Abstract
The Canadian Association of Radiologists Incidental Findings Working Group consists of both academic subspecialty and general radiologists and is tasked with adapting and expanding upon the American College of Radiology incidental findings white papers to more closely apply to Canadian practice patterns, particularly more comprehensively dealing with the role of ultrasound and pursuing more cost-effective approaches to the workup of incidental findings without compromising patient care. Presented here are the 2021 Canadian guidelines for the management of pancreatic incidental findings. Topics covered include anatomic variants, fatty atrophy, pancreatic calcifications, ductal ectasia, and management of incidental pancreatic cysts.
Introduction
In 2017, the Canadian Association of Radiologists (CAR) received feedback from its membership that there was a need for a Canada-specific set of incidental findings guidelines which would acknowledge the unique distribution of imaging modalities and resource limitations faced by radiologists and healthcare systems as a whole in this country. The CAR Incidental Findings Working Group was formed in February 2018 and tasked with either adapting pre-existing American College of Radiology (ACR) guidelines to meet the concerns of the CAR membership or authoring new guidelines.
The Working Group consists of both academic subspecialty and general radiologists practicing in Canada. Major international guidelines reviewed included the American College of Radiology white papers,1-3 American Gastroenterological Association Institute Guideline, 4 the European Pancreatic Cyst Guidelines, 5 and the International Consensus Guidelines for the Management of IPMN and MCN of the Pancreas. 6 Regional practice guidelines within Canada were also assessed by the working group. These guidelines formed the basis of the CAR incidental findings working group recommendations following review of areas of discrepancy between Canadian practice patterns, difficulties in following guidelines as written, or areas in which an alternative approach was more cost-effective without compromising patient care. In order to support any changes, a literature database in the cloud was established consisting of the references used by the ACR committees, and additional papers were added which had become available since those publications providing supporting evidence of any proposed changes. The literature review demonstrated that while many recommendations from the ACR white paper in particular applied well to Canadian practice, the management of incidental cystic lesions of the pancreas remains divisive and challenging in a health care system facing increasing issues balancing the cost of medical imaging with the probability of significant disease. All management recommendations were discussed as a group for consensus.
Per standard CAR process for the approval of guidelines, a draft version of the article was posted on the CAR website for member feedback and peer review prior to submission for publication; all member comments were addressed prior to article submission.
While the flowcharts and tables presented are based on the best available scientific evidence there remain continued significant gaps in the literature: ultimately this document reflects consensus recommendations rather than a fully evidence-based standard of care.
Anatomic Variants Mimicking Disease
There are several known congenital and acquired conditions of the pancreas that can simulate a pancreatic mass, the so called “pseudomass.”7-11 Both focal chronic pancreatitis and autoimmune pancreatitis can appear mass-like, mimicking pancreatic adenocarcinoma.9,12 A smooth tapering pancreatic duct passing through the mass-like area or absence of ancillary signs such as an abrupt pancreatic duct cut off, double duct sign (associated dilated CBD with the dilated pancreatic duct), or upstream pancreatic gland atrophy help distinguish between a pseudomass and true pancreatic adenocarcinoma.13,14
Juxtapapillary duodenal diverticula arise near the ampulla of Vater and protrude into the pancreatic head. These typically have an air fluid level leading one to the correct diagnosis, but if completely fluid filled these diverticula can be mistaken for a true mass. Multiplanar reformats and CT with oral contrast usually allow one to see the communicating channel with the duodenal lumen. 10 Rarely, these can cause obstructive jaundice thought to be secondary to local inflammation and fibrosis affecting the common bile duct (Lemmel’s syndrome). 15
Normal pancreatic lobulations may also be misconstrued for pathology; pancreatic lobulations are defined as outpouching of the pancreatic parenchyma greater than 1 cm in relation to the anterior superior pancreaticoduodenal artery8,9,16 (Figure 1). An outpouching along the anterior surface of the pancreatic body to the left of the superior mesenteric vessels is called the tuber omentale.8,9 Intrapancreatic splenosis (or intrapancreatic accessory spleen) commonly presents as a well-defined mass measuring 1-3 cm in the pancreatic tail, appearing identical to normal splenic tissue on all modalities including the typical heterogeneous early enhancement of normal splenic tissue.9,11 When in doubt, intrapancreatic splenosis can be confirmed with Technetium-99 m heat denatured RBC SPECT/CT nuclear medicine scan with a high degree of sensitivity and specificity. 11
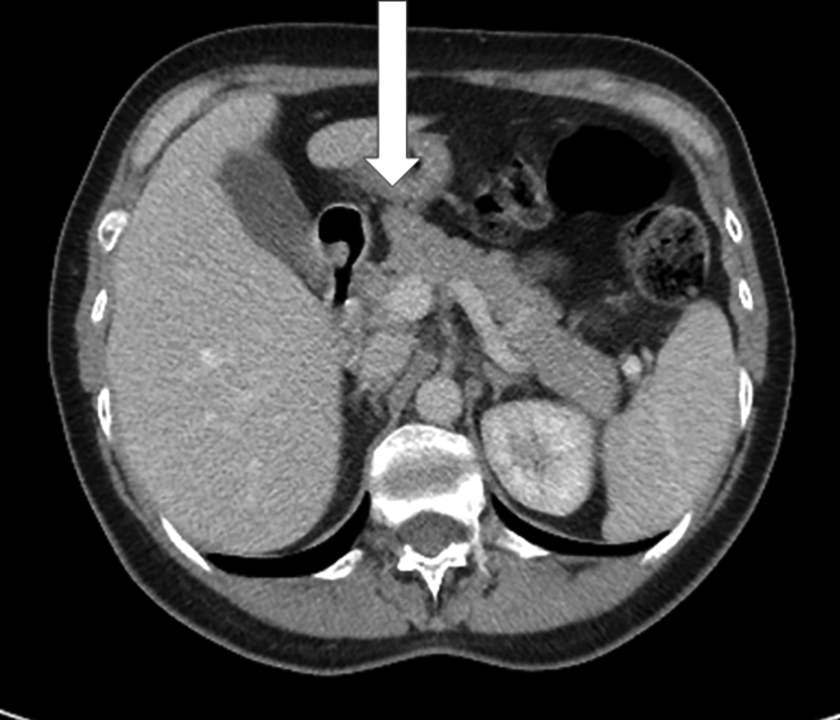
Normal pancreatic lobulation.
Pancreatic Lipomatosis
The most frequent pathologic finding in the adult pancreas is some degree of lipomatosis, otherwise known as fatty replacement or fatty infiltration. In a pathologic series of over a 100 cases, all but 6 had some degree of fatty replacement. 17 The degree of fatty replacement can be highly variable and often demonstrates a patchy heterogeneous distribution throughout the gland. This can result in a “two-tone” pancreas effect due to the difference in appearance of the normal pancreas in contrast to those areas with fatty infiltration. The patterns of distribution of fatty replacement are important to recognize to prevent the misdiagnosis of residual normal pancreatic tissue for a pancreatic mass. Four different types of uneven pancreatic lipomatosis have been described (Figure 2): replacement of the head with sparing of the uncinate process and peribiliary region, body, and tail; replacement of the head, neck, and body, with sparing of the uncinate process and peribiliary region, and tail; replacement of the head, including the uncinate process, and sparing of the peribiliary region, body, and tail; and total replacement of the pancreas with sparing of the peribiliary region.18,19 While pancreatic lipomatosis has been suggested to increase the risk of pancreatic cancer, screening for pancreatic cancer is not currently recommended in patients with pancreatic lipomatosis. MRI may be considered if the fatty infiltration does not match 1 of the 4 common patterns of lipomatosis, but such imaging should be the exception rather than the rule.
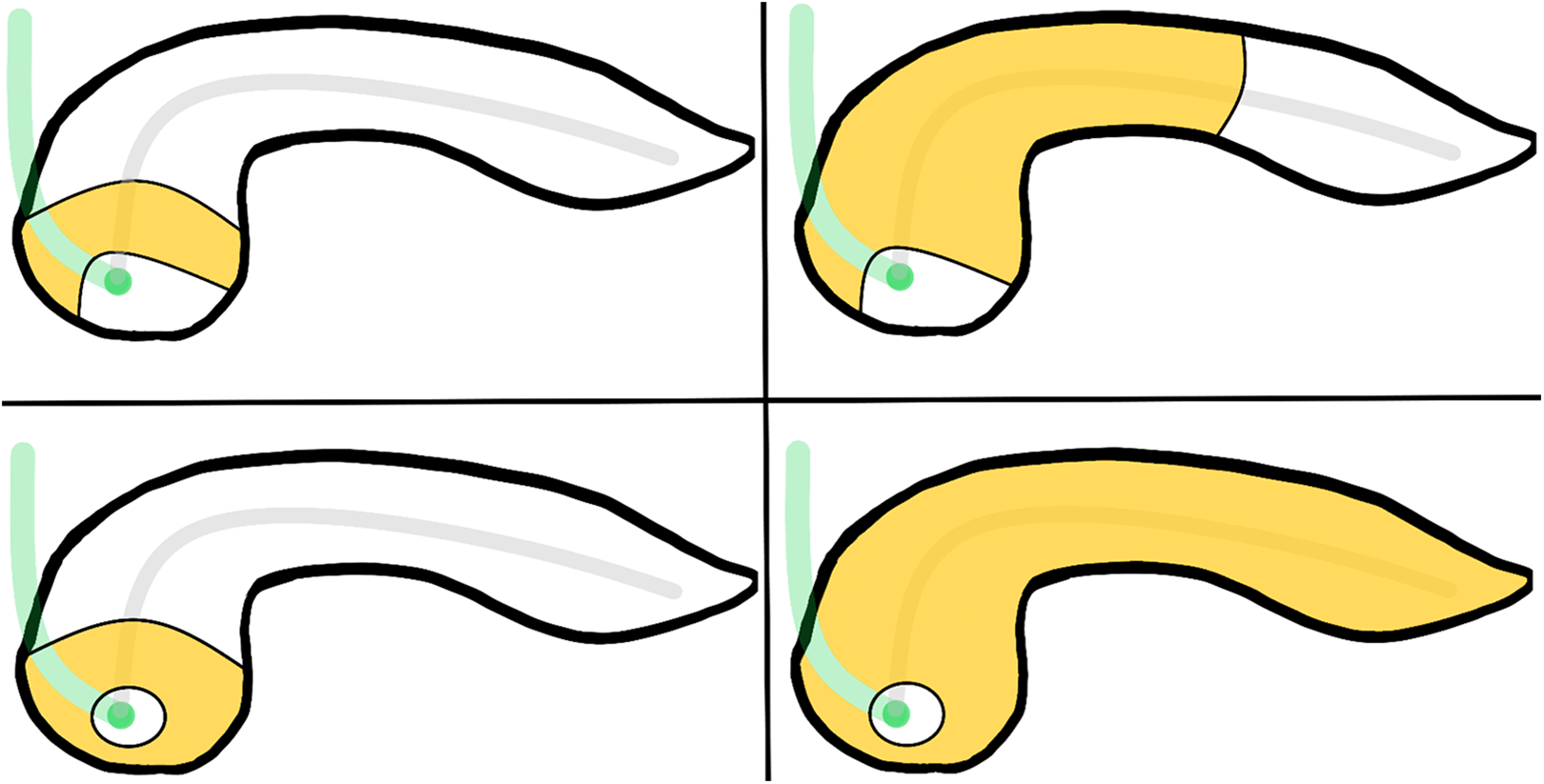
Uneven pancreatic lipomatosis involving only the pancreatic head (A), the pancreatic head, neck and body (B), the pancreatic head and uncinate process (C), and the entire pancreas (D). Note continued sparing of the peribiliary region in all 4 patterns of lipomatosis.
Pancreatic lipomatosis is not limited to the senescent pancreas, demonstrating a significant correlation with obesity, type II diabetes mellitus (DM), and severe generalized atherosclerosis. It is also highly associated with chronic pancreatitis, any pancreatic duct-obstructing process, and genetic syndromes such as cystic fibrosis and Shwachman—Diamond syndrome. 20 Due to the high association between fatty pancreas with metabolic syndrome, obesity and type-II DM, the term non-alcoholic fatty pancreas disease (NAFPD) has been gaining recognition and is often concomitantly associated with non-alcoholic fatty liver disease (NAFLD). 21 The reporting radiologist may choose to include the presence of pancreatic lipomatosis in their radiology report impression, particularly in the younger patient population, as it may lead the ordering physician to discuss important lifestyle changes for the patient.
Pancreatic Calcifications
When focal calcification is identified within the pancreas, the radiologist should first identify and follow the course of the splenic artery to ensure such calcifications are not vascular in origin. The differential diagnosis of pancreatic calcifications includes calcification following pancreatitis, calcification within a neoplasm, or idiopathic ductal calculi unrelated to pancreatitis.22,23 Typically, there will be some other abnormality suggestive of a mass in the case of intra-neoplastic calcium, and the presence of calcification associated with a contour abnormality or focal change in parenchymal appearance should prompt dedicated imaging with multiphasic pancreatic CT or MRI. 23 The primary diagnostic dilemma for radiologists encountering pancreatic calcification, however, is when to diagnose chronic pancreatitis.
Pancreatitis is the most common cause of pancreatic calcification, and chronic pancreatitis secondary to alcohol consumption has historically been felt to be more likely than other causes to result in foci of calcification, although other factors such as genetic predisposition and smoking may play a role.22,23 It is important to recognize that 1 or 2 punctate calcifications incidentally found within the pancreas are not diagnostic of chronic pancreatitis alone. There currently are no evidence-based criteria or consensus guidelines for when pancreatic calcification should prompt an imaging diagnosis of chronic pancreatitis, but limited studies suggest that the number, size and distribution of the calcifications are important. Calcifications can be classified as punctate when < 3 mm or coarse when
The Working Group recommends that when calcifications are identified, an attempt should be made to localize them (ductal versus parenchymal), characterize them as punctate (< 3 mm) or coarse (
Pancreatic Ductal Dilatation
Differences in pancreatic ductal diameter between men and women are minimal and likely not clinically significant, but previous studies have shown that the mean duct diameter does increase slightly with age. Ductal diameter may increase up to 0.5-0.7 mm between the second and eighth decades.27,28 At autopsy, a study of 112 patients showed that 18 had a main duct diameter of > 4 mm (11 of the patients being > 70 years of age), but it is uncommon for the duct to reach this calibre in vivo in imaged patients without pancreatic disease. 17 Prior research has shown mean pancreatic duct diameters ranging between 2.0-2.6 mm in elderly patients, up to 3.4 mm at 2 standard deviations above the mean, and this has led to a consensus that 3.5 mm represents a reasonable cut-off for diagnosing ductal dilatation. These same studies showed that ductal calibre may gradually increase from the tail through the head.24,27-30
The Working Group recommends diagnosing ductal ectasia only when the duct is
Management of Pancreatic Cysts
Incidental pancreatic cystic lesions are increasingly being diagnosed as the use of medical imaging increases, with prevalence rates ranging from 2-49% in the literature.5,31-37 The vast majority of incidental pancreatic cysts have no malignant potential, being either developmental or the sequelae of prior inflammation. However, mucinous pancreatic neoplasms—a small subset of simple cystic pancreatic lesions—have been associated with an increased risk of malignancy; rates of invasive carcinoma in mucin-containing cystic pancreatic neoplasms approach 36% in some studies. The challenge of balancing the cost to work up incidental cystic pancreatic lesions with the probability of developing an associated pancreatic malignancy has resulted in a variety of specialty-based, pan-specialty, and geographic guidelines implemented across the world. As no single guideline has been adopted or agreed upon by the majority of specialties, following extensive literature review the CAR Incidental Findings Working Group has decided to recommend a Canadian-specific approach to the management of incidental pancreatic cysts in an attempt to balance the costly workup of an overwhelmingly benign and common finding.
Overall, our recommendations blend the 2017 International Consensus Guidelines, the 2017 ACR white paper on incidental pancreatic cystic lesions, and the 2015 American Gastroenterology Association guidelines with additional consideration of work up cost within the Canadian healthcare system. Compared to existing major guidelines, the CAR Incidental Findings Working Group recommendations make 3 significant additions: simple cystic pancreatic lesions 5 mm or less in longest maximum diameter (in any plane) do not require follow-up imaging, an upper age limit of 75 years for follow-up examinations has been established, and follow-up imaging has been limited to a total of 5 years for the majority of patients.
Criteria for Follow-Up
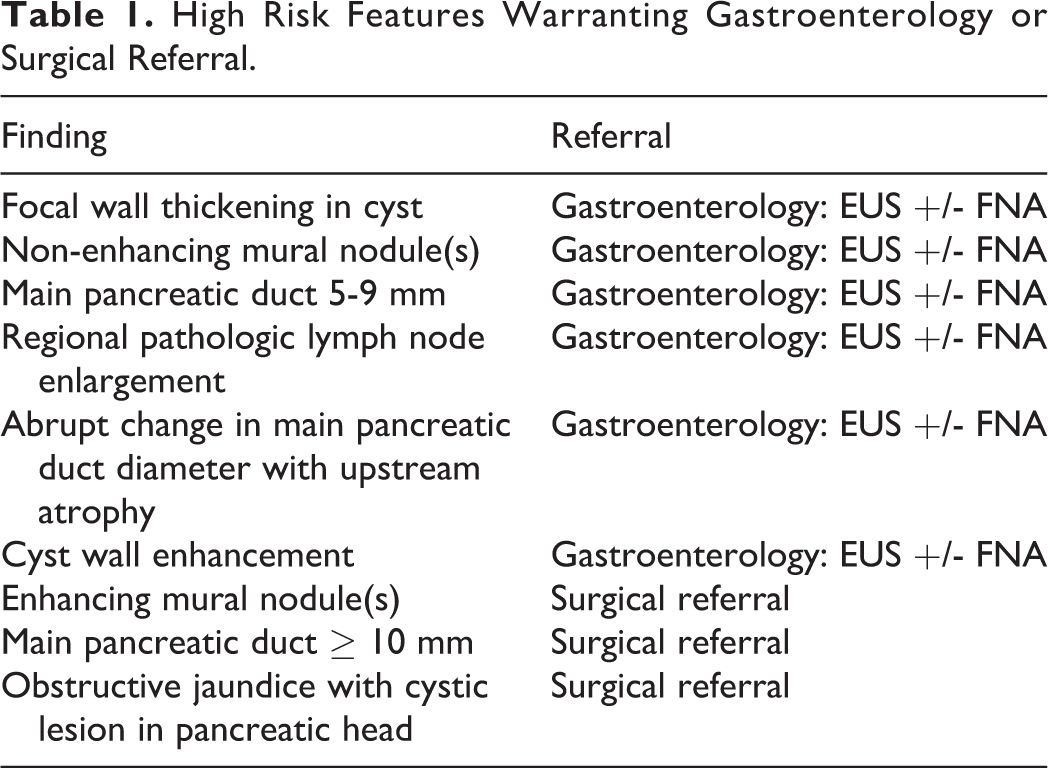
The CAR Incidental Findings Working Group recommendations apply to patients with simple cystic lesions of the pancreas. High risk features are generally consistent between all guidelines and are outlined in Table 1; patients with any high risk finding on imaging or history should be referred to gastroenterology for endoscopic ultrasound (EUS) for further characterization and/or assessment with fine needle aspiration (FNA) or directly to hepatobiliary surgery for consideration of surgical resection. The recommendations also only apply to patients between the ages of 40 and 75 years. Patients less than age 40 with a simple pancreatic cystic lesion should also be referred to gastroenterology for a complete risk assessment and possible EUS. The need for imaging follow-up in patients over the age of 75 years must be predicated on the potential for medical or surgical intervention combined with the patient’s probability of malignancy. Most patients over the age of 75 are unlikely to require follow-up imaging for small, simple cystic lesions with no other clinical or imaging risk factors. Suspected cystic lesions incidentally detected on ultrasound or CT should be assessed with non-emergent contrast-enhanced pancreatic MRI and MRCP to confirm internal simple cystic nature. This study can then either serve as the initial baseline examination or to direct referral to gastroenterology or hepatobiliary surgery depending on the findings. Non-simple cystic lesions (internal septations, nodularity, endoluminal debris, et cetera) are excluded from the follow-up recommendations in this document and warrant gastroenterology referral. For patients with multiple simple cystic pancreatic lesions, the most concerning lesion by size or morphology should be used to determine subsequent management.
High Risk Features Warranting Gastroenterology or Surgical Referral.
Simple Cystic Lesions Less Than or Equal to 5 mm in Maximal Size Do Not Require Follow-Up
In autopsy series up to 49% of patients have pancreatic cysts up to 5 mm in size, with studies in the literature documenting growth rates for such tiny lesions of 0.4 mm per year and a malignancy rate of up to 1% over 10 years. 37 In general, abdominal MR examinations on 1.5 T magnets are performed with T2 slice thickness ranging from 4 to 6 mm. MRCP sequences use thinner slice acquisition, down to 0.9 mm in typical clinical practice. As the growth rate of these lesions is well below the threshold for detection, even over multiple years, and the probability of malignancy in such lesions is markedly low, the Working Group does not recommend follow-up of simple cysts 5 mm or less in size. 38
Patients Over 75 Years Do Not Require Follow-Up
The 2017 ACR white paper was the first major guideline to suggest an upper age limit for pancreatic cystic lesion assessment at 80 years. Prolonged screening results in increased imaging demand with likely minimal clinical impact. While 1 study demonstrated an increased risk of malignancy in patients over the age of 80, 39 the benefit of medical and/or surgical management in this population must be assessed on an individual case basis, and not through inclusion in population-based guidelines. The European guidelines recommend imaging follow-up until patients are “unfit for surgery;” however in a system based predominantly on primary care assessment such as Canada, such statements place an unreasonable burden on primary care providers to fully evaluate the potential for surgical treatment in the elderly population. While a patient with few or no comorbidities, patients with isolated tail lesions (potentially warranting distal pancreatectomy rather than pancreaticoduodenectomy), or other similar clinical situations may warrant follow-up beyond the upper limits recommended depending on local gastroenterology and surgical practice, such patients will be outliers overall relative to the general population. Due to the higher prevalence of comorbid conditions with increasing age, and the high morbidity associated with pancreatic surgery, the Working Group was comfortable with a consensus age cut off at 75 years of age to balance the increasing but still small risk of malignancy in this patient population with the overall awareness of system resources and decreasing gains of imaging follow-up over time.
CAR Incidental Findings Working Group Recommendations
Incidental Simple Pancreatic Cyst > 0.5-2.4 cm, Age 50-75 Years
Simple pancreatic cysts detected on CT, MR, or US commonly represent sequelae of prior inflammation or benign retention cysts,40,41 but imaging cannot differentiate such post-inflammatory change from mucinous neoplasms including intraductal papillary mucinous neoplasm (IPMN) (Figure 3). In the absence of concerning features outlined in Table 1, the CAR Incidental Findings Working Group recommends a follow-up IV contrast enhanced MR pancreas with MRCP within 1 year of discovery. If the lesion remains unchanged at 1 year, follow-up limited pancreatic cyst follow-up MR examinations are recommended every 2 years for a total of 5 years or until the patient reaches age 75, whichever occurs first. If the lesion undergoes growth over 3 mm in longest dimension (in any plane) from the baseline examination at any of the 2 subsequent follow-up examinations or develops suspicious features (Table 1), gastroenterology referral for consideration of EUS +/- FNA is recommended. If a new lesion is discovered on a follow-up examination meeting follow-up criteria (≥5 mm, no suspicious features), the new lesion should be considered an index lesion and follow-up performed accordingly.
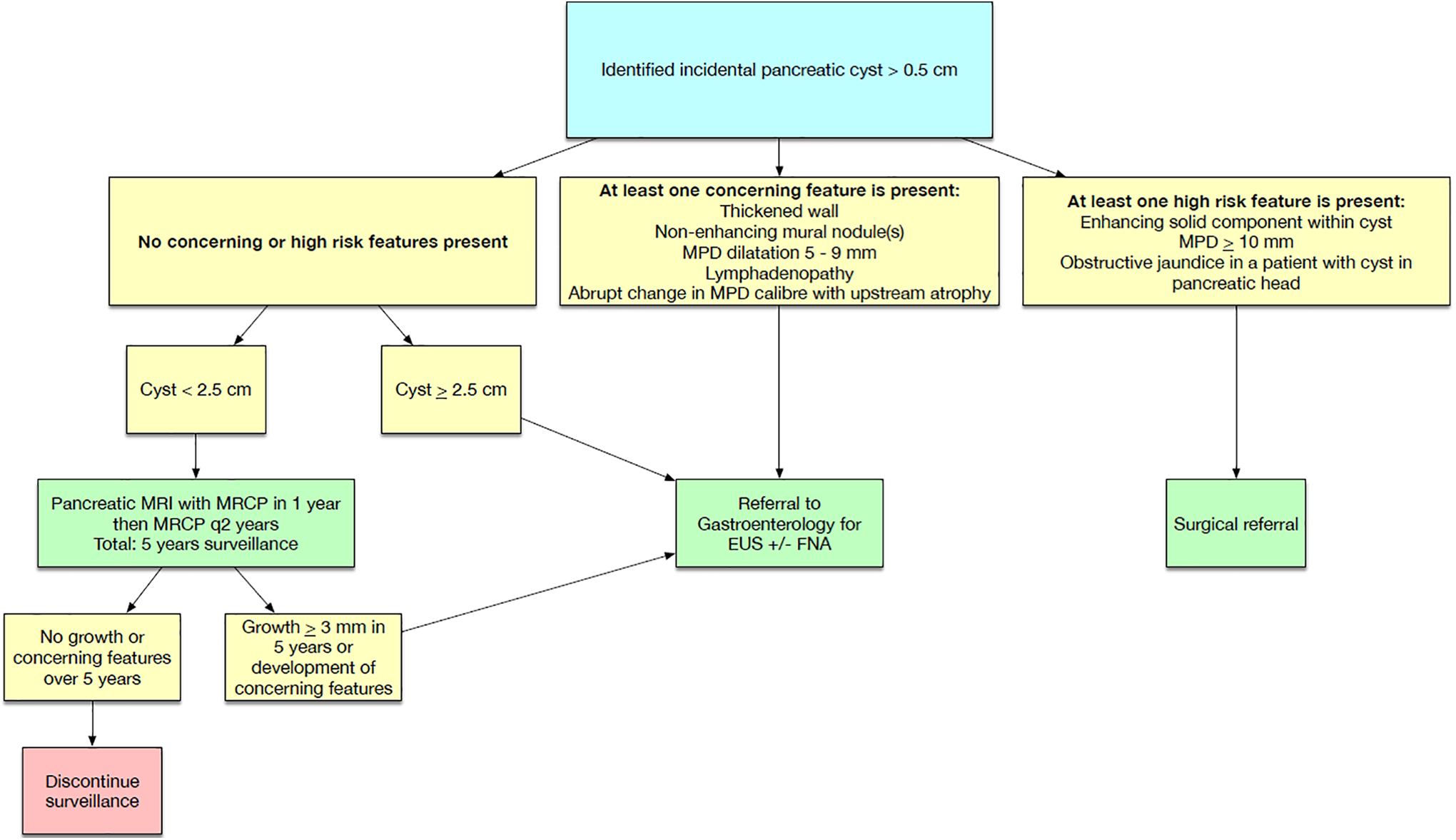
Flowchart for simple pancreatic cyst > 0.5-2.4 cm, age 50-75 years.
Incidental Simple Pancreatic Cyst > 0.5-2.4 cm, Age 40-49 Years
Simple pancreatic cysts detected on CT, MR or US in a younger age group are more likely to be post-inflammatory than in the older patient population, 42 but due to the increased risk of developing a treatable neoplasm over time the CAR Incidental Findings Working Group recommends a follow-up limited pancreatic cyst examination every 2 years for a total of 10 years once the initial IV contrast enhanced pancreas MR with MRCP has been completed (Figure 4). As in the older patient group, any growth over 3 mm in longest dimension (in any plane) or development of suspicious features compared to the baseline MR examination warrants gastroenterology referral for consideration of EUS +/- FNA to exclude a mucinous neoplasm. If a new lesion is discovered on follow-up examination which meets the follow-up criteria (> 5 mm, no suspicious features), the new lesion should be considered an index lesion and follow-up performed accordingly.
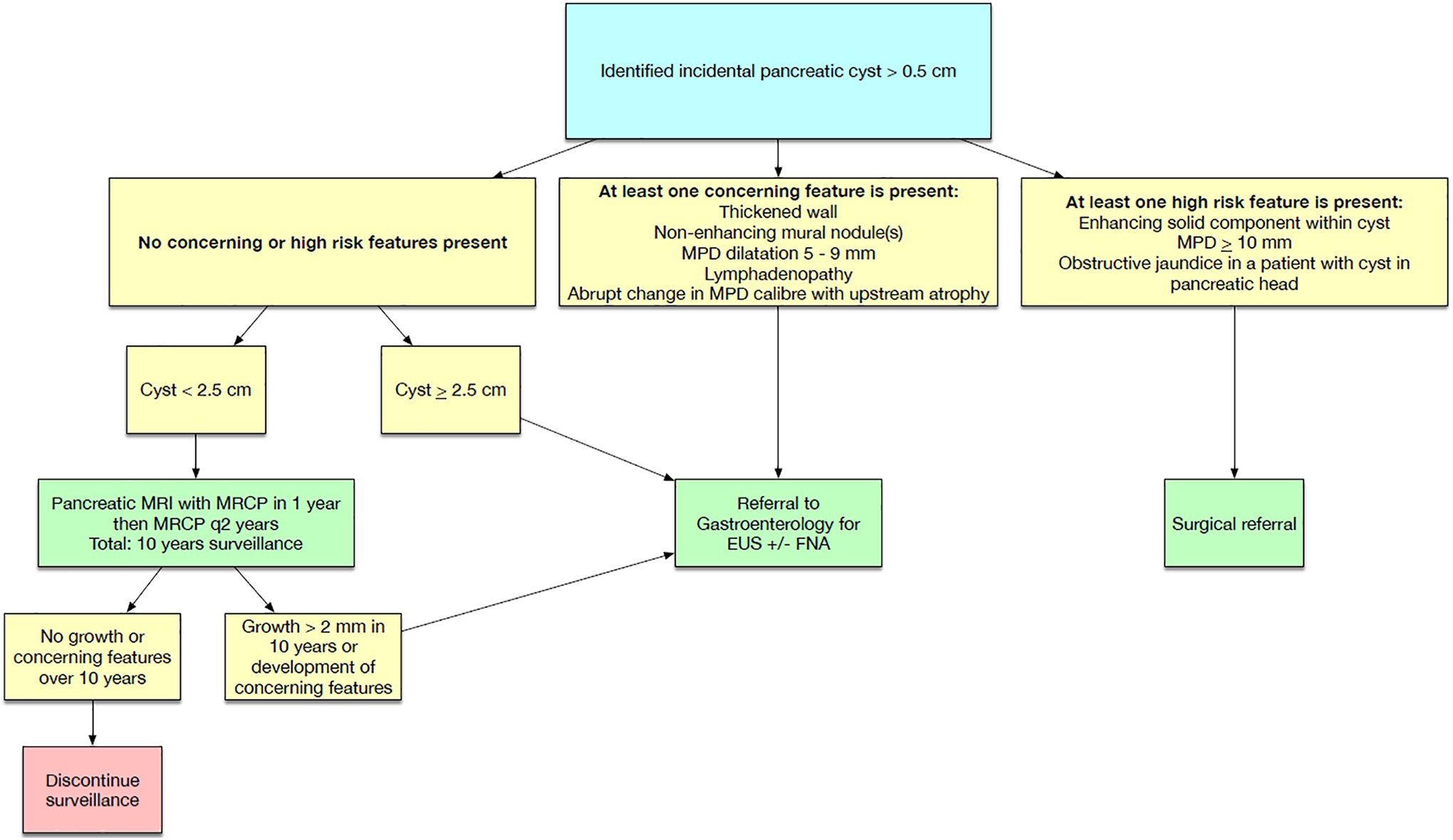
Flowchart for simple pancreatic cyst > 0.5-2.4 cm, age 40-49 years.
Simple Pancreatic Cysts, Age < 40 Years
Although the majority of such lesions are post inflammatory in patients less than 40 years of age, the CAR Incidental Findings Working Group recommends gastroenterology referral for all such patients to assess for common and uncommon causes of pancreatitis, as well as determination of the need for further diagnostic workup of such lesions (EUS, imaging follow-up,
Simple Pancreatic Cysts > 2.4 cm, Any Age
While the most current systematic review performed by the American Gastroenterology association described an increased risk of malignancy with simple cysts ≥ 3 cm (odds ratio [OR], 2.97; 95% confidence interval [CI], 1.82-4.85), 43 the results of the working group stakeholder survey found a significant minority of physicians performing or ordering EUS prefer a lower threshold of 2.5 cm for referral to initiate early discussion and longitudinal management. The CAR Incidental Findings Working Group recommends all patients with simple cystic lesions over 2.4 cm be referred to gastroenterology for consideration of EUS +/- FNA evaluation due to the high risk of mucinous neoplasm or symptomatic sequelae of pancreatitis.38,44
Summary of Recommendations
When pancreatic calcifications are identified, the description should include location (ductal versus parenchymal), size (punctate [< 3 mm] or coarse), number of calcifications, and presence or absence of other features of chronic pancreatitis (atrophy, cysts, or ductal ectasia). An impression of the likelihood of chronic pancreatitis may then be given based on the constellation of findings.
Pancreatic lipomatosis is common, especially as patients age, and does not warrant specific imaging follow-up or screening for malignancy.
Diagnose ductal ectasia if the MPD measures > 3.5 mm or if the upstream duct is more dilated than the downstream duct, which may indicate obstruction due to a stricture or calculus. Depending on the clinical scenario, this often will require further investigation with either multiphasic pancreatic CT or MRI/MRCP.
Simple pancreatic cystic lesions are an increasingly common cause of imaging assessment. The CAR Incidental Findings Working Group presents a series of Canada-specific follow-up recommendations which differ from the ACR, AGA, and ESR guidelines. These include 5-year surveillance, maximum age of 75 years for screening, no follow-up for simple cystic lesions ≤5 mm in longest dimension, and gastroenterology referral for cysts > 2.4 cm or with any worrisome feature(s).
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr. Fung received honoraria from the College of Physicians and Surgeons of Alberta for serving as a CPSA councilor. Dr. Wong is a member of the Advisory Boards for Takeda and for Boston Scientific; and received honoraria from Takeda, Boston Scientific, Ferring, and Allergan. Dr. Kirkpatrick received honoraria from Siemens Healthineers for CME activities; and received honoraria from the Manitoba College of Physicians and Surgeons for serving on program review committee.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
