Abstract
Breast cancer screening guidelines vary for women at intermediate risk (15%-20% lifetime risk) for developing breast cancer across jurisdictions. Currently available risk assessment models have differing strengths and weaknesses, creating difficulty and ambiguity in selecting the most appropriate model to utilize. Clarifying which model to utilize in individual circumstances may help determine the best screening guidelines to use for each individual.
This is a visual representation of the abstract.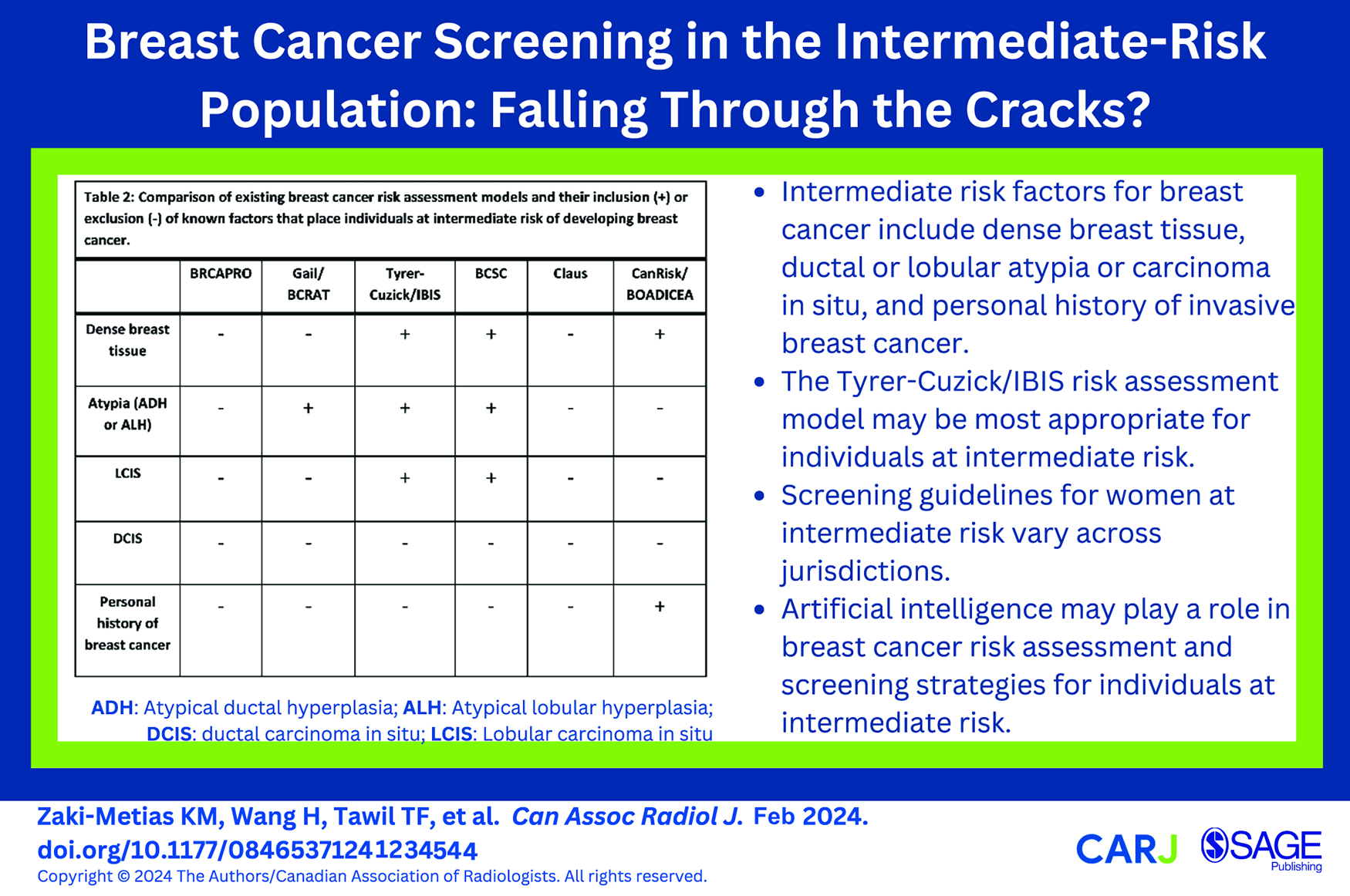
Introduction
Screening mammography reduces breast cancer-related morbidity and mortality.1,2 Significant strides have been made over the past 2 decades in developing recommendations for women (The authors recognize that gender is a spectrum, however, the term women is used when citing research as all data has been reported as women.) identified at high-risk for developing breast cancer. Guidelines recommend early screening with both digital mammography and breast magnetic resonance imaging (MRI) for those identified to be at 20% or greater lifetime risk of developing breast cancer.1-3 Women at higher-than-average risk for breast cancer may also have supplemental screening with ultrasound, including automated and handheld whole breast ultrasound (WBUS), and contrast-enhanced mammography (CEM).4,5 However, despite advances in technology and screening guidelines for the average and high-risk populations, there remains a gap in the literature and guidelines pertaining to screening those at intermediate risk of breast cancer.
Lifetime risk categories are designated as average, intermediate, and high risk and are utilized by current population-based screening guidelines to outline recommendations for screening (Table 1). Average-risk individuals have less than 15% lifetime risk of developing breast cancer, and high-risk individuals have greater than 20% risk of developing breast cancer during their lifetime. Individuals at intermediate risk of developing breast cancer are defined as those with 15% to 20% lifetime risk. 2 Risk factors that confer this degree of risk include dense breast tissue, a personal history of lobular carcinoma in situ (LCIS), atypical ductal hyperplasia (ADH), or atypical lobular hyperplasia (ALH), or a prior diagnosis of ductal carcinoma in situ (DCIS) or invasive breast cancer. 2 Personal history of DCIS, LCIS, ADH, or ALH are known risk factors for developing invasive breast cancer. Additionally, women with a personal history of DCIS have been found to have an increased long-term risk of developing invasive breast cancer and an increased risk of breast cancer-related mortality relative to the general population. 6
Lifetime Risk Categories Utilized in Breast Cancer Risk Stratification.

Dense breast tissue is an established independent risk factor for developing breast cancer; extremely dense breast tissue confers a 100% increased risk relative to women with less dense breast tissue.2,6-9 Individuals with dense breasts are categorized as intermediate risk, which is defined by the American Cancer Society (ACS) as a 15% to 20% lifetime risk of breast cancer.1,4,10 Using the American College of Radiology (ACR) BI-RADS categories, this includes breast density categories C (heterogeneously dense) and D (extremely dense).4,10 Currently, mammography is the sole imaging modality proven to decrease breast cancer-related mortality. However, increased breast density can lead to decreased interpretational accuracy and, therefore, decreased sensitivity for breast cancer with mammography. 1
The purpose of this manuscript is to: (1) conduct a review of several breast cancer risk assessment models in the context of intermediate risk and (2) summarize the existing global screening guidelines for individuals at intermediate risk.
Risk Assessment Models
Several studies have evaluated the performance of 6 risk assessment models: BRCAPRO, Gail/Breast Cancer Risk Assessment Tool (BCRAT), Tyrer-Cuzick/International Breast Cancer Intervention Study (TC/IBIS), Breast Cancer Surveillance Consortium (BCSC), Claus, and CanRisk/BOADICEA (Breast and Ovarian Analysis of Disease Incidence and Carrier Estimation Algorithm) (Appendix). Each of these breast cancer risk assessment models incorporates different variables to calculate risk, with each model focusing on varying aspects of risk. Some of these variables include dense breast tissue, personal history of DCIS, LCIS, ADH, or ALH, and history of invasive breast cancer. Table 2 summarizes inclusions and exclusions of these factors by each model.
Comparison of Existing Breast Cancer Risk Assessment Models and Their Inclusion (+) or Exclusion (-) of Known Factors That Place Individuals at Intermediate Risk of Developing Breast Cancer.
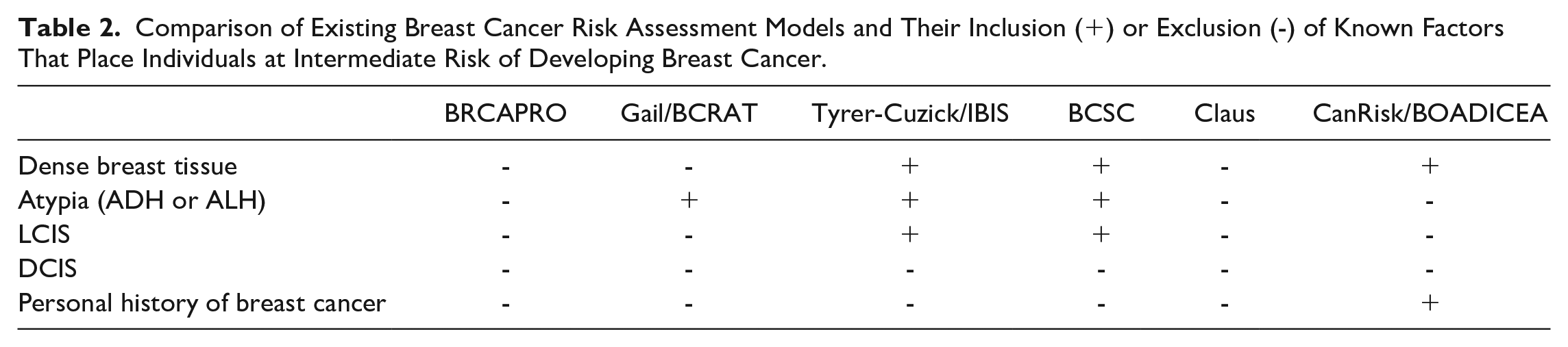
Previous research found the BRCAPRO model to be well calibrated among a population of Ashkenazi Jewish and non-Ashkenazi Jewish women, as well as in women with one first- or second-degree family member with breast cancer.3,6 However, the BRCAPRO model was found to overpredict the number of cancers in women without first- or second-degree family members with breast cancer, and for women with one first- or second-degree family member. It also underpredicted the number of cancers in women with more than 2 first- or second-degree family members with breast cancer.3,6 A strength of the BRCAPRO risk model is its ability to identify the probability of carrying a BRCA1/2 gene mutation. However, it should be noted that the model excludes other non-hereditary genetic risk factors.3,6 In relation to the intermediate risk group, the BRCAPRO risk model does not take breast tissue density or a personal history of DCIS, LCIS, atypia, or a personal history of breast cancer into account.3,11-13
The Gail/BCRAT risk assessment model was also found to be well calibrated in both Ashkenazi Jewish and non-Ashkenazi Jewish women, and for women with no breast cancer family history or with one first degree family member. 11 The Gail/BCRAT model was tested for Asian and Pacific Islander, Black, and Hispanic women using data from the Women’s Health Initiative. While the Gail/BCRAT model is the only model validated for use in Black women without a prior biopsy, it may underestimate risk in Black women with previous biopsies.14,15 Validation for other racial or ethnic groups, including Hispanic women, and risk among women with 2 or more affected family members is not yet available. 11 In Hispanic women born outside the United States, a study by Banegas et al 15 using the Hispanic Risk Model (HRM) has reported higher risk than the Gail/BCRAT risk model for foreign-born women. In relation to the intermediate risk group, the Gail/BCRAT risk assessment model considers a personal history of atypia but does not incorporate whether patients have personal history of invasive breast cancer, breast tissue density, or personal history of LCIS or DCIS.3,11-13
The TC/IBIS risk assessment model has been found to be well-calibrated for the Ashkenazi Jewish women. It was found to overpredict breast cancer risk in non-Ashkenazi Jewish women and the number of cancers in women with one or more affected family members. 11 Monticciolo et al 7 found that the TC/IBIS model was the most comprehensive and most consistently accurate of the risk assessment models, however, it is time-intensive for both patients and providers to complete. A study by Valero et al 16 found that the TC/IBIS risk assessment model overestimated the risk for women with prior LCIS and recommended that it not be used for this patient population. In the context of evaluating factors that would place an individual at intermediate risk of developing breast cancer, the TC/IBIS risk model considers breast density and personal history of LCIS or atypia.3,11-13 As the TC/IBIS risk model assesses the risk of developing both DCIS and invasive breast cancer, these factors are not included in risk assessment calculations.12,17
The BCSC model was found to have the highest discriminatory accuracy among women with known increased mammographic breast density. A study by Vachon et al 18 found that breast density and genetic variation in the BCSC are important risk factors in estimating breast cancer risk with the BCSC model. In relation to the intermediate risk group, the BCSC model considers factors including breast density and personal history of atypia and LCIS. It does not include personal history of DCIS or invasive breast cancer in the risk calculation.3,11-13
The Claus risk model was found to have 2 significant challenges in that it underpredicted the number of cancers but had higher positive and negative predictive values. The Claus model should only be used in individuals with a family history of breast or ovarian cancer.11-13 The Claus model considers first- and second-degree relatives, and does not consider any non-hereditary risk factors. 19 In relation to the intermediate risk group, the Claus risk model does not consider any of the factors placing individuals at intermediate risk including dense breast tissue, personal history of DCIS, LCIS, ADH, or ALH, or a personal history of breast cancer.3,11-13
The CanRisk/BOADICEA model estimates both lifetime breast cancer risk and probabilities of carrying loss-of-function variants that may predispose to breast cancer.20-22 This risk model has been found to be well-calibrated in women of European backgrounds and with Ashkenazi Jewish heritage; however, it has not been validated for Black, Asian, or Hispanic ethnicities and may not be reliable in these and other populations of non-European ancestry.20,23 In the context of intermediate risk factors, the CanRisk/BOADICEA model incorporates breast density and a personal history of invasive breast cancer, although only the risk of a contralateral breast cancer can be calculated in individuals with a history of invasive breast cancer.21,22 However, this model does not consider personal history of LCIS, ADH, or ALH, and should not be used to estimate risk in patients with a personal history of DCIS.3,12,13,24
Each model has unique strengths and weaknesses that may drive its use in specific populations, such as those at intermediate risk (Table 2). The TC/IBIS and BCSC models include most of the factors for the intermediate group (Table 2), suggesting that these models may be the most appropriate for use in the intermediate-risk population. The Claus and BRCAPRO models include none of the aforementioned intermediate risk factors and, as such, may be less suitable for comprehensive risk assessment in these individuals. Ultimately, the most appropriate risk assessment model depends on each individual’s specific risk factors and validation for use in that population.7,25
Risk Assessment Model Utilization
The American College of Radiology (ACR), Canadian Association of Radiologists (CAR), and Canadian Society of Breast Imaging (CSBI) recommend that all women should undergo a formal breast cancer risk assessment by the age of 30 to determine an optimal screening strategy and need for supplemental screening based on individual risk.4,9,26 This recommendation is emphasized for Black and Ashkenazi Jewish women, who are at higher risk of developing breast cancer at a younger age and for genetic mutations relative to the general population (ie, those at average lifetime risk). 9
Existing data suggests a gap in knowledge and education of primary care providers who refer their patients for screening mammography, with between 4% and 51% of referring providers utilizing breast cancer risk assessment models. Furthermore, there is a lack of understanding of the purpose of risk assessment models and the application of screening guidelines based on risk stratification. 27 Future work may consider exploring methods to increase awareness regarding the use and application of risk assessment models by referring physicians, in addition to examining barriers faced when adopting the use of such models (Table 2).28,29
Existing Guidelines for the Intermediate Risk Population
Breast cancer screening guidelines for patients at intermediate risk vary not only between jurisdictions, but also between agencies, provinces, territories, or states, within the same country. Although there are well-established recommendations for screening individuals at average and high risk, there are gaps in recommendations for those at intermediate risk.
United States of America
Recommendations regarding screening for individuals at intermediate risk in the United States of America are generally unclear, but the use of more advanced imaging modalities such as MRI may provide benefit, based on results of the American College of Radiology Imaging Network (ACRIN) 6666 trial.1,30 In 2007, the ACS recommended neither for nor against screening MRI in women with intermediate risk for breast cancer given limited supporting evidence. Since then, studies have found that women at intermediate risk may benefit with supplemental screening such as with breast MRI. 2 In 2009, many states suggested women with dense breasts discuss supplemental screening with their physicians after the passing of breast density notification legislation in the state of Connecticut. 2 In 2018, the ACR recommended annual screening breast MRI for individuals with dense breast tissue, citing the increased risk for developing breast cancer among women with dense breasts.3,10 Digital breast tomosynthesis (DBT), also known as 3D mammography, has been shown to increase cancer detection rates in women with increased breast density compared to digital 2D mammography alone. 31 WBUS has been demonstrated to decrease interval cancer detection rates and, while it is less sensitive than breast MRI, it can be used in settings where breast MRI is contraindicated, cannot be performed, or is inaccessible. 31 Of note, supplemental ultrasound following a negative screening mammogram may increase the false positive rate and lower the screening positive predictive value, so an informed discussion and shared decision-making between a patient and their physician is recommended to discuss potential risks and benefits of supplemental WBUS after negative screening mammography. 31 Similarly, the United States Preventive Services Task Force (USPSTF) states that while there is data suggesting ultrasonography, MRI, or DBT may detect additional breast cancer in individuals with dense breasts, there is still insufficient evidence to recommend this for screening when taking into account the benefits of increased sensitivity and the potential harms of increased false positive test results.32-34
In May 2023, the ACR updated their recommendations regarding women at intermediate risk. Individuals with dense breasts, a history of LCIS, ADH, or ALH, or those with a prior diagnosis of breast cancer with dense breast tissue are now recommended to undergo annual supplemental screening MRI. 9 For intermediate-risk women in whom MRI is contraindicated or not available, CEM may be a suitable alternative supplemental screening modality, however, further studies including the Contrast-Enhanced Mammography Imaging Screening Trial (CMIST) will shed light on the utility of CEM for screening and performance relative to DBT. 9 The ACR indicated that DBT and screening ultrasound, both handheld and automated, showed no difference in average- and intermediate-risk women with dense breast tissue.9,35
Canada
In 2018, the Canadian Task Force on Preventive Health Care (CTFPHC) released their recommendations on breast screening which remained unchanged from the recommendations made in 2011. However, there are shortcomings to the CTFPHC recommendations, such as the exclusion of recent observational research and newly demonstrated flaws in one of the large randomized controlled trials. 4 The CAR recommendations, endorsed by the CSBI, are annual screening of asymptomatic individuals beginning age 40, and for those with dense breasts (BI-RADS categories C and D), supplemental screening with ultrasound or CEM should be offered, where available. 4 The CAR recommendations do acknowledge that an optimal timeline has not yet been established for supplemental screening. 4 CEM has been found to have increased sensitivity relative to full-field digital mammography in the intermediate-risk population with dense breasts (BI-RADS categories C and D). 5 Furthermore, the use of CEM in this population may preclude the need for supplemental screening with ultrasound, as no added benefit has been demonstrated. 5 CEM is almost as sensitive as MRI, much less expensive, and more accessible, however, it has not yet been approved by Health Canada for breast cancer screening. 4 DBT may be considered as a useful tool in patients with dense breast tissue and is recommended for use in routine screening; however, it is not recommended as a supplemental screening modality in place of MRI or ultrasound. 4
The Clinical Advisory Committee on Breast Cancer Screening and Prevention, containing experts from Ontario and Québec, recommends individuals with an intermediate risk of breast cancer, which they define as 17% to 30%, receive a screening mammogram every 1 to 2 years beginning at age 40. 36 If their breast density is determined to be greater than 75%, akin to BI-RADS category D (extremely dense), an annual mammogram is recommended with the suggestion to consider complementary annual ultrasound and clinical breast examination. 36
In September 2023, the CAR published updated breast imaging referral guidelines addressing the need for supplemental screening for those at intermediate risk. In women with extremely dense (ACR BI-RADS category D) and heterogeneously dense (ACR BI-RADS category C) breast tissue, supplemental screening is recommended, although no specific screening modality is suggested given variations in regional resources and capacity limitations. 26 Women with previous diagnosis of a high-risk lesion (LCIS, ADH, or ALH) and patients with a history of breast cancer with breast conserving surgery are recommended annual screening with digital mammography or DBT, with the addition of annual screening MRI or ultrasound (if MRI is contraindicated) for those with extremely dense breast tissue. 26
Europe
Breast cancer screening guidelines are variable among the various European countries. However, the European Commission Initiative on Breast Cancer (ECIBC) does not recommend that individuals with increased breast density, who are therefore at intermediate risk of breast cancer, undergo automated breast ultrasound (ABUS), conventional hand-held ultrasound, or MRI over mammography alone. 33 The ECIBC addresses the possibility of tailored screening with DBT in addition to 2D mammography in women with intermediate risk but does not give a recommendation. 33 The European Society of Breast Imaging (EUSOBI) guidelines, updated in 2022, recommend supplemental screening breast MRI be offered every 2 to 4 years for women with extremely dense breasts (BI-RADS category D) between the ages of 50 and 70.4,37 However, for individuals with heterogeneously dense breasts (BI-RADS category C), they state that there is not enough evidence to recommend supplemental screening MRI in this population.4,37
United Kingdom
In the United Kingdom, mammograms are offered every 3 years in the National Health Service Breast Screening Programme (NHSBSP) between ages 50 and 70. 38 In 2019, an external review was completed for the UK National Screening Committee regarding breast density and supplemental screening with ultrasound, however, no changes to the current guidelines were made, citing issues with cost effectiveness and consistent methods of density measurement. 39
Australia
Australia’s national screening program BreastScreen offers biennial mammography to individuals aged 50 to 74. 40 BreastScreen is age-based, however supplemental screening is available in some jurisdictions based on risk factors such as history of benign breast disease, family or personal history of breast or ovarian cancer, or genetic markers indicating higher risk of breast cancer.40,41 Outside of the BreastScreen program, the Australian government’s eviQ guidelines recommend annual screening mammography for women aged 40 to 49 at intermediate risk (17%-30%) with supplemental screening including MRI, DBT, or ultrasound.40,42
The screening guidelines for individuals at intermediate risk vary between jurisdictions (Table 3). There is significant heterogeneity within the guidelines and this lack of consensus can be cause for confusion, and therefore create additional barriers to adequate screening for individuals at intermediate risk. There is an opportunity for significant improvement by creating consensus around the guidelines for screening individuals at intermediate risk of breast cancer.
Comparison of Existing Breast Cancer Screening Guidelines for Women at Intermediate Risk of Breast Cancer and Their Inclusion (+) and Exclusion (-) Various Supplemental Screening Modalities by Country/Region.
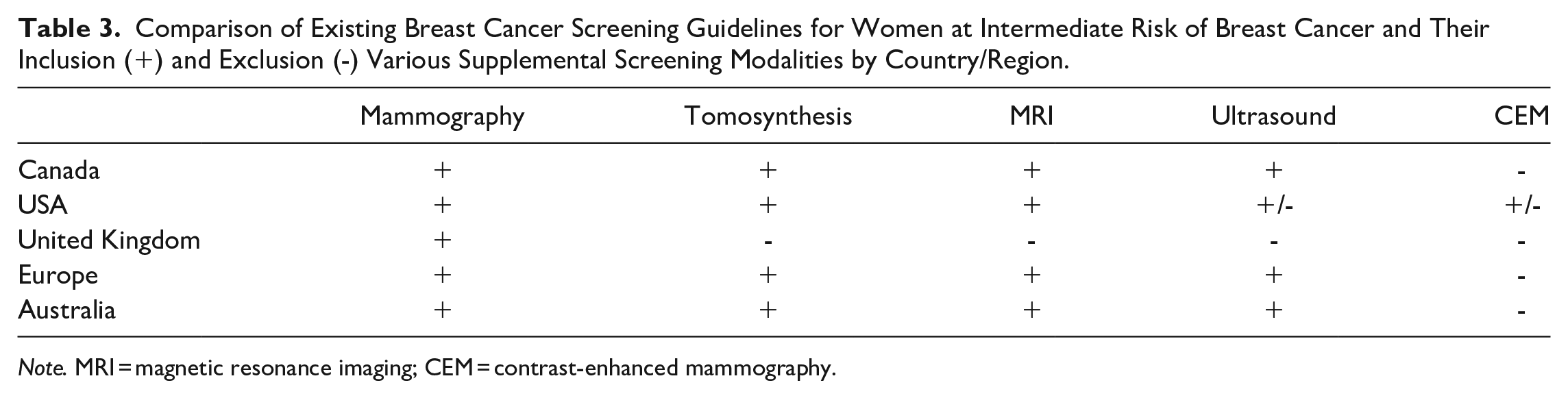
Note. MRI = magnetic resonance imaging; CEM = contrast-enhanced mammography.
Future of Breast Cancer Screening in the Intermediate-Risk Population
Continued research is necessary to improve guidelines for the population at intermediate risk for developing breast cancer. Further development of a risk-adjusted, personalized screening approach can be especially beneficial for individuals with intermediate risk for breast cancer. 43 To achieve this, future studies evaluating and improving risk models are required. Existing risk models have been evaluated based on 6-year risk and additional studies are required for longer-term risk estimates. 11
Ongoing studies assessing breast cancer risk assessment, including MyPebs in Europe, WISDOM in the USA, and PERSPECTIVE in Canada, are evaluating a novel approach to breast cancer screening based on an individual’s personal risk, compared to the current method of population age-based screening.44-46
The evolving nature of individual risk should also be considered. The risk of developing breast cancer can evolve throughout the various stages of an individual’s life. Currently, professional societies recommend women undergo formal breast cancer risk assessment prior to the age of 30. However, the lifetime risk calculated at age 30 may change considerably in the following decade prior to the initiation of mammographic screening for those deemed average risk. For example, a genetic mutation or new breast cancer may be discovered in an immediate family member. An individual’s risk may further change throughout the screening years, or with subsequent pregnancies or menopause.
Further research on screening modalities, including their sensitivity and specificity in women with dense breasts, is also warranted. The use of artificial intelligence (AI) to determine breast density offers another opportunity for improvement because visual assessment of breast density is limited by inter- and intra-reader variability.8,25,40 Available data on AI-based risk prediction tools suggest that they are already proving a useful supplement and outperform traditional risk assessment models.43,47,48 Future work exploring the limitations of risk assessment by demographic characteristics is ongoing, as are investigations into the logistics and barriers to AI implementation and widespread adoption of risk-based screening versus population-based screening. Furthermore, ongoing research is exploring the integration of polygenic risk scores and single nucleotide polymorphisms (SNPs) into personalized risk assessment, prognostication, and treatment of breast cancer overall and of specific subtypes.49,50 Finally, it is important to maintain the participation of experts in breast cancer diagnosis and treatment, including radiologists, in research regulation and design, as the lack of input from radiologists and other breast cancer experts has been an issue cited in critiques of some screening guidelines.32,41
Conclusion
Individuals with factors placing them at intermediate risk of developing breast cancer do not fit into existing screening guidelines for the average or high-risk population. Geographic jurisdictions’ and professional societies’ screening recommendations vary considerably for the intermediate-risk population. Some of the currently available breast cancer risk assessment models consider factors such as breast density and personal history of atypia or carcinoma in situ. However, none of the models are truly comprehensive in assessing intermediate risk. Future work on the integration of AI-based technologies with risk assessment models may enhance our ability to risk stratify patients and offer appropriate screening guidelines. Furthermore, the involvement of radiologists as clinical content experts in breast cancer screening, diagnosis, and treatment in future research and development of breast cancer screening guidelines is critical to ensure the best preventative care for patients and to close the gap for oft-overlooked populations such as those at intermediate risk.
Footnotes
Appendix
Summary of the Variables Considered by the Various Breast Cancer Risk Assessment Models.
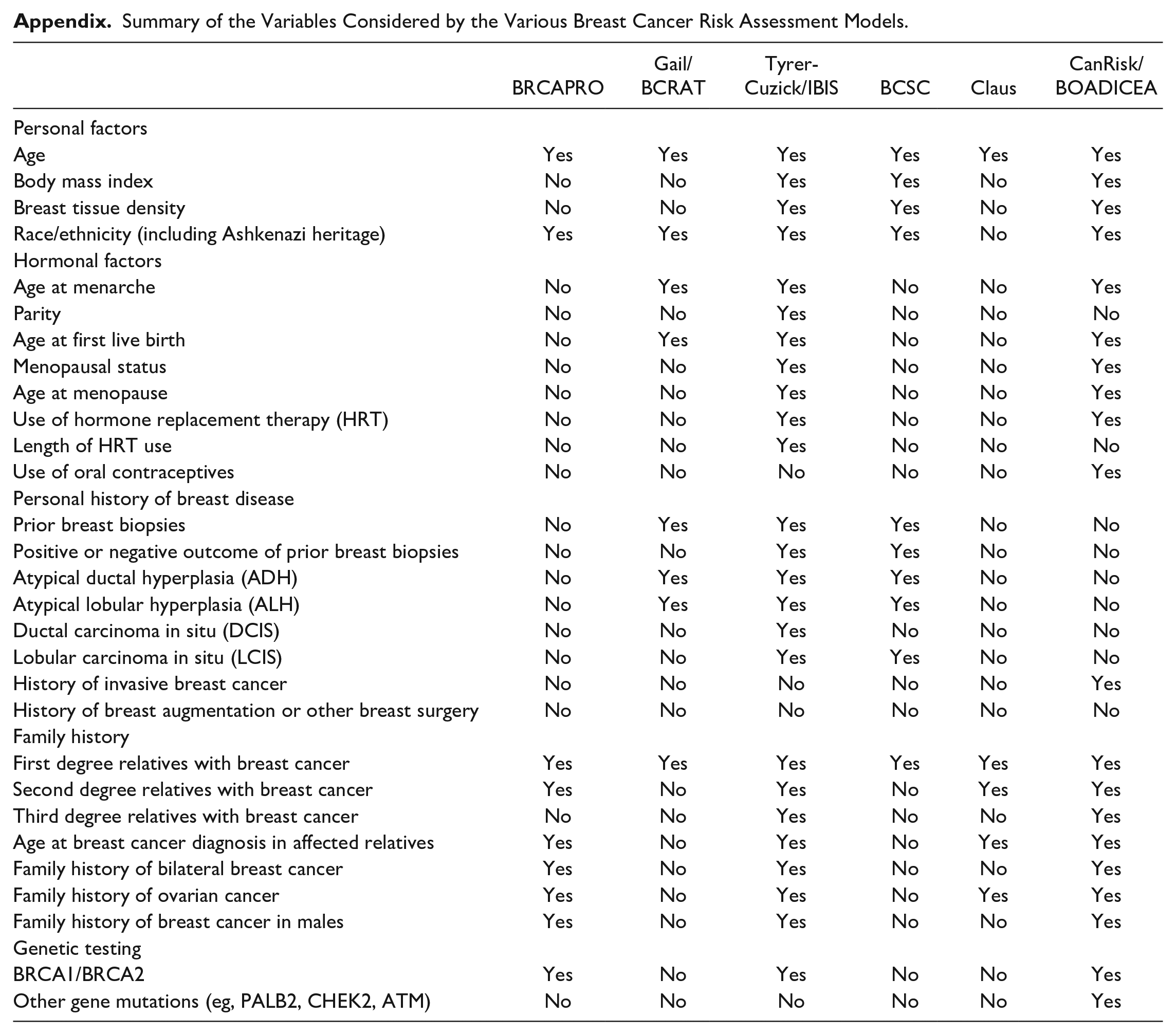
| BRCAPRO | Gail/BCRAT | Tyrer-Cuzick/IBIS | BCSC | Claus | CanRisk/BOADICEA | |
|---|---|---|---|---|---|---|
| Personal factors | ||||||
| Age | Yes | Yes | Yes | Yes | Yes | Yes |
| Body mass index | No | No | Yes | Yes | No | Yes |
| Breast tissue density | No | No | Yes | Yes | No | Yes |
| Race/ethnicity (including Ashkenazi heritage) | Yes | Yes | Yes | Yes | No | Yes |
| Hormonal factors | ||||||
| Age at menarche | No | Yes | Yes | No | No | Yes |
| Parity | No | No | Yes | No | No | No |
| Age at first live birth | No | Yes | Yes | No | No | Yes |
| Menopausal status | No | No | Yes | No | No | Yes |
| Age at menopause | No | No | Yes | No | No | Yes |
| Use of hormone replacement therapy (HRT) | No | No | Yes | No | No | Yes |
| Length of HRT use | No | No | Yes | No | No | No |
| Use of oral contraceptives | No | No | No | No | No | Yes |
| Personal history of breast disease | ||||||
| Prior breast biopsies | No | Yes | Yes | Yes | No | No |
| Positive or negative outcome of prior breast biopsies | No | No | Yes | Yes | No | No |
| Atypical ductal hyperplasia (ADH) | No | Yes | Yes | Yes | No | No |
| Atypical lobular hyperplasia (ALH) | No | Yes | Yes | Yes | No | No |
| Ductal carcinoma in situ (DCIS) | No | No | Yes | No | No | No |
| Lobular carcinoma in situ (LCIS) | No | No | Yes | Yes | No | No |
| History of invasive breast cancer | No | No | No | No | No | Yes |
| History of breast augmentation or other breast surgery | No | No | No | No | No | No |
| Family history | ||||||
| First degree relatives with breast cancer | Yes | Yes | Yes | Yes | Yes | Yes |
| Second degree relatives with breast cancer | Yes | No | Yes | No | Yes | Yes |
| Third degree relatives with breast cancer | No | No | Yes | No | No | Yes |
| Age at breast cancer diagnosis in affected relatives | Yes | No | Yes | No | Yes | Yes |
| Family history of bilateral breast cancer | Yes | No | Yes | No | No | Yes |
| Family history of ovarian cancer | Yes | No | Yes | No | Yes | Yes |
| Family history of breast cancer in males | Yes | No | Yes | No | No | Yes |
| Genetic testing | ||||||
| BRCA1/BRCA2 | Yes | No | Yes | No | No | Yes |
| Other gene mutations (eg, PALB2, CHEK2, ATM) | No | No | No | No | No | Yes |
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
