Abstract
In the immunocompromised setting, there are distinct radiologic findings of primary central nervous system lymphoma (PCNSL), including necrotic ring-enhancing lesions, increased propensity for intralesional haemorrhage, and multiplicity. In this clinical context, advanced imaging with MR perfusion, spectroscopy, and diffusion-weighted imaging can be used to increase accuracy in the diagnosis of lymphoma over mimics such as high-grade glioma, metastases, or infection. This review summarizes the histology and pathophysiology of PCNSL in immunodeficient hosts, which provide a basis for its imaging appearances, prognosis, and treatment. This discussion is important for the general radiologist as the incidence of immunodeficiency-related PCNSL may be increasing.
This is a visual representation of the abstract.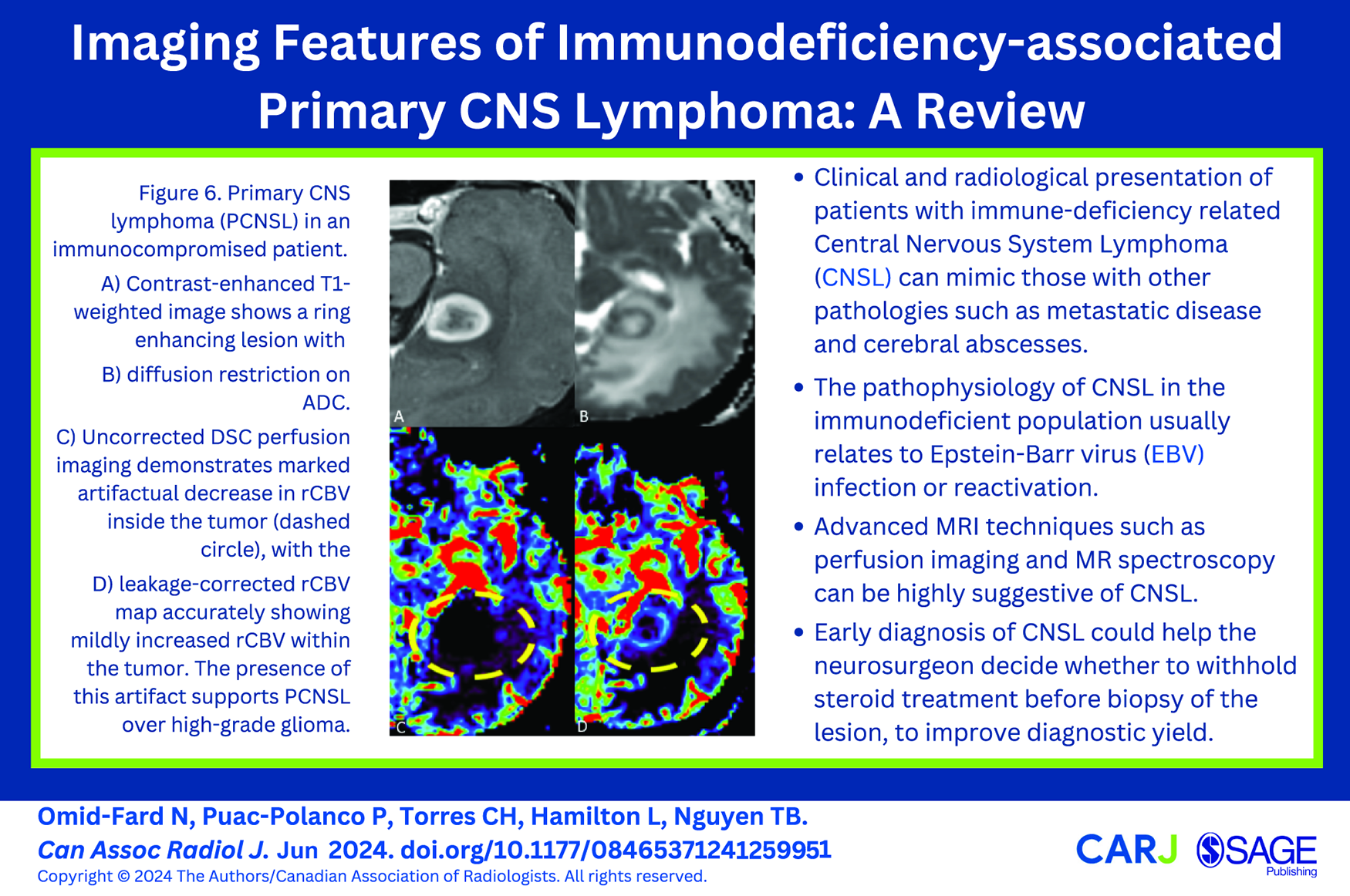
Introduction
Immunodeficiency-associated primary central nervous system lymphoma (PCNSL) is a distinct and rare clinical entity, typically related to the Epstein-Barr virus (EBV). 1 EBV-related lymphoproliferative disease most commonly involves the lymphatic system, bone marrow, lung, and abdomen. 2 In a minority of cases, the central nervous system (CNS) can be involved, with an estimated incidence of 7% to 15% in the post-transplant context. 3 Timely diagnosis is essential, especially in patients taking immunosuppressive medications, as withdrawal can sometimes lead to lymphoma regression. 4
This review will highlight the spectrum of imaging findings in immunodeficiency-associated PCNSL, along with an overview of the pathophysiology, histopathology, treatment, and prognosis. Imaging protocols and techniques will also be discussed.
Disease Classification
The World Health Organization 2021 classification identifies 4 main groups of CNS lymphoma 5 : (1) Primary diffuse large B-cell lymphoma of the CNS; (2) Intravascular large B-cell lymphoma; (3) Lymphomatoid granulomatosis (LG); and (4) Immunodeficiency-associated CNS lymphoma. The latter category includes human immunodeficiency virus (HIV)-associated and immunosuppressive treatment-related PCNSL, of which post-transplantation lymphoproliferative disorder (PTLD) is the most well-described and can present with primary CNS involvement. PTLD is becoming more relevant as the number of surviving organ transplant recipients increases, along with a greater incidence of PCNSL. 6 Finally, a lesser studied group of patients with immunodeficiency-associated PCNSL is those under prolonged immunosuppressive treatment due to underlying autoimmune disorders. 7 Primary immunodeficiency syndromes are scarcely studied as a separate category in relation to PCNSL due to their rarity, but imaging appearances have been described similar to the autoimmune group. 8
Pathophysiology
The predominant mechanism by which lymphoma develops in immunocompromised individuals is based on EBV infection. This double-stranded deoxyribonucleic acid (DNA) herpes virus has infected 90% of the world’s population, spreading often occultly via oral secretions. 3 After the initial infection of epithelial and predominantly B lymphocytes, the virus enters a latent phase as an episome adjacent to the host DNA, 9 limiting its detection by the immune system. Latently infected B-cells can later reactivate and proliferate through production of viral oncoproteins and microribonucleic acids that alter the cell cycle, promote infected cell proliferation and turnover, and interfere with tumour suppressor genes.3,10 In immunocompetent individuals, EBV infection is kept under control by EBV-specific CD8+ T cells that target infected B-cells, but lack of this response in immunosuppressed individuals leads to autonomous proliferation and eventually lymphoproliferative disease. 3 The histological features of immunocompetent versus immunodeficient PCNSL are contrasted in Figure 1.
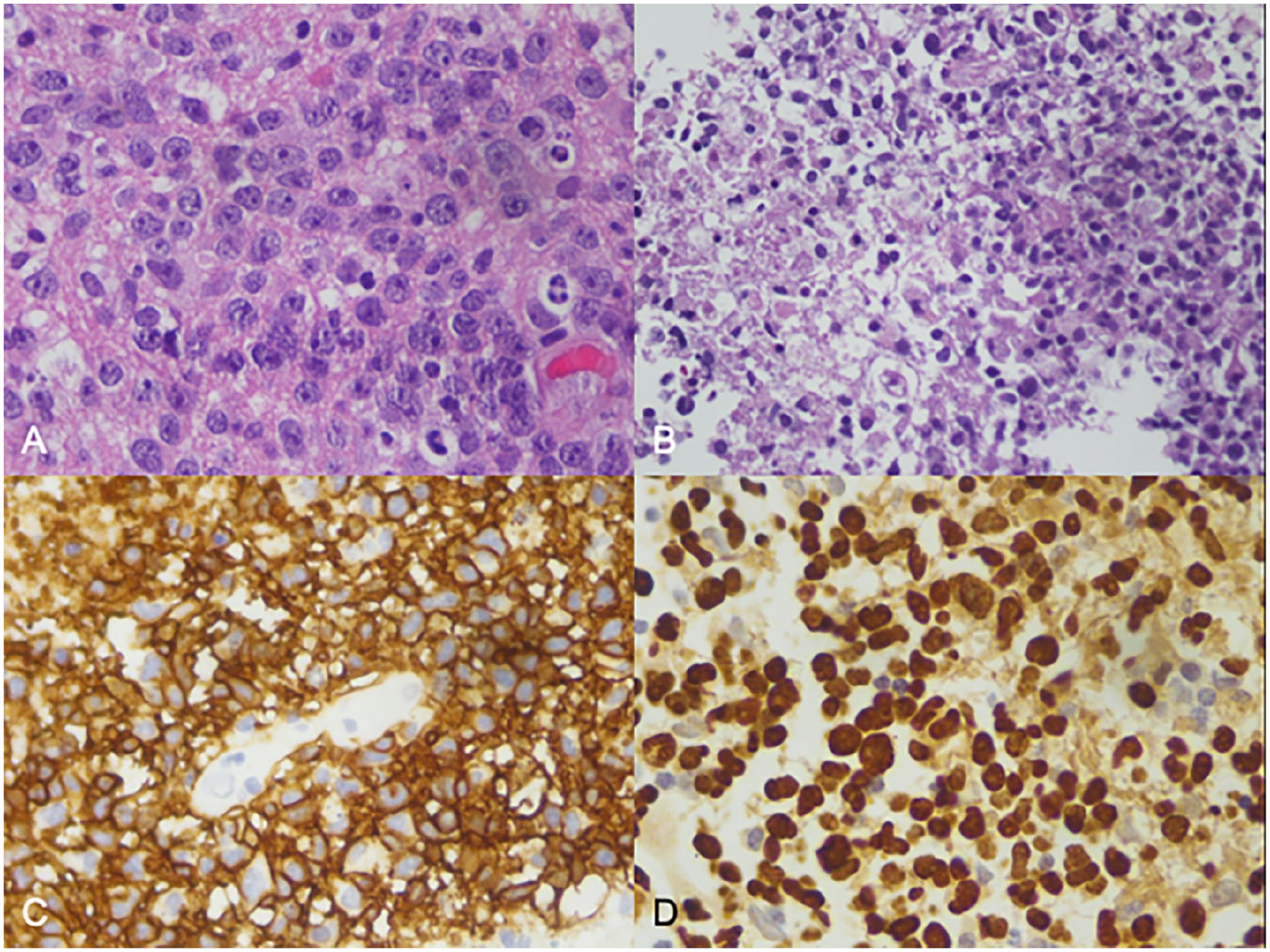
Histopathology of immunocompetent versus immunodeficiency-associated PCNSL. (A) Example of diffuse large B-cell lymphoma (DLBCL) in an immunocompetent individual with sheets of dyscohesive large cells with minimal cytoplasm and large nuclei with prominent nucleoli (H&E). (B) Example of EBV-positive DLBCL in immunocompromised individual (post-transplant lymphoproliferative disease) with area of pink coagulative-type necrosis within tumour (left side image; H&E). (C) Immunohistochemistry for B-cell marker CD20 shows diffuse strong membranous staining of the lymphoma cells. (D) Chromogenic in situ hybridization with EBV RNA probe (EBER) demonstrates diffuse strong nuclear staining of the lymphoma cells in an immunocompromised individual.
Immunocompetent PCNSL (Figure 2) is generally unrelated to EBV, in contrast to its immunodeficient counterpart, but it is nonetheless important to note that not all tumours in the immunocompromised population test positive for EBV. In fact, 20% to 40% of PTLD are EBV negative, and this proportion has been increasing. 3 This finding can potentially be explained by the “hit-and-run” hypothesis, which predicts continued oncogenesis in cells even if EBV DNA is cleared from them. 9 However, other mechanisms could also account for these EBV-negative cases, which demonstrate differing pathology, including more monomorphic cell lines and more T-cell origin lymphoma in the context of PTLD. 3 Furthermore, rare cases of Human Herpesvirus-8 related PTLD have been described. 3
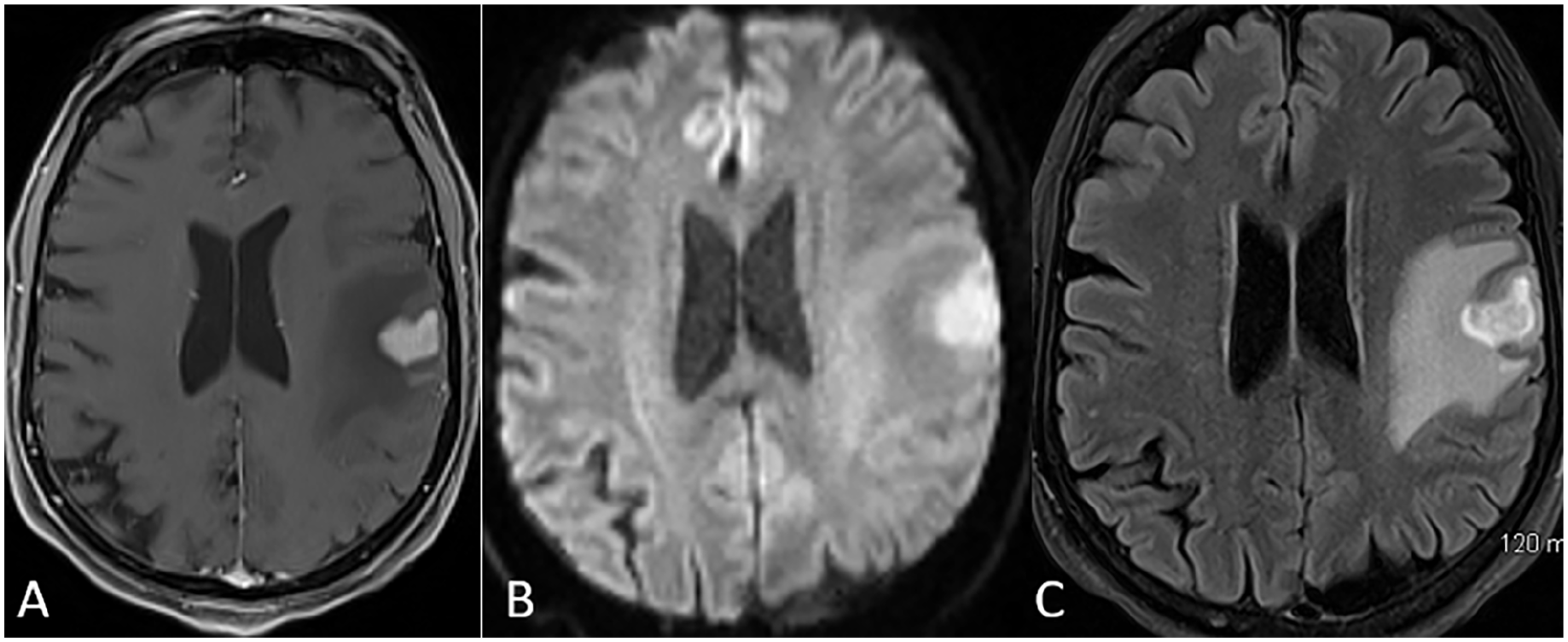
Characteristic features of immunocompetent primary CNS lymphoma on MRI. (A) Contrast-enhanced T1-weighted axial images show a homogenously enhancing mass in the left subcortical frontal lobe. (B) DWI (B value 1000 s/mm²) image shows avid diffusion restriction (verified on ADC, not shown). (C) T2-weighted FLAIR shows areas of intermediate to low signal within the mass and moderate surrounding vasogenic oedema.
While the above pathophysiology forms the basis of all types of immunodeficiency-related PCNSL, phenotypic differences exist in biochemistry, histopathology, clinical presentation, and imaging appearance. Detailed pathological and clinical features are beyond the scope of this review but have been previously described. 3 A brief discussion is provided below.
Acquired immune deficiency syndrome (AIDS)-related PCNSL (Figures 3 and 4) is rare and tends to manifest late in the disease course, unlike its systemic counterpart. 11 As an AIDS-defining illness, lymphoma typically occurs systemically and earlier in the disease process with a median CD4 count of 189 × 106/L. However, PCNSL occurs with a median CD4 count of 30 × 106/L and is often comorbid with opportunistic infections. The mechanism is usually secondary to a weakened immune response and prior EBV infection, with the direct oncogenic effect of HIV-related B-cell hyperactivation considered less contributory. 11 Incidence of PCNSL in HIV patients has significantly decreased with modern antiretroviral therapies. 11
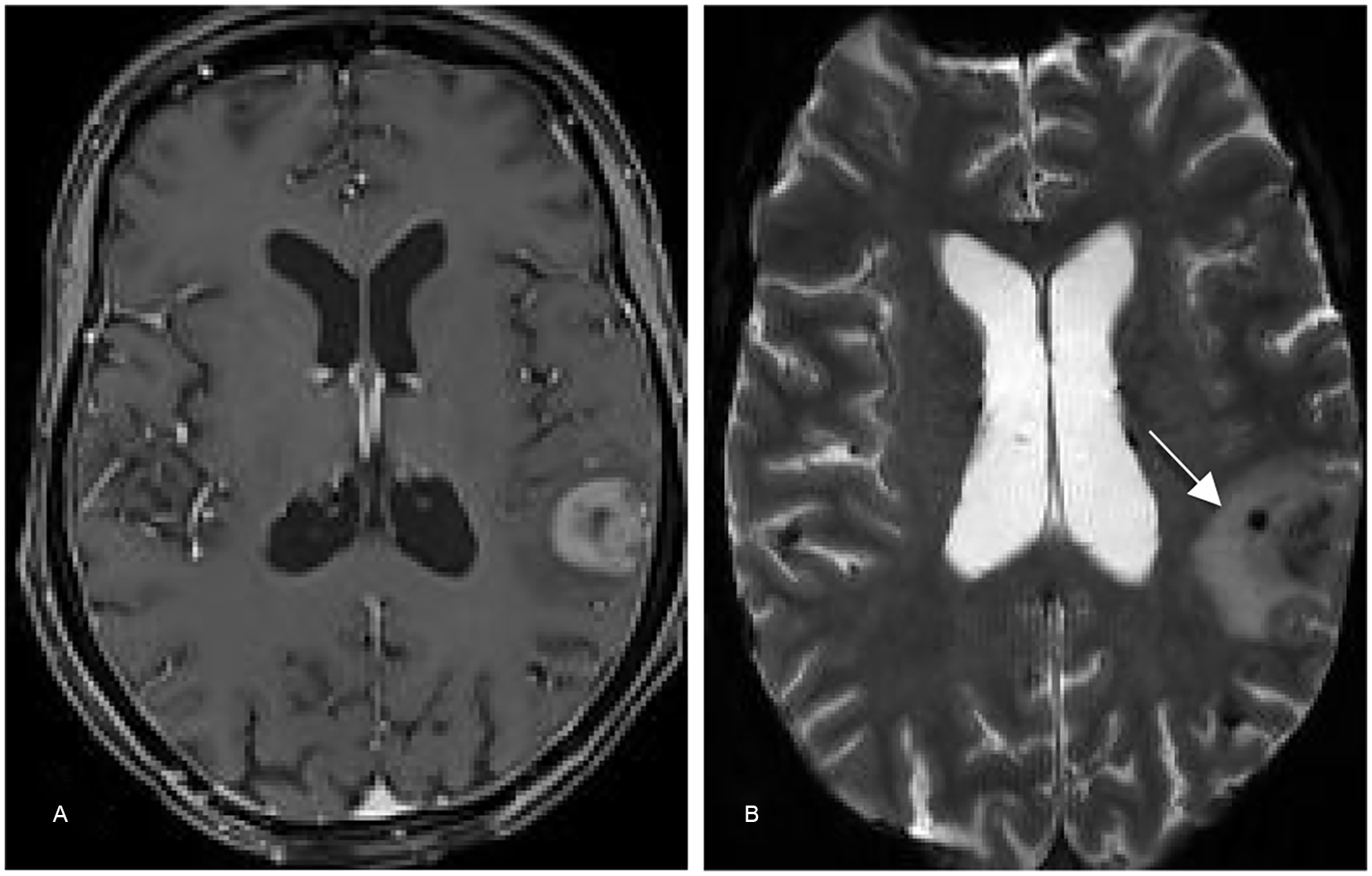
EBV+ Diffuse large B-cell lymphoma in a 45 year-old HIV+ female, noncompliant with medications, who presented with seizures and hallucinations. Unfortunately despite treatment she experienced rapidly progressive cognitive decline and delirium, dying 1 month after the biopsy. (A) Contrast-enhanced T1-weighted image shows avid ring enhancing lesion in the left temporal lobe. (B) Blooming artifact seen within the lesion (arrow) on GRE indicating microhaemorrhage.
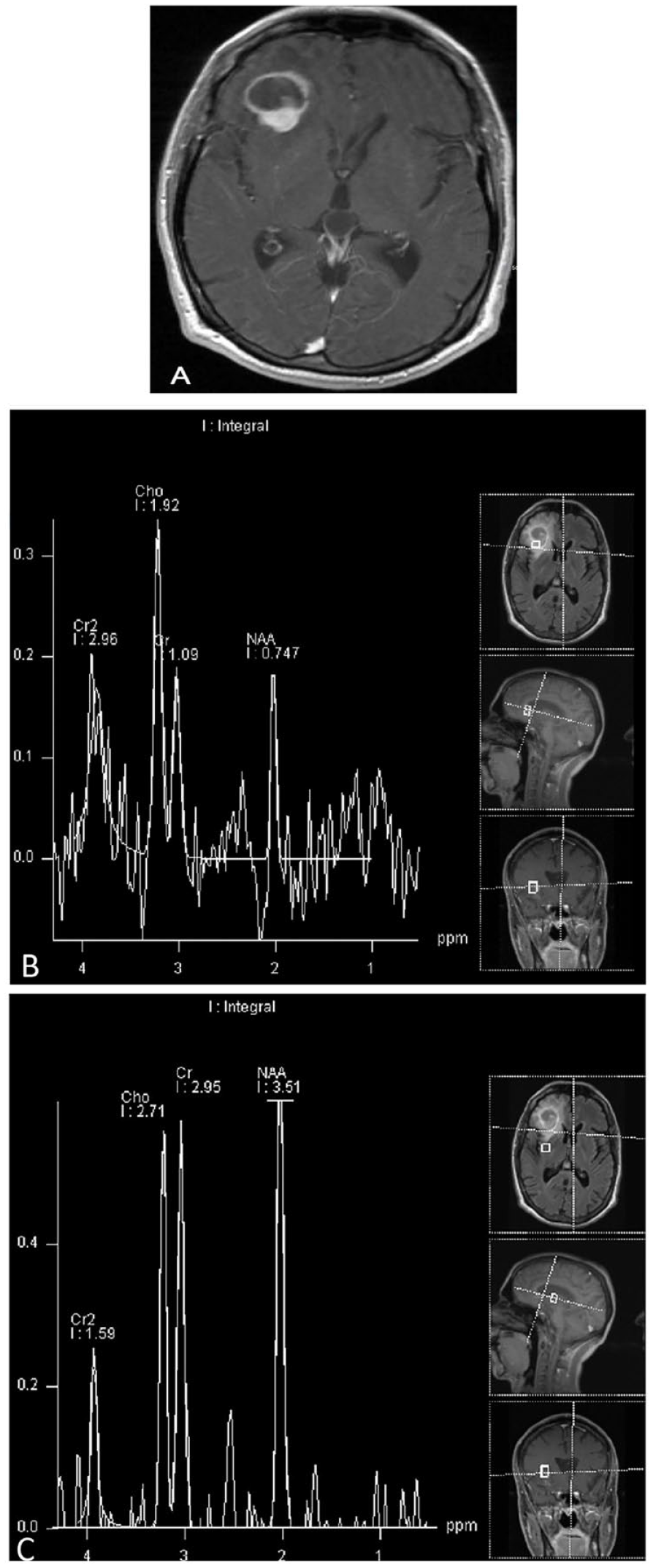
Aggressive EBV negative B-cell non-Hodgkin lymphoma in a 35 year-old male with advanced HIV, non-compliant with treatment, presenting with 3 to 4 months of profound personality changes. Unfortunately, despite radiotherapy he died 1 month later with CNS depression and other HIV complications including opportunistic infections. (A) Contrast-enhanced T1-weighted sequence showing a ring enhancing lesion in the right frontal lobe with posterior nodular component. (B) Single voxel MR spectroscopy (TE 135 ms) demonstrating elevated choline/creatine and choline/NAA ratios within the enhancing components, but not surrounding the tumour (C).
Lymphomatoid granulomatosis (LG) is an angiocentric form of lymphoproliferative disease that can involve the CNS concurrently with other extranodal organ systems, universally affecting the lungs, 12 although case reports of primary CNS LG exist. 13 LG is theorized to stem from the B-cell proliferative response to EBV antigen stimulation but with prominent angiocentric T-cell infiltrates.12,14 Historically associated with the immunocompromised state, LG is now defined as an immunocompetent disease and can be seen in middle-aged adults or post-treatment immune reconstitution syndrome of patients with AIDS.3,14 In the immunocompromised setting, the LG appearance is considered a subtype of a polymorphic lymphoproliferative disorder. 15
Transplant-related lymphoproliferative disease occurs after years of immunosuppressant use, allowing EBV-infected B-cell proliferation. 3 CNS PTLD (Figure 5) presents later than other types of PTLD and is EBV+ in most cases. 3 The potency of immunosuppression and the type of medication used influence the risk of PTLD, with some drugs conferring greater risk specifically for CNS involvement, such as Belatacept, a human IgG1 immunoglobulin selectively blocking the process of T-cell activation to prevent transplant organ rejection. 3 Host factors are also important, with young (<18 years) and old (>60 years) Caucasian males at highest risk for PTLD. 3 However, primary exposure to EBV from the allograft in a seronegative organ recipient is the most critical risk factor. 3
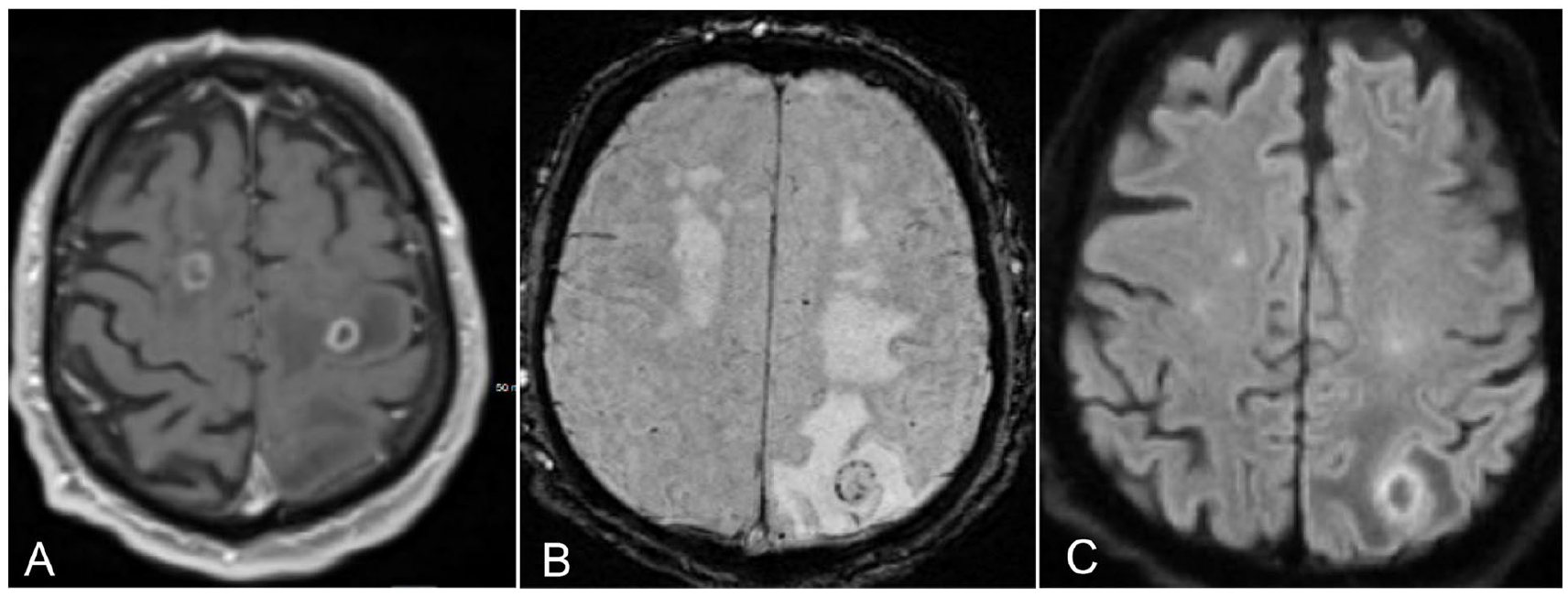
EBV positive diffuse large B-cell lymphoma in a 72 year-old male with renal transplant presenting with seizure, progressive right arm weakness, aphasia, and confusion. The patient was on cyclosporine, cellcept, and prednisone. (A) Contrast-enhanced T1-weighted image shows ring enhancing lesions in the bilateral frontal lobes. (B) SWI sequence shows blooming artifacts within the left parietal lesion, consistent with microhaemorrhage. (C) DWI (B-value 1000 s/mm²) shows diffusion restriction within the solid component of the lesion (verified on ADC, not shown).
Autoimmune disease-associated PCNSL has not been well-studied (Figure 6). A recent meta-analysis showed the most common autoimmune conditions in a cohort of PCNSL patients to be systemic lupus erythematosus, multiple sclerosis, myasthenia gravis, and rheumatoid arthritis, although sample sizes were small. 7 Presumably, the disease pathophysiology relates to immunosuppressant drug use dose and type. However, the inflammatory effect of the disease itself should be considered as well. 7 For instance, type 1 diabetes is the most prominent autoimmune condition in the general population, yet no significant association with PCNSL has been found. 7 The low inflammatory activity of this disease is, however, confounded by the rare requirement for immunosuppressives.
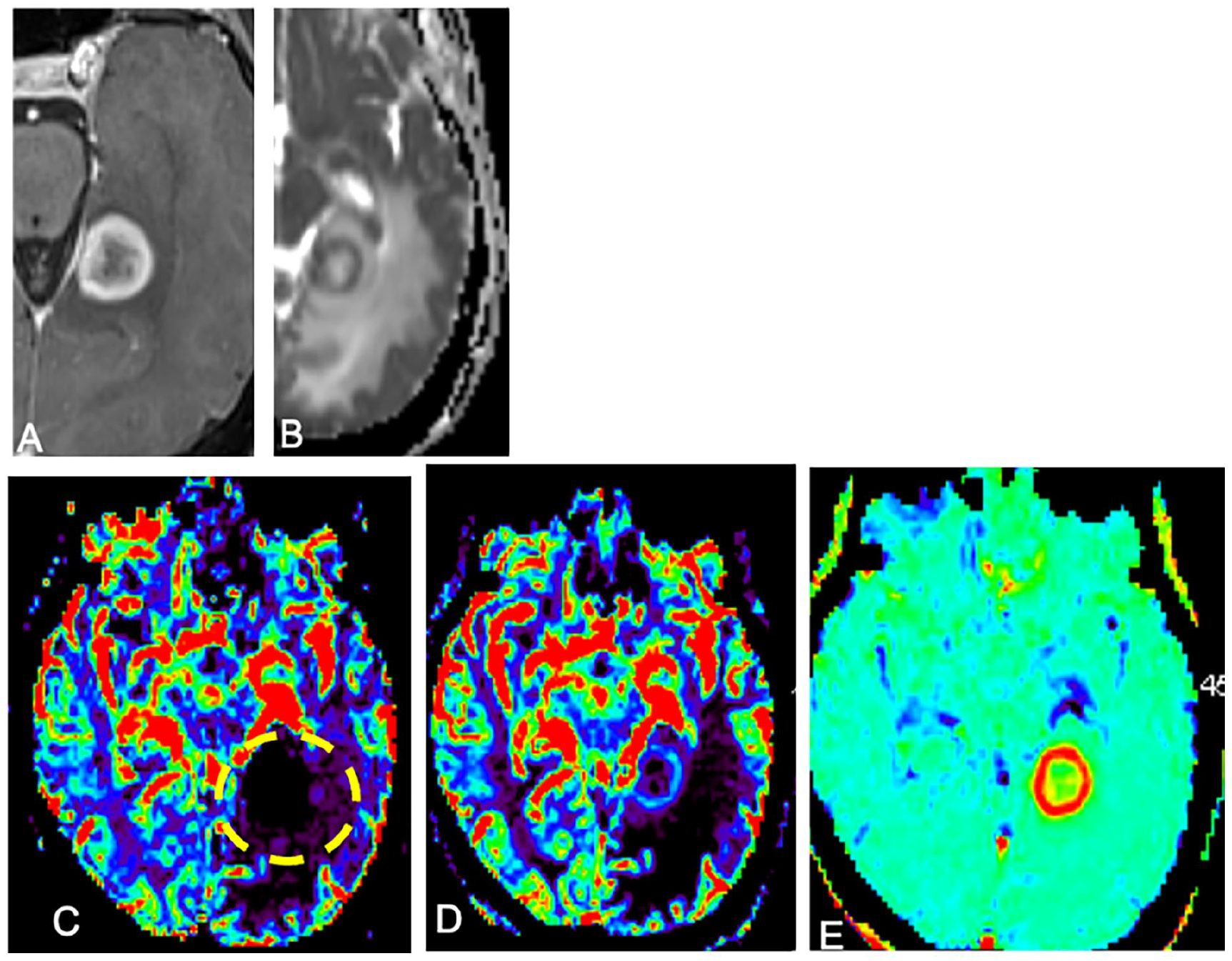
Diffuse large B cell lymphoma (EBV negative) in a 69 year old female with systemic lupus erythematosus and nonspecific interstitial pneumonitis treated with mycophenolate mofetil and prednisone. (A) Contrast-enhanced T1-weighted image shows a ring enhancing lesion with (B) diffusion restriction on ADC. (C) DSC perfusion imaging demonstrates marked artifactual decrease in uncorrected rCBV inside the tumour (dashed circle), with the (D) leakage-corrected rCBV map accurately showing mildly increased rCBV within the tumour. This is due to the effect of high vascular permeability (elevated K2, mapped in (E)) leading to blunting of T2*-weighted signal-intensity loss secondary to extravascular gadolinium T1 effects.
Imaging Appearances
The imaging features of immunocompetent PCNSL have been previously documented, 16 highlighted by Figure 2. Periventricular or superficial parenchymal lesions with moderate to marked homogeneous contrast enhancement are typical. Secondary intraocular involvement has been described in 25% of cases. 16 On unenhanced CT, hypercellularity leads to iso- to hyperdense lesions. On MRI, hypercellularity often leads to T2 hypointensity relative to gray matter and diffusion restriction in the enhancing component. 16 There are distinct radiologic findings of PCNSL in the immunocompromised setting, summarized in Table 1, including (1) presentation with multifocal lesions; (2) lesions containing necrotic cores leading to ring enhancement; and (3) increased propensity for intralesional microhaemorrhage, 16 which can be shown with gradient echo planar (GRE) or susceptibility weighted imaging (SWI). Basal ganglia and corpus callosal involvement have also been described, 16 but are nonspecific.
Distinguishing Features of Immunocompetent Versus Immunodeficiency-Associated Primary CNS Lymphoma.
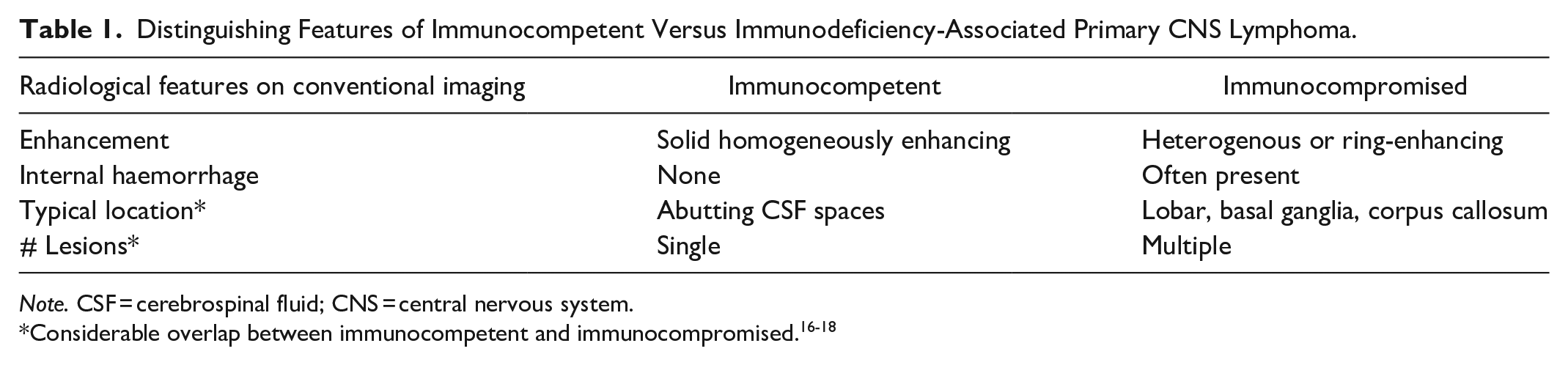
Note. CSF = cerebrospinal fluid; CNS = central nervous system.
Thus, immunosuppressed PCNSL can have a similar appearance to metastatic disease or cerebral abscesses on conventional imaging but might be differentiated from them with additional sequences. On SWI, the presence of a dual rim sign, defined as an outer hypointense ring with adjacent inner hyperintense ring, can support pyogenic abscess. 19 Apparent diffusion coefficient (ADC) values of the enhancing component of PCNSL are generally lower than for high-grade glioma (HGG), which is a useful differentiating feature. 20 On dynamic susceptibility contrast (DSC) perfusion-weighted imaging (Figure 6), an intermediate degree of relative cerebral blood flow (rCBV) increase is expected (ie, less than HGG). The rCBV of PCNSL can be underestimated due to the T1 effects of interstitial gadolinium from leaky tumour vessels. 21 This can lead to a significant discrepancy between the uncorrected rCBV and the leakage corrected rCBV. The uncorrected rCBV is the most accurate perfusion parameter to differentiate between lymphoma and glioblastoma since T1 leakage effects are less pronounced in HGG and do not lead to an artifactual decrease in rCBV. 22
Dynamic Susceptibility Contrast Perfusion Parameters
A GRE acquisition following an injection of 0.1 mmol/kg of gadolinium is the most common technique used. Post-processing model-based leakage correction can be performed to obtain both corrected and uncorrected rCBV. As discussed above, compared to HGG, rCBV is typically lower in PCNSL (particularly uncorrected) since neoangiogenesis is infrequently seen in lymphoma but the leakage coefficient (K2) is higher due to increased vascular permeability. 22 While tissue permeability has traditionally been mapped using T1-based dynamic contrast enhancement (Ktrans), at our institution T2*-based dynamic susceptibility contrast is preferred due to faster acquisition time within the first pass, obtaining the related K2 coefficient, which is used for leakage correction of the rCBV maps. 23
MR Spectroscopy
MR Spectroscopy (MRS) can be a useful adjunct to narrow the differential in ambiguous cases of PCNSL (Figure 4). While the choline (Cho)/creatine (Cr) ratio of typical PCNSL has been found to be higher than atypical cases, 24 the real value of MRS is in distinguishing lymphoma from other entities. For example, higher Cho/Cr and Cho/N-acetylaspartate (NAA) ratios can differentiate PCNSL versus tumefactive demyelination (using intermediate echo time, TE 144 ms) 24 and the presence of amino acid signals can suggest an abscess. 25 MRS parameters can also be used to predict the likelihood of PCNSL over glioblastoma or metastasis, particularly when combined with perfusion imaging. For example, Cho/NAA has been found to be lower in PCNSL compared to HGGs (TEs 35/135 ms) due to increased neuronal loss causing a greater decrease in NAA in the latter. 26 The presence of MRS abnormality outside the region of the mass can suggest an infiltrative glioma instead of PCNSL. Advanced imaging features are summarized in Table 2.
Advanced Imaging Features to Distinguish Primary CNS Lymphoma From Mimics.

Note. PCNSL = primary CNS lymphoma; HGG = high-grade glioma; DWI = diffusion-weighted imaging; rCBV = relative cerebral blood volume; MRS = magnetic resonance spectroscopy; Cho = Choline; NAA = N-acetylaspartate.
Nuclear Medicine
The role of nuclear medicine in the initial diagnosis of PCNSL is limited, although sometimes used. Fludeoxyglucose F18 positron emission tomography (PET) will show intense activity in the solid portions, in contrast to toxoplasmosis, but can be obscured by adjacent gray matter activity. 31 C-11 methionine PET can overcome this limitation given its low background brain parenchymal uptake. Differentiating PCNSL from toxoplasmosis can also be done using Thallium-201 imaging, which shows avid uptake in the former but not the latter. 31 Added benefits of PET are concurrent assessment of the whole body to find extra-CNS disease and detecting residual disease after treatment.
Treatment and Prognosis
The treatment of EBV-induced PCNSL is constantly evolving given the lack of specific therapies. First, a histopathological diagnosis must be obtained, usually through neurosurgery, but in some cases through CSF sampling or biopsy of other sites when there is extra-CNS disease. 32 Steroids should be avoided unless urgently needed to reduce mass effect, as they can reduce diagnostic yield and blunt future cytotoxic responses. 32 This highlights the importance of accurate diagnosis by the radiologist. In general, the treatment is based on age and performance status with the goal of prolonging survival while minimizing toxicity. Induction therapy consists of high-dose methotrexate (MTX) as the backbone, which can be used alone or with other multi-drug regimes (eg, procarbazine, vincristine, rituximab, etc.). The use of multi-drug regimes in combination with MTX improves the response rates than MTX alone. 33 In immunocompetent PCNSL, the role of radiotherapy and surgery is considered controversial. 34 The chemosensitive nature of the disease generally limits the role of surgery to biopsy. Adjuvant whole brain radiotherapy (WBRT) is occasionally considered to increase progression-free survival. However, the benefits may be limited when compared to the risk of morbidity. 34 WBRT is currently discouraged, and it is only used in patients with contraindications for other therapies. 35 In immunocompromised PCNSL, the first step to treatment is to reconstitute the immune system, whether through antiretroviral therapy in the AIDS setting, or tapering of immunosuppressive medications. This, in combination with high-dose methotrexate or rituximab, can achieve complete remission. 32
Consolidation therapy aims to maximize the elimination of microscopic residual disease, which leads to improved overall response rates (ORR) to induction therapy. Consolidation regimes include chemotherapy with stem cell rescue or high-dose cytarabine ± etoposide. Reduced dose WBRT can also be used as a consolidation regime, but it is still under ongoing investigation due to the high rate of neurotoxicity. 36
The majority of patients relapse within the first 2 years despite a 70% to 90% response rate to high-dose MTX-based induction therapy. 37 Patients have a median overall survival of 40 to 70 months and 2 months following relapse in patients without any salvage therapy. 38 Treatment of relapsed or refractory disease is generally considered for a rechallenge with high-dose MTX or an alternative aggressive systemic therapy, followed by consolidation therapy. 39
Biology characterization of PCNSL through molecular profiling has led to the development of novel targeted therapies. These new targeted drugs (ibrutinib, lenalidomide, and pomalidomide) aim to inhibit key signalling pathways for lymphoma cells, activate the viral lytic cycle that is susceptible to antiviral therapies, boost the immune response of patients, or have an indirect immune modulation of the tumour microenvironment. 40 Lastly, despite the lack of clinical trials, anti-CD-19-directed chimeric antigen receptor T-cell (CAR T) therapy appears to be an effective and safe treatment option for primary relapsed/refractory PCNSL. 41
Conclusion
The clinical and radiological presentation of patients with immune-deficiency related PCNSL can mimic those with other pathologies such as metastatic disease and cerebral abscesses. A detailed clinical history might reveal an immunodeficiency state such as organ transplant or autoimmune disorder. Advanced MRI techniques such as perfusion imaging can be highly suggestive of PCNSL when a low uncorrected rCBV and high K2 values are present at the enhancing rim of the lesion. This could help the neurosurgeon decide whether to withhold steroid treatment and to proceed with biopsy of the lesion. The pathophysiology of PCNSL in the immunodeficient population usually relates to EBV infection or reactivation, with EBV positivity highly suggestive on pathology. Modern therapies allow for prolonged survival, provided that the host’s immune system can be reconstituted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
