Abstract
The integration of Digital Breast Tomosynthesis (DBT) and Artificial Intelligence (AI) represents a significant advance in breast cancer screening. This combination aims to address several challenges inherent in traditional screening while promising an improvement in healthcare delivery across multiple dimensions. For patients, this technological synergy has the potential to lower the number of unnecessary recalls and associated procedures such as biopsies, thereby reducing patient anxiety and improving overall experience without compromising diagnostic accuracy. For radiologists, the use of combined AI and DBT could significantly decrease workload and reduce fatigue by effectively highlighting breast imaging abnormalities, which is especially beneficial in high-volume clinical settings. Health systems stand to gain from streamlined workflows and the facilitated deployment of DBT, which is particularly valuable in areas with a scarcity of specialized breast radiologists. However, despite these potential benefits, substantial challenges remain. Bridging the gap between the development of complex AI algorithms and implementation into clinical practice requires ongoing research and development. This is essential to optimize the reliability of these systems and ensure they are accessible to healthcare providers and patients, who are the ultimate beneficiaries of this technological advancement. This article reviews the benefits of combined AI-DBT imaging, particularly the ability of AI to enhance the benefits of DBT and reduce its existing limitations.
Introduction
Breast cancer continues to be a major health challenge worldwide, affecting approximately 2 million women each year. It is the leading cause of cancer-related deaths in women despite the widespread implementation of screening programs. 1 Mammography, the principal method for breast cancer screening, is crucial for early detection. 2 However, it has limitations; while 2D mammography detects about 87% of breast cancers, 3 it often fails in women with dense breasts, which can mask cancer signs on mammograms. 4 This is particularly problematic given that nearly half of all women undergoing screening have dense breasts. 5 Mammograms not only miss certain cancers but also have a high rate of false positives, 2 which can lead to unnecessary further testing and anxiety. Moreover, mammograms can identify precancerous conditions that might never become problematic yet often result in treatment due to our inability to predict the risks of non-intervention.6,7 This highlights the need for advancements beyond traditional digital mammography.
Digital Breast Tomosynthesis (DBT), a newer technology, improves upon traditional methods by obtaining multiple images from different angles, reducing the issue of tissue overlap inherent in 2D mammography.8,9 DBT has demonstrated increased cancer detection rates with an incremental cancer detection rate of 1.2 to 2.7 cases per 1000 screenings with a variable impact on recall rates. 10 Combined with synthetic 2D imaging, this technique offers slighter greater radiation dose compared to standard mammography, addressing initially raised concerns about increased radiation exposure.11,12
The shift from 2D mammography to DBT in the United States is evident, 13 with a significant increase in facilities adopting DBT over the past 8 years. Despite its advantages, DBT faces challenges, such as higher costs, greater storage requirements, and longer times needed for image interpretation, which may hinder its widespread adoption. 10
On the other hand, integrating Artificial Intelligence (AI) in medical devices, particularly in radiology, is reshaping diagnostics. 14 This article reviews the benefits of combined AI-DBT imaging particularly the ability of AI to enhance the benefits of DBT and reduce its existing limitations.
DBT: Enhancing Detection Through AI-Computer-Aided Diagnosis (AI-CAD) Software
The use of computer-aided diagnosis (CAD) tools in mammography is not novel, with over 2 decades of clinical application. 15 Originally, CAD served as an adjunct tool, offering a “second look” to improve detection rates. 15 Early CAD systems relied on manually defined image features and basic classifiers, often inadequate for complex diagnostic tasks and has been demonstrated to increase false positives. 15 In contrast, AI-powered CAD, specifically deep learning-based AI-CAD, utilizes advanced multi-layer neural networks that autonomously learn from large datasets to identify relevant features. 16 This advancement has marked a new phase in CAD’s evolution, improving clinical decision support throughout various stages of patient care. 17 Within the context of DBT, AI-CAD not only holds the potential to distinguish between normal and abnormal findings and improve disease detection but also improves workflow efficiency by managing and prioritizing reading tasks. 10 Figure 1 outlines the key potential benefits of this synergistic technological advancement.
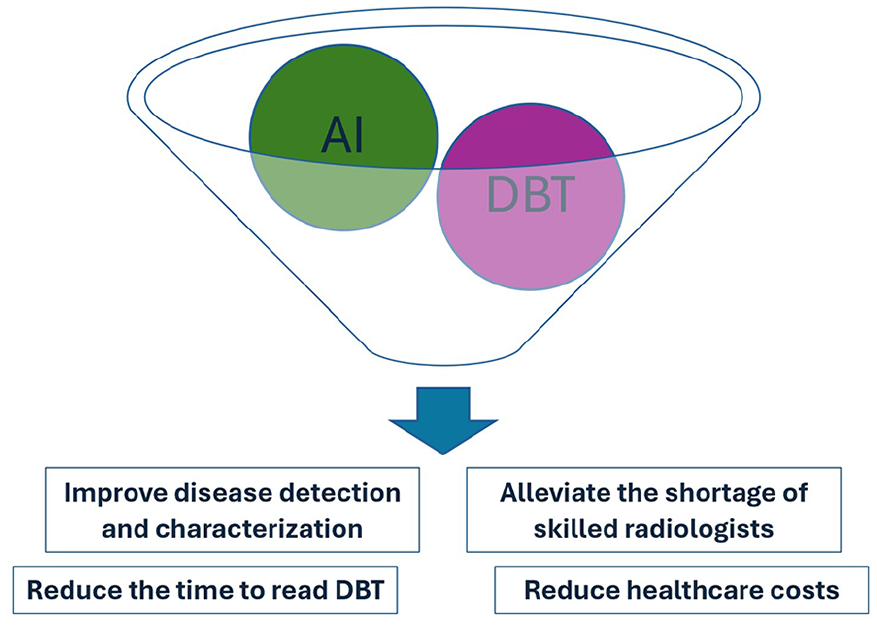
Key potential benefits of this synergistic technological advancement.
Unpacking How AI Enhances DBT: Clarifying the Mechanism
Deep learning algorithms in Digital Breast Tomosynthesis (DBT) scrutinize each image slice for signs of abnormality, focusing on specific features such as calcifications, soft tissue masses, asymmetries, distortion, or a combination of them. The AI system assigns scores to identified lesions, with the lesion score reflecting the algorithm’s confidence that a lesion is malignant based on image analysis. 18 In contrast, the case score evaluates the overall likelihood that the entire DBT exam reveals a malignant lesion, offering a broader assessment rather than focusing on individual lesions. 18
The AI software calculates scores by comparing detected lesions against a database of biopsy-proven cancerous lesions. 19 In that way, the AI algorithms can identify abnormalities and assess the likelihood of malignancy, providing critical support in clinical decision-making. For example, a lesion scoring 80% is deemed more suspicious than 80% of the malignant lesions in the database, signalling a high concern for potential malignancy. A lower score indicates less suspicion compared to the database’s malignant lesions, but it does not rule out the possibility of cancer. The lesion score primarily aids radiologists by highlighting areas that might require more detailed examination and potentially further testing. It’s crucial to recognize that a 100% lesion score does not confirm cancer, as AI cannot replace tissue sampling. Figures 2 to 4 display examples of the AI-DBT tool.
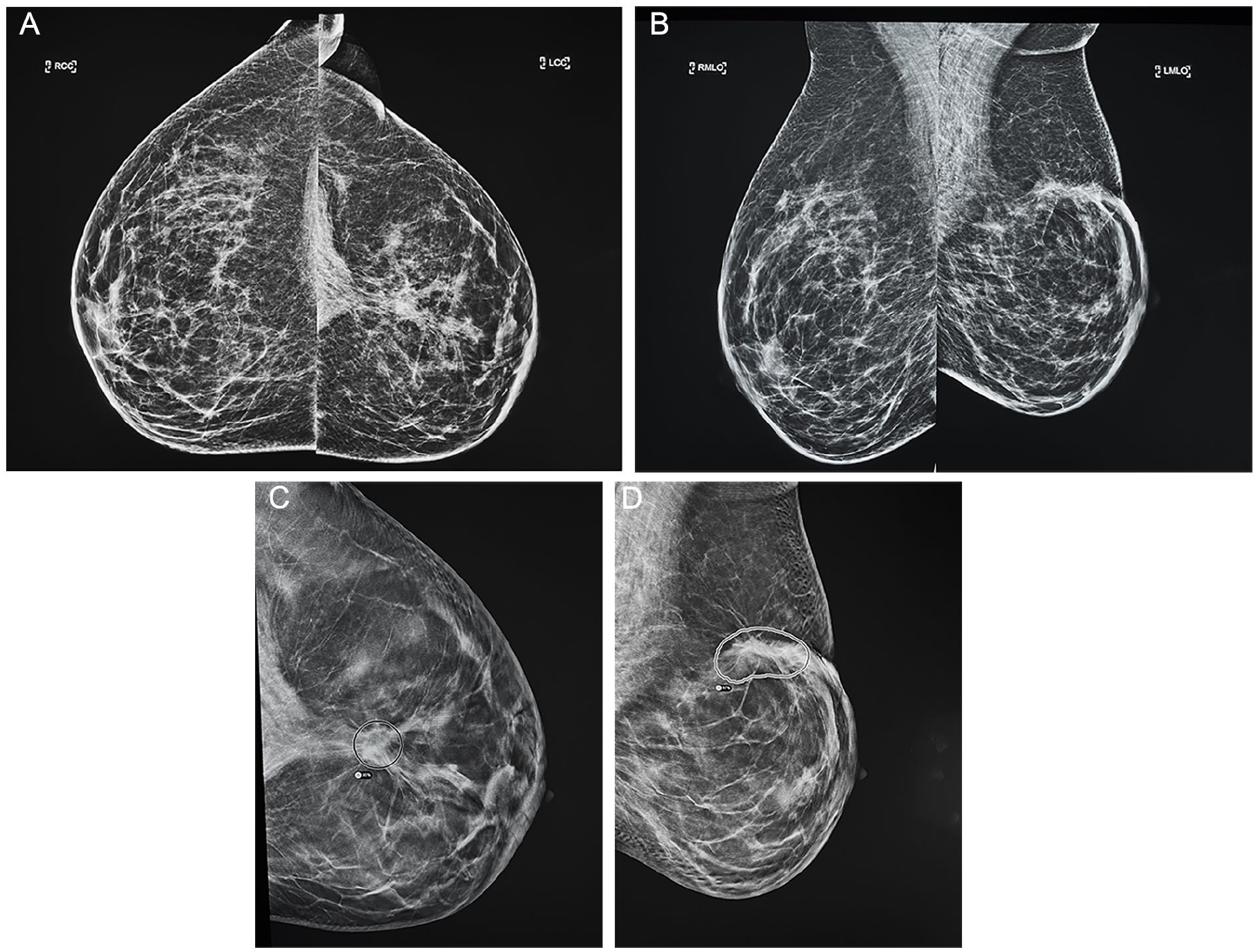
A 49-year-old patient with a history of left malignant lumpectomy was assessed using AI-DBT imaging, which indicated a high-priority case with up to a 90% lesion score. Given the patient’s history, we could classify the finding as benign. (A, B) Bilateral craniocaudal and mediolateral oblique mammogram views showed left-sided postoperative changes. (C) AI-DBT imaging on the craniocaudal showed a 90% lesion score. (D) AI-DBT imaging on mediolateral oblique view showed an 87% lesion score.
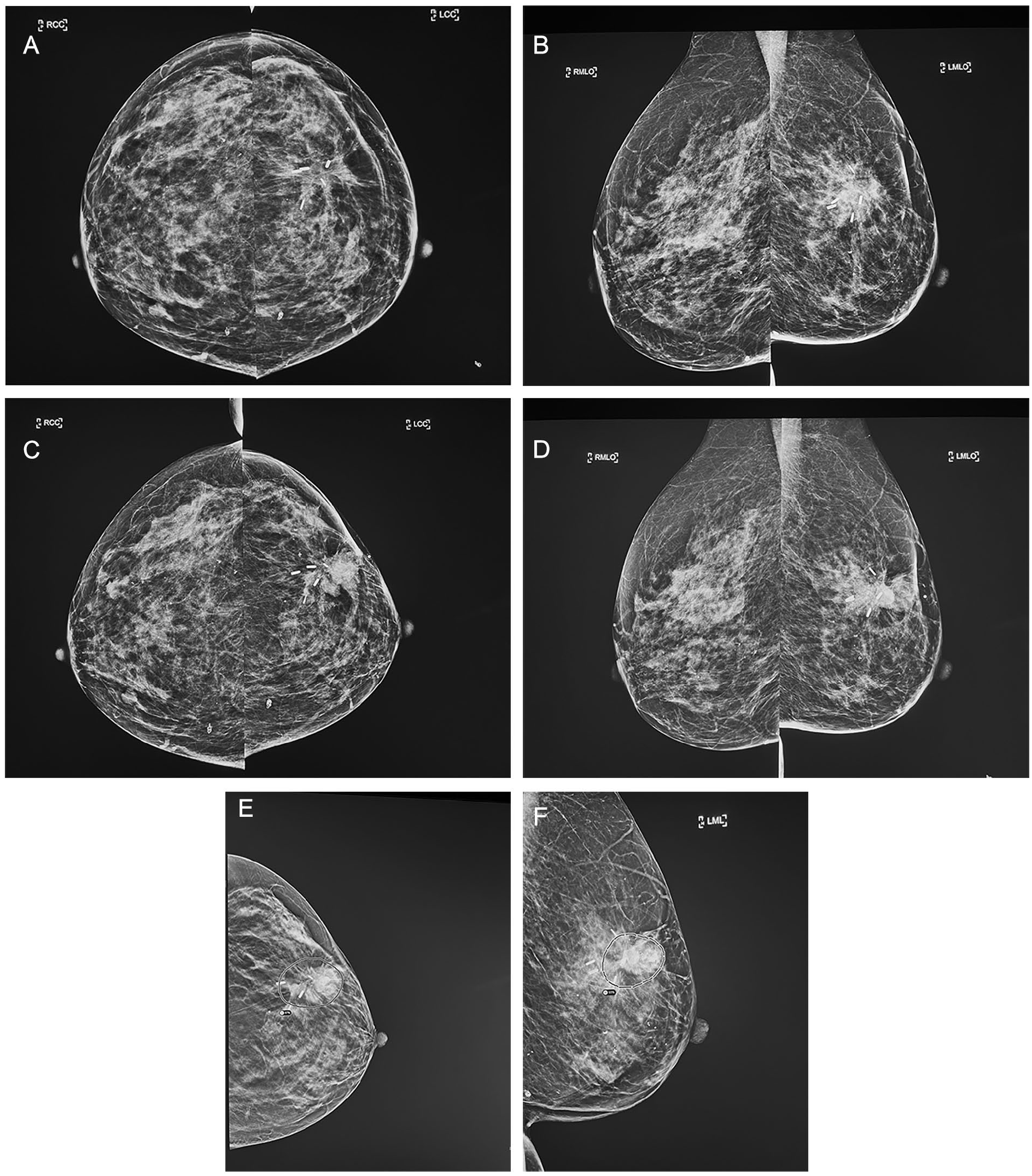
A 52-year-old patient with a previous history of left malignant lumpectomy presenting with a biopsy-proven left side recurrence, highlighted as a high-priority case by the AI-DBT imaging, with up to 93% lesion score. (A, B) Bilateral craniocaudal and mediolateral oblique mammogram 3 years ago showed postoperative changes in the left breast. (C, D) Recently, bilateral craniocaudal and mediolateral oblique mammogram views demonstrate, at the left surgical site, a development of an equal density irregular mass with associated internal amorphous calcifications (E) AI-DBT imaging on the craniocaudal showed an 87% lesion score. (F) AI-DBT imaging on mediolateral oblique view showed a 93% lesion score.
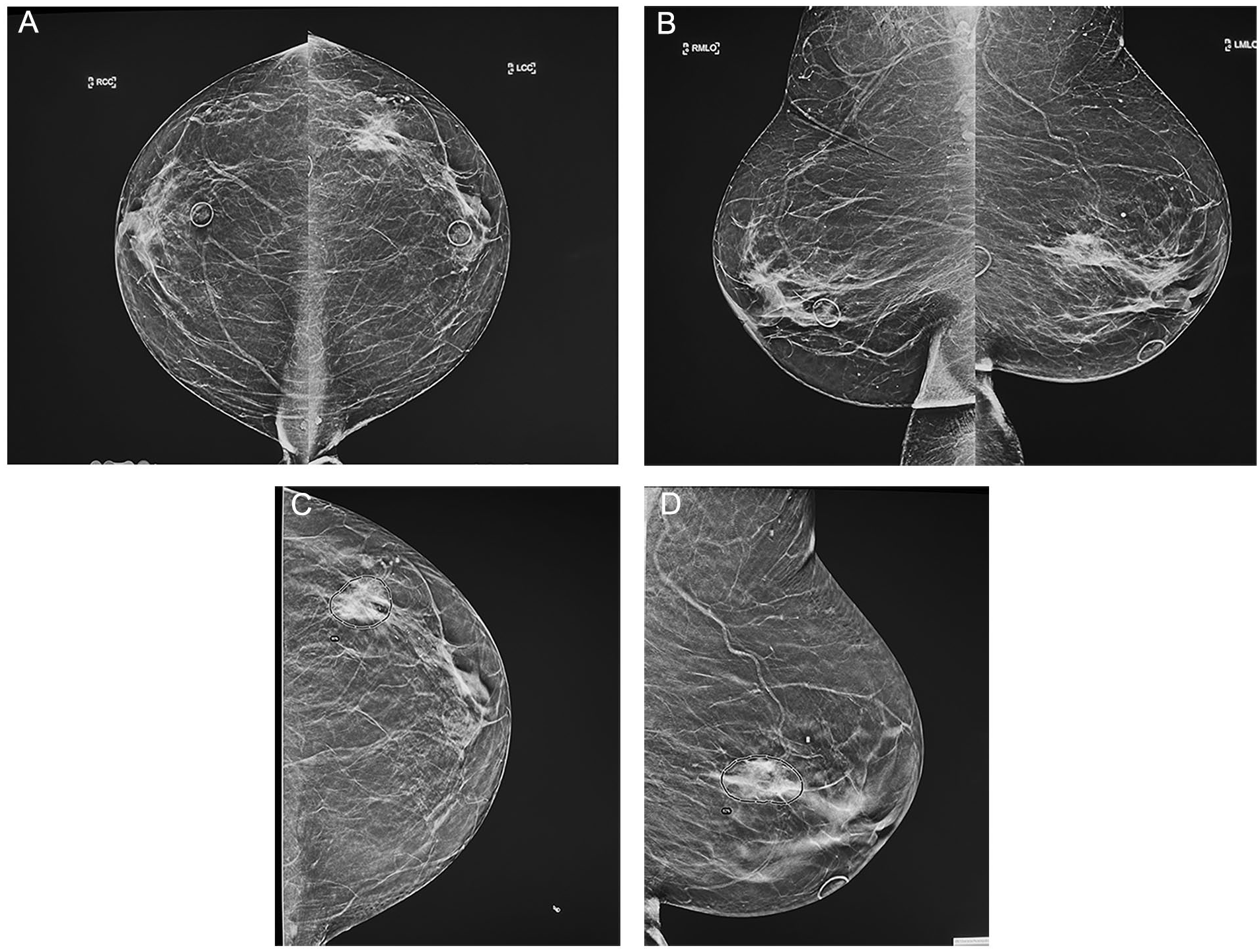
A 77-year-old patient with a clinical area of concern in the left breast, which biopsy-proven malignancy, depicted as a high-priority case by the AI-DBT imaging with up to 90% lesion score. (A, B) Bilateral craniocaudal and mediolateral oblique mammogram views showed underlying clinical concern in the left outer breast, BB-marked, an equal-density irregular and spiculated mass with associated fine and pleomorphic calcifications extending beyond the mass. (C) AI-DBT imaging on the craniocaudal showed a 90% lesion score. (D) AI-DBT imaging on mediolateral oblique view showed an 62% lesion score.
Risk scores may be vendor specific depending on the on the predetermined datasets used to develop the thresholds for malignancy for image analysis. 19 Furthermore, mammography systems and patients’ demographics may also influence risk scores.20,21 Therefore, close liaison with the vendor is needed for the user to understand and optimize performance when using different AI-CAD systems.
Moreover, modern AI-CAD systems assist radiologists by triangulating findings across multiple mammographic views. 22 This capability can significantly streamline the diagnostic process, making it more efficient by reducing the time needed for radiologists to match lesions across different images.
Additionally, to simulate clinical practices where radiologists compare current examinations with prior ones to identify temporally stable or growth findings, a temporal DBT lesion-processing module has been proposed. This module automatically contrasts 2 screening exams to factor in lesion growth or stability during evaluation, showing significant improvement in breast cancer detection performance. In external testing, PriorNet demonstrated superior performance with an AUC of 0.896 (95% CI: 0.885-0.896), outperforming the baseline models, which achieved AUCs of 0.846 (95% CI: 0.846-0.847) and 0.865 (95% CI: 0.865-0.866), both with
Streamlining Workflow With AI-CAD in Radiology
In radiology departments, AI-CAD system outputs are being used as practical filters within radiologists’ worklists to streamline and prioritize case reviews. 24 This integration allows radiologists to sort cases based on the number of findings, allowing them to choose to address simpler cases with no findings before moving on to more complex ones with multiple findings or vice versa.
Additionally, technologists can leverage the AI-CAD scores after images are captured to prompt radiologists for expedited reviews, potentially enabling quicker or even same-day evaluations for patients. This proactive strategy can significantly patient anxiety linked to waiting for recall decisions and enhance adherence to screening program protocols.25,26
Integrating AI With DBT in Clinical Practice
Several methods have been proposed to harness AI-driven CAD to boost DBT effectiveness, as described below and outlined in Figure 5.
One approach is to use AI as a second reader. It can replace one of the radiologists in the double-reading process, which is common in many European countries or integrating AI as an auxiliary tool within the single-view reading framework commonly done in North America, where it would act as a supplementary reviewer.
Alternatively, AI could be considered for use as a standalone diagnostic tool, operating independently from radiologists, a bold step that could transform diagnostic practices by fully utilizing AI’s analytical capabilities.
Additionally, AI systems can serve as a triage tool, acting as gatekeepers by separating cases based on findings. This helps prioritize reader workflow, enabling radiologists to concentrate on cases with a higher probability of malignancy, thus improving diagnostic accuracy. However, unlike when AI is used as a standalone modality, it does not remove the need for radiologists to serve as screening readers.
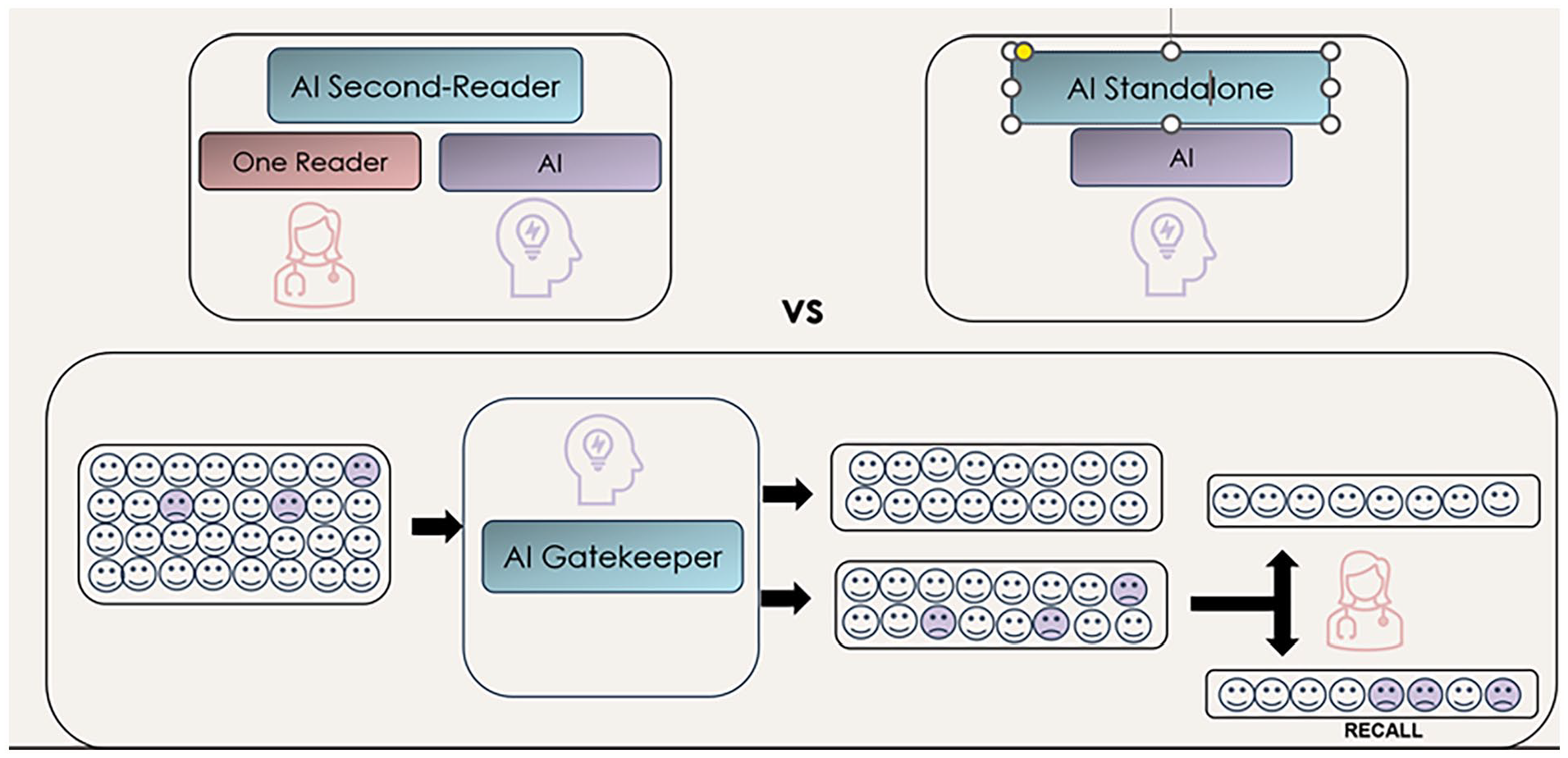
Strategies for integrating AI with DBT in clinical practice.
AI as the Second Reader in DBT
Double reading in breast cancer screening involving 2 radiologists has proven to identify more cancers than single-reader methods. 27 Despite its effectiveness, this approach can increase false-positive recalls and adds significant burdens to screening processes due to the heightened workload. 27 In addition, its feasibility depends on the availability of skilled radiologists which are frequently in short supply.
The evidence summarized in Table 1A and B from retrospective studies,28-32 which compare AI performance against radiologists of varying experience levels, shows that AI-CAD systems used as second readers in DBT either meet or surpass the sensitivity and specificity of traditional double-reading methods. Regarding recall rates, the single study included in this review
29
found that adding AI as a supplementary tool in a single-reading setting was noninferior to radiologists in recall rates (
Evidence Summary of the AI-DBT Imaging as a Second Reader.
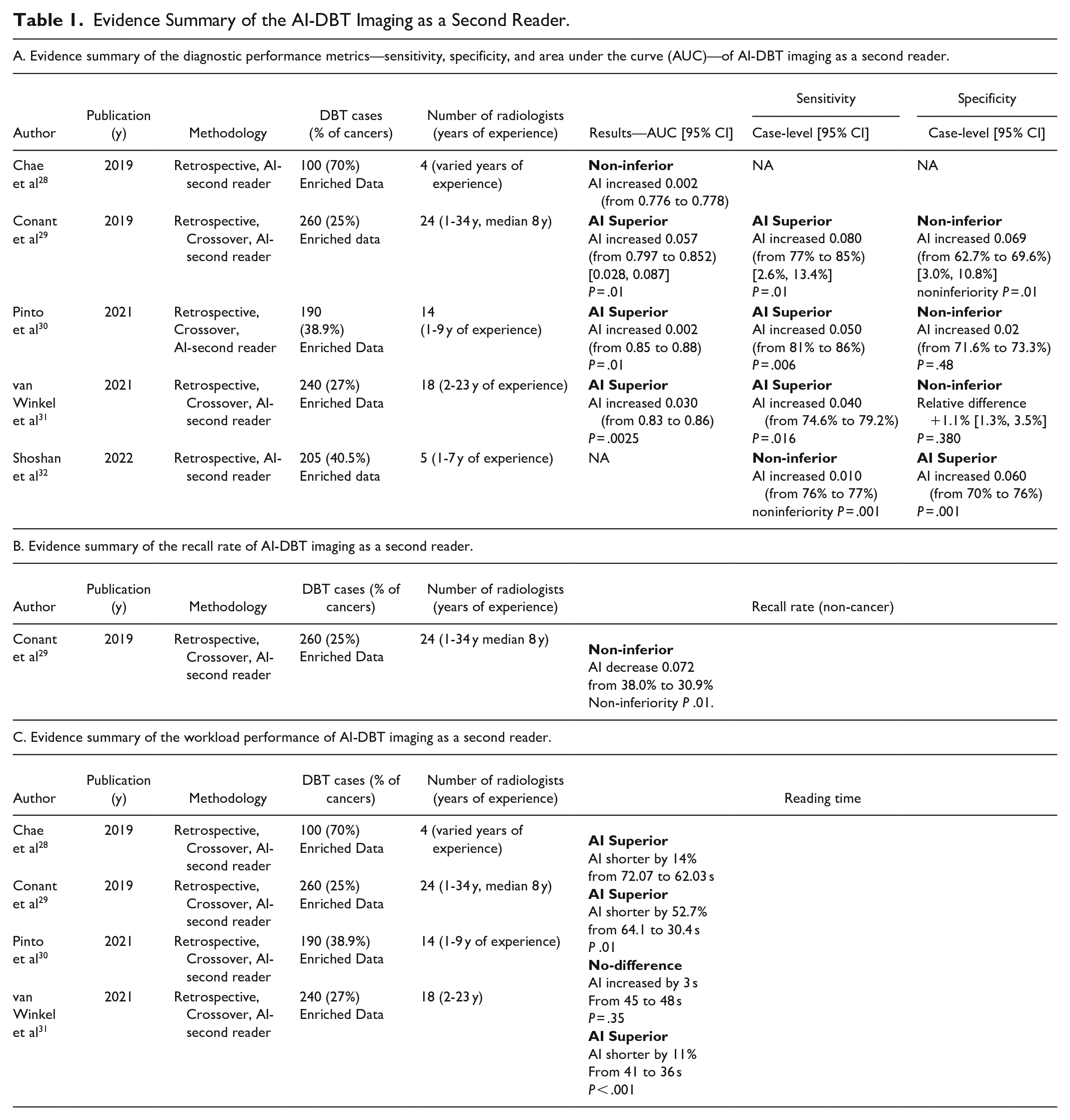
Additionally, study results demonstrate that employing AI as a second reader can significantly improve workflow efficiency. Using AI to instantly highlight the lesions and provide a lesion score, can reduce the time required to review each DBT scan by 11% to 52.7%. It can address a major limitation of DBT, marking a substantial step forward in streamlining breast cancer screening processes (Table 1C).
AI and DBT as Standalone Screening Modalities
Employing AI as a standalone modality in breast cancer screening could offer a cost-effective solution by eliminating completely the need for radiologists to read the screening studies, enabling them to concentrate solely on the subsequent workup with additional images and/or biopsies of findings flagged by the AI. 34 This approach could significantly ease the current shortage of breast imaging specialists. However, standalone AI systems have not yet received FDA approval, as its present approved use is in a supportive role in conjunction with radiologists. 35
Furthermore, adopting AI as an independent reader introduces substantial regulatory challenges, particularly regarding the medicolegal implications of AI errors. 34 In cases of misdiagnosis, determining liability is complicated. Questions arise about whether the responsibility lies with the radiologist, whose name appears on the report, or the AI system itself. Since the development of AI involves multiple contributors, assigning accountability can be problematic.
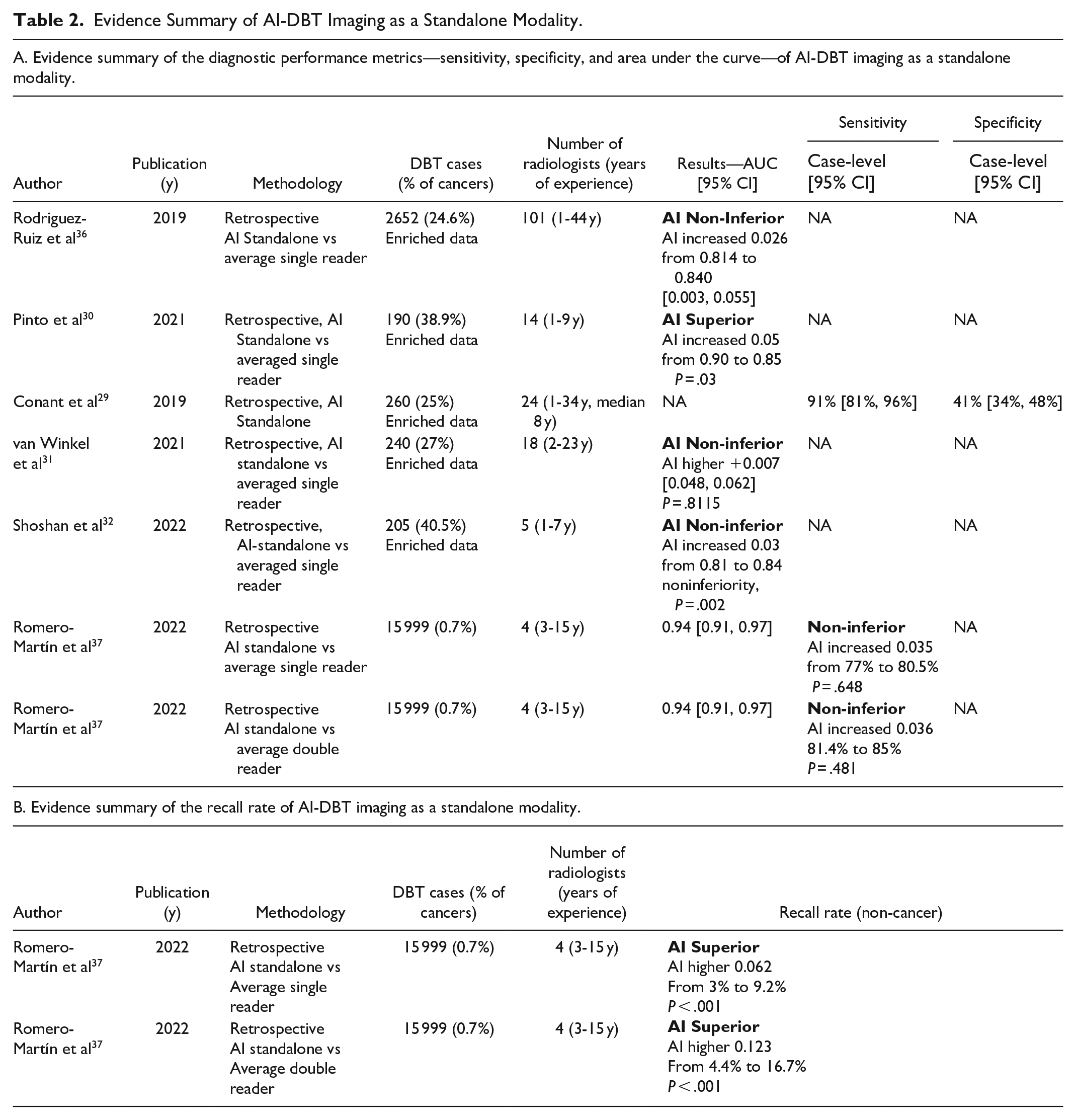
Despite these challenges, evidence summarized in Table 2A and B29-32,36,37 indicates that AI-CAD systems used as standalone modalities in screening are at least as effective, if not superior, to radiologists in terms of performance. These findings align with a recent published polled data from 16 studies, which included 1 108 328 examinations in 497 091 women. 38 Additionally, studies suggest that using AI as a standalone reader in DBT settings at least matches human readers’ capabilities and significantly reduces recall rates. This could potentially lower patient anxiety, which has shown to be a significant deterrent to screening participation and reduce unnecessary follow-up tests and biopsies, thus cutting healthcare costs.25,26
Evidence Summary of AI-DBT Imaging as a Standalone Modality.
AI as a Gatekeeper in DBT Screening
The use of AI-CAD as a gatekeeper in breast cancer screening is primarily based on the low incidence of breast cancer within the screening population, which can contribute to reduced radiologists’ accuracy. 39 An AI system, if finely tuned for higher sensitivity, could effectively filter out a large number of DBT exams that are likely to be normal. This would lessen the screening burden on radiologists, allowing them to focus on cases with a higher likelihood of cancer, thereby improving the radiologist’s diagnostic performance. In this scenario, AI serves as a triage tool that prioritizes cases for reading based on the findings. However, unlike when AI is used as a standalone modality, it does not remove the need for radiologists to serve as screening readers.
Raya-Povedano et al 40 reviewed 15 987 DM and DBT examinations (which included 98 screening-detected and 15 interval cancers) and demonstrated that, regardless of whether double or single reading protocols were used, implementing AI to filter out normal DBT screens could significantly reduce the radiologists’ workload by approximately 70%. This adjustment also led to a lower recall rate in the double reading scenario without compromising the sensitivity of the screenings.
Additionally, comparing DM and DBT across 31 000 screening cases using a double reading approach, 40 using AI as a gatekeeper reduced the reading time by about 30%, improved sensitivity by 25%, and decreased the recall rate by 27%. This evidence underscores the potential of AI to streamline the screening process effectively while maintaining or even enhancing diagnostic accuracy.
Expanded Applications of AI-CAD in DBT
AI-CAD on Synthetic Images
AI technology also uses CAD-based methods to map areas of suspicion identified in DBT slices onto a two-dimensional synthetic digital mammography (SDM) image, marking potential abnormalities. 41 This approach has demonstrated improved sensitivity while maintaining specificity comparable to traditional methods.42,43 Additionally, it has been shown to decrease the workload for radiologists by reducing reading times by between 23.5% and 29.2% without impacting recall rates.42,43
AI Simplifying DBT Slice Review
AI has further been applied to reduce the number of DBT slices that radiologists need to review. This is achieved by generating fewer, thicker slices, approximately 6 mm each, instead of 1 mm each, intended to shorten interpretation time. A study conducted by Sauer et al 44 evaluated this strategy using an enriched dataset comprising 111 DBT scans, including 70 cancer cases and reviewed by 3 radiologists with varying levels of experience. The results confirmed that diagnostic accuracy remained consistent, and the goal of reducing reading time was effectively achieved.
Challenges in Integrating AI and DBT into Clinical Practice
Incorporating AI with DBT into clinical settings presents several challenges that hinder its rapid adoption and impact patients’ trust in physicians and healthcare institutions. These challenges include:
Limited Generalizability of Studies: Images from DBT vary based on the manufacturer, showing differences in angular range, acquisition methods, pixel binning, and the reconstruction techniques used. 45 There is a scarcity of validated multicentric studies that involve medical devices with varying pixel parameters 45 and encompass diverse socio-demographic groups, including different ages and ethnicities. 46 This lack of broad representation affects the generalizability and transferability of results.
Transparency and Traceability Issues: AI systems often lack clear traceability, particularly within the neural network’s hidden layers, known as the “black box” problem.47,48 This limits the research’s reproducibility and hampers clinicians’ and patients’ acceptance of AI algorithms.
Evidence of Clinical Benefit: There is an absence of evidence showing that AI integration leads to improved outcomes, such as reduced breast cancer mortality. 49 Studies often focus only on diagnostic performance metrics, which can introduce lead-time bias with early detection but might not necessarily translate into improved survival rates.
Reimbursement: As AI systems increasingly perform functions traditionally done by physicians, addressing reimbursement issues becomes vital. These systems can significantly alter cost structures and job roles within healthcare, necessitating a comprehensive evaluation of reimbursement strategies. Policymakers and healthcare leaders must engage in extensive screening program-level discussions to adapt effectively. This ensures that reimbursement policies are equitable and align with the broader goals of healthcare systems, facilitating a smooth integration of AI technologies while maintaining the quality of care. 50
Additionally, specific technical challenges arise in the development of AI algorithms for DBT compared to digital mammography (DM) 10 :
Computational Demands: Developing an AI algorithm for DBT requires significantly more computational power.
Complex Annotation Tasks: Annotations for DBT must be made across multiple slices, unlike DM which typically involves 1 or 2 images. This task is further complicated by the generally lower resolution of tomosynthesis sections compared to DM images.
Limited Training Data: There is a scarcity of de-identified public DBT datasets available for AI training, which is much smaller compared to those available for DM. This limitation restricts the development of robust AI algorithms.
While these challenges are significant, ongoing research and studies have begun addressing them. However, a major issue remains with the critical appraisal of available evidence and its integration into clinical practice. This challenge is largely driven by the “laboratory effect,” where outcomes from studies using “enriched data”—datasets disproportionately weighted with malignant cases—may not accurately reflect real screening scenarios. Such data can skew the performance metrics of AI tools and radiologists, often showing higher recall rates in study settings compared to real-world clinical environments.
Future Directions of AI and DBT in Breast Cancer Detection
The future of breast cancer detection is intricately linked to continuous innovations in Digital Breast Tomosynthesis (DBT) and Artificial Intelligence (AI) technologies. For the findings from existing studies to be deemed reliable and applicable on a broader scale, there is a crucial need for validation within actual population-based screening scenarios. This will involve conducting prospective, multicentric research that includes a variety of equipment providers to ensure the universal applicability of the results. Additionally, it is vital to incorporate considerations of ethnoracial diversity to promote equal access to these advanced technologies, facilitating their widespread adoption across varied patient demographics. 51 Moreover, it is imperative to assess the impact of these technological advancements not just on detection metrics but also on patient survival outcomes and their cost-effectiveness. Understanding these aspects will provide a more comprehensive view of their practical value in healthcare settings, helping to shape future strategies for breast cancer screening.
Conclusion
The implementation of advanced models combining DBT and AI in breast cancer screening holds the potential to positively impact several levels of healthcare delivery. For radiologists, this integration could significantly reduce workload and fatigue, which are common in routine clinical tasks. Health systems could see improved workflows and easier adoption of DBT, particularly in regions experiencing shortages of breast radiologists. For women undergoing screening, this technology could minimize unnecessary recalls and reduce anxiety and the need for follow-up procedures and biopsies without compromising diagnostic accuracy. However, bridging the gap between the complex realm of AI algorithms and their practical clinical applications demands further research. This is crucial to ensure that breast cancer screening effectively benefits from these technological advancements.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
