Abstract
Health leaders must consider various solutions to alleviate the healthcare management burden within Canadian hospitals. Increased access and rapidity of Infectious Diseases (IDs) testing via multiplex Point-of-Care Tests (POCTs) is an example. Twenty-four interviews assessed the facilitators and barriers to implementing such tests in the inpatient hospital setting in Quebec, Ontario, and British Columbia. The perspective of health leaders, including provincial laboratory officials, medical and laboratory directors, POCT managers, physicians, and nurses, revealed the following major themes as being relevant to the sustainable adoption and implementation of a multiplex POCT for IDs: (1) current diagnostic methods and experience with POCTs; (2) disease burden and level of health system integration affecting a hospital; (3) the management capacity of hospitals; and (4) the anticipated impact on healthcare management efficiency. The study provides structure for health leaders to evaluate and plan for necessary organizational transformation for adopting multiplex POCTs.
Introduction
In Canada, the decentralization of diagnostic tests in the form of multiplex Point-of-Care Tests (POCTs) for Infectious Diseases (IDs) is a relevant topic for healthcare management, 1 with wide-ranging operational implications that could address the concerning wait-times for patients, burnout in the healthcare workforce, 2 and Antimicrobial Resistance (AMR) and Stewardship (AMS) efforts, 3 especially during infection outbreaks when infection prevention decisions are critical. 4 Equity and access to care and improved spatial justice are additional important Canadian healthcare priorities.5-7
The objective of a POCT is to obtain results with greater speed and efficiency, for timelier decision-making, by removing the need for specimen transportation to a central laboratory. 8 Specimens can be analyzed by an individual without laboratory science credentials. 9 Examples of POCTs that have been implemented in Canada include those assessing a few specific molecules or biomarkers, such as blood gases, glucose, pregnancy status, drug intake, and SARS-CoV-2 antigens.1,10 Previous studies have reported how the implementation of such POCTs required the assessment of the following factors: funding, staffing, training, documentation, quality assurance, standardization, and oversight.1,11-13 In the case of multiplex identification panels that can detect multiple analytes simultaneously, assessing readiness for adoption and implementation can be even more challenging.14,15 However, doing this exercise is important for health leaders seeking solutions to addressing the healthcare management burden of hospitals through a rapid diagnosis, improved bed allocation, and more judicious use of antimicrobials, in line with AMS efforts.15-19
Multiplex POCTs have been tested in the context of sexually transmitted and blood-borne infections for their diagnostic performance and suitability within Canadian outreach community settings. They showed excellent specificity and sensitivity, and optimized patient care (e.g., same-day testing and care coordination).20,21 In the United States, multiplex molecular POCTs for IDs have been implemented in hospitals, with evidence supporting efficient patient care management and improved patient outcomes. 22 To the co-authors’ knowledge, no studies have yet investigated feasibility, preparedness, and potential impact of Canadian hospitals considering the implementation and adoption of multiplex POCTs for use within inpatient settings. This lack of evidence prevents health leaders from confidently determining if such change would improve hospital efficiency and strengthen AMS. To bridge this gap, this study explored the barriers, facilitators, and anticipated impact of implementing multiplex POCTs for the diagnosis of IDs within Canadian inpatient hospital settings.
Methods
An environmental scan methodology was selected, 23 using exploratory semi-structured interviews.24,25 The areas of exploration and the thematic analysis were guided by the Practical, Robust Implementation and Sustainability Model (PRISM). 26 PRISM supports the investigation of barriers and facilitators to the sustainable implementation of a health intervention, such as a POCT. 26 “POCTs” were defined for participants as testing performed outside the central laboratory, while “multiplex POCTs for IDs” as POCTs that simultaneously test a panel of multiple potential viral/bacterial infection causes.
Ethics Review
The study was approved by Veritas Independent Review Board (Veritas IRB Inc, Kirkland, Quebec). All steps of the research were conducted in accordance with the Declaration of Helsinki. 27
Sampling Criteria
Three provinces in Canada were prioritized: Quebec (QC), Ontario (ON), and British Columbia (BC), as they account for 76% of the Canadian population. 28 Based on previous literature, 29 the following stakeholder groups were included: laboratory directors, POCT coordinators/site supervisors/committee members/chairs, administration representatives, and nurses/physicians involved in the diagnosis or treatment of IDs in the inpatient hospital setting. Exclusion criteria were: >50% of professional time spent researching/teaching, currently retired, self-rated knowledge level of POCT as “none” or “basic,” self-rated knowledge level of multiplex POCTs for IDs as “none,” and no experience using/implementing a POCT in the last 3 years. Purposive sampling criteria included a variety of hospital types (e.g., university centres/tertiary hospitals vs local hospitals with limited inpatient care) and targeted stakeholders for each province. 30 A sample size of 24, with an ideal quota of 8 participants per province and 12 per hospital type, was determined a priori based on studies demonstrating data saturation after 10-13 interviews in a relatively homogenous group and a minimum of 20 interviews to identify common themes among sub-groups.31,32
Recruitment
Potential participants were identified through professional listings of diagnostic test users, decision-makers, and healthcare professionals, derived from pharmaceutical and health research industry-related organizations in compliance with the ESOMAR/ICC International Code on Market, Opinion and Social Research and Data Analytics. 33 Potential participants were sent an IRB-approved invitation to the study explaining its purpose and method. The invitation included a secure link for a brief eligibility screening, systematically applying research criteria, followed by a consent form and opt-in/out for eligible participants. Purposive sampling was applied through continuous monitoring of screening responses and coordination of remaining recruitment to obtain desired sub-group quotas. All interviewees provided informed consent prior to participation. Participants received $150 in financial compensation for their time upon study completion.
Data Collection
Semi-structured interview guides were developed in English, in line with study objectives and areas of exploration. 34 Questions were open-ended with follow-up probes to ensure comprehensive capture of participants’ perspectives.24,25 The interview guide was pilot tested on 4 participants to ensure rich and relevant answers were obtained in alignment with the study objectives and a 45-60 minutes completion time. The final interview guide was translated into French. All interviews were conducted in English or French (participant preference) by co-author MA (MPH, female researcher, trained in qualitative methods, experienced in public health, performance, and quality improvement research). Interviews were conducted on-line using Zoom video communication software. The moderator adjusted the pre-determined questions to the participants’ responses and asked spontaneous open-ended questions for in-depth exploration of unanticipated topics and/or perspectives.24,25 The moderator did not have an established relationship with the participants, and participants did not know anything about the moderator prior to the interview. A study recruiter was included only for the introduction of interviews, to ease coordination. No repeat interviews were carried out. One person refused participation without reason, and a second individual cancelled prior to the interview due to other commitments hindering participation. All interviews were audio-recorded, transcribed, and joined with notes taken by the moderator. French transcriptions were translated into English for analysis.
Analysis
A preliminary coding tree was developed based on the areas of exploration and used by the researcher/moderator to code transcripts in NVivo 12 Pro (2017, Lumivero). Transcripts were subsequently reviewed for familiarity and then coded by one coder (co-author MA). The process allowed for new codes to be created, transformed, and validated to ensure that the emerging and final coding tree was appropriately grounded in the interview content. 35 Final coding was reviewed to identify relationships between codes and themes/sub-themes according to what emerged from the data, in guidance with the PRISM model and areas of exploration.26,36 Hence, a mix of inductive and deductive reasoning approaches drove analysis. 37 Findings were collectively interpreted by the multidisciplinary group of co-authors (with backgrounds in POCT, IDs, biochemistry, public health, health systems research, and performance/quality improvement) to triangulate the multiple perspectives of the stakeholders. 38
Results
The sample (n = 24) included 2 laboratory leaders overseeing POCT at the provincial level (BC = 2), 10 directors responsible for leading institutional-level POCT use (QC = 2; ON = 6; BC = 2), 5 managers for department-level POCT use (QC = 2; ON = 3), and 7 hospital employees as end-users (QC = 2; ON = 2; BC = 3). The majority (n = 18) worked in urban hospitals; the remaining worked in rural hospitals (n = 4) or overseeing diverse hospital types (n = 2). Perspectives included those of laboratory representatives, nursing staff, and physicians.
The integrated perspectives of participants revealed the following major themes relevant to the sustainable adoption and implementation of a multiplex POCT for IDs: (1) current diagnostic methods and experience with POCTs; (2) disease burden and level of health system integration affecting a hospital; (3) the management capacity of hospitals in terms of their human resources, space, and information technology; and (4) anticipated impact on healthcare management efficiency (Figure 1). The identified barriers (Table 1) and facilitators (Table 2) associated with each of these major themes are presented with representative quotes. Major Themes Related to the Perceived Sustainability of Adopting and Implementing a Multiplex POCT for Infectious Diseases Barriers to a Sustainable Adoption and Implementation of a Multiplex POCT for IDs Facilitators to a Sustainable Adoption and Implementation of a Multiplex POCT for IDs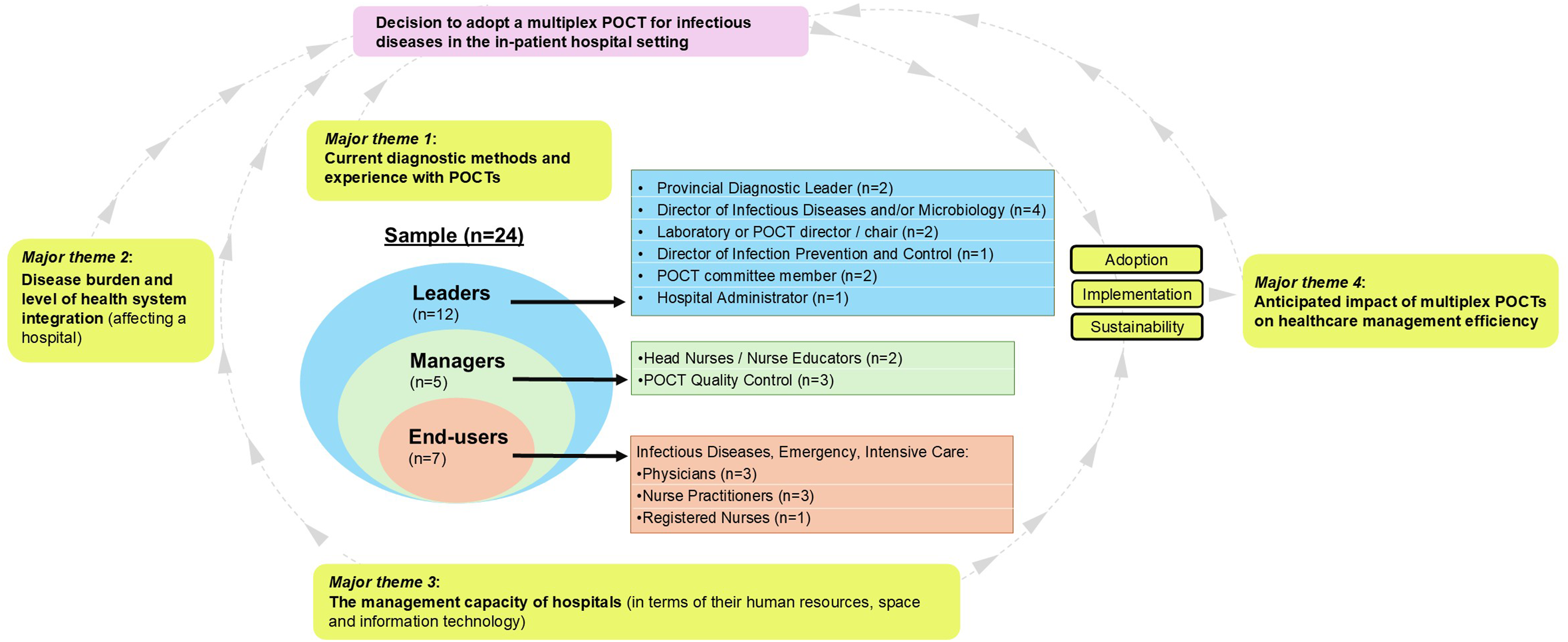


Current Diagnostic Methods and Experience With POCTs
Currently established laboratory practices and varying levels of familiarity and experience with POCTs were found to shape health leaders’ openness towards adopting a multiplex POCT for IDs. Substantial advantages were perceived by participants who worked in rural hospitals relying solely on specimen transportation to an external laboratory by shuttle or airplane, and thus facing significant hurdles (e.g., traffic and challenging routes) to rapid diagnosis. Contrastingly, lesser benefits were perceived by participants working in urban hospitals with well-established internal transportation systems and laboratory processes offering access to timely results for a lesser cost. Experience with POCT molecular assays for SARS-CoV-2 respiratory virus during the pandemic was associated with positive outlooks on the adoption of multiplex POCTs for IDs, as participants felt non-laboratory personnel were able to demonstrate appropriate use and reporting. Contrastingly, participants without much experience with POCTs demonstrated a greater challenge in recognizing the value of adopting such technology. Many laboratory representatives expressed skepticism towards the diagnostic performance of multiplex POCTs, especially with current limited human resources dedicated to training and quality assurance outside the laboratory. This latter factor was considered an important barrier to adoption, necessitating substantial organizational transformation.
Disease Burden and Level of Health System Integration
Disease burden, described in terms of frequency of occurrence and hospitalization requirements, affected the perceived cost-effectiveness of a POCT by health leaders. In urban hospitals, a higher frequency of ID occurrences due to high population density was noted necessitating powerful laboratory testing with greater throughput capacity than possible with a multiplex POCT. However, molecular POCTs were perceived as particularly valuable for IDs with high severity (e.g., respiratory syncytial virus) where downstream savings in hospitalization can be demonstrated. However, health system fragmentation in Canada was perceived as causing substantial variance in POCT standards, with no clear national guidance currently available. Standardization efforts for POCT use in BC were reported as important next steps in facilitating a cohesive provincial implementation of any POCT.
The Management Capacity of Hospitals
The current healthcare management burden of Canadian hospitals, reflected in the shortage of staff and heavy workloads, was reported as an important barrier to the sustainable adoption and implementation of a multiplex POCT for IDs. Training and audit concerns were voiced due to the anticipated steep learning curve related to molecular testing and the risk to laboratory accreditation if POCT protocols are not followed by non-laboratory personnel. This perceived risk was especially noted in busy hospital units with limited storage and greater chances for cross-contamination. A well-established workflow and direct POCT supervisor-staff relationships were discussed as important strategies to overcome these barriers. Further, the capacity to interconnect POCTs with the charting system of a hospital and central laboratories was discussed as necessary for efficient disease documentation and reporting aimed at alleviating healthcare management burden.
Anticipated Impact on Healthcare Management Efficiency
Participants recognized that multiplex POCTs for IDs could positively impact numerous aspects of hospitals’ management processes, including improved bed flow, cost reduction related to specimen transport, reduced risk of exposure to communicable diseases for employees, and increased equity in access to rapid diagnosis. However, participants pointed out that multiplex POCTs could not support the assessment of resistance profiles of identified pathogens as accurately as current laboratory equipment, which is necessary for the optimal use of antimicrobials.
Discussion
Based on the results of this study, the following items need to be considered by health leaders when assessing the relevance and impact of a molecular-based multiplex POCT for IDs within an inpatient hospital setting in Canada: 1. The development of a compelling business case that outlines the operational capacity and capability at the system level; 2. Cost analysis encompassing capital costs and ongoing operational requirements for POCT implementation; 3. Organization readiness emphasizing: a. Human resources available for the performance of POCT; b. IT expertise to integrate technology within current infrastructures; c. Governance structures, including national standards for POCT complexity grading and appropriate use and oversight; d. Available space for storage of POCT and appropriate decontamination; e. POCT quality assurance processes; f. Capabilities for training and oversight; g. Existing familiarity and clinical experience with POCTs; h. Culture of openness towards change, innovation and quality improvement; 4. Risk assessment and mitigation strategies related to the possibility of negative testing outcomes; 5. Value associated with capability to provide rapid molecular testing for syndromic infections to interpret surveillance and preparedness strategies during emergency response events (e.g., improved patient flow and bed allocation, infection prevention and control decisions, and facilitation of stewardship efforts); and 6. Workflow assessment, including clear delegation of roles and responsibilities leading to the obtainment of POCT results.
Our results suggest that the benefit of using a multiplex POCT for IDs outweighs the cost in rural hospitals because of the substantial delays to test results, compared to urban hospitals with in-house laboratories. However, urban hospitals may still benefit from complementary use of molecular POCTs during seasonal outbreaks. 4 Importantly, molecular-based multiplex POCTs for IDs are not meant to replace a controlled laboratory testing environment, and peer comparisons are necessary for verification of end-user competence and instrumentation accuracy. This point is especially relevant to address concerns around transitioning testing from controlled laboratory environments to clinical spaces, including the skepticism of laboratory directors related to the performance and cost-benefit analysis of POCTs compared to laboratory-based tests. 12
Recommendations
We recommend the following prioritized actions to facilitate the sustainable adoption and implementation of molecular multiplex POCTs for IDs in the inpatient hospital setting within Canada: 1. Recruiting IT expertise within hospitals to help connect existing information system and clinical workflows with the POCT; 2. Standardizing protocols at the national and organizational level, related to POCT complexity evaluation, approved POCTs, and enforced quality assurance processes. Such efforts have been undertaken in BC to facilitate system integration
39
; 3. Revision of workflow with respect to the storage, training, use, oversight, and quality assurance of the POCT, ensuring a supportive relationship between POCT supervisors and end-users is established to reduce the risk of negative testing outcomes.
These actions must leverage strong collaboration between the laboratory, clinical, and hospital leaderships to ensure alignment of policies and procedures and ultimately reduce the potential risk for negative outcomes.
Limitations
This study did not fully represent the wide range of hospitals in Canada, nor rural settings, as sampling focused on the three most populated provinces. While purposive sampling was performed, participants were recruited from established lists associated with the use of diagnostic tests, which can explain larger representation of laboratory personnel. Data saturation was reached, but inclusion of participants from other provinces and stakeholder groups may have expanded the results further. Participants did not review transcripts for comments or corrections, nor did they provide feedback on the findings.
Conclusion
This original qualitative research study offers evidence on a hospital management innovation that aims to facilitate the diagnosis of infections and resulting inpatient flow in Canadian hospitals, especially during outbreaks. The study was novel in exploring the anticipated requirements and impact of implementing a molecular multiplex POCT for use by non-laboratory personnel in Canadian hospitals. Health leaders may use this evidence to design local assessment tools and strategic interventions when considering the implementation of higher complexity POCTs for IDs, as part of improved healthcare management strategies.
Footnotes
Acknowledgements
We acknowledge the support provided by bioMérieux Canada for generating interest for this study and would like to thank all participants for their time and reflections.
Ethical Considerations
This study and related materials (study protocol, recruitment material, informed consent, and research tools) were reviewed and approved by an independent review board (IRB Veritas) with the following study number: GR-26-011 and tracking number: 024-3291-18112-1.
Consent to Participate
All study participants provided informed consent prior to participating in the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by independent medical education funds provided by bioMérieux Canada.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MA and PL are employees of AXDEV Group Inc. JS is the clinical lead for the POCT Expert panel for the Ontario Laboratory Medicine Program. JI and KW have no conflicts of interest to disclose.
Data Availability Statement
The interview guide with questions and probes can be accessed as by contacting the corresponding author. The coding tree can be accessed by contacting the corresponding author.
