Abstract
Fungal rhinosinusitis (FRS) includes non-invasive and invasive subtypes with the latter having significant morbidity and mortality. This systematic review aims to identify the imaging features most correlated with invasive fungal rhinosinusitis (IFRS) and present a checklist of these features to aid diagnosis. PubMed, Embase, CENTRAL, and Science Direct were searched from inception to May 2023, in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. Primary research articles published in English describing the imaging features of IFRS were included. The systematic review was conducted in accordance with the PRISMA guidelines. Forty-eight articles were identified for inclusion. Six studies examined radiological features in acute invasive fungal rhinosinusitis (AIFRS), and 9 studies of chronic invasive fungal rhinosinusitis (CIFRS). A majority of studies did not specify whether IFRS cases were acute or chronic. On CT, bony erosion and mucosal thickening were the most common features. Other features include nasal soft tissue thickening, nasal cavity opacification, opacification of the affected sinus, and perisinus soft tissue infiltration. Extra-sinus extension was commonly observed on MRI, most often invading intraorbitally and intracranially. Other sites of extra-sinus extension included the cavernous sinus, pterygopalatine fossa, infratemporal fossa, masticator space, and facial soft tissue. IFRS is a condition with potential for high morbidity and mortality. Several radiological features are highly suggestive of IFRS. Early identification of high-risk radiological features using a checklist may aid prompt diagnosis and early treatment. Future research investigating the radiological differentiation between IFRS and other significant pathology including bacterial orbital cellulitis would be beneficial.
Introduction
Fungal rhinosinusitis (FRS) is a broad term encompassing a disease spectrum ranging from fungal colonization of the nasal cavity and paranasal sinuses to invasive and potentially fatal disease with extra-sinus extension. FRS has become increasingly prevalent over the past 2 decades in the context of increased awareness and the high prevalence of diabetes.1,2
FRS may be acquired incidentally or iatrogenically. Fungal spores are commonly inhaled, and if not repelled by the respiratory mucociliary escalator are then phagocytosed by macrophages and dendrites. However, in immunocompromised individuals fungal spores may invade and proliferate within the mucosa, resulting in invasive disease. Invasion of neurovascular structures may progress to thrombosis and ischemia, enabling further spread beyond the sinuses into adjacent soft tissue and bony structures. Additionally, prior surgery or mucosal injury may also allow seeding of spores.3,4
The most recognized classification system, initially formulated by DeShazo et al in 1997 and further defined by The International Society for Human and Animal Mycology, categorizes FRS into invasive and non-invasive types based on histopathological evidence of fungal invasion into host tissue. Invasive subtypes include acute invasive (also known as fulminant) FRS (AIFRS), chronic invasive FRS (CIFRS), and chronic granulomatous invasive FRS (CGIFRS). Aspergillus and mucoraceae are most commonly implicated in AIFRS, while the majority of CIFRS AND CGIFRS are secondary to aspergillus. Saprophytic fungal infestation, fungal ball, and eosinophilic FRS, which includes allergic fungal rhinosinusitis (AFRS), represent non-invasive subtypes.5,6
Invasive FRS (IFRS) is a condition with high morbidity and mortality. The diagnosis is made through a combination of clinical assessment, radiology, and pathology, with a gold standard of histopathological analysis on biopsy. Accurate radiological identification may facilitate early diagnosis, allowing prompt treatment. We aim to identify the imaging features most strongly correlated with IFRS and present a checklist of these features to aid diagnosis.
Method
A systematic search of the databases PubMed/MEDLINE, Embase, CENTRAL, and Science Direct was conducted from inception to May 2023, in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 2020 guidelines. Clinical trials registries, as well as the reference lists of relevant articles were also searched. MeSH terms and keywords utilized in the search included “fungal sinusitis,” “acute fungal sinusitis,” “chronic fungal sinusitis,” “unilateral fungal sinusitis,” “bilateral fungal sinusitis,” “mucormycosis sinus disease,” “findings,” “signs,” “features,” observations,” and “manifestations.”
Inclusion criteria were primary research articles examining a population diagnosed with invasive FRS with inclusion of relevant imaging findings, published in the English language, with a full-text version available. Conference abstracts, reviews, case reports, and editorial articles were excluded. The risk of bias of each included article was assessed using the Critical Appraisal Skills Programme (CASP) checklist tool. Determination of eligibility, risk of bias evaluation, and data extraction were performed in duplicate (A.C. and J.P.). Ethics approval was gained from the Central Adelaide Local Health Network (CALHN) Research Ethics Committee (reference number 17939).
Results
A total of 3734 articles were identified during the search. After abstract screening and application of eligibility criteria, 48 relevant articles were identified for inclusion. (Supplemental File 1). Risk of bias for all included studies was variable (Supplemental File 2).
Invasive Fungal Sinusitis
Acute Invasive Fungal Sinusitis
Acute IFRS (AIFRS) is defined as the presence of fungal hyphae within sinonasal mucosa, submucosa, vasculature or bone, in the setting of 1 month or less of sinusitis symptoms.5,6 AIFRS often affects diabetic or immunosuppressed patients, although it has been reported in the immunocompetent population. Symptoms of sinusitis such as nasal obstruction, rhinorrhea, facial pain, fever, and headache are typical. Symptoms of orbital or intracranial extension include proptosis and diplopia.7,8 Prompt diagnosis is essential to prevent fungal invasion into surrounding structures including the orbit and central nervous system and associated high morbidity and mortality rates. 1 Age and sex characteristics of included study cohorts are detailed in Table 1.
Age and Sex Characteristics of IFRS Cohorts.

Imaging
Computed Tomography
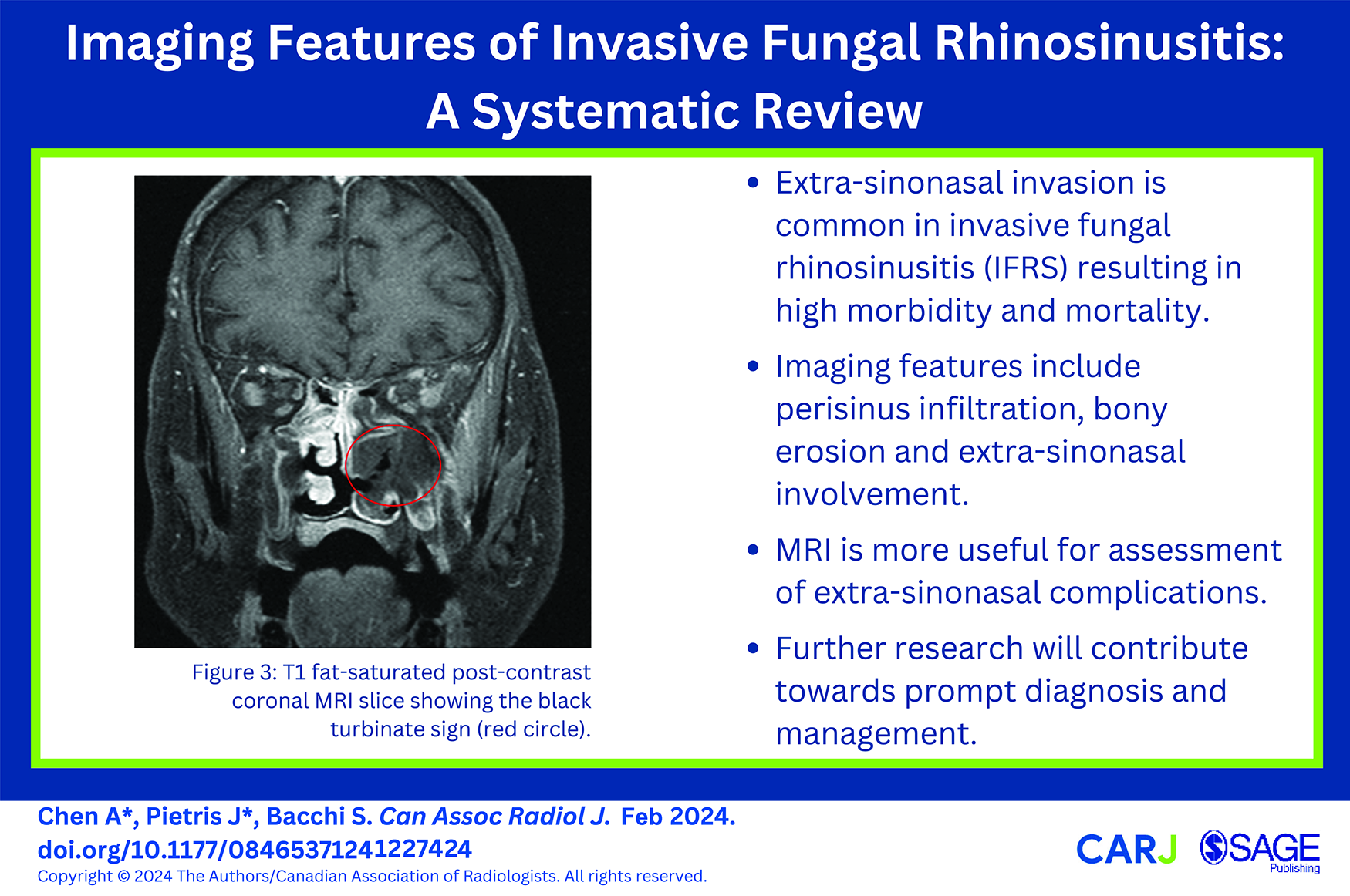
Nine included studies analyzed the radiological features associated with AIFRS. On computed tomography (CT), mucosal thickening and bony erosion are typical. Other associated findings include nasal soft tissue thickening, nasal cavity opacification, opacification of the affected sinus, perisinus soft tissue infiltration, and extra-sinus invasion (most commonly intraorbital or intracranial) (Figure 1). 3 CT provides good definition of bony structures and demonstrates subtle bony erosion and periostitis. However, infection may spread intraorbitally through small osseous vascular channels without frank bony destruction. 9 The sensitivity of bony destruction as a radiological characteristic of AIFRS is variable; among the included studies, reports of bony erosion ranged from 0% to 57% of cases.10-13 CT has limited sensitivity for assessment of extra-sinonasal involvement when compared to magnetic resonance imaging (MRI). 9 Given the combination of non-specific CT findings and the potential for paranasal sinus disease to appear mild, a high index of clinical suspicion for AIFRS should lower the threshold for further investigation such as MRI and endoscopy.
On the left is an axial CT at the level of the maxillary sinus demonstrating opacification of the right maxillary sinus and infiltration of the pre-antral fat (blue arrow) and, to a lesser extent, the retro-antral fat (yellow arrow). On the right is an axial CT bone window at the same level demonstrating subtle bony erosion (blue arrows) of the right maxillary sinus.
Magnetic Resonance Imaging
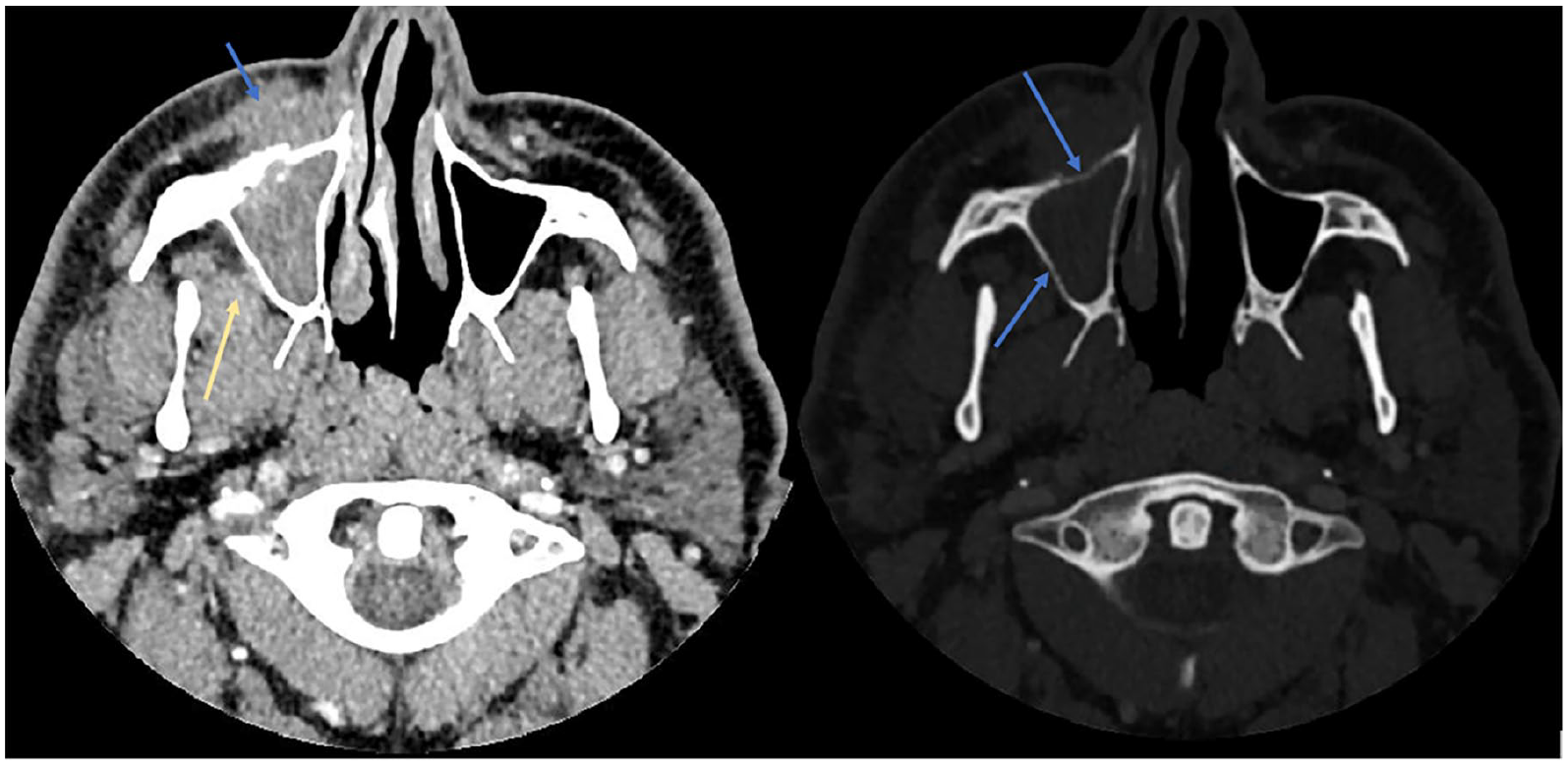
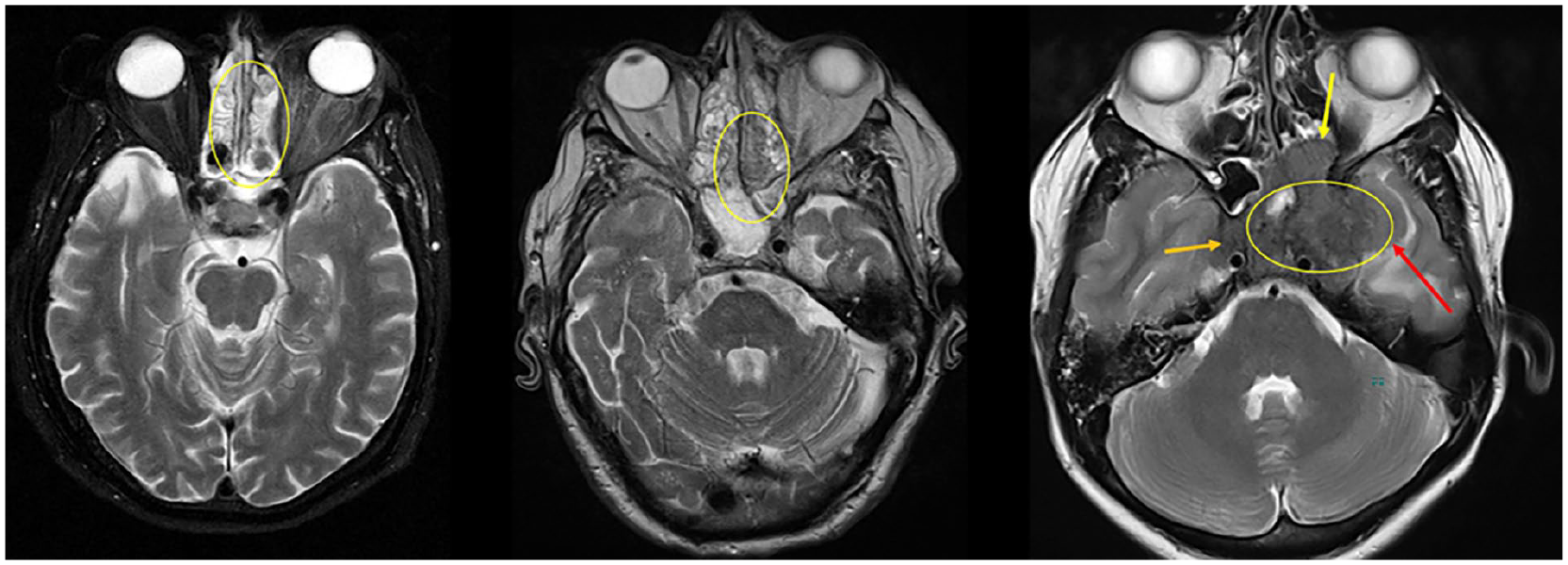
Classically, the appearance of fungal infection on MRI has been defined as both T1 and T2 hypointense. 14 Fungal hyphae are typically T2 hypointense, as they have paramagnetic properties. However, T2 signal intensity within the paranasal sinuses is variable as it is influenced by the protein and water content of secretions (Figure 2). Loss of contrast enhancement on MRI correlated with endoscopic findings of AIFRS in 77% of cases reported by Groppo et al, 15 and 48% of cases reported by Choi et al. 13 Kim et al 16 found that postoperative residual loss of contrast enhancement was associated with poorer outcomes. Non-enhancing hypointense mucosa (the “black turbinate sign”) indicates necrosis (Figure 3). However, Han and Escott 17 identified this sign in 30% of immunocompetent patients without IFRS. As such, the black turbinate sign should be interpreted only in conjunction with other radiological and clinical findings.
Axial T2 MRI slices demonstrating the spectrum of T2 signal intensity dependent on the hyphae content of secretions. On the left is T2 fluid-hyperintense IFRS, in the middle intermediate or heterogeneous T2 signal intensity, and on the right low T2 signal intensity.
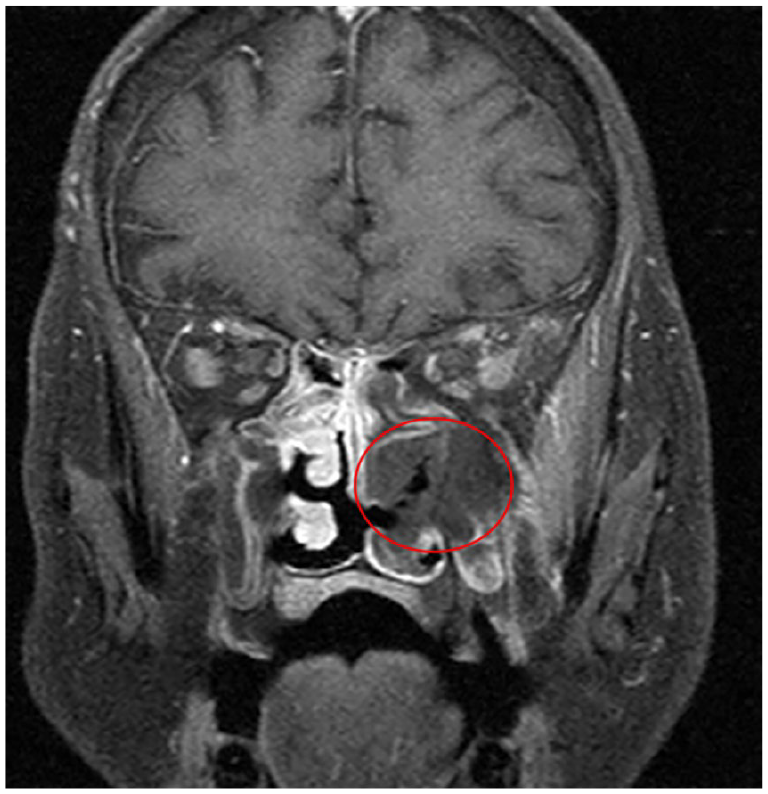
T1 fat-saturated post-contrast coronal MRI slice showing the black turbinate sign (red circle).
MRI is superior to CT for evaluation of extra-sinonasal invasion. The T1 weighted image (T1WI) is the key sequence for depicting infiltration of fat planes. Orbital apex involvement is a risk factor for poor visual outcomes. 18 Intracranial complications including meningitis, intracranial collection, encephalitis, dural venous sinus thrombosis, and cerebral infarct are well demonstrated on MRI. 9 The incidence of extra-sinonasal involvement for extra-sinonasal sites varies as follows: cavernous sinus 17% to 50% (Figure 4), intracranial 13% to 50%, ICA occlusion 13% (Figure 4), orbit 38% to 65%, and infratemporal fossa 39% to 74%.10,11,13,19,20
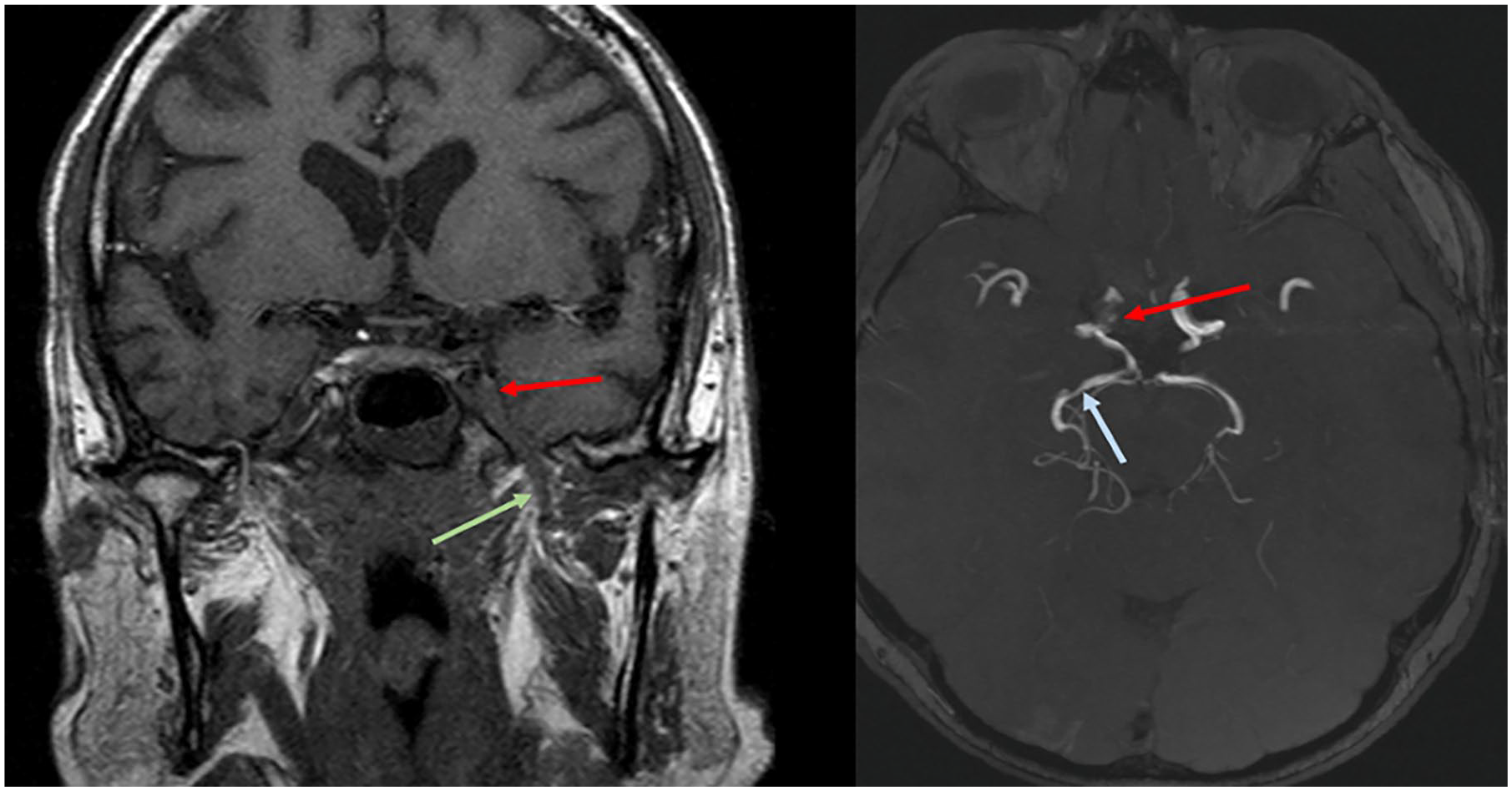
On the left, a coronal MRI slice shows spread of FRS disease into the cavernous sinus (red arrow) and perineural spread along the V3 division of the trigeminal nerve (green arrow). On the right, an axial post-contrast MRI slice demonstrates narrowing of the diseased right internal carotid artery (red arrow) and a skip lesion of the posterior cerebral artery more distally (blue arrow).
Chronic Invasive Fungal Rhinosinusitis
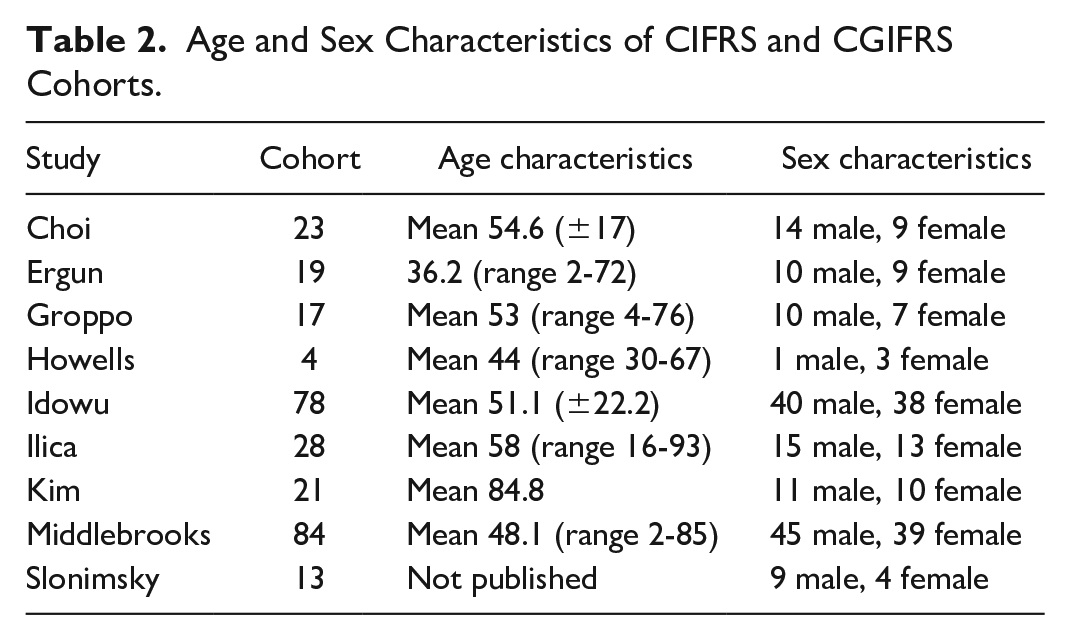
CIFRS is defined as an insidious destructive process occurring over more than 12 weeks. Histologically, it demonstrates dense aggregates of hyphae, vascular invasion, and chronic inflammation with involvement of local structures. Risk factors include immunosuppression, acquired immunodeficiency syndrome (AIDS), diabetes, and corticosteroid use. 5 Patients typically present with symptoms of chronic sinusitis. Additional features include facial swelling, visual impairment, and focal neurological deficits.8,21 Age and sex characteristics of included study cohorts of both CIFRS and CGIFRS are detailed in Table 2.
Age and Sex Characteristics of CIFRS and CGIFRS Cohorts.
Imaging
Computed Tomography
Six included studies described radiological features associated with CIFRS. The most common features of CIFRS appreciable on CT are bony erosion and hyper-attenuating soft tissue within the paranasal sinuses, which appears mass-like. 3 Sclerosis of the paranasal sinus wall is seen in 53% of cases. 22 Subtle perimaxillary fat infiltration is also associated with chronic invasive disease (Figure 5).3,21,23
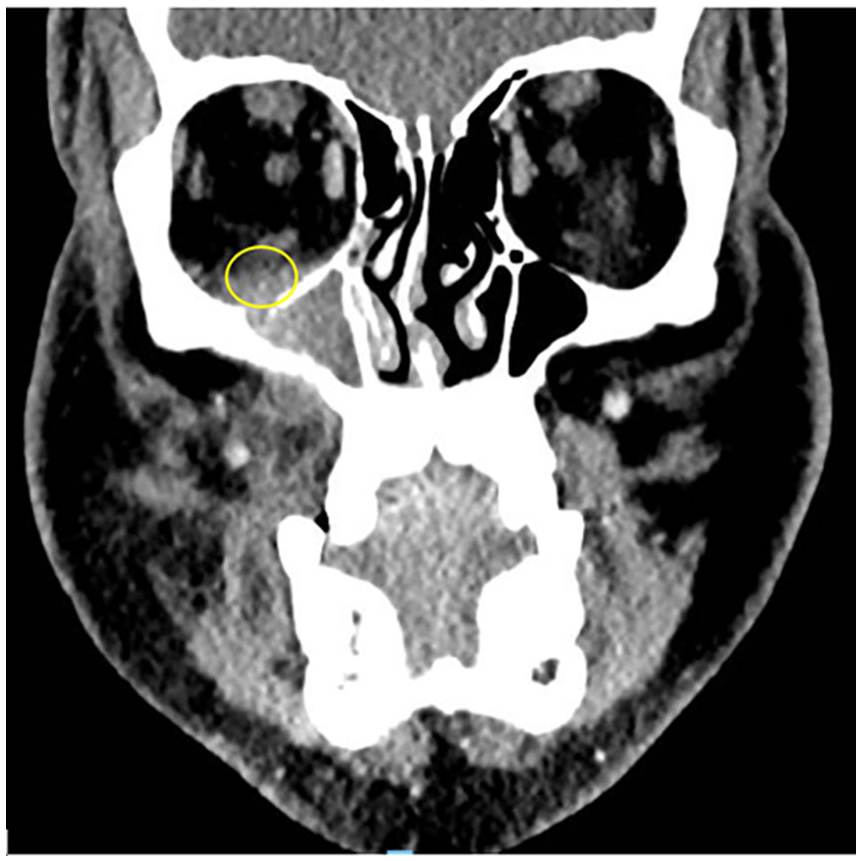
Coronal CT slice showing effacement of the peri-sinus fat plane intraorbitally but extraconally (yellow circle). This is a sensitive marker for IFRS.
Magnetic Resonance Imaging
As with AIFRS, T1 and T2 signals within the sinuses vary depending on the protein and water content of secretions, and presence of fungal elements. T2 hypointense fungal elements are reported in several cohorts of included studies, ranging from 49% to 94% of cases.22,24,25 Ill-defined T2 hyperintense edema is well demonstrated on fat suppressed MRI images. The presence of protein in secretions also increases the T1 signal. Hyperintensity of secretions is more typical of chronic rather than acute IFRS, and usually reflects heavy metal deposition.9,26
The maxillary and sphenoid sinuses are the most common sites of CIFRS infection. Extra-sinonasal sites of involvement include the orbit (ranging from 8% to 71% of cases), the pterygopalatine fossa (76%-100% of cases), and the masticator space (86% of cases). Additionally, the cavernous sinus (15%-70% of cases) and the internal carotid artery (12% of cases) are also commonly reported sites of involvement.22,24,27,28
Chronic Granulomatous Invasive Fungal Rhinosinusitis
It remains contentious whether CGIFRS is a distinct clinical entity from CIFRS. Clinical manifestations of CGIFRS are similar to CIFRS. Differentiation between the 2 is made on a histopathological basis, with CGIFRS defined by formation of non-caseating granulomas. CGIFRS typically onsets over 12 weeks or longer and incidence is higher in India, Pakistan, and Africa.3,5,25,29 See Table 2 for age and sex characteristics of included studies’ cohorts.
Imaging
Computed Tomography
There are limited published data on the radiological characteristics of CGIFRS. Zhou et al compared 11 cases of CGIFRS to 44 cases of CIFRS. Radiological features assessed on CT included bony destruction (27% of cases), orbital or optic nerve abnormality (27% of cases), and CNS abnormality (45% of cases). However, it is important to note the presence of these features did not have a statistically significant correlation with a diagnosis of CGIFRS. 27 Cho et al described 4 patients with CGIFRS. CT demonstrated a mass forming pattern, bony erosion, and sclerosis in all 4 patients. Fungal ball was seen in 50% of patients. A 2010 study by Reddy et al included patients with CGIFRS and CIFRS. In 17 patients with CGIFRS or CIFRS, CT demonstrated homogeneous isodensity to hyperdensity (compared to muscle) within the paranasal sinuses in 94% of patients. 25
Magnetic Resonance Imaging
Extra-sinus invasion was commonly found on MRI in a study published by Cho et al, 28 involving the orbit in 75%, pterygopalatine fossa in 75%, masticator space in 50%, and skull base in 75%. Similarly, Reddy et al 25 observed a lack of expansion of the sinuses in all patients, bony erosion in 94% of patients, homogeneous post contrast enhancement in 94%, and an extra-sinus component of disease exceeding the size of the intra-sinus component in 88%.
Discussion
The currently available literature identifies a range of radiological features associated with IFRS. On CT scans, bony erosion was the feature most commonly associated with IFRS, identified in 14 included studies. Mucosal thickening was also described in 9 included studies. MRI signals of patients with IFRS were most commonly isointense on T1 image sequences, and T2 hypointense. The black turbinate sign was associated with IFRS in 7 included studies. Extra-sinonasally, the orbit and the cavernous sinus were the most common sites of infection extension, described in 18 and 16 included studies respectively.
Radiological involvement of the orbit includes orbital apex enhancement in 9% to 90%, extraocular muscle involvement (47%-73%), orbital wall invasion (60%), orbital cellulitis (58%), globe proptosis (28%-54%), retro-orbital fat stranding (50%), superior ophthalmic vein thrombosis (10%-28%), optic neuritis (25%), ophthalmic artery thrombosis (14%), globe deformity (5%-12%), and orbital compartment syndrome (8%).33,37,39,44,45,47-49 Other orbital manifestations of IFRS described by Hada et al 44 globe tenting, subperiosteal collections, intraorbital collections, and optic neuropathy secondary to optic nerve involvement.
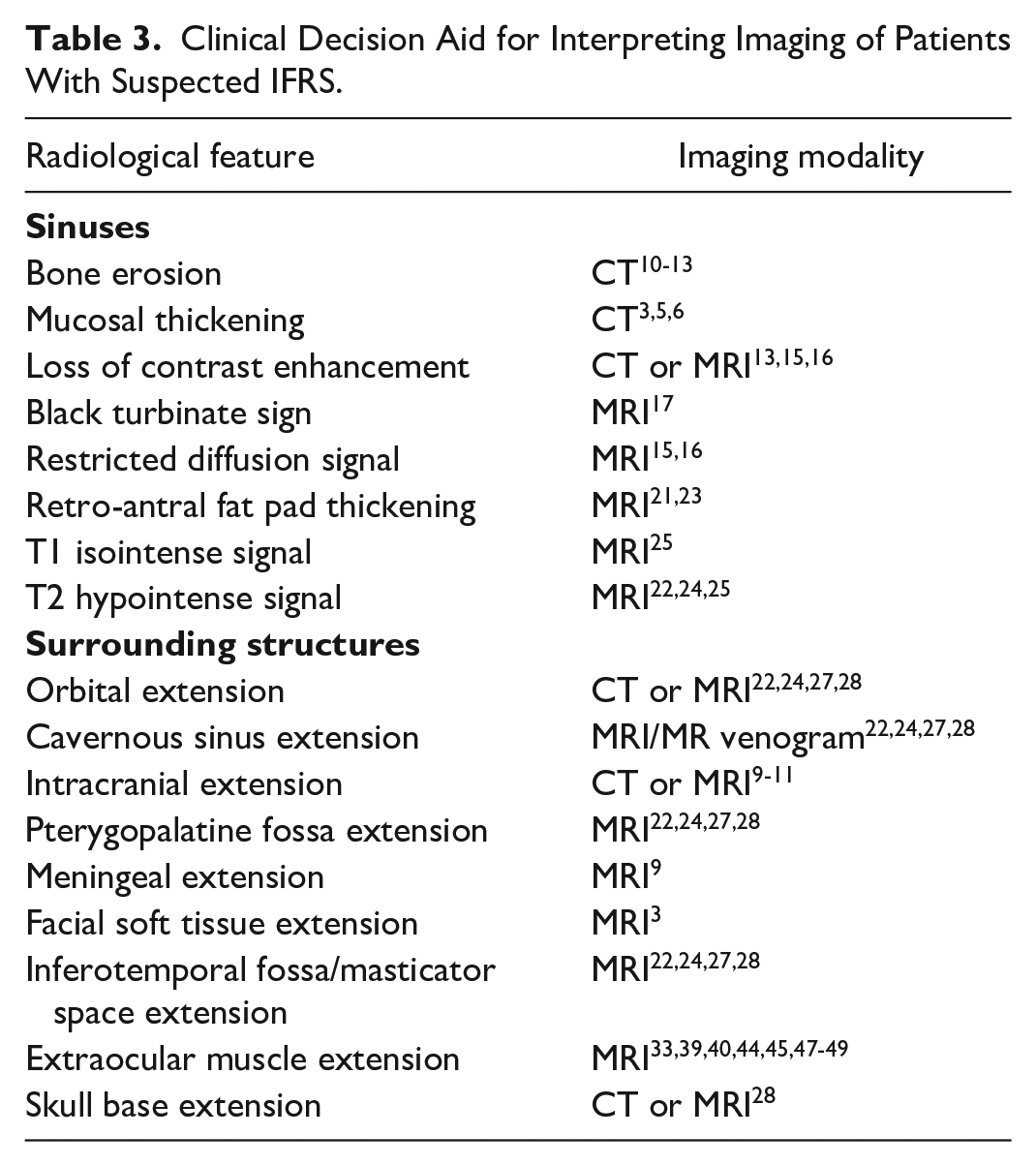
Table 3 lists the most common radiological features in IFRS which can be utilized as a checklist to assist clinicians in diagnosis.
Clinical Decision Aid for Interpreting Imaging of Patients With Suspected IFRS.
Rhino-Orbital-Cerebral Mucormycosis in the COVID-19 Era
Since the COVID-19 pandemic engulfed the world in early 2020, the world has seen a surge in cases of fungal infections of the head and neck, primarily rhino-orbital-cerebral mucormycosis (ROCM).30-36 This surge has largely been attributed to the pre-existing pandemic of poorly controlled diabetes, as well as an uptick in corticosteroid and oxygen use as part of the COVID-19 infection management algorithm. 52 Many recent studies analyze the imaging features of IFRS in the setting of active or recent COVID-19 infection. Comparisons have been drawn between these COVID-associated cases of IFRS and AIFRS, however, the majority of studies have not definitively categorized COVID-associated IFRS as a subtype of AIFRS or as a separate clinical entity.
Maxillary and ethmoid sinuses are the most commonly involved infection sites.37-40 Other sites of involvement include the orbit (27%-92% of cases), intracranial (2%-60% of cases), the cavernous sinus (8%-100% of cases), the infratemporal fossa (21%-71% of cases), and the internal carotid artery in 6% to 60% of cases.38,39,41-51 The distinction between imaging modalities for identifying COVID-associated ROCM is poorly defined in the current literature.
Imaging features are similar to those associated with IFRS, including loss of contrast enhancement in 28% to 65% of cases, and bony erosion in 33% to 88% of cases.40-43,47,48,52,53 There are limited data comparing the imaging findings of isolated ROCM with COVID-associated ROCM.
Multiple staging systems for ROCM severity have been proposed. Kumar et al and Singh et al both proposed similar 3 tier staging systems for COVID-associated ROCM, categorizing cases based on sinonasal involvement, orbital involvement, and central nervous system involvement.45,54 Kumar et al 55 proposed a scoring system based on MRI findings, consisting of a total of 60 points including 20 points for sinonasal, 20 points for orbital, 10 points for soft tissue, and 10 points for cerebral involvement. Patil et al 52 proposed a similar points-based system of based on sinus involvement, marrow edema, orbital and periorbital extension, optic nerve involvement, and intracranial extension.
Limitations and Future Research
This systematic review has several limitations. Publication bias may be introduced as studies with significant findings are more likely to be published. Many included studies examined populations of small sample size. The variable risk of bias of the included studies also contributes to the overall risk of bias of this review. There is significant heterogeneity among the cohorts analyzed in each included study. The reporting of radiological features of IFRS varies considerably between studies, limiting the ability to compare results and the subsequent generalizability of conclusions drawn. Bacterial sinusitis is not within the scope of this review. This represents a significant limitation.
Further research is required to better define differentiation criteria for the diagnosis of CIFRS and CGFRS. From a clinical perspective, analysis of imaging features that may aid in reliably differentiating IFRS from other pathology such as bacterial orbital cellulitis, inflammatory conditions such as IgG4-related orbital inflammation, and infiltrative or malignant pathology will aid the decision making of clinicians when triaging the urgency for biopsy and surgical management.
Conclusion
FRS can be classified into invasive and non-invasive types, with 5 subtypes including AIFRS and CIFRS. Imaging findings on CT most strongly associated with IFRS are bony erosion, mucosal thickening, and sclerosed sinus walls. On MRI, T1 signal isointensity and T2 signal hypointensity are frequently observed. Extra-sinonasal extension of both acute and chronic IFRS is common and is best demonstrated on MRI. Extension most often affects the orbit, manifesting in a variety of radiological features including orbital apex syndrome and extraocular muscle enlargement. In cases suspicious for IFRS, clinicians should have a low threshold for further investigation with endoscopy and biopsy. We present a simple radiological checklist to aid imaging interpretation in such cases. Future research should target radiological differentiation between IFRS and other significant pathology including bacterial orbital cellulitis.
Supplemental Material
sj-docx-1-caj-10.1177_08465371241227424 – Supplemental material for Imaging Features of Invasive Fungal Rhinosinusitis: A Systematic Review
Supplemental material, sj-docx-1-caj-10.1177_08465371241227424 for Imaging Features of Invasive Fungal Rhinosinusitis: A Systematic Review by Anni Chen, James Pietris, Stephen Bacchi, WengOnn Chan, Alkis J. Psaltis, Dinesh Selva and WanYin Lim in Canadian Association of Radiologists Journal
Supplemental Material
sj-docx-2-caj-10.1177_08465371241227424 – Supplemental material for Imaging Features of Invasive Fungal Rhinosinusitis: A Systematic Review
Supplemental material, sj-docx-2-caj-10.1177_08465371241227424 for Imaging Features of Invasive Fungal Rhinosinusitis: A Systematic Review by Anni Chen, James Pietris, Stephen Bacchi, WengOnn Chan, Alkis J. Psaltis, Dinesh Selva and WanYin Lim in Canadian Association of Radiologists Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
