Abstract
Objectives:
The purpose of this article is to review the embryology of the lower limb arterial anatomy along with common variants and their clinical relevance.
Design:
Embryologic variations of the lower limb arterial system may be explained by i.) persistence of primordial arterial segments, ii.) abnormal fusion, iii.) segmental hypoplasia/absence, or a combination of both. Persistent sciatic artery, corona mortis, and popliteal entrapment syndrome will also be discussed with associated symptoms, and potential complications.
Conclusion:
Knowledge of these variations is essential for surgical and endovascular management as failure to recognize them can result in complications.
Introduction
Lower limb vascular embryology is a challenging topic due to lack of detailed studies and the numerous possible congenital anatomical variations that exist. This review highlights the anatomy and embryology of lower limb arteries and their anatomical variations. This includes variants such as persistent sciatic artery, corona mortis, and variations in branching pattern of the popliteal arteries and popliteal entrapment syndrome. Clinical presentation and key clinical considerations are described. Knowledge of these variations is crucial for accurate diagnostic radiology reports and success of endovascular and open surgical management.
Normal Anatomy
Aorta
The abdominal aorta branches into the right and left common iliac arteries at the L4/L5 lumbar vertebrae,1,2 which in turn each bifurcate into the external and internal iliac arteries anterior to the sacroiliac joints.1,2
Internal Iliac Artery
The internal iliac artery continues inferomedially and splits at the greater sciatic foramen into the anterior and posterior branches.2,3 The anterior division supplies the pelvis, and visceral organs.2,3 It includes the superior vesical, obturator, middle rectal, inferior vesical, internal pudendal, and inferior gluteal arteries.2,3 In females, the inferior vesical artery may be replaced with the vaginal artery.2,3 In men, the anterior division frequently gives rise to the prostatic artery, either directly or via a branch vessel. The posterior division divides into the iliolumbar, lateral sacral, and superior gluteal arteries.2,3
External Iliac Artery
The external iliac artery forms the common femoral, deep circumflex iliac artery, and inferior epigastric arteries. It takes an anterior course along the medial border of the psoas major muscles and exits the pelvic girdle posterior to the inguinal ligament.1,2,4 At a third of the distance between the pubic tubercle to the anterior superior iliac spine the external iliac artery crosses the inguinal ligament and becomes the common femoral artery. The inferior epigastric artery arises from the medial side of the distal external iliac artery, while the deep circumflex iliac artery originates from the lateral side.1,2,4 The inferior epigastric artery supplies the rectus abdominus muscles, while the deep circumflex iliac artery supplies the iliacus and internal oblique muscles, and iliac fossa.1,2,4 Variants of the internal iliac and external iliac arteries are beyond the scope of this review.
Common Femoral Artery
The common femoral artery (CFA) begins distal to the inguinal ligament as a continuation of the external iliac artery. On angiography, the branching of the inferior epigastric artery is sometimes used as a marker for the origin of the CFA instead of the inguinal ligament as the inguinal ligament is not seen angiographically. The CFA courses anterior to the femoral head and divides into the deep femoral artery (profunda femoris) and superficial femoral artery (SFA) (Figure 1). Small branching vessels of the CFA include the superficial epigastric, superficial circumflex iliac artery and external pudendal arteries.
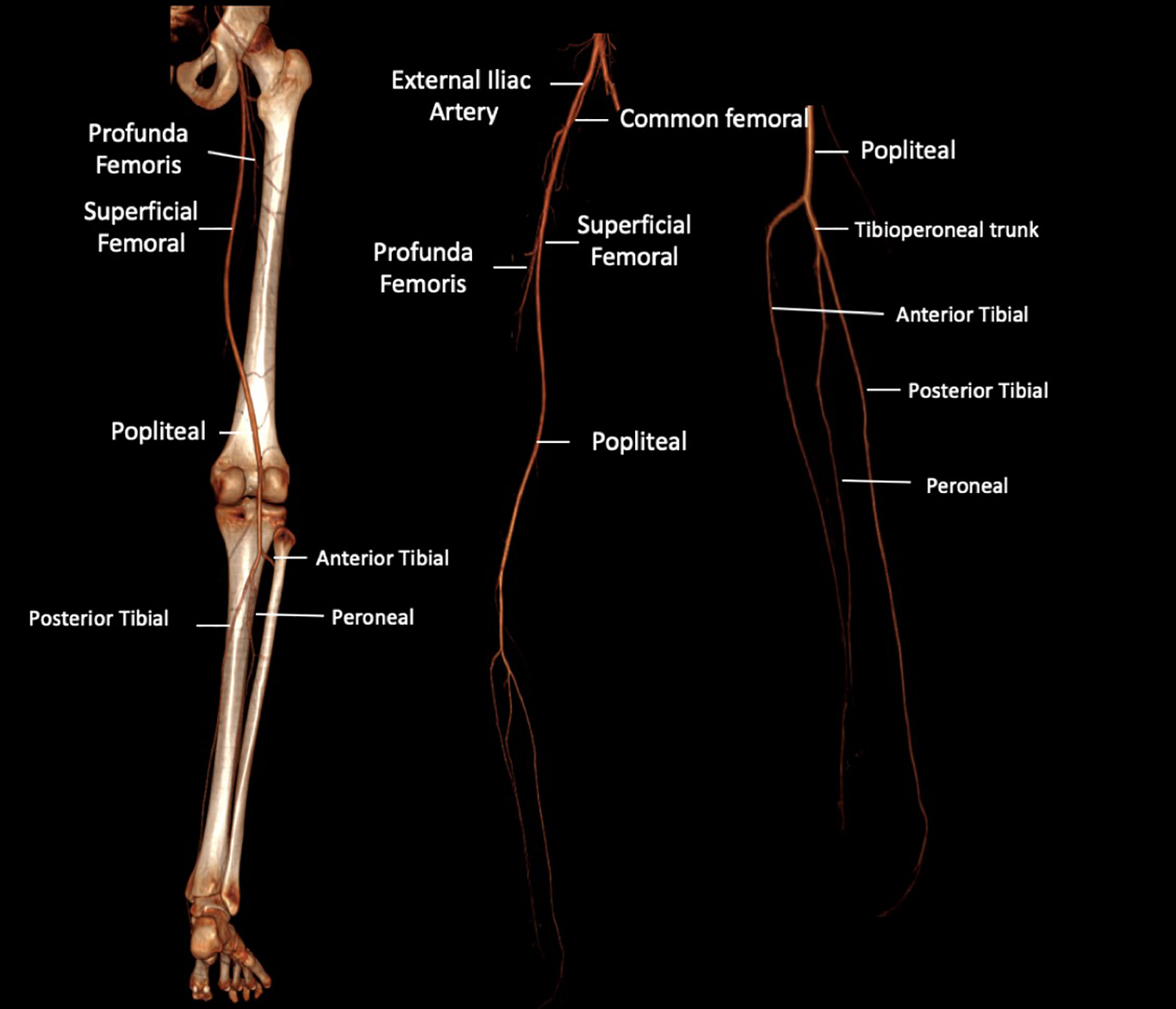
Normal branching of the lower limb arteries.
The SFA continues distally through the adductor canal and becomes the popliteal artery at the adductor hiatus. The SFA supplies blood to the lower leg while the deep femoral artery supplies blood to the hip and thigh. The descending genicular artery is a small branching vessel that arises from the SFA prior to entering the adductor canal. It supplies blood to the knee joint.1-4 Other smaller perforating vessels include the superior medial genicular artery, and superior lateral genicular arteries.
The deep femoral artery branches into medial femoral circumflex, lateral femoral circumflex, and terminal perforating vessels. The medial circumflex supplies the posterior femur, femoral head/neck, abductor muscles (longus, magnus, and brevis), gracilis, and pectineus muscles. The lateral circumflex supplies knee extensor muscles (vastus lateralis, vastus medialis, vastus intermedius, rectus femoris). Terminal perforators (usually 3) of the deep femoral artery supply the hamstring muscles (semitendinosus, semimembranosus, short and long head of biceps femoris) and the femur.2,3
Popliteal Artery
The popliteal artery travels within the popliteal fossa and bifurcates into the anterior tibial artery and tibioperoneal trunk. 5 The tibioperoneal trunk then divides into the posterior tibial artery and peroneal (fibular) artery. The popliteal artery has multiple smaller branches such as genicular arteries (inferior medial and inferior lateral) and sural arteries.2,5 Genicular arteries supply blood to the knee.2,5 Sural arteries supply numerous muscles including the soleus, gastrocnemius, plantaris, and distal hamstrings.2,5
Anterior Tibial Artery
The anterior tibial artery supplies the anterior compartment of the lower limb.2,6 It courses anterior to the popliteus muscle passing through the interosseus membrane in between the tibia and fibula.2,6,7 After it is through the interosseus membrane, it travels between the tibialis anterior and extensor digitorum longus muscles.2,6,7 At the anterior ankle, it becomes the dorsalis pedis artery.2,6,7 It subsequently branches into the medial malleolar, lateral malleolar, medial tarsal, lateral tarsal, and arcuate arteries.
Posterior Tibial Artery
The posterior tibial artery travels within the deep posterior compartment of the leg along the border with the superficial compartment.2,7,8 At the level of the ankle, it travels posterior to the medial malleolus and into the tarsal tunnel.2,8 It gives off the medial plantar, lateral plantar, and medial calcaneal arteries. 7
Peroneal Artery
The peroneal artery arises from the tibioperoneal trunk and supplies the muscles of the lateral compartment of the lower leg (peroneus brevis, and peroneus longus).2,9 It courses in between tibia and fibula along the interosseus membrane.2,9
Embryology
Vascular development
The vascular tree is developed through a dynamic process with constant remodeling through a balance of two distinct but interlinked phenomena; vasculogenesis and angiogenesis.10-14
Vasculogenesis is the process in which vascular tubes are created de novo. Mesenchymal endothelial cell precursors (angioblasts and hemangioblasts) form condensations which develop sinusoidal tubes with blood-filled lumens. 10 These merge and expand to form vessels. The early vascular tree is developed largely by vasculogenesis.
Angiogenesis is the process of vascular growth through formation of new vessels from the pre-existing primitive vascular tree by sprouting, splitting and/or intercalated growth. Organ-specific differentiation of the vascular tree includes pruning, fusion or regression of vessels, associated with mechanisms such as disassembly of junctions, cell retraction, endothelial cell migration, and (very rarely) apoptosis. Through an interaction between these processes the final adult vascular network is formed. The process of vasculogenesis, angiogenesis and vascular maturation occurs in a tightly regulated and predictable pattern, but changes in this process can lead to clinically significant variations in anatomy.10-13
Development of Lower Limb Arteries
The branches of the lower abdominal aorta begin to form at the 6 mm stage embryo (35-37 days gestational age).15-17 As the segmental (intersomitic) arteries of the lower aorta begin to coalesce to form the main extraembryonic vessel, the umbilical artery is formed. It continues in a transverse direction and when it reaches the dorsal aspect of the Wolffian duct, the axial artery branches off. 15 The axial artery is the first artery to penetrate the lower limb (Figure 2), and is located in the center of the developing limb bud.15,16,18 It is also known as A. ischiadica/sciatic artery, since it mainly follows the course of the sciatic nerve. 18
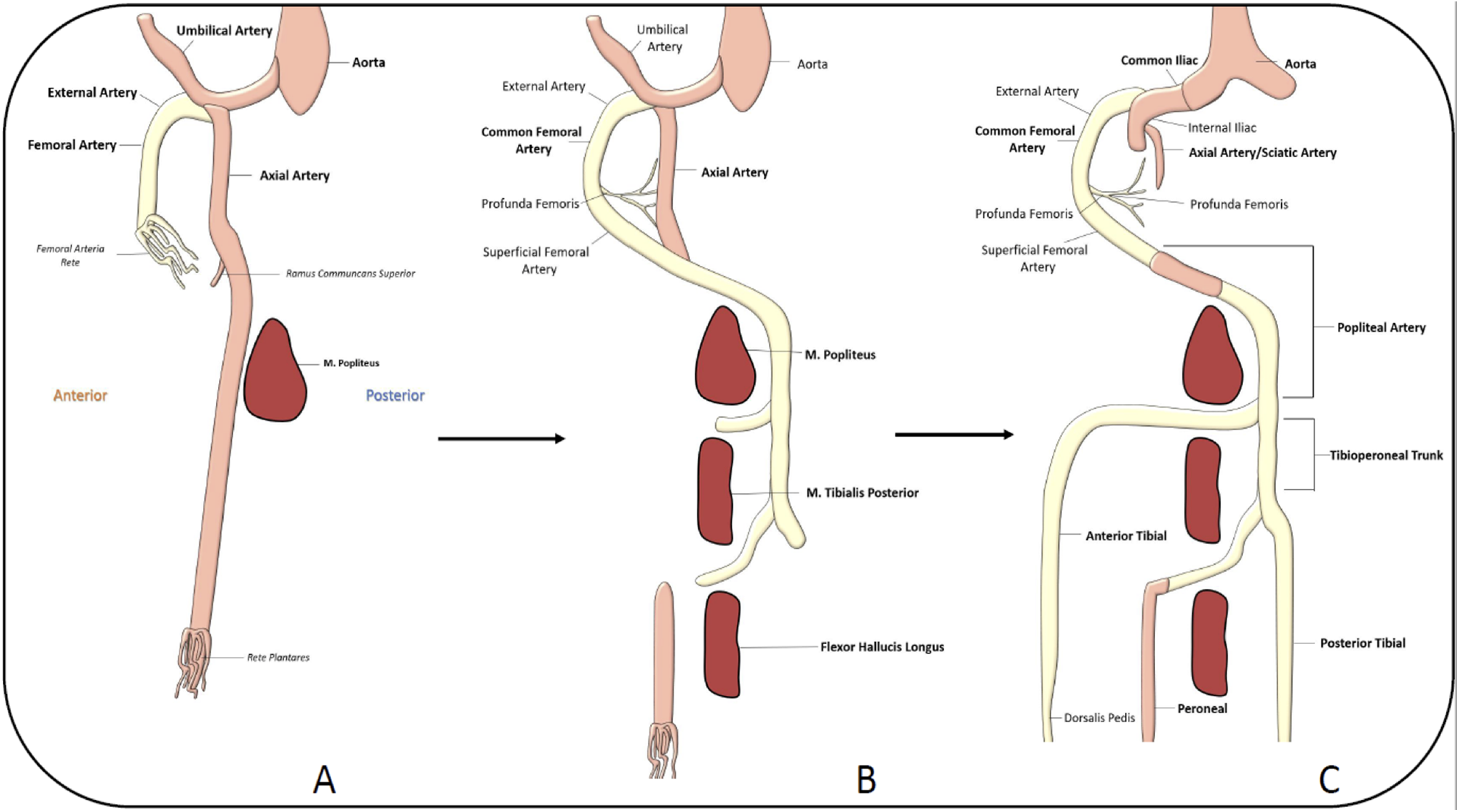
Embryological development of lower limb arterial anatomy. A, The axial artery is the first artery to penetrate the lower limb. The femoral artery begins to penetrate later during embryological development. B, It joins the axial artery at the level of the ramus communicant superior and becomes the major source of blood flow to the lower limb. Concurrently, the middle segment of the axial artery regresses. C, The remnant of the axial artery is the sciatic artery. The anterior tibial and posterior tibial arteries form through vasculogenesis, while the distal peroneal artery is a remnant of the original axial artery.
However, immediately thereafter, a second leg artery branches off the umbilical artery more proximally and gives rise to external iliac and femoral arteries. Key points to highlight include: The axial (sciatic) artery is the main artery of the early leg bud. As the limb grows the axial artery passes into the lower limb along medial thigh and then posteriorly to the lateral leg. It extends to the sole where it forms an arterial plantar plexus. Later in development through remodeling it regresses. The proximal (sciatic) part later receives a more specialized function as it forms the accompanying artery of the sciatic nerve.15,16 External iliac/femoral artery penetrate into the lower limb All distal vessels arise by complex remodeling of the axial artery and the vascular plexus of the distal leg bud.15,17
The popliteal portion of the axial artery lies at the ventral side of the developing popliteal muscle, while in the adult it is situated dorsal to the popliteus muscle. Therefore, the name A. poplitea profunda has been suggested for the embryonic section of the axial artery. 15 The final popliteal artery (dorsal to the popliteus muscle) is derived from separately developing vascular beds from the superficial femoral artery.
In 12 mm embryos (39-42 day gestational age), the skeletal mesenchymal condensations are apparent. Below the knee, the position of the embryological axial artery is between the tibia and fibula, which prompted the name interosseous artery. It runs at the ventral aspect of the limb and is later found between interosseous membrane and tibialis posterior muscle. More proximally, the femoral artery has formed a lateral and a medial extension. The lateral joins the axial (sciatic) artery and takes over the perfusion of the distal leg, while the proximal/sciatic part of the axial artery becomes reduced to the accompanying artery of the sciatic nerve. The medial extension of the femoral artery represents the anlage of the A. descendens genus and the R. saphenus. Together, this shows that while the main nerve supply of the leg is dorsal, the sciatic nerve, the main blood supply is shifted to the ventral side, the femoral artery. 15
At the 14 mm embryo stage (42-45 day gestational age), the dense vascular supply of the knee starts to develop. Thereby, complex vascular remodeling takes place, which shifts the “deep” popliteal segment of the axial artery into the final position dorsal to the popliteus muscle.9-11 Distal to the knee, the interosseous segment of the axial artery develops a perforating artery into the extensor compartment of the lower leg (ramus perforans cruris), the anlage of the anterior tibial artery. Simultaneously to the regression of the interosseous artery, new vessels arise by angiogenic mechanisms, and form the anlagen of the posterior tibial and peroneal arteries.
Molecular Control of Vessel Formation
The molecular control of vascular development is not well understood. There is a complex interplay between growth factors and inhibitors.10-12,19 Vascular remodeling requires sprouting of new vessels, intussusceptive growth, regulation by hemodynamic factors, growth and inhibitory factors, and tissue environment. Principal growth factor that regulates vessel formation is vascular endothelial growth factor (VEGF). It is an important growth factor that promotes endothelial growth, differentiation, migrations, and suppresses apoptosis. There are numerous other signaling pathways which modulate VEGF-A activity including angiopoietin/Tie2, Notch, hedgehog protein etc.11,19 Variations in these processes through failure of regulation, genetic predisposition, and environmental factors (such as hypoxemia) can lead to variations in vessel growth and patterning. This includes vessel duplication, hypoplasia, or aplasia.10,19
Variations of Vessels
There are numerous anatomical variations of the lower limb arteries, and can often impact the success of surgical reconstructions, endovascular intervention, embolectomy, or diagnosis of arterial injury. 20 The vast majority of these variations can be explained by a combination of persistent primitive arterial segments, abnormal fusions, and segmental hypo or aplasia. 20 Anatomical variations of the femoral artery are rare and include absent profunda femoris artery, persistent sciatic artery and duplication of superficial femoral artery.
Absent Profunda Femoris
An absent profunda femoris artery is a rare variation which occurs in approx. 0.4% of the population. 21 Literature reports 65-80% of these are bilateral.22,23 In these cases, the arteries of the thigh are usually supplied either directly from the common and superficial femoral artery or persistent sciatic artery (if present). Clinically, patients are normally asymptomatic, and it is discovered incidentally.
Duplicated Superficial Femoral Artery
Only 5 cases of Duplicated superficial femoral artery have been reported in literature.24,25 The duplicated vessel generally begins near the abductor hiatus and converges at the level of the popliteal artery.24,25 Embryologically, as the femoral artery grows through angiogenesis and vasculogenesis it comprises a plexus of vascular channels called the femoral artery rete. 24 These channels combine with the ramus communicans superior, which is a branch of the axial artery to form the profunda femoris. 24 Duplication of the SFA results from non-union of the femoral artery rete plexus distal to the ramus commiunicans superior. This results in formation of 2 vessels which subsequently combine at the primitive axial artery to form the popliteal artery. 24
Persistent Sciatic Artery
The persistent sciatic artery (PSA) occurs when the embryological sciatic artery fails to regress (Figures 2 and 3).26-28 This occurs when there is hypoplasia or incomplete formation of the femoral artery. The reasons for insufficient femoral artery development are not well understood, but likely based on failure of complex molecular interactions. 27 The PSA occurs with an estimated incidence of 0.03-0.06%. 26 It begins as a branch of the internal iliac artery and passes through the greater sciatic foramen. It follows the path of the sciatic nerve, and communicates directly to the popliteal artery. Bower et al. suggested a classification that describes the persistent sciatic artery as complete or incomplete. 28 The complete type occurs when the main arterial supply to the lower limb is through the PSA, and the SFA is typically hypoplastic. The incomplete type occurs when the main arterial supply to the lower limb is through the SFA and the sciatic artery is hypoplastic and/or terminates in the thigh. 28
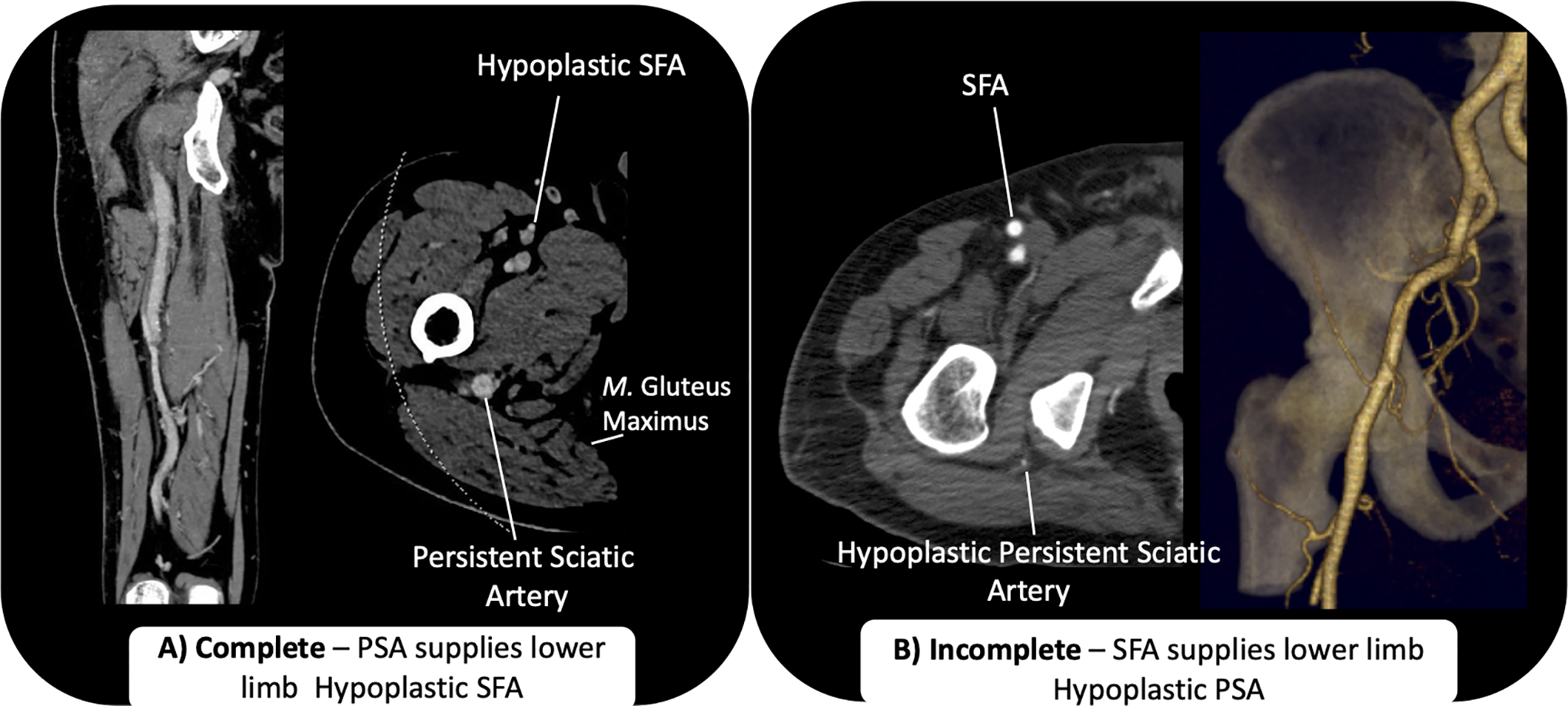
Persistent Sciatic Artery is a branch of the internal iliac artery. It passes through the greater sciatic foramen. It travels along the sciatic nerve deep to the gluteus maximus muscle. PSA can be complete or incomplete. A, In complete PSA, the PSA is the dominant artery that supplies the lower limb and communicates with the popliteal artery. The femoral artery is hypoplastic. B, In incomplete PSA, the PSA is hypoplastic, while the femoral artery supplies the lower limb.
Clinically, most PSA patients are asymptomatic. They may present with an absent femoral pulse if the patient has a hypoplastic or absent femoral artery. If symptomatic, patients generally present with non-specific gluteal pain or a buttock mass. They may also present with aneurysm formation, lower limb ischemia, or peripheral neuropathy with compression of the sciatic nerve. A pathognomonic clinical exam finding of PSA is the “Cowie Sign,” in which a normal popliteal and pedal pulse can be palpated with an absent/diminished femoral pulse. 29 Aneurysms occur in 48% of PSA patients, generally forming near the buttock region, between the piriformis muscle and posterior aspect of the greater trochanter of the femur (Figure 4).26-28 This may be palpated as a pulsatile mass near the gluteal region. The exact cause of the aneurysms is unknown; one hypothesis is persistent sciatic artery is overstretched in between the piriformis muscle and greater trochanter.26-28 Over time this results in vessel wall weakness and aneurysm formation. Another hypothesis is that due to the anatomical location, the PSA endures repetitive trauma as it is constantly pressed against the hip, sacrospinal ligament, and piriformis muscle during hip flexion. This results in vessel wall weakness by destruction of the media/adventitia and aneurysm formation.26,27 A third theory is that congenital alterations lead to reduced elasticity in the arterial wall and lead to aneurysm formation. 29 However, aberrant molecular signaling, e.g. induced by shear stress (Kang et al., 2019), has not been investigated yet. 30
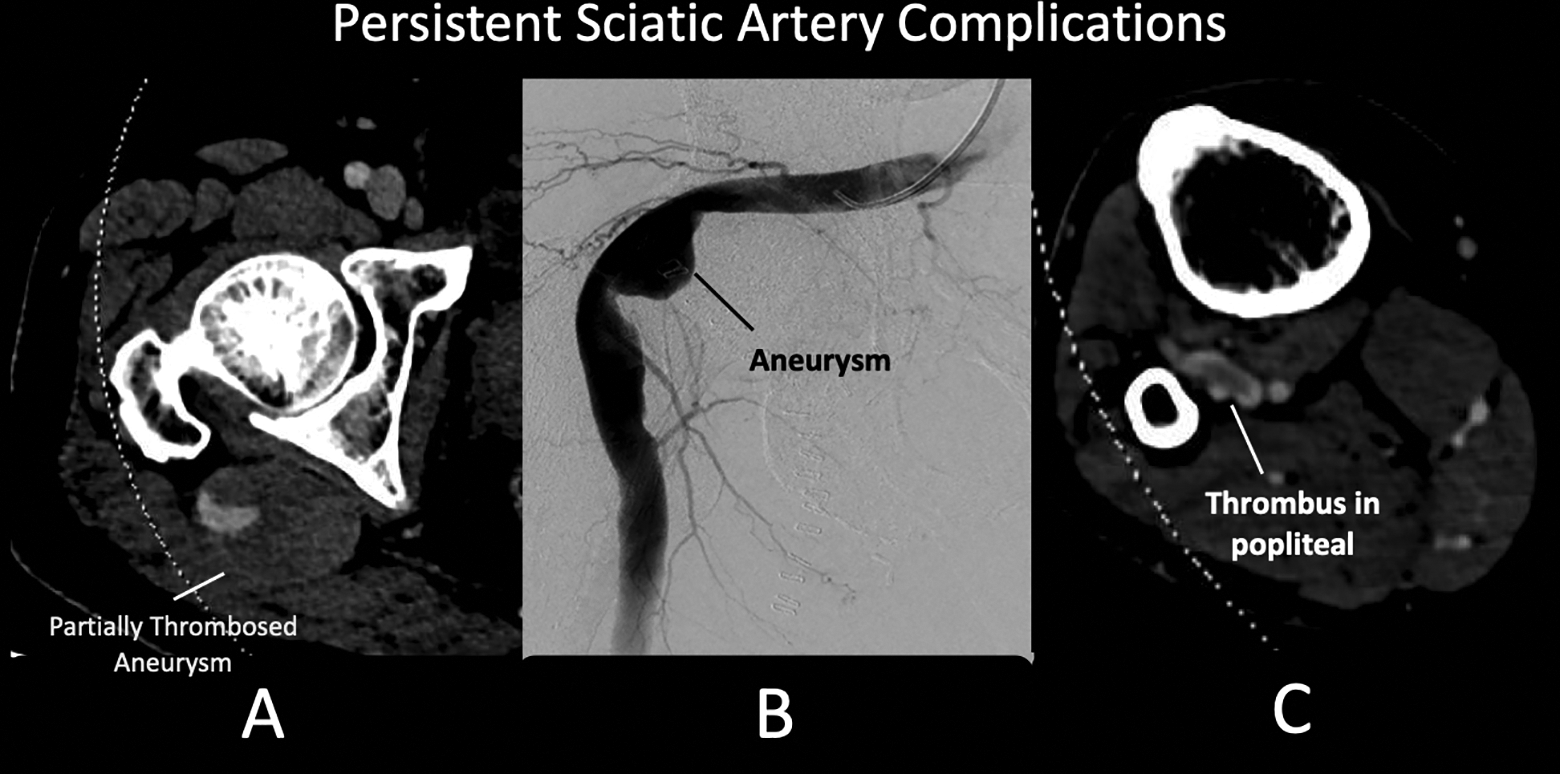
Complications of persistent sciatic artery. A, CT angiogram that demonstrates a partially thrombosed aneurysm along the right gluteal region. B, The aneurysm can be seen on conventional angiography. C, Thrombus of the popliteal artery extending into the trifurcation, this likely embolized from the aneurysm.
Treatment is generally reserved for symptomatic patients and dependent on the complication. Generally, PSAs that are asymptomatic are not treated. Asymptomatic PSAs may be followed up with physical exam and duplex ultrasound. 29 In patients with aneurysms, the PSA is occluded with either a surgical or endovascular approach. Surgical options include ligation of the PSA, or excision of the aneurysm with an interposition graft. If the SFA is hypoplastic, an additional bypass may be required to maintain blood flow in the distal limb. Endovascular options include coiling, Amplatzer plug placement or insertion of a covered stent graft.26-29,31
Corona Mortis
Corona mortis is a vascular connection between the internal and external iliac vessels (Figure 5). It is generally a direct connection between the obturator artery and either the external iliac or inferior epigastric artery.32,33 It is Latin for “Crown of Death,” and is clinically relevant because transection of the corona mortis can cause the vessel to retract inferiorly or through the obturator foramen, severely compromising vascular control for hemostasis. Pelvic osteotomies using the medial approach, as well as acetabular fracture repair using the ilioinguinal approach, pose the highest risk for severing a corona mortis vessel. Additionally, the corona mortis usually crosses the superior pubic rami. Fractures of the superior pubic rami can lead to transection of the corona mortis and may result in life threatening bleeding (Figure 5).32,33 The embryological basis is not well understood, variations in branching patterns can be from abnormal angiogenesis. One theory suggests this can occur from hypoxia, low oxygen levels allow for stabilization of hypoxia inducible factor-1α, which subsequently upregulates vascular endothelial growth factor (VEGF) and nitric oxide synthase expression. The nitric oxide causes dilation of vessels which causes extravasation of plasma proteins. Ultimately, this leads to protease and matrix metalloproteinase expression. 33 These protein complexes lead to abnormal vessel formation. 33
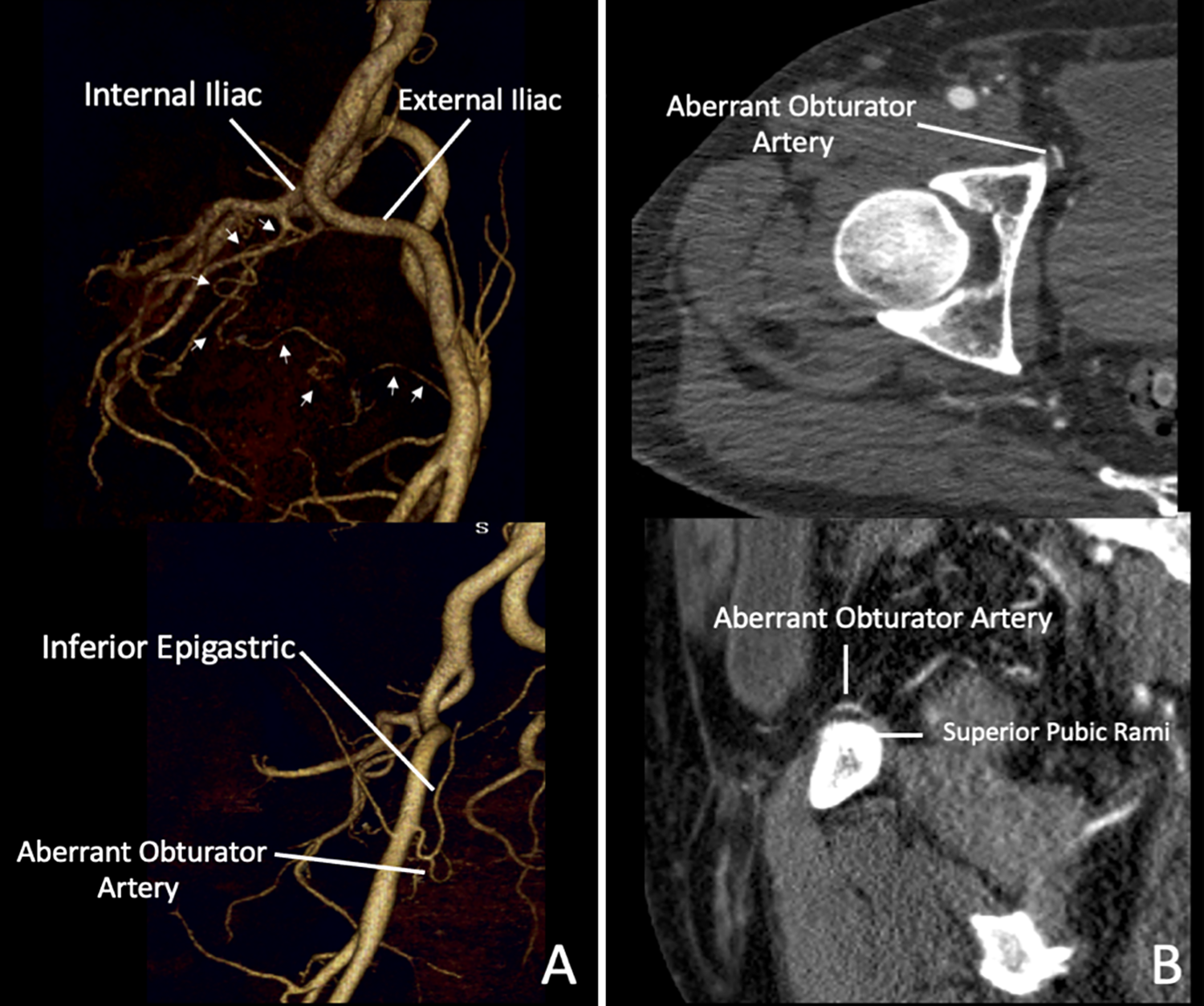
Corona Mortis—3D reconstructions of a CT angiogram demonstrate an (A) aberrant obturator which directly anastomoses the internal and external iliac artery (arrows). B, It is in close proximity of the aberrant vessel to the superior pubic rami. Fractures of the superior pubic rami or pelvic osteotomies may transect the corona mortis. If transected, it can retract into the pelvis resulting in life threatening bleeding.
Iliac Artery Aneurysms
Risk factors including genetic predisposition, smoking, advanced age, and hypertension contribute to the pathogenesis of aneurysms. However, the onset and distribution of aneurysms may depend on fetal factors. For example, isolated iliac artery aneurysms are rare compared to abdominal aortic aneurysms and combined aortoiliac artery aneurysms. Case series in the literature suggests that less than 10% of iliac artery aneurysms occur in the internal iliac artery, while less than 1% occur in the external iliac artery, and 89% occur in the common iliac arteries. 34 One explanation of the lower rate of aneurysm in the external iliac artery is that during fetal development the umbilical artery and internal iliac artery are the main continuation of the aorta. They have a large caliber whereas the external iliac artery is a branch of the umbilical artery. The higher mechanical load associated with placental circulation may cause early structural changes by disruption of elastogenesis and deposition of microcalcification.35,36 These minor disruptions may result in aneurysm formation later in life. The external iliac artery does not contribute to placental circulation and is not subject to dramatic flow and calibre changes seen in the aorta, common iliac and internal iliac arteries. 35
Variations in Popliteal Artery
Variations of the popliteal artery branching patterns are common and have been reported in 7.2-12% of studied cases.20,37-39 Kim et al. proposed a classification system which divides the branching patterns into 3 categories. Type I branching pattern was defined when the division of the popliteal artery occurred below the level of the tibial plateau in the region of the popliteus muscle (Figure 6). Type II branching occurred when the bifurcation was high, above the level of the tibial plateau (Figures 6 and 7). Type III branching involved a hypoplasia or aplasia of one of the vessels (Figures 6 and 8).37-39
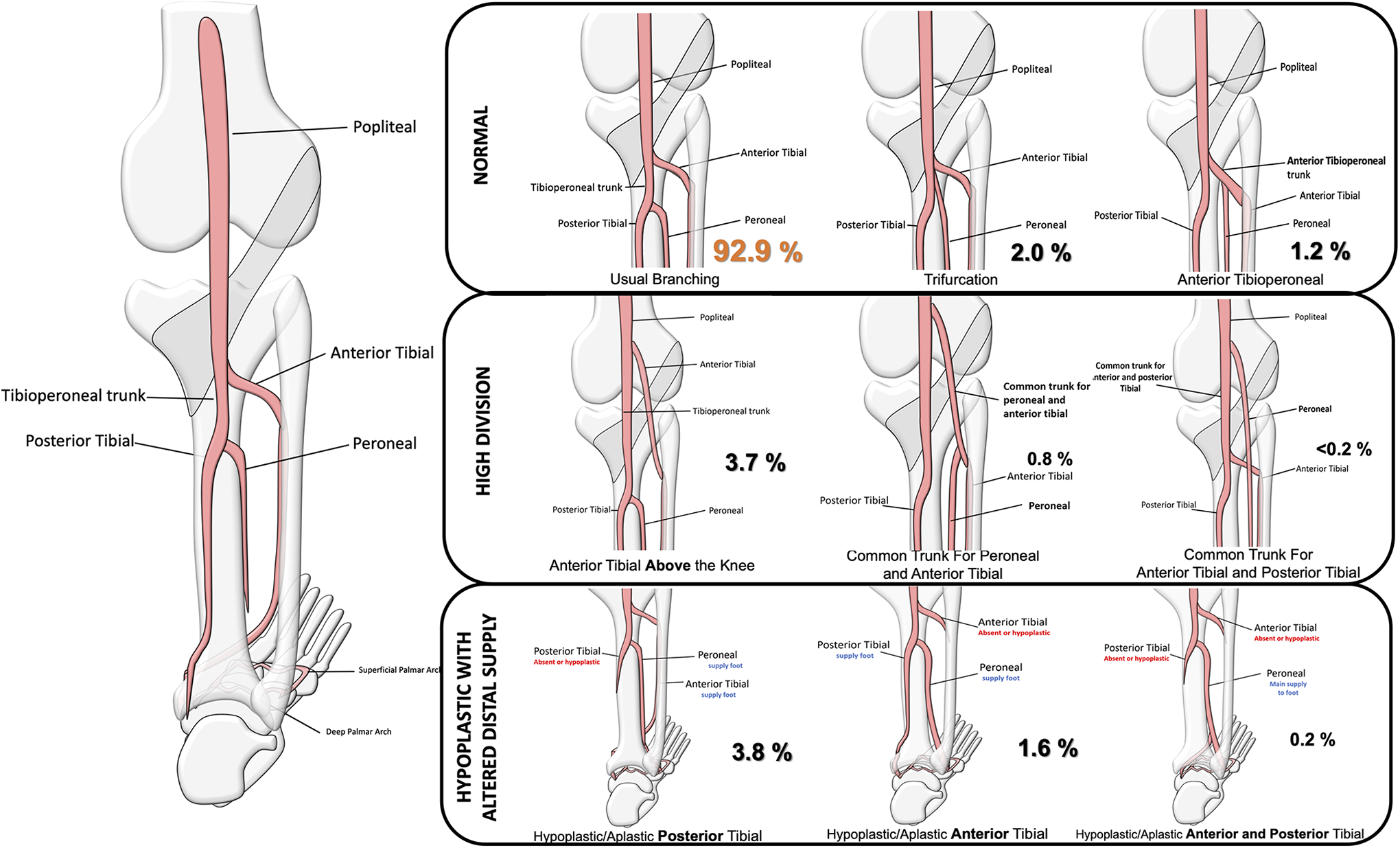
Variation of branching pattern of the popliteal artery. Type I (Normal) branching pattern occurs when the division of the popliteal artery occurred below the level of the tibial plateau in the region of the popliteus muscle. Type II (High Division) branching occurs when the bifurcation was high, above the level of the tibial plateau. Type III (Hypoplastic with altered distal supply) branching involves a hypoplasia or aplasia of one of the vessels.
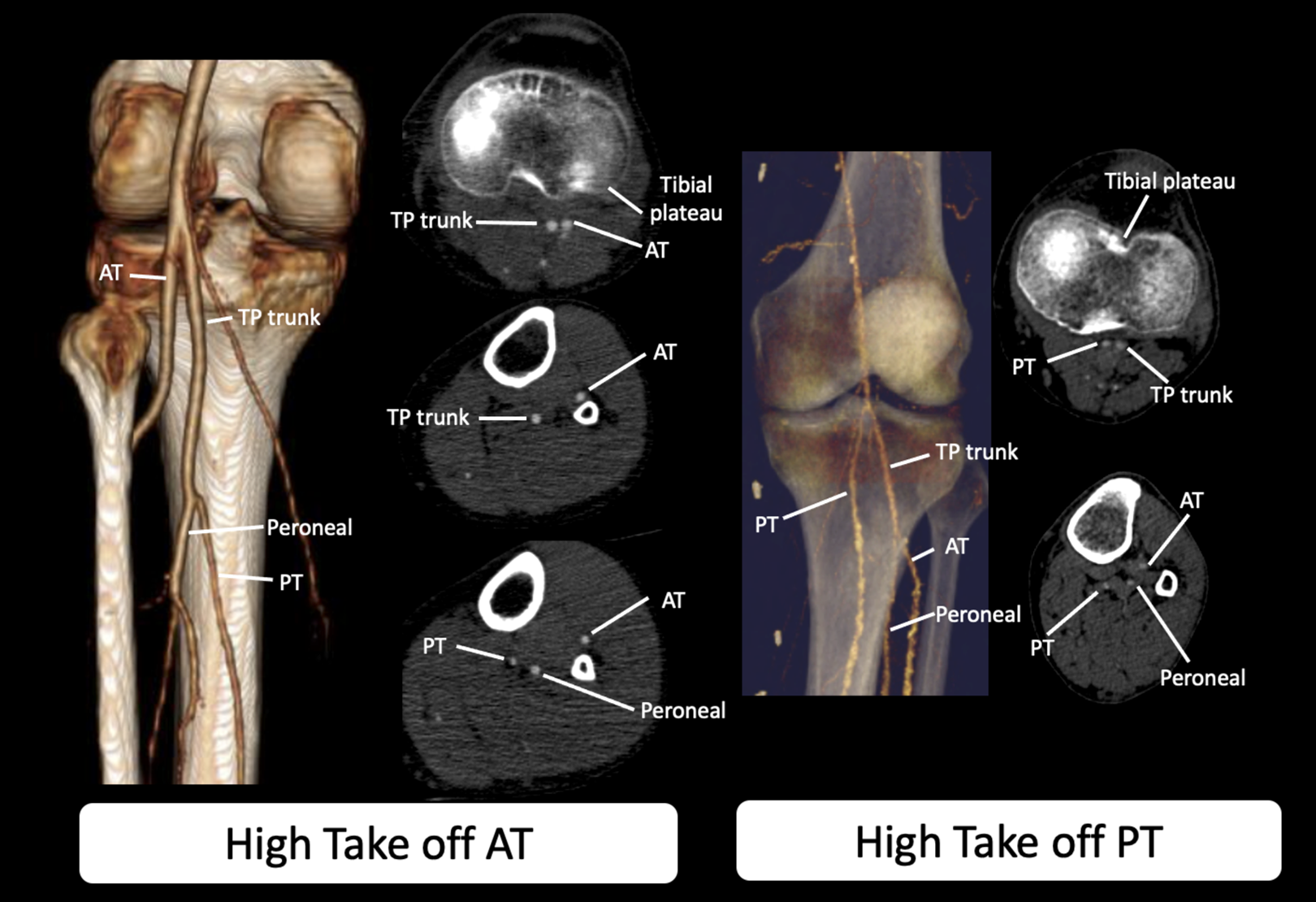
CT angiogram demonstrating Type II branching of the popliteal artery in 2 different patients. The panel on the left demonstrates high take off of the anterior tibial artery. The panel on the right demonstrates a high take off of the posterior tibial artery. In this case the anterior tibial and peroneal share a common trunk. Note the close proximity of the AT (panel on left) and PT (panel on right) to the superior cortex of the tibia. There is risk of injury to these vessels during knee arthroscopy or high tibial osteotomies.
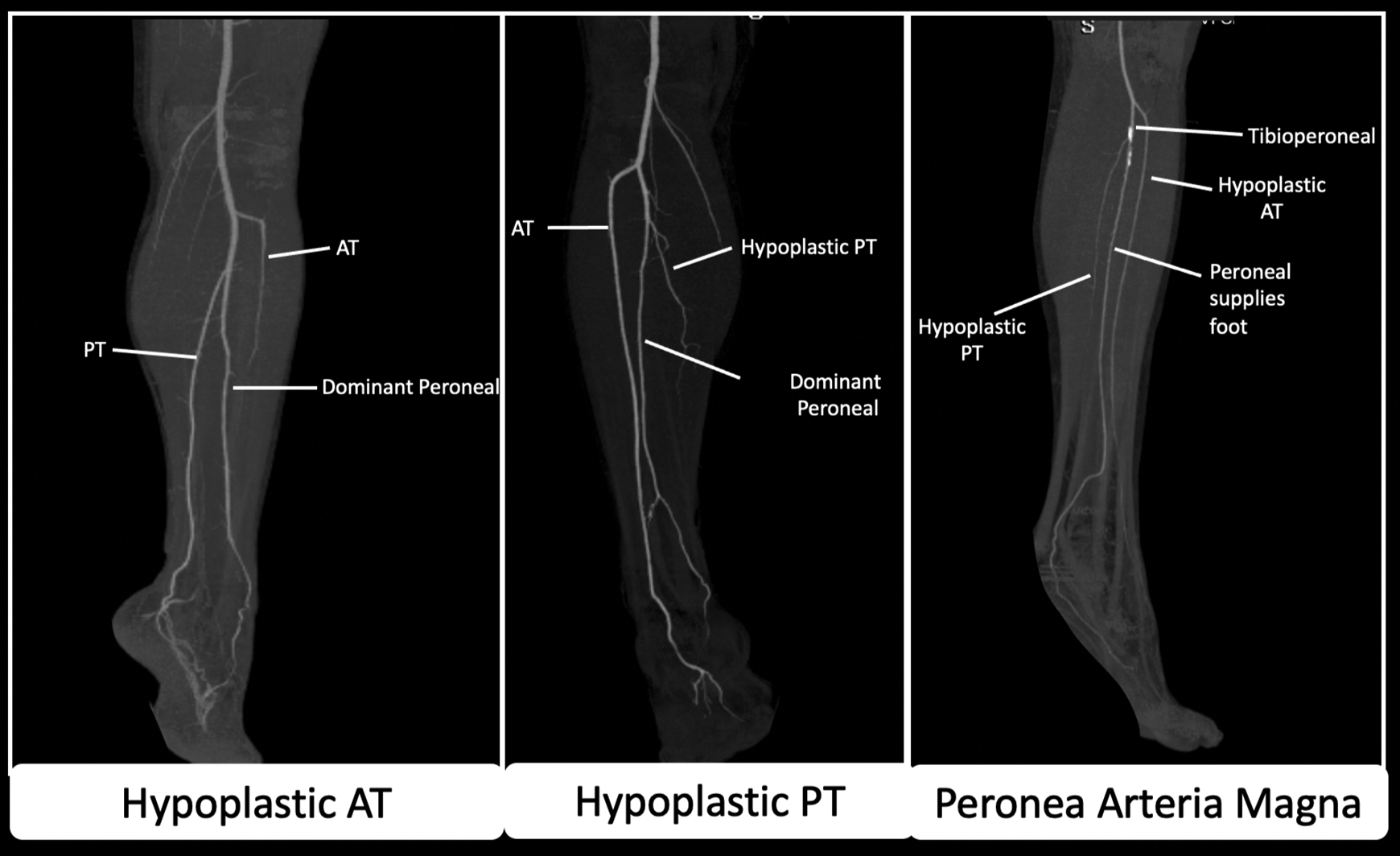
MIP reconstructions of CT angiograms demonstrate Type III branching pattern of the popliteal artery. This includes hypoplastic anterior tibial with dominant peroneal (left), hypoplastic posterior tibial with dominant peroneal (middle), and peronea arteria magna (right). Peronea Arteria Magna is both hypoplastic anterior tibial and posterior tibial arteries with a dominant peroneal artery solely supplying the foot.
Type III A-C anomalies are clinically important as the peroneal artery has an important role in supplying blood to the foot. For instance, femoro-distal popliteal and tibial arterial reconstructions which are anastomosed to a hypoplastic artery may not allow for adequate perfusion of the distal extremity. The Type III C anomaly also known as Peronea Arteria Magna is particularly important (Figures 6 and 8). In this anomaly, the peroneal artery solely supplies the pedal circulation. 40 If these variations are not recognized prior to peroneal artery harvest for surgical procedures such as the fibular free flap, then they may lead to limb ischemia. 38 Type II (High Division) of the popliteal artery can result in a high origin of the anterior tibial or posterior tibial arteries. In this variant the tibial artery divides above the level of the popliteus muscle and courses anterior to the popliteus muscle with direct contact with the superior cortex of the tibia. 41 Given the close proximity to the superior tibial cortex, it is at risk of injury during knee arthroscopy, or high tibial osteotomies in which the posterior tibial cortex is cut with either an oscillating saw, osteotome, or drill. This can lead to complications such as arterial transections, pseudoaneurysms, thrombosis and acute limb ischemia. 41
Popliteal Entrapment Syndrome
Popliteal entrapment can lead to vascular compromise (stenosis/occlusion) of the popliteal artery.20,42-45 It is due to an abnormal relationship between the popliteal artery and the gastrocnemius muscles that is caused by abnormal popliteal fossa embryological development.42,46
The popliteal artery normally passes in between the 2 heads of the gastrocnemius muscles. An aberrant course of the popliteal artery or gastrocnemius muscles, hypertrophy of the gastrocnemius muscles, or development of a fibrous band can all result in tissue that crosses the artery and results in arterial compression.
Approximately 60% of patients are less than 30 years of age with a 15:1 male predilection. Patients generally present with calf claudication but can also present with critical limb ischemia. On physical exam, pulses can disappear with either dorsiflexion or plantar flexion. The classification of popliteal entrapment syndrome can be divided into 6 different types, as follows (Figures 9–11):
Popliteal artery is medial to a
Popliteal artery is medial to an
Additional slip of gastrocnemius muscle which originates from the lateral or medial femoral condyles compresses the popliteal artery
Popliteal artery entrapped by the Popliteus muscle
External compression of popliteal artery
Functional or acquired, and results from hypertrophy of muscles (Figure 11).
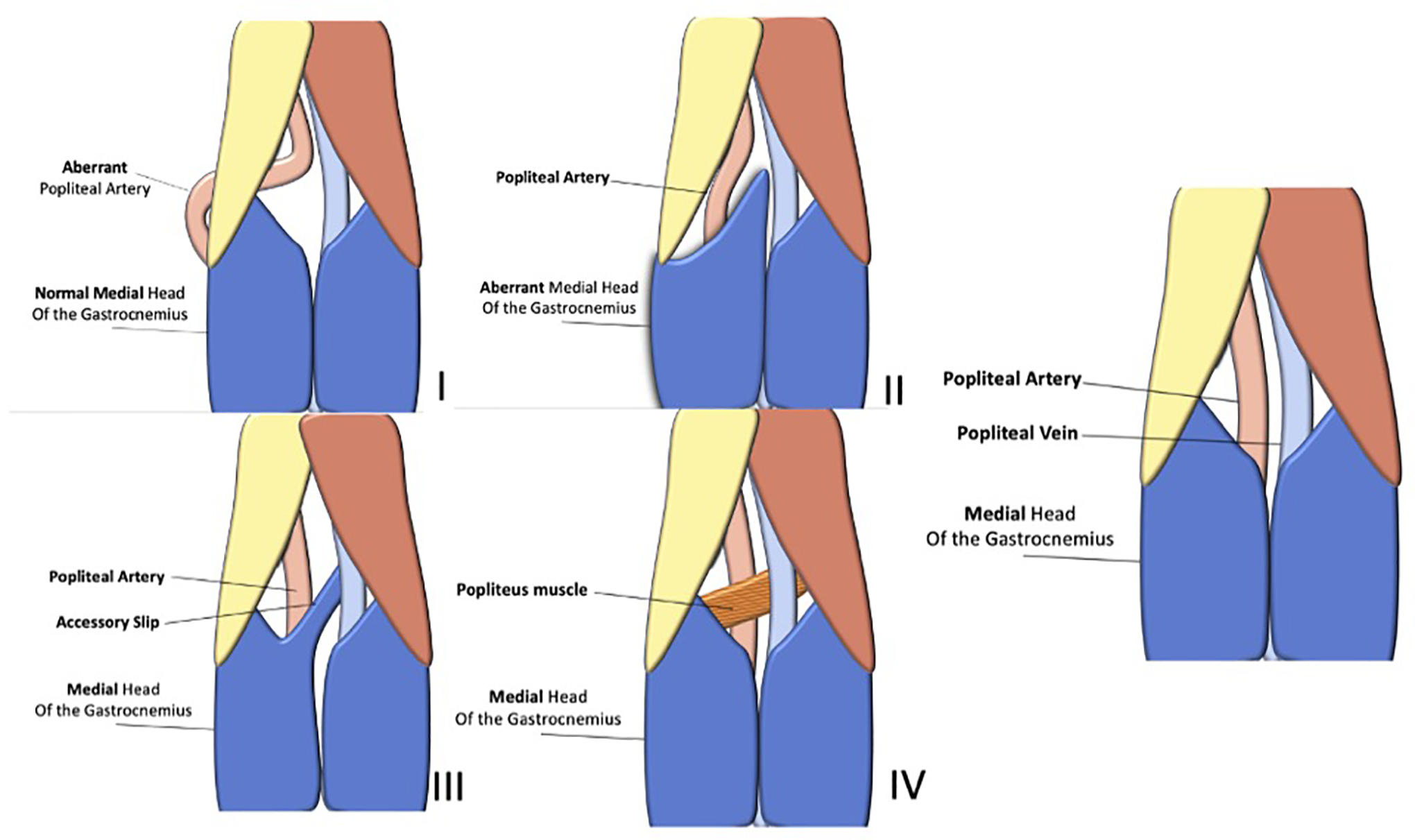
Classification of popliteal entrapment syndrome.
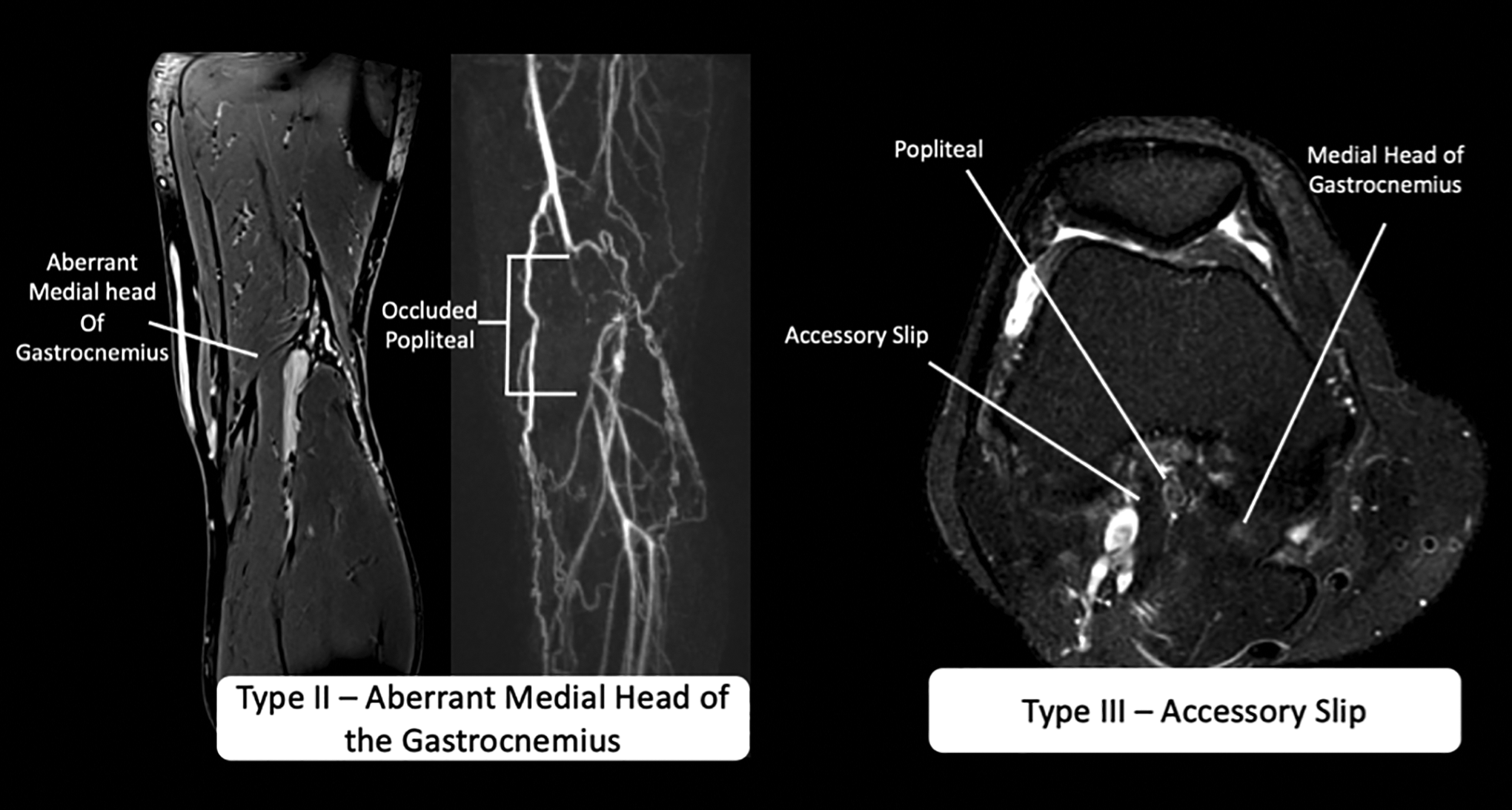
T1 fat saturation post gadolinium MRA demonstrates type II (left) and type III (right) popliteal artery entrapment. The left image demonstrates an aberrant lateral origin of the medial head of the gastrocnemius muscle. The 3D angiogram reformats demonstrate short segment occlusion of the popliteal artery in this region. The right image demonstrates an accessory slip of the medial head of the gastrocnemius muscle. The popliteal artery is occluded in between the accessory slip and the origin of the medial head of the gastrocnemius muscle.
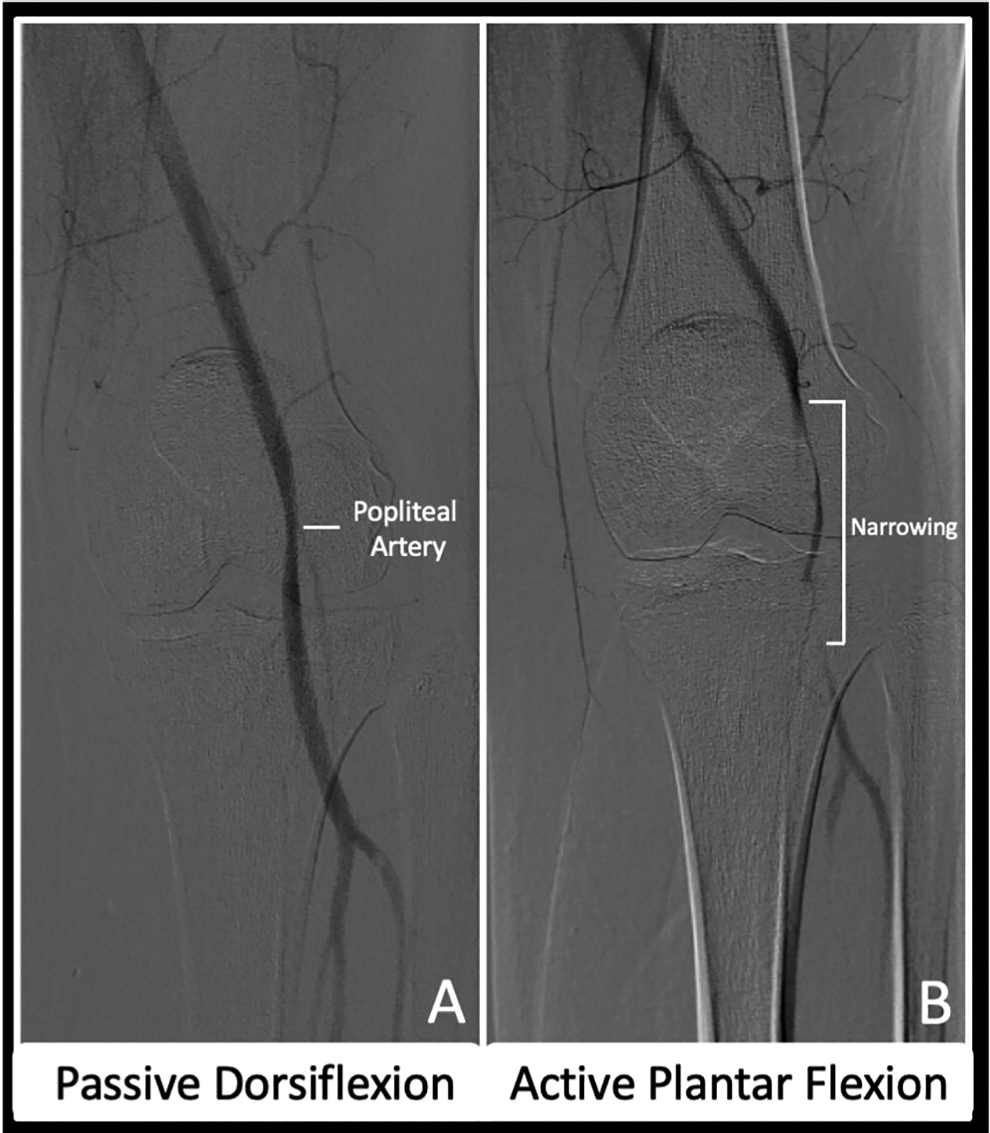
Type VI Popliteal Entrapment Syndrome. Conventional digital subtraction angiogram demonstrates mild narrowing of the popliteal artery with passive dorsiflexion and severe narrowing/occlusion with active plantar flexion.
Embryologically, the proximal medial head of the gastrocnemius muscle arises from the posterior aspect of the fibula and lateral tibia.2,46,47 With further limb development and medial rotation and extension of the knee, the medial head of the gastrocnemius migrates medially across the popliteal fossa.46-48 It ultimately attaches to the posterior surface of the medial femoral condyle.46-48
At the same time, the complex rearrangement of the popliteal artery is also occurring. As described above, the initial “deep” popliteal artery is located anterior to the popliteus muscle, while the definite vessel is located posterior to the muscle. As the axial artery extends further into the lower limb, through remodeling the deep popliteal artery regresses while the new vessel is formed by angiogenic mechanisms.
If the final popliteal artery forms before the migration of the medial head of the gastrocnemius muscle, the artery is displaced medially along with the muscle as it migrates medially.46,48 This forms type I popliteal entrapment. In type II entrapment, the popliteal artery is still medially displaced, due to an abnormal positioned medial head of the gastrocnemius muscle. The medial head is lateral to the medial femoral condyle (generally intercondylar region) due to incomplete migration. 46 In type III entrapment, a portion of the medial head completely migrates while the other part of it does not (called the accessory slip). 46 The accessory slip can either arise from the medial or lateral femoral condyles. The definitive popliteal artery generally passes in between the 2 heads of the medial gastrocnemius. In type IV, the embryological popliteal artery (ramus perforans cruris) remains as the popliteal artery, therefore the popliteal artery is located anterior to the popliteus muscle. In type V entrapment, there is compression of the popliteal vein in addition to the artery by any of the above mechanisms. In type VI entrapment, no anatomical abnormality is seen. It is termed “functional,” and believed to result from hypertrophy of the medial gastrocnemius muscle, which compresses the posterior aspect of the popliteal artery during plantar flexion. 46
Conclusion
Variations of arterial lower limb anatomy are often explained by a combination of persistent primitive arterial segments, abnormal fusion, segmental hypoplasia or absence of vessels. Failure to recognize congenital variations can result in procedural/surgical complications including limb ischemia, life threating hemorrhage, or a failure of intended surgery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
