Abstract
This is a visual representation of the abstract.
Introduction
Peripartum cardiomyopathy (PPCM) is a rare cardiomyopathy that affects women in late pregnancy or in the post-partum period. 1 The Heart Failure Association of the European Society of Cardiology defined PPCM in 2010 as an idiopathic cardiomyopathy presenting with heart failure secondary to left ventricular systolic dysfunction toward the end of pregnancy or months following delivery, where no other cause of heart failure is found. 2 No specific test exists for PPCM, and it remains a diagnosis of exclusion with poorly understood pathophysiology, variable clinical presentation and evolution of disease that is difficult to predict. 3
The incidence of PPCM varies between 1 and 100 per 10 000 live births, depending on the geographic region.4,5 PPCM has a mortality rate of 3% to 40% and is an important and steadily increasing cause of pregnancy-related morbidity and mortality worldwide.6,7 However, the recovery rate after delivery is higher than other causes of cardiomyopathy. Adverse cardiac outcomes such as persistent left ventricular systolic dysfunction, cardiac failure, heart transplant, and death occur in patients diagnosed with PPCM.7,8 CMR plays an important role in the diagnosis and management of non-ischaemic cardiomyopathies such as PPCM, with LGE providing important prognostic information.9,10
However, there is variability in the reported LGE prevalence in PPCM in the literature and the pattern of LGE is not well described. Haghikia et al evaluated 34 patients with PPCM and reported LGE in 71% 11 whereas Xu et al evaluated 84 patients with PPCM, with 45% of patients demonstrating LGE. Schelbert et al reported a very low prevalence of LGE in PPCM (5% [2/40] at baseline, 13% [5/40] in total, including at 6-month follow-up CMR). 12 Few studies have evaluated strain patterns in PPCM. Xu et al demonstrated that myocardial strain parameters were worse in PPCM patients with LGE than those without, which correlated with worse patient outcomes. 7
We aimed to evaluate the prevalence and common patterns of LGE, and to compare myocardial strain in patients with PPCM compared to healthy female controls.
Material and Methods
Patient Selection and Inclusion Criteria
This retrospective, single-centre cohort study was approved by the local institutional review board. CMR reports from January 2010 to February 2018 were retrospectively searched using the keywords “peripartum cardiomyopathy” and “post-partum cardiomyopathy.” Our search identified 59 potential patients whose electronic patient records (EPR) were further reviewed to collect additional information about their history, risk factors, and diagnosis. All patients had echocardiographic evidence of impaired left ventricular function during their initial presentation in the peripartum period. Inclusion criteria were based on the Heart Failure Association of the European Society of Cardiology that defined PPCM as idiopathic cardiomyopathy manifesting as heart failure secondary to left ventricular systolic dysfunction with a left ventricular ejection fraction (LVEF) <45% at the end of pregnancy or in the first 6 months following delivery and no history of heart diseases. 2 Exclusion criteria included factors that could have contributed to or explained their cardiomyopathy, such as familial dilated cardiomyopathy, arrhythmogenic cardiomyopathy, acute myocarditis, coronary artery disease, etc. Exclusion criteria also included lack of clinical follow-up at our institution and/or no additional information in the electronic patient record other than the CMR. Thirty-two patients were included in the study, and their initial CMR during the post-partum period was evaluated (Figure 1). One patient declined intravenous contrast administration. Ventricular function, LGE, and myocardial strain patterns of patients with PPCM were compared to a control group composed of 26 healthy women who were referred for CMR on the basis of screening for cardiomyopathy due to positive family history or other clinical features to suspect a cardiomyopathy, but had completely normal MRI findings. Baseline and follow up transthoracic echocardiographic data in the PPM cohort was reviewed to determine if there was functional improvement.

Flowchart for identification of PPCM cohort.
MRI Protocol
All study subjects were imaged with a cardiomyopathy protocol using 1.5 or 3 T MRI scanners (Magnetom Avantofit; Siemens Healthcare, Erlangen, Germany, Magnatom Verio; Siemens Healthcare, Erlangen, Germany). After localizers, ECG-gated 2-chamber, 3-chamber, 4-chamber, and short axis oblique cine steady state free precession (SSFP) stacks were acquired. Nulling times were individually adjusted to optimize the nulling of normal myocardium. To identify areas of myocardial inflammation/scarring, late gadolinium enhancement (LGE) images (Gadobutrol [Gadovist]; Bayer Healthcare, Berlin, Germany) were acquired 10 to 15 minutes after injection of gadolinium-based contrast at 0.15 mmol/kg body weight.
Cardiac MRI Analysis
Dedicated cardiac post-processing software (Circle CVI version 5.17.0) was used to determine ventricular volumes, ejection fraction, left ventricular mass, and myocardial strain values. Body surface area calculated by the Mosteller equation was used to index ventricular volumes and mass. Myocardial strain analysis in the short axis and long axis views was performed in all 32 patients and compared to a control group of 26 healthy women who underwent CMR (Circle CVI version 5.17.0).13,14
Two radiologists independently assessed LGE patterns on a per segment basis according to the American Heart Association (AHA) 17 segment model in 31 patients who underwent contrast enhanced CMR. Discrepancies were resolved by consensus review.
Statistical Analysis
Continuous variables were reported as the median (interquartile range [IQR]) with minimum and maximum values for non-normally distributed and mean and standard deviation for normally distributed data (Shapiro-Wilk test for normality). The Mann-Whitney U test served for statistical comparison of strain and MRI variables between the PPCM and control groups, if non-normal distribution of data. A paired T-Test or Wilcoxon Signed Rank test was performed to assess baseline and follow-up transthoracic echocardiography variables for the PPCM cohort, based on normality of the data. Statistical significance was determined by two-tailed P < .05. Those without follow-up data were not included in the analysis. All statistical analyses were performed with STATA software (Version 17.0).
Results
The initial cardiac MRI keyword search identified 59 patients. Twenty-seven patients were excluded after factors were identified on their EPR that could explain or contribute to their cardiomyopathy (Figure 1).
Thirty-two women with PPCM (mean age 42 ± 6 years) were included (Table 1) and compared to a control group of 26 healthy women (mean age 43 ± 14 years). The median duration between delivery and CMR was 177 days (IQR 68.5-404.5).
Patient Demographics and Clinical Characteristics at the Time of PPCM Diagnosis (n = 32).

Note. Data are listed as mean ± standard deviation or n (%). BMI = body mass index; BSA = body surface area; DCM = dilated cardiomyopathy; CVA = cerebrovascular accident; GDM = gestational diabetes mellitus; SGA = small for gestational age; NICU = neonatal intensive care unit; PPCM = peripartum cardiomyopathy; NYHA = New York Heart Association; LBBB = left bundle branch block; RBBB = right bundle branch block; ECG = electrocardiogram.
Seventeen patients (53%) were primigravid, 5 (16%) gravida 2, 2 gravida 3 (6%), 4 gravida 4 (13%), 2 gravida 5 (6%), and 1 patient gravida 6 and 8 respectively (3%) at the time of diagnosis. No patients had been diagnosed with PPCM during an earlier pregnancy. Twenty-nine patients (90.6%) presented with heart failure and 5 (15.6%) with arrhythmia. Comorbid conditions included diabetes (6.25%), hypertension (18.7%), and a family history of DCM (12.5%). Fifteen patients (46.8%) were admitted during their pregnancy for cardiovascular complications including heart failure requiring diuretic therapy (n = 13), arrhythmia (n = 5), hypertension (n = 2), and cerebrovascular accident (n = 1) and obstetric complications, including as pre-eclampsia (n = 8), gestational diabetes (n = 3), post-partum haemorrhage (n = 1), and premature rupture of membranes (n = 1).
Transthoracic echocardiography at presentation demonstrated mean left indexed ventricular end-diastolic volume (LVEDVi) and LVEF of 85.2 ml/m2 and 28.5%, respectively (Table 2). Twenty-six patients in the PPCM cohort were followed up with a transthoracic echocardiogram, with a median duration between the initial and follow-up echocardiogram of 417 days (IQR 262-699). Twenty-four (92%) of these patients had an improvement in LVEF (mean LVEF 28% ± 1.91% at diagnosis and 45% ± 3.0% at follow-up, P < .001). At the most recent clinical follow-up (median duration between diagnosis and follow-up of 1385 days [IQR 401-2044]), 3 patients (9%) had persistent left ventricular dysfunction (defined as LVEF <45%), and 3 patients (9%) had persistent palpitations. One patient with persistent left ventricular dysfunction had LGE, whilst all 3 patients with persistent palpitations had LGE, 2 of which were mid-wall linear LGE and one with only right hingepoint LGE. There were no major adverse cardiac events, defined as death, non-fatal myocardial infarction, or unplanned revascularization 15 in the PPCM cohort.
Baseline and Follow-Up Transthoracic Echocardiography.
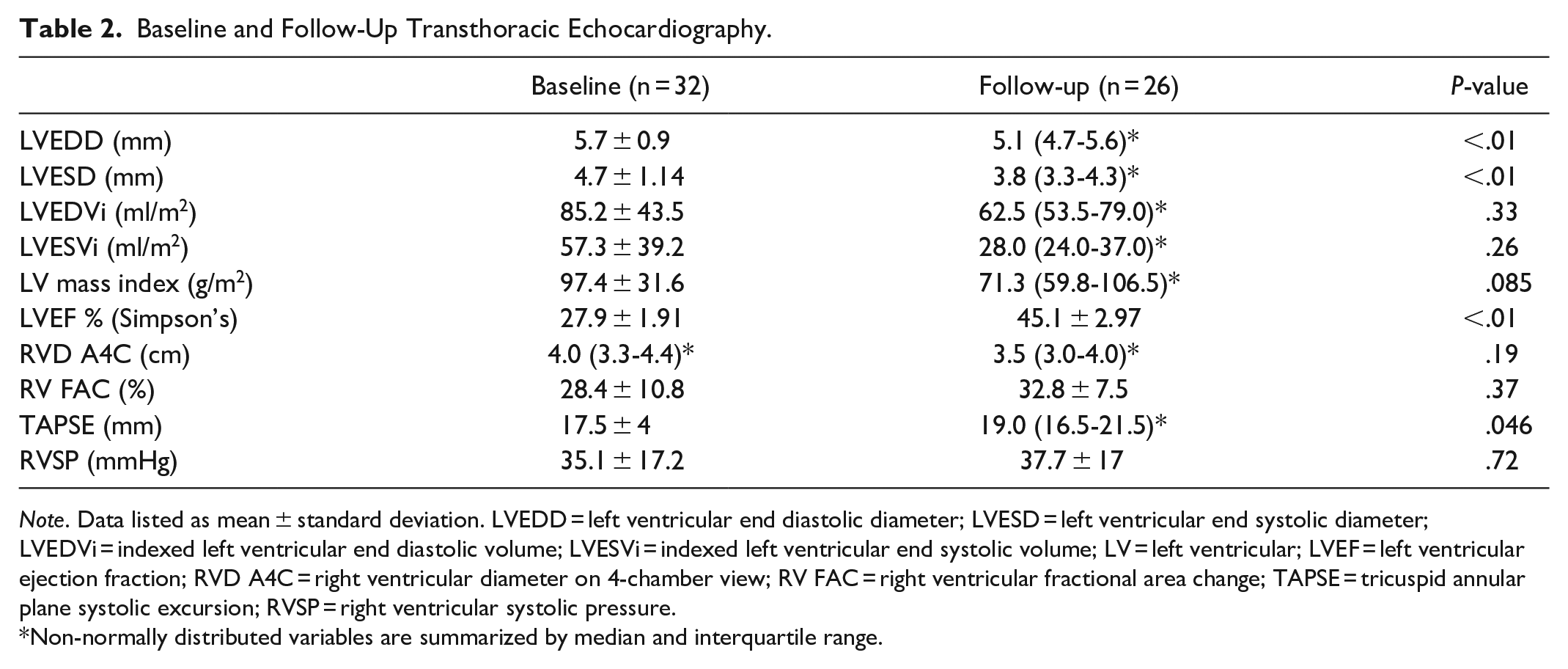
Note. Data listed as mean ± standard deviation. LVEDD = left ventricular end diastolic diameter; LVESD = left ventricular end systolic diameter; LVEDVi = indexed left ventricular end diastolic volume; LVESVi = indexed left ventricular end systolic volume; LV = left ventricular; LVEF = left ventricular ejection fraction; RVD A4C = right ventricular diameter on 4-chamber view; RV FAC = right ventricular fractional area change; TAPSE = tricuspid annular plane systolic excursion; RVSP = right ventricular systolic pressure.
Non-normally distributed variables are summarized by median and interquartile range.
Women with PPCM had significantly lower LVEF by MRI (median 37.5%, 25.5%-45.5% vs 60.5%, 57.0-63.0, P < .001) and higher LV end-diastolic volumes (median 108 ml/m2, 93.5-135.5 ml/m2 vs 76 ml/m2, 71.0-88.0, P < .001). Right ventricular indexed end-diastolic volumes and ejection fractions were similar for both groups.
Women with PPCM had significantly reduced global LV circumferential strain (mean −11.1 ± 4.5 vs −19.2 ± 1.6, P < .01), reduced global LV radial strain (mean 16.0 ± 8.1 vs 33.2 ± 4.5, P < .001), and reduced global LV longitudinal strain (mean −11.4 ± 4.2 vs −18.6 ± 1.4, P < .001), compared to controls (Table 3).
MRI Strain, Ventricular Size and Function, and LGE in PPCM Compared to Control Group.
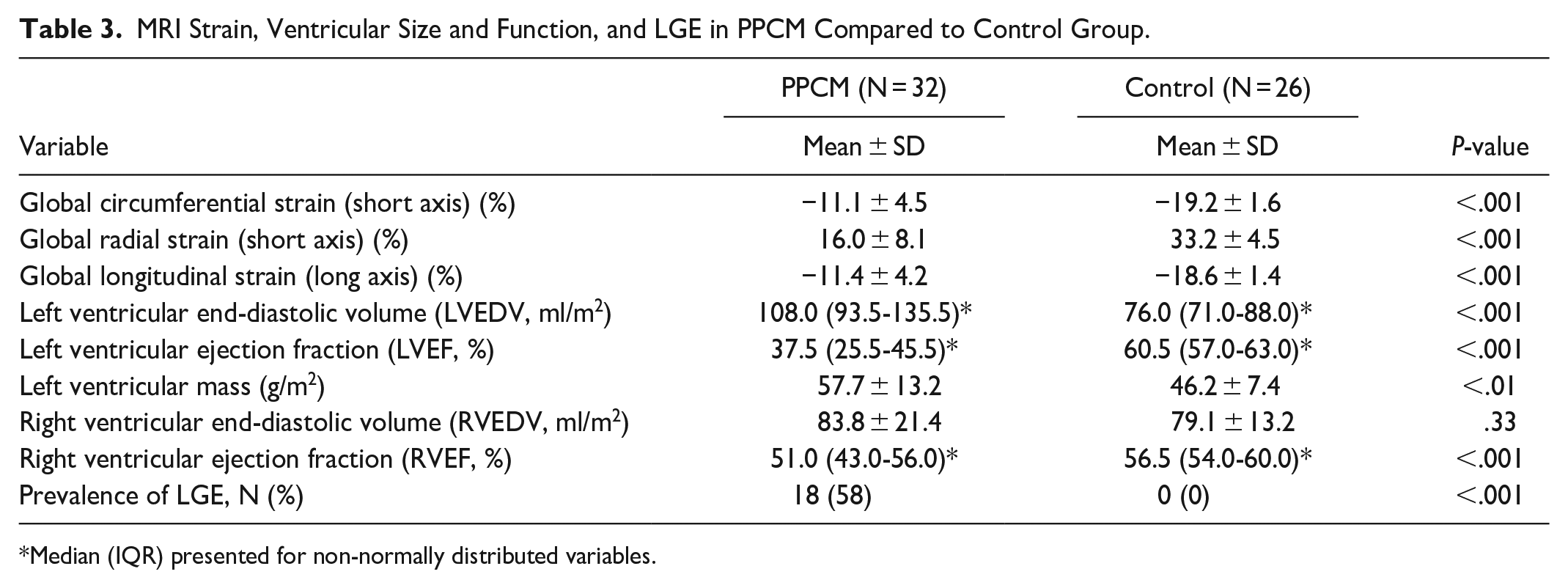
Median (IQR) presented for non-normally distributed variables.
One out of the 32 patients declined intravenous contrast. Eighteen patients (58%) had non-ischaemic pattern LGE, with no (non-hingepoint) LGE in the controls. LGE was present in the basal anteroseptum, basal inferoseptum, basal inferior, mid anteroseptum, mid inferoseptum, and mid inferolateral segments (Figure 2). LGE was most prevalent in the basal anteroseptum (n = 12) and mid anteroseptum (n = 8) as shown in Figure 2.
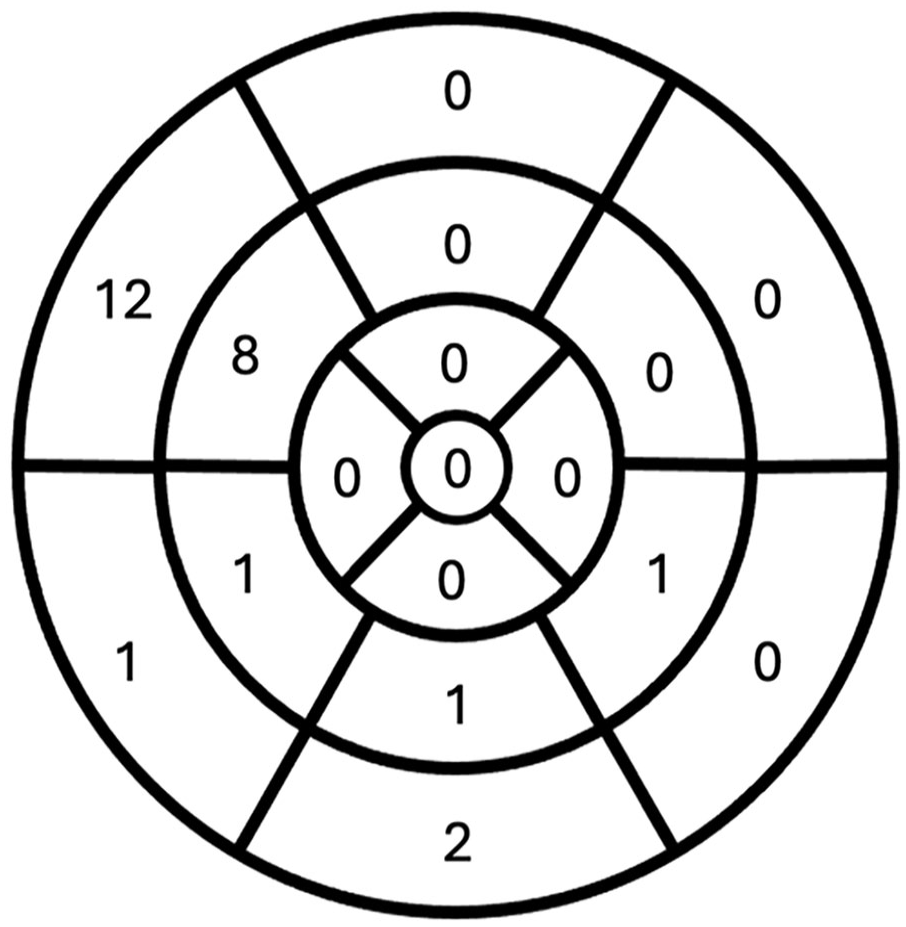
LGE distribution by AHA segments in the PPCM cohort.
LGE patterns observed included linear mid-wall (n = 8, 26%) most frequently as shown in Figure 3, then linear subepicardial (n = 6, 19%), and right ventricular side of the interventricular septum (n = 3, 10%), non-specific right inferior hingepoint (n = 13, 42%). Six out of 26 women in the control group had only right inferior hingepoint LGE (23%) and none had non-hingepoint LGE.
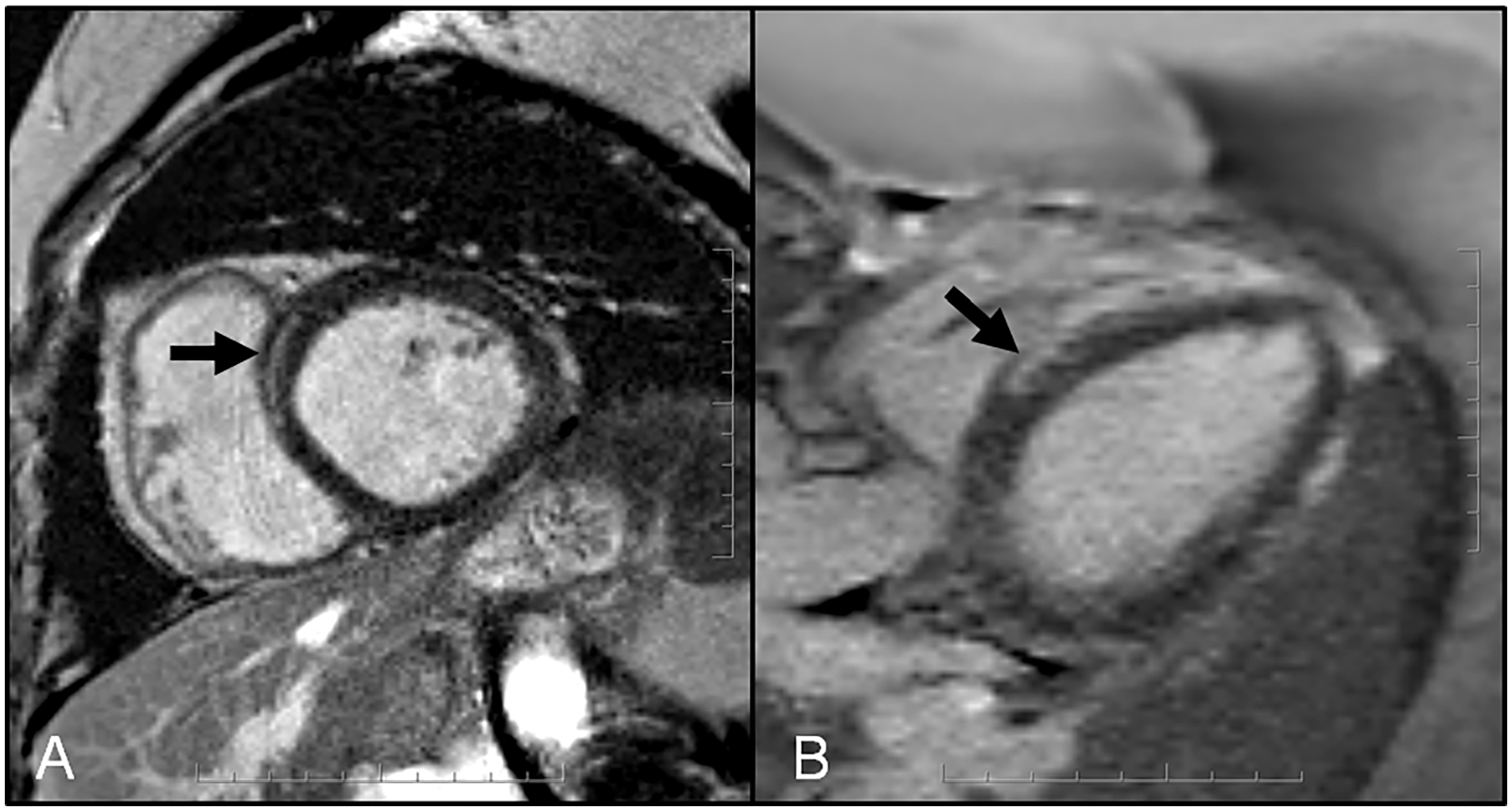
35-year G2P2 with PPCM—Short axis (A) and 3 chamber (B) late gadolinium enhancement (LGE) images depicting mid-wall enhancement in the basal anteroseptum.
Discussion
The presence of LGE is associated with worse outcomes across non-ischaemic cardiomyopathies, including those with PPCM. 10 There is variable reported LGE prevalence and the LGE pattern in patients with PPCM is not well established. Halliday et al noted that the location of LGE is important and is a better prognostic factor than the extent of LGE. 16 Our study is the first to clearly state that a linear LGE pattern is most common and that LGE was most frequently seen in the basal and mid anteroseptum. However, our sample size was too small with too few cardiovascular events to correlate pattern and distribution of LGE with patient outcomes. Petryka-Mazurkiewich et al evaluated the structural differences in patients with PPCM and idiopathic DCM and observed that myocardial strain was more abnormal in patients with DCM than PPCM. They also reported that PPCM and DCM patients with LGE had more dilated ventricles with reduced function than those without LGE. 17 However, ability to differentiate DCM from PPCM may be challenging as there are similar features.
Eighteen (58%) patients in the PPCM cohort had LGE, which is higher compared to other recently published studies by Petryka-Mazurkiewicz et al (43%) and Xu et al in (45%); but less than reported by Haghikia et al (71%).7,11,17 Schelbert et al in contrast reported a low prevalence (12.5%) of LGE in their prospectively recruited cohort of 40 women diagnosed with PPCM (2 women had LGE at baseline CMR, 3 had LGE detected at 6 month follow up CMR). 12 We did our best to exclude patients who were more likely, on the basis of clinical presentation, biomarkers, and LGE pattern to have acute myocarditis rather than PPCM. We acknowledge that the potential inadvertent inclusion of patients with other diagnoses other than PPCM in published studies may be a contributing factor to the highly variable LGE prevalence rates.
PPCM diagnosis is also challenging due to overlapping features with non-ischaemic dilated cardiomyopathy that can also have a mid-wall, linear LGE pattern. Peri-partum cardiomyopathy may represent a non-ischaemic dilated cardiomyopathy that occurs during pregnancy and the post-partum period. The pregnancy state may potentially unmask or trigger another type of non-ischaemic cardiomyopathy. As we learn more from prospective studies and international multi-site registries, we may better understand the pathophysiology of PPCM.
Isaak et al evaluated the diagnostic and prognostic value of CMR in patients with acute stage PPCM compared to healthy pregnant women. Similar to Isaak et al, PPCM patients in our cohort had reduced left ventricular function, increased left ventricular volumes, increased left ventricular mass, and abnormal strain parameters compared to healthy control group. Isaak et al observed impaired right ventricular systolic function with normal right ventricular volumes in patients in the acute stage of PPCM, which normalized on follow-up CMR. 18 Our patient population had normal median right ventricular systolic function and volumes but the majority of the PPCM cohort were not scanned during the acute phase.
This is a retrospective single-centre study with potential selection bias as patients were identified using keywords to search radiology reports from January 2010 to February 2018. Our patient population may have had more severe cardiovascular dysfunction or symptoms to warrant a CMR. In addition, a relatively small patient cohort, long interval between delivery and baseline MRI, and incomplete clinical and imaging follow-up data limit the ability to correlate the temporal changes in LGE, strain patterns, and functional data with patient outcomes. However, other studies have found significant value in CMR in PPCM patients for both the diagnosis and prognostication. CMR is helpful in suspected PPCM cases not only to rule out other causes of cardiac dysfunction (such as acute myocarditis) but also valuable to provide accurate biventricular size and functional assessment, strain analysis for earlier detection of cardiac dysfunction, LGE detection, which has been shown to offer prognostic value correlating with worsening ventricular function over time or lack of functional recovery.7,11,18 Furthermore, our control group consisted of healthy women who were not pregnant or postpartum. This limits our ability to draw conclusions regarding comparisons with a cohort of women with uncomplicated pregnancy or women in the post-partum period. Another limitation is that parametric mapping and T2 weighted imaging, which is standard practice today, were not performed in most PPCM patients (n = 27). This precludes identification of interstitial fibrosis and/or myocardial oedema that can be present even in the absence of LGE. Future multicentre research involving larger patient populations is needed to confirm our findings.
Conclusion
PPCM remains a diagnosis of exclusion and poses a diagnostic dilemma for referring clinicians. Myocardial strain was significantly reduced in patients with PPCM compared to the control group. The prevalence of LGE in our cohort was higher than reported in some recently published studies. LGE was most often linear and most prevalent in the basal and mid anteroseptum in our study. This LGE pattern and distribution may be nonspecific in isolation but could suggest PPCM in the correct clinical context along with abnormal CMR strain values. Larger prospective studies are needed to validate these findings.
Footnotes
Acknowledgements
We would like to express our gratitude to all those who contributed to the completion of this research and to our families for their continued support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
