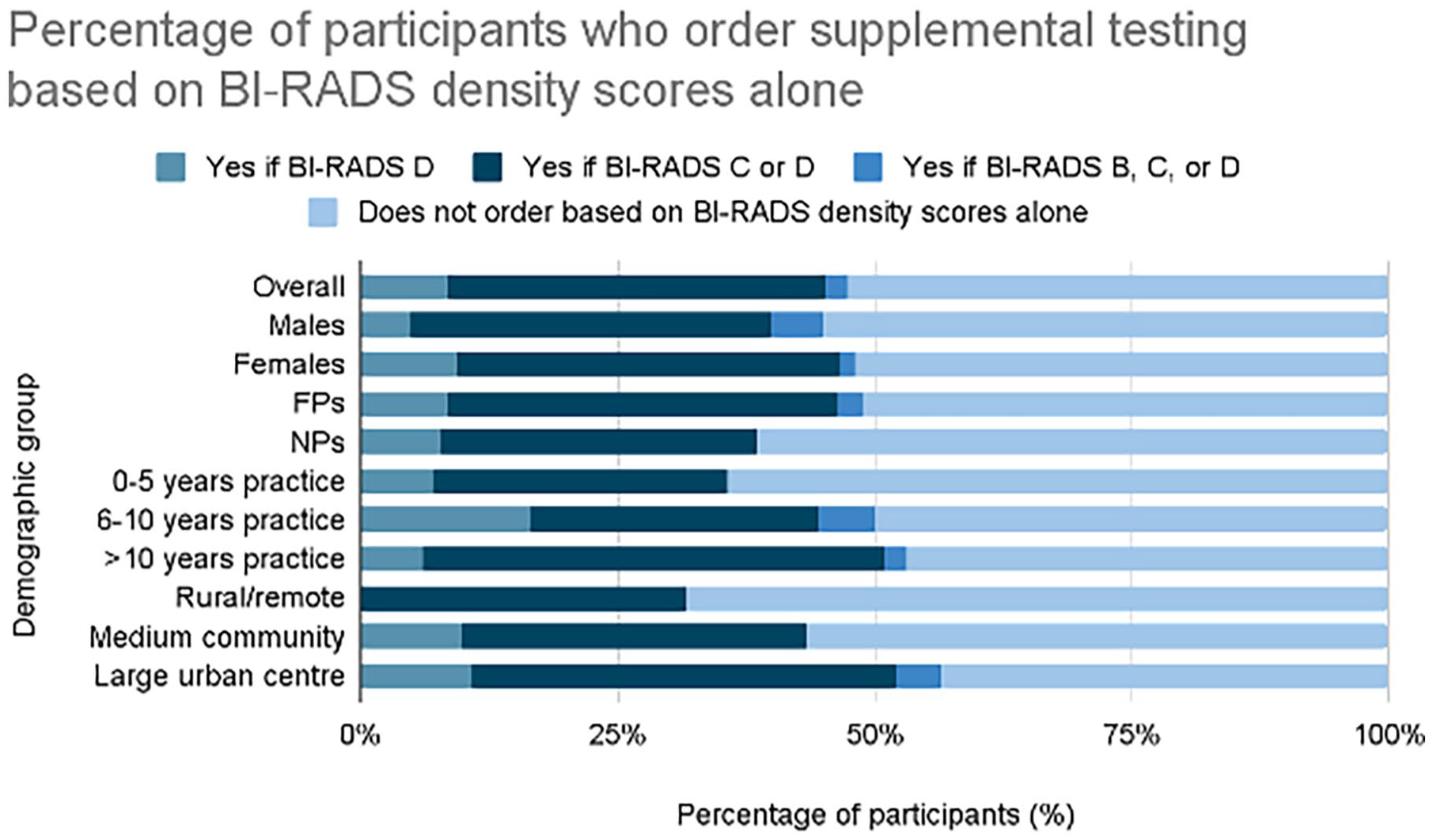
Screening practices based on BI-RADS density scores, stratified by participant demographics.
Knowledge and Attitudes
Overall, 57% (n = 55) of the participants were aware of the increased risk of breast cancer with higher breast density and 43% (n = 41) were unaware or unsure. Eighty percent (n = 77) of participants felt that they needed more education about the significance of breast density and supplemental breast screening.
When asked how often they have discussions with their patients about breast density, 24% (n = 23) responded always, 54% (n = 52) responded sometimes, 16% (n = 15) responded rarely, and 6% (n = 6) responded never. When asked how often they routinely review BI-RADS density scores on screening mammography reports with their patients, 33% (n = 31) indicated always,
52% (n = 49) indicated sometimes, 13% (n = 12) indicated rarely, and 3% (n = 3) indicated never.
Discussion
This prospective cross-sectional survey evaluated how primary care providers in BC utilize BI-RADS density scores in breast cancer screening for average risk, asymptomatic patients with normal screening mammography. The results of this study demonstrate there is a substantial proportion of primary care providers who do not order supplemental testing based on BI-RADS density scores alone, with another considerable, albeit comparatively smaller, proportion who order supplemental testing for patients with BI-RADS density scores C or D. For providers who order supplemental testing, the majority would order a breast ultrasound only and a smaller number order additional or alternative tests, such as a breast MRI. By far, the most common reasons cited for not ordering supplemental testing were current BC guidelines recommending against doing so and lack of availability of supplemental testing. Interestingly, there were no clinically significant differences in practice patterns when broken down by participant demographics. Furthermore, the results of this study demonstrated variability in how often providers discuss and review BI-RADS density scores with their patients. Overall, these findings demonstrate there are practice pattern variations between primary care providers in BC and their decision to order supplemental testing based on BI-RADS density scores alone.
The results of this study bring attention to a knowledge gap that exists among many primary care providers in BC regarding the increased risk of breast cancer with higher breast density and areas of uncertainty in the role of supplemental testing. The majority of primary care providers in the study endorsed the need for more education around breast density. A 2019 survey assessing physician knowledge of supplemental testing following reports of breast density scores in New York found 62% of respondents were not aware of the increased risk of breast cancer with higher breast density, 67% felt they would benefit from more education around breast density and supplemental testing, and only 25% reported having discussions about breast density with their patients. 7 This knowledge gap and uncertainty exist despite evidence linking breast density with breast cancer risk. A recent systematic review and meta-analysis showed that patients with BI-RADS D scores have a 1.83-fold higher risk of developing breast cancer compared to patients with BI-RADS B scores. 5 In addition, sensitivity of mammography has been shown to decrease with increased density of breast tissue, dropping from 81% to 93% for BI-RADS A, to 84% to 90% for BI-RADS B, 69% to 81% for BI-RADS C, and 57% to 71% for BI-RADs D. 6 Patients could conceivably be at higher risk for developing breast cancer or have small cancers missed if their screening mammography reported a higher BI-RADS density score. Yet, the current BC Cancer Breast Screening guidelines do not recommend supplemental testing based on BI-RADS breast density scores alone. 8 The results of this study are relatively similar to findings in a previous study conducted at the Mayo Clinics in Minnesota, Florida, and Arizona that aimed to evaluate primary care providers’ awareness and familiarity with dense breasts and supplemental breast screening. The study conducted at these clinics found only 26% of respondents offered supplemental testing to all patients with dense breasts reported on their mammogram reports, while 47% offered supplemental testing based on additional risk factors. 11 When assessing comfort levels in discussing breast density with patients, 47% of respondents felt uncomfortable or only slightly comfortable doing so. 11
It has been argued that there are inherent limitations of supplemental testing including false positives, overdiagnosis, and limitations in sensitivity. 8 Overdiagnosis could increase costs on the healthcare system as well as cause unnecessary anxiety for patients, potentially resulting in more harm than benefit. A study conducted in the United States (US) with patients 50 to 74 years of age who were simulated to receive biennial mammography alone, mammography plus ultrasound for those with extremely dense breasts, or mammography plus ultrasound if they had heterogeneously or extremely dense breasts, found that supplemental ultrasound imposed a higher cost overall. 12
Of 1000 women who received a supplemental ultrasound for heterogeneously or extremely dense breasts, 354 had false-positive biopsy results which had an estimated additional cost of $560 000. 12
Supplemental ultrasound averted 0.36 additional breast cancer deaths and led to an increase of 1.7 quality-adjusted life years per 1000 women. 12 It is based on these and other findings that supplemental testing for patients with dense breasts on mammography using breast ultrasounds is not currently recommended on a population level in the US.12,13
On the other hand, benefits of supplemental testing include earlier detection of cancers or detection of cancer after a negative screening mammography, which may potentially allow for earlier treatment and better prognosis.6,8 A large randomized controlled trial conducted with Japanese women between the ages of 40 and 49 without a history of breast cancer found that patients who received both mammography and ultrasound had a higher sensitivity rate, albeit lower specificity rate, of detecting breast cancer compared to mammography alone. 14 That trial concluded that ultrasound could be an effective screening tool for early detection of breast cancer, especially in people with dense breast tissue. 14 Additionally, a study in BC evaluating breast ultrasound screening in women with dense breasts (BI-RADS C or D) and negative mammograms found a breast cancer detection rate of 7 in 1000 cases screened. 15 Their findings support the implementation of supplemental breast ultrasounds for breast cancer screening in patients with dense breasts. 15
Beyond ultrasound as a supplemental imaging modality for breast cancer detection, there are other modalities that can be used such as Digital Breast Tomosynthesis (DBT), Contrast-Enhanced MRI (CE MRI), Contrast-Enhanced Mammography (CEM), and Molecular Breast Imaging (MBI). 16 DBT, approved by the FDA in 2011, improves cancer detection by reducing tissue overlap, particularly in heterogeneously dense breasts, but its efficacy is limited in extremely dense tissue, often necessitating additional screening. 17 CE MRI, is another option, particularly for detecting biologically aggressive cancers, minimizing overdiagnosis of less aggressive forms, and is particularly suited for high-risk women. 18 CEM combines functional imaging with standard mammography, using iodinated contrast to highlight abnormal vascular patterns, enhancing cancer visibility. 19 MBI, leveraging radionuclide uptake in cancerous tissues, provides high cancer detection rates, especially in dense breasts, though concerns about radiation exposure limit its use. 20 Regardless of modality, all women with dense breasts may benefit from supplemental screening after mammography or DBT.
Furthermore, it is important to examine the reasons underlying BC’s current position against the routine use of supplemental imaging modalities. This would provide critical insight into the variability in their implementation across regions. Issues such as primary care physician shortages can impact referral rates to supplemental imaging. 21 Limited and variations in healthcare funding and spending across different parts of BC could make the high cost of supplemental imaging a potential strain on resources, while incomplete coverage may lead to financial burdens and inequitable access for patients. 22 Additionally, access to advanced imaging is hampered by infrastructure shortages in rural areas and a lack of radiologists particularly in underserved regions. The absence of standardized guidelines further complicates implementation, creating uncertainty among practitioners and resulting in inconsistent use across different areas.
Limitations
This study has several limitations inherent to survey-based research, including reporting and selection bias. Not all participants answered every question, potentially skewing responses, though most questions still had 97% participation. The predefined answer options may have influenced responses, especially regarding decision-making for supplemental testing, despite providing an open-ended field to capture additional insights. Categorizing open-ended responses into themes could introduce bias and subjectivity, though using two independent reviewers aimed to reduce this. Additionally, only organizations that consented participated. The impact of non-response bias, as data from non-participants were not captured, could disproportionately affect the results if the perspectives of non-responding organizations differ substantially from those that consented to participate. Such bias may limit the generalizability of the findings to all primary care providers in BC.
Future Considerations
Further research is needed to evaluate the utility of supplemental testing in breast cancer screening for patients with higher BI-RADS density scores. To shed further light on this issue, a randomized study is currently underway to evaluate the value of breast ultrasound as a supplemental test for patients with increased breast density. 8
Conclusion
This study demonstrated that there are variations in how primary care providers in BC utilize the BI-RADS density scores reported on normal screening mammography of average risk, asymptomatic patients in their clinical practice. Factors that may explain this finding include the current BC Cancer Breast Cancer Screening guidelines not recommending supplemental testing, the lack of availability of supplemental testing, and primary care providers’ uncertainty about the role of supplemental testing for patients with dense breasts. The results of this study highlight not only the need for better, higher-quality evidence to standardize clinical practice guidelines for risk management of patients with dense breasts, but also the need for more supplemental breast cancer screening resources to help advance breast cancer screening in BC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
