Abstract
Positron emission tomography/magnetic resonance (PET/MR) imaging has gone through major hardware improvements in recent years, making it a reliable state-of-the-art hybrid modality in clinical practice. At the same time, image reconstruction, attenuation correction, and motion correction algorithms have significantly evolved to provide high-quality images. Part I of the current review discusses technical basics, pre-clinical applications, and clinical applications of PET/MR in radiation oncology and head and neck imaging. PET/MR offers a broad range of advantages in preclinical and clinical imaging. In the preclinic, small and large animal-dedicated devices were developed, making PET/MR capable of delivering new insight into animal models in diseases and facilitating the development of methods that inform clinical PET/MR. Regarding PET/MR’s clinical applications in radiation medicine, PET and MR already play crucial roles in the radiotherapy process. Their combination is particularly significant as it can provide molecular and morphological characteristics that are not achievable with other modalities. In addition, the integration of PET/MR information for therapy planning with linear accelerators is expected to provide potentially unique biomarkers for treatment guidance. Furthermore, in clinical applications in the head and neck region, it has been shown that PET/MR can be an accurate modality in head and neck malignancies for staging and resectability assessment. Also, it can play a crucial role in diagnosing residual or recurrent diseases, reliably distinguishing from oedema and fibrosis. PET/MR can furthermore help with tumour characterization and patient prognostication. Lastly, in head and neck carcinoma of unknown origin, PET/MR, with its diagnostic potential, may obviate multiple imaging sessions in the near future.
This is a visual representation of the abstract.
Introduction
Positron emission tomography (PET) and magnetic resonance imaging (MR) have been in the field of clinical imaging for more than 3 decades. However, hybrid PET/MR is the most recent combination in non-invasive diagnostic molecular imaging, and its literature on various technical innovations, preclinical as well as clinical applications has rapidly increased since its introduction. Similar to the other hybrid molecular imaging, it provides morphological information alongside metabolic activity or other cellular-level characteristics of tissues and offers the possibility of integrated quantification. Through this combination, PET-imaging—with different radiopharmaceuticals—was able to expand into different /new oncologic but also non-oncologic imaging, and MR gained capabilities to visualize molecular-based information beyond morphology. Although PET/MR is not widely available worldwide, based on several and partly site-specific reasons, and while it has found its place into only few guidelines, there are noticeable pieces of evidence that support its utilization in clinical practice. In this 2-part review article, we are covering the most prominent current applications of PET/MR in different fields of medical practice. In the first part, the technical basics of PET/MR as well as preclinical are being discussed. Moving on to clinical applications then, its value in radiation medicine and the head and neck region are reviewed in this part (part I), whereas other clinical applications will be discussed in part II, including PET/MR in chest, cardiac, gastrointestinal, hepatobiliary-pancreatic, genitourinary, gynaecology, and haematology applications.
Technical Basics
The current performance of integrated clinical PET/MR is achieved with recent hardware and software improvements, and the related technical basics are briefly discussed here.
System Hardware Design
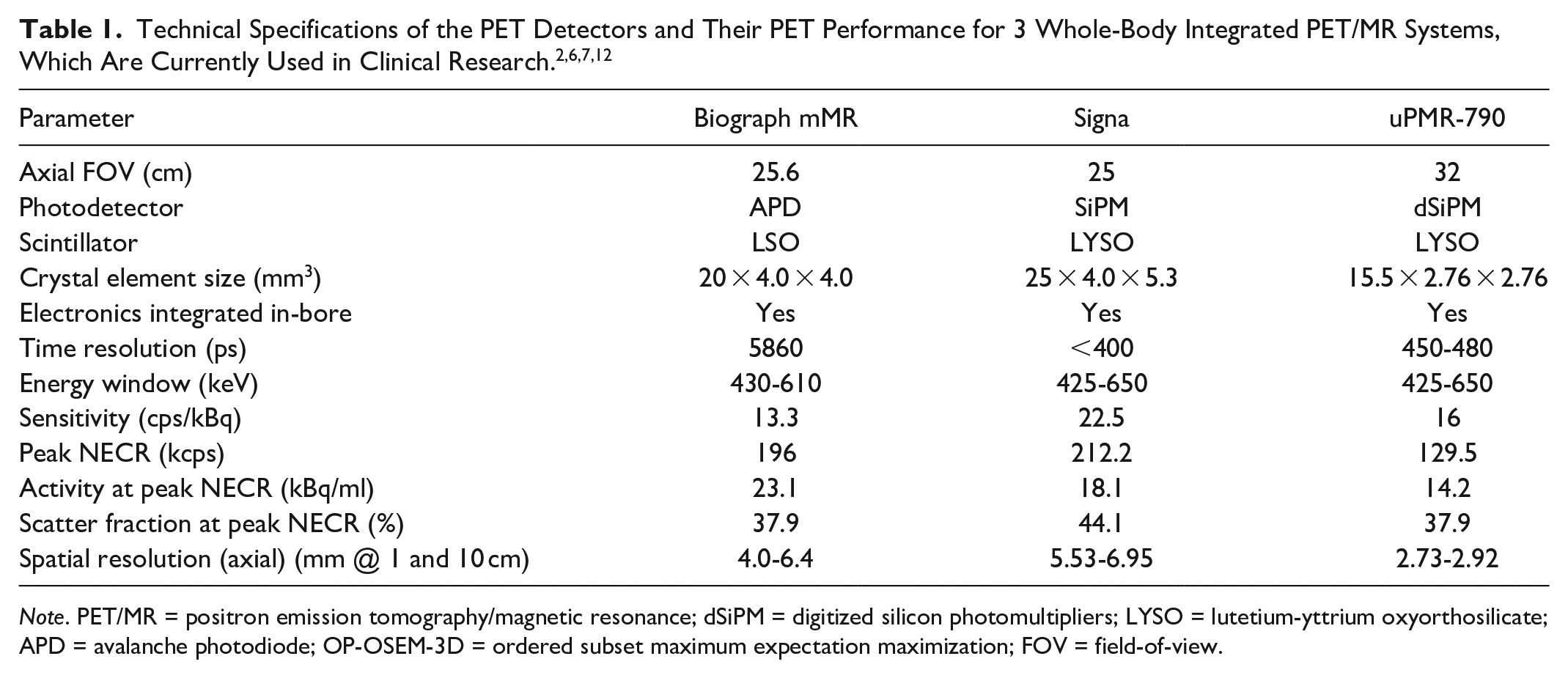
In all current clinical PET/MR systems, both modalities are truly integrated into one housing so that they operate simultaneously. Earlier on, the 2 imaging modalities were used sequentially when introduced by Philips in 2009 at Mount Sinai Medical Center, NY., and Hôpitaux Universitaires de Genève, Geneva. 1 In less than a year, in 2011, Siemens Healthcare released the first simultaneous imaging results from the first commercial clinical integrated PET/MR (Biograph mMR based on Verio platform) at the Technische Universitat Munchen, Germany. 2 Thereafter, in 2013, GE Healthcare provided a comparable simultaneous PET/MR system based on the Signa Discovery-750 platform with the addition of the smart time-of-flight feature.3,4 Most recently, United Imaging Healthcare introduced the third simultaneous PET/MR, the uPMR-790 system with similar features to the Biograph and Signa systems. 5 The MR component usually has similar specifications across different PET/MR vendors, such as a 60 cm-patient bore, a superconducting magnet with a field strength of 3.0T, whole-body gradient coil systems (44-50 mT/m amplitude, 200 T/m/s slew rate), and a Tx/Rx radiofrequency (RF) body resonator.2,6,7 Similar to a stand-alone MR system, the PET/MR uses dedicated receiver RF resonators to acquire high spatial and temporal resolution images for each region of the body. This requires RF-phased receivers’ arrays with at least >8 channels, especially for neural and cardiac imaging.8-10 Normally, the PET gantry is placed between the shield of the RF body resonator and the gradient coils. However, concerning PET components such as the PET detector, each vendor uses different technology, arrangements, and specifications (Table 1). Perhaps the currently most desirable technology is a combination of digitized silicon photomultipliers and detectors with lutetium-yttrium oxyorthosilicate crystals. The digitized silicon photomultipliers provides a faster response compared to the avalanche photodiodes and, hence, allows time-of-flight imaging. Furthermore, the lutetium-yttrium oxyorthosilicate crystals include retroreflector technology that enhances PET sensitivity through faster scintillation decay, resulting in a higher count rate. 11 However, avalanche photodiode’s were the first detectors introduced in PET/MR and are still working reliably in the field.
Note. PET/MR = positron emission tomography/magnetic resonance; dSiPM = digitized silicon photomultipliers; LYSO = lutetium-yttrium oxyorthosilicate; APD = avalanche photodiode; OP-OSEM-3D = ordered subset maximum expectation maximization; FOV = field-of-view.
PET Image Reconstruction, Attenuation, and Motion Correction
PET images are commonly reconstructed with the robust algorithms—ordered subset maximum expectation maximization, which preserves the spatial resolution and incorporates correction for scatters and random coincidences. 13 Additionally, the PET image contrast and sharpness can be enhanced by applying the MR-guided reconstruction technique, where the anatomical information aids the reconstruction (Figure 1).14,15 The photon attenuation caused by MR components in the PET field-of-view, such as resonators, headphones, eye-tracking, the patient table, etc., is usually corrected using CT-based attenuation correction (AC) maps of coefficients at 511 keV energy level. 16 AC can also be performed using a point source transmission-based AC map 17 or rotating transmission source, 18 improving the accuracy of the AC and resolving the beam hardening effect in CT-based AC. Meanwhile, the AC of the patient’s body (tissue, water, fat, and bone) has evolved from MRAC acquisitions (ie, dual-echo Dixon and ultra-short echo time) to AI-based pseudo-CT-maps (Figure 2). 19
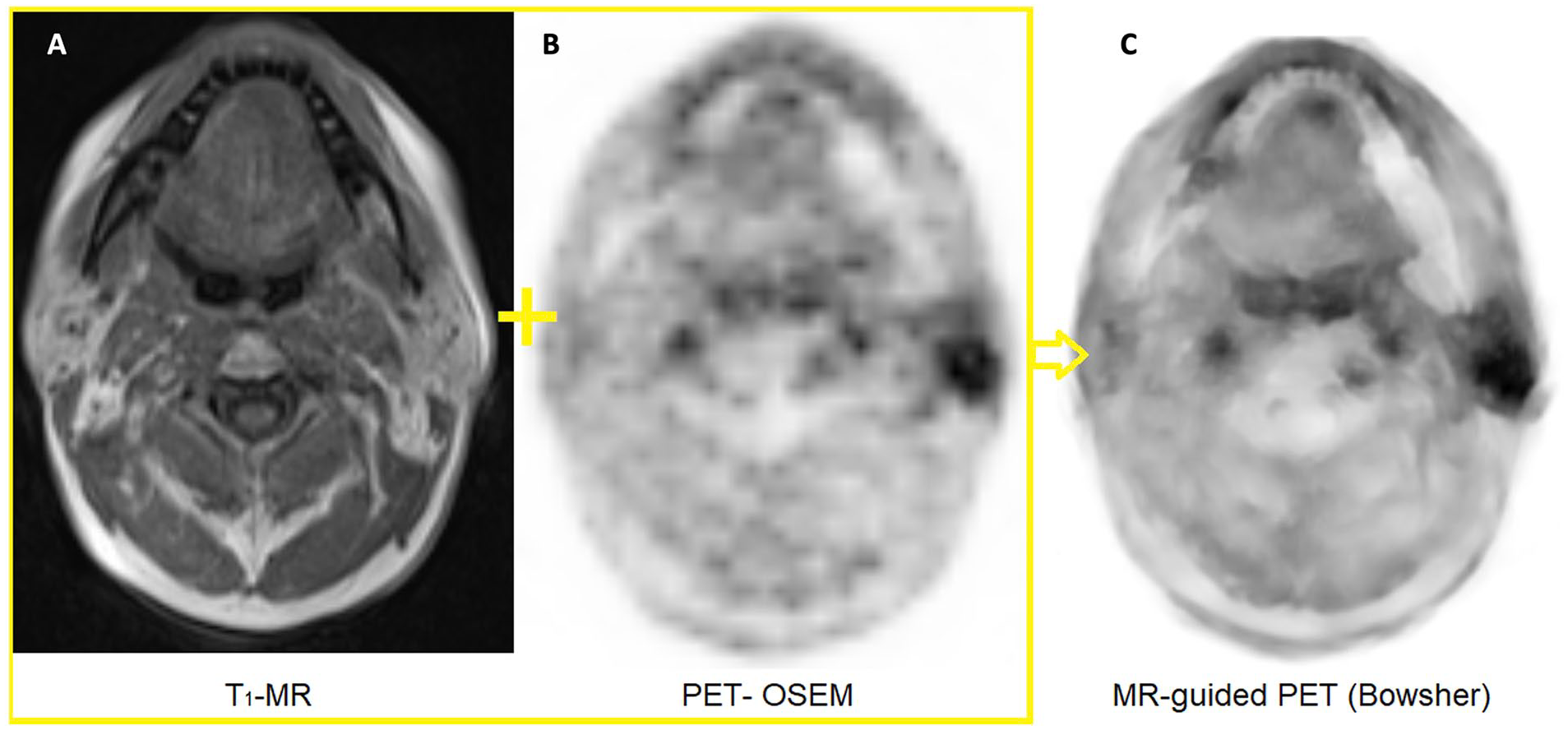
Bowsher MR-guided PET reconstruction technique utilizes T1-MR image (A) and OSEM PET image (B) to produce sharp and high contrast PET image (C).
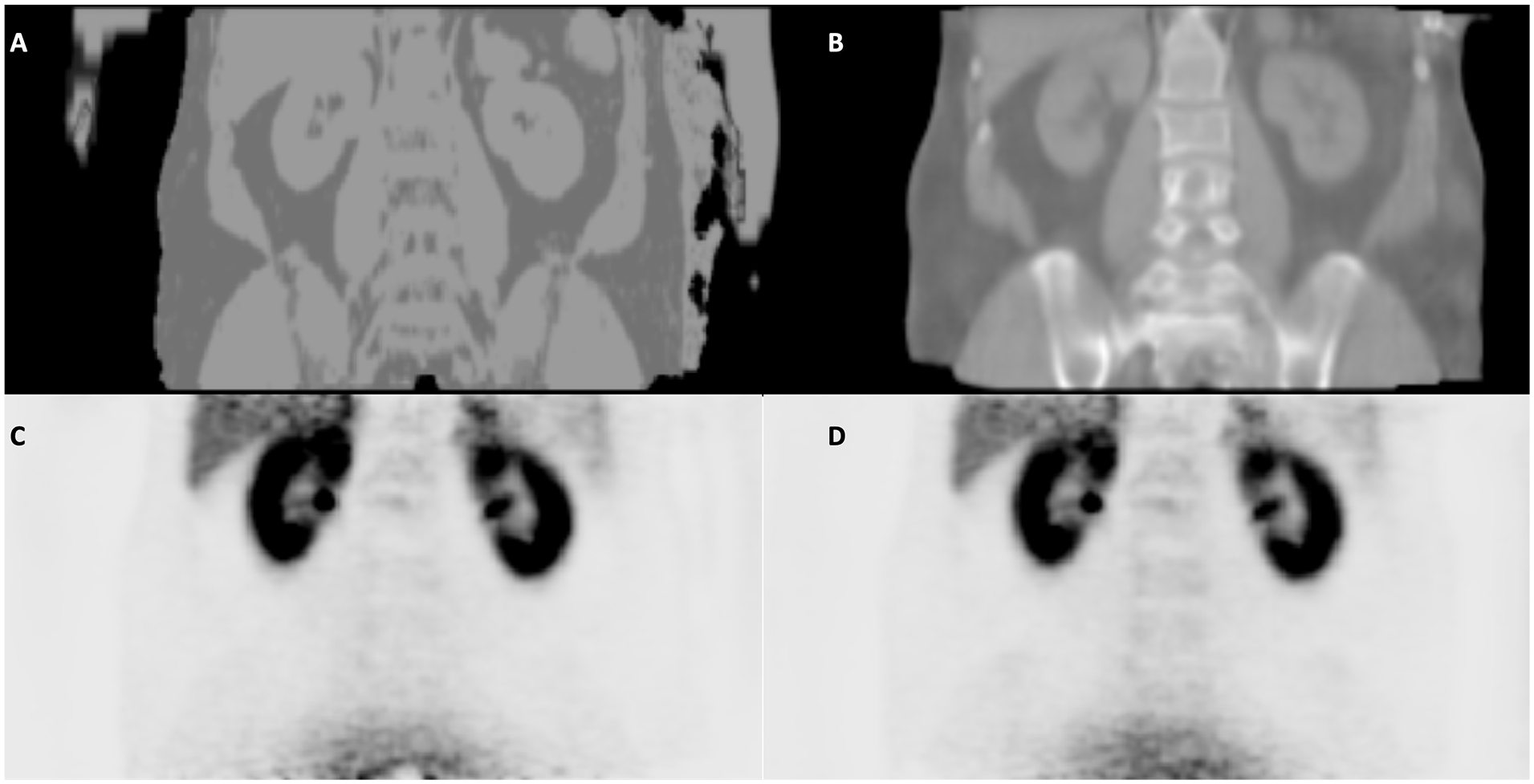
The conventional Dixon-based attenuation correction (AC) map is shown in (A) along with its corresponding PET image (C). The latest technique, AI-based AC, that generates pseudo-CT mimicking the CT-based AC map, is shown in (B) and its PET image (D).
Similar to stand-alone imaging modalities, respiratory and cardiac motions in PET/MR are great sources of degradation of the image quality. For motion correction, several techniques are available for the PET/MR, including PET data-driven motion correction,20,21 MR-based 3D radial stack-of-stars spoiled gradient-echo sequence, and device-aided motion correction (ie, pilot-tone). 22
Preclinical Applications
The development of hybrid PET/MR systems started with small, MR-compatible PET detectors and PET detector rings that were first evaluated in murine models of disease.23-25 In addition to being a technology development platform, preclinical PET/MR can provide new insight into animal models of disease and facilitate the development of methods that inform clinical PET/MR.
Small Animal PET/MR
Small animal PET/MR typically requires dedicated systems that can acquire images with increased spatial resolution and sensitivity. Fortunately, there are numerous examples from both commercial vendors26-32 and instrumentation labs.33-35 Simultaneous PET/MR can improve animal welfare with shorter acquisition times and fewer tail vein punctures. Likewise, simultaneity is a necessity for PET/MR studies that seek to measure complementary functional information or account for motion with the help of information from MR. Conversely, sequential PET/MR systems and acquisitions may benefit from larger axial and transaxial field-of-view, accommodating larger animals and multi-animal beds with higher PET sensitivity.
Small animal PET/MR benefits from the large number of mouse and rat models in which acute and chronic diseases can be induced surgically, chemically, genetically, or arise spontaneously. It can aid in the development of new PET tracers that require biodistribution and pharmacokinetic studies prior to more expensive large animal models and human studies.
Preclinical PET/MR is useful for delineating different anatomical regions of the brain as well as for the development of synergistic PET/MR methods with ex vivo tissue validation (Figure 3A). 36 In addition to anatomical information, MR can provide quantitative and functional measurements of relaxation, diffusion, perfusion, blood oxygen level-dependent (BOLD) signal, chemical composition, and contrast agents. Simultaneous small animal PET/MR has been used to show simultaneous activation in different brain regions with BOLD functional MR and 18F-fluorodeoxyglucose ( 18 F-FDG) PET37,38 and evaluate PET/MR contrast agents for sensing extracellular pH. 39 By combining quantitative information from PET and MR, we have gained new insights into cancer cell proliferation40,41 and tumour metabolism (Figure 3B). 42
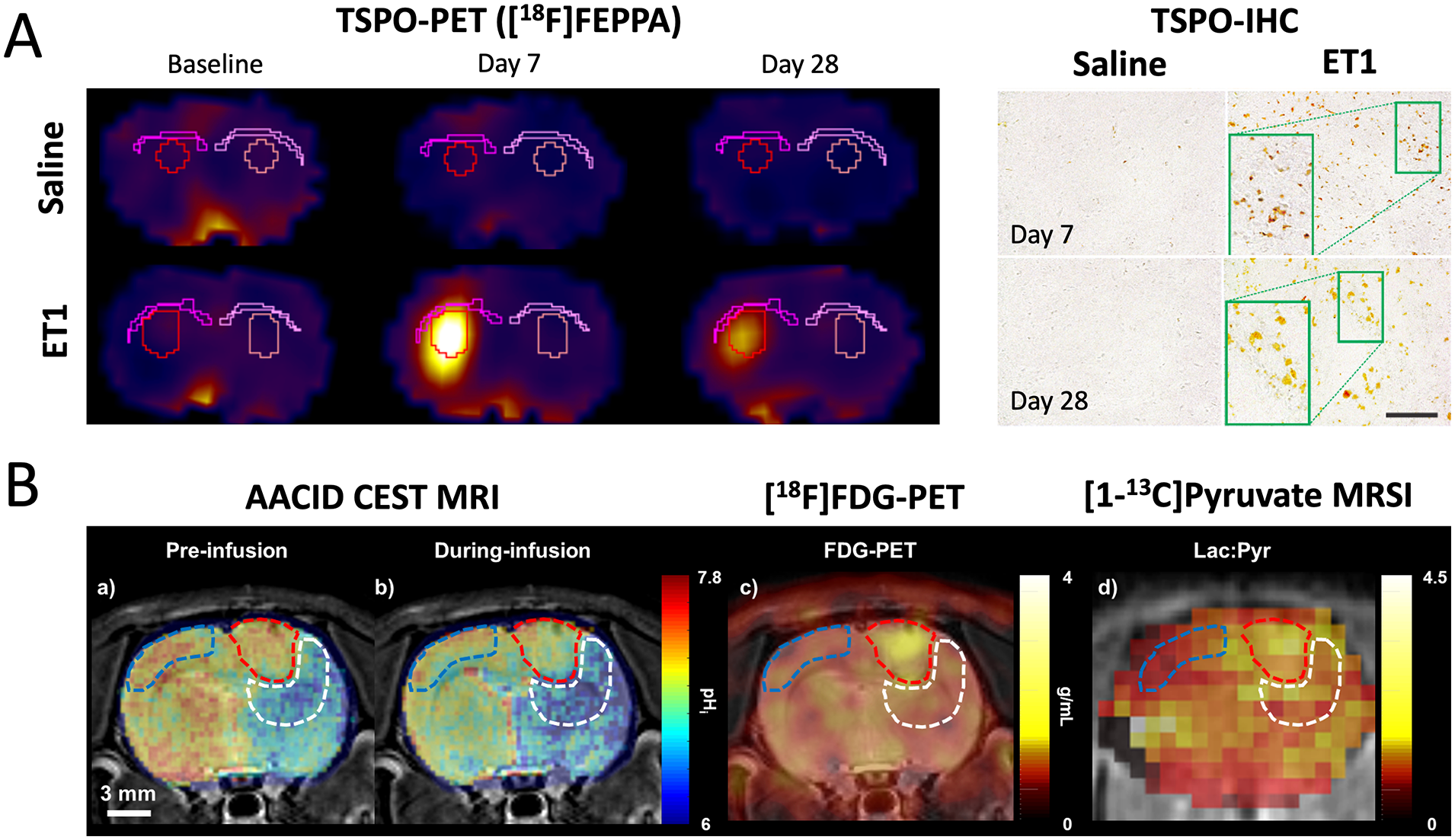
Small animal PET/MR examples. (A) Evaluation of a PET tracer targeting the translocator protein (TSPO) overexpressed in activated microglia. Using an endothelin-1 (ET1) rat model of stroke, Al-Khishman et al were able to show elevated TSPO-PET expression in the infarct with confirmatory TSPO immunohistochemistry (TSPO-IHC) in corresponding tissue. MR helped guide the delineation of anatomical structures in the brain, demonstrating that TSPO was not present in remote white matter despite evidence of activated microglia overexpressing MHCII in immunohistochemistry. The figure was adapted from Al-Khishman et al 36 and used under CC-BY 4.0. (B) Using a chemical exchange saturation transfer (CEST) MR method sensitive to intracellular pH (pHi) known as amine and amide concentration-independent detection (AACID), Qi et al demonstrated that an infusion of glucose could drive a decrease in tumour pHi that was correlated with both 18F-FDG PET and hyperpolarized [1- 13 C]pyruvate MRSI measurements of glucose metabolism. This work was performed in a C6 rat model of glioma with sequential acquisitions in a small animal PET system (Siemens Inveon DPET), small animal 9.4T MR (Agilent), and clinical 3T MR (GE MR750). The figure was adapted from Qi et al 42 with permission from Springer Nature.
Large Animal PET/MR
Although more expensive and challenging, large animal PET/MR can improve our understanding of disease pathology and serve as a platform to develop and translate PET/MR methods to clinical research and care. Despite the increased infrastructure and costs related to large animal imaging, there are many benefits to simultaneous large animal PET/MR on clinical scanners. By using a clinical PET/MR, researchers have access to a wide range of MR pulse sequences with the opportunity to directly translate PET/MR methods and findings to human research protocols. Large animals also have anatomy and physiology that are closer to humans. Spatial and temporal fusion of MR and PET datasets can help mitigate cardiac/respiratory motion and show the relationship of PET tracer uptake to numerous MR measurements.
Large animal PET/MR has been used to investigate myocardial infarction and heart failure,43-46 cardiotoxicity related to radiotherapy,47,48 and to develop methods that combine information from MR and PET for improved quantification49,50 (Figure 4). When acquired on a clinical PET/MR, methods developed in large animals can be directly translated into human studies.51,52
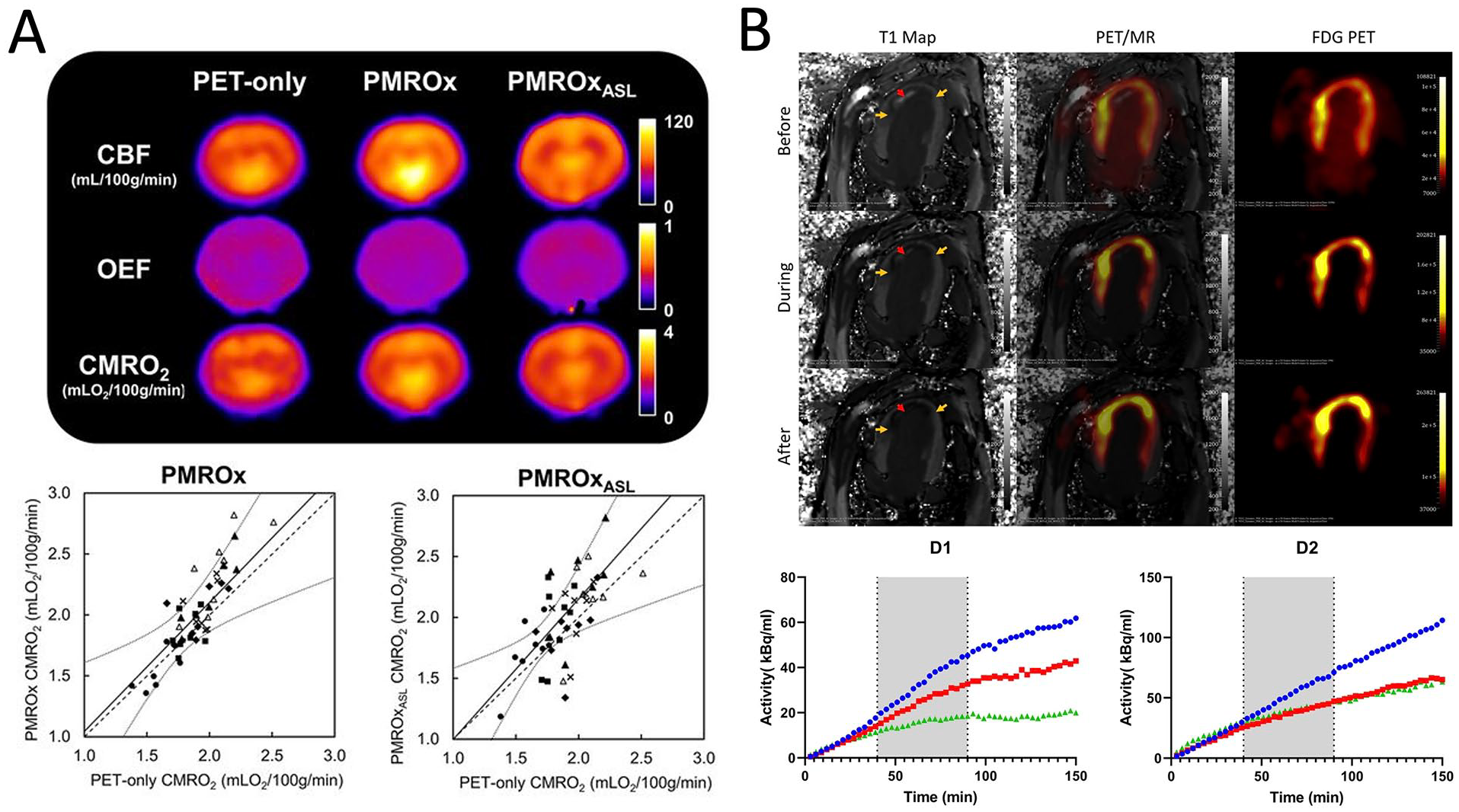
Large animal PET/MR examples. Both examples have informed subsequent human studies on the same 3T clinical PET/MR (Siemens Biograph mMR). (A) Using a porcine model, Narciso et al validated a novel PET/MR method that greatly simplifies the acquisition of cerebral metabolic rate of oxygen (CMRO2) maps with a dynamic acquisition of 15O-O2-PET along with simultaneous MR measurements of whole-brain CMRO2 and voxel-wise cerebral blood flow (ASL). The PET/MR approaches (PMROx and PMROxASL) were strongly correlated with the gold-standard PET-only approach. Figure from Narciso et al 53 © SNMMI. (B) In a canine model of myocardial infarction, Wilk et al were able to evaluate the efficacy of myocardial glucose suppression using a heparin injection and lipid infusion (grey time span) during a 150-minute constant infusion of MR contrast agent (Gd-DTPA) and 18F-FDG. Dynamic PET and quantitative MR were acquired throughout the infusion protocol. Combined with tissue validation, they were able to demonstrate the limitations of myocardial glucose suppression for detecting inflammatory cells with 18F-FDG PET in the myocardium. The figure was adapted from Wilk et al 54 and used under CC-BY 4.0.
Overall, preclinical PET/MR can play an important role as a platform for discovery science, illuminating and expanding the capabilities of clinical PET/MR.
Radiation Medicine Applications
The effectiveness of external beam radiotherapy hinges on the ability to direct radiation dose to tumours while avoiding dose to critical normal structures. Advances in medical imaging science and radiotherapy technologies evolve hand in hand as each demands breakthroughs and creates opportunities in the other.
Imaging acquired as part of staging can provide important qualitative information for target delineation. However, dedicated imaging acquired specifically for radiotherapy planning provides incremental value, considering quantitative and spatial information that may not be available otherwise. A dedicated PET/CT simulator and planning MR acquisition allows images to be acquired in the same position as the planning CT, allowing for superior registration and image fusion, implementation of motion management techniques and opportunities for biology-guided radiotherapy. Standardized acquisition and segmentation protocols (eg, windowing and colour scale, mode of scanning) improve how the images can be interpreted and used in quantitative applications to the radiotherapy process.
Target delineation accuracy is the first step in which PET and MR data have already played an important role in the radiotherapy process. 18F-FDG PET is the most commonly used tracer. Clinical studies have demonstrated clinical impact beyond standard planning CT data in selected circumstances such as non-small cell lung cancer (NSCLC) with adjacent atelectasis, gastrointestinal malignancies such as the oesophagus, pancreas, and anal cancers to support involved node delineation and demarcation infiltration boundaries. Other tracers, such as 68Ga-DOTATATE for skull base meningiomas, as well as prostate-specific membrane antigen (PSMA) tracers for prostate cancer with oligometastasis suitable for ablative radiotherapy, among others, are increasingly being studied. 55 Dedicated planning MR is more frequently incorporated into the radiotherapy workflow. Especially for treatment techniques that demand high accuracy in target delineation, such as stereotactic radiotherapy for many anatomic sites (eg, liver, pancreas, spine, anal rectum, central nervous system), volumetric modulated arc therapy (VMAT), and intensity-modulated radiation therapy (IMRT) techniques, the incorporation of dedicated MR with or without contrast is already part of the standard of care in many state-of-the-art radiotherapy centres.
Beyond morphologic and spatial localization of the tumour, perhaps the most powerful feature PET and MR can bring to the radiotherapy workflow is the ability to capture functional characteristics at baseline and in response to treatment that is otherwise not possible with other modalities. For example, the use of 18F-fluoromisonidazole or 18F-Fluoroazomycin arabinoside ( 18 F-FMISO/18F-FAZA) PET and MR acquired with diffusion-weighted imaging (DWI) and the derived apparent diffusion coefficient (ADC) can both point toward potential hypoxic regions. Changes in 18F-FDG uptake can reflect radio-responsiveness. Similarly, large ADC values have been found to correlate with radiotherapy responders. Dynamic contrast-enhanced (DCE) has also been investigated as a biomarker for radiotherapy but requires contrast administration during MR. The use of these data can identify biological sub-volumes that benefit from differential dosing in an area of investigation. 56
Radiotherapy, in its classical form, involves a treatment plan designed upfront and delivered over time. Adaptive radiotherapy refers to the changes in the treatment plan in response to positional and volume changes along the treatment course and is an area of active adoption and investigation. The launch of the MR-Linac, the integration of MR with linear accelerator into a single platform, enables online visualization and adaptation and is purpose-built. By seeing in real time, tumours near dose-limiting structures can now receive higher, optimal doses that may not be possible otherwise. Biology-guided adaptive radiotherapy represents the next logical step. Van Houdt et al described the value of different MR-based metrics for this purpose, including many tumour sites such as gastrointestinal malignancies, cervix, prostate, and brain metastases, using DWI, T2w, and T1w data. 56 PET-Linac has recently been launched as well, allowing PET profiles or biological signatures to act as a fiducial, providing the opportunity for multiple sites to be treated at once. 57
The integration of PET/MR with linear accelerators into a single unit, enabling simultaneous acquisition of both tissue morphology and tumour metabolism in treatment, is again expected to provide the greatest potential of providing unique biomarkers for treatment guidance. Yan et al provided a good overview of the considerations from patient positioning, target delineation, and efficacy evaluation to patient surveillance. 58 Their early work suggested that treatment-induced changes at 48 to 72 hours could predict long-term responses and subsequent metabolic remission in lymphoma. Zamboglou et al demonstrated the feasibility of integrating PSMA PET and MR-based focal dose escalation in radiotherapy while maintaining favourable toxicity and quality of life. 59 In another study, Gouel et al provided early data contrasting signals from 18F-MISO PET, 18F-FDG PET, and contrast-enhanced MR to characterize hypoxic tumours in oropharynx cancers. 60 Nanoparticles providing dual-modality probes will likely play an important role in clarifying novel biology-guided radiotherapy. Whether the promise of enriching the imaging signals to guide radiotherapy will translate into improved outcomes requires active investigation.
Head and Neck Applications
Overall, in head and neck malignancies, 18F-FDG PET/MR has been shown to be of value, while there are also limited, albeit promising results for PET/MR using radiotracers beyond 18F-FDG, such as 68Ga-labelled fibroblast activation protein inhibitor ( 68 Ga-FAPI). 61 For cancer staging and resectability assessment, several studies showed that 18F-FDG PET/MR is potentially superior compared to other imaging modalities (eg, MR or PET/CT),62,63 though most studies indicated 18F-FDG PET/MR is a highly accurate modality with more or less comparable accuracy to 18F-FDG PET/CT in overall staging.64-67 The main superiority of 18F-FDG PET/MR is in primary or recurrent tumour characterization, as well as locoregional staging.63,68,69 For instance, both primary and recurrent head and neck squamous cell carcinoma have characteristic PET/MR features, which generally include an intermediate signal intensity on T1w, T2w, or fat-saturated T2w sequences, moderate enhancement after administration of contrast media, restricted diffusivity on ADC maps, and increased standardized uptake value (SUV) on 18F-FDG PET images. In the previous investigations that compared the accuracy of PET/MR in locoregional staging with PET/CT, conflicting results have been reported in head and neck cancer. However, there was a trend toward enhanced evaluation of locoregional spread with the utilization of PET/MR. 68 For example, Chan et al prospectively studied 113 patients with primary nasopharyngeal carcinoma. 63 They reported that 18F-FDG PET/MR was significantly more accurate than 18F-FDG PET/CT in terms of defining the extent of the primary tumour, by being more reliable in depicting intracranial invasion, perineural infiltration, infra-orbital fissure involvement, and bone marrow invasion to the clivus. Interestingly, 18F-FDG PET/MR could also correct false-positive findings of the head and neck MR when evaluating tumour extension into the nasal cavity or oropharynx. In addition, for N staging assessment, they reported that 18F-FDG PET/MR was more sensitive than head and neck MR and 18F-FDG PET/CT alone. In particular, 18F-FDG PET/MR was useful for distinguishing retropharyngeal lymph node metastasis from adjacent nasopharyngeal tumours.
As a unique advantage, PET/MR is able to acquire a dedicated head and neck MR protocol tailored to imaging the respective clinical question. 70 In addition, whole-body PET/MR can provide a one-stop-shop assessment in patients with lower neck tumours (eg, in the hypopharynx) or lymph node metastases (cervical levels III/IV) who need whole-body staging due to the high risk of distant metastases. 71 Also noteworthy, in patients with risk factors are allergy against gadolinium-based contrast media, PET/MR can be safely performed without contrast agents, being still as accurate as contrast-enhanced PET/CT imaging based on advanced optional sequences, that is, fat-suppressed T2w pulse sequence. 72 Moreover, although the literature in this regard is scarce, it seems there is a recent shift toward PET/MR imaging-guided estimation of gross tumour volume for radiotherapy planning in head and neck carcinomas. 68 As an example, in their investigation, Samołyk-Kogaczewska et al studied head and neck squamous cell carcinoma in a limited population and showed that the combination of PET/MRI could provide more information during target tumour mass segmentation in radiotherapy planning of patients than other standard imaging techniques, being able to enhance the accuracy of target volume delineation. 73
In addition, PET/MR can significantly help in the diagnosis of residual or recurrent disease, reliably distinguishing them from oedema and fibrosis.68,74-77 In particular, PET/MR with DWI was shown to be highly accurate in evaluating head and neck squamous cell carcinoma residual disease or recurrence and could facilitate salvage surgery planning in the irradiated neck.68,77 In a prospective investigation by Becker et al, they reported a high diagnostic accuracy for PET/MR with DWI in the detection of recurrent or residual disease following radiotherapy. Sensitivity, specificity, positive predictive value, and negative predictive value of PET/MR were 97%, 92%, 93%, and 97% per patient and 93%, 94%, 91%, and 95% per lesion, respectively. They also found an excellent agreement between PET/MR-based and histopathology-derived local stages of the resected tumours.
Patel et al recently showed that by using Neck Imaging Reporting and Data System (NI-RADS) criteria in PET/MR interpretation, they were able to evaluate treatment failure for the primary tumour, neck lymph nodes, and combined sites. 78 Ashour et al also reported a similar experience by implementing MR’s DWI and T2w signal intensity alongside PET in NI-RADS criteria, resulting in an improved NI-RADS accuracy. 79 Moreover, the combination of PET-derived + MR-derived parameters (eg, metabolic tumour volume [MTV] and total lesion glycolysis [TLG] + ADC) was reported to have prognostic value in predicting later recurrence of the disease, which can signify the value of PET/MR as one-session hybrid imaging. 80 In addition, various 18F-FDG PET/MR-derived parameters (eg, information from DWI and DCE, as well as PET SUV) may help assess tumour differentiation in head and neck malignancies, aiding in patient management and prognostication.81,82 Lastly, in patients with head and neck carcinoma of unknown origin, PET/CT and MR are both present in the diagnostic workup algorithm, in which PET/MR, with its diagnostic potential in this clinical scenario, may obviate 2 imaging sessions in the near future.83-85
Summary
To summarize part I of our review, the literature supports the notion that PET/MR can provide the field of medical practice with accurate information, having unique advantages that make it stand out from other imaging techniques in various preclinical and clinical scenarios. The hardware and image reconstruction advancements have helped PET/MR to enhance its capabilities and provide reliable characteristics considering both molecular and morphological aspects of the target tissues. Also, preclinical PET/MRs have facilitated the development of methods informing clinical PET/MR. Small and large animal devices play an important role as a platform for discovery science, illuminating and expanding the capabilities of clinical PET/MR. In addition, they have been able to provide new insight into animal models of disease.
In clinical applications, in radiation medicine, PET/MR could improve the ability to capture functional characteristics at baseline and in response to treatment that would otherwise not be possible with other modalities. Different introduced radiotracers (eg, 18F-FDG, 18F-FMISO) alongside different sequences of the MR’s component can precisely visualize various characteristics of the tumours, such as radio-responsiveness and potential hypoxic regions, and help identify biological sub-volumes that benefit from differential dosing in an area of investigation.
For head and neck applications, PET/MR was shown to be of value in cancer staging and resectability assessment. The main superiority of PET/MR is in primary or recurrent tumour characterization, as well as locoregional staging. In addition, whole-body PET/MR can provide a one-session assessment in patients with lower neck tumours or lymph node metastases who need whole-body staging due to the high risk of distant metastases.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.