Abstract
Due to the major improvements in the hardware and image reconstruction algorithms, positron emission tomography/magnetic resonance imaging (PET/MR) is now a reliable state-of-the-art hybrid modality in medical practice. Currently, it can provide a broad range of advantages in preclinical and clinical imaging compared to single-modality imaging. In the second part of this review, we discussed the further clinical applications of PET/MR. In the chest, PET/MR has particular potential in the oncology setting, especially when utilizing ultrashort/zero echo time MR sequences. Furthermore, cardiac PET/MR can provide reliable information in evaluating myocardial inflammation, cardiac amyloidosis, myocardial perfusion, myocardial viability, atherosclerotic plaque, and cardiac masses. In gastrointestinal and hepato-pancreato-biliary malignancies, PET/MR is able to precisely detect metastases to the liver, being superior over the other imaging modalities. In genitourinary and gynaecology applications, PET/MR is a comprehensive diagnostic method, especially in prostate, endometrial, and cervical cancers. Its simultaneous acquisition has been shown to outperform other imaging techniques for the detection of pelvic nodal metastases and is also a reliable modality in radiation planning. Lastly, in haematologic malignancies, PET/MR can significantly enhance lymphoma diagnosis, particularly in detecting extra-nodal involvement. It can also comprehensively assess treatment-induced changes. Furthermore, PET/MR may soon become a routine in multiple myeloma management, being a one-stop shop for evaluating bone, bone marrow, and soft tissues.
This is a visual representation of the abstract.
Introduction
In the previous part of our review, 1 we showed that positron emission tomography/magnetic resonance imaging (PET/MR) is a hybrid imaging modality that provides the field of medical practice with accurate information, having unique advantages that make it stand out from other imaging techniques. The hardware and image reconstruction advancements have helped PET/MR to build on a strong foundation for acquisition and quantification, whereas preclinical PET/MRs have facilitated the development of methods informing clinical PET/MR.
In the first part of this review, the technical basics of PET/MRI, preclinical applications, and clinical applications of PET/MRI in radiation oncology and head and neck imaging were discussed. In radiation medicine, PET/MR could bring the ability to capture unique characteristics for treatment guidance that were otherwise not possible with other single-imaging modalities. It has also been shown to precisely visualize various characteristics of the tumours, such as radio-responsiveness and potential hypoxic regions, and help identify biological sub-volumes that benefit from differential dosing. In head and neck applications, PET/MR was shown to be of value in cancer staging and resectability assessment, being mainly superior in primary or recurrent tumour characterization, as well as locoregional staging. In addition, whole-body PET/MR could provide a one-session assessment in patients with lower neck tumours or lymph node metastases who need whole-body staging due to the high risk of distant metastases. Moreover, PET/MR (particularly multi-parametric PET/MR with diffusion-weighted imaging [DWI]) could significantly help in the diagnosis of residual or recurrent disease, reliably distinguishing them from oedema and fibrosis.
In part II, we will continue to cover the current clinical applications of PET/MR. This part includes PET/MR applications in the chest, cardiac, gastrointestinal, hepatobiliary-pancreatic, genitourinary, gynaecology, and haematology fields.
Chest Applications
PET/MR has potential clinical applications in the chest, particularly in the oncology setting. 2 The ability of MR to provide superior soft tissue resolution can offer increased accuracy in assessing the involvement of structures in the mediastinum and pleura in malignant disease. However, challenges persist, notably in detecting small lung lesions, where PET/MR is inferior to computed tomography (CT) or PET/CT due to factors like cardiac and respiratory motion artefact, low proton density in the air-filled lungs and fast signal decay.3,4
Studies highlight a common theme of PET/MR’s struggle in detecting non-avid sub-centimetre nodules, with detection rates decreasing as nodule size decreases. In 126 patients with primary abdominal malignancy, Biondetti et al demonstrated a per-patient sensitivity of 28% and per-nodule sensitivity of 12%. 5 Raad et al identified 89 nodules (2 mm-1 cm) in 43 patients on the CT component of PET/CT not identified on PET/MR, and Sawicki et al identified 42 lung nodules not detected on PET/MR but identified on PET/CT (2-7 mm) in 51 patients with a range of tumour types.6,7
Having said that, while some studies demonstrate improved sensitivity with larger nodules (eg, 70% detection rate >7 mm), there has been a limited reported impact of missed small nodules on PET/MR on clinical management, possibly due to the benign nature of many of the nodules or patient selection bias (often patients included had more advanced stage cancer). 5 Noteworthy in this regard, in the abovementioned study by Raad et al, although the non-avid small nodules were missed by PET/MR, 97% of these nodules did not grow in the follow-up, indicating their benign nature. Also, there is always the clinical discussion in what way <5 mm pulmonary nodules should be clinically acted upon other than surveillance.
Efforts to enhance detection include utilizing standard MR sequences like T2wHASTE and T1 VIBE post-contrast, with variations such as StarVIBE showing potential. 8 Additionally, ultrashort or zero echo time (UTE/ZTE) MR sequences have been explored, yielding promising results in improving detection rates, especially for smaller nodules. This allows high-resolution structural information with improved signal-to-noise and contrast-to-noise ratios due to very short echo time. Burris et al assessed 82 nodules in 8 patients with extra-thoracic malignancies using PET/MR with free breathing UTE and dual-echo 3D GRE lung imaging immediately after PET/CT. 9 The detection rate was significantly higher for UTE compared with dual-echo GRE imaging for nodules at least 4 mm (82% vs 34%) and in nodules without 18F-fluorodeoxyglucose (18F-FDG) uptake (68% vs 22%). Zeng et al evaluated 209 patients with a range of malignancies with 18F-FDG PET/MR using diagnostic Dixon and ZTE under respiratory gating, simultaneously with PET acquisition, comparing with CT chest within 1 month of PET/MR. A total of 227 lung lesions (1.1-30.3 mm) were identified in 113 patients. The detectability score for PET/ZTE was significantly higher than for PET/Dixon for all subgroups based on size, as well as improved differentiating of lung lesions. Detectability was >80% for lung lesions >4 mm and >93% for lesions >6 mm. 10
In lung cancer evaluation, PET/MR shows promise, offering comparable performance to PET/CT in staging, particularly in early-stage disease. Heusch et al showed PET/MR agreed with PET/CT in T staging in 16/16 patients, and there was no statistically significant difference in nodal staging. 11 In a study of 84 patients with non-small cell lung cancer (NSCLC), Kirchner et al showed that T staging was concordant in 38/39 patients and N staging concordant in 83/84 patients. 12 While several studies demonstrate concordance between PET/MR and PET/CT in NSCLC staging, 13 discrepancies exist in some cases, attributed to differences in size assessment and uptake times. Schaarschmidt et al compared the staging of 77 patients with NSCLC using PET/CT and thoracic PET/MR. 14 Differences in staging were present in 35% of patients. However, overall, PET/MR has shown substantial agreement with PET/CT in determining resectability and staging, with potential management implications. 15 The ability of PET/MR to assess metastasis to organs like the brain, liver, and adrenal glands, along with potential prognostic insights and treatment response assessment, enhances its clinical utility.
In summary, while challenges persist in small lung nodule detection, PET/MR offers advancements, particularly with the utilization of UTE/ZTE sequences. In lung cancer assessment, PET/MR demonstrates comparable performance to PET/CT, though standardization and optimization of protocols are essential considerations. 2 The ability of PET/MR to provide detailed anatomical evaluation and assess local disease invasion to pleura and mediastinum, as well as metastases to extrathoracic organs (eg, adrenals), underscores its potential clinical benefits. 16 Ongoing refinement and standardization efforts are crucial to fully realize the clinical utility of thoracic PET/MR.
Cardiac Applications
Cardiac PET/MR applications include the evaluation of myocardial inflammation, cardiac amyloidosis, myocardial perfusion, myocardial viability, atherosclerotic plaque, and cardiac masses (Table 1).
Cardiac PET Tracers.
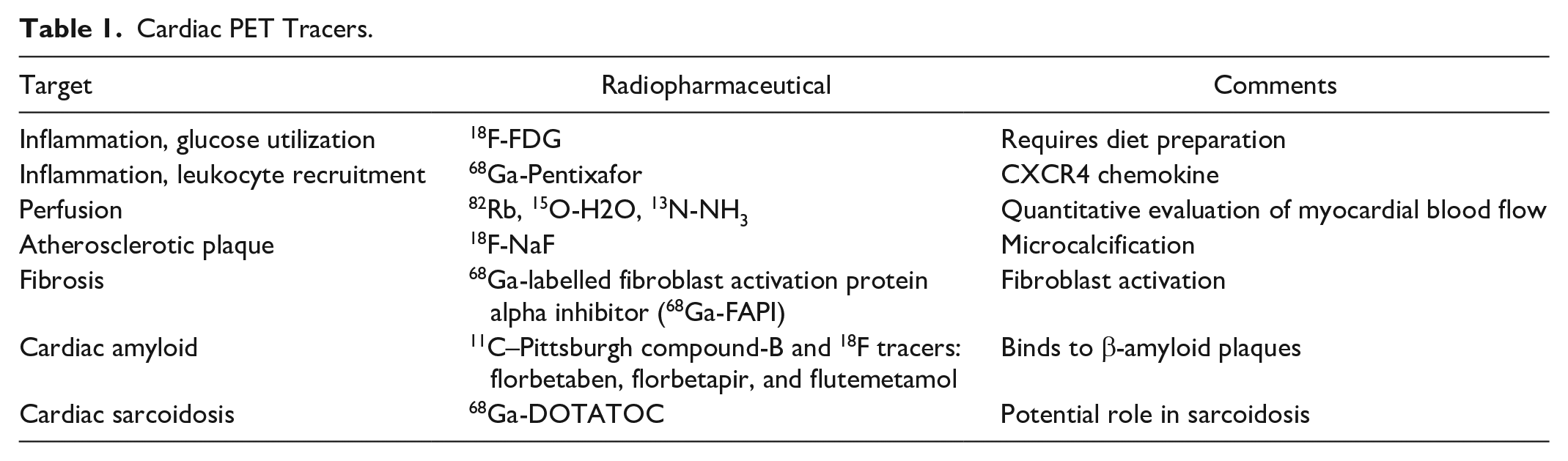
Inflammation
18F-FDG PET/MR can be used to evaluate and quantify metabolic activity in the context of myocardial inflammation (Figure 1) following a high-fat and low-carbohydrate diet to suppress physiologic myocardial glucose uptake by shifting myocardial metabolism from glucose to free fatty acids. 18F-FDG PET and cardiac MR provide complementary information for the diagnosis and prognosis of cardiac sarcoidosis (Figure 2).17-19 Combined PET/MR has a lower radiation dose, shorter total imaging duration, and higher diagnostic test performance for cardiac sarcoidosis compared to standard-of-care evaluation with separate imaging tests. 20 Combined 18F-FDG PET/MR is also useful in the evaluation of non-ischaemic myocardial inflammation in the setting of myocarditis post-COVID-19 21 and after vaccination. 22
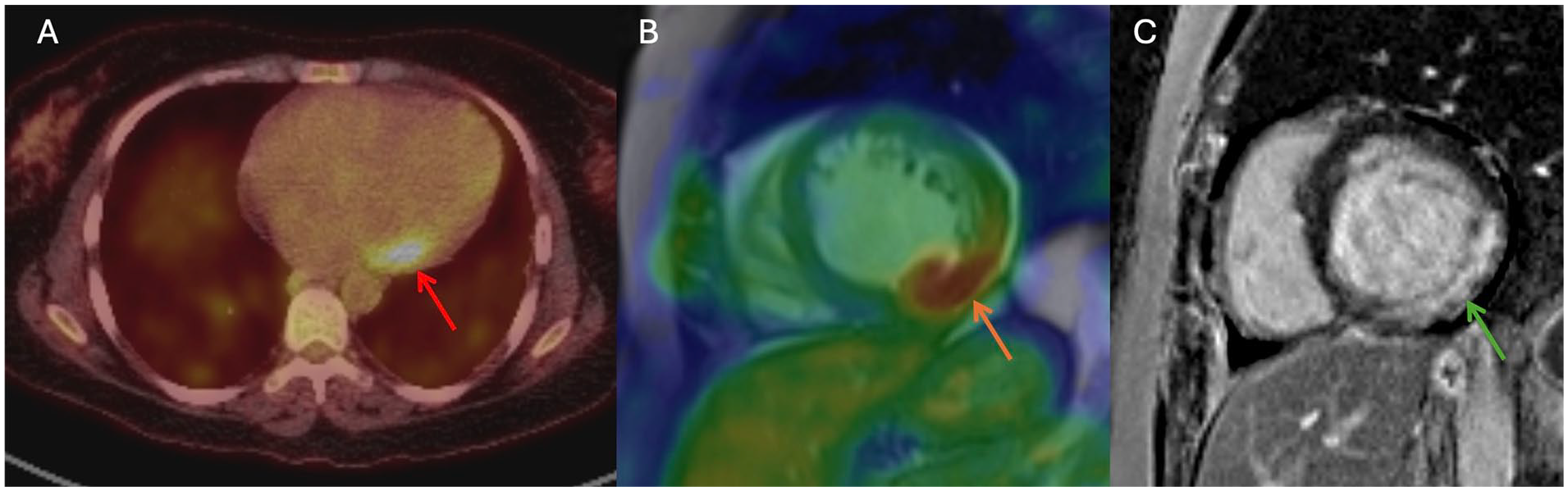
Myocarditis with focal 18F-FDG uptake on PET/CT at the basal lateral wall (red arrow, A), co-localizing 18F-FDG uptake and late gadolinium enhancement (LGE; orange arrow, B) on combined PET/MR (LGE, green arrow, C).
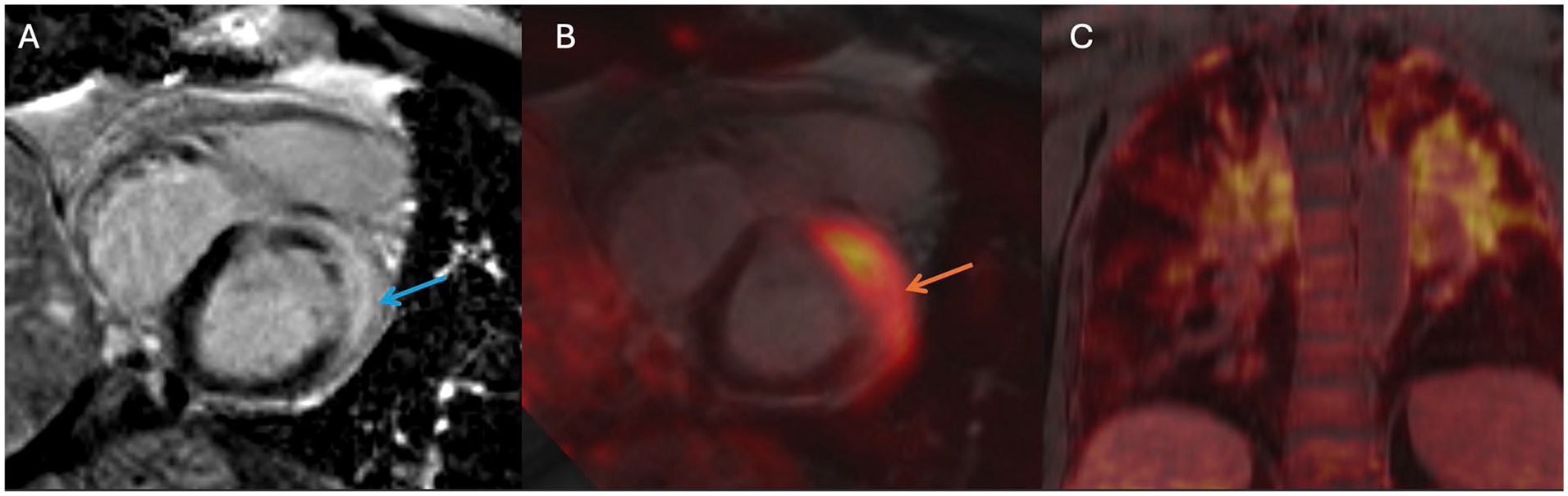
Cardiac sarcoidosis with transmural late gadolinium enhancement at the basal anterior and anterolateral wall (blue arrow, A) colocalizing with 18F-FDG uptake (orange arrow, B) on combined PET/MR and extensive 18F-FDG uptake in the lungs (C, in keeping with extra-cardiac sarcoidosis).
Cardiac Amyloid
Cardiac MR has high diagnostic test performance for cardiac amyloidosis but cannot reliably distinguish between transthyretin (TTR) and light chain (AL) amyloid. PET radiotracers that selectively bind to β-amyloid plaques were initially established for the diagnosis of Alzheimer’s disease but have also been explored for imaging of cardiac amyloidosis, including 11C-Pittsburgh B and 18F-florbetapir.23,24 Preliminary PET/MR studies indicate the uptake of 18F-sodium fluoride (18F-NaF) in patients with TTR amyloid correlating to late gadolinium enhancement (LGE) on cardiac MR. 25
Ischaemia and Myocardial Perfusion
Cardiac MR and PET are both used for evaluating stress and rest myocardial perfusion and for quantitative evaluation of myocardial blood flow. 26 Tracers utilized in PET perfusion include 15O-water, 82Rb-RbCl, and 13N-NH3. Combined cardiac PET/MR could combine the strengths of each modality, though clinical adoption has been limited.
Viability
Viability assessment seeks to identify and differentiate infarcted myocardium from viable tissue. For cardiac MR, viability is typically assessed using LGE and described as the percentage of wall thickness involved. For PET, viability is typically evaluated using 18F-FDG following insulin and oral glucose loading to standardize glucose uptake. In this scenario, the absence of 18F-FDG uptake implies infarcted, non-viable tissue. Combined PET/MR evaluation of viability prior to revascularization correlates with regional wall motion after acute myocardial infarction. 27
Fibrosis
Fibrosis is typically evaluated using cardiac MR LGE, native T1w mapping or extra-cellular volume fraction. 28 More recently, there has been growing research on 68Ga-labelled fibroblast activation protein alpha inhibitor (68Ga-FAPI) in the detection of early stages of cardiac remodelling, particularly after myocardial infarction. 29
Atherosclerotic Plaque
Atherosclerotic plaque can be evaluated using MR (including plaque morphology and luminal stenosis) and PET (including 18F-NaF for evaluation of microcalcifications). Combined coronary PET/MR has been reported using 18F-FDG and 18F-NaF. 30 Novel tracers include 68Ga-Pentixafor, which targets the chemokine receptor CXCR4 and has been shown to bind to atherosclerotic plaque and infarcted myocardium. 31
Gastrointestinal and Hepatobiliary-Pancreatic Applications
So far, PET/MR’s capabilities for imaging hepatobiliary-pancreatic (HBP) and gastrointestinal (GI) malignancies remain mostly understudied. However, some of the few results already available underline PET/MR’s potential in these 2 systems.
One of the most researched areas, and very relevant for GI cancers and pancreatic tumours, is PET/MR’s capability for detecting metastatic disease to the liver. While PET/MR’s superiority over PET/CT in this field is mostly MR-related, 32 PET surpasses MR’s capability to detect nodal disease and extrahepatic metastasis, 33 also relevant for these populations. For colorectal cancer (CRC), a recent meta-analysis showed a per-patient sensitivity and specificity of 94% and 89%, respectively. 34 Meanwhile, somatostatin-receptor (SSTR) imaging with PET/MR using hepatobiliary contrast agents was proven superior to contrast-enhanced PET/CT for neuroendocrine tumour (NET) metastases. 35 Also, in the field of hepatic metastases, PET/MR was able to predict responders from non-responders to Yttrium-90, specifically in CRC, when dose volumetry was performed within 72 hours of radioembolization. 36
There are still scarce studies evaluating PET/MR in primary liver tumours like hepatocellular carcinoma (HCC) and cholangiocarcinoma. However, as contrast-enhanced MR is the imaging method of choice for HCC 37 and one of the 2 suggested by the National Cancer Care Network (NCCN) for cholangiocarcinoma, the synergy of PET and MR could be exploited in specific cases (Figures 3 and 4). For example, in HCC, 18F-FDG-avidity was associated with early recurrence after liver transplant. 38 Furthermore, PET/CT showed higher sensitivity than stand-alone CT or MR in early detection of recurrence after radiofrequency ablation. 39 Meanwhile, in cholangiocarcinoma, 18F-FDG PET and MR were found to improve the overall diagnostic accuracy of staging. 40 Further studies leveraging MR’s superior liver diagnostic performance with the various non-18F-FDG radiotracers available (ie, choline, prostate-specific membrane antigen [PSMA], FAPI, or CXCR4) will further define PET/MR’s capabilities in HCC and other liver lesions.
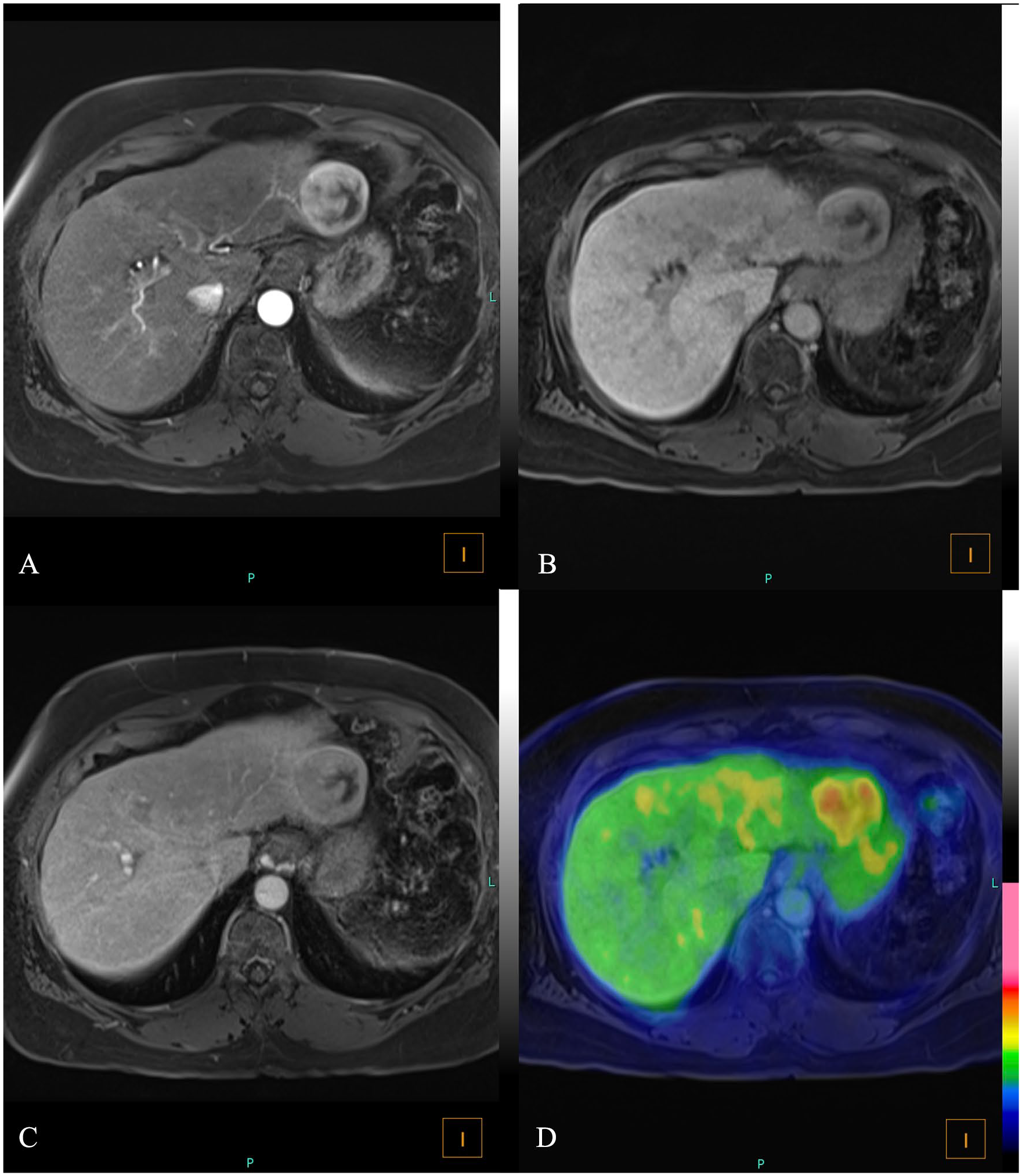
Arterial (A), portal (B), and delayed (C) phases in MR show a large segment 2 liver lesion with delayed capsule enhancement compatible with hepatocellular carcinoma. Fused images (D) show 18F-FDG uptake by the lesion.
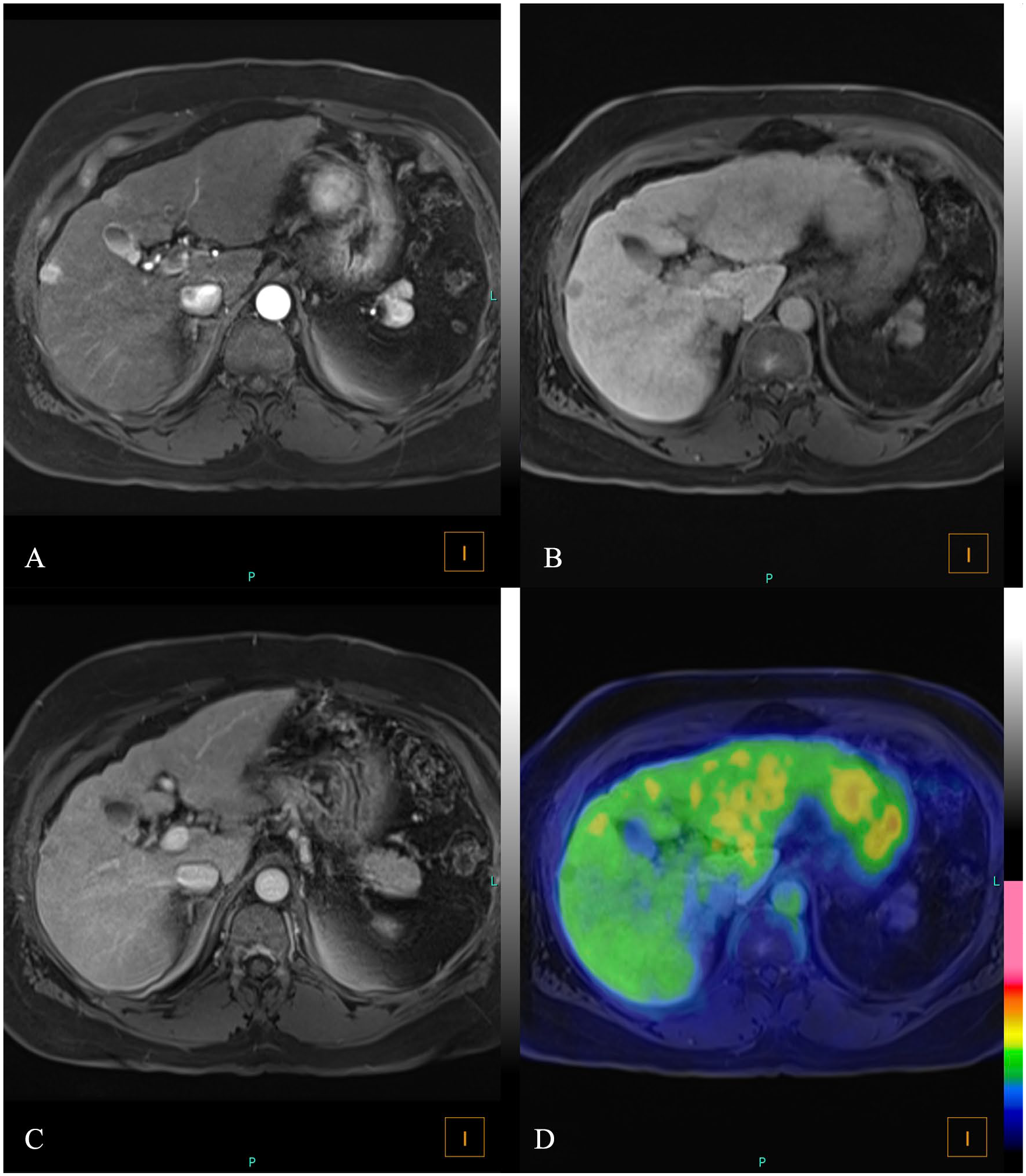
Arterial (A), portal (B), and delayed (C) phases in MR show a small subcapsular segment 5 liver lesion with delayed capsule enhancement compatible with hepatocellular carcinoma. Fused images (D) show absence of 18F-FDG uptake by the lesion, underscoring the value of the hybrid MR component in lesion characterization.
In GI, both PET and MR already play important roles in staging, treatment response, and surveillance. 41 For CRC, PET/CT is already indicated to rule out further metastases in surgical candidates and possible recurrence with elevated carcinoembryonic antigen (CEA) and normal conventional studies. 42 Hence, combining PET and MR offers a one-stop shop for these patients. 41 Similarly, in rectal and anal canal cancer, MR is the method of choice for local extension, while PET has shown superiority over CT and MR for nodal and whole-body staging, thus again supporting the role of PET/MR in these patients. 41
Pancreatic ductal adenocarcinoma is another field that needs to be investigated in more detail. Nevertheless, studies already showed that PET/CT or PET/MR could change the management of patients by up to 20% at staging 43 and that a ratio between the metabolic tumour volume and minimum apparent diffusion coefficient (ADC) in PET/MR was a strong predictor of advanced TNM and an independent predictor of progression-free survival (PFS). 44
Genitourinary and Gynaecology Applications
PET/MR serves as a comprehensive diagnostic modality in malignancies for which PET and MR are individually indicated, such as prostate cancer (Figure 5) and gynaecologic malignancies (Figure 6), particularly endometrial and cervical cancer. When performed simultaneously, the 2 modalities can offer synergistic information, enhancing diagnostic accuracy. The high soft-tissue and spatial resolution of MR is crucial for the evaluation of local tumour extent, while PET is superior at detecting nodal and distant metastatic disease. 45
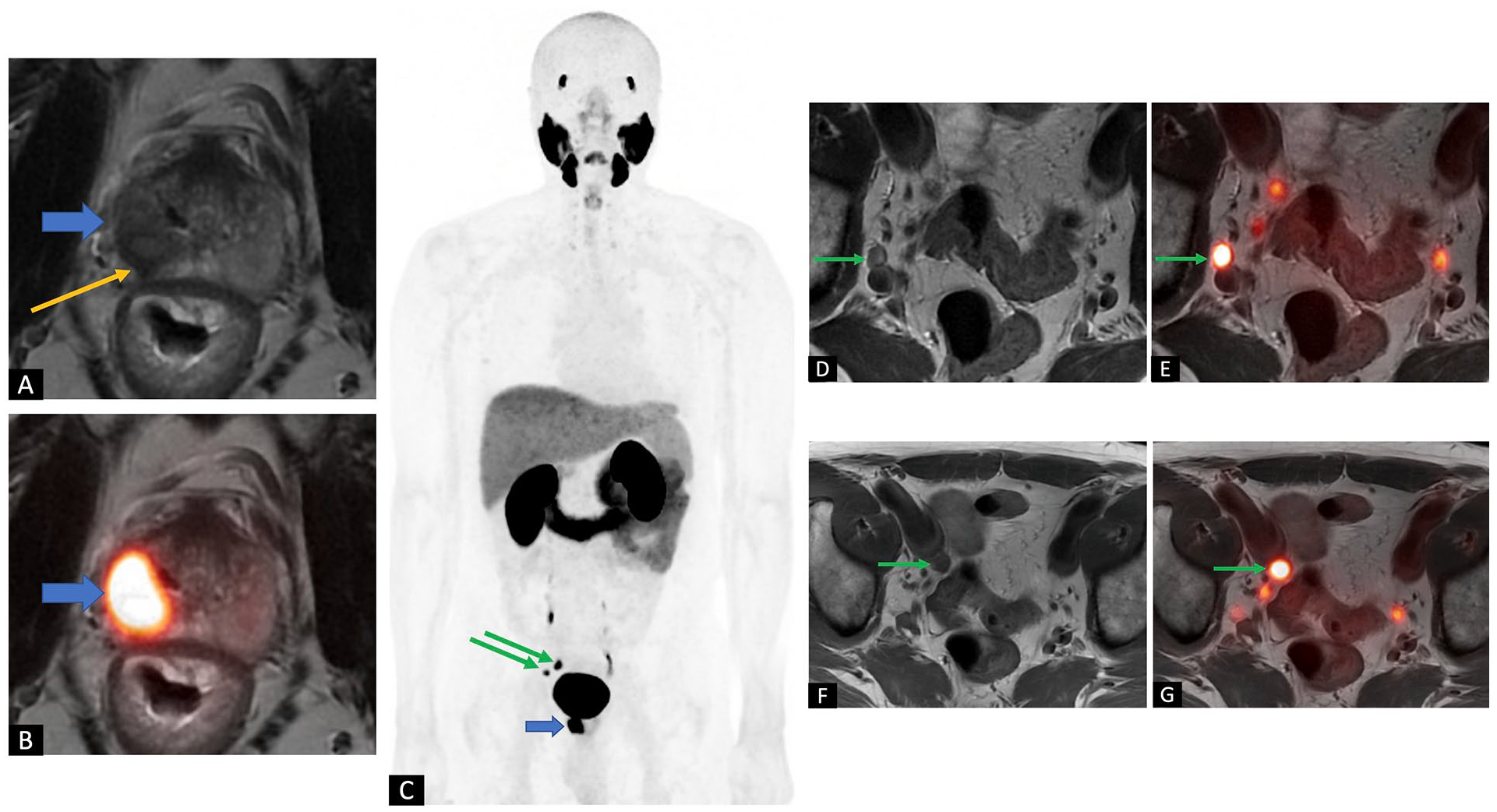
A 59-year-old male was referred for 18F-DCFPyL PET/MR for initial staging of high-risk prostate cancer (Gleason score 4 + 4) from transrectal ultrasound-guided biopsy with a relatively low serum PSA of 5 ng/mL. (A) Axial high-resolution T2w image of the prostate gland shows a large T2w hypointense tumour involving the right peripheral zone (blue arrow) with right-sided extra-capsular extension (yellow arrow). Seminal vesicles (not shown) were not involved. (B) Axial fused PET/MRI image shows intense PSMA uptake in the primary tumour (blue arrow). (C) Maximum intensity projection PET image shows suspicious right pelvic nodes (green arrows) in addition to the primary malignancy (blue arrow). No distant metastatic disease was evident. (D, F) Axial T2w and (E, G) axial fused PET/MR images show intense uptake in the right internal and external iliac nodes (green arrows), measuring 8 and 11 mm, respectively, consistent with nodal metastases. Overall staging by PET/MR: T3a N1 M0.
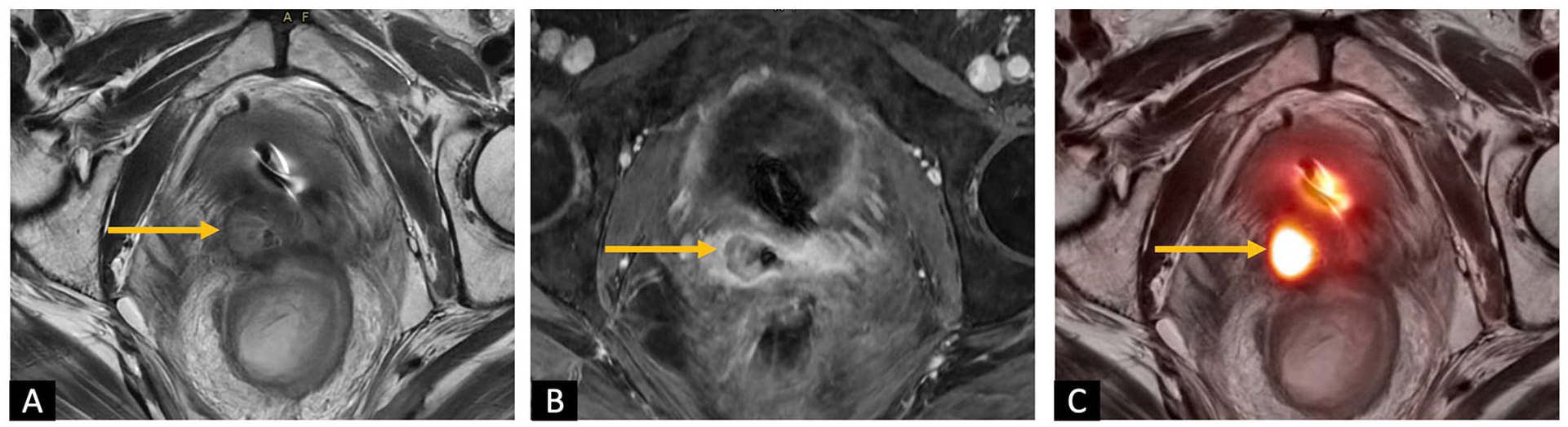
A 55-year-old female with a history of stage II grade 3 endometrial cancer resected 11 years ago with adjuvant external beam radiation therapy and subsequent diagnosis of squamous cell carcinoma of the rectum. The patient was referred for fullness at the right vaginal cuff concerning recurrent disease. (A) Axial T2w, (B) axial T1 post-contrast, and (C) axial fused 18F-FDG PET/MR images of the pelvis show a hypoenhancing right vaginal mass with intermediate T2w signal and intense 18F-FDG uptake, consistent with disease recurrence (yellow arrow). Biopsy confirmed recurrent endometrial cancer.
At the initial staging of cervical cancer, 18F-FDG PET/MR outperforms MR alone for local disease evaluation (detection of parametrial, vaginal, and deep cervical stromal invasion) and for the detection of distant metastases.46,47 In endometrial cancer, 18F-FDG PET/MR similarly allows for a comprehensive and more accurate evaluation than each modality alone; the incorporation of dynamic contrast enhancement is particularly helpful in endometrial cancer to assess the myometrial depth of invasion.48,49 The simultaneous PET/MR acquisition can outperform PET/CT for the detection of pelvic nodal metastases, which may be explained by the high sensitivity of the long PET acquisition acquired on the region of interest simultaneously along the dedicated pelvic MR. 50 The combined functional information obtained from both modalities (eg, tumour maximum standardized uptake value [SUVmax]/ADC ratio, tumour volume on MR, along with metabolic tumour volume [MTV] and total lesion glycolysis [TLG] on PET) is more predictive of pelvic nodal metastases and lymphovascular invasion relative to the parameters obtained from each modality alone.46,49,51 Similarly, this combined information can provide a higher accuracy for radiation planning, as well as for the detection of local disease recurrence.45,52
Given its high diagnostic precision, PSMA PET using one of the US Food and Drug Administration (FDA)-approved PSMA targeting molecules (68Ga-PSMA-11, 18F-Piflufolastat, and 18F-Flotufolastat) has been rapidly incorporated into the management paradigm of prostate cancer. Two scenarios particularly benefit from integrated PSMA PET/MR. First, the initial staging of unfavourable intermediate-risk disease and higher, for which both scans are indicated, and the second is early biochemical recurrence.53-55 At initial staging, simultaneous PET/MR provides accurate, comprehensive staging in one scan, with multiparametric MR being the standard of care for the assessment of extracapsular extension and seminal vesical invasion and PET being the gold standard for the detection of metastatic disease. 56 Retrospective studies suggest enhanced accuracy of integrated PSMA PET/MR for the assessment of local disease extent relative to each modality alone. 57 In early biochemical recurrence, PSMA PET is highly accurate for the detection of metastatic disease, while the prostate bed might be masked by the urinary excretion of the PSMA radiotracer, particularly in 68Ga-PSMA studies. The MR component of the scan, on the other hand, can offer the advantage of higher sensitivity for the detection of local recurrence in the prostate bed, PSMA PET has been shown to increase the specificity and positive predictive value in this setting.55,58
Haematology Applications
In haematologic oncology, PET/MR applications have been primarily investigated on 18F-FDG PET/MR value in lymphoma and myeloma. Although not yet a standard test, several applications exist where PET/MR may be of benefit. In lymphoma, 18F-FDG PET/CT is the standard-of-care imaging in managing the vast majority of patients.59,60 Since MR offers superior soft-tissue evaluation in many regions (eg, brain, head and neck, liver), there is growing evidence supporting its utilization as the anatomical component of choice alongside 18F-FDG PET.61-63 Combined 18F-FDG PET and MR can significantly enhance diagnosis, particularly in detecting extra-nodal involvement, that is, bone marrow. 64 PET/MR application can be signified by knowing that bone marrow assessment is a major challenge for current standard-of-care imaging while being significantly crucial in patient management, and bone marrow biopsy is required for definitive diagnosis in various scenarios.65,66 Since there is often no morphological correlate on CT, and with the known accuracy of MR in bone marrow assessment, performing PET/MR instead of PET/CT can be of great value, increasing diagnostic accuracy and confidence.64,67,68 Additionally, the MR component can aid where 18F-FDG PET is inconclusive, that is, after chemotherapy or granulocyte colony-stimulating factors administration when a diffuse 18F-FDG uptake is common. 69 PET/MR can also comprehensively assess treatment-induced changes in tumour metabolism, vascularity, and cellularity. 63 Moreover, since lymphoma can occur at young ages, MR is a satisfactory hybrid component in children and young adults. PET/MR can lead to a significant dose reduction compared to PET/CT.70-72 Although there can also be downsides, such as longer examination times than for PET/CT, in a robust multi-centre investigation, authors showed that by limiting the sequences (their suggestion: opting for non-enhanced T2w transverse sequences with fat saturation), we can still yield highly accurate detection rates. 73 Therefore, 18F-FDG PET/MR may eventually become the hybrid imaging of choice in radiation-sensitive populations, as well as patients requiring lifelong imaging follow-up or active surveillance. 74 Lastly, 18F-FDG PET/MR have the potential to be the standard-of-care imaging in lymphomas with variable 18F-FDG avidity, that is, mucosa-associated lymphoid tissue (MALT) lymphoma.75,76 DWI and ADC maps provide an additive value to 18F-FDG PET, that is, to evaluate MALT treatment response or rule out disease transformation (eg, chronic lymphocytic leukaemia to diffuse large B-cell lymphoma).77,78
Similar to lymphoma, 18F-FDG PET/MR is currently not standard imaging for multiple myeloma. However, since both 18F-FDG PET/CT and MR are recommended in various clinical scenarios, 18F-FDG PET/MR may soon become a routine in multiple myeloma management where available. 79 It can be a one-stop shop for evaluating bone, bone marrow, lymph nodes, and soft tissues. Additionally, MR parameters can be used as a surrogate marker for 18F-FDG uptake. 80 To predict disease progression from precursor stages, also, both 18F-FDG PET positivity and the number of focal lesions on MR have been shown to be prognostic, making hybrid 18F-FDG PET/MR a promising modality. 81
Lastly, looking beyond 18F-FDG, new applications for PET/MR are emerging in haematologic malignancies. In particular, the body of literature regarding CXCR4 imaging is growing, and some promising findings support the utilization of PET/MR in lymphomas/leukaemias with low to moderate 18F-FDG avidity. In MALT lymphoma, initial experiences have shown that CXCR4 PET/MR can be of value in newly diagnosed patients and can provide an excellent tumour-to-background contrast. 82 In mantle cell lymphoma, Mayerhoefer et al showed that 68Ga-Pentixafor PET/MR could be superior to MR alone for the evaluation of complete remission, especially at the end of treatment. 83 Furthermore, the feasibility of 68Ga-Pentixafor PET/MR was also established in patients with chronic lymphocytic leukaemia and acute myeloid leukaemia. 84 Thus, further investigations on non-18F-FDG PET/MR applications may report additional benefits for PET/MR in the field of haematology in the near future.
Summary
In summary, PET/MR has experienced major improvements in recent years, making it a reliable state-of-the-art modality in medical practice. Thanks to the advanced MR sequences (eg, UTE/ZTE), PET/MR seems to show increasing potential in chest applications. In lung cancer evaluation, PET/MR shows promise, offering accurate and comparable overall staging. Also, the ability of its MR component to provide superior soft tissue resolution can offer increased accuracy in assessing the malign involvement of structures in the mediastinum and pleura. Furthermore, PET/MR is capable of accurately assessing distant organ metastasis, along with providing potential prognostic insights and treatment response assessment. In the field of cardiology, cardiac PET/MR can provide reliable information in evaluating myocarditis, amyloidosis, myocardial perfusion, myocardial viability, atherosclerotic plaques, and cardiac masses. In GI and HBP malignancies, 18F-FDG PET/MR can precisely detect primary tumours and metastases, particularly in the liver. In addition, the utilization of SSTR PET/MR and hepatobiliary contrast agents was proven to be highly accurate in the assessment of NETs. Furthermore, in rectal and anal canal cancer, MR is the method of choice for local extension, while 18F-FDG PET has shown superiority over other modalities for nodal and whole-body staging, supporting the role of PET/MR in these patients. Considering genitourinary and gynaecology applications, 18F-FDG PET/MR is a comprehensive diagnostic method, especially in prostate, endometrial, and cervical cancers. The high soft-tissue and spatial resolution of MR is crucial for the evaluation of local tumour extent, while PET is superior at detecting nodal and distant metastatic disease. Hence, when performed simultaneously, the 2 modalities can offer synergistic information, enhancing diagnostic accuracy. Lastly, PET/MR can significantly enhance the diagnosis of haematologic malignancies, particularly in detecting extra-nodal/osseous involvement and assessing post-treatment changes, and is a one-stop shop for evaluating bone, bone marrow, and soft tissues.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
