Abstract
This is a visual representation of the abstract.
Introduction
Thoracic computed tomography (CT) is a common diagnostic test most often performed in the evaluation of patients with signs or symptoms suggestive of cardiopulmonary disease. Thoracic CTs frequently reveal clinically significant incidental findings, including incidental pulmonary nodules (IPN), which are found in approximately one-third of all thoracic CT scans. 1 In a study from the United States (US) between 2006 and 2012, the annual rate of thoracic CT increased from 15.4 to 20.7 per 1000 person-years, and the rate of identified nodules increased from 3.9 to 6.6 per 1000 person-years. It was estimated that more than 4.8 million Americans had at least one thoracic CT scan and 1.57 million had a nodule identified, among whom, 63 000 received a new lung cancer diagnosis within 2 years. 1 Similar increases in overall CT use have been documented in Canada 2 and a recent North American study of medical imaging rates demonstrated a 5% to 15% increase in per capita chest CT in Ontario every year between 2000 and 2016. 3
The literature reports variability in the malignancy risk of IPNs. In a prospective observational study of an IPN program in Mississippi (US), 5% of patients were found to have lung cancer. 4 Meanwhile, a review of patients with indeterminate pulmonary nodules measuring 7 to 20 mm in diameter reported the risk of malignancy to vary from 5% to 60%. 5 Regardless, few patients with IPNs receive required follow-up.6,7 Preliminary results from the Prospective Watch the Spot study in the US in patients with IPNs ≤15 mm demonstrated that 30% of patients eventually diagnosed with lung cancer had stage migration to >cT1b (>20 mm) by the time the cancer was diagnosed due to heterogenous nodule management and imperfect adherence to follow-up recommendations.8,9 Appropriate management of IPNs is crucial, 10 given the association between early lung cancer diagnosis and improved survival rates.11-14 This underscores the importance of standardized, equitable, and resource-responsible processes to manage patients with IPNs. 15
We recently explored the current state of IPN management in Canada through a literature review and interviews with 20 multidisciplinary experts from specialties involved in IPN detection and assessment across 5 provinces in Canada. 16 This study highlighted variability in radiology reporting of IPN, inconsistent management of IPN, and poor communication between healthcare providers and between healthcare providers and patients. 16 In the current study, we use multidisciplinary expert consensus to make actionable recommendations that could improve IPN management in Canada.
Methods
A modified Delphi technique 17 (Figure 1) was performed to achieve expert consensus on recommendations for optimal IPN management in Canada in line with the Ethical Delphi Manual 18 and informed by the Guidance on Conducting and REporting DElphi Studies, CREDES (Supplemental Appendix 1). 19 The Delphi technique is a widely used group communication process for gathering the views of experts on a specific topic and is commonly used in healthcare research.19,20

The Delphi study process.
Expert Working Group and Panellists
Experts from Canada with experience in lung nodule management were recruited to form a multidisciplinary expert working group (EWG) in September 2022 to discuss opportunities for improvement in IPN management in Canada. Targeted participants included radiologists, respirologists, thoracic surgeons, epidemiologists, medical oncologists, radiation oncologists, and primary care physicians identified by professional networks. Snowball sampling was used for recruitment, first with leaders in the Canadian Association of Thoracic Surgeons, the Canadian Society of Thoracic Radiology, the Canadian Thoracic Society, and Lung Cancer Canada. Panellists from the EWG were contacted via email communication by one researcher. A 2-hour EWG discussion was held based on a survey that was previously circulated to experts to understand the priority topics and key recommendations (Supplemental Appendix 2). Following the discussion, the EWG agreed to participate in a Delphi panel to refine and establish recommendations on IPN management in Canada.
Questionnaire Development, Data Collection, and Analysis
The Delphi questionnaire consisted of 10 questions with multiple statements on priority areas identified in our recent literature review (Supplemental Appendix 2). 16 Two content experts (C.F. and D.M.) with experience with Delphi panels pilot tested the questionnaire. Panellists received the questionnaire via email and independently completed each round. Rounds 1, 2, and 3 were completed during a 12-day window in December 2022, January 2023, and February 2023, respectively. The questionnaire used a 5-point Likert scale (1 = extremely agree, 5 = extremely disagree) to increase the response rate and quality. 21 Panellists were encouraged to suggest additional recommendations for consideration, which were incorporated as new statements in subsequent rounds (Supplemental Appendix 2).
Consensus was achieved if a statement had an ≥80% response rate among panellists and the range (ie, the difference between highest and lowest ratings) was either 0 (strong consensus) or 1 (consensus). The median for each statement was also assessed to understand the level of agreement or importance. Panellists were asked to rank the level of importance only for certain questions to prioritize specific aspects that are particularly relevant to IPN management, minimizing potential survey fatigue and streamlining data analysis.
In the first round, statements based on previously identified opportunities for improvement in IPN management 16 were presented to panellists to assess their agreement. In the second round, results from the first round were presented for each statement, and panellists were asked to score their level of agreement. For the third round, only questions where more than half of the statements did not achieve consensus were included (Supplemental Appendix 2). After the third round, the anonymized results were circulated to all panellists for review. To finalize the topics of priority from the Delphi survey, the emergent themes were presented and validated by the panellists.
Data were reviewed, and in cases where a panellist did not respond to a survey round, they were excluded from the analysis. Following review of data, outliers were removed from the dataset to reduce the influence of extreme values on the analysis. This is an accepted technique in the Delphi process to minimize bias that would disproportionately affect the group’s consensus or divergence on a given issue.22,23 For each statement, outliers were identified through the sorting method where the data set values were sorted in ascending order, the distance of each response from the central tendency of the data was calculated, and up to 2 responses were eliminated.
Following data analysis, experts participated in a 1-hour debriefing meeting on topics where there was no consensus. While this meeting did not impact the Delphi process results, the discussion from the meeting helped to frame the reasons for which there was a lack of consensus. Findings discussed herein are recommendations that had agreement with strong consensus (range in ratings of 0) or no consensus (range in ratings of 2, 3, or 4).
Ethics Approval
Ethics approval was not required for participation in the Delphi panel process and completion of surveys.
Results
Delphi Survey
Twelve experts were invited to participate in the panel, of which 11 (91.7%) agreed to participate. Eight (66.7%) of these panellists participated in the post-survey debriefing meeting. Panellists were from 4 provinces (British Columbia [n = 1], Nova Scotia [n = 2], Ontario [n = 5], Quebec [n = 3]) and included 3 radiologists, 3 respirologists, 2 thoracic surgeons, 1 primary care physician, 1 medical oncologist, and 1 epidemiologist.
In round 1 of the survey, 10/11 (90.9%) panellists participated; in rounds 2 and 3, 11/11 (100%) participated. Consensus was achieved for 183/217 (84.3%) statements included in the Delphi questionnaire (Figure 2, Table 1). Full results are provided in Supplemental Appendix 2.
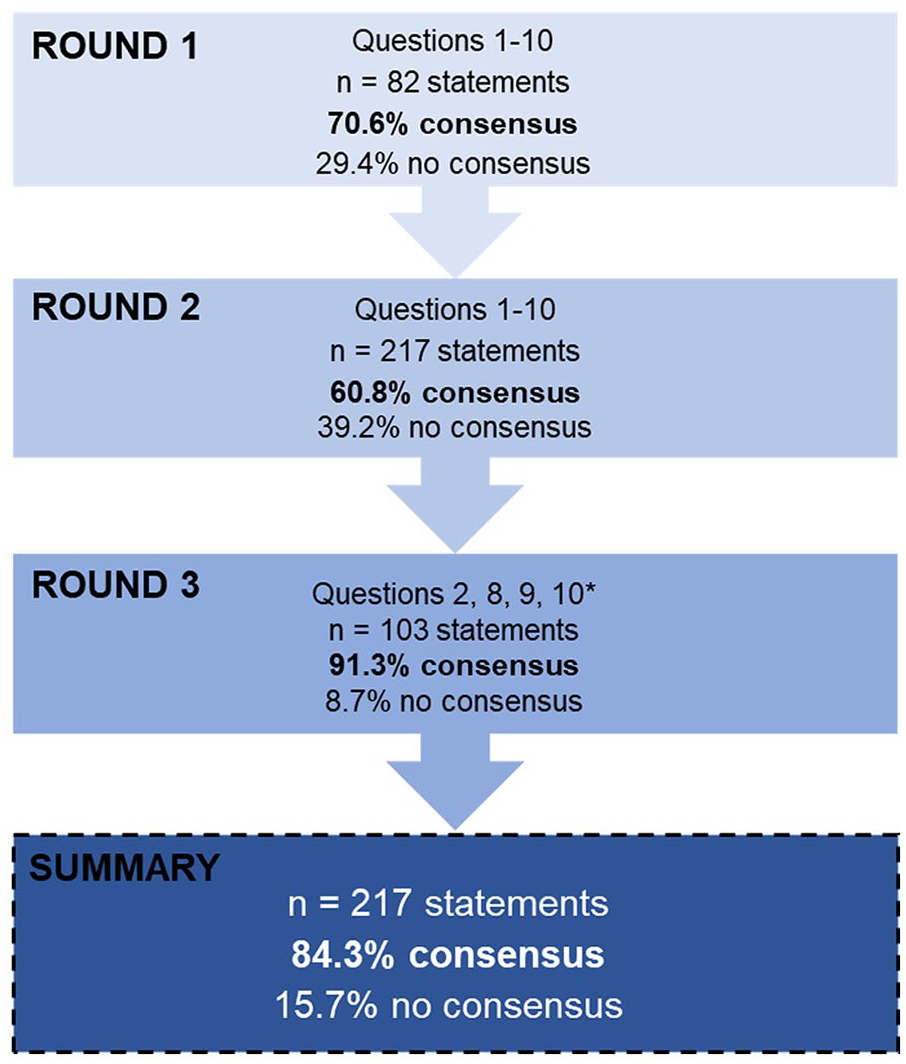
Flow diagram outlining the 3 survey rounds of the Delphi study.
Summary of Delphi Process Results.
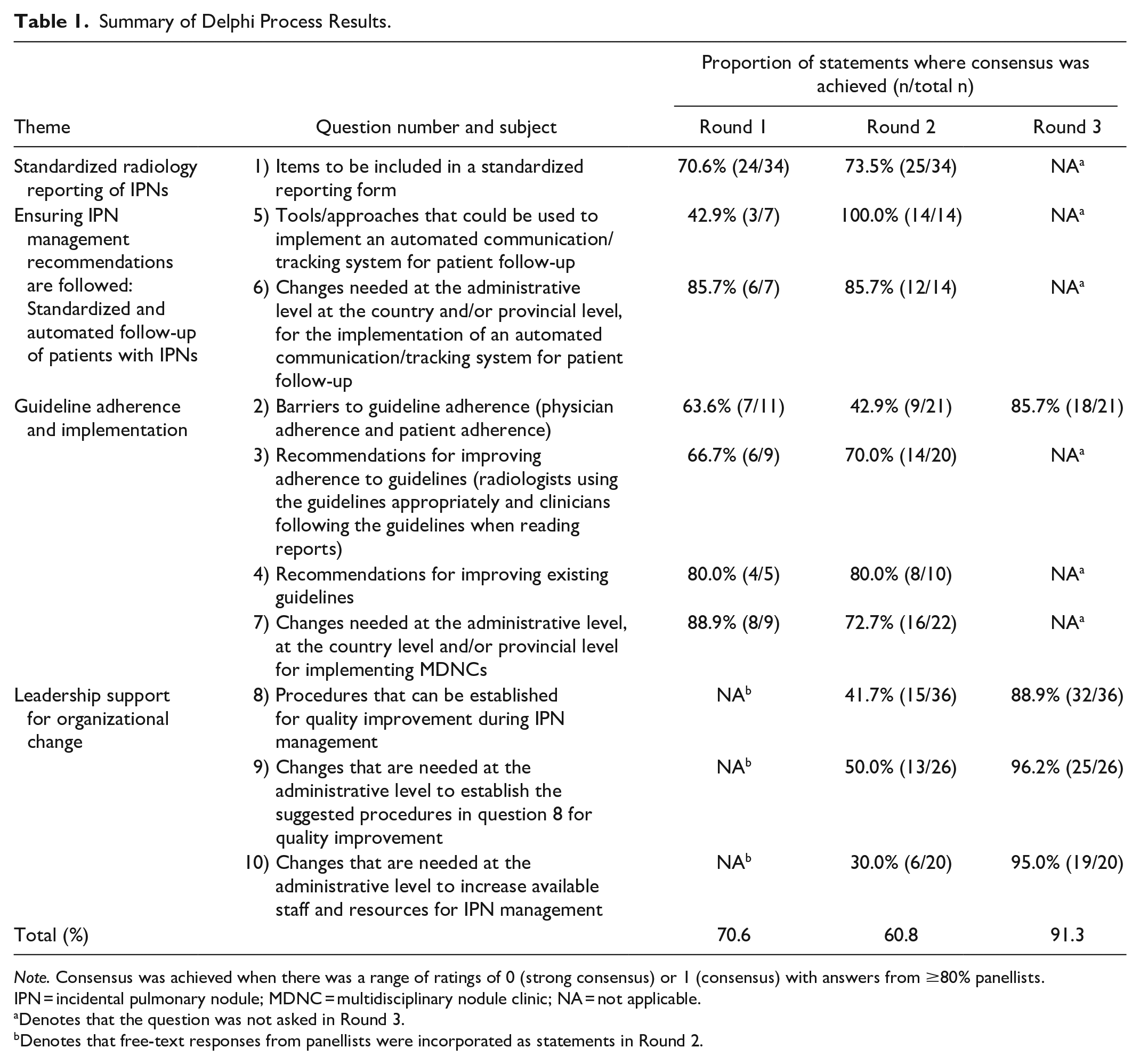
Denotes that the question was not asked in Round 3.
Denotes that free-text responses from panellists were incorporated as statements in Round 2.
Standardized Radiology Reporting of IPNs
The Likert scores and the level of consensus regarding the radiology report are presented in Figure 3. For all nodule sizes, 11/11 (100%) panellists strongly agreed that a summary of findings and follow-up recommendations should be included in the CT report. There was also consensus that reporting requirements include change in nodule diameter and, for nodules over 8 mm, the presence of suspicious features. During the Delphi panel, it was noted that radiology reports should be simple and user-friendly, as in structured lung cancer screening programs.
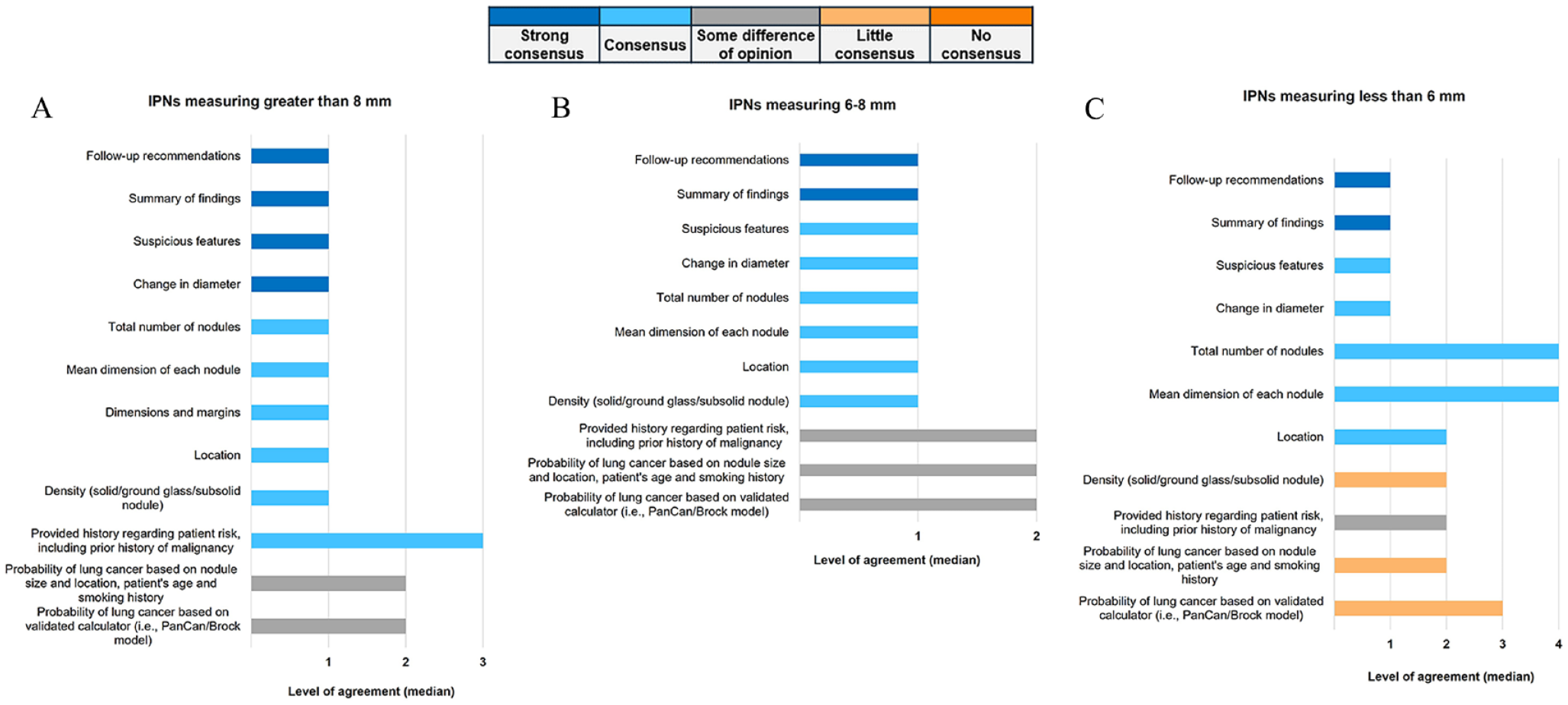
Items that should be included in standardized radiology reporting form when reporting IPNs measuring: (A) >8 mm, (B) 6-8 mm, and (C) <6 mm.
For CT scans with smaller-sized nodules, there was less consensus on additional items to include in the radiology report, such as patient risk assessment for nodules 8 mm or less, or the density of nodules under 6 mm. During the Delphi panel, 2 radiologists expressed difficulties in accurately assessing the density of small nodules (under 6 mm), therefore limiting the usefulness of including this level of detail.
For all nodule sizes, there was also no consensus on the inclusion of the probability of lung cancer, either in broad categories or numerically based on a calculator such as the McWilliams/PanCan lung nodule risk calculator, 24 in the standardized radiology report. This was explored in the debriefing session where it was noted that patient demographics (eg, smoking history and prior cancer history) influence the likelihood of malignancy but that this information is often not known to the reporting radiologists, further limiting the use of validated risk calculators by radiologists. It was also noted that the likelihood of malignancy is not the only variable affecting the management strategy.
Ensuring IPN Management Recommendations Are Followed: Standardized and Automated Follow-Up of Patients With IPNs
All panellists (11/11, 100%) strongly agreed on the importance of 2 key elements for patient follow-up and communication; first, they agreed that imaging departments would ideally automatically book follow-up imaging appointments or nodule clinic appointments. Second, they agreed on implementing a centralized alert system25,26 with closed-loop communication features (ie, ensuring that information is conveyed, acknowledged, and correctly confirmed among those providing care; Figure 4). Panellists acknowledged various barriers to these quality initiatives in the subsequent debrief.
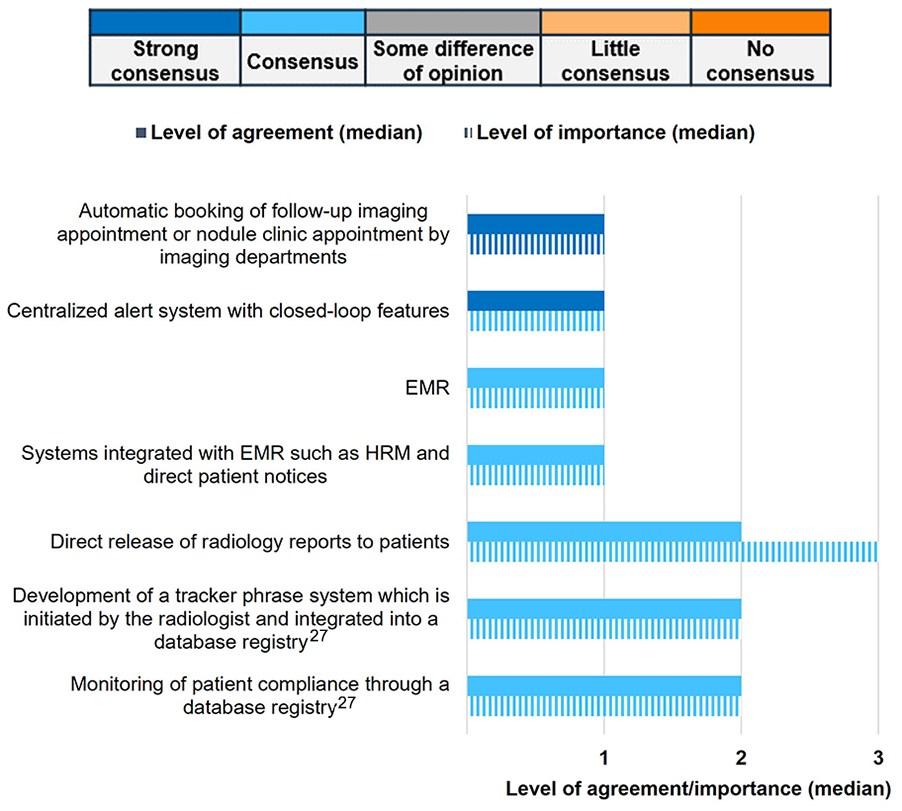
Tools/approaches that could be used to implement an automated communication/tracking system for patient follow-up.
Panellists universally (11/11, 100%) accepted 2 factors affecting patient follow-up (Figure S1B). First, they recognized that patients not being informed of the presence of an IPN was a barrier. Second, they acknowledged that direct release of radiology reports to patients could be a potential tool used to implement an automated communication system for patient follow-up (Figure 4). During the Delphi panel discussion, panellists noted the potential ethical and logistical concerns of direct report release to patients if support mechanisms are not in place. The panellists highlighted that reports should include a description of the findings in simple language that is clear and easily understandable to healthcare providers regardless of their specialty. Reports should also have specific guidance to health care providers regarding the most appropriate next step to manage the detected lung nodules (eg, no further imaging follow-up, repeat low-dose CT, or referral to specialized lung nodule clinic for diagnostic work up).
Regarding the use of virtual care for the management of patients with nodules or the ability to provide advice remotely, consensus was not reached. During the debriefing meeting, it was noted that disagreement might stem from differing opinions on whether patients with IPNs can be effectively managed virtually or if in-person visits are always necessary. Additionally, it was suggested that the diversity in answers reflects the heterogeneity in both nodule and patient characteristics.
Guideline Adherence and Implementation
Panellists unanimously accepted (11/11, 100%) that evidence-based guidelines (eg, IPN consensus guidelines produced by the Fleischner Society 28 or the British Thoracic Society 29 ) are important. They also universally agreed (11/11, 100%) on the significance of standardized reporting and standard terminology, (10/10, 100%; Figure 5A and Figure S2) such as is described in the Fleischner Society IPN Guidelines (FSG) 28 and the Fleischner Society Glossary. 30 However, there was little consensus regarding the establishment of guideline adherence as a requirement for radiologist reimbursement or financial incentives for providing guideline-concordant care. In the Delphi panel, panellists noted that radiologists should not always strictly adhere to guidelines, as there may be situations where a more nuanced approach is most appropriate.
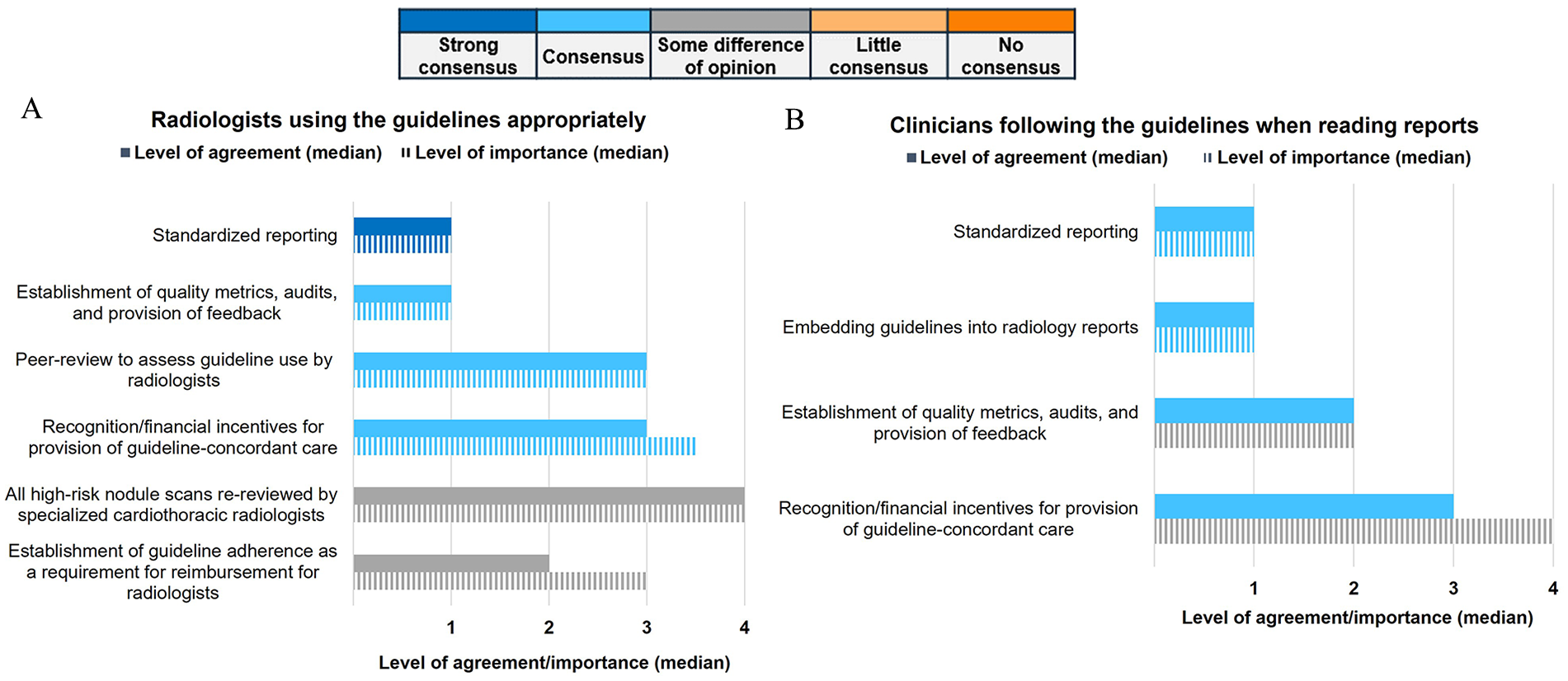
Recommendations for improving adherence to guidelines for (A) radiologists using the guidelines appropriately and (B) clinicians following the guidelines when reading reports.
In the debriefing meeting, panellists discussed barriers to physician adherence to guidelines. It was expressed that guidelines can be intricate and lack the necessary decision aids to support clinicians in their application. Panellists expressed that radiology reports often do not provide guidance on next steps. The expert panel echoed this concern, suggesting the need for specification and guidance on which physician is responsible for applying the guidelines, such as the radiologist or the ordering physician. Moreover, the panellists advocated for radiological data to be synoptically reported and for patients to obtain direct referrals to expert IPN management from radiology rather than primary care alone, to mitigate the inconsistent actioning of report results.
In the debriefing session, panellists also discussed having all high-risk nodule scans re-reviewed by specialized cardiothoracic radiologists, as this did not achieve consensus during the Delphi process. While the panellists expressed enthusiasm for the concept, it was seen as impractical. Conversely, a respirologist shared insights on an initiative aimed at enhancing nodule management, which involved implementing a consistent and standardized review process with cardiothoracic radiologists. The panellists acknowledged that this approach might not be universally feasible, which may have contributed to the observed lack of consensus. It was noted that in some Canadian centres, second opinions from subspecialist thoracic radiologists are routinely obtained for patients referred to a specialist for nodule investigation.
There was no consensus that patient follow-up, in terms of patients missing appointments, was a barrier to patient adherence (Figure S1B). In the debriefing session, panellists suggested that the differing experiences may have contributed to divergent responses in the Delphi panel. For example, a radiologist may not be frustrated about a patient missing an appointment, especially if they are aware that a nodule is not of high concern. However, a primary care physician may feel stressed by the patient’s absence, or a respirologist may be frustrated if the patient misses their follow-up CT appointment.
Leadership Support for Organizational Change
Panellists (11/11, 100%) strongly agreed that leadership support for organizational change is needed at the administrative level to implement an automated system to improve IPN management. Panellists also ranked this support as an extremely important factor for improvement (Figure 6). All panellists (11/11, 100%) strongly agreed on the necessity of establishing standardized protocols, recommendations, as well as patient and referral pathways for quality improvement in IPN management at the provincial and regional levels (Figure S4B and C). At all governmental levels (ie, regional, provincial, and national), panellists agreed that administrative (10/10, 100%) and financial support (9/10, 90%) are necessary for quality improvement (Figure S5A-C). At the provincial and regional levels, panellists (10/10, 100%) universally agreed that availability of data and access to all prior imaging is needed for quality improvement (Figure S5B and C).
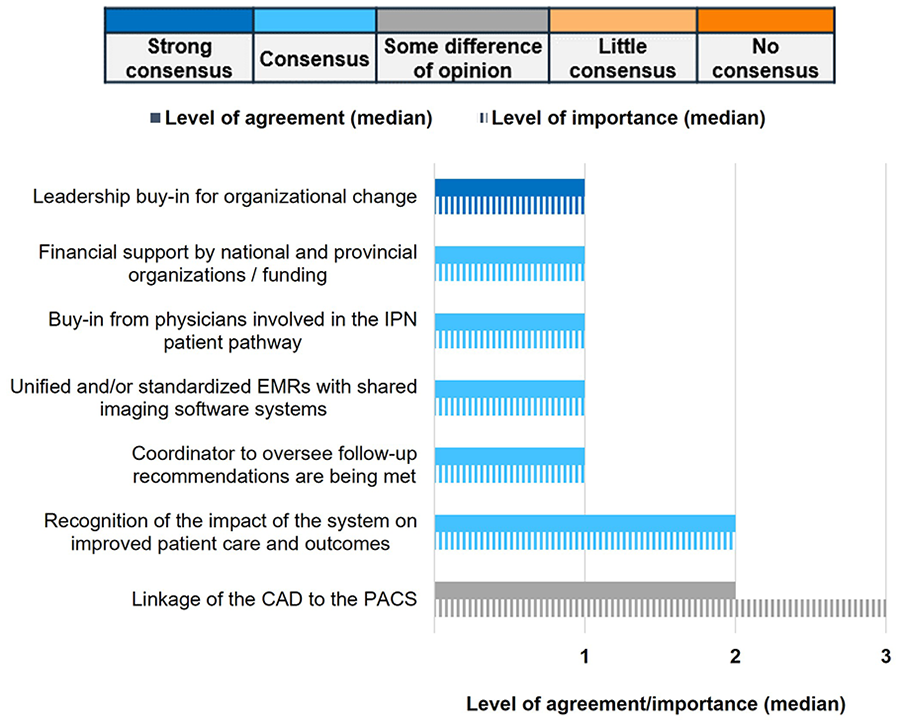
Changes needed at the administrative level for the implementation of an automated communication/tracking system for patient follow-up.
Across all levels of government, panellists concurred that funding and awareness of IPN management should be established at the administrative level to increase staff and resources (10/11, 90.9%; Figure S6A-C). All panellists (11/11, 100%) also strongly agreed on the need for leadership support for organizational change at the administrative level to implement multidisciplinary nodule clinics (MDNCs) and agreed with the importance of this recommendation (Figure S3). Panellists (10/11, 90.9%) also agreed that training and mentoring of physicians should be established at all levels (Figure S6A-C), although no specific examples were suggested.
A recommendation that did not reach consensus was the need for funding of tools, such as computer-aided detection (CAD), at the regional level (Figure S6C). Similarly, there was no consensus on whether CAD should be integrated with the picture archiving and communication system (PACS). There was also no consensus on the importance of this topic (Figure 6). During the Delphi panel, one panellist noted that CAD is often already linked to PACS. It was also noted that optimal IPN management requires the ability to compare current imaging in relation to previous scans, as monitoring changes in nodules over time is key to evaluating the risk of malignancy. Some provinces have implemented province-wide imaging archives facilitating comparison while, in other centres, comparison is difficult due to incompatible systems within a single province (or even city) and high rates of private imaging, with imaging not included in the provincial archive. Additionally, it is important to inform health authorities about potential patient safety and inefficiency concerns arising from barriers in comparing imaging performed on the same patient at different institutions. As such, information technology solutions to satisfy this need are necessary. However, panellists expressed a sense of overall powerlessness to implement the required improvements for organizational change. They emphasized the need for assistance from hospital administration and health authorities to enhance IPN management through, for example, facilitating communication and data sharing between different PACS systems.
Discussion
In this study, we used a modified Delphi process with a panel of experts in pulmonary nodule management to identify recommendations for improving IPN management in Canada. The main recommendations that emerged from this expert consensus included standardized radiology reporting of IPNs, standardized and automated follow-up of patients with IPNs, guideline adherence and implementation, and leadership support for organizational change.
While the expert panel overwhelmingly agreed that including a summary of IPN findings and follow-up recommendations in standardized radiology reports is essential, the level of consensus regarding the need for specific nodule details varied by nodule size. At all nodule sizes, there was consensus that change in size should be reported and, for nodules measuring at least 8 mm, nodule density and suspicious features should be reported. Panellists were concerned that including specific details for smaller nodules might lead to unwarranted follow-up investigations or interventions, especially given that most nodules under 6 mm are benign and do not require further action. 28 It is noteworthy that the experts in our study did not reach consensus on the inclusion of probability estimates of malignancy in standard radiology reports. In the follow-up debriefing meeting, panellists attributed this lack of consensus to differing opinions on its potential impact on the subsequent steps in the management pathway.
Nevertheless, the overall consensus regarding the value of comprehensive information in radiology reports in the form of a summary of findings and follow-up recommendations underscores its importance for facilitating appropriate follow-up care.31,32 In many instances, radiologists note nodules but appropriate follow-up is not performed. 32 As such, there is a clear need for explicit actionable items and recommendations by the radiologist in the report. 31 This can help overcome shortcomings in follow-up and improve monitoring of pulmonary nodules.
The expert panel agreement with the use of consistent terminology (eg, as described by the Fleischner glossary 30 or Fleischner Society IPN Guidelines 28 ) in standardized radiology reporting, aligns with the literature.7,33 Radiology terminology is often susceptible to misunderstandings.14,34-36 Thus, consistent terminology has the potential to minimize misunderstandings and improve guideline use, thus promoting more consistent and accurate IPN management.28,37 Standardized reporting also has the potential to address the over-investigation of IPNs, whereby patients may be subjected to unnecessary diagnostic procedures and interventions. Carter et al 38 demonstrated that decision-oriented reporting systems for lung cancer screening can help minimize false-positive results, and noted that radiology reports should also be structured for IPN management. Similarly, Woloshin et al 37 surveyed clinicians in the US comparing a standard thoracic CT report to a structured thoracic CT report with probability estimates for malignancy and management recommendations on follow-up strategy for patients with IPNs and found that more clinicians followed the correct management strategy with the structured report. Structured radiology reports can also reduce missing information, increase patient follow-up and quality of care, and improve clinicians’ response, as documented in US studies.37,39,40 The literature includes several examples of structured IPN reports.28,37
In addition to standardized reporting, panellists also made recommendations on guideline adherence. Panellists noted that IPN guidelines, such as those produced by the Fleischner Society 28 or the British Thoracic Society, 29 should serve as tools to guide decision-making, rather than rigid mandates. However, they highlighted the lack of relevant patient information provided to radiologists and uncertainty about the next steps as barriers to physician adherence to guidelines and consistent IPN management. Currently, the FSG for IPN management are based on nodule size, require knowledge of the patients underlying lung cancer risk, and are only applicable to patients ≥35 years who are not at risk for metastatic disease or opportunistic infection.28,41
Key recommendations for ensuring patient follow-up included considering automatic booking of follow-up imaging and a centralized alert system within existing IPN programs. Multiple studies have documented that missed and delayed follow-up of IPN is common.7,42-44 Panellists highlighted that enhancing communication systems among physicians and between physicians and patients can address delays in follow-up imaging, which has been documented to have a positive impact in the Canadian health system. For example, Walker et al 45 assessed the impact of an electronic platform whereby primary care providers could send questions along with relevant documentation (eg, laboratory values and imaging reports) to a selected radiology subspecialty. The platform resulted in improved patient management and eliminated unnecessary imaging tests. Similarly, in the US, an electronic communication tool enabling radiologist-generated alerts for IPN follow-up recommendations improved timely IPN follow-up. 46
While panellists recommended automatic booking of follow-up imaging, in some health jurisdictions in Canada without an IPN program, there is no pathway for a radiologist to arrange a repeat CT scan without a repeat referral from a specialist or primary care physician. This is partially due to concerns about conflicts of interest and income-generating implications under fee-for-service systems. 47 This highlights that implementing automatic booking for IPN requires collaboration with health authorities to design a process that maintains patient safety and meets health care legislation requirements.
Panellists also highlighted that the direct release of radiology reports to patients would be a beneficial tool to improve patient-provider communication. The direct release of radiology reports could encourage patients to actively engage in their healthcare, advocate for themselves, and promote shared decision-making.48-50 It has also been shown to improve patient outcomes. 49 In one survey that assessed the accessibility and importance of portal-released radiology reports, 88% of patients reported that the ability to access reports was important. 51 Additionally, 88% of referring physicians reported that releasing reports to patients was useful. 51 This approach may further streamline the development of structured and standardized reports, making them more readily accessible to patients and less prone to misinterpretation. However, panellists also noted the importance of ensuring that there are appropriate supports and processes in place for patients who have questions about their imaging findings to ensure that patients are appropriately supported and that providers are not overwhelmed.
Panellists recognized the need for administrative support as a critical component of quality improvement initiatives and to increase staff and resources at all administrative levels. Specifically, they recommended leadership participation and organizational change for implementing MDNCs. Studies in Ontario demonstrated the benefits of rapid assessment clinics and lung cancer multidisciplinary clinics (MDC), which streamline the care of patients with a new lung cancer diagnosis to facilitate concurrent oncology consultation, and improve timeliness of care and clinical outcomes. 52 These models also demonstrated improved patient communication, physician collaboration, and better quality of care. 53 This highlights that support from leadership with organizational and administrative change in the Canadian healthcare system can lead to improved outcomes.
Several statements did not reach consensus, primarily due to concerns regarding the feasibility of implementing certain changes. These include standardized national referral pathways, the development of guidelines by national organizations, and establishing a uniform PACS system. This can be partially explained by the decentralized nature of healthcare in Canada, which is predominantly delivered through provincial jurisdictions. Consequently, changes at the national level may not be feasible without coordination and agreement among all provinces and territories.
Limitations
In terms of study limitations, we first note that the geographical representation was somewhat limited. Second, there was a limited number of panellists from primary care. Third, some of the panellists’ recommendations may be difficult to implement, such as the automatic booking of follow-up imaging. Fourth, the Delphi process relies on expert opinion. Recommendations have not yet been assessed for their feasibility within the Canadian healthcare system, where much of healthcare is delivered outside of an academic centre, in rural areas, and where specialist access is limited. Moreover, due to the nature of the Delphi panel process, experts were not able to engage in direct discussion and debate regarding their opinions. As a result, it is possible that some statements that did not initially receive consensus may have eventually achieved consensus if given the opportunity for further discussion.
The debriefing session following the Delphi panel allowed a better understanding of disagreement. The session offered insights into the differing ways the statements in the Delphi panel were interpreted and the potential implications associated with those interpretations. This helped clarify the reasons behind the varied responses from panellists, which were often influenced by their clinical backgrounds and expertise in different medical specialties. Therefore, it is important to interpret recommendations with caution as they may warrant further evaluation and validation.
Conclusion
Based on this Delphi study, key recommendations for improving IPN management in Canada include standardized radiology reporting of IPNs, standardized and automated patient follow-up, guideline adherence and implementation, and leadership support for organizational change. It underscored the importance of clear communication, imaging reports that provide specific guidance, automatic booking of follow-up imaging appointments, and administrative backing in improving the quality of care for patients with IPNs. Future research should explore how to appropriately implement these recommendations and assess their effectiveness in clinical practice.
Supplemental Material
sj-docx-1-caj-10.1177_08465371241257910 – Supplemental material for Recommendations to Improve Management of Incidental Pulmonary Nodules in Canada: Expert Panel Consensus
Supplemental material, sj-docx-1-caj-10.1177_08465371241257910 for Recommendations to Improve Management of Incidental Pulmonary Nodules in Canada: Expert Panel Consensus by Geneviève C. Digby, Stephen Lam, Martin C. Tammemägi, Christian Finley, Carole Dennie, Stephanie Snow, Jeffrey Habert, Jana Taylor, Anne V. Gonzalez, Jonathan Spicer, Jyoti Sahota, Danielle Guy, Paola Marino and Daria Manos in Canadian Association of Radiologists Journal
Supplemental Material
sj-pdf-2-caj-10.1177_08465371241257910 – Supplemental material for Recommendations to Improve Management of Incidental Pulmonary Nodules in Canada: Expert Panel Consensus
Supplemental material, sj-pdf-2-caj-10.1177_08465371241257910 for Recommendations to Improve Management of Incidental Pulmonary Nodules in Canada: Expert Panel Consensus by Geneviève C. Digby, Stephen Lam, Martin C. Tammemägi, Christian Finley, Carole Dennie, Stephanie Snow, Jeffrey Habert, Jana Taylor, Anne V. Gonzalez, Jonathan Spicer, Jyoti Sahota, Danielle Guy, Paola Marino and Daria Manos in Canadian Association of Radiologists Journal
Footnotes
Acknowledgements
All authors contributed significantly to all aspects of the paper: conceptualization; research; data analysis and interpretation; manuscript writing, review, and editing. Victor Genestier, employee from Amaris Consulting, provided valuable manuscript preparation assistance and editing review. Medical writing and editorial assistance were funded by AstraZeneca.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.C.D., S.L., M.C.T., C.F., C.D., S.S., J.H., J.T., A.V.G., J.S., and D.M. received unrestricted support for meetings, planning and data collection, manuscript writing and editing, participating in the Delphi panel, and consulting fees from AstraZeneca. G.C.D. received research funding from MaRS/Merck & Co., Inc. and from Pfizer Inc.; honoraria from Merck & Co., Inc. and GSK Medical; and has received grants outside the submitted work paid directly to Queen’s University from the Ontario Lung Association and Ontario Thoracic Society. G.C.D. serves as the Canadian Thoracic Society Choosing Wisely Canada Working Group chair. J.S. received consulting fees from AstraZeneca to support the conduction of this study and was an employee of Amaris Consulting at the time of this study. D.G. and P.M. received consulting fees from AstraZeneca to support the conduction of this study and are employees of Amaris Consulting. The authors have no other conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was fully funded by AstraZeneca.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
