Abstract
An acute aortic syndrome (AAS) is an important life-threatening condition that requires early detection and management. Acute intramural hematoma (IMH), aortic dissection (AD) and penetrating atherosclerotic ulcer (PAU) are included in AAS. ADs can be classified using the well-known Stanford or DeBakey classification systems. However, these classification systems omit description of arch dissections, anatomic variants, and morphologic features that impact outcome. The Society for Vascular Surgery and Society of Thoracic Surgeons (SVS-STS) have recently introduced a classification system that classifies ADs according to the location of the entry tear (primary intimomedial tear, PIT) and the proximal and distal extent of involvement, but does not include description of all morphologic features that may have diagnostic and prognostic significance. This review describes these classification systems for ADs and other AAS entities as well as their limitations. Typical computed tomography angiography (CTA) imaging appearance and differentiating features of ADs, limited intimal tears (LITs), IMHs, intramural blood pools (IBPs), ulcer-like projections (ULPs), and PAUs will be discussed. Furthermore, this review highlights common imaging interpretation pitfalls, what should be included in a comprehensive CTA report, and provides a brief overview of current management options.
Introduction
Acute aortic syndromes (AAS) can lead to rapid clinical deterioration and require immediate detection and management. 1 The clinical entities of aortic dissections (AD), intramural hematomas (IMH), and penetrating atherosclerotic ulcers (PAU) cannot be differentiated on the basis of their associated symptoms, which are similar, often involving chest or back pain. This highlights the importance of imaging, particularly with computed tomography angiography (CTA) as an early assessment tool to detect the presence of the various AAS entities and further classify and describe them.2,3 CTA is considered the first-line imaging modality for AAS due to its high spatial resolution and diagnostic accuracy, fast acquisition and processing time, widespread availability, and easier patient access and tolerance compared to transesophageal echocardiography (TEE) or magnetic resonance imaging (MRI).4,5 A protocol of non-enhanced CT, used to detect acute IMH, followed by CTA should be used to increase diagnostic accuracy and detection of AAS.5-8 Multidetector CT with multiplanar and volume rendering reconstructions nicely demonstrate aortic branches and the site and suitability of stent graft landing zones, which helps guide management decision making. 9 Although transthoracic echocardiograms (TTE) or TEE may be suitable in emergency situations, they do not provide optimal visualization of the distal ascending aorta, aortic arch, and are also operator dependent.4,9,10 Echocardiography may be done acutely for diagnostic purposes (useful to rule in but not to rule out AD), but does not eliminate the need for cross-sectional imaging to determine the extent of the dissection and branch vessel involvement. TEE is critical intraoperatively to visualize aortic valve function and TTE may be useful in follow-up if there are ongoing root and valve concerns.4,10
Cardiac motion artifacts, especially in patients with tachycardia, may mimic a dissection and electrocardiogram (ECG) gated imaging should be used to accurately assess the aortic root and coronary arteries where possible (Figure 1).5-8,11 However, some centres may not have the capability for ECG-gated CTA, and in these instances a non-gated CTA with optimization of scan speed is usually sufficient to exclude AAS. Post-processing using multiplanar and volume rendered reconstructions should also be performed to ensure adequate detection and characterization of aortic lesions.6,8,11 3D anatomic visualization of ADs is also possible by MRA but with lower spatial resolution. 12 However, MRI surveillance may be an option for follow-up of chronic ADs in order to avoid cumulative radiation.4,5 MRI may be considered to exclude AAS in younger or pregnant patients with underlying aortopathy, such as Marfan syndrome or Ehlers-Danlos syndrome.5,10
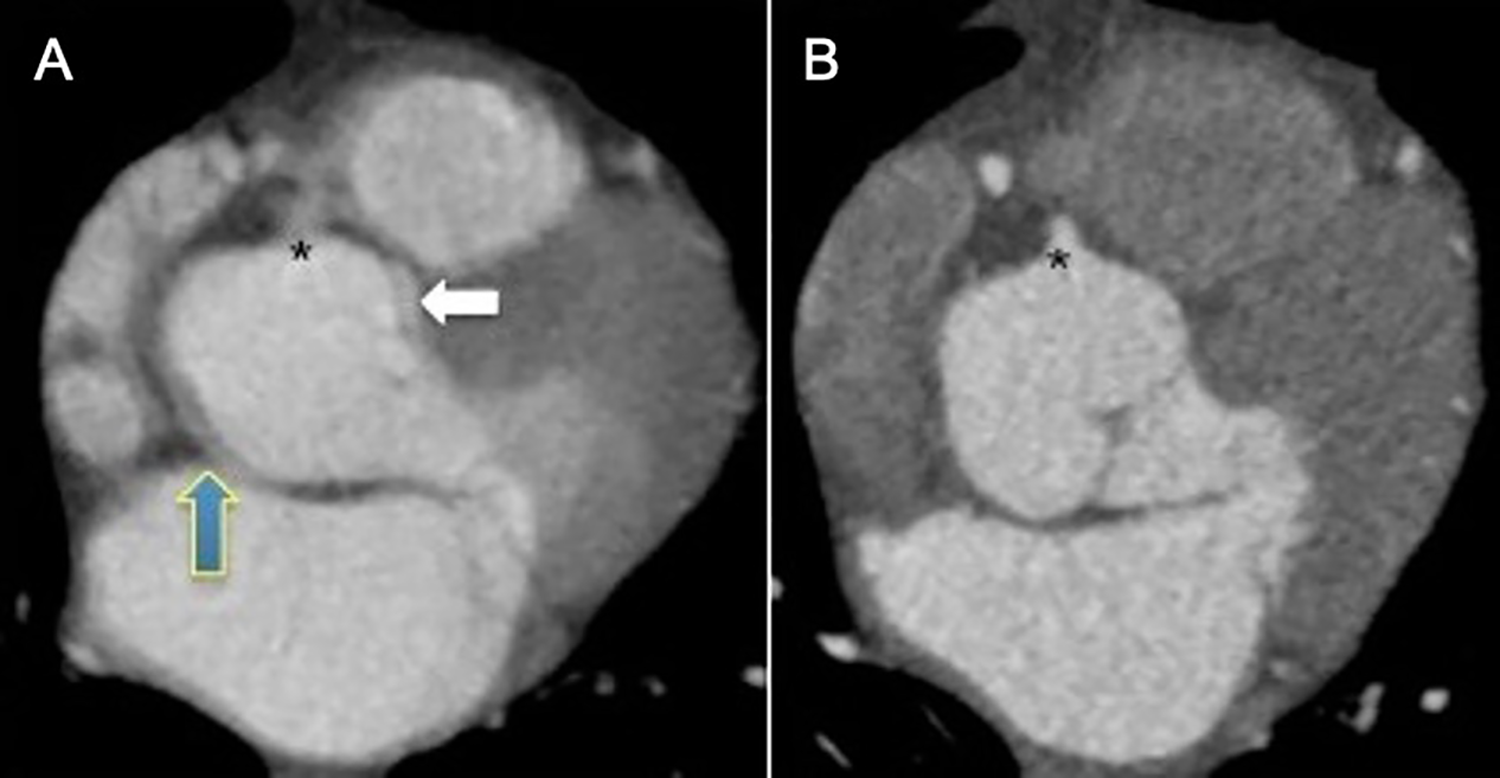
Cardiac motion artifacts eliminated with ECG-gated CTA. A, Non-ECG gated CTA demonstrating cardiac motion blurring at the level of the aortic root (blue and white arrows). The double edges of the aortic root may be mistaken for aortic dissection flap by inexperienced readers. B, ECG-gated CTA in the same patient performed 1 day later to confirm absence of aortic dissection. The aortic root and the right coronary ostium (*) are better visualized on the ECG-gated CTA due to elimination of cardiac motion blurring. Case courtesy of Dr. Mini Pakkal.
Various classification systems, such as the Stanford, 13 and DeBakey 14 classification systems have been used to characterize AAS entities. 12 The Stanford classification system is widely used for ADs and IMHs and categorizes patients into those who benefit from primary urgent surgical management and those who may not (Figure 2). 13 The DeBakey classification system is used exclusively to classify ADs omitting description of other AAS entities (Figure 2). 14 More recently, the Society for Vascular Surgery and Society of Thoracic Surgery (SVS-STS) have introduced a new classification system based on description of the entry tear (primary intimomedial tear, PIT) location as well as proximal and distal extent of the AD (Figure 3). 15 This classification system aims to address the limitations of previous classifications, (i.e., allows description of arch dissections, IMHs, and, PAUs) while facilitating accurate anatomic description of the AAS entity to help guide management, which includes medical, surgical, and endovascular interventions. 15
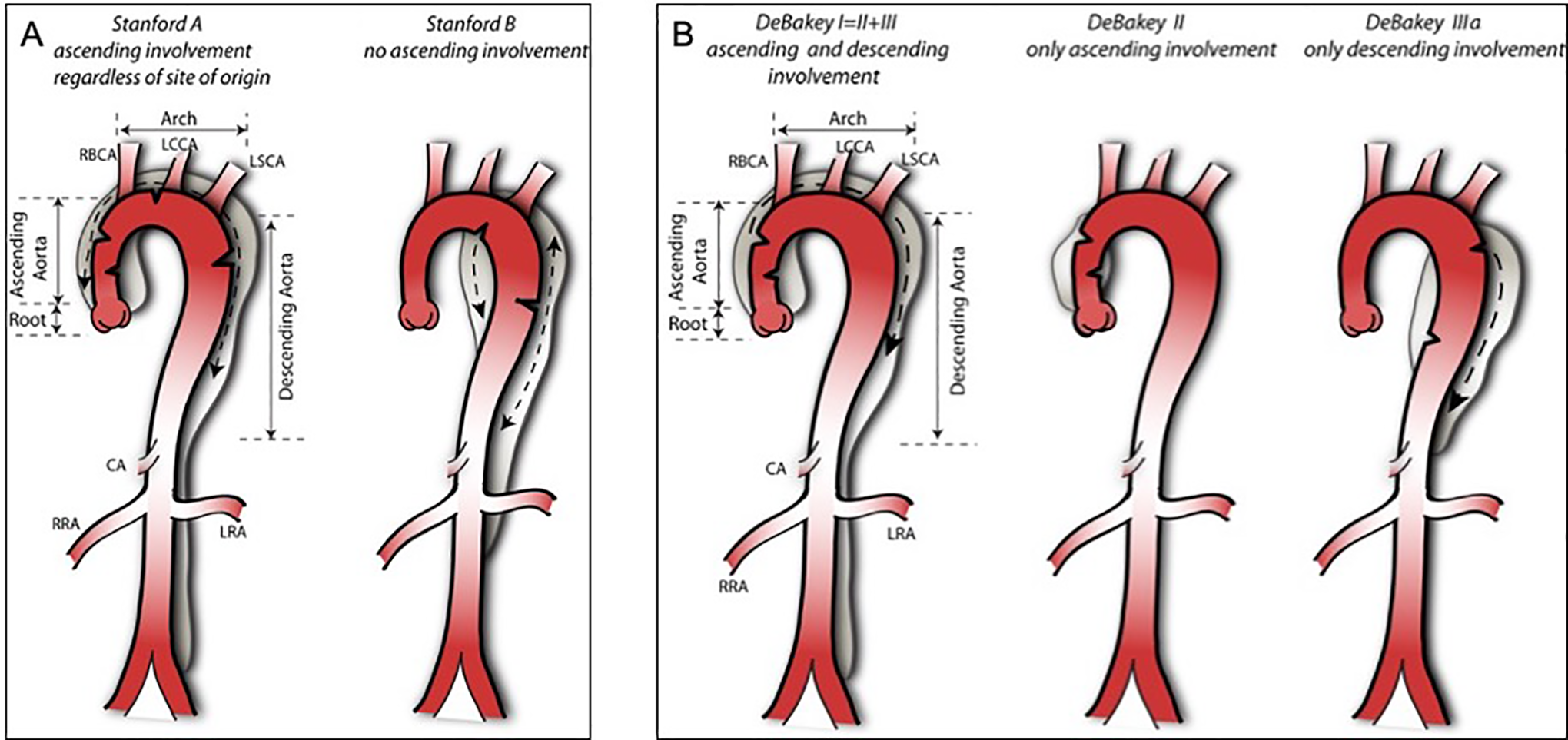
Stanford and DeBakey classifications of aortic dissections. A, Stanford classification: all dissections involving the ascending aorta (up to the origin of the right brachiocephalic artery) are type A dissections. Dissections that do not involve the ascending aorta are type B, the majority of which involve the descending thoracic aorta distal to the left subclavian artery. However, the left subclavian artery is not the anatomic boundary between stanford type A and B dissections, but rather type A dissections involve the ascending aorta, and type B do not. B, DeBakey classification: type I originates in the ascending aorta and extends antegrade to involve at least the aortic arch and typically the descending aorta. Type II originates in, and is limited to the ascending aorta. Type III originates in the descending aorta distal to the left subclavian artery, and typically extends antegrade to involve the thoracoabdominal aorta (type IIIa dissection is limited to the thoracic aorta, type IIIb dissection extends below the diaphragm). RBCA indicates right brachiocephalic artery; LCCA, left common carotid artery; LSCA, left subclavian artery; CA, celiac artery; RRA, right renal artery; LRA, left renal artery. Images courtesy of Dr. Kun Huang.
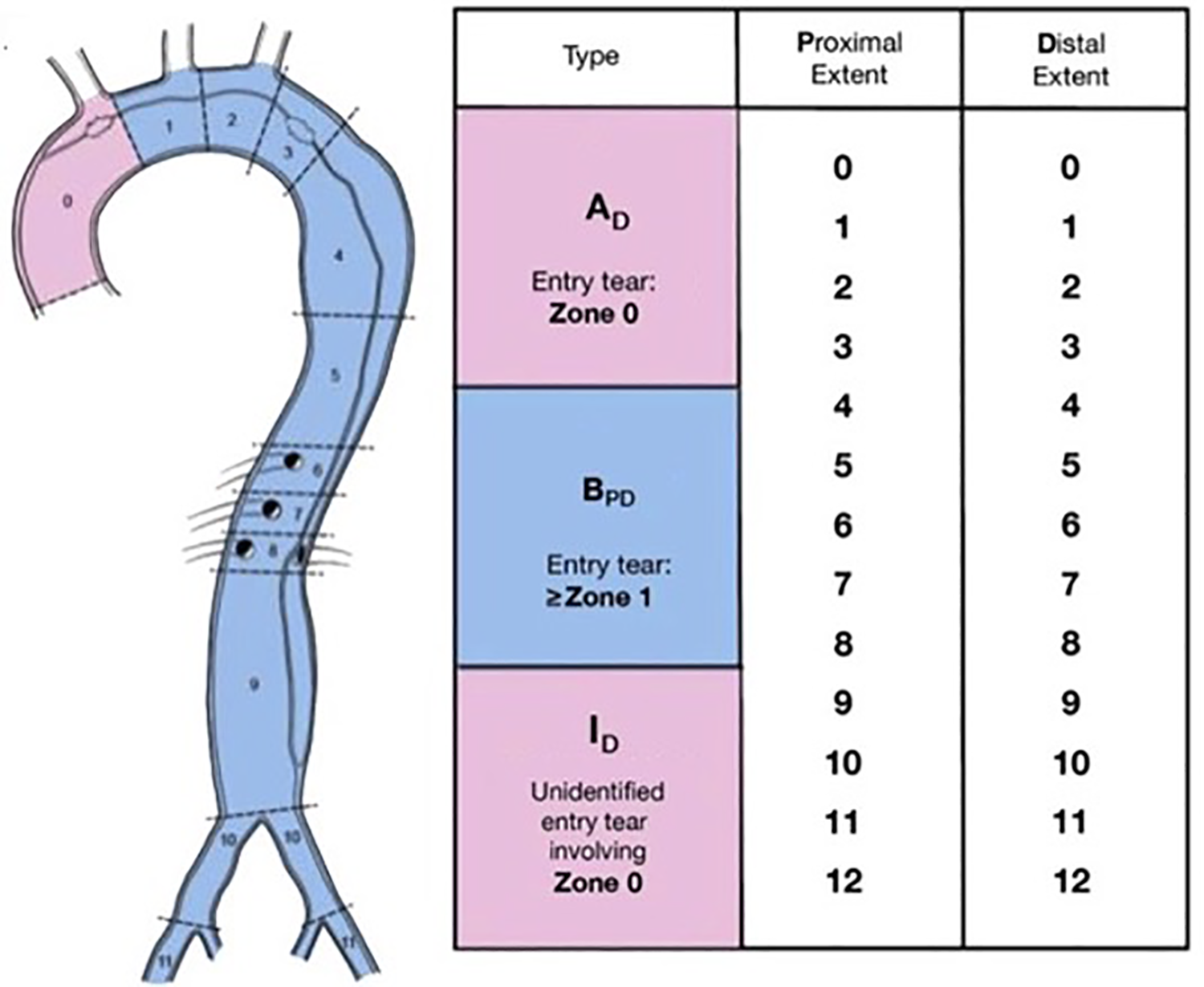
Society for Vascular Surgery-Society of Thoracic Surgeons (SVS-STS) aortic dissection classification system. Zones of attachment for reporting thoracic endovascular aortic repair (TEVAR) as originally described by Fillinger et al, 2010. Type A originates in zone 0, type B originates in zone 1 and beyond, and unidentified entry tears are indeterminate (I). Adapted from Lombardi et al, 2020, reprinted with permission.
The objective of this paper is to present an overview of the classification systems and typical CT appearances of AAS, including imaging interpretation pitfalls and imaging findings that have prognostic significance. Furthermore, this review highlights what should be described in a comprehensive CTA report and provides a brief overview of current management options.
Aortic Dissections
Definition
ADs are defined by a PIT involving the intimomedial layer of the aorta creating 2 pressurized channels. 16 The channels are comprised of the inner, typically smaller true lumen (TL) and the outer, typically larger false lumen (FL).2,16 The FL is within the aortic media (Table 1).
Acute Aortic Syndromes and Entities With Key Imaging Features and Classifications Systems Used to Describe them.

Classification
The Stanford and DeBakey classifications systems aid in standardized multidisciplinary communication and management decisions of ADs because they are widely used by the surgical community.13,14,17 The Stanford system classifies dissections into type A, which involve the ascending aorta, and type B, which do not involve the ascending aorta. 13 However, the Stanford classification (Figure 2) does not define the distal extent of the dissection, which is described in the DeBakey classification. 13 The DeBakey system classifies dissections into type I, which is a dissection that involves the ascending aorta, aortic arch, and the descending aorta, type II, which is limited to the ascending aorta, and type IIIa and b, which are dissections involving the descending aorta alone and dissections in the descending and abdominal aorta, respectively (Figure 2). 14 However, ADs limited to the arch and dissections with retrograde extension into the arch but not involving the ascending aorta, are not described by either classification system and this has led to uncertainty with regard to description, management, and prognosis (Figure 4).17-20 The term “non-A non-B” or “type C” have been used to describe this subset of dissections, or Type B with arch involvement, or simply arch dissection.18-21 In contrast, the SVS-STS classification describes arch ADs because it divides the thoracoabdominal aorta into zones utilized in thoracic endovascular aortic repair (TEVAR) reporting standards (Figure 3).15,22 This nomenclature divides the aorta into zones (0-11), specifying that type A dissections originate in zone 0 (ascending aorta) and type B dissections originate in zone 1 or beyond.15,22 Furthermore, the extent of dissection is also designated by zone number, such that a type A dissection with distal extension to zone 6 would be denoted as A6. 15 Type B dissections are further subdivided into proximal and distal dissection zones, such that B0,9 would represent a retrograde type B dissection originating in zone 1, extending proximally into zone 0 and distally to zone 9 (Figure 3). 15
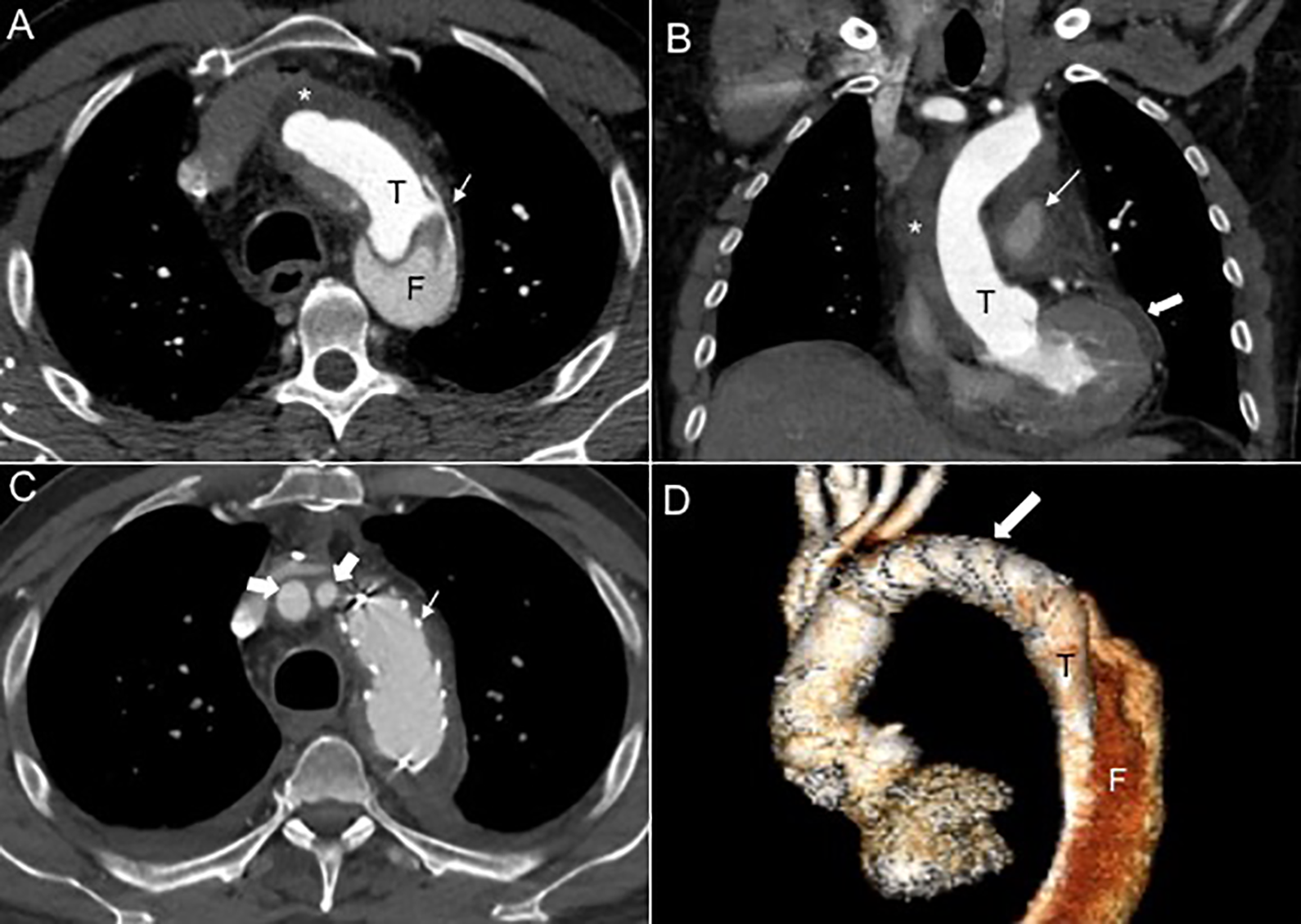
Primary intimomedial tear (PIT) in the descending aorta with retrograde extension of aortic dissection (AD) to the ascending aorta. Stanford type A, B0, 10. Pre (a, b) and post (c, d) total arch and frozen elephant trunk (FET) repair. A, Axial CTA showing the true lumen (T), the false lumen (F) with complete thrombosis of the false lumen in the ascending aorta (*), and the PIT in the descending aorta (white arrow). B, Coronal CTA showing the true lumen, thrombosis of the false lumen (*) in the ascending aorta, hematoma surrounding the pulmonary artery (thin white arrow), and hemopericardium (thick white arrow), which are known complications of ADs due to rupture. C, Post-repair axial CTA shows evidence of arch graft replacement with FET technique (thin white arrow) and patent RBCA and LCCA (thick arrows). D, Post-repair volume rendered image showing the graft replacement of the ascending aorta and aortic arch and endovascular stent-graft (white arrow) in the proximal descending thoracic aorta that sealed the PIT. The RBCA and the LCCA arise from a common re-implanted graft from the right anterior aspect of the ascending aorta. The distal extent of the AD was the common iliac arteries (not shown).
Imaging Findings
ADs appear most commonly as a double-barrel lumen consisting of a TL separated from an FL with a distinct intimal flap defect (Figures 4 and 5).6,7 Typically, the PIT is located in areas of greatest hydraulic stress, such as the right lateral wall of the ascending aorta or proximal segment of the descending aorta close to the ligamentum arteriosum in the descending aorta. 4 There may be multiple sites of intimal injury and CTA allows for thin-section imaging to capture intimal defects as small as 1 mm.23,24 In the early angiographic phase, the TL is more intensely opacified due to faster flow compared to the FL. 6 Typically, the FL is larger than the TL and crescent shaped (Figures 4 and 5). 6 Rarely, a three-channel AD or an aorta with several FLs may exist. 6 Anatomic variants, such as the vertebral artery originating directly from the aortic arch, can have clinical implications. 25
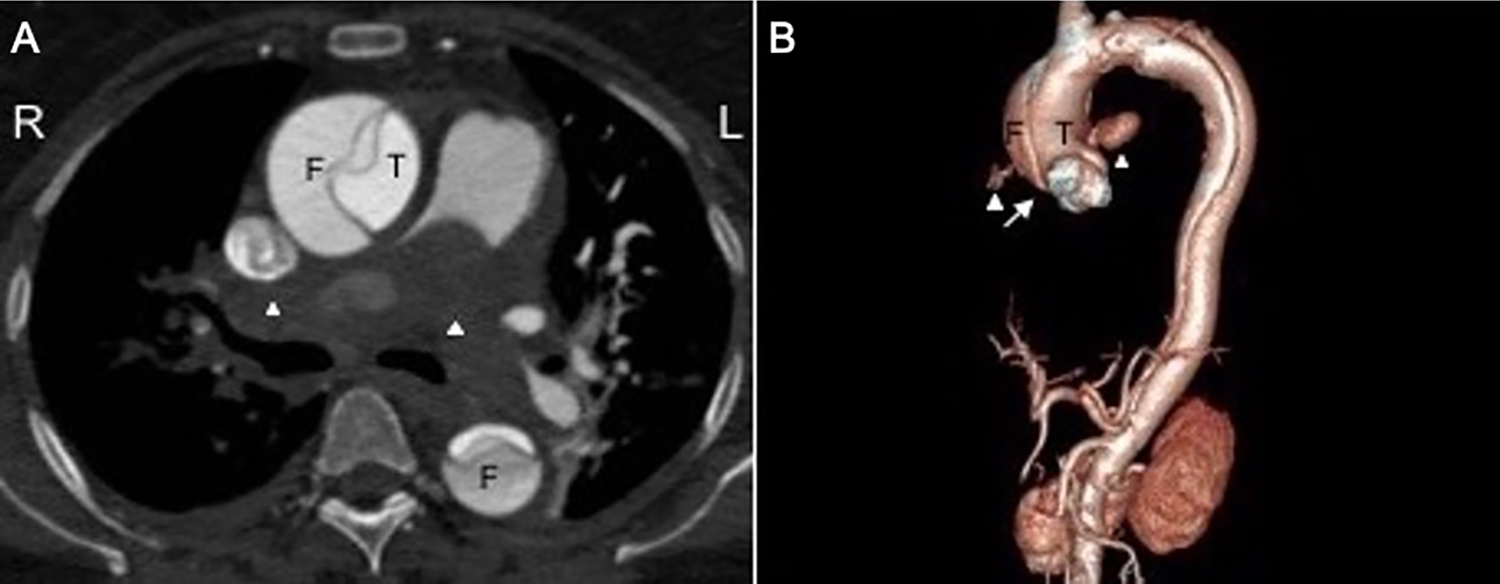
Sleeve hematoma secondary to ruptured aortic dissection mimicking large saddle pulmonary embolism. Stanford type A, DeBakey I, A11. A, Axial contrast-enhanced CT chest shows dissection flap in the ascending aorta separating the true lumen (T) and false lumen (F). Significant narrowing of the lumen by an apparent filling defect in the right and left pulmonary arteries (white arrow heads) due to a sleeve hematoma that resulted from the ruptured dissection tracking along the shared adventitia between the aorta and pulmonary arteries, thereby mimicking pulmonary embolism (PE). The hematoma from ruptured aortic dissections tends to be smooth, hyperdense, circumferential, and contiguous and may even extend into the segmental and subsegmental level and into alveolar air spaces in contrast to acute PE. Significant luminal compression of the pulmonary arteries may occur as in this case with large volume hematoma. B, Volume rendering demonstrates the type A dissection with PIT in the ascending aorta (not shown) extending retrograde to the sinotubular junction (white arrow) but not involving the aortic root. Contrast-filled outpouchings represent points of rupture from the ascending aorta both anteriorly and posteriorly (white arrowheads). The distal extent of the dissection was the external iliac arteries (not shown).
Imaging Interpretation Pitfalls
Technical factors that may impede adequate evaluation of the dissection include insufficient contrast enhancement and streak artifacts from metal or dense contrast in the left innominate vein.6,26 Aneurysms and diverticula can also present a challenge and may fold back against the aortic wall creating an illusion of an intimal flap on axial imaging. 7 Similarly, periaortic structures may also superimpose onto the aorta and create a double lumen appearance.6,26 Ruptured type A dissection can cause a sleeve hematoma external to the lumen of the pulmonary arteries along the shared adventitia that can be mistaken for pulmonary embolism (Figure 5).27-29 The hematoma from ruptured AD tends to be smooth, hyperdense to blood pool, circumferential and contiguous and may even extend into the segmental and subsegmental level and into alveolar air spaces in contrast to acute PE. 29 Sleeve hematomas along the pulmonary arteries most often result from a posterior aortic wall rupture. 29
Reporting
In CTA reports of ADs, it is important to comment on the location and size of the PIT, extent and direction of propagation, as well as location and size of re-entry tears. 23 It is important to identify all branch vessels and their degree of patency and lumen of origin, size of the aorta, degree of thrombosis of the FL, and presence of periaortic or pleural fluid.4,11 Most type A dissections require immediate surgical intervention and the degree to which a type A dissection extends into the aortic root, aortic valve, aortic arch, coronary arteries, and great vessels impacts surgical management options and prognosis. 2
High risk features of type B dissections should be described in CTA reports and include a large entry tear (>10 mm), PIT in the lesser curvature of the arch, an initial aortic diameter of >4 cm at first baseline CTA, and a partially thrombosed FL with increased intraluminal pressure are associated with risk of subsequent rupture.4,6,11,15,30-32 Partial thrombosis of the FL in the descending aorta can lead to FL expansion and TL compression causing collapse and subsequent branch vessel and organ malperfusion.10,33 Complete thrombosis of the FL is typically a favorable imaging finding associated with good prognosis.34,35 Furthermore, increase of ≥ 5 mm in maximal aortic diameter in any segment after any form of management and the source of persistent flow to the FL must be reported on follow-up studies. 15
ADs in the descending aorta with retrograde dissection limited only to the arch have better outcomes than those with retrograde extension into the ascending aorta (Figure 4). 36 Thus, differentiating retrograde extension to the arch versus ascending aorta is important to describe in CTA reports. 36 A PIT located in the distal arch or partial thrombosis of the FL increases the likelihood of retrograde extension into the ascending aorta and poor aortic remodeling, thus patients must be monitored closely. 37 Retrograde extension to involve the arch or ascending aorta confers higher risk of neurologic, cardiac, and organ malperfusion complications compared to isolated type B dissections and also warrant closer clinical monitoring (Figure 4).19,38,39
Management
According to current guidelines, first line treatment for type A antegrade ADs is a supra-coronary ascending aorta graft replacement, intimal resection, and if necessary, aortic valve repair or replacement of the aortic root. 40 Extension of the repair into the aortic arch, including with the frozen elephant trunk technique has become increasingly used in patients with arch pathology (arch tear or aneurysmal arch) and may be beneficial in patients with distal malperfusion (Figure 4). 41 Acute type B dissections are typically medically managed unless complications develop such as aortic rupture, shock, periaortic hematoma, refractory hypertension or pain, or malperfusion syndromes causing end-organ, visceral, or lower limb ischemia.4,11,20,33,40 Certain predictors of poor aortic remodeling and subsequent need for open or endovascular surgery, sare also increasingly used in the absence of the above-mentioned complications as indications for surgical intervention in the acute setting. The literature currently suggests first-line conservative therapy for arch dissections followed by TEVAR or conventional surgery. This is done to induce FL thrombosis and help re-expand the TL through stent grafting.11,17,18,21
Intramural Hematomas
Definition
Historically, IMHs were thought to arise from the spontaneous rupture of the vasa vasorum leading to hemorrhage within the aortic media in the absence of a PIT.12,38,42-44 With current high spatial resolution CT imaging, they are now thought to be secondary to small intimal tears that thrombosed, causing aortic wall hemorrhage without a re-entry tear (Table 1).12,38,43,44 IMHs may also be iatrogenic secondary to percutaneous catheter manipulation or caused by acute trauma, such as motor vehicle crashes. 45
Classification
The Stanford classification system categorizes type A IMHs as those involving the ascending aorta and type B IMHs as those that do not, similar to ADs.9,44 IMHs occurring in the aortic arch may be described as “type B with arch involvement.”9,45 The SVS-STS classification can also be used for IMH. 15
Imaging Findings
On unenhanced CT, IMHs appear as hyperdense, smooth, crescentic or circular shape typically with aortic wall thickening ≥ 5 mm and wall attenuation of 39-72 HU.1,4,45 There is no associated dissection flap and no flow in the false channel differentiating IMH from ADs (Figure 6).1,4,9,38,42 On contrast-enhanced imaging the aortic wall hemorrhage/hematoma does not enhance. 11 The presence of intimal calcifications can help distinguish an IMH from an intraluminal thrombus, as IMHs tend to displace intimal calcifications toward the center of the aortic lumen whereas calcifications remain on the outer border of luminal thrombi. 38
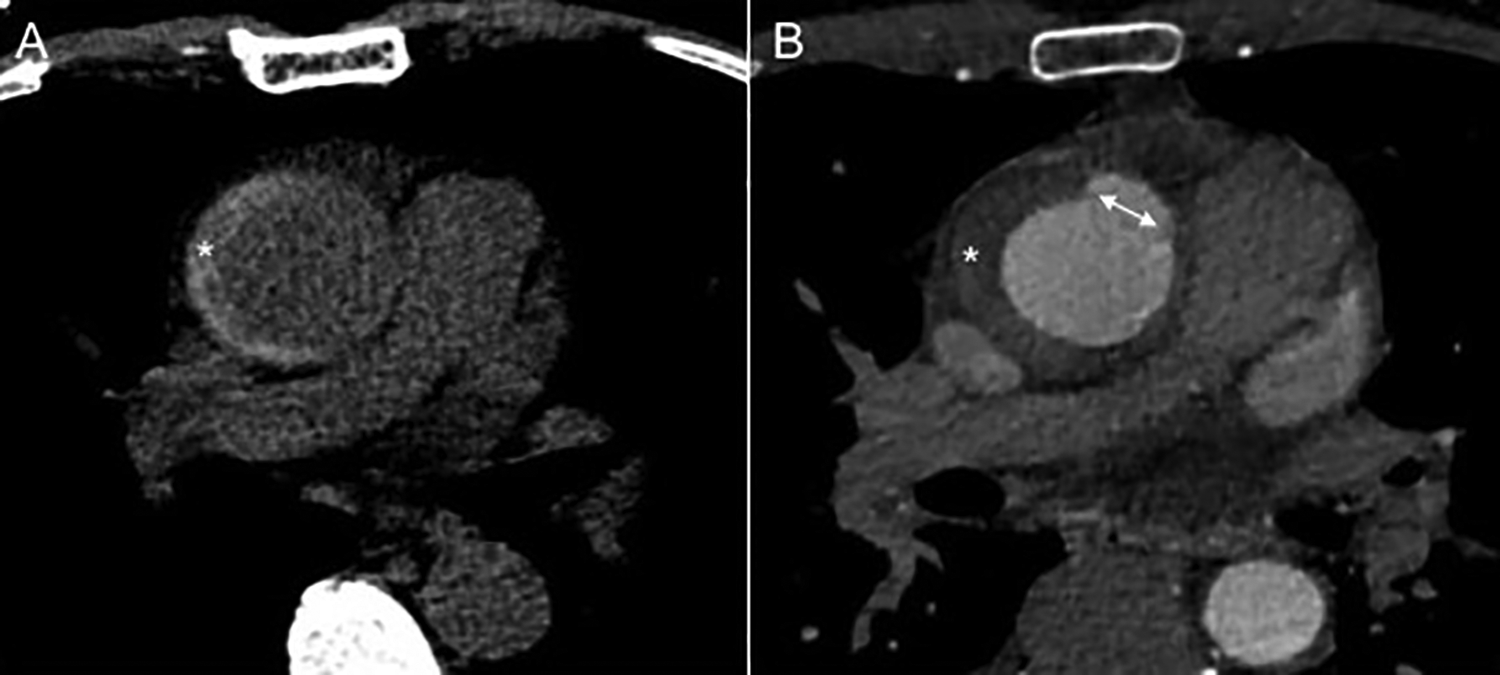
Acute intramural hematoma (IMH) with development of ulcer-like projections (ULP). Stanford type A, A2. A, A hyperdense crescent (*) on non-contrast imaging consistent with acute type A IMH in the ascending aorta extending to the arch. B, ULP (double-headed white arrow) in the ascending aorta that developed near the IMH (*) on subsequent CTA imaging one week later.
IMHs in the ascending aorta and aortic arch are at higher risk of developing an ulcer-like projection (ULP) due to the greater hydraulic wall shear stress in these locations (Figure 6). 38 ULPs are localized blood-filled pouches protruding from the TL into the thrombosed FL, and this indicates the existence of a flow channel between the 2 lumens, typically through a tear >3 mm in diameter (Table 1).6,45 Intramural blood pools (IBP) also known as aortic branch artery pseudoaneurysms (BAP), are also outpouchings of localized contrast pools inside the IMH and form secondary to the damage caused by IMH propagation across branch vessels, most commonly in the descending aorta (intercostal, lumbar arteries etc.) (Table 1).6,43,46 They may be partially or completely torn and so the communication between the IBP and TL may be absent or typically <2 mm in diameter.6,43,46
Imaging Interpretation Pitfalls
It can be difficult to distinguish IMHs from ADs with thrombosed FL. However, delayed venous phase imaging can aid in this differentiation but may require recall of the patient for this delayed phase scan as it is not part of standard CTA protocols. 4 Furthermore, a thrombosed FL in an AD tends to have a spiral course longitudinally in the aorta whereas an IMH maintains a constant circumferential relationship throughout. 7 Special attention must be paid in the setting of a hyperattenuating crescent in association with an aneurysmal dilation, as this is a sign of impending rupture and the presence of an IMH is most likely a secondary complication of the pre-existing aneurysm (38). If the PIT was missed on the baseline CTA, it can be difficult to distinguish an evolving PIT from a ULP. 47 IBPs typically have a narrower neck than ULPS and this may help to differentiate the two. Rarely an aortic abscess can mimic a low density IMH but is often associated with periaortic fat stranding due to inflammation. 7
Reporting
CTA reports for IMH should describe maximum hematoma thickness, maximum aortic diameter, presence of IBPs or ULPs, ULP diameter and depth, and presence or absence of pericardial effusion, pleural effusion, or periaortic hematoma.38,42,43 A PIT in the context of IMH or thrombosed FL must also be localized as it can direct the course of treatment, especially when TEVAR is being considered (6).
Management
The Stanford classification system classifies IMHs and ADs in a similar fashion. However, their management is not necessarily the same. 38 IMHs may progress to ADs, aneurysms or pseudoaneurysms and lead to branch vessel compromise or eventual rupture.4,38,45 Only 10% of thoracic IMHs resorb over time.5,11 Even after resolution of the IMH, long-term surveillance is recommended.5,11
Maximal aortic diameter and luminal diameter at the level of maximal IMH thickness as well as maximal axial thickness of the hematoma have prognostic implications.4,11 Features correlating with worse outcomes for IMH include involvement of the ascending aorta, a maximal diameter of ≥55 mm in the ascending aorta or ≥40 mm in the descending aorta, increased wall hematoma/thickness ≥10 mm, presence of ULPs, periaortic hemorrhage, or pleural effusion.4,11,42,43,45 In particular, ULPs in the ascending aorta and aortic arch are at risk of progressing to dissection, aneurysms, or rupture. 43 IBPs decrease the probability of IMH resorption over time (43, 46). However, they often have lower risk of complications compared to ULPs.43,46
Surgery has been suggested as first-line management for type A IMHs similar to ADs. However, optimal timing remains uncertain.4,11,20,45 There is some evidence to show that non-urgent surgery or even medical management are options in high-risk patients with multiple co-morbidities.9,43,46 Furthermore, an uncomplicated asymptomatic type A IMH with no evidence of organ malperfusion, or progressive dilatation may be suitable for medical management with close follow-up, although this topic remains controversial and may have varying natural history depending on the ethnicity of the study population.1,44,48 It is important to distinguish between type A IMHs and type A ADs, as urgent surgery is not indicated in all type A IMH cases, although a low threshold for intervention should be considered in patients with high risk imaging features. 1 Type B IMHs are managed non-surgically with continued surveillance unless complications arise. In such instances, they are treated with surgical repair or endovascular treatment.42,43 Poor prognostic factors for type B IMH are age >70 years, cardiac tamponade, presence of ULPs, increased initial maximal aortic diameter, and maximal hematoma thickness.4,49
Limited Intimal Tears
Definition
LITs, also known as “incomplete dissections,” are tears through the intima and superficial media without the creation of a FL.7,50 LITs are often located in the ascending aorta and associated with aortic dilatation (Table 1).51,52
Classification
LITs are classified into type A (ascending aorta) and B (transverse arch and descending aorta) as per the Stanford classification, but can also be described using the SVS-STS classification.15,52
Imaging Findings
LITs are best seen with 3D volume rendered images with ECG-gating especially when in the ascending aorta (Figure 7). 52 LITs are characterized by a stellate or linear intimal tear associated with minimal amounts of blood in the dissected aortic wall.6,50 They are often associated with focal linear filling defects, small amounts of local IMH under the lesion edges, and an eccentric aortic bulge or minor contour abnormality at the base of the intimomedial defect. 52 They are referred to as a “mushroom” cap lesion based on appearance on volume rendered reconstructions.6,51,52 Interestingly, most patients with type B LITs (unlike ADs and IMHs) have aneurysmal aortas. 52
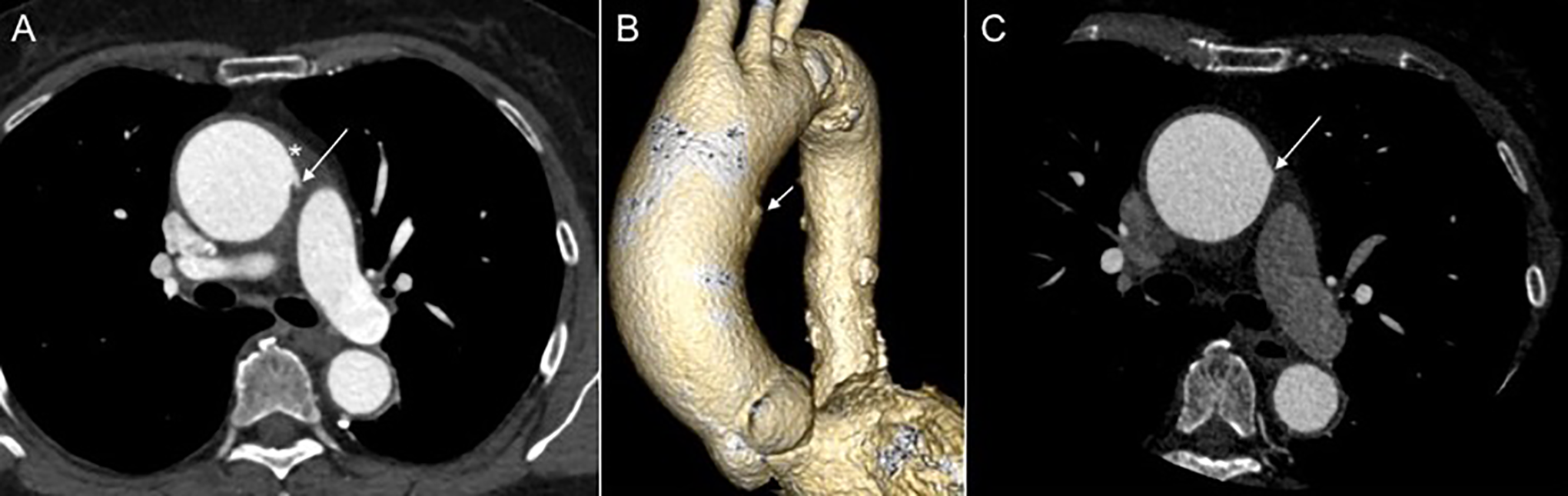
Acute limited intimal tear (LIT) in the ascending aorta. Stanford type A, A0. A, Axial CTA an outward “bulge” (arrow) characteristic of a LIT in a dilated ascending aorta (45 mm). A hyperdense crescent representing intramural hematoma (IMH) is surrounding the LIT (*). B, 3D volume rendered image shows an outward bulge consistent with LIT along the left aspect of the ascending aorta (white arrow). Note the denser calcified atherosclerotic plaques in the arch and descending aorta. C, Follow-up axial CTA 2 months later showing a significant size reduction of both the IMH and LIT (white arrow).
Imaging Interpretation Pitfalls
It can be difficult to distinguish LITs from other aortic pathologies on imaging, especially if the radiologist is unfamiliar with this entity.7,52 LITs are typically misclassified as IMH or PAU, due to their localized pathology. However, most often there is no associated hematoma or atherosclerotic disease accompanying the intimal defect of a LIT.12,52,53 The presentation of AD symptoms without evidence of a dissection flap on imaging should alert radiologists to the possibility of the presence of a LIT.50,54 This is especially true if there is an aortic aneurysm, significant unexplained aortic valve regurgitation, or pericardial effusion. 50
Reporting
These entities are typically underdiagnosed due to misclassification or lack of detection. 52 Although, misclassification as another aortic lesion may not have detrimental consequences, understanding the pathophysiology, imaging, and outcomes of LIT is important to enable accurate diagnosis and appropriate treatment. 52 These entities may progress to AD, rupture and cause cardiac tamponade, thus making accurate and timely detection critically important. 50 LIT size, location and associated IMH should be described in CTA reports.
Management
In contrast to a type A ADs, type A LITs seem to respond positively to medical management. 52 However, there are limited long term outcome data and surgical repair remains the current mainstay of treatment. 52
Penetrating Atherosclerotic Ulcers
Definition
A PAU typically occurs in the setting of pre-existing atherosclerotic disease and forms when an ulcerated atheroma erodes the inner arterial wall creating a hematoma in the intimomedial layer (Table 1). 3 They predominantly occur in the descending aorta and arch, and rarely in the ascending aorta.4,12,55
Classification
PAUs can also be described using the SVS-STS classification.4,15,50 The SVS-STS classification also facilitates description of the proximal and distal extent of an IMH that be associated with the PAU. 15
Imaging Findings
PAUs are crater-like ulcers with irregular margins protruding beyond the adventitial borders of the aorta. They are often seen in the descending aorta and surrounded by atherosclerotic plaque.4,7,11 They are typically seen on CTA as contrast enhanced outpouchings of the descending aortic wall often beyond the normal adventitial boundary with adjacent sub-intimal hematoma (Figure 8).2,11 There are often multiple sites present and each can range from 2-25 mm in diameter and 4-30 mm in depth. 11
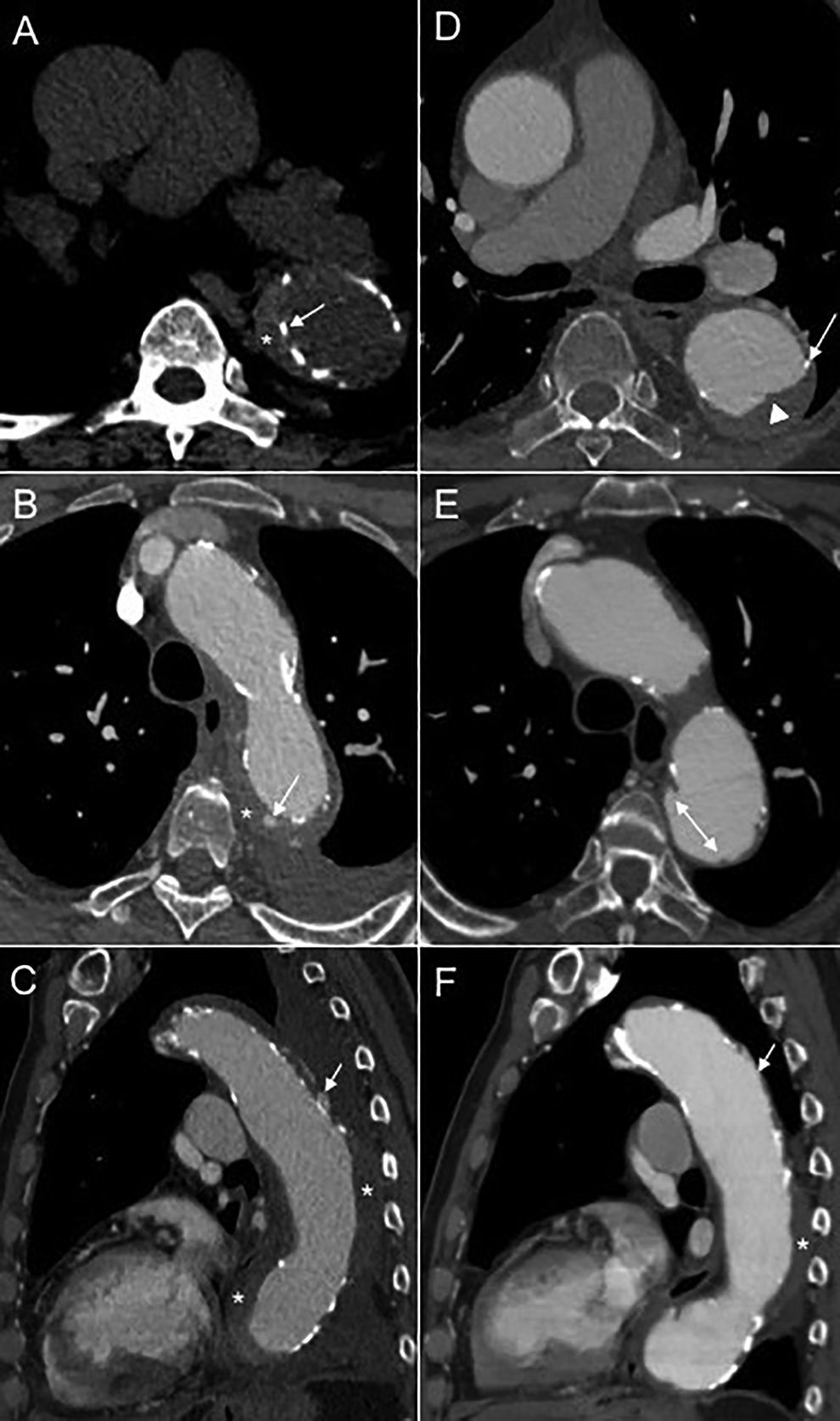
Acute intramural hematoma (IMH) secondary to enlarging penetrating atherosclerotic ulcer (PAU) in descending aorta. Stanford type B, B3,10 PAU3. A, A hyperdense crescent (*) on axial non-contrast CT imaging consistent with acute type B IMH in the descending aorta. The intimal calcifications (white arrow) are displaced medially confirming hematoma in the medial layer that is typical of IMH. B, CTA demonstrates a tiny focal ulcerated atherosclerotic plaque (white arrow) in the proximal descending aorta that may be the source of the acute IMH (*) that increased in size on follow-up with appearance of a PAU (shown in E). Note the calcified atheroma in the descending aorta, typically seen with PAUs. There is also a small left pleural effusion. C, Sagittal CTA view of the aorta shows the development of an intramural blood pool (IBP) (white arrow) within the IMH (*). Note the small left pleural effusion. Follow-up CTA 4 months later (D-F). D, the IMH has resolved at this location in the descending aorta and a luminal thrombus (white arrowhead) has developed. The intimal calcifications (white arrow) are not displaced medially therefore, the new thrombus is luminal rather than in the medial layer as expected for IMH. Notice the irregular internal contour of the luminal thrombus in contrast to the IMH (typically a smooth hyperdense crescent). E, The PAU (white double headed arrow) has enlarged over time at 4-month follow-up. F, Sagittal CTA view shows the IBP (white arrow) has resolved over time at 4-month follow-up (in contrast to ulcer-like projections) and the IMH has decreased (*).
Imaging Interpretation Pitfalls
Differentiating a PAU from ULP is often quite challenging. This is because PAUs may be associated with the presence of an adjacent IMH, similar to ULPs (Figure 8).9,43 However, ULPs are typically identified on follow-up imaging rather than initial CTA and are usually found in the ascending aorta where there are fewer atherosclerotic plaques. 43 In contrast, PAUs are commonly in the descending aorta and surrounded by atherosclerotic plaques. 43 Mycotic aneurysms are saccular dilatations that can be mistaken for a PAU, although calcifications and thrombus are not as frequently associated. 7
Reporting
PAUs with initial diameter >20 mm or neck >10 mm and those associated with IMH are at higher risk of progression to pseudoaneurysm, AD and rupture and may benefit from early intervention.9,56,57 Therefore, measurement of the size of the PAU is helpful to include in CTA reports to direct appropriate management.
Management
Among the various AAS entities, PAUs are at the highest risk of aortic rupture. 4 Type A PAUs are especially at high risk of rupture and require urgent intervention. 4 Type B PAUs are medically managed unless there are signs and symptoms of hemodynamic instability, persistent pain, aortic rupture, distal embolization, presence of periaortic bleeding, or pleural effusion.6,9,58 Complications of PAUs include development of IMH, AD, Pseudoaneurysm, or rupture.3,11 TEVAR is the preferred treatment option because most patients have multiple comorbidities and are at advanced age.3,9,20,42,58 It is an effective treatment for type B PAU when pain and hypertension are difficult to control or when there are signs of expansion or interval growth.5,20
Conclusion
Various AAS entities are described using multiple classifications systems. The Stanford and DeBakey classifications are widely used, especially among cardiothoracic or cardiovascular surgeons, although, they have limitations. The SVS-STS classification addresses many of the limitations of the other classification systems but is only recently published and may not be relevant to physicians who are not experts in the diagnosis and management of aortic diseases including AAS. The SVS-STS classification, however, is a robust and easy to use classification system that not only aids in description and management of clinical cases but also facilitates accurate description of aortic pathologies in study populations for comparative outcome research studies. CTA plays an important role in identifying the spectrum of AAS entities based on characteristic imaging features and should be ECG-gated where possible. It is important to recognize the differentiating features on CTA that separate one AAS entity from another as well as morphologic features that carry prognostic significance. It is also important to be aware of interpretation pitfalls and to accurately describe the AAS entities in radiology reports with as much detail as possible to help guide appropriate patient management.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
