Abstract
Acute mesenteric ischemia (AMI) is an uncommon yet highly lethal cause of acute abdomen in the emergency setting. Computed tomography (CT) imaging, in particular a biphasic protocol consisting of angiographic and venous phase scans, is widely used to corroborate non-specific clinical findings when suspicions of AMI are high. Techniques such as low kilovoltage peak scanning, dual energy acquisition, or a combined arterial/enteric phase can improve iodine conspicuity and evaluation of bowel enhancement. Biphasic CT with CT angiography is mandatory to directly assess for the 3 primary etiologies of AMI—arterial, venous, and non-occlusive mesenteric ischemia (NOMI), and the CT angiographic findings may be the first visible in the disease. In addition, numerous non-vascular CT findings have also been reported. Bowel wall thickening, mesenteric stranding, and ascites are common but non-specific findings that correlate poorly with disease severity. Pneumatosis intestinalis and portomesenteric venous gas, while not pathognomonic for ischemia, are highly specific in cases of high clinical suspicion. Bowel wall hypoenhancement is an early and specific sign but requires a protocol optimizing iodine conspicuity to confidently identify. Finally, intraperitoneal free air and solid organ infarcts are also highly specific ancillary findings in AMI. AMI occurs as a complication in 10% of small bowel obstruction (SBO) patients, and understanding imaging findings of ischemia in the context of SBO is necessary to aid in treatment planning and reduce over- and under-diagnosis of strangulation. Familiarity with the imaging features of ischemia by radiologists is vital to establish an early diagnosis before irreversible necrosis occurs.
Introduction
Acute mesenteric ischemia (AMI) accounts for <1% of all causes of acute abdomen in the emergency setting.1-4 Nonetheless, it is associated with high mortality (50–90%) owing to its non-specific clinical presentation resulting in delayed diagnosis.1-3,5 AMI is distinguished from chronic mesenteric ischemia (CMI), which is caused by gradual stenosis of the mesenteric vasculature—usually by atherosclerosis—and is not acutely fatal.6,7 Radiologists play an essential role in prompt diagnosis of AMI as imaging is highly sensitive and specific and typically needed to corroborate non-specific clinical findings.
Background and Pathophysiology
The gastrointestinal (GI) tract is predominantly perfused by the celiac artery, superior mesenteric artery (SMA), and inferior mesenteric artery (IMA).8-10 The celiac artery and its branches are the primary blood supply for the stomach, proximal duodenum, and spleen.8-10 The SMA supplies most of the small bowel, as well as the colon to the level of the splenic flexure.8-10 The IMA, the smallest of the 3 vessels, supplies the descending colon, sigmoid colon, and the proximal third of the rectum.8-10
Extensive collateralization between the 3 arteries ensures continuous blood supply even if one vessel is compromised. In the chronic setting, typically at least two of the three vessels must be occluded or critically stenosed before ischemia develops. 10 The SMA and celiac artery primarily anastomose through an arcade formed by the gastroduodenal and pancreaticoduodenal arteries. 10 The middle colic artery of the SMA and the left colic artery of the IMA anastomose at the splenic flexure, a watershed territory known as the “Griffith point.” (10) The ileocolic, right, middle, and left colic arteries also anastomose in a continuous arcade called the marginal artery of Drummond. 10 The more centrally located arc of Riolan (mesenteric meandering artery) connects the middle and left colic arteries. 10 The superior rectal artery of the IMA anastomoses with the middle and inferior rectal arteries (arising from the internal iliacs), the so-called “Sudeck point.“ (10) New collateral vessels can develop and existing collateral vessels can enlarge if there is progressive vascular compromise, such as with atherosclerosis. 10
Imaging Stages of Arterial Occlusive or Nonocclusive Mesenteric Ischemia. a
aIn contrast, venous occlusive ischemia commonly results in early bowel wall thickening, mesenteric edema, and ascites due to congestion.
Imaging Protocol
Given its widespread availability, fast scan time, and diagnostic efficacy, multidetector CT (MDCT) has become the primary imaging modality used to confirm the diagnosis when clinical suspicion is present. 3
Sample Mesenteric Ischemia CT Protocol.

Flush with 40 ml normal saline at same rate.
May administer water as neutral oral contrast if patient can tolerate it.
Calculate abdominal aortic arrival time (Tarr) with a test bolus.
Arterial/enteric phase: Slice thickness 0.6–1.25 mm; scan at Tarr + 20–25 seconds (consider scanning this phase with low kVp [≤100] or dual energy acquisition).
Venous phase: Slice thickness 2.5–5.0 mm; scan at Tarr + 65– 75 seconds.
Abbreviations: I, iodine; kVp, kilovoltage peak; mg, milligrams; ml, milliliters; Tarr, arrival time; s, seconds.
A triphasic protocol with the addition of a non-contrast phase was initially thought to increase sensitivity and interobserver agreement when assessing for bowel wall hypoenhancement, and may also aid in identification of submucosal hemorrhage but a subsequent study found no benefit beyond the traditional biphasic exam for diagnosing ischemia and it has not been as widely adopted.19-21
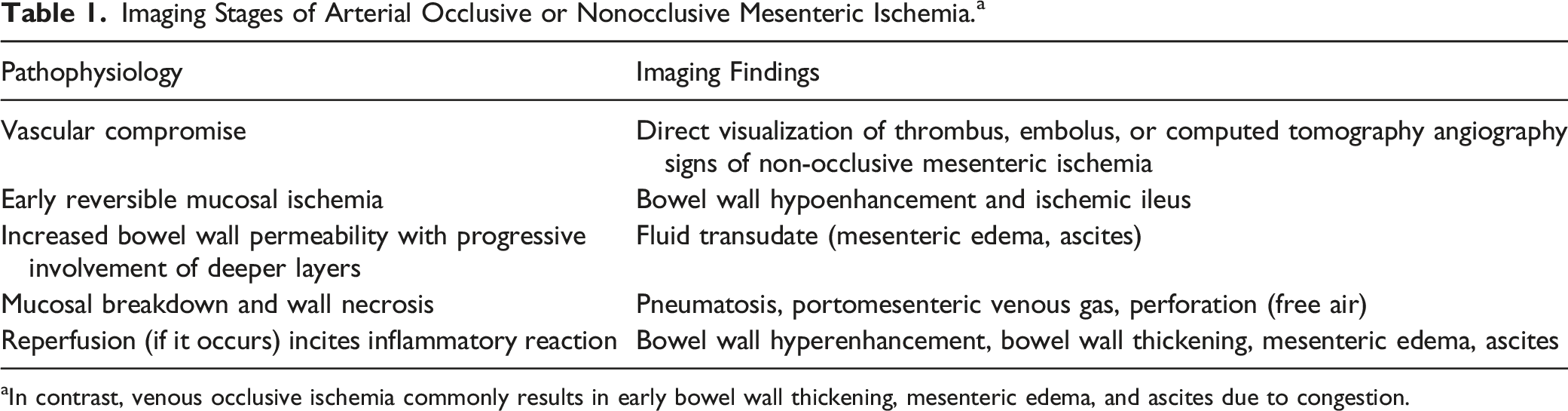
Dual-energy CT (DECT) is an emerging tool for the evaluation of AMI. Low keV virtual monoenergetic image (VMI) reconstruction (40–60 keV) can approach the k-edge of iodine (33.2 keV) and will thus increase the conspicuity of iodinated contrast (Figure 1).22-24 DECT can also improve the evaluation of the arterial vasculature by more vividly highlighting intraluminal contrast, which can be helpful in cases where angiographic images are degraded by suboptimal contrast bolus injection or timing.22-24 Mesenteric emboli. A) Coronal maximum intensity projection (MIP) image shows extensively calcified branches of the superior mesenteric artery filled with emboli (curved arrow). While some loops in the right abdomen enhance normally (arrowhead), others on the left are hypoenhancing and ischemic (arrow). B) Transverse dual energy combined arterial/enteric phase CT image demonstrates the profound contrast difference between the normal (arrowhead) and hypoenhancing ischemic (arrow) loops. Note that the ischemic bowel is thin-walled and that the decreased enhancement could prove challenging to identify without first using normal bowel as an internal control to compare to.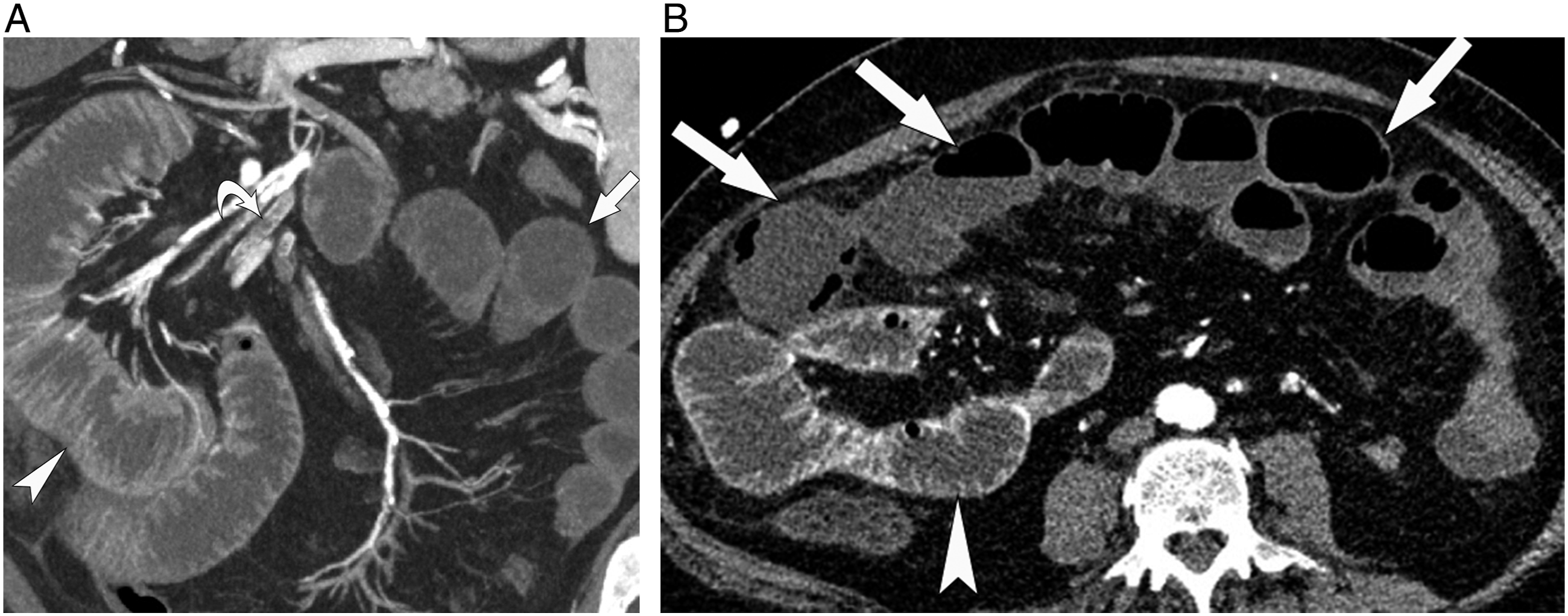
Administration of positive oral contrast should be avoided as it may mask bowel wall enhancement. 25 The use of a neutral or negative oral contrast such as low-density barium sulfate or water can be considered to distend the small bowel to better evaluate mural pathology, but this may be impractical in the acute setting as it can significantly delay scan time and can be poorly tolerated by patients who are often critically ill.15,25,26
Causes of Primary Acute Mesenteric Ischemia
The etiology of primary AMI is traditionally divided into occlusive or non-occlusive mesenteric ischemia (NOMI). Occlusive etiologies are further divided into arterial or venous.
Arterial
Arterial AMI is the most common, representing 50–75% of all cases of AMI.1,2 The extent of ischemia can be variable and depends primarily on the location of occlusion/stenosis, clot burden, and the robustness of collateral circulation. 14
Embolic AMI accounts for 40–50% of all cases of AMI. 2 The SMA is particularly susceptible to embolic phenomena owing to its relatively large caliber and narrow takeoff from the abdominal aorta.1,10 Emboli frequently lodge 3–10 cm from the SMA ostium near or distal to the origin of the middle colic artery.1,4,10,14 Emboli are most often cardiac in origin; less commonly they can arise from the aorta or from within the SMA itself such as from a partially thrombosed SMA aneurysm.5,10,14 Around 20–50% of patients have multiple SMA emboli or synchronous emboli to the solid organs such as the spleen and kidneys, and a third have an antecedent embolic event.1,10,27,28 The clinical presentation is often fulminant and characterized by severe abdominal pain “out of proportion” to physical examination, diarrhea, and hematochezia. 1
Superior mesenteric artery thrombosis is responsible for 20–35% of AMI and up to 80% of patients have a history of CMI. 1 A common trigger for thrombotic AMI is rupture of an existing atherosclerotic plaque.5,14 The site of obstruction is usually more proximal compared to embolic AMI—often at or close to the SMA origin—with consequently greater extent of ischemia.1,5,14 Symptom onset in thrombotic AMI is more insidious compared to that of embolic AMI, given increased collateralization, and is typically superimposed on existing CMI symptoms such as postprandial abdominal pain and food aversion. 10
Dissection is another cause of arterial AMI; the SMA can arise from a poorly perfused false lumen in aortic dissection, a dissection flap from aortic dissection can propagate into the SMA, or a focal dissection can occur directly within the SMA itself. 5 Dissection and other etiologies such as fibromuscular dysplasia and vasculitis combined make up less than 5% of cases. 14
Occlusive emboli or thrombi appear as abrupt non-opacification of an arterial vessel which is termed the “vessel cut-off sign” (Figure 1).5,14 SMA thrombosis or embolism in a patient with clinical suspicion for AMI is a highly specific finding (specificity up to 100%).13,16,28-30 In cases of acute-on-chronic thrombosis, existing calcified and non-calcified atherosclerotic plaque is often present within the SMA, as well as the abdominal aorta and other major arteries, and can be extensive.5,14 An investigative pitfall in arterial AMI can occur when evaluating MIP reconstructions. While MIP reconstructions allow for rapid examination of the mesenteric arcade, it can also underestimate the burden of disease (Figure 2) as any contrast around a thrombus will be projected and could mask pathology. Correlation with axial and multiplanar angiographic images is always necessary for accurate evaluation of the vasculature. Interpretative pitfalls: Underestimating thrombus on maximum intensity projection (MIP) images. Sagittal MIP (A) and multiplanar reformation (MPR) (B) of the aorta show how much more conspicuous a large embolus (arrows) in the superior mesenteric artery is on the MPR. As an MIP will project the brightest pixel in the volume of tissue reconstructed, if there is any contrast on either side of the thrombus it will project forward on the MIP and mask the clot. MIP images are excellent for a rapid assessment of the mesenteric arcade but can underestimate clot burden.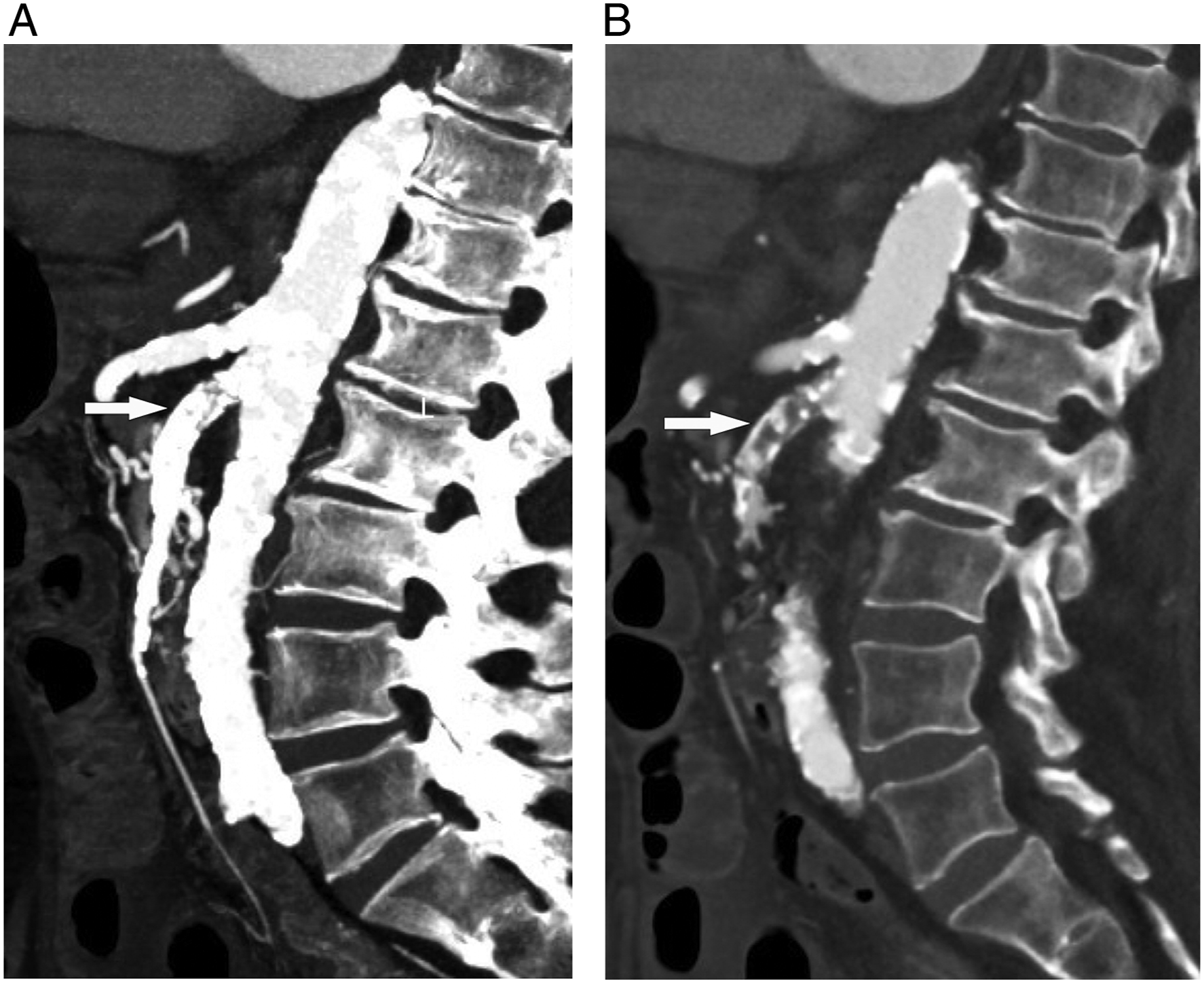
Non-Occlusive
Comprising 20–30% of cases, NOMI occurs as a result of mesenteric hypoperfusion caused by decreased cardiac output with no evidence of vascular occlusion.1,2,27,31 A 50% decrease in SMA blood flow initially results in compensatory vasodilation, but if hypoperfusion persists, sympathetic stimulation results in mesenteric vasoconstriction causing increased arterial resistance and further decrease in perfusion. 27 This change can happen as early as 10 minutes following hypoperfusion. 31 This response is often a reaction to a systemic trigger event such as recent major surgery, hypotension, sepsis, various types of shock, and drug interaction.1,32,33 Vasoconstriction can be reversed if blood flow is rapidly restored, but after 30 minutes of hypoperfusion the response can be irreversible, even with 100% restoration of flow.27,34 NOMI is associated with poor prognosis and high mortality as it typically affects the elderly, those who are critically ill, and those who have significant existing comorbidities.1,27,31 Clinical findings are non-specific and typical AMI symptoms such as abdominal pain may be absent or only present in advanced disease.1,35 Patients are frequently altered or sedated and unable to characterize their symptoms, making early recognition and diagnosis even more challenging. 1
In NOMI, typical findings of arterial vasoconstriction are seen such as focal narrowing of the SMA branch origins, “pruning” of the mesenteric arcade, and vasospasm resulting in a “beaded” or “string of sausage” appearance to the mesenteric vasculature (Figure 3).27,35-37 SMA caliber can also be significantly reduced in NOMI patients compared to prior CT or healthy controls and an SMA diameter of under 4 mm supports the diagnosis.
37
As NOMI often occurs in low-flow states, both the portomesenteric and systemic venous vasculature may appear diminutive or collapsed, but resuscitation prior to CT means this is not always the case.
5
The distribution of disease is often widespread, but areas of necrosis are often non-continuous and first present in the watershed territories.
37
Non-occlusive mesenteric ischemia (NOMI). Volume rendered reconstructions of a CT angiogram (CTA) before (A) and after (B) the patient developed urosepsis and was administered vasopressors. The patient developed non-occlusive ischemia and demonstrates profound vasospasm and pruning of the mesenteric vascular arcade. The mid-to-distal superior mesenteric artery (arrow) is barely visible as a result, and the splenic artery (arrowhead) is severely spasmed. C) A transverse image in the same patient shows typical findings of hyperenhancing adrenal glands (arrows), periportal edema (arrow), and hypoperfusion of the liver and spleen (*). D) Volume rendered CTA in a different patient outlines other major angiographic findings which can be seen in NOMI, including narrowing of mesenteric branch origins (arrowheads) and beading of the spasmed superior mesenteric artery (arrow).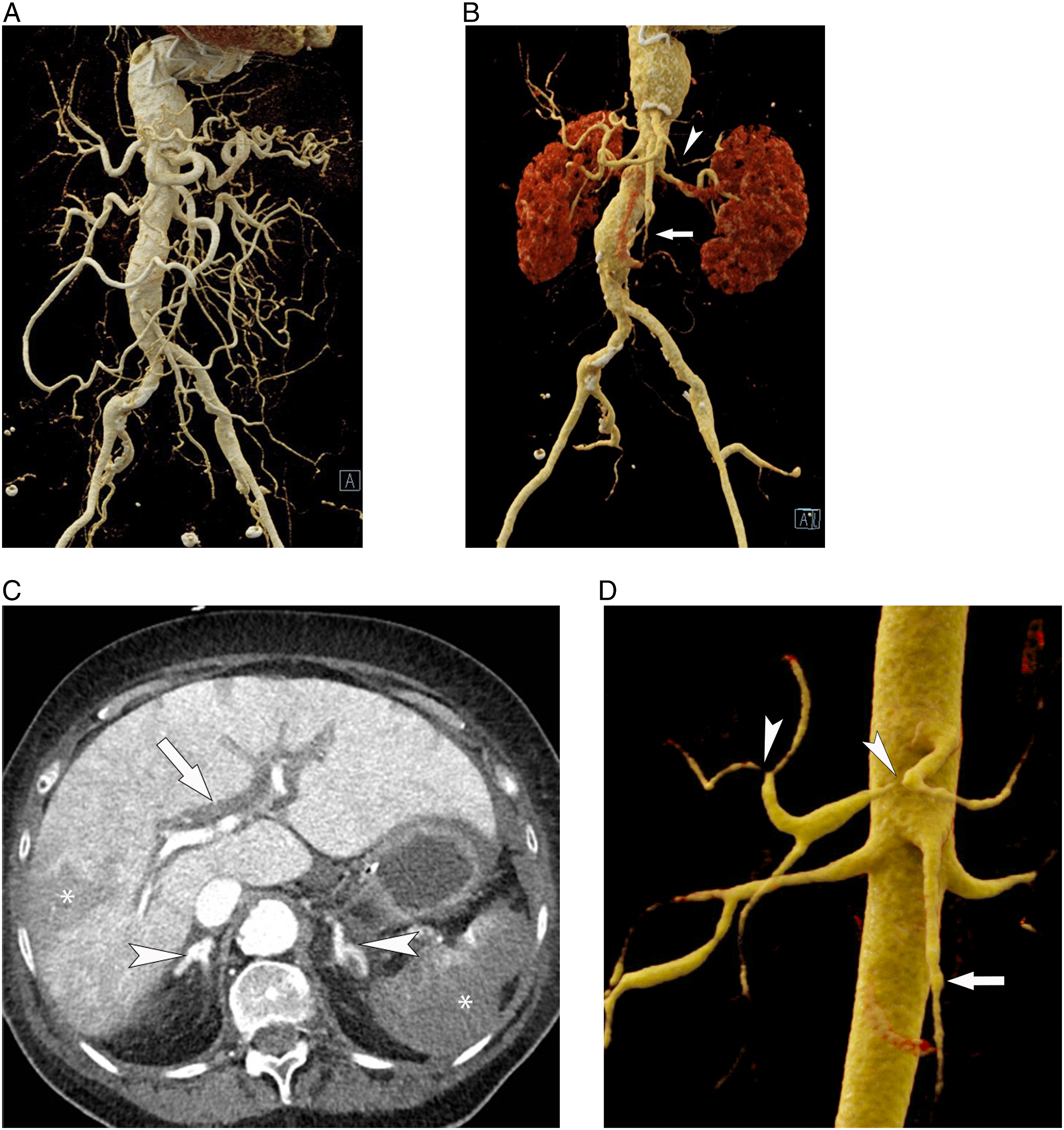
Differentiating NOMI from arterial or venous AMI is important as these are high surgical risk patients and reperfusion therapy is generally first attempted.27,31,33
Venous
Venous AMI is the least common of the 3 primary etiologies and comprises 5–20% of cases.1,5 As many as 60% of patients have a prior history of peripheral vein thrombosis or pulmonary embolus.10,14 While thrombosis can occur idiopathically or secondary to intra-abdominal pathology such as acute pancreatitis or recent surgery, many patients have systemic conditions resulting in a hypercoagulable state. 14
Mesenteric venous thrombus (MVT) appears as nonopacification or an intraluminal filling defect within the mesenteric veins (Figure 4). Non-occlusive thrombus typically presents as a filling defect with a rim of contrast opacification, an appearance which has been found to be over 90% specific for MVT.5,14,16 The presence of a superior mesenteric vein (SMV) thrombus is highly specific in patients with clinical suspicion for AMI at 94–100%.13,16,28,29 The affected vein is often enlarged and there can be engorgement of the upstream venous tributaries secondary to increased venous pressure.
5
The distribution is often initially segmental and slowly progressive, first affecting the smaller upstream veins before propagating into the SMV proper.5,14 Mesenteric venous thrombosis. Coronal reconstruction of a CT with intravenous contrast in the venous phase shows extensive thrombosis of the entire superior mesenteric venous arcade (arrowheads) and multiple loops of thickened small bowel which are poorly enhancing (arrows) within a large volume of ascites (*).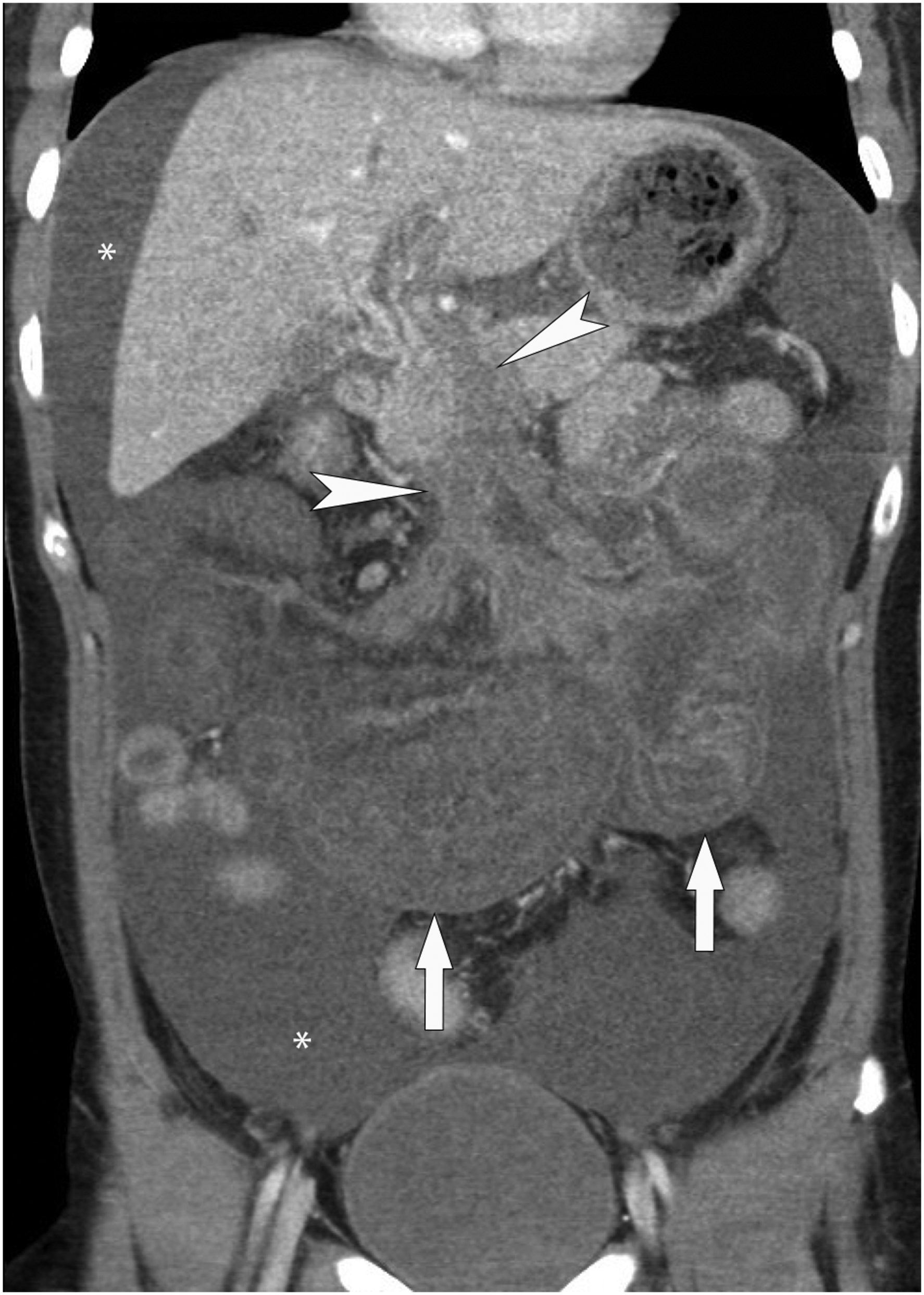
Even in cases of extensive clot burden, development of ischemia is usually more gradual compared to arterial AMI or NOMI and infarction typically does not occur unless there is extensive involvement of the upstream peripheral arcade or vasa recta branches.10,14 Primary involvement of the inferior mesenteric vein is also atypical. 14 Unlike the rather fulminant presentation seen in arterial AMI patients, venous AMI patients can present 1–4 weeks after onset with non-specific symptoms such as diffuse abdominal pain and diarrhea.1,14
Non-Vascular CT Findings
It should be noted that sensitivities and specificities of the various CT signs of ischemia in the literature for the last 2 decades are usually determined in a selected patient population where ischemia is clinically suspected, either due to pain out of proportion to physical findings, abdominal pain with biochemical evidence of ischemia (e.g., elevated lactate, metabolic acidosis), or abdominal pain with risk factors for ischemia (prior ischemia, hypercoagulable state, atrial fibrillation, significant peripheral vascular disease).
Pneumatosis Intestinalis and Portomesenteric Venous Gas
Pneumatosis intestinalis (PI) is the presence of gas dissecting through the layers of the bowel wall, which can extend into the mesenteric venous system and eventually into the portal vein.
PI and portomesenteric venous gas are present in 6–43% and 3–36% of AMI cases, respectively.11,13,16,28,29 The presence of PI does not always indicate irreversible transmural infarction. Wiesner et al found that 30% of their patients with portal venous gas and PI had partial mural rather than transmural infarction, a finding also corroborated by Kernagis et al which found that 40% of their study population with PI had viable bowel during surgery.38,39 Duron et al 40 also found that 47% of patients who still had viable bowel during surgery had PI on imaging. The reason may be that air can enter the bowel wall from mucosal breakdown prior to irreversible necrosis. Wiesner et al 38 found that more extensive “band-like” PI had greater association with transmural infarction (90%) compared to more isolated “bubble-like” PI (70%), and that PI in combination with portomesenteric venous gas was more strongly associated with transmural infarction (91%).
PI and portomesenteric gas in isolation are non-specific for AMI as they can also occur secondary to a large array of etiologies including benign causes.41,42 When AMI is suspected clinically, however, or when it is associated with an elevated lactic acid level, the presence of PI is highly specific for AMI (81–100%).13,14,16,21,28,29,41 PI seen in conjunction with other findings of AMI also increases specificity. Kirkpatrick et al 13 found that bowel wall hypoenhancement with PI has a sensitivity of 42% but a specificity of 97–100%.
An investigative pitfall is the presence of “pseudopneumatosis” where locules of gas trapped between bowel wall and intraluminal fluid can be mistaken for intramural air (Figure 5). Although sometimes challenging, pseudopneumatosis can be distinguished from true PI if the locules of gas are only associated with the dependently located intraluminal fluid. Interpretative pitfalls: Pseudopneumatosis. A) Transverse unenhanced CT image of a patient with small bowel ischemia and pneumatosis intestinalis (arrow). Note that air is seen both dependently and non-dependently within the wall of the involved bowel loop. B) Close-up view of the cecum from a CT with oral contrast where air trapped along the wall by liquid fecal material was mistaken for pneumatosis in a patient with right lower quadrant pain. When the bowel wall is thin, differentiation between air internal to the wall versus within the wall can be challenging, but true pneumatosis should not be related to the level of fluid within the bowel. In this case, there is no suspicion of pneumatosis above the air-fluid level.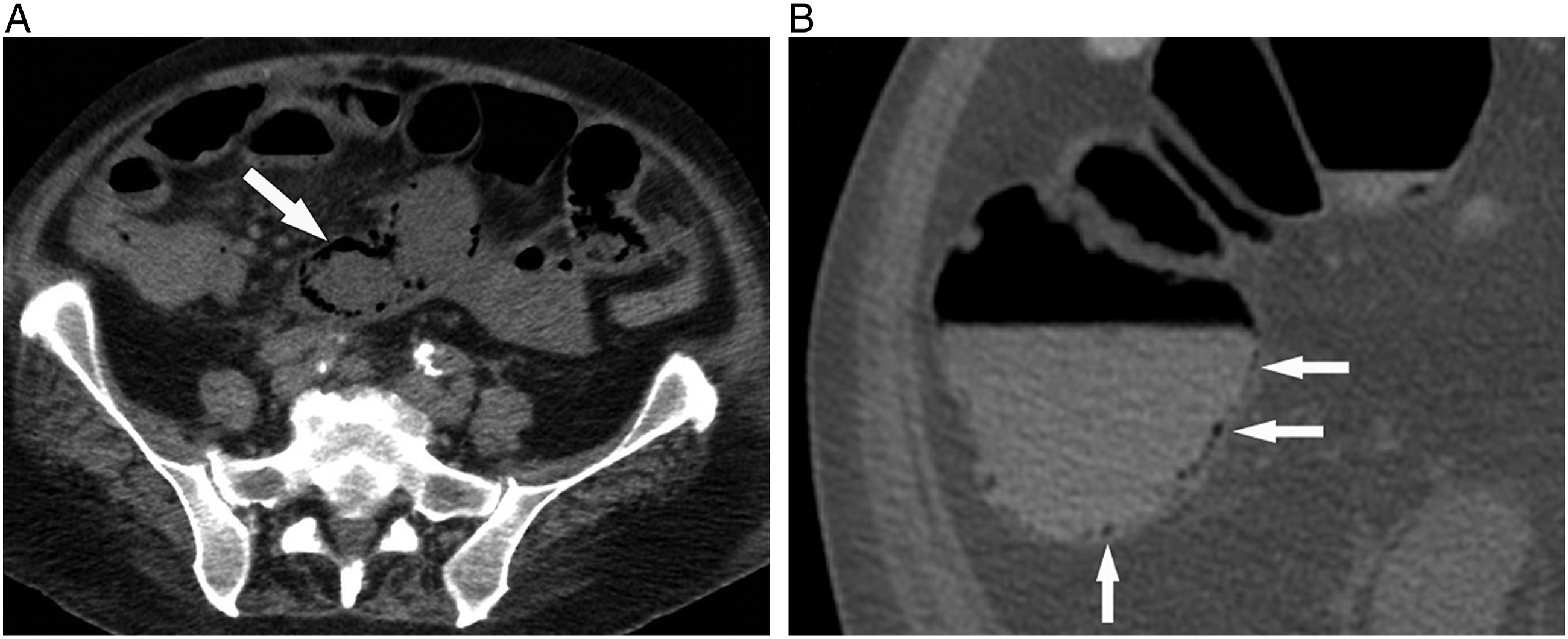
Changes in Bowel Wall Thickness
The prevalence of bowel wall thickening varies widely and has been found to be present in 26–96% of AMI cases.5,11,13,16,28 A common definition is >3 mm in adequately distended small bowel.13,28 Assessment of bowel wall thickening can also be qualitative and usually done in comparison to normal bowel.
Bowel wall thickening is more common with venous AMI, where it can progress to >15 mm although 8–9 mm is more typical.5,43 In this case, venous compromise with preserved arterial flow results in vascular engorgement, submucosal edema, and cyanosis giving a trilaminar appearance of a hypoenhancing submucosa sandwiched between hyperenhancing mucosa and muscularis propria/serosa (“target sign”).11,14,44 Ischemia can eventually occur as progressive and prolonged wall thickening and edema results in increased intramural pressure and eventual compromise in arterial perfusion. 5
While some authors have found sensitivities of 79–85%, bowel wall thickening is typically one of the least specific findings of AMI and can be found in multiple non-ischemic conditions.11,13,16,28,29 The presence and degree of bowel wall thickening also poorly correlate with disease severity, and many cases of arterial occlusive ischemia demonstrate no thickening at all (Figure 1).11,43
The presence of intramural hemorrhage can also cause bowel wall thickening, and this causes hyperattenuation of the thickened wall on nonenhanced CT.11,43,45 Intramural hemorrhage can occur as a consequence of ischemia, and has been described as a poorly sensitive (5–18%) but specific finding (90–98%) when AMI is clinically suspected. 14 A potential pitfall, however, is in patients anticoagulated for atrial fibrillation who present with abdominal pain that could be secondary to primary intramural hemorrhage from over-anticoagulation versus secondary hemorrhage from ischemia if under-anticoagulated and experiencing systemic emboli. Short segment involvement or thickening >1 cm are both more predictive of primary intramural hemorrhage rather than secondary hemorrhage from ischemia. 45
Bowel wall thinning is seen in arterial AMI and is caused by destruction of the intramural musculature and nerves causing loss of tone and progressive bowel dilation. 14 Wall thinning can appear markedly pronounced being termed “paper-thin” walls. 11 The finding has poor inter-observer agreement since normal walls can be quite thin, and this is a very challenging sign to prospectively identify.14,46
Decreased or Absent Bowel Wall Enhancement
Bowel wall hypoenhancement is a critical early finding of AMI.26,47,48 As with wall thickening, assessment of bowel wall enhancement is typically qualitative and is usually done in comparison to normal bowel (Figure 1).
Although prospectively identified in less than half of patients in many studies, bowel wall hypoenhancement is generally a highly specific imaging finding for AMI at 93–99% and the reason why maximizing enhancement of the normal bowel wall with optimized protocols is so important.13,16,24,29,30,46,49-51 One investigative pitfall is the presence of diffusely hypoenhancing or non-enhancing bowel, which can be overlooked as “normal” if there is no normally enhancing bowel for comparison (Figure 6). Interpretative pitfalls: diffuse nonenhancement. Transverse portal venous phase CT image in a patient with a large superior mesenteric artery embolus (not shown) demonstrates nonenhancement of every visible bowel loop with the exception of the descending colon (arrowhead). Some of the bowel blends in with the ascites and is difficult to even discretely identify. When the abnormal enhancement is so diffuse, it may actually be overlooked or dismissed as a suboptimal contrast bolus in error.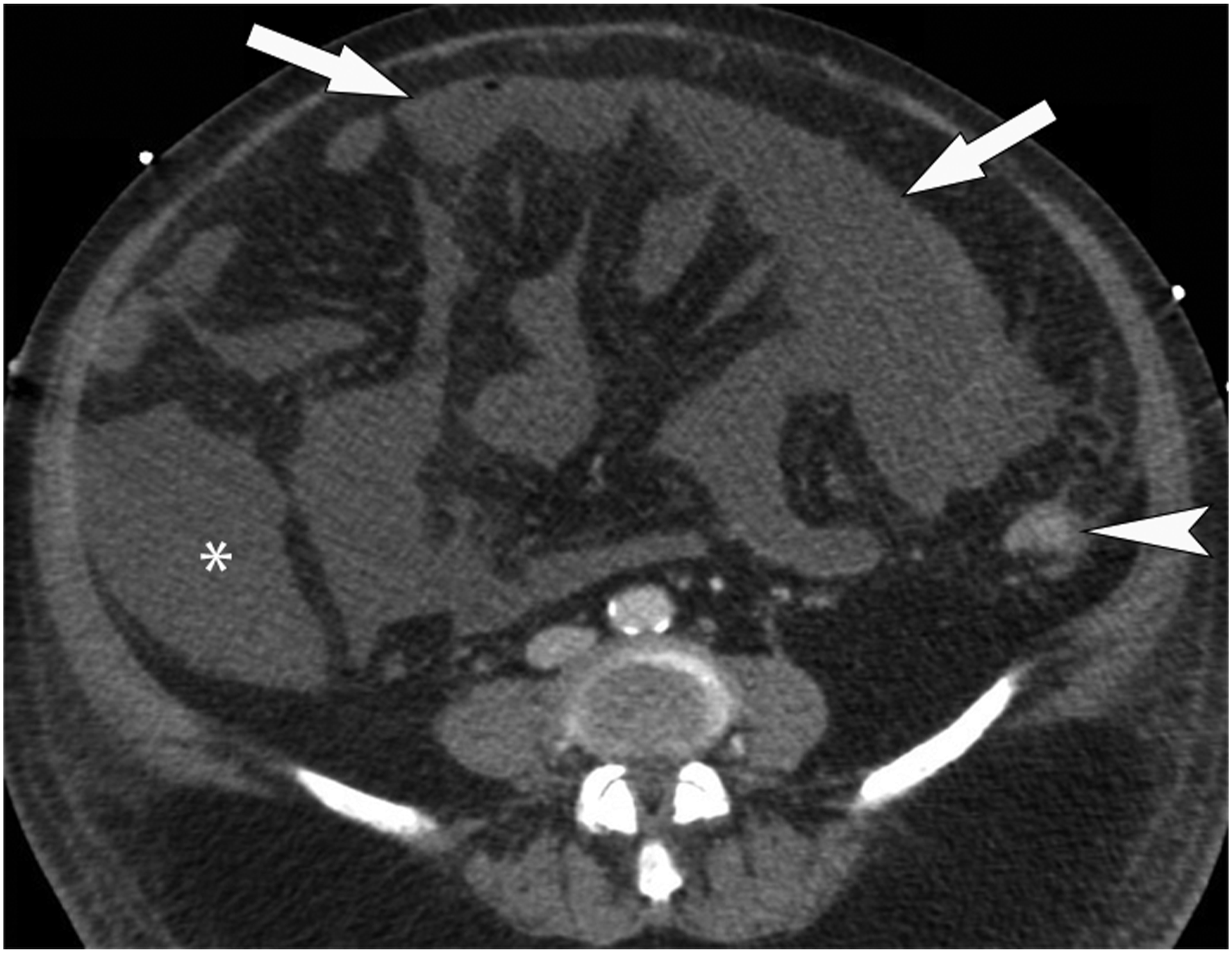
Paradoxically, the bowel wall can sometimes appear hyperenhancing in AMI; this is typically seen in venous AMI secondary to vascular congestion, reperfusion injury, and strangulated bowel obstruction.8,11,43
Bowel Luminal Dilation
Luminal dilatation and associated bowel air-fluid levels are another early finding present in 21–91% of cases of AMI and found more often in arterial AMI (Figures 1 and 7).11,13,14,28,47,52 The definition of “dilation” is typically small bowel over 2.5–3.0 cm in maximum diameter.13,14,28,47 The proposed pathophysiology is the interruption of normal bowel peristalsis and hypotonia, which can be reactive to ischemic insult or secondary to damage to the intramural musculature and nerves.11,14 Reported sensitivity and specificity of this finding are highly variable at 39–67% and 29–83%, respectively.13,16,21,28-30 As dilatation is so common with other diseases, its main utility is in drawing attention to involved bowel loops which can then be interrogated for other more specific signs. Reperfusion injury. Maximum intensity projection (MIP) reformations of mesenteric CT angiograms (CTAs) performed before (A) and after (B) embolectomy. A) On the initial CTA, the superior mesenteric artery (SMA) terminates abruptly at the level of the embolus (arrow). B) Following embolectomy, the vessel now is widely patent (arrow). C) Preprocedure transverse combined arterial/enteric phase CT shows multiple nonenhancing small bowel loops in the pelvis (arrows). D) Following embolectomy, a venous phase transverse CT shows these same loops (arrows) are now thickened and hyperenhancing (arrows) amongst mesenteric edema and ascites. Hyperenhancement can thus paradoxically be a sign of ischemia.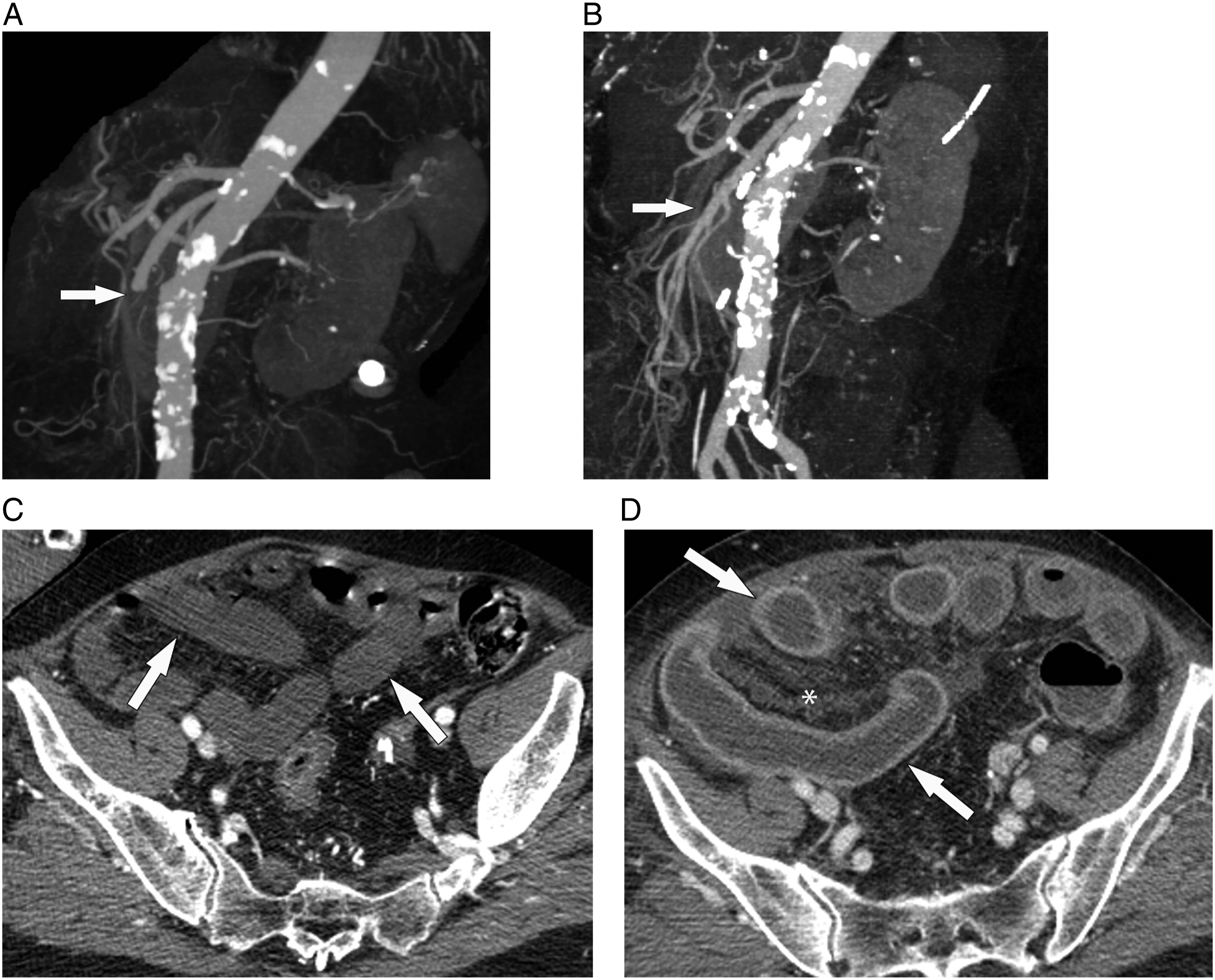
Mesenteric Fat Stranding and Ascites
Mesenteric fat stranding and ascites likely occur secondary to transudation of fluid caused by increased bowel wall permeability, vascular congestion, or transmural ischemia.5,11 They have been found to be sensitive but not specific signs of AMI even in patients where it is clinically suspected, with a sensitivity and specificity of 71–81% and 33–47% for ascites and 75–88% and 28–61% for mesenteric stranding, respectively.13,16,28-30 As with bowel wall thickening, the presence of mesenteric fat stranding and ascites poorly predicts disease severity. 43
Similar to bowel dilatation, mesenteric edema and ascites commonly occur with other diseases and are best used to direct attention to nearby bowel loops which might show other more specific signs of AMI (Figures 4, 6, and 7).
Reperfusion
Reperfusion of prior ischemic bowel can paradoxically worsen an ischemic insult, a process known as reperfusion injury (Figure 7). 37 As previously discussed, AMI increases bowel wall vascular permeability and causes mucosal disruption. 1 The reintroduction of oxygen-rich blood leads to infiltration of neutrophils with secretion of pro-inflammatory mediators and free radicals in the wall, causing further damage.1,31,38,53 This can lead to shock, DIC, and multiorgan failure. 53 Late reperfusion is associated with higher risk of reperfusion injury. 33
Outcome in NOMI can depend on whether there is effective or ineffective reperfusion. 37 Effective perfusion is associated with hyperenhancing bowel, bowel wall thickening, ascites and mesenteric fat stranding.14,27,33,37,54 The presence of effective reperfusion is associated with better prognosis and decreased mortality. 37 The presence of normal mesenteric vasculature with bowel wall thickening, a sign of effective reperfusion, is associated with significantly lower mortality in NOMI patients. 37 CT findings associated with ineffective reperfusion and poorer outcomes include bowel wall thinning, bowel hypoenhancement, diminished mesenteric vasculature, and decreased systemic and portomesenteric venous caliber.27,33,37,55 Given global hypoperfusion, solid organ ischemic changes are also seen in NOMI ranging from changes of the “CT hypoperfusion complex” such as hyperenhancement of the kidneys and adrenal gland, to wedge-shaped areas of infarction affecting the spleen, liver, and kidneys, to rarer entities such as ischemic cholecystitis or pancreatitis.27,56
Other Findings
Pneumoperitoneum is an ominous sign of bowel perforation secondary to transmural infarction and is seen in 21% of AMI patients; it is highly specific at 94–97%.13,16,28,29,37,52 Solid organ infarcts are frequently seen in AMI patients, particularly those with embolic AMI or NOMI; as such, its presence is highly specific as an ancillary finding in patients clinically suspected of having AMI at 94–96% (Figure 8).10,13,16,27-29 Solid organ infarcts. A) Maximum intensity projection (MIP) sagittal reformation of a CT angiogram (CTA) of the abdominal aorta shows opacification of the superior mesenteric artery (arrowhead) but complete occlusion of the celiac axis (arrow) in a postoperative patient who developed abdominal pain. B) Transverse venous phase CT image demonstrates splenic infarction (arrow) and multiple areas of hepatic infarction (arrowheads) later confirmed at autopsy.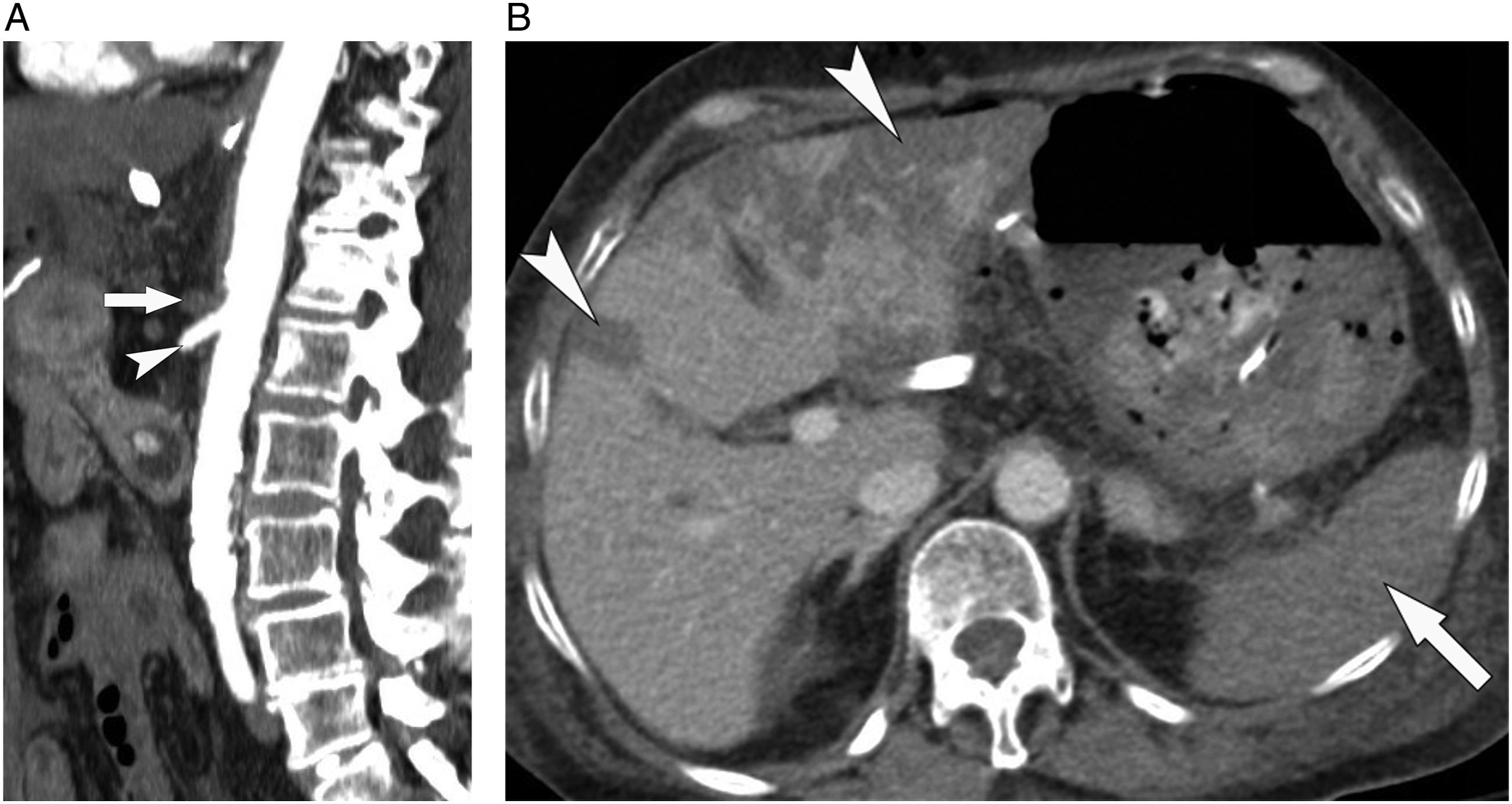
Secondary Mesenteric Ischemia
AMI can complicate small bowel obstruction (SBO) and is termed strangulation, affecting 10% of patients and usually those with closed-loops.
43
A closed-loop obstruction occurs when 2 high-grade transition points exist, usually in close proximity to one another (Figure 9).43,57,58 Closed-loop obstructions are at higher risk of AMI secondary to extrinsic compression of the vascular stalk, twisting of the mesentery causing first venous and later arterial vascular occlusion, and intraluminal dilation of the small bowel resulting in increased intramural pressure and decreased arterial perfusion.11,57 In addition to closed-loop obstruction, a similar process can occur with volvulus, intussusception, or a high-grade stenotic lesion such as a stricturing malignancy.11,57 Closed-loop small bowel obstruction. A) Coronal reconstruction of a portal venous phase CT with intravenous contrast shows dilated upstream loops of small bowel with normal, thin, enhancing walls (arrowhead), in contrast to the dilated closed loop where the wall is poorly enhancing and thickened distally (arrow). Free fluid (*) is seen in the mesentery and peritoneal cavity. B) Transverse image in the same patient demonstrates the transition point, secondary to an adhesion (arrow), and the proximal and distal ends of the hypoenhancing closed loop (*).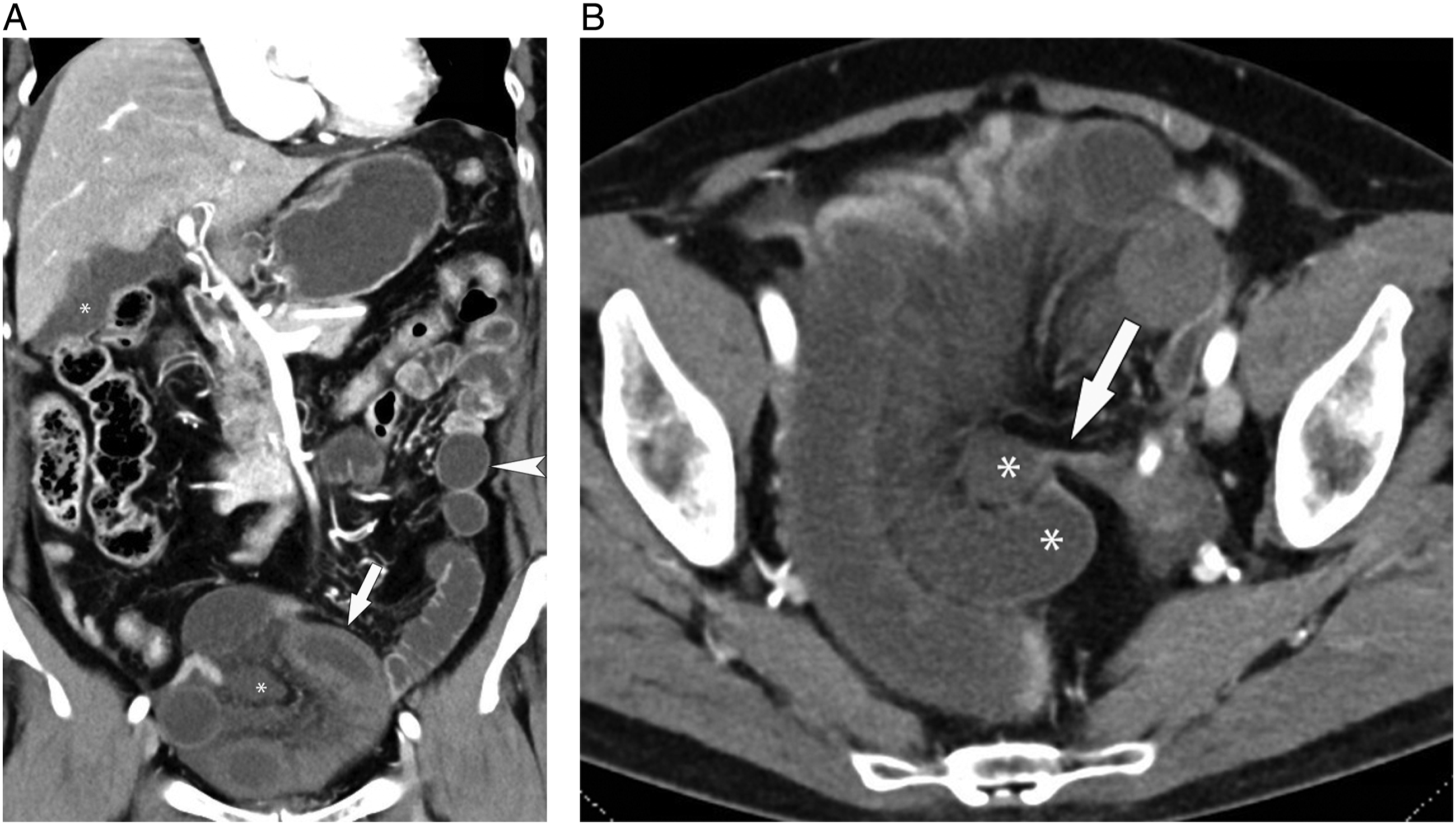
Identifying ischemia associated with an SBO can be challenging since every non-vascular sign of AMI has been described as associated with strangulation at some point in the literature, and yet many (e.g., mesenteric edema, ascites, wall thickening) can be seen when ischemia is not present and are simply due to congestion.48–51,57–61 There are a few key points which have been borne out over multiple studies. First, the absence of any mesenteric or mural findings has a high negative predictive value for strangulation (i.e., no bowel wall thickening or hypoenhancement, and no mesenteric edema or ascites).26,59,61 Second, bowel wall hypoenhancement in SBO is the most reliable sign of secondary ischemia.60,61
In summary, an evidence-based way to approach these cases is to a) have a high index of suspicion for ischemia with any closed-loop SBO, b) any other SBO with no other bowel wall or mesenteric findings has a low likelihood of strangulation, c) the more signs of ischemia present, the greater the level of concern, and d) bowel wall hypoenhancement should be considered the most reliable indicator of strangulation.
Conclusion
The greatest impact radiologists can make in AMI is to establish a diagnosis early and before irreversible necrosis. CT angiographic findings of thrombosis, emboli, or NOMI are specific but require an adequate arterial phase acquisition. Bowel wall hypoenhancement is also an early and specific sign but requires a protocol optimizing iodine conspicuity to confidently identify, and techniques such as using dual energy, low kVp scanning, or a combined arterial/enteric phase can be helpful. A properly optimized biphasic exam combined with knowledge of all of the potential signs of ischemia will help speed diagnosis and reduce mortality.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
