Abstract
The liver, spleen, and kidneys are the commonest injured solid organs in blunt and penetrating trauma. The American Association for the Surgery of Trauma (AAST) Organ Injury Scale (OIS) is the most widely accepted system for categorizing traumatic injuries. Grading systems allow clear communication of findings between clinical teams and assign a measurable severity of injury, which directly correlates with morbidity and mortality. The 2018 revised AAST OIS emphasizes reliance on CT for accurate grading; in particular regarding vascular injuries. Dual-Energy CT (DECT) has emerged as a promising tool with multiple clinical applications already demonstrated. In this review article, we summarize the basic principles of CT attenuation to refresh the minds of our readers and we scrutinize DECT’s technology as opposed to conventional Single-Energy CT (SECT). This is followed by outlining the benefits of various DECT postprocessing techniques, which authors of this article refer to as the 3Ms (Mapping of Iodine, Material decomposition, and Monoenergetic virtual imaging), in aiding radiologists to confidently assign an OIS as well as problem solve complex injury patterns. In addition, a thorough discussion of changes to the revised AAST OIS focusing on definitions of key terms used in reporting injuries is described.
This is a visual representation of the abstract.
Introduction
Trauma is the leading cause of death in the younger population (<45 years of age) and the fourth overall leading cause of death. 1 Clinical examination in polytrauma patients is exceedingly unreliable and imaging advancement has led multidetector CT to become the imaging modality of choice in hemodynamically stable trauma patients. 1 This is evident in its ability not only to accurately diagnose the various spectrum of injuries but also help inform therapeutic decisions, for example, in cases of active hemorrhage. Single Energy Computed Tomography (SECT) has long been validated as the reference standard diagnostic imaging modality in acute clinical settings.2,3 Its accessibility and high diagnostic accuracy have made it a first-line tool in major emergency departments (EDs) worldwide.4,5 Most abdominal SECT scans are acquired at 120 kVp, producing a polyenergetic X-ray beam with an average energy of 75 to 76 keV and an energy peak of 120 kVp. 6 Dual Energy Computed Tomography (DECT) has been used in clinical practice since 2006, with some of the initial limitations compared with SECT, such as relatively increased radiation dose, excessive noise, inferior spatial resolution, and longer acquisition time, addressed by advances in CT technology. 7
Principles of Attenuation
CT images represent the attenuation of X-ray photons by different body constituents, with attenuation expressed in Hounsfield Units (HU). 8 Two dominant physics principles account for X-ray attenuation; the Photoelectric effect and Compton effect. SECT is limited by its sole reliance on CT HU for quantitation. 9 This explains why, depending on their relative densities, 2 materials of differing composition such as calcium and iodine may yield the same measurable HU when subjected to a single X-ray beam but produce different HU values when exposed to different energy levels at DECT; independent of their density value. Because Iodine and calcium have high atomic number (Z), they are more susceptible to the photoelectric effect. Contrarily, soft tissue demonstrates weak photoelectric effect and less variation of attenuation values with different energy levels. 10
DECT Principles
DECT assesses attenuation by materials when submitted to high (140-150 kVp) and low energy (80-100 kVp) photon beams and acquires 2 datasets for the imaged anatomical range. The term DECT is a misnomer as each beam is polyenergetic, composed of photons of varying energies centred around a kV peak, hence it is also known as spectral CT. Postprocessing manipulation is utilized to reconstruct 3 different types of images: mixed images, material specific images, and virtual monoenergetic images (VMI). 11
Images derived from a combination of the high and low energy datasets are created and are called mixed, blended, or weighted images. Using varying percentages of the high and low kVp datasets, the resultant images simulate SECT images acquired at 120 kVp12,13 and are sent to the Picture Archiving and Communication System (PACS) for interpretation.
The next step entails creating DECT material-specific images after evaluating interaction of all body constituents with the high and low energy levels. These images are generated through differential attenuation analysis to obtain a dual-energy index. Postprocessing software is used to calculate the attenuation properties of each voxel at low and high energy and a mathematical algorithm is used to determine the proportion of dominant materials within the voxel based on a 3-material decomposition technique, for example, to evaluate the representation of iodine, calcium, or fat in each voxel. 7 Once the proportions are known, different sets of images are generated after subtracting each constituent. The virtual non-contrast (VNC) images are constructed after the voxels containing iodine are excluded from the image. Similarly, iodine-only images and virtual non-calcium images can be generated. It is also possible to superimpose a color-coded iodine overlay map in addition to quantifying the amount of iodine in a specific region of interest to assess for enhancement. By way of identification of tissue composition, DECT has placed itself in a superior position over SECT for several applications.
DECT allows the extrapolation of images for various single-energy levels. The resultant VMI simulates the image that would be obtained if the scanner were to emit a true monoenergetic beam of X-rays. Most postprocessing software is capable of extrapolating energy levels ranging from 40 to 200 keV, beyond the effective energy level of both X-ray tubes, by using variable fractions of the data acquired by each tube. 14 Using low energy images improves the contrast-to-noise ratio by increasing the attenuation of materials with high atomic numbers such as iodine and calcium at the expense of decreased spatial resolution and increased noise. On the contrary, high-energy images improve signal-to-noise ratio and notably ameliorate the effect of streak artifact but with decreased attenuation of body constituents at higher energies, thereby limiting soft tissue contrast resolution.
Workflow
Certain considerations should be given to the impact postprocessing of DECT images has on the department’s workflow. Image reconstruction is performed using a third-party postprocessing software. Reconstruction is either created manually by technologists/radiologists or automatically created by the software and sent to PACS for reporting.
Manual reconstruction is operator-dependent but has the advantage of specific case optimization as the reader feels appropriate. However, radiologists should be aware that this process can be time-consuming and could induce negative repercussions for the department’s throughput.
Automated reconstructions are sent directly to PACS which saves time and increases utilization for timely image interpretation. It also offers predefined standardized image reconstruction for all performed scans. Drawbacks include “image overload” and the need for larger storage capacity. Additionally, the default vendor optimization including window/level of greyscale and overlay images may not appeal to all radiologists.
DECT Systems
Various technical approaches have been employed to acquire DECT datasets. These include dual-source, fast kilovoltage-switching, and multilayered-detector scanners.
Dual-Source
Dual-source scanner with dual detector arrays utilizes 2 X-ray sources and 2 data-acquisition systems mounted on the gantry in an orthogonal fashion. 8 Each X-ray source is equipped with its own high voltage generator allowing independent control of the X-ray tube potential and current. High energy dataset is obtained at 120 or 140 kVp and low energy dataset acquired at 80 or 100 kVp. 15 Using 2 separate X-ray sources allows beam filtration and current modulation in each tube resulting in optimization of image quality. However, the dual-source images suffer from limited temporal and spatial registration because the high and low energy scans are acquired at slightly different times. Furthermore, the available field of view is limited to 33 cm somewhat hindering evaluation in patients with large body habitus.
Single-Source With Fast Kilovoltage Switching
This prototype employs a single source scanner with a single detector layer. It is contingent on a single X-ray source with fast switching between 2 kilovoltage settings (80 and 140 kVp) at intervals of 0.5 ms during a single gantry rotation to generate high and low energy X-ray acquisitions. 15 Unlike the dual-source scanner, the tube current remains constant for both energy levels and cannot be altered simultaneously. The exposure time ratio is varied between the 80 and 140 kVp acquisitions (typically 65% of exposure time at 80 kVp and 35% at 140 kVp) in order to optimize the contrast-to-noise ratio. The detector system has a fast-sampling capability to acquire the alternating high and low energy data. The advantages of the fast kilovoltage switching scanner include good temporal and spatial registration. Furthermore, the available field of view is increased to 50 cm for image analysis. Drawbacks include limited spectral separation between the high and low energy datasets. Additionally, the noise to signal ratio is higher on images obtained at lower energy levels as the tube current cannot be modulated at the same time the peak voltage is altered.
Single-Source With Dual-Detector Layers
This scanner detector system is modified with 2 scintillation layers arrayed one on top of each other to receive separate high and low energy data from a single X-ray source. 15 The top detector layer captures low energy data whilst the bottom layer captures high energy data. This prototype offers excellent temporal and spatial registration but has limited energy separation with substantial spectral overlap.
Radiation Dose
When first introduced, concerns were raised about DECT’s radiation dose being substantially higher than SECT. However, introduction of iterative reconstruction techniques, improved detector efficiency, and additional spectral filtration facilitated DECT acquisition at a radiation dose equivalent to or even less than SECT.16-18 Assumptions that DECT imparts twice the radiation dose are false since the total dose used to obtain both datasets is split between X-ray beams when using dual-source or fast-kV switching techniques. 11 Data from the high and low-kVp acquisitions at DECT yield blended images similar to SECT with equivalent image noise levels, in addition to generating reconstructed images that allow analysis of material composition. DECT’s ability to create virtual nonenhanced images may obviate the need for nonenhanced image acquisition, substantially lowering radiation dose for multiphase examinations. 11
Clinical Applications of DECT in Solid Organ Injury (SOI)
Material Decomposition
Material decomposition techniques enable differentiation of materials of differing chemical compositions. The breakdown of tissues into basic constituents allows separation of materials such as water, fat, iodine, and calcium within each voxel. 19 Some of these constituents may have the same attenuation value on SECT due to their reliance on the X-ray attenuation coefficient. However, the production of virtual material-specific images by DECT is instead dependent on the volume fraction and physical density of materials contained in each image voxel. 19 A designated software is used to calculate the attenuation properties of each voxel at low and high energy and determine the proportion of dominant materials within the voxel. 11 Once the proportions are known, different sets of images can be generated after subtracting or isolating each constituent. VNC images are created after the voxels containing iodine are identified and excluded.
Unenhanced images are invaluable in confirming the hyperattenuating nature of acute blood products within solid organs (intraparenchymal hematoma) or the abdominopelvic cavity. 11 Similarly virtual iodine-only and non-calcium images can aid in the differentiation of vascular injury (contrast) from calcification or exogenous/surgical material or even determining foreign body composition in the setting of penetrating trauma.
Virtual Monoenergetic Images (VMI)
Low-keV virtual monoenergetic imaging accentuates Iodine-rich tissues, improves contrast conspicuity, and consequently increases the sensitivity for detecting solid organ parenchymal lacerations. 20 On the other hand, a high keV monoenergetic application is utilized to reduce streak and blooming artifact caused by metallic orthopedic implants, endovascular stents, embolization material, and surgical packing material after damage control laparotomy.21,22 This facilitates the interrogation of adjacent organs and vessels for signs of injury in the setting of trauma.
Iodine Mapping
Employment of iodine overlay mapping can be very useful in confirming active extravasation of contrast indicative of acute vascular injury. Additionally, the application is valuable in differentiating hematoma from active bleeding on occasions where only a single-phase portal venous study is acquired. In the absence of an arterial-phase study; accumulation of Iodine within the hematoma on the Iodine overlay map confirms brisk active bleeding.
To aid familiarity and effortless acquaintance with the main postprocessing advantages of DECT, the authors of this article refer to these as the
Summary of Advantages of DECT’s Post-Processing
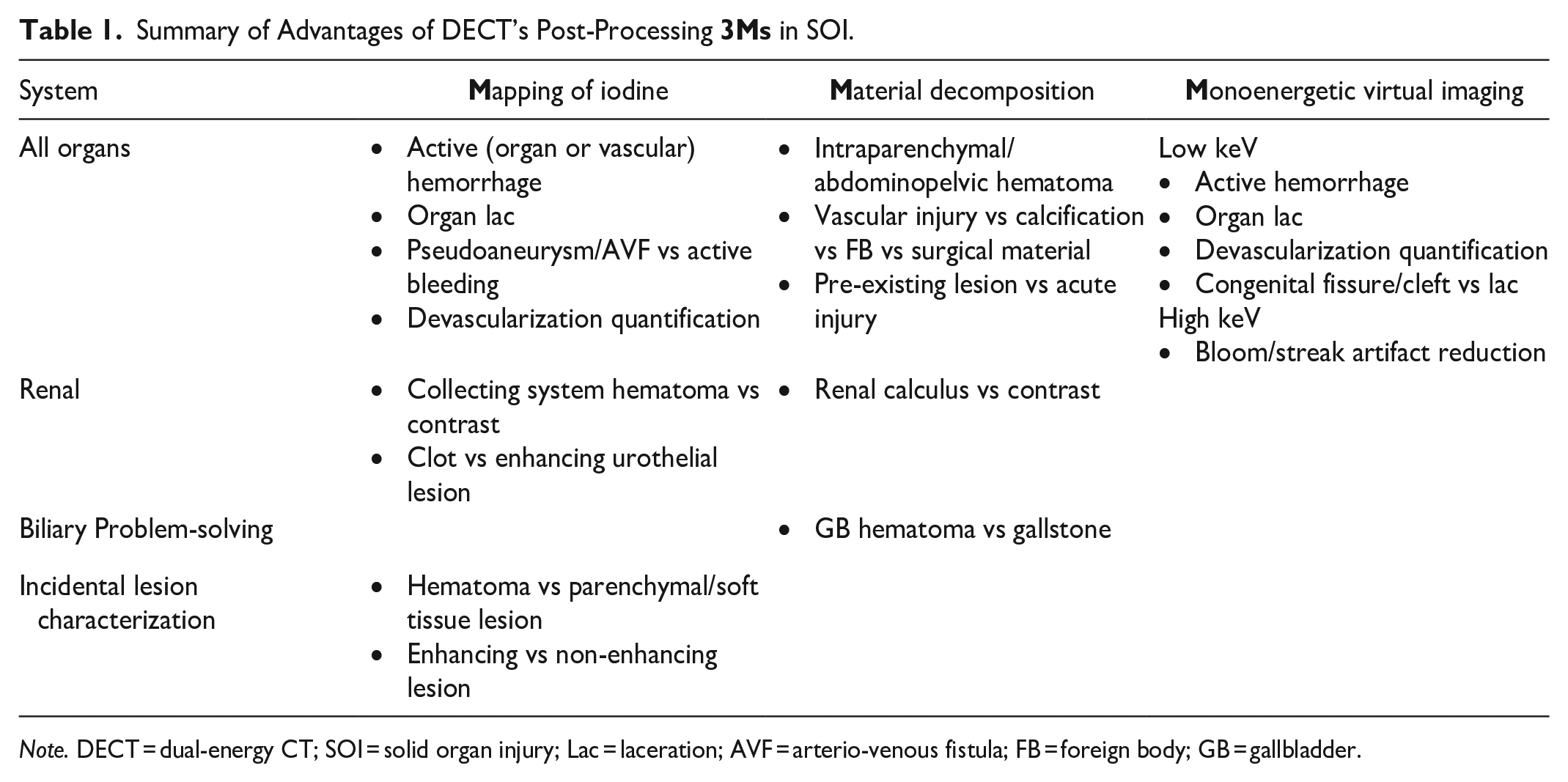
Note. DECT = dual-energy CT; SOI = solid organ injury; Lac = laceration; AVF = arterio-venous fistula; FB = foreign body; GB = gallbladder.
Solid Organ Injury (SOI)
The liver, spleen, and kidney are the commonest injured solid organs in blunt and penetrating trauma. 1 Most traumatic abdominal injuries presenting to ED are due to high velocity blunt trauma with motor vehicle collision the most frequent mechanism. 23 The most common causes of penetrating abdominal trauma are stab wounds and gunshots. 24 Grading systems with standardized nomenclature and assignment of measurable severity of injury exist, allowing prompt communication between the radiologist and clinical teams. Increasing grades of injury correlate with increasing mortality, operative rates, and hospitalization costs. 25
American Association for the Surgery of Trauma (AAST) Organ Injury Scale (OIS)
Moore et al 26 published the OIS for liver, spleen, and kidney trauma on behalf of the AAST. The OIS was devised for facilitating clinical research and reflecting on patient outcome. The grading system for liver and spleen was updated in 1994. 27 In recent years, management strategies for SOI have shifted toward non-operative management (NOM) for all but the most severe injuries, with reliance on CT for diagnosis and classification. At the other end of the spectrum, immediate damage control surgery (DCS) has been introduced for unstable severely injured patients to mitigate delays aiming to save lives with rapid control of hemorrhage, followed by intensive care support prior to definitive surgical repair. Kozar et al 25 revision of the OIS for SOI in 2018 reflects evolution of the grading system and its increased dependence on imaging.
The 2018 OIS now includes 3 sets of criteria; imaging, operative, and pathologic, whereas previous grading relied solely on operative criteria. 25 The other most significant change to the 2018 OIS is the incorporation of CT diagnosed vascular injury, including contained injuries such as pseudoaneurysm or arteriovenous fistula and non-contained active bleeding/contrast extravasation.28-30 A contained vascular injury appears on CT as a focal collection of vascular contrast that follows blood-pool density and decreases in attenuation with delayed imaging. Non-contained active bleeding however, is depicted as vascular contrast, focal or diffuse, that increases in size and/or attenuation with delayed imaging. Active bleeding may remain within the organ capsule or extend beyond to the peritoneal cavity. To maintain consistency, the same terminology for vascular injuries is used for all SOIs. Notably, the presence of multiple grade I/II injuries to the same organ or involving bilateral organs in the case of kidneys advances the overall grade level by one up to grade III.
The expansion of the AAST classification to include CT-identified injury patterns removes ambiguity in assigning grades and maintains inter-rater reliability. 31 The revision committee acknowledged that the grade of injury, in the presence of vascular trauma, maybe higher than the previously described parenchymal-based injury. Also, the committee recognized that vascular injuries are associated with higher NOM failure rate. Table 2 summarizes definitions for pertinent terminology described in the updated AAST OIS and Table 3 illustrates significant recent changes to the OIS.
Definition of Key Terms Described in the 2018 American Association for the Surgery of Trauma Organ Injury Scale.

Note. AVF = arteriovenous fistula.
Changes to the 2018 American Association for the Surgery of Trauma Organ Injury Scale.
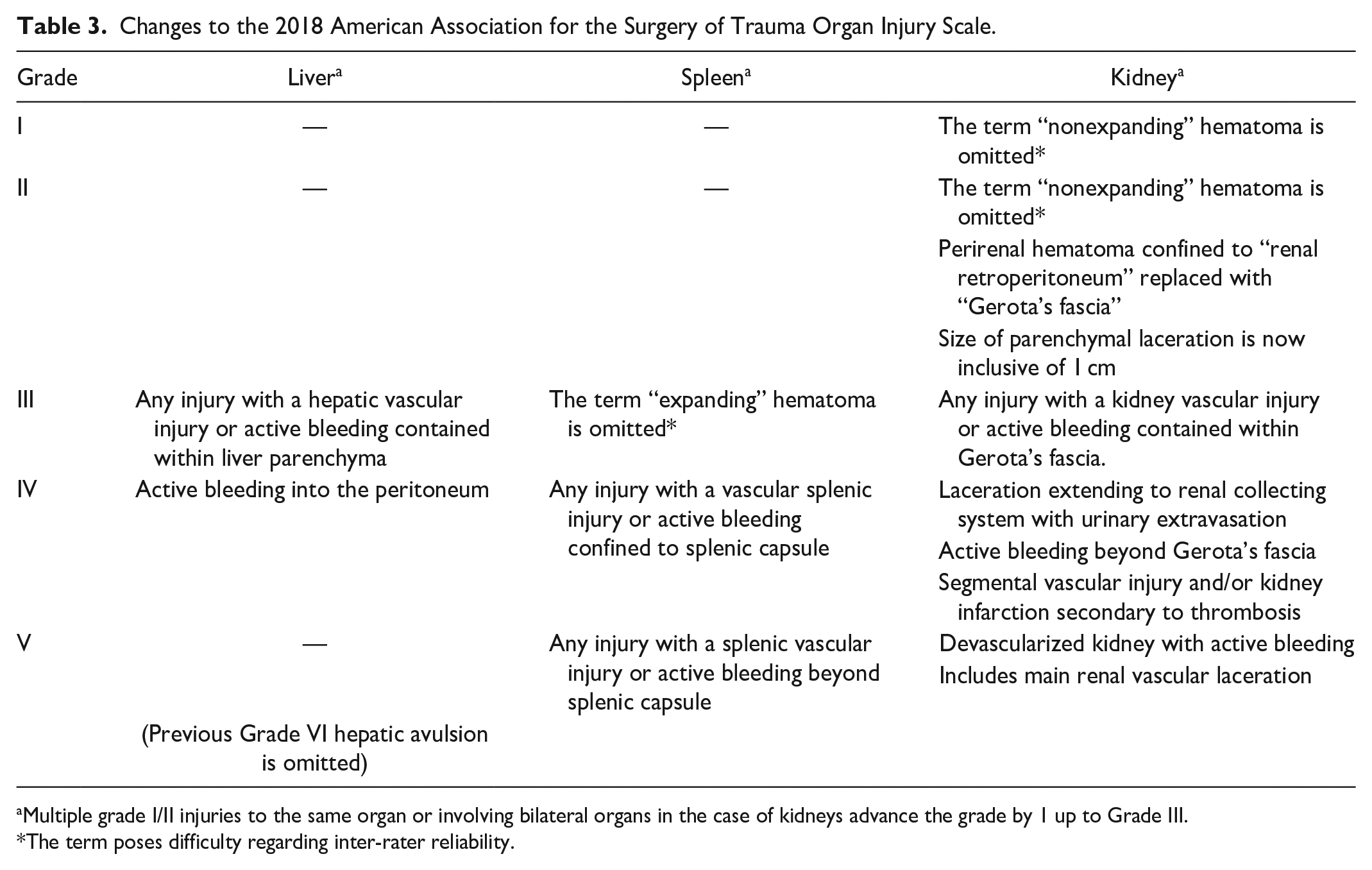
Multiple grade I/II injuries to the same organ or involving bilateral organs in the case of kidneys advance the grade by 1 up to Grade III.
The term poses difficulty regarding inter-rater reliability.
CT Technique
The use of CT in the setting of blunt trauma has been encouraged since early 1980s. 32 The American College of Radiology’s appropriateness criteria recommends Multidetector CT (MDCT) of the abdomen and pelvis with IV contrast enhancement as the imaging modality of choice for hemodynamically stable trauma patients. 33 MDCT is fast, widely available and provides both anatomic and functional data. It diagnoses the entire spectrum of SOI with accuracy and helps formulate therapeutic decisions in cases of severe SOI or active hemorrhage. 34 In a selected group of hemodynamically unstable patients who respond to resuscitation, CT is considered appropriate to facilitate clinical decision-making whilst the patient is closely monitored. 1
The 2018 revised OIS recommends dual-phase CT imaging, including arterial and portal venous phases, for accurate diagnosis of vascular injuries of the spleen, liver, and kidney. Dual-phase imaging allows temporal vascular assessment at 2 distinct points in time, shown to increase the sensitivity of depicting vascular injuries and providing an overall better diagnostic performance in evaluating SOI than either phase alone. Optional 5 to 10-minute delay imaging is considered in cases of suspected active bleeding. In addition, when kidney injury is known or suspected, delayed excretory phase imaging should be obtained. 25
It is worth noting that there continues to be heterogeneity amongst centres regarding the best CT protocol in the setting of trauma. At the authors’ institution, dual-energy MDCT scanners are used for all trauma imaging. The radiologist provides preliminary verbal interpretation to the trauma team at the CT scanner console for life-threatening injuries. SECT abdominopelvic arterial phase imaging (35 seconds post-injection) is acquired as part of a high-pitch CT angiogram of the chest, abdomen and pelvis followed by abdominopelvic DECT acquisition (70 seconds post-injection) with an optional 5 or 10-minute delayed phase abdominopelvic SECT in suspected active bleeding or renal collecting system injury respectively. There is no need to acquire DECT imaging in all phases. Utilizing portal venous acquisition is preferred due to its preferential parenchymal enhancement making best use of DECT benefits. Multiplanar reformats including axial, sagittal, and coronal as well as bone reconstructions are created. Images including dual-energy applications are automatically sent to PACS workstations ready for dedicated reads.
For follow-up imaging, particularly in those patients managed non-operatively, most institutions prefer to perform a dual-phase study to better delineate vascular and parenchymal complications. 35
AAST Scale
A summary of imaging criteria for the revised 2018 AAST OIS is illustrated in Table 4. 25
Summary of Imaging Criteria for 2018 AAST OIS.
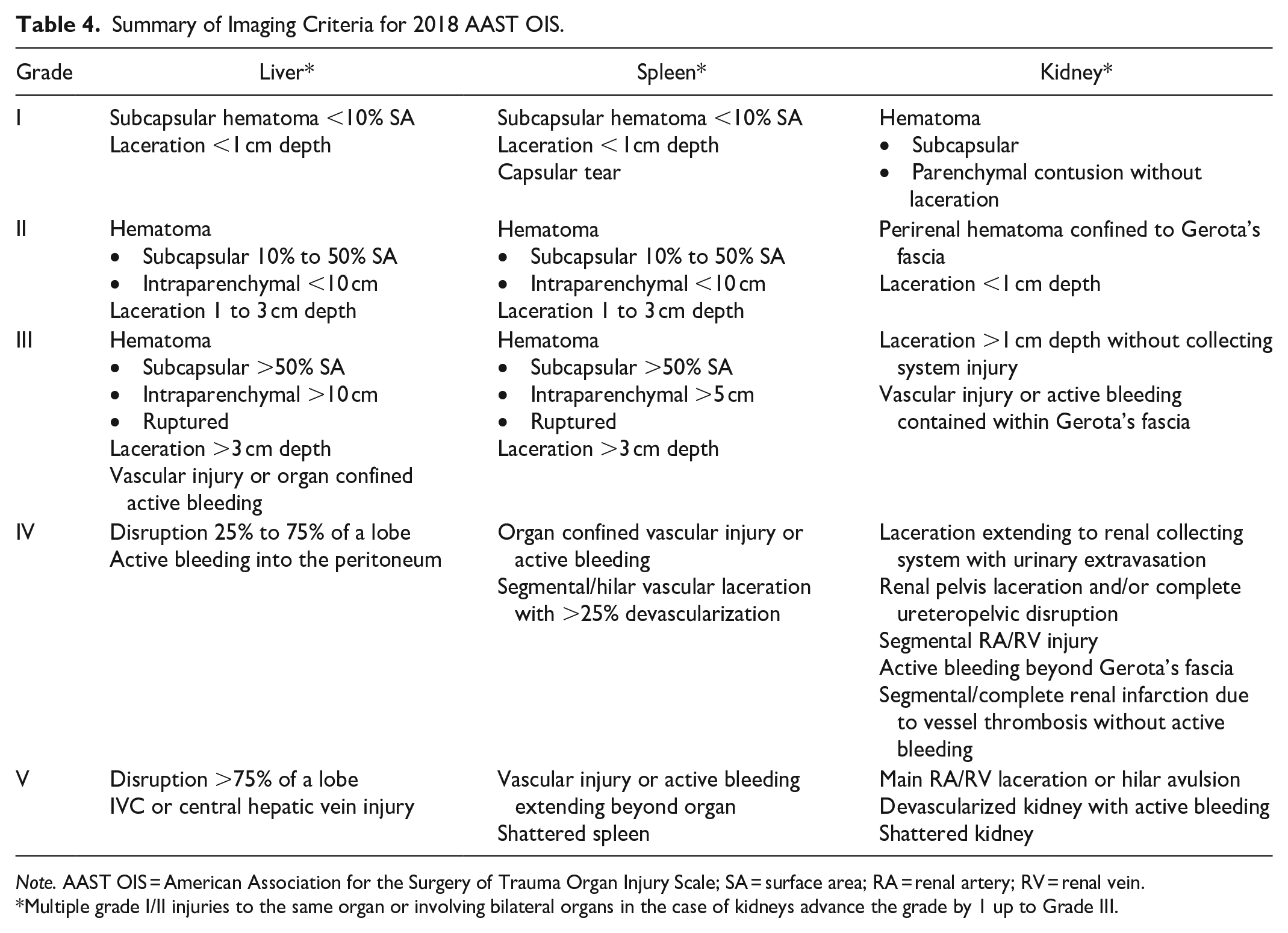
Note. AAST OIS = American Association for the Surgery of Trauma Organ Injury Scale; SA = surface area; RA = renal artery; RV = renal vein.
Multiple grade I/II injuries to the same organ or involving bilateral organs in the case of kidneys advance the grade by 1 up to Grade III.
Over the next few paragraphs, we endeavor to illustrate to our readers the embodiment of various DECT applications in identifying different grades of SOI.
Liver Injury
Surgical intervention was the primary treatment option for liver injuries until late 1980s. 36 A significant increase in selective NOM for blunt hepatic injury ensued in 2 major USA trauma centres from mid-1990s onward.37,38 A key component to this shift was attributed to the ability of CT to accurately diagnose and assess the severity of liver injury. Nowadays, over 80% of adult blunt liver injury is managed non-operatively.
DECT Applications in Liver Injury
VNC imaging can be utilized to depict the presence of and location of blood products for accurate grading of injury and guidance of subsequent patient management. Low-keV VMI exploits Iodine k-edge by increasing its attenuation coefficient for better delineation of vascular and parenchymal injuries. Additionally, Iodine overlay mapping improves the conspicuity of low-grade parenchymal lacerations and vascular injuries. Figures 1 and 2 illustrate the role of DECT in depicting low- and high-grade liver injuries respectively.
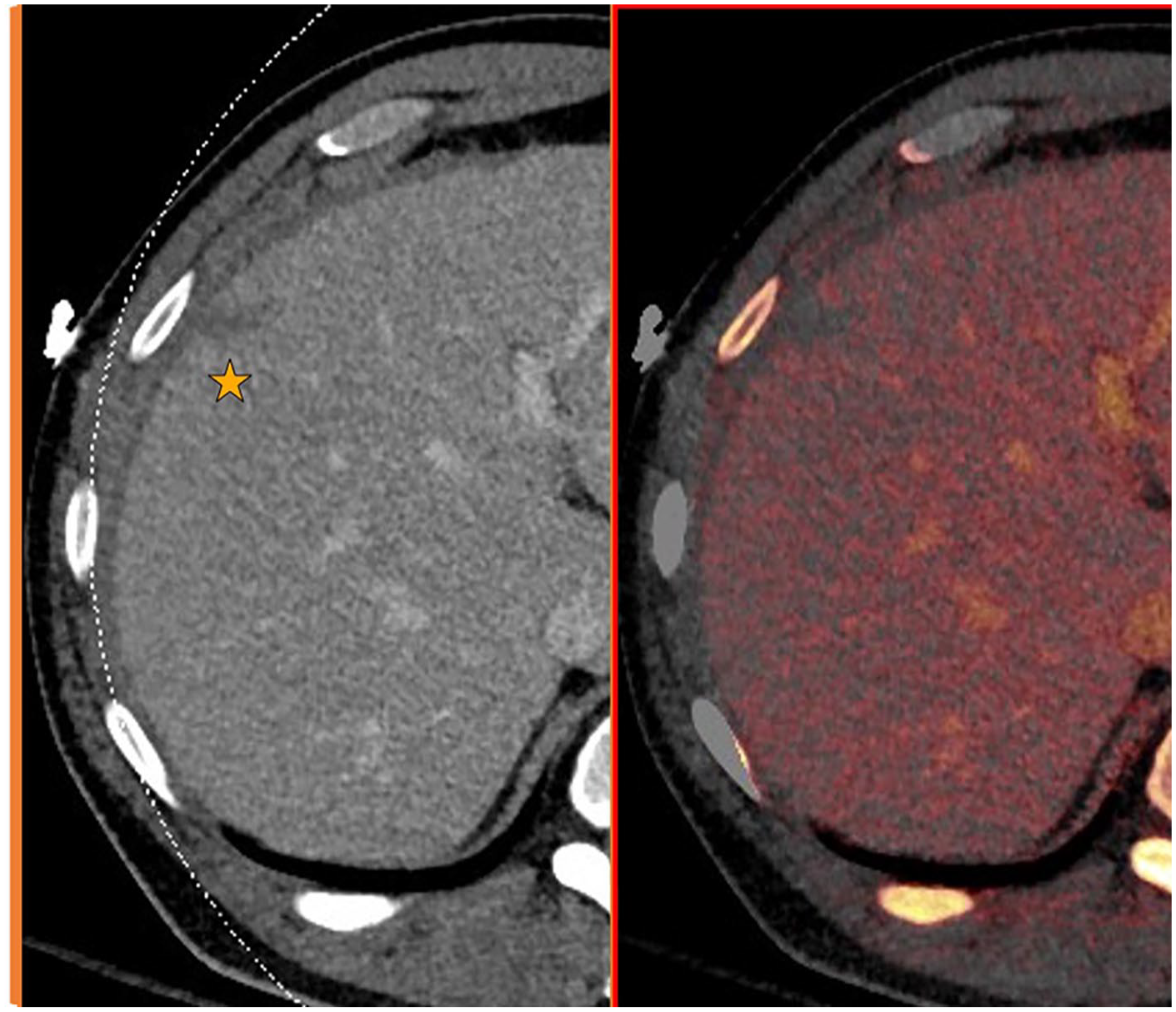
Contrast enhanced (portal venous) and Iodine overlay axial images of the right lobe of the liver.
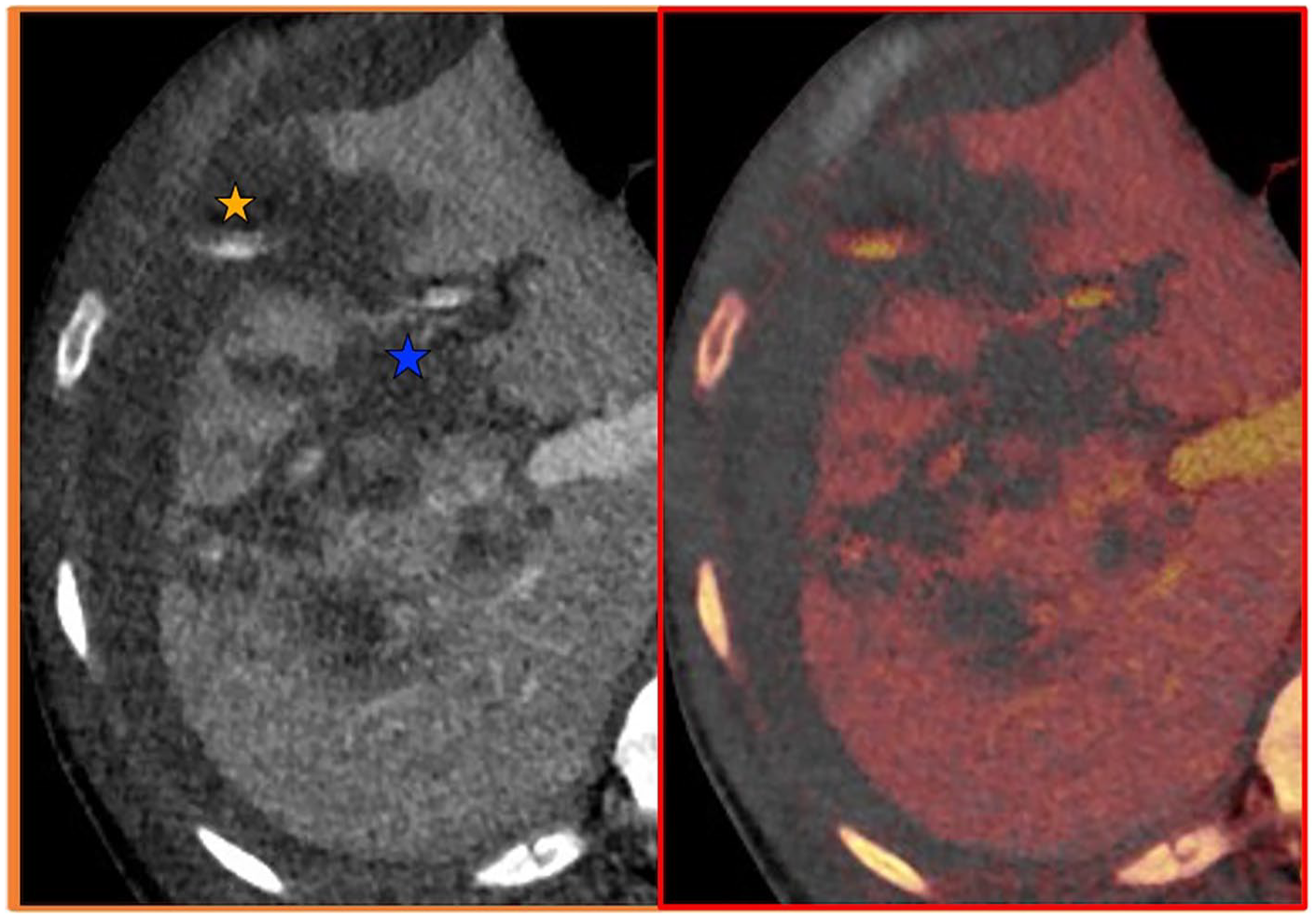
Contrast enhanced (portal venous) and Iodine overlay axial images of the liver.
Splenic Injury
The spleen is among the commonest injured organs in blunt abdominal trauma, accounting for up to 49% of all visceral injuries.39,40 Splenectomy was the traditional treatment of splenic injury for around 100 years. 36 King and Schumacher’s description of post-splenectomy lethal sepsis in 1952 shed light on the immunological role of the spleen in preventing septicemia 41 paving the way for NOM of splenic injuries. Nowadays >80% of adults who sustain blunt splenic trauma are managed non-operatively, 36 although the presence of vascular injuries means operative management is more likely.
DECT Applications in Splenic Injury
Similar to liver injuries, various DECT applications are utilized for easier identification and grading of splenic injuries associated with trauma. Figures 3 and 4 exhibit low- and high-grade splenic injuries respectively. Differentiating the type of vascular injury whether due to pseudoaneurysm/arteriovenous fistula formation or active bleeding is an additional asset of employing DECT. Furthermore, the Iodine overlay map can problem-solve linear hypodensities as either congenital splenic clefts or true lacerations particularly with concomitant injury to other organs.
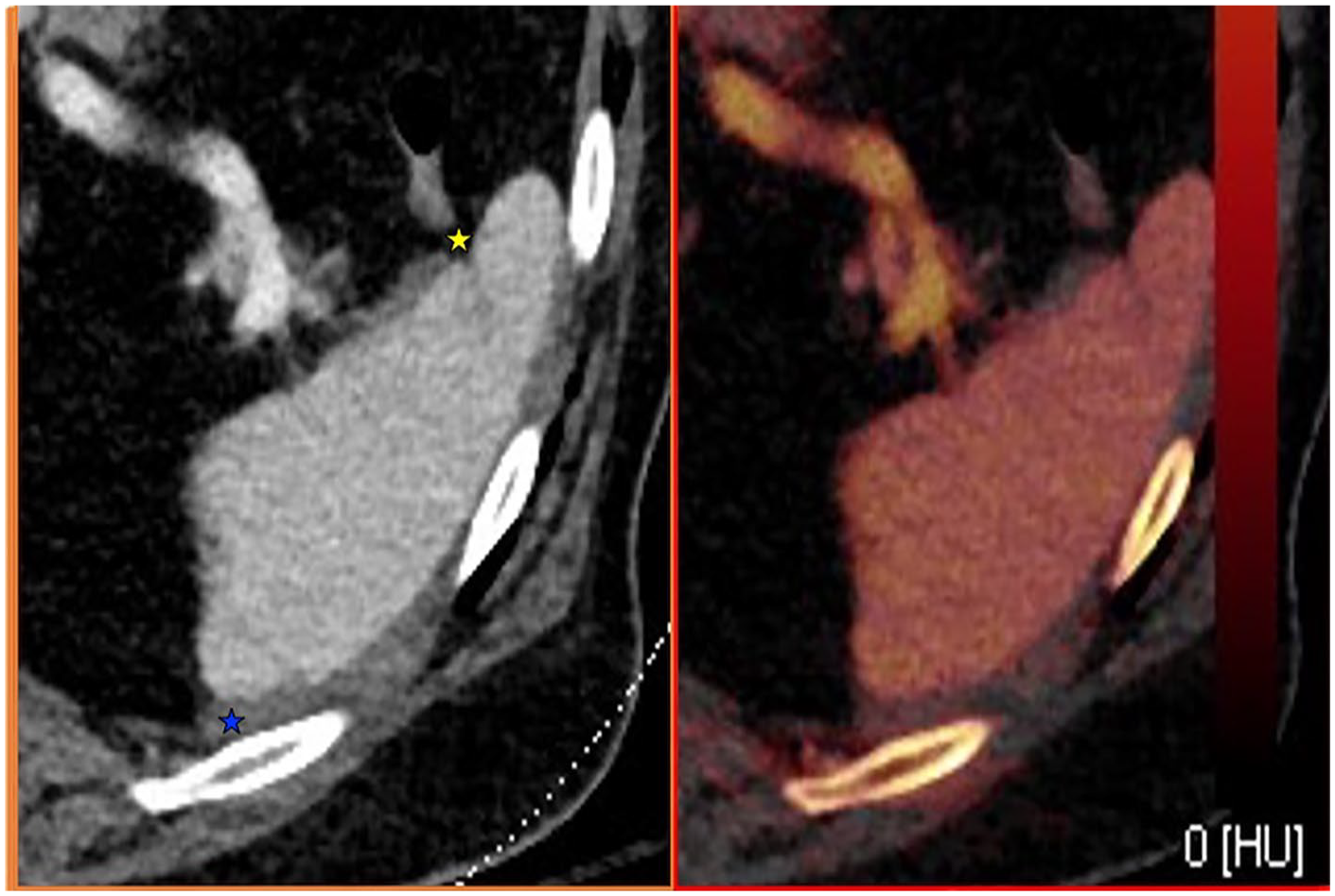
Iodine overlay and contrast enhanced (portal venous) axial images of the spleen.
From left to right—Arterial, portal venous, and Iodine overlay axial images of the spleen.
Kidney Injury
Eighty-nine percent of all renal injuries are attributed to blunt trauma. 42 Approximately 10% of patients with blunt abdominal trauma sustain renal injury, and around 6% of patients presenting with a penetrating mechanism have a renal component to their injury. 43
NOM was the standard of care for renal injuries well into the 20th century. 36 This was challenged by a 1963 study which recommended more operative management. 44 However, this was disputed by a later dated study (1989) which projected successful conservative treatment of the vast majority of its recruited patients. 45
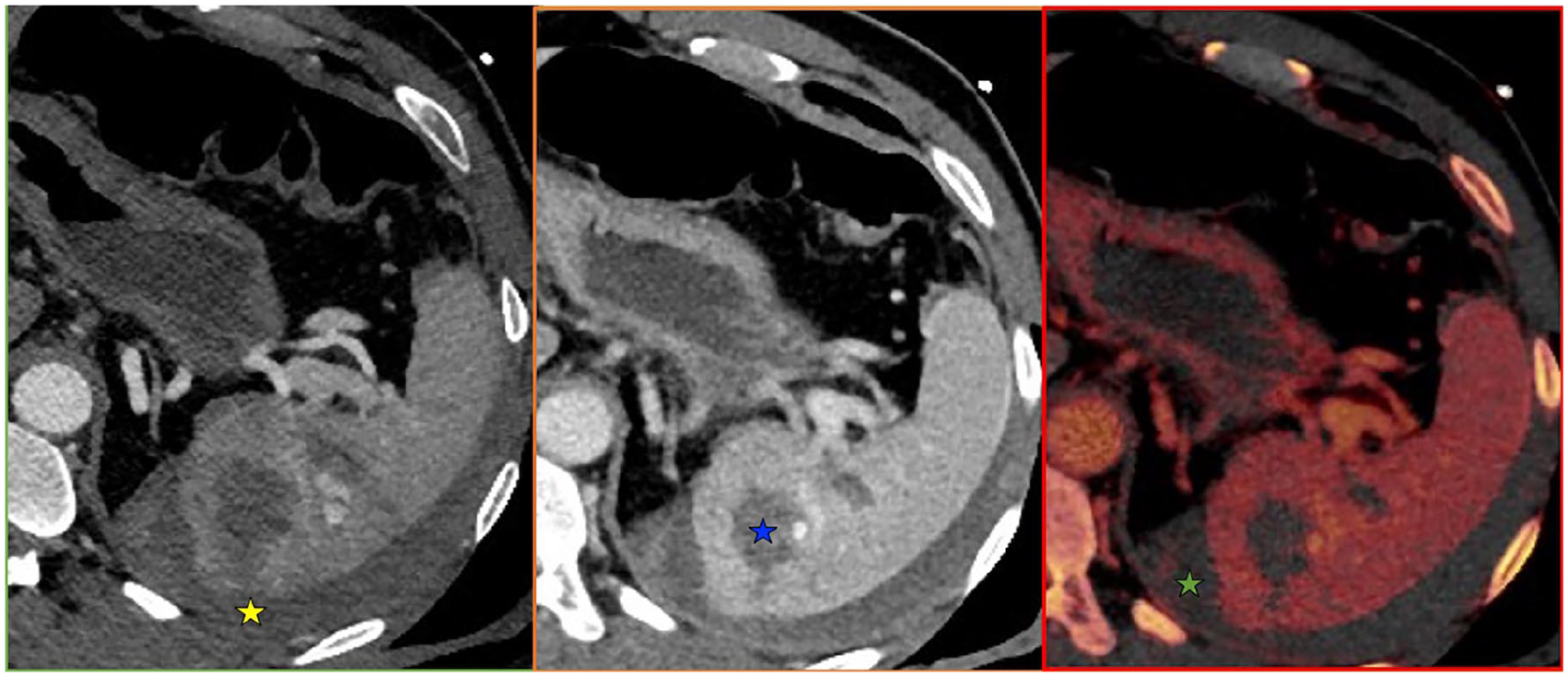
DECT Applications in Renal Injury
Over and beyond the various aforementioned applications which can be similarly utilized in renal injuries; the adoption of material decomposition can differentiate renal calculi from excreted contrast (Figure 5) or vascular injuries. Iodine overlay imaging improves the conspicuity of collecting system injuries, demonstrates with clarity devascularized renal parenchyma and, in conjunction with VNC imaging, can differentiate parenchymal hematoma from an enhancing lesion. Examples of low- and high-grade renal injuries are provided in Figures 6 and 7.
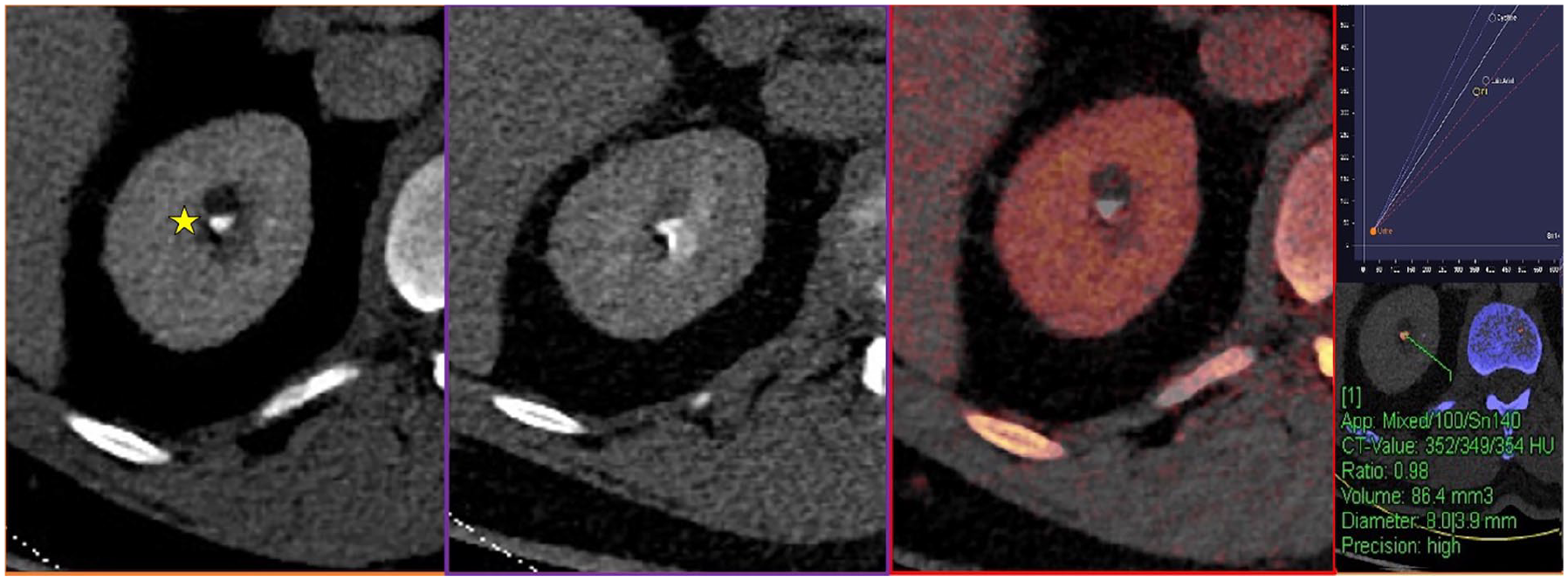
Images from left to right—Contrast enhanced (portal venous phase), delayed (excretory phase), Iodine overlay map, and DECT application for calculus composition.
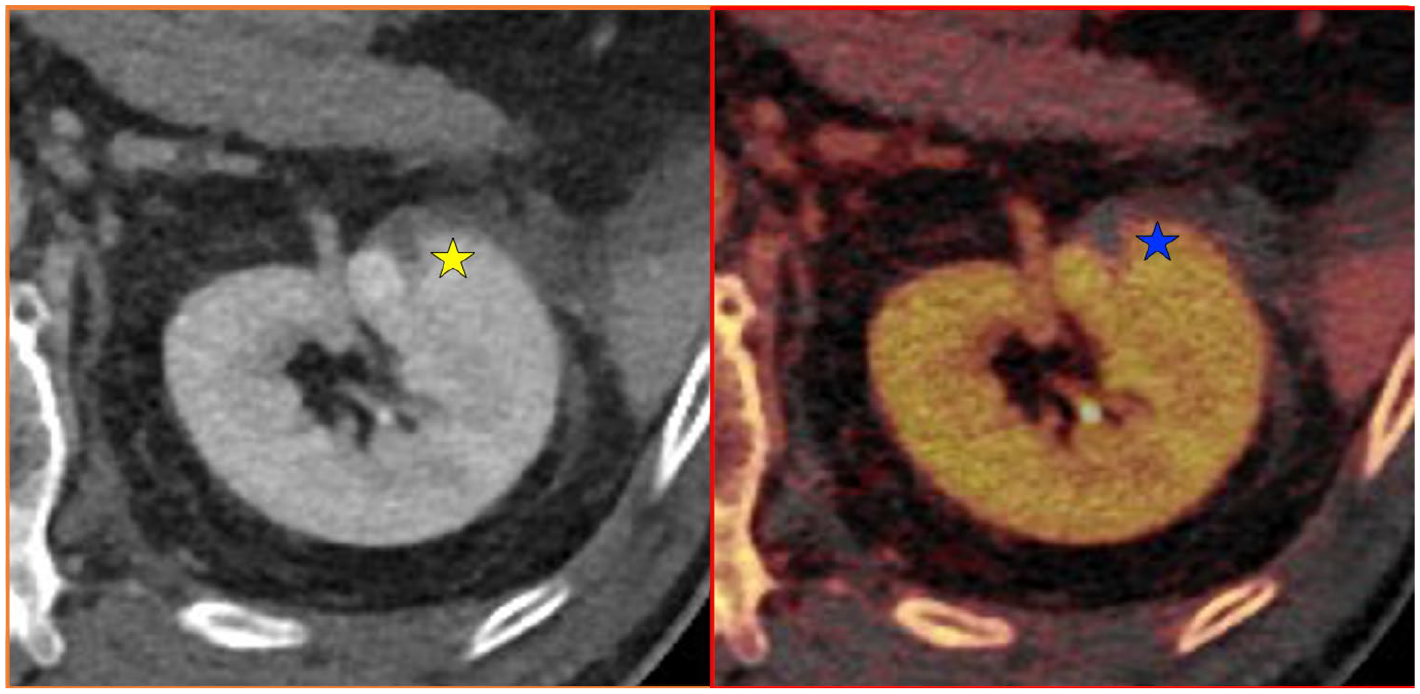
Contrast enhanced (portal venous) and Iodine overlay map axial imaging of the left kidney.
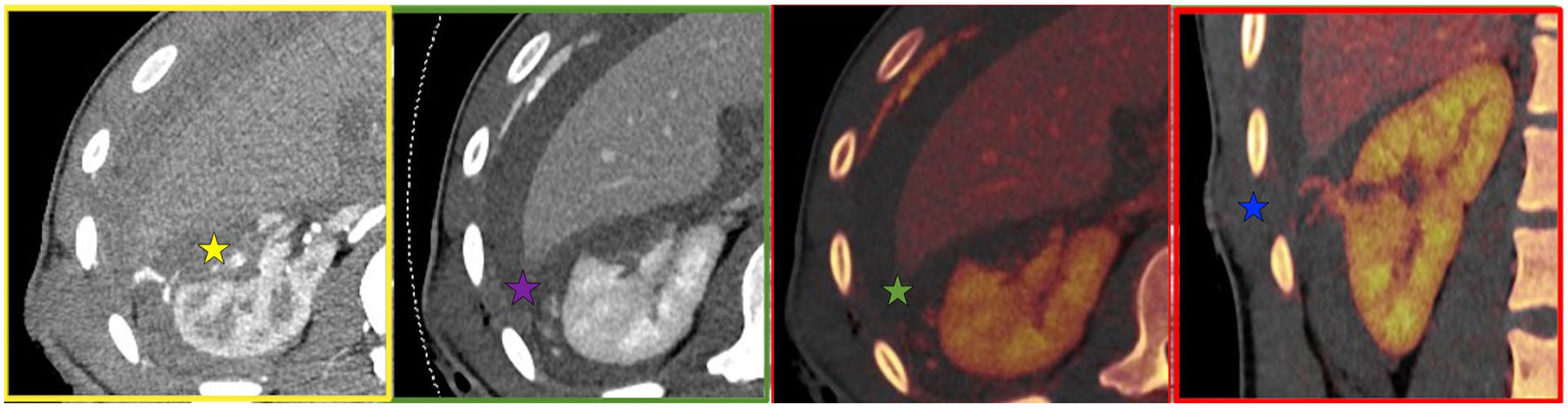
From left to right—Right kidney axial image acquisition in the arterial and portal venous phases. Iodine overlay map of the same kidney in the axial and coronal reconstruction planes.
Conclusion
A preference for selective NOM signifies an important evolution in the care of trauma patients in recent years. The updated AAST OIS confirms increasing dependence on CT for accurate diagnosis and classification of SOI, with accurate grading key to predicting patient outcomes. As a result, radiologists have a duty to adapt accordingly to image interpretation and become familiar with the updated grading systems. DECT plays a role in improving detection of injuries and diagnostic confidence, helping radiologists to accurately characterize SOI and assign an appropriate OIS.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
