Abstract
Acute stroke is a widespread, debilitating disease. Fortunately, it also has one of the most effective therapeutic options available in medicine, endovascular treatment. Imaging plays a major role in the diagnosis of stroke and aids in appropriate therapy selection. Given the rapid accumulation of evidence for patient subgroups and concurrent broadening of therapeutic options and indications, it is important to recognize the benefits of certain imaging technologies for specific situations. An effective imaging protocol should: 1) be fast, 2) easily implementable, 3) produce reliable results, 4) have few contraindications, and 5) be safe, all with the goal of providing the patient the best chance of achieving a favorable outcome. In the following, we provide a review of the currently available imaging technologies, their advantages and disadvantages, as well as an overview of the future of stroke imaging. Finally, we offer a perspective.
Acute ischemic stroke (AIS) is a widespread, debilitating disease, globally affecting approximately 9.5 million people per year. 1 In the US and Canada, it is a leading cause of death and disability. 2 Fortunately, it also has one of the most effective therapeutic options available in medicine, endovascular treatment (EVT), with an astonishingly low number needed to treat of 2.6 for patients with large vessel occlusions (LVO). 3 Prior to EVT, intravenous (IV) thrombolytics comprised the standard of care and, currently, if patients are eligible, both are often used in combination to achieve and maintain vessel patency. 4
The AIS population is heterogenous, however in the Canadian-led HERMES meta-analysis of the large EVT trials5-9 (one of which was also Canadian-led), the investigators did not identify any subgroups in which the treatment was not effective. 10 These initial trials, however, involved highly selected patient cohorts and were thus able to show a clear benefit, albeit at the cost of excluding many patients who may have benefited from treatment.7,11 Since then, many trials have been done or are being conducted on patients who fall outside of these strict inclusion criteria. The BASICS (ClinicalTrials.gov Identifier: NCT01717755) and BEST 12 trials examined the effect of EVT on vertebrobasilar occlusions versus best medical management and found no significant difference in patient outcome, in contrast to a registry-based study. 13 However, both trials were relatively small and limited to crossover, and there was no evidence of a detrimental effect of EVT. The late window trials DAWN and DEFUSE 3 used CTP and MRI-based imaging criteria to show the efficacy of EVT in patients presenting beyond the 6-hour time window,14,15 which have been adopted in many clinics around the world. Recently, the focus has shifted to medium vessel occlusions (MeVOs); data from non-randomized studies suggest that EVT of MeVOs is both possible and safe,16-19 however high-level evidence from randomized trials is lacking. Furthermore, trials investigating the effect of EVT on outcome in patients with large baseline infarctions and minor stroke are currently underway (SELECT-2 (ClinicalTrials.gov Identifier: NCT03876457), TESLA (ClinicalTrials.gov Identifier: NCT03805308), TENSION (ClinicalTrials.gov Identifier: NCT03094715), LASTE (ClinicalTrials.gov Identifier: NCT03811769) and MOSTE (ClinicalTrials.gov Identifier: NCT03796468)). Researchers often use the term “futile treatment” when referring to EVT that did not lead to an improvement in outcomes, and patient selection criteria are applied to identify patients who are most likely to benefit from EVT, thereby also aiming to keep the “futile” EVT procedures at a minimum. This approach is however problematic, since the decision to treat or not to treat with EVT is made based on available information prior to treatment, and there are several factors after the treatment decision has been made that also influence outcomes, such as reperfusion quality, stroke unit care, and post-stroke complications. Even in cases in which EVT does not lead to an improvement in outcome, the complication rate of the EVT procedure appears to be low and there is no evidence of harm in patients with failed EVT compared to those who are not treated with EVT. 20 A synopsis of the current American and Canadian evidence-based imaging-related guideline recommendations can be found in Table 1.
COR: class of recommendation (I: strong benefit, IIa: moderate; IIb weak; III: no benefit/harm); LOE: level of evidence (A: high quality, B-R/B-NR: moderate quality; C-LD: studies with limitation of design or execution; C-EO: consensus of expert opinion); EVT: endovascular treatment; mRS: modified Rankin scale; NIHSS: National Institutes of Health Stroke Scale; ASPECTS: Alberta Stroke Program Early CT Score; ACA: anterior cerebral artery; PCA: posterior cerebral artery; M2/M3: second and third segments of the middle cerebral artery; LVO: large vessel occlusion
While EVT is highly effective in (re)-opening the vessel, it must be done in a timely manner. Indeed, an estimated 2 million neurons die for every prolonged minute of vessel occlusion, highlighting the acute, time critical nature of this disease (“time is brain”). 22 As such, it is crucial to minimize the steps between stroke onset and recanalization.
Acute stroke due to non-traumatic hemorrhage represents 10-15% of all stroke cases, however is associated with higher rates of mortality and morbidity. 23 Imaging plays a major role in the differentiation of diagnosis between AIS and stroke due to hemorrhage and thereby aids in the triage of patients for appropriate therapy selection. It has evolved rapidly over the years, from a simple non-contrast computed tomography (NCCT) scan performed within a week of onset to sophisticated software programs that allow for quasi-immediate automatic brain parenchyma assessment and detection of the occluded vessel. These technological developments are iteratively occurring within an adaptive framework in response to broadening therapeutic indications. At the same time, there has been an increase in imaging technology accessibility, allowing diagnostic stroke workups to be performed at smaller tertiary or more rural clinics. As a result, multiple imaging strategies exist, largely dependent on institutional resources, regional guideline recommendations, and, to some extent, stroke physician preferences. Regardless, the goal has remained the same: to quickly diagnose and appropriately treat the patient for the best chance of achieving a favorable outcome.
Goals of Imaging in Acute Stroke
When a patient presents to the emergency department with suspected stroke, neuroimaging is paramount to confirm the diagnosis and helps physicians to make timely treatment decisions by excluding the presence of hemorrhage or other stroke mimics, visualizing the occlusion site, and assessing the extent of tissue damage/ischemic changes. All this information is necessary for appropriate patient triage and therapy selection, which is arguably the most important role of neuroimaging in stroke. To this end, most AIS imaging protocols include parenchymal imaging, vascular imaging, and some form of functional imaging to assess for irreversibly damaged versus potentially salvageable tissue (i.e., so-called “core” versus penumbra); otherwise described as the assessment of the 3 Cs: core, clot, collaterals.
In addition, acute stroke imaging findings can also help predict outcomes following treatment and may guide further patient management in the early post-acute phase, e.g., by identifying potential sources of stroke or underlying vessel pathology. While CT remains the fastest and most widely available imaging modality, magnetic resonance imaging (MRI) is also employed for the AIS workup, particularly for certain patient subgroups (e.g., those presenting in the late time window). 24 As in many aspects of medicine, there is no one-size-fits-all strategy; each step of the decision pathway, including choice of imaging, is based on a culmination of factors, including local resources. However, from our own experience, it is useful to standardize acute stroke imaging within one institution so that the physicians on call and imaging technicians know exactly what to do, which streamlines workflows and avoids confusion and subsequent time delays.
In the following, we present an overview of the currently available state-of-the-art stroke imaging modalities, their advantages and disadvantages (Table 2), and their role in acute treatment decision-making, outcome prognosis, and post-acute management. Due to the in our opinion limited applicability of MRI for the acute stroke situation, we have placed the focus on CT-based protocols. We also provide a brief discussion on the future of stroke imaging technology and how that could impact our current protocols and practices.
Advantages and Disadvantages of Various Acute Stroke Protocols.
CT: computed tomography; CTA: computed tomography angiography; NCCT: non-contrast CT; spCTA: single phase CTA; mCTA: multiphase CTA; CTP: CT perfusion; MRI: magnetic resonance imaging; ASPECTS: Alberta Stroke Program Early CT Score; AVM: arteriovenous malformation; MeVO: medium vessel occlusion; LVO: large vessel occlusion; EVT: endovascular treatment; TOF: time of flight
* Often cited as an advantage over iodinated contrast CTA-studies, however a meta-analysis could not detect an increase of acute kidney injury in patients who received contrast 28
Imaging Techniques Used for Treatment Decision-Making in Acute Ischemic Stroke
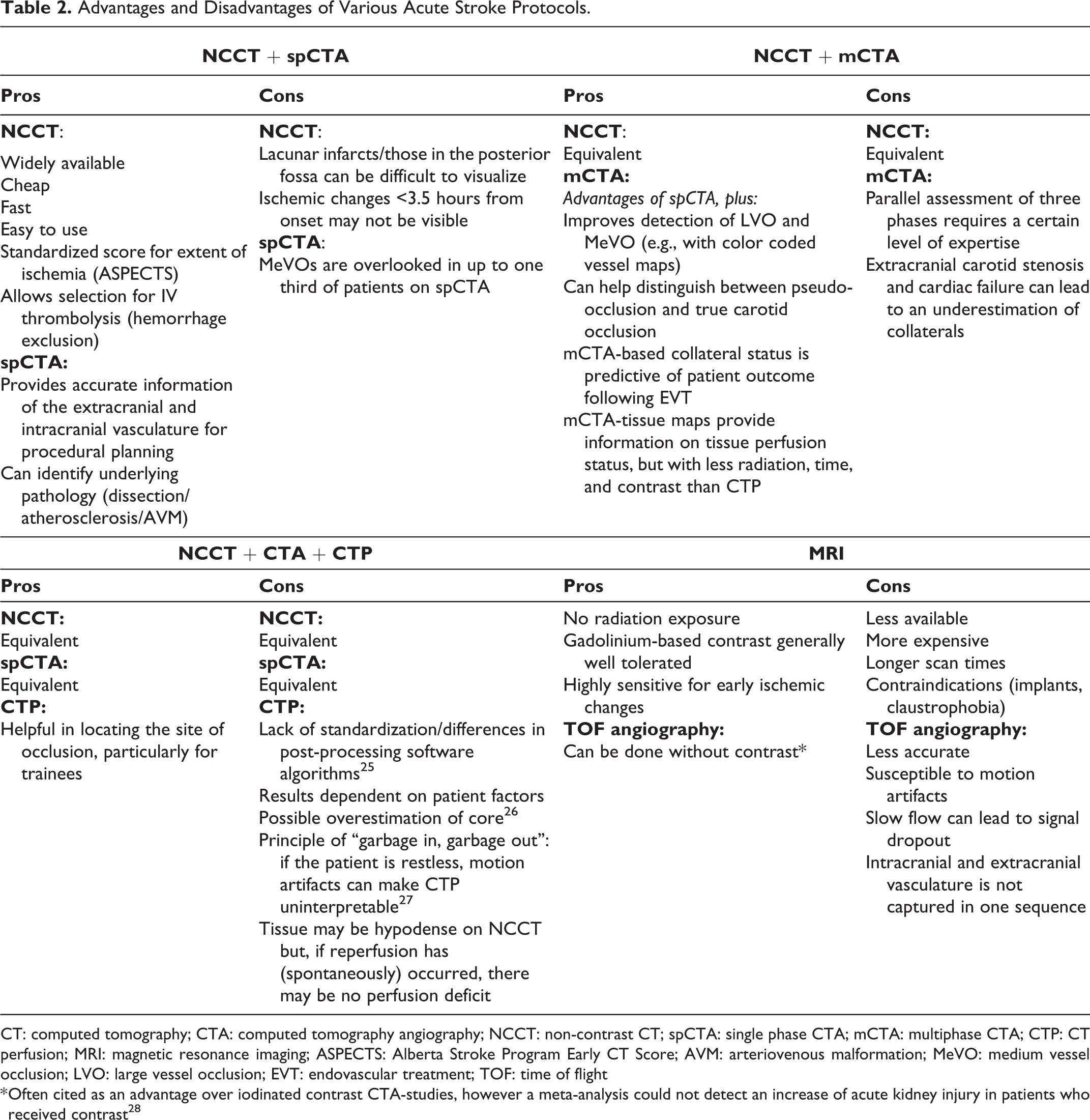
The information needed for treatment decision-making that can be obtained from parenchymal imaging includes a) exclusion of intracranial hemorrhage (ICH), and b) exclusion of extensive ischemic changes (usually assessed with the Alberta Stroke Program Early CT Score [ASPECTS], Figure 1), as both these findings would preclude treatment with IV thrombolysis and EVT.
Exemplary NCCT with the labeled ASPECTS regions (no early ischemic changes = ASPECTS 10). M1: anterior middle cerebral artery (MCA) cortex/frontal operculum; M2: MCA cortex lateral to insular ribbon/anterior temporal lobe; M3: posterior MCA cortex/posterior temporal lobe; M4: anterior MCA territory superior to M1; M5: lateral MCA territory superior to M2; M6: posterior MCA territory superior to M3; I: insula; C: caudate; L: lentiform nucleus; IC: internal capsule. Figure modified from aspectsinstroke.com.
CT Based
As previously mentioned, NCCT remains the primary imaging modality for the diagnosis of AIS. 29 It is fast, widely available, and allows for easy imaging of critically ill patients. Furthermore, CT scanners are improving, producing higher-quality images at lower radiation doses.
Newer machine-learning algorithms 38 and attenuation-based biomarkers such as net water uptake provide a more granular picture of developing hypodensity (as opposed to the binary nature of ASPECTS) and are also gaining attention. 39 When employed in combination with “classic” radiological features of AIS such as a hyperdense vessel sign, 40 NCCT-based assessment provides a powerful first imaging step in the AIS diagnostic workup.
MRI Based
In some cases (and if available), MRI-based imaging can be advantageous over CT, despite longer acquisition times (MRI has been shown to delay door to needle times in a quasi-randomized trial 41 ) and more complex monitoring systems. For example, when the NCCT is negative, yet the clinical suspicion of AIS remains high.
In patients presenting in the >4.5-hour time window or those with unknown time of onset, fluid-attenuated inversion recovery (FLAIR)/DWI mismatch is commonly used to determine eligibility for IV thrombolytics.
Vascular Imaging
The information needed for treatment decision-making that can be obtained from vascular imaging includes a) identification of a vessel occlusion and its location, since this will decide whether a patient is amenable for EVT or not, and b) assessment of the aortic arch and cervical arteries, which is important for the choice of access site and appropriate devices in case EVT is performed. For hemorrhagic stroke, vascular imaging is useful for the detection of underlying pathology.
CT Based
CT angiography (CTA) provides an iodine contrast-based visualization of the cerebral and cervical vasculature, usually from the aortic arch to the vertex.
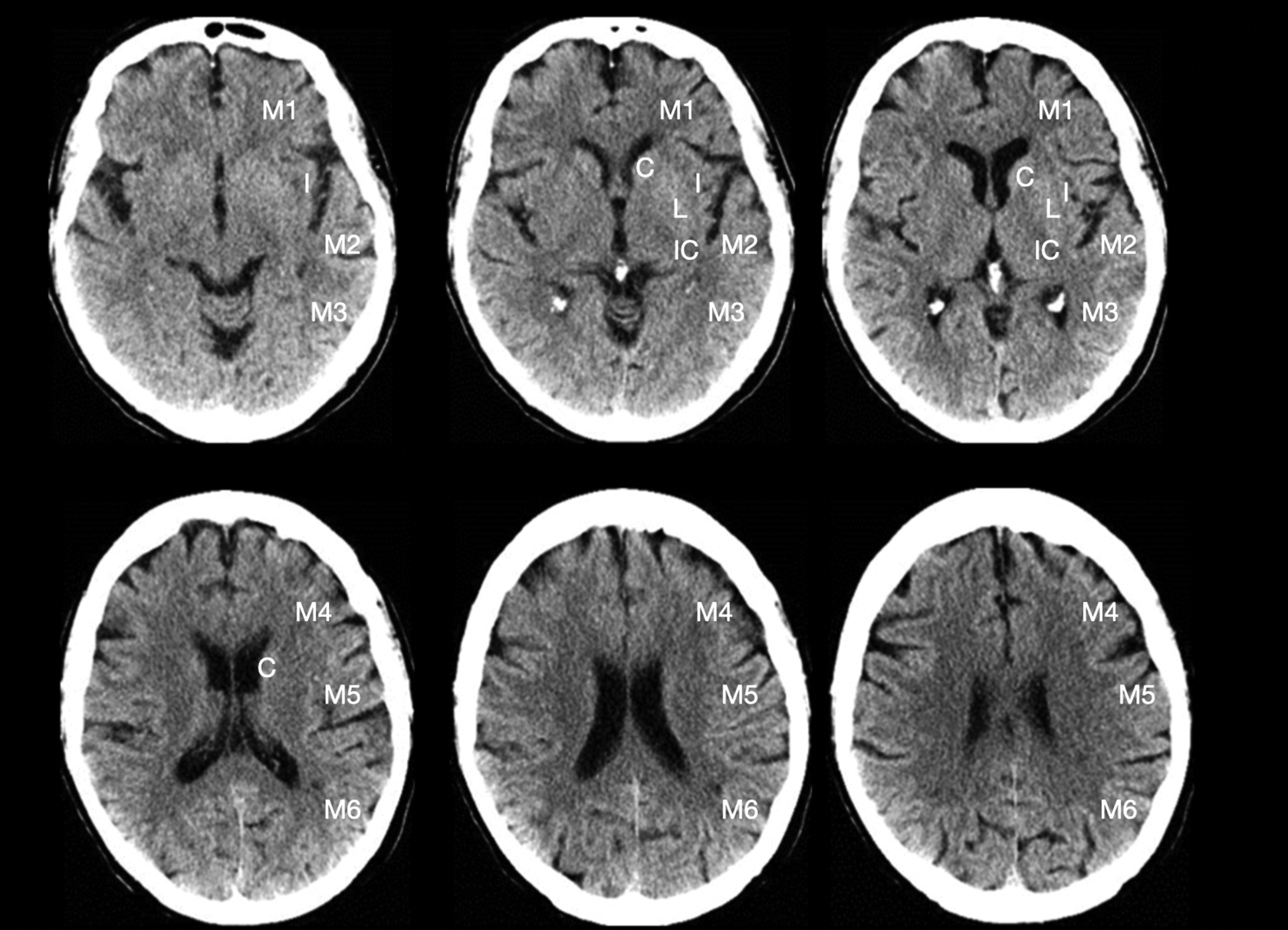
Typical imaging protocol in patients with suspected acute ischemic stroke using multiphase CTA. First, NCCT is obtained to rule out hemorrhage and provide an estimate of early ischemic parenchymal changes (A-C). mCTA is then performed: The first phase scans the extracranial vasculature from the aortic arch to vertex. The second and third phases scan only the intracranial vasculature (skull base to vertex). The delayed filling of collateral vessels in the second and third phases (black circles) point toward the site of occlusion (in this case a left-sided M2 segment middle cerebral artery occlusion) and allow for estimation of the collateral blood supply of the ischemic parenchyma. Detecting this occlusion on the first phase, which is the only available phase in regular single-phase CTA, could have been challenging. Blue areas of the middle images indicate the axial coverage of each phase. NCCT: non-contrast CT; mCTA: multiphase CTA.
Advanced Imaging for AIS
The goal of functional imaging in AIS at baseline is to assess the extent of ischemic core and the “penumbra” (tissue at risk, which is not yet irreversibly damaged and would benefit from reperfusion). 54 The current belief is that immediate reperfusion saves penumbral tissue, but not core, and reperfusing the latter therefore does not result in improved outcomes but may cause harm due to an increased risk of reperfusion hemorrhage. The current practice is to proceed with treatment when there is a small ischemic core and a large penumbra (“target mismatch profile”) and to forego treatment when a large core and little or no penumbra is present.
CT based
mCTA
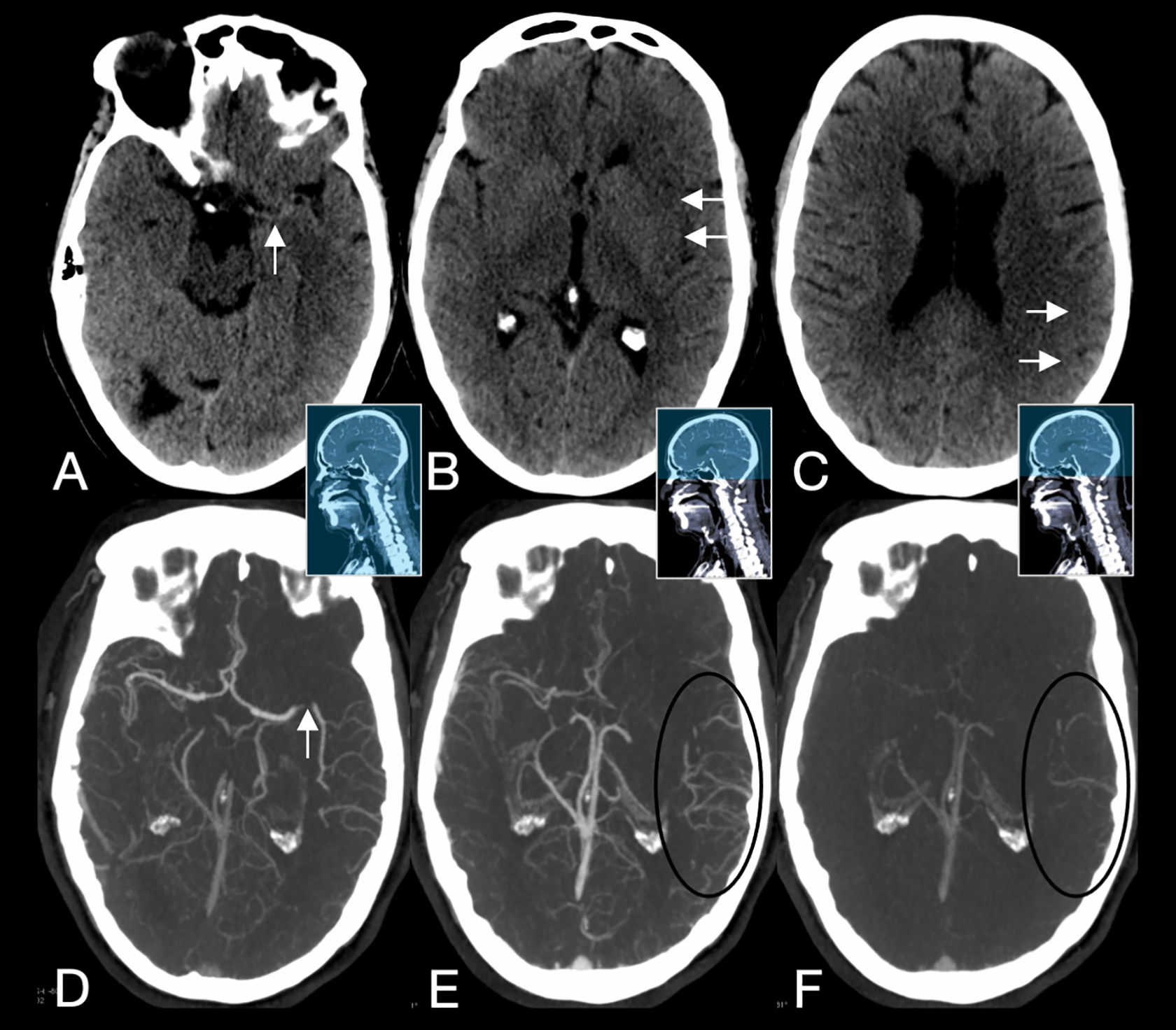
mCTA displays pial-arterial collateral filling in a time-resolved fashion, which can be translated into a 3-point score (good vs. intermediate vs. poor collaterals) 45 (Figure 2). mCTA collateral status is predictive of patient outcome following EVT, 45 as was shown in the ESCAPE trial; 5 patients with good mCTA collaterals were more likely to benefit from EVT, even if the ASPECTS was low. 55 mCTA collateral status also predicts outcome following IV thrombolysis. 56 mCTA is robust and easy to implement; however, extracranial carotid stenosis and cardiac failure can lead to an underestimation of collaterals. 45 Collateral scoring on conventional mCTA requires simultaneous/side-by-side reading of all 3 phases, which assumes a certain level of expertise. To facilitate mCTA readings, a novel display format has been developed in which time variant color maps are superimposed to create a single image (Figure 3). 57 These have been shown to further improve prediction outcome in patients with AIS due to LVO compared to conventional mCTA collateral grading. 58
mCTA-based color summation maps facilitate vessel occlusion detection by superimposing all 3 phases (A-C) onto a single, color-coded image (D). The first, early arterial phase (A) is represented in red, the second, late arterial phase (B) is shown in green, and the third, venous phase (C) is shown in blue.
CT perfusion maps
During CT perfusion (CTP), the brain is scanned multiple times after administration of a bolus of IV contrast. Manufacturer-specific software algorithms then produce color coded output maps that reflect hemodynamics and changes in cerebral blood volume (CBV) in comparison to the contralateral, unaffected side. These most commonly include mean transit time (MTT), cerebral blood flow (CBF), time to maximum (Tmax), and CBV, and rely on the central volume principle of CBF = CBV/MTT. 59 Together, they help to identify the site of vessel occlusion, as well as unmask ischemic changes that are not immediately visible on the NCCT, both of which can be particularly useful for trainees. CTP-derived parameters like the hypoperfusion intensity ratio (HIR) can also give an estimate of the collateral status. HIR is defined as the ratio of volume with a Tmax greater than 10 seconds (i.e., severe hypoperfusion) to the volume of tissue with a Tmax greater than 6 seconds 60 and has been used to estimate the infarct progression rate for decision making in potential transfer scenarios. 61
NCCT, CTA, and CT perfusion (CTP) now comprise the “state-of-the-art-advanced imaging” protocol of many stroke centers. Whole brain CTP is used to estimate “core” and penumbra volume, which aids in patient selection for EVT, particularly those with an onset or last known well of greater than 6 hours.4,14,15 Importantly, CTP is not necessary for clinical decision-making in patients presenting within 6 hours from last known well and obtaining CTP should never delay IV thrombolysis or EVT.
The disadvantages of CTP are partially a result of the longer acquisition, processing, and interpretation times. Furthermore, CTP represents a snapshot in time and is dependent on patient cardiac function and blood pressure. 62 Concomitant extracranial carotid stenosis can lead to hypoperfusion of the supplied hemisphere and result in under/overestimation of the core and penumbra, respectively.63,64 Previous infarcts and white matter disease can also confound the results. 65
Postprocessing algorithms employ varying thresholds to display the degree of mismatch between infarct core and penumbra, by providing volumes for each that could give the readers a false sense of precision. Brain tissue with CBF values ≥5 mL/100 g/min have been shown to regain function if reperfusion is rapidly achieved. 66 As a result, these measures are at most a probabilistic estimate of tissue that will proceed to infarction if the vessel is not recanalized, otherwise previously referred to as severe ischemic tissue of unknown viability (sit-UV). 26
mCTA tissue maps
An alternative to perfusion-like imaging can be achieved with mCTA-derived tissue maps. Since mCTA allows for whole brain time resolved images of the pial vessels, information on tissue perfusion status can be extracted using a discrete 4-point time intensity curve. These maps are similar to conventional CTP maps (Figure 4), but require a substantially lower radiation dose, less contrast, and do not result in prolonged imaging times. A recent study by Wu et al observed that mCTA-based perfusion models were able to predict infarct core, penumbra, and perfusion status using machine learning, with comparable accuracy to CTP. 48
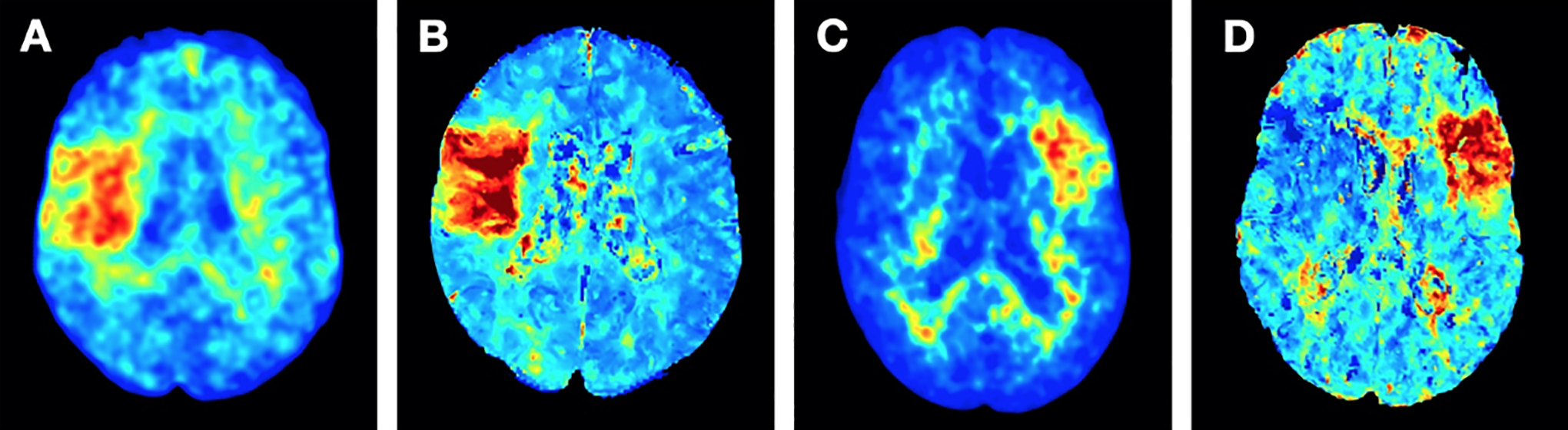
Right-sided M2 (A, B) and left-sided M3 occlusion (C, D). mCTA tissue maps (A, C) show a clear change of color in the affected parenchyma, thereby allowing for fast and accurate vessel detection, similar to CTP maps (B, D). mCTA: multiphase CTA; CTP: CT perfusion.
An Optimal Acute Stroke Imaging Protocol: Our Perspective
At the current moment, many institutions choose a combination of imaging techniques for the diagnosis, treatment selection, and monitoring of AIS patients. The choice varies dependent on patient factors (e.g., comorbidities, time from onset), as well as on institutional resources and expertise. Prior to the administration of IV thrombolytics, for example, only the possibility of hemorrhage needs to be ruled out; it is not necessary to wait for vascular imaging. In most hospitals, CT is still the imaging modality of choice; not only is it the fastest, most widely available technology, but it also has relatively few contraindications and is often more easily implemented than MRI. Indeed, an ideal stroke imaging technology should fulfill the following criteria: 1) it should be fast, 2) it should be easily implementable, 3) it should produce reliable results, 4) it should have very few contraindications, and 5) it should be safe. The additional information that can be gained from more advanced imaging techniques needs to be balanced against the time delays and postprocessing-related noise that come along with them (Figure 5).
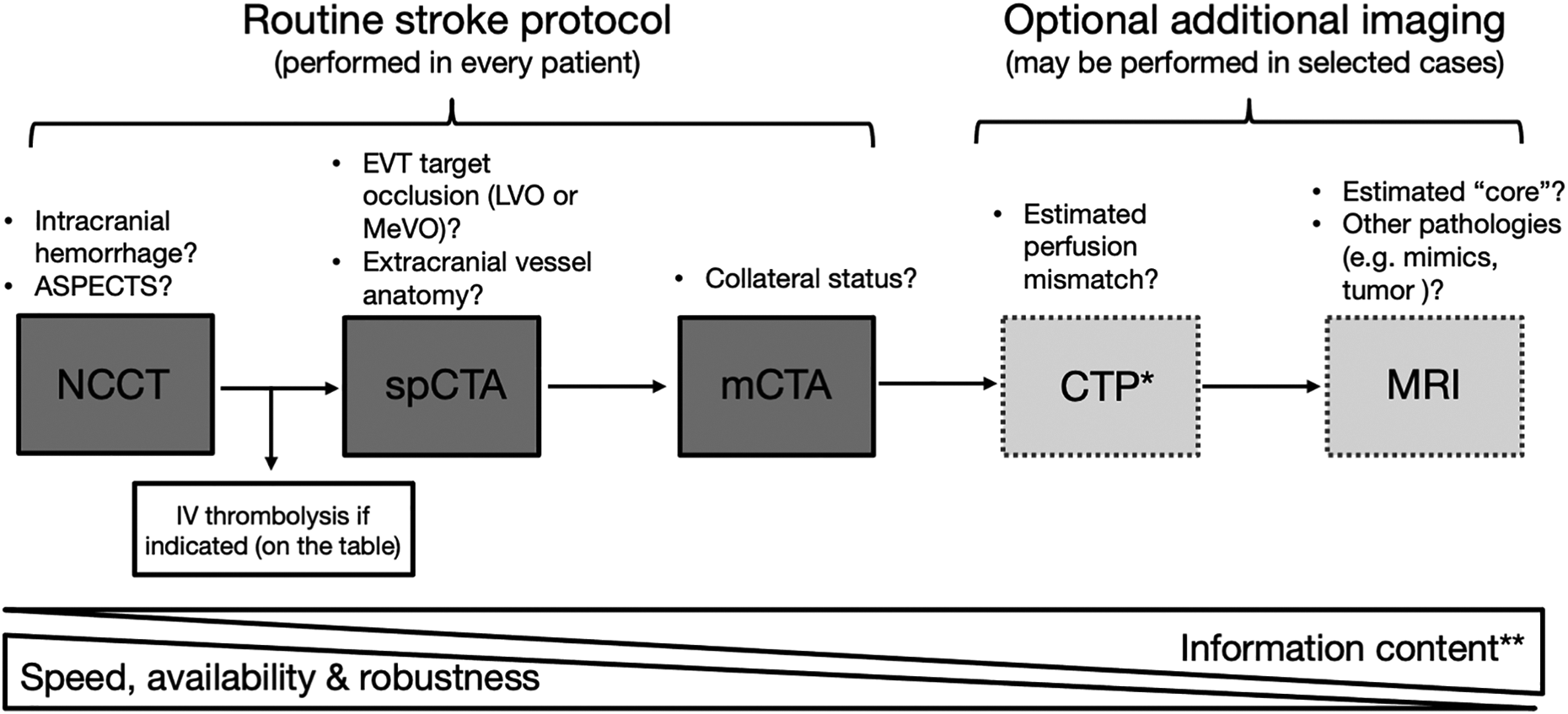
Every stroke protocol should include an NCCT and a form of extracranial + intracranial vascular imaging (single-phase or multiphase CTA). The speed and accuracy that can be achieved with these modalities, in conjunction with the patient’s clinical presentation and history, is sufficient in the vast majority of cases for appropriate therapeutic decision-making. NCCT: non-contrast CT; spCTA: single phase CTA; mCTA: multiphase CTA; CTP: CT perfusion; MRI: magnetic resonance imaging; ASPECTS: Alberta Stroke Program Early CT Score *CTP can be useful in that it can aid trainees in identification of the occluded vessel. **Increased information content may not always be relevant; knowing the exact volume of an ischemic lesion is neither possible with our current technology, nor is it pertinent to patient treatment.
At the current moment, CT seems to provide the most reasonable compromise/balance of these factors. Figure 6 outlines a current guideline-based imaging protocol. However, as our knowledge of stroke increases, along with broadening therapeutic options and indications, it is important to recognize the benefits of certain imaging technologies for specific situations. For the Canadian healthcare infrastructure and other countries with similarly wide-spread geographies, for example, the transfer of AIS patients from rural hospitals to EVT-capable hospitals often results in substantial time delays. While CTP is recommended for the late time window (>6 hours) by the current guidelines, in our experience mCTA is of at least equal value, requiring no post-processing and being both more robust and inclusive when selecting patients for treatment.
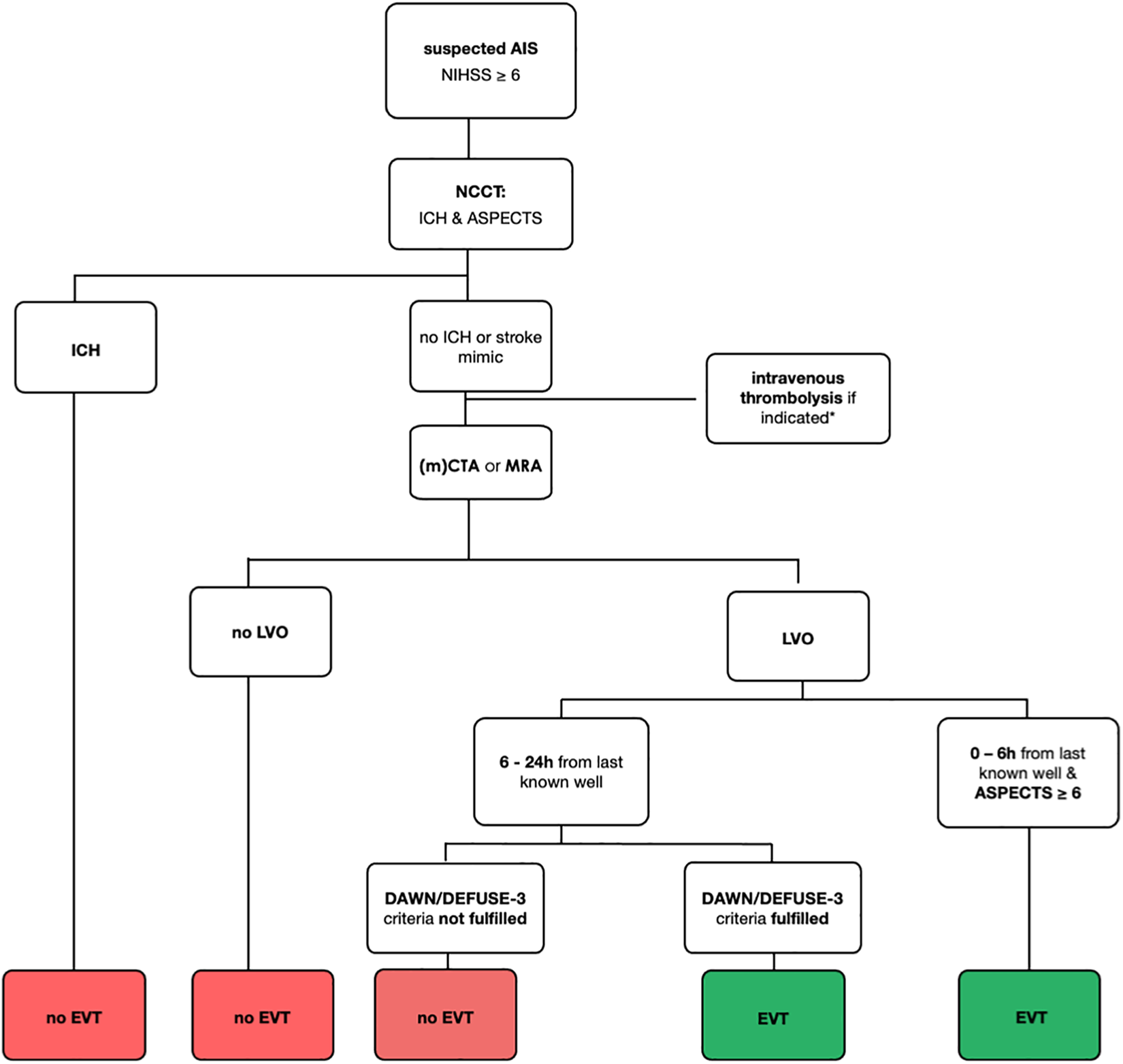
Flowchart of current guideline-based recommendations for AIS decision-making. If the protocol takes place at a center participating in a randomized clinical trial, randomization should occur when possible. AIS: acute ischemic stroke; NCCT: non-contrast CT; ICH: intracranial hemorrhage; mCTA: multiphase CTA; LVO: large vessel occlusion; EVT: endovascular treatment; ASPECTS: Alberta Stroke Program Early CT Score.* For intravenous thromboylsis eligibility criteria, see current guidelines.4,21
Importance of Acute Stroke Imaging Findings in the Post-Acute Phase
Neuroimaging is not only crucial for acute treatment decision-making; it can also be used to guide post-acute treatment and secondary stroke prevention in several ways.
Stroke Pattern
The patterns of ischemic lesions on MRI imaging can help to differentiate between large vessel or microvascular/embolic disease, which will result in different secondary stroke prevention regimens.
Pre-Existing Cerebrovascular Disease
MRI sequences such as gradient echo and susceptibility weighted imaging can elucidate etiologies of microbleeds and provide useful clues for diagnosis, such as cerebral amyloid angiopathy, which may lead to a more detailed diagnostic work-up and influence further management.
Extracranial Carotid Artery Disease
Extracranial MRA including vessel wall MRI and even routine CTA allow for plaque/vessel wall characterization for prognostication purposes, which can help in the investigation of stroke etiology (e.g., degree of carotid stenosis, carotid webs, symptomatic non-stenotic carotid disease [SyNC])67-69 and guide secondary prevention measures. 70 Transcranial Doppler is a noninvasive, real-time assessment of cerebrovascular function without the need of contrast medium and can also provide information regarding vessel wall changes and carotid plaque morphology, certain features of which are associated with a higher risk of (recurrent) stroke. 67
New developments in the Pre-Hospital Setting
Despite the major strides made for the treatment of AIS in recent years, time still remains a limiting factor. Indeed, since 2015, many hospitals have had success in streamlining their workflows to shorten the time from admission to recanalization, thereby achieving higher rates of good functional outcome. 71 However, the time from activation of emergency medical services to hospital admission will always remain variable and more complex to control. Nevertheless, imaging in the field could help first responders make critical decisions regarding transfer of a patient (e.g., to a primary stroke center versus a comprehensive stroke center with EVT capabilities). There are currently a number of technologies under development, including mobile stroke units (MSUs) and specially designed stroke helmets for the detection vessel occlusion and assessment of tissue perfusion. MSUs were first piloted in Germany in 2010 and are rapidly gaining attention.72-74 They are equipped with an on-board CT and are often accompanied by a stroke physician; patients can be screened for hemorrhage which would allow earlier IV thrombolysis administration, while some are outfitted with CTA capabilities, further aiding in patient triage. 75 “Stroke helmets” employ electromagnetic, 76 ultrasound, 77 microwave, 78 and EEG 79 based technologies (among others) to differentiate between ischemic and hemorrhagic stroke, or other stroke mimics. Such technologies could help to substantially shorten the pre-hospital interval, thereby likely improving patient outcomes. Currently, however, most of these devices are in the early investigational phase.
Future Directions of Imaging
Finally, artificial intelligence and machine learning are rapidly changing the way image-based diagnosis and processing are performed. Recently, Wu et al developed a machine learning approach for the automatic segmentation of infarct lesions on NCCT images that had good agreement with stroke volumes as determined on DWI-MRI. 38 As previously mentioned, another study demonstrated that mCTA-based automated prediction of ischemic brain tissue fate was feasible, with comparable results to CTP imaging. 48 Deep convolutional networks have also been used to detect the presence of LVO on mCTA. 80 Once fully developed and validated, these technologies will help to avoid misdiagnosis and delayed diagnosis of AIS, thereby reducing treatment delays, guiding patient triage and transfer, and ultimately improving patient outcomes, which is particularly valuable in the Canadian healthcare infrastructure with its wide-spread geography, in which the transfer of AIS patients from rural hospitals to EVT-capable hospitals is both time and resource-consuming and results in patients being treated far away from their homes and families.
Conclusion
Stroke imaging technology has advanced rapidly in recent years and will likely continue to do so. This is in part due to a need to adapt to novel treatment options, but also a result of the rise of artificial intelligence, machine learning, and automation. However, it is important to remember that outcomes are a result of a complex interaction of variables, from patient history to post-therapeutic rehabilitation strategies. While we may have a multitude of options at our disposal, imaging represents only one facet of the stroke story. Clinical presentation and imaging studies are complementary diagnostic measures we can continue to integrate and use to further improve our ability to offer this powerful treatment to as many patients as possible.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

