Abstract
Digital breast tomosynthesis (DBT) is gradually being implemented in routine clinical breast imaging practice. The technique of image acquisition reduces the confounding effect of overlapping breast tissue, which substantially affects cancer detection, abnormal recall, and interval cancer rates in a screening/ surveillance setting. In a diagnostic setting, tomosynthesis also allows for improved lesion localization and characterization over conventional imaging, which potentially improves the accuracy and improved workflow efficiency. To optimize the utility of tomosynthesis, imagers should be aware of the pertinent aspects of image acquisition as it relates to interpretation, the appearance of benign and malignant pathologies, and sources of possible misinterpretation. This article aims to provide a practical knowledge base of DBT and demonstrate its potential benefits when incorporated into routine clinical practice.
Introduction
Digital breast tomosynthesis (DBT) is gradually being incorporated in routine clinical practice around the world, with over 9,000 accredited DBT units in the United States as of October 2020. 1 The Canadian Association of Radiologists (CAR) published guidance on breast imaging and intervention in late 2012 including a description of DBT and its applications and limitations. At that time, DBT was in the active stages of testing and early stages of clinical use, and the CAR guidance noted that it was unclear what the role of DBT would be in general population screening, subgroup screening, and diagnosis. 2
The diffusion of technology within Canada has been slow due to the lack of appropriate fee guide, lack of funding and implementation challenges. The country’s first reported installations for clinical use in 2014 were in Toronto’s Princess Margaret Cancer Centre, British Columbia and Women’s Breast Health Centre in Ottawa. Since then, major academic centers and others in Canada have adopted this technology.3,4
While conventional 2D mammography is recognized as the primary modality for breast cancer screening, its limitations about breast density, false positives, and cancer detection have been well established. The ability of DBT to address these limitations has led to its incorporation into many routine clinical practices. Nevertheless, there is hesitancy among some radiologists to adopt DBT due to concerns of prolonged reporting times and difficulty with image interpretation.5,6 DBT is often called a “3D mammogram,” which is a misnomer; DBT is simply a mammogram that looks at the breast through different angles and should be referred to as “tomo,” “tomosynthesis,” or just “DBT” and is not a 3D technique. This article aims to provide a practical knowledge base of DBT and demonstrate its potential benefits when incorporated into routine clinical practice.
Technique
The acquisition of the DBT image involves the rotation of the X-ray tube in an arc or a sweep either in a continuous to step-and-shoot technique to obtain projection images. The sweep angle varies between 15 and 50°, depending on the manufacturer, smaller sweep angles providing better in-plane resolution while larger sweep angles allow better out-of-plane resolution. 7 The obtained projection images are then reconstructed into 0.5-1 mm-thick image sections parallel to the detector using the filtered back projection and iterative technique. The total number of reconstructed images depends on the thickness of the compressed breast and are viewed as a stack of images that can be scrolled (Figures 1 and 2).
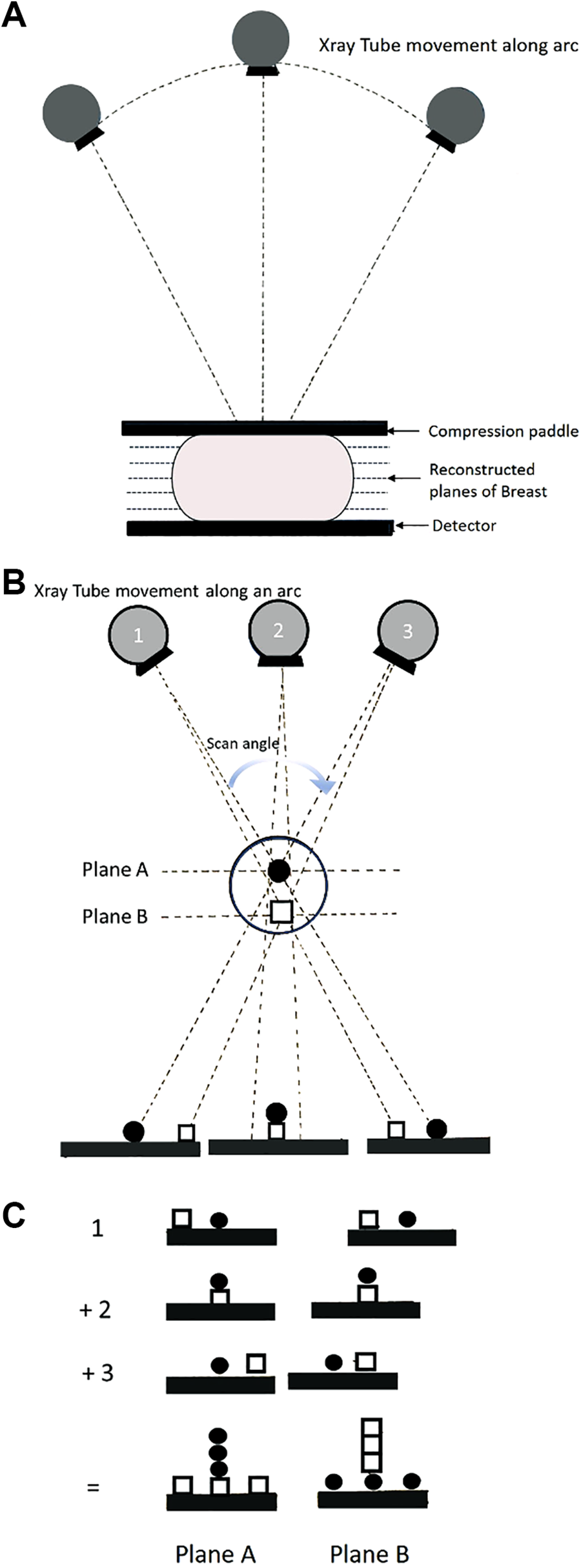
Illustration demonstrating basic principle of digital breast tomosynthesis. A, Multiple projection images are obtained at different angles as the X-ray tube moves along an arc. Projection images are then reconstructed into thin slices. B, Tube moving through different objects in different planes demonstrating the in-plane objects conspicuously while blurring out other planes. C, The “sphere” is in-plane in Plane A is Enhanced while the “square” is blurred. The “square” is in-plane in Plane B is enhanced while the “sphere” is Blurred. (Artist: Dr Tanvi Jakhi MD).
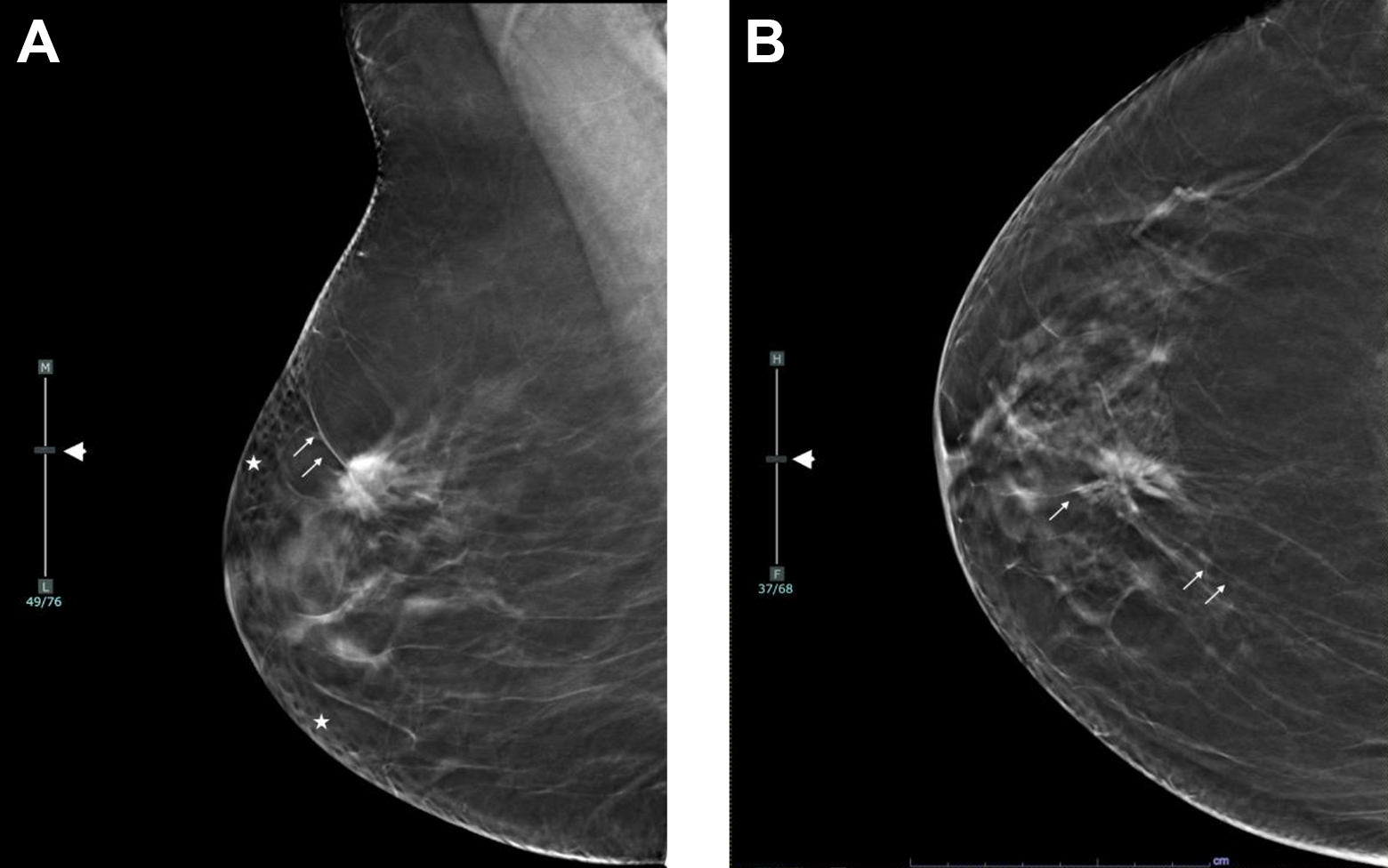
BI-RADS descriptors for lesion location. Right spiculated mass upper medial, middle third quadrant, 11 o clock, 5 cm from nipple. A, DBT right Mediolateral Oblique view slice 49/76 demonstrated on the scroll bar (short arrow). B, DBT right cranio-caudal view slice 37/68 demonstrated on the scroll bar (short arrow) other structures: skin pores seen as lucencies (stars). Spicules (arrows).
The “combo-mode” is when DM and DBT are acquired combined technique providing the ability to compare the information provided by both techniques but results in nearly an increase in the radiation dose by a factor of 2.25 compared DM alone but is still well within the U.S. FDA limit of 3 mGy per view single mammogram. 8
The DBT data set can be reconstructed into 2-dimensional “synthesized mammogram” (SM). SM could avoid additional DM required to complement DBT and reduce almost 50% the radiation dose. Recently, a polled data showed similar performance metrics using DBT with SM images compared with the combo-mode. 9 Therefore, the absorbed dose and compression time can be significantly reduced in DBT+SM procedures compared to the combo mode and have gained widespread acceptance in screening programs across the United States and Europe.
Choi et al 10 studied quantitative analysis of radiation dosage data between different imaging studies showed that image acquisition with DBT+SM could be accomplished with lower absorbed radiation doses and achieve higher image quality than DM alone.
Screening/Surveillance
Cancer Detection Rate
Several studies, including systematic reviews and meta-analysis, have reported that DBT has a higher cancer detection rate (CDR) compared to DM with increased detection of invasive carcinomas compared to ductal carcinoma in situ (DCIS).9,11-18 Alabousi et al 9 reviewed 42 studies involving 2,606,296 patients and showed that CDR was higher in combined DBT+DM (6.36/1,000) and combined DBT+SM (7.40/1,000) compared to DM alone (4.68/1,000). Invasive CDR was also higher in combined DBT+DM (4.53/1,000) and combined DBT+SM (5.68/1,000) compared to DM alone (3.42/1,000). It is noteworthy that the invasive carcinomas detected by DBT appear to be those associated with a more favorable prognosis as such as tubular, papillary, and mucinous carcinomas and luminal A molecular subtypes.14-18 Therefore, long-term follow-up studies are needed to determine if the increased CDR observed with DBT would translate into a change in population mortality.
Recall Rates and Positive Predictive Value (PPV)
DBT has been associated with decreased recall rates compared to DM. This is most evident with the ability of DBT to resolve asymmetries/ focal asymmetries. However, higher recall rates have been observed with masses and architectural distortions, particularly on initial screens, secondary to the unmasking effect with DBT.19-21 In their meta-analysis, Marinovich et al 11 showed a more significant reduction in recall rate with DBT in American studies than European/Scandinavian studies.
Alabousi et al 9 reported a lower recall rate in combined DBT+SM (42.3/1000) while no difference was identified in the recall rate of combined DBT+DM (64.6/1000) or DBT alone (82.4 /1000) compared with DM alone. PPV was higher in both combined DBT+DM (10.0%), and combined DBT+SM (16.0%) compared to DM alone (7%). No difference in PPV was identified between DBT and DM alone. 9 In addition, according to the Giess et al 22 shows that DBT improved PPV3 for biopsy by as much as 20%.
Effect of Breast Density
In a multi-institution study of over 452,320 examinations the CDR and PPV1 for recall was seen to increase in both dense and non-dense breasts with DBT. 23 The benefit was greatest in heterogeneously dense breasts and not statistically significant in extremely dense breasts. This suggests that DBT may not detect some breast cancers in extremely dense breasts, particularly if not associated with architectural distortion or microcalcifications (Figure 3).
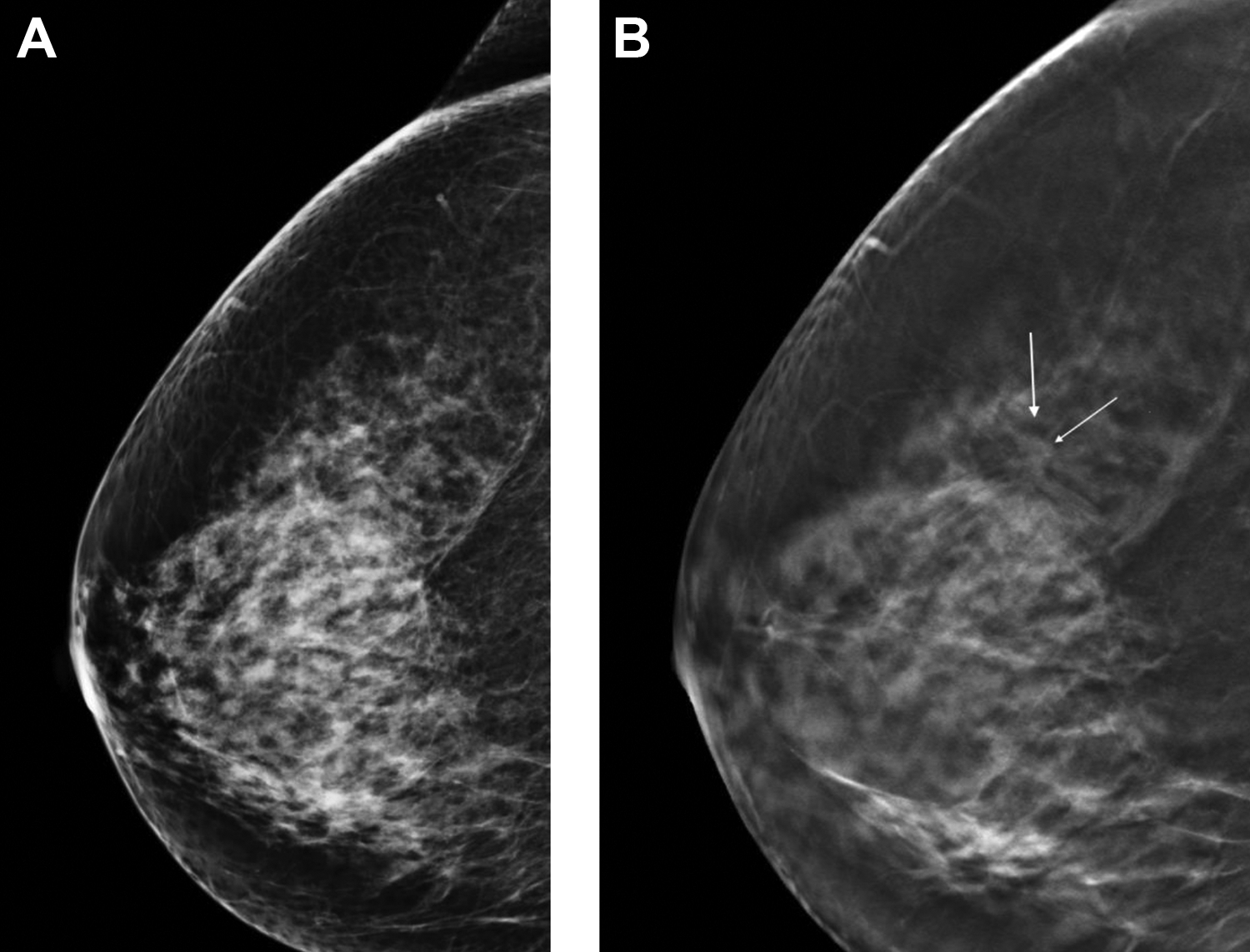
Benefit of DBT in dense breast tissue. A, Right 2D cranio-caudal view showing heterogeneously dense tissue (BI-RADS C), with no evidence of a mass. B, Corresponding DBT cranio-caudal view shows a spiculated mass (arrows) in the upper outer quadrant of the breast. Final pathology: invasive ductal carcinoma.
Interval Cancer Detection Rate
An interval cancer is one that arises after a screen that is interpreted as normal before the subsequent screen. These cancers may present symptomatically or may be asymptomatic, detected by another modality (e.g., US or MRI). They are associated with increased mortality compared to screen-detected cancers with less favorable tumor characteristics. 24
A prospective, population-based screening trial (Malmo Breast Tomosynthesis Screening Trial) compared with a large contemporary control group screened with digital mammography showed a lower interval cancer detection rate. 25 These results are concordant with other previous published studies.26,27 The interval cancers’ characteristics (i.e., the proportion of invasive, size, grade, and nodal status) with DBT screening were similar to those identified in previous DM-only rounds. 25 More robust data would be required to evaluate the benefit of DBT in reducing the interval cancer rates.
Sustainability of the Benefits of DBT in Screening Setting
Most significant performance improvements with DBT compared with DM have been observed with initial mammographic screens. On subsequent screening rounds, women aged 40 to 79 years with heterogeneously dense breasts and women aged 50 to 79 years with scattered fibroglandular density were seen to benefit most from DBT, with both lower recall rates and higher cancer detection rates. 28
More recently, Conant et al 29 compared the DBT outcomes over 5 years and multiple rounds to the outcomes of DM. They demonstrated that favorable outcomes of DBT were sustained over multiple years. DBT also associated with the detection of a higher proportion of cancers with poor prognosis than digital mammography, contrary to the belief that DBT only cancers are low grade and well-differentiated.
The Tomosynthesis Mammographic Imaging Screening Trial (TMIST) is the first randomized controlled trial that will provide many answers pertaining to the performance of digital breast tomosynthesis and the subgroups of women that it will be most beneficial. 30 TMIST will enroll a proposed 165,000 healthy women ages 45 to 74 throughout multiple centers across North America and other international sites. TMIST is designed to evaluate if DBT reduces the rate of advanced breast cancers, defined as metastases, positive nodes and/or invasive tumor greater than or equal to 2 cm and/or estrogen receptor-negative and progesterone-receptor negative and/or HER2-positive tissue equal to 1 cm or greater. This will address the question of possible overdiagnosis/overtreatment and may help researchers tailor future screening to a woman’s individual risk.
Multimodality Screening
The prospective Adjunct Screening with Tomosynthesis or Ultrasound Trial (ASTOUND) evaluated the incremental cancer yield with DBT versus US. 31 It showed that DBT detected more than 1-half of the cancers that were occult on DM but were detected on US in women with dense breast tissue with fewer incremental false-positive results. The added cancer detection with the US was accompanied by an increased false positive rate and lower PPV on biopsy than mammography. Supplemental contrast-enhanced MRI (CE-MRI) showed incremental cancer detection rate and reduced interval cancer rates when performed after DM and DBT. 31
There has been increased interest in supplementary screening with MRI in women with extremely dense breast tissue. In a multicenter study (ECOG-ACRIN) evaluating women with dense breasts who underwent both DBT and abbreviated MRI in 2 rounds of annual screening, 32 MRI demonstrated at higher invasive CDR than DBT (11.8 vs 4.8 invasive cancers per 1000 women, respectively), with no interval cancers observed on follow-up. While abbreviated MRI showed higher sensitivity than DBT (95.7% vs. 39.1%, respectively), DBT retained higher specificity (97.4% vs. 86.7%, respectively).
Surveillance of Breast Cancer Survivors With DBT
Surgery and radiation treatment may lead to scarring and tissue retraction, thereby reducing mammography sensitivity, which can obscure or mimic cancers at DM. Bahl et al 33 showed that the addition of DBT to the surveillance of survivors led to a reduction in the abnormal interpretation rate and higher specificity with no significant differences in the cancer detection rate between the 2 groups. The benefit of adding DBT to SM or DM showed a significant reduction in recall rates compared with DM regardless of mammographic density in this population 34 (Figure 4).
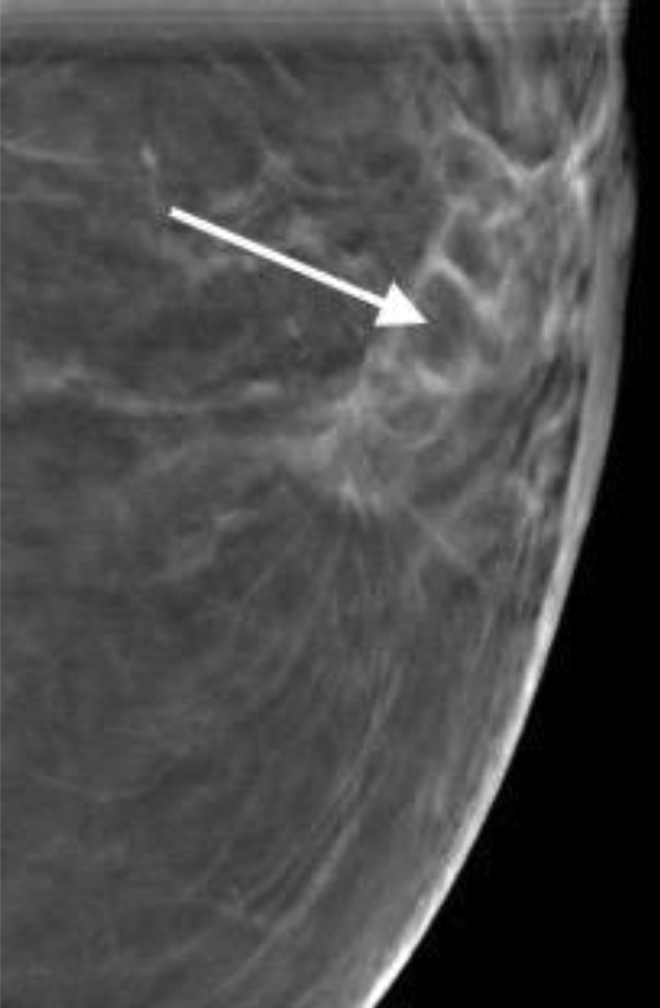
Left CC DBT showing a post-operative scar with central fat (arrows).
Diagnostic Mammography
DBT has been shown to improve diagnostic accuracy for noncalcified lesions 35 with a reduction in the need for additional mammographic views, recommendations for ultrasound,36,37 and BI-RADS 3 classification. 38 Lesion resolution is substantially affected by the angle of the arc through which the x-ray tube moves. Greater arc angles produce thinner slices, which produce less tissue overlap, improving in-plane focus and increasing lesion conspicuity when reconstructed.
Two View DBT
Cancers may be seen only on 1 view or have increased conspicuity on 1 view, such as invasive lobular carcinomas highlighting that 2-view DBT is better than a single view. Rafferty et al 39 reported that 15% of the lesions (both benign and malignant) were seen better on the craniocaudal view.
Triangulation
Accurate assessment of abnormal mammograms is dependent on correct triangulation. Errors in triangulation lead to misdiagnoses and potentially missed cancers, as the assessment is focused on the incorrect region of the breast. Assessments are particularly challenging when dealing with abnormalities visualized only on a single projection, often requiring additional images as 90-degree views and tangential views. With the use of DBT, these other traditional additional views may not be necessary in many cases, permitting an expedited assessment.
The first step while reviewing the DBT images is to identify the plane and position of the nipple to avoid confusion with triangulation. Once the lesion is localized using scroll/localizer bar on the image stack, knowing the position of the nipple can allow the imager to determine if the lesion is superior or inferior on the CC projection, and medial or lateral on the MLO projection.
The imager should be aware that if the nipple axis is eccentric or not in the center of the breast, the nipple will not localize to the center of the stack. Furthermore, the nipple may be rolled superior /inferior on the CC projection and medial /lateral on the MLO projection. This can be further confounded by adding extra images on the side of the breast with the compression paddle during post-processing in some reconstruction algorithms (Figure 2).
Spot compression views (SCV)- Studies demonstrate that replacing supplemental views by DBT improved the diagnostic accuracy of noncalcified lesions. 35 DBT has been found at least equivalent to additional imaging views, especially for the mammographically detected asymmetric densities.40,41 Use of DBT leads to decreased need of additional views for noncalcified findings and can also reduce the use of ultrasound and biopsy 42 (Figure 5).
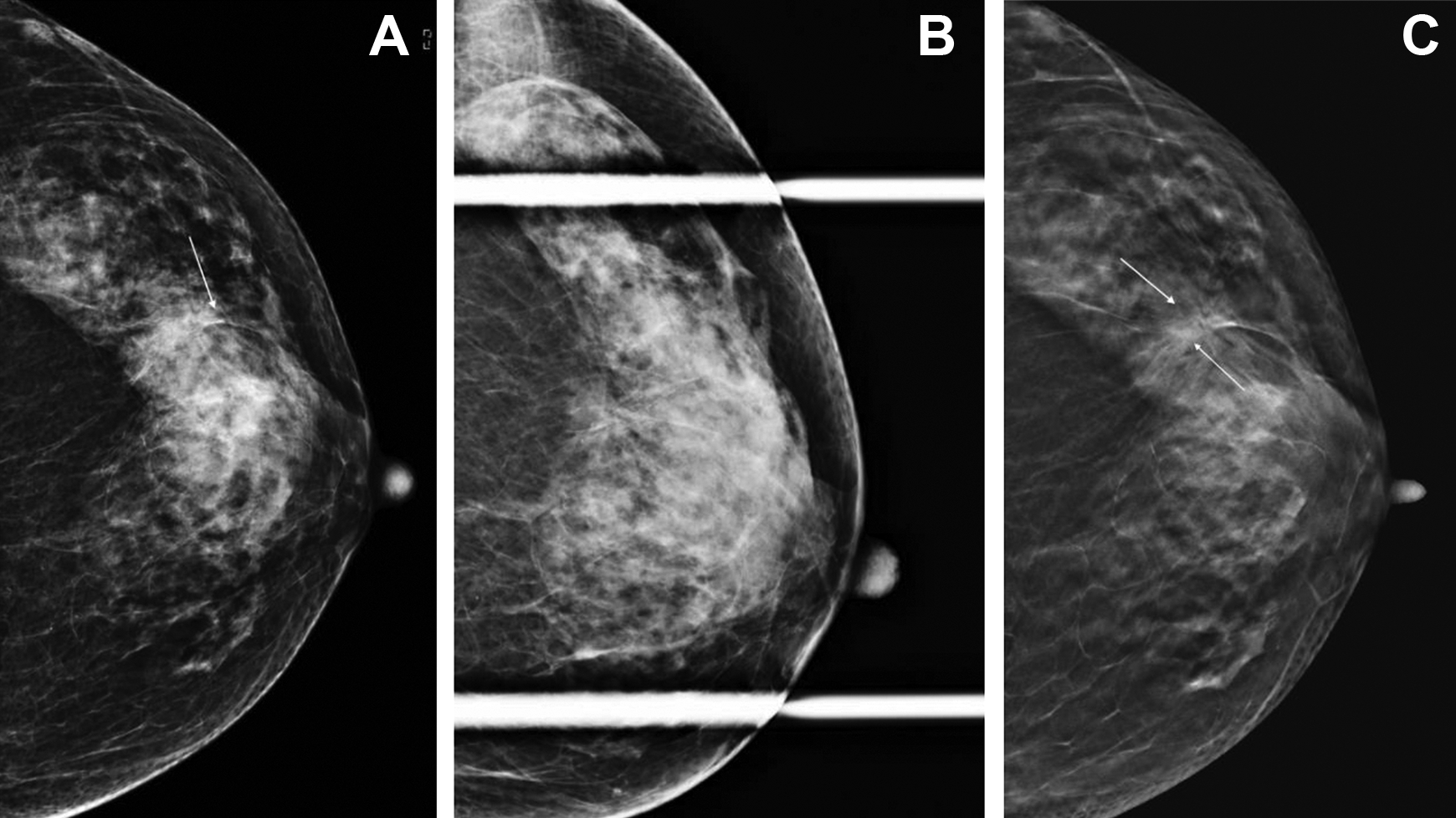
Role of spot compression views. A, Left 2D cranio-caudal view showing subtle architectural distortion (arrows) in the lateral breast. B, Left 2D spot compression view shows resolution of the distortion. C, Corresponding left DBT cranio-caudal view shows a spiculated mass (arrow) in the upper outer quadrant. Final pathology: invasive ductal carcinoma.
Spot Compression DBT Views
DBT can be used with spot compression views to assess a focal area and is based on the reader’s discretion. However, when used in this manner, the potential benefits of offering the DBT to the entire breast will be lost, particularly in determining the extent of the disease. If spot compression DBT is performed in addition to global DBT, there is a substantial increase in the radiation dose. 43
Report Organization and Lexicon
DBT reporting should follow the DM reporting as per the 5th edition of the BI-RADS Atlas 44 along with the Digital Breast Tomosynthesis Guidance supplement to the ACR BI-RADS released in 2020. 45
Findings identified on DBT slices should be described with standard nomenclature and indicate the general location in the breast with the addition of the slice numbers where the abnormality is in focus (Figure 2).
Appearance of Noncalcified Lesions on DBT
Masses
DBT enables detecting both benign and malignant masses by increasing lesion conspicuity with its superior in-plane assessment allowing better characterization of shape, margins, and fat content. Masses identified on DBT should be managed as with DM. All well-circumscribed masses are not benign, and hence appropriate diagnostic workup should be performed with ultrasound and further as necessary.
Breast cancers tend to be less dense on DBT than on DM due to the nature of the technique. Fat within encapsulated masses usually indicates benign cause. However, fat is often visualized in malignant masses on DBT, which could lead to a false-negative misinterpretation. Breast cancers engulf surrounding fat as they grow, and this is better appreciated in DBT. Therefore, low density or fat within a mass should not deter biopsy if the mass shows other suspicious features.
DBT can be an important staging tool given that there are controversies in the use of magnetic resonance imaging in the staging of newly diagnosed breast cancer.46,47 Cancer measurements with DBT correlate to pathology more accurately than DM 48 (Figure 6). In women with breast cancer, DBT allows better margin assessment, extent of cancer-associated distortions, multifocal or multicentric disease, and contralateral disease compared to DM. 49 It is noteworthy that while measuring the cancers, only the core nucleus should be measured, and the long spicules should not be included in the measurements.
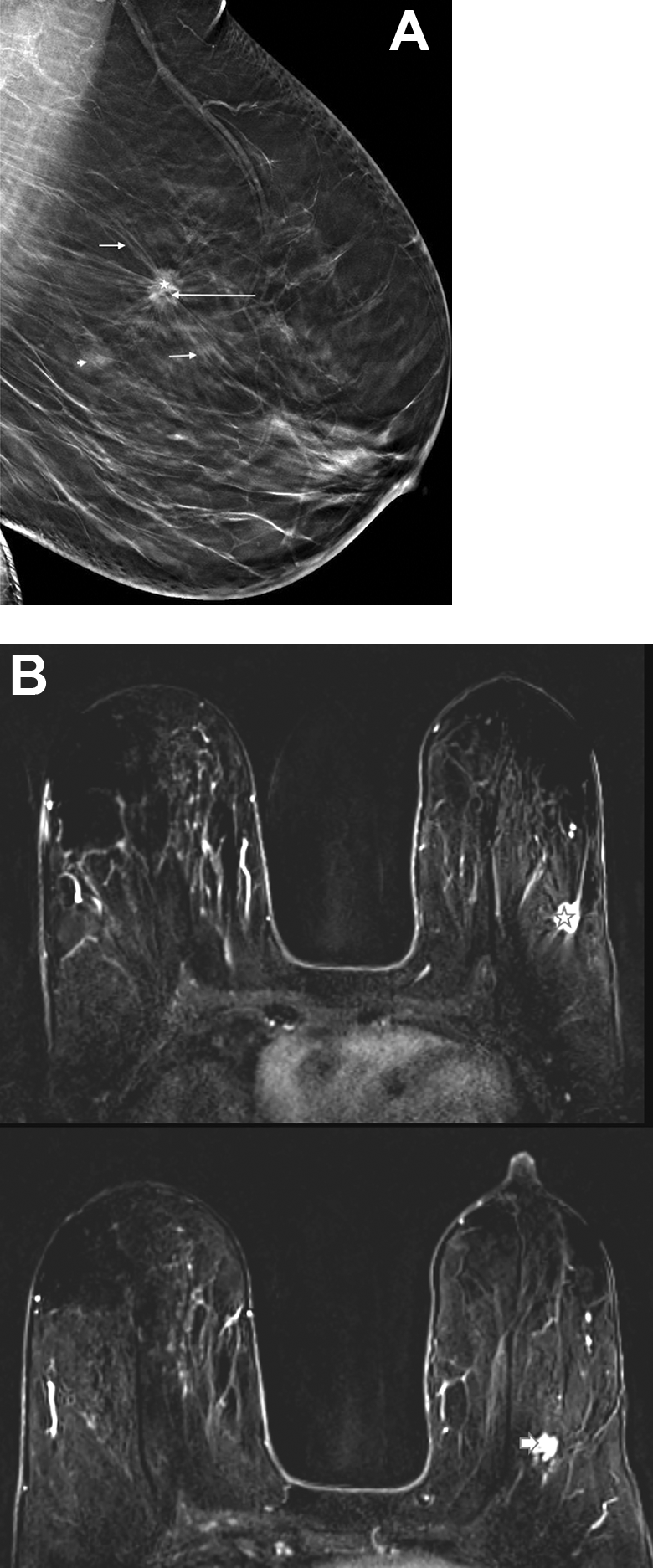
A, Left MLO DBT showing the classic appearance of an invasive mammary carcinoma with long slender spicules (short arrows), irregular margins, and intralesional fat (long arrow). The measurement of the mass should include only the central mass (star) and not the long spicules. Additional out of plane mass (arrowhead) seen postero-inferior to the index mass. B, DCE-MR showing the spiculated mass in the upper outer quadrant (star) and the smaller enhancing mass postero-inferior (short arrow) to the index mass. Final pathology: multifocal invasive ductal carcinoma.
Architectural Distortion
Architectural distortion is better visualized with DBT than with DM and, although there is decreased overall recall rate with DBT, the recall for architectural distortion is significantly increased with DBT. 18 This leads to increased detection of benign and malignant lesions.
DBT-detected distortion has been associated with a relatively high probability of malignancy, with a PPV of up to 50%. 16 Architectural distortion is a common presentation of noncalcified cancers on DBT and was shown to be the primary findings in 12% of the subtle cancers detected on DBT. 50 Among the cancers that manifested as architectural distortion at DBT, 50% were occult on DM, and 20% were classified as single view asymmetry or focal asymmetry on DM. 50 DBT detected architectural distortion should undergo tissue sampling with DBT guided biopsy if not accounted for by prior history of surgery or trauma. Readers should look for converging lines, especially at the fat-glandular interface looking for subtle cancer that may otherwise be hidden in the underlying dense tissue especially at the apex of the breast where summation artifact is commonly seen 43 (Figure 7).
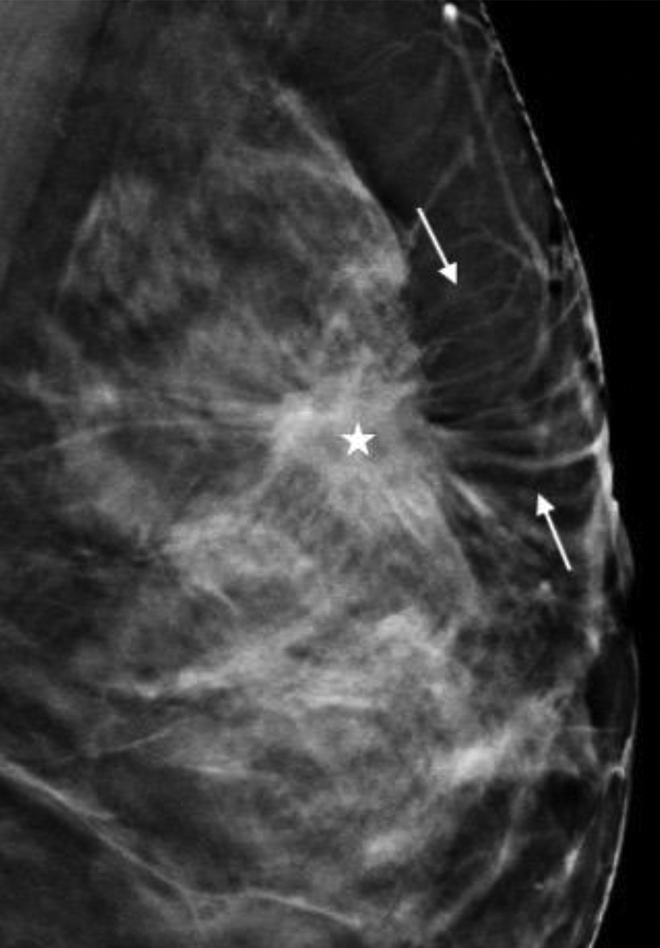
DBT image showing mass (star) at the fat-glandular interface with radiating spicules (between arrows) extending toward the skin surface giving a “rising sun” appearance.
Up to 87% of invasive lobular cancers tend to present with architectural distortion and, therefore, are better detected by DBT.12,51,52 The DBT extent of these cancers, which tend to be multifocal, multicentric, and bilateral, also closely correlates with final histology. 51
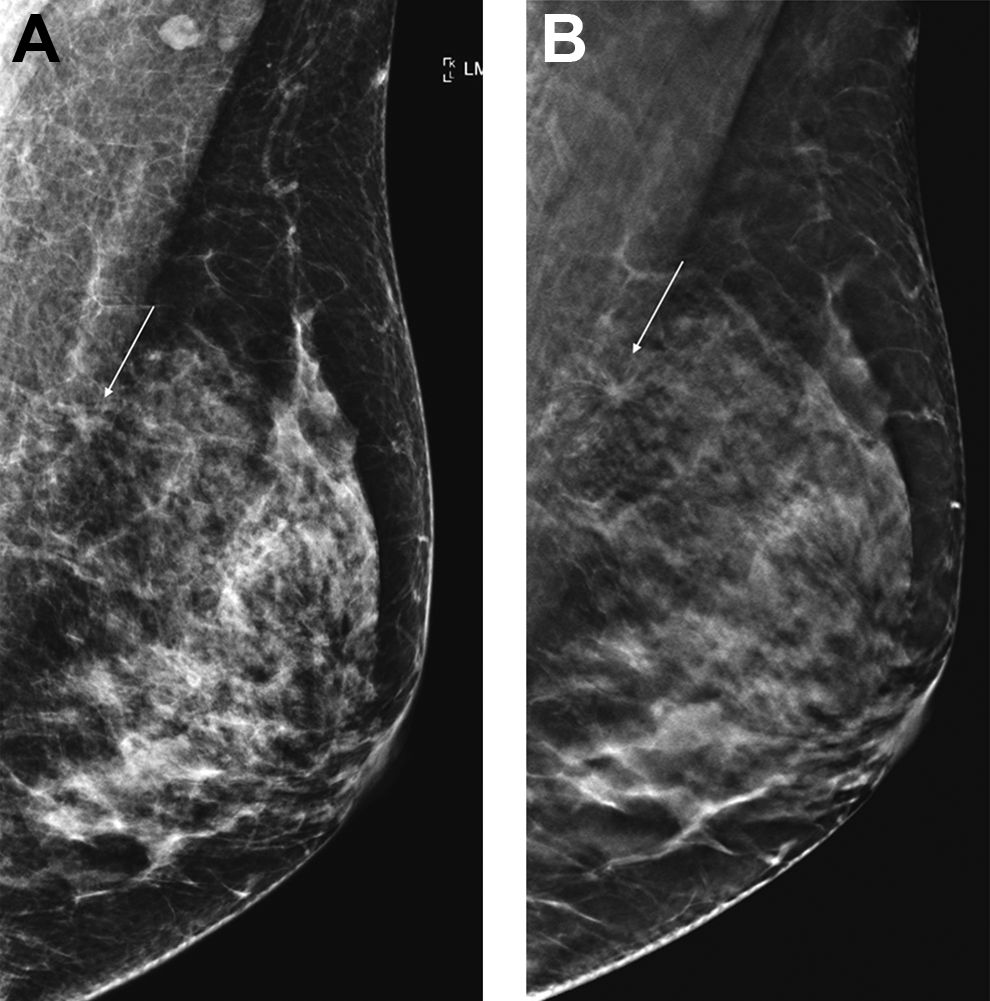
DBT also leads to increased detection and biopsy of nonmalignant architectural distortions, most commonly radial scars, which account for at least 33.2% of architectural distortions detected with DBT 53 (Figure 8).
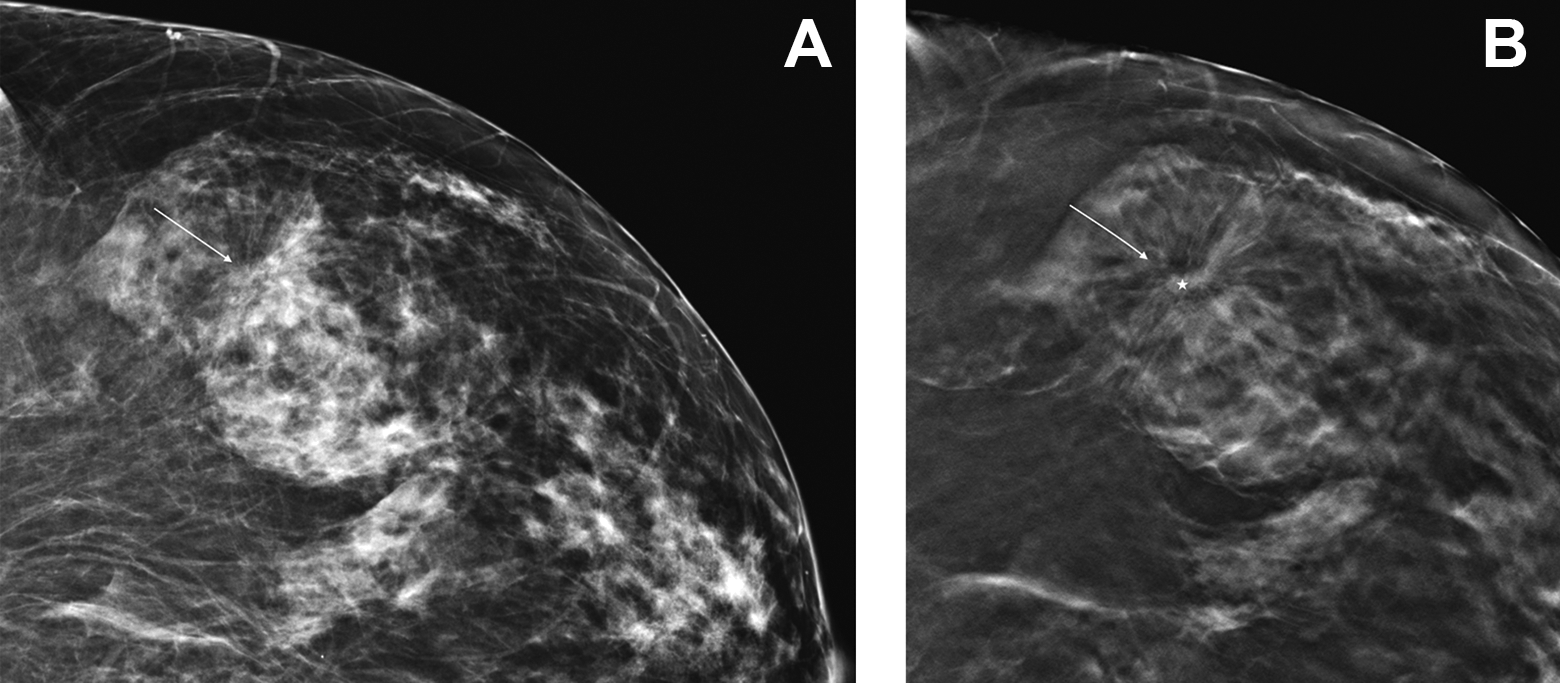
Detection of subtle architectural distortion. A, 2D cranio-caudal view of the lateral left breast shows subtle architectural distortion. B, On the corresponding DBT in the same projection and location, the architectural distortion is now easier to appreciate with fine spicules (arrow) and a central nidus (star). Final pathology: radial scar.
Asymmetries
DBT is of great value in the assessment of asymmetries. It helps resolve asymmetries produced by over-lapping tissue, decreasing unnecessary workup, intervention, and BI-RADS 3 recommendations. A retrospective analysis comparing DBT and DM showed that the screening recall rate was significantly lower with DBT than with DM for asymmetries (13.3% vs. 32.2%, respectively) and focal asymmetries (18.2% vs. 32.2%), which would have otherwise attributed to the false-positive recall rate. 16 Thus, DBT allows increased specificity and reduced recall rates for mammographically detected asymmetries. Breast cancers that would otherwise be classified as asymmetry are likely to be reclassified as masses on DBT, thereby increasing the specificity (Figures 9 and 10).
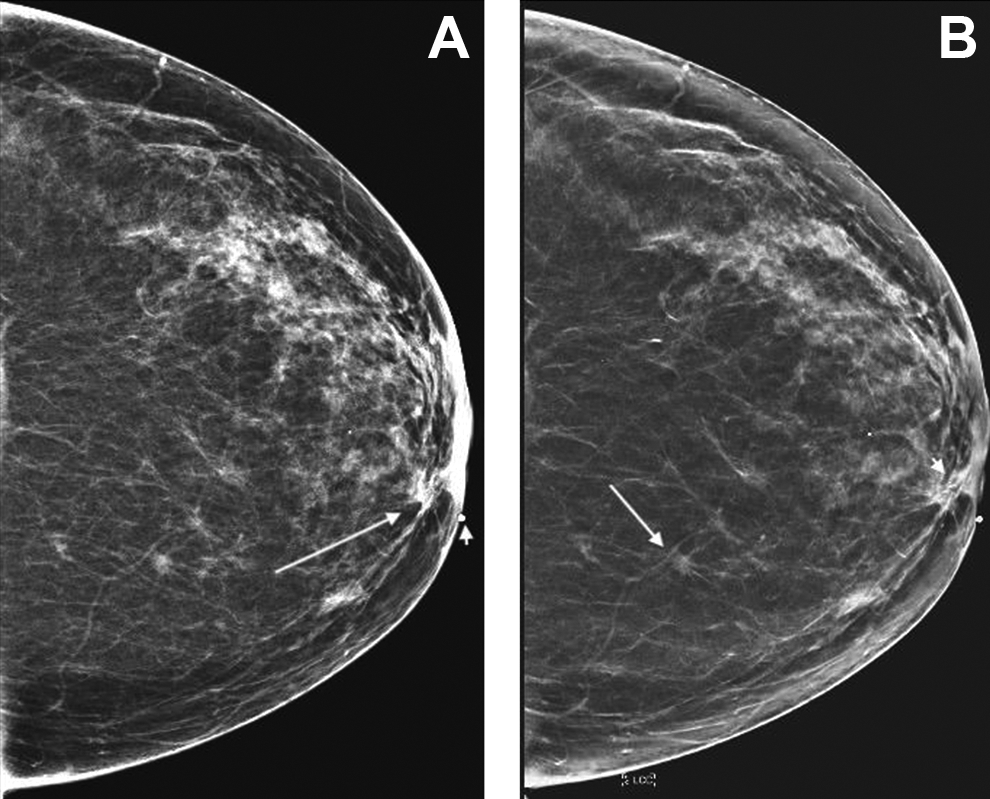
Benefit of synthesized mammogram. A, 2D Craniocaudal view of the left breast shows a medially located asymmetry (arrow) adjacent to the nipple. The asymmetry is palpable and marked by a radio opaque marker (short arrow). B, Synthesized 2D mammogram shows a small spiculated mass (short arrow) adjacent to the nipple corresponding to the palpable finding and the asymmetry seen on the 2D image. An additional fine spiculated mass (long arrow) in the posterior third breast is identified which was not well appreciated on the 2D image. Final pathology: multifocal invasive ductal carcinoma.
Upgrade of BI-RADS descriptors on DBT. A, 2D left mediolateral oblique view shows an asymmetry (arrow) overlapping the edge of the pectoralis muscle. This would be classified as BI-RADS 0, with further workup Indicated. B, DBT image shows that the asymmetry seen on 2d Corresponds to a Spiculated mass (arrow). This can now be classified as BI-RADS 5. Final pathology: well- differentiated invasive ductal carcinoma with tubular features.
Appearance of Calcified Lesions on DBT
The back-projection algorithm that is used in the reconstruction of SM enhances the appearance of calcifications. A retrospective reader study of 198 patients 54 suggests that DBT plus SM may be sufficient in diagnosing calcifications without DM, but, so far, due to lack of sufficient evidence, additional dedicated higher spatial resolution magnifications views should be performed to avoid missing or misdiagnosing calcifications.
The shift of voxels during tube motions accompanied by the degree of optical density of the calcifications may cause blurring of the calcifications. Hence, when DBT plus SM is performed without DM, it is essential to assess images for motion which may obscure the calcifications in the projection that the motion occurred. 55 Detecting motion on DBT can be difficult, and 1 has to carefully scroll the image stack to look for signs of motion such as the “slinky” or zipper out of plane artifact.55,56 Reviewing the projection images can also allow the detection of motion.
“Declustering” effect may make perception of especially small groups of calcifications difficult. Slicing a group of calcifications may show only a few calcifications in each plane. This is contrary to the conventional assessment, which views the group in its entirety. This can be overcome by slabbing the slices to form thicker planes, thereby including more calcifications in each plane (Figure 11).
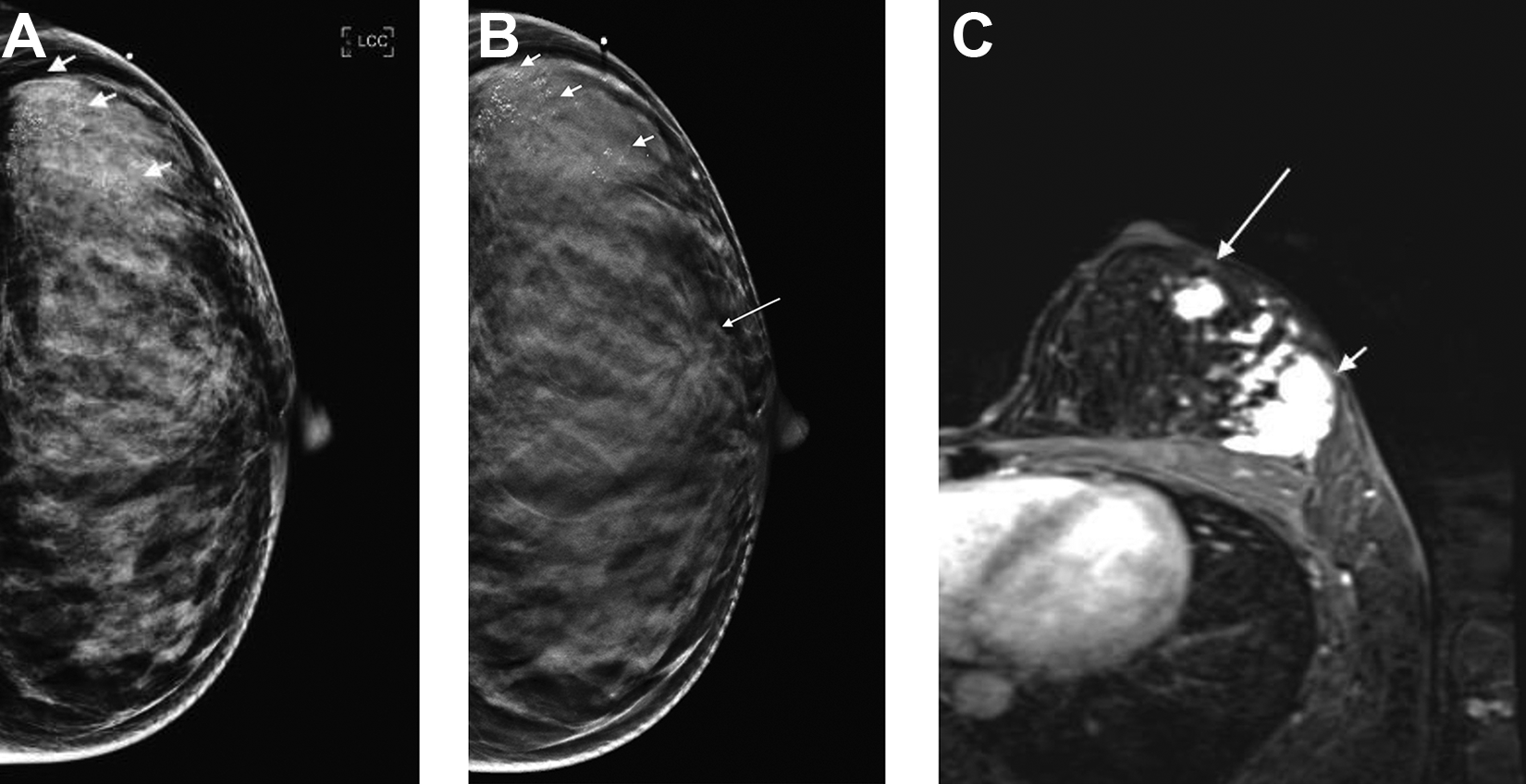
Calcifications. A, Left 2D craniocaudal views shows numerous groups of granular microcalcifications (short arrows), in a palpable region, on the background of dense breast tissue. B, Corresponding DBT shows that the microcalcifications (short arrows) appear sharper and brighter compared to the 2D image. An additional spiculated mass (long arrow) is now seen in the lateral periareolar region. C, Axial CE-MRI of the left breast shows an enhancing mass (short arrow) corresponding to the area of the microcalcifications and an additional mass anteriorly (long arrow) corresponding the spiculated mass seen on DBT. Final pathology: invasive ductal carcinoma with high grade ductal carcinoma in situ.
Pseudocalcifications may also be seen on SM due to algorithm attenuating speckle or quantum noise as well as structural noise from overlapping structures, e.g. Cooper’s ligaments. Careful scrolling confirms the absence of these pseudocalcifications on the stack or projection images. Binning of pixels, patient motion or declustering on the other hand can lead to disappearing subtle calcifications on SM. Both these scenarios highlight the importance of reporting SM in accompaniment of the DBT stack images and never as stand-alone images. 57
Benign Calcifications
DBT allows easier localization of skin calcifications, avoiding the need for complex tangential views, etc. It should be noted that the skin calcifications may be seen deeper than expected and not on the first or last image of the stack due to the breast curvature and, more importantly, due to the post-processing algorithms, which add extra slices on the side of the compression paddle especially when calcifications are closer to the compression paddle.
Vascular calcifications appear as parallel lines associated with curved tubular structures, which are much easier to identify with DBT due to decreased overlap.
Rim calcifications associated with oil cysts or fat density are better seen with DBT as well as soft tissue masses associated with dense popcorn calcifications. When unsure, magnification views should be performed, especially in cases with different varieties of suspected adenosis.
Malignant Calcifications
Morphological description and distribution are in accordance with the standard BI-RADS lexicon. The multifocality and distribution are better depicted by DBT, allowing a better assessment of the extent of disease. 58 More importantly, underlying associated masses may be seen with DBT, especially in dense breast tissue, which may upgrade the BI-RADS descriptors and category (Figure 11).
Other Considerations
DBT-Guided Biopsy
As more and more women access DBT in the future, DBT only findings will increase, which would require DBT guided biopsy.
Advantages of DBT guided biopsy59-61:
Z-axis or depth information can be obtained without the need for stereotactic imaging pairs.
DBT allows the full detector field for imaging during DBT biopsy as opposed to a small window with conventional DM stereotactic systems.
DBT-guided procedures have also been shown to have a decreased total biopsy time by more than half compared with conventional DM stereotactic biopsies.
The need for fewer exposures with DBT guidance leads to an overall decrease in radiation dose compared to DM stereotactic biopsies despite the additional glandular dose associated with each individual DBT acquisition during DBT-guided biopsies.
Training
The Food and Drug Administration requires 8 hours of additional training for Mammography Quality Standards Act (MQSA)-qualified radiologists to interpret DBT images in clinical practice. 1 CAR DBT guidelines also recommend that radiologists get an initial 8 hours of specific tomosynthesis training similar to the ACR requirement. Technologists and medical physicists should also obtain appropriate training. 62 The effect of the performance is based on the readers’ knowledge, formal training, and familiarity with the new technique. Lack of appropriate training can often lead to an inappropriate interpretation of findings, undermining the technique’s benefits, which can lead to resistance in adopting the technique. This can lead to a steep learning curve effect.
The screening recall rate was most improved among non—breast imaging specialist radiologists and in women with nondense breasts, 63 which may indicate that DBT would be a good tool for the relatively less experience breast radiologist or non-breast imaging radiologist and would have relatively less impact on highly experienced breast radiologist.
Workflow
Reading time/interpretation time is expected to decrease with the increased radiologist’s experience. 64 In addition, advances in technology with machine-learning computer-aided detection algorithms highlighting lesions on the DBT stack are promising in improving faster review with increased radiologist performance.50,65,66
Cost Effectiveness
Whether DBT could be a cost-effective alternative replacement for DM in a population screening program is a matter of ongoing investigations and depends on the cost to the program. 67 Several studies have shown DBT to be cost-effective, particularly in dense breasts.68,69 Cost of equipment, availability, storage of data, and ability of biopsy of DBT-only findings are also a consideration in cost-effectiveness to a breast imaging department.
Artifacts
Some unique artifacts are associated with DBT, which may lead to the degradation of images that may obscure important details on a DBT image. Most of the artifacts are due to the technique and the processing. 57
Conclusion
There is current evidence-base suggesting that DBT is a potentially useful screening allowing higher PPVs, higher CDRs, reduced recall rates, and lower interval cancer rates. (Table 1) However, it is still unclear whether the increased cancer detection rate in the screening setting with DBT is related to overdiagnosis or will have a long-term impact on a patient’s survival. Before guidelines have strong evidence to support the use of DBT on a population basis, longer-term DBT outcome data is needed.
Benefits of Digital Breast Tomosynthesis.

In the diagnostic setting, DBT has shown improved ability to accurately characterize and localize lesions, with a marked benefit on workflow and efficiency.
Its limitation in the performance in extremely dense breast tissue indicates the need for supplementary screening despite DBT.( Table 2) DBT remains a morphologic-based imaging modality, and supplementary screening CE-MRI has shown better performance and promise, although it is not abundantly available. Contrast-enhanced spectral mammography and contrast-enhanced DBT may be a plausible alternative to provide functional information similar to CE-MRI 70 and needs to be explored in larger population-based trials, especially in the subgroup of dense breast tissue. DBT is ultimately a compression technique, and poor compression may preclude visualization of a cancer, especially in the background of dense breast tissue just as with DM. Cancers can look different on DBT and they may not be perceptible or discerned as abnormalities on the images to the untrained eye. Attention to radiologist training will continue to improve the accuracy and performance of screening and diagnostic mammography with DBT. It must be a serious consideration to any practice considering the implementation of DBT.
Limitations of Digital Breast Tomosynthesis.

Footnotes
Acknowledgments
Medical illustration by Tanvi Jakhi MD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
