Abstract
interRAI instruments consist of clinical information systems able to support integrated care. Through a scoping review, we describe how interRAI instruments are used: (1) as interventions (implementation category) and (2) to evaluate interventions (efficacy category) in older adults. In accordance with the PRISMA-ScR framework, we searched 6 databases and conducted dual-independent screening, with conflicts resolved by a third reviewer. Data extraction followed an identical procedure. The review yielded 64 manuscripts, including 43 and 21 categorized as studies of efficacy or implementation, respectively. Findings indicate that interRAI systems are consistently utilized to evaluate or enhance participant-centred outcomes across diverse healthcare settings in 17 countries, with a particular emphasis on home and long-term care. interRAI is a versatile system with the potential to form the foundation of an integrated clinical information system. This review provides a basis for future research testing novel intervention strategies with interRAI systems.
Introduction
Population aging is a worldwide phenomenon. Although rates vary by country, it is expected that by 2050, the size of the aging population will have more than doubled from 900 million in 2015 to 2.1 billion.1,2 Such a demographic shift reflects an increase in life expectancy and a decrease in annual birth rates. 3 Unfortunately, life quality and longevity are not synonymous, meaning that many older adults spend their final years living with health-related issues that increase their need for healthcare. Subsequently, there is an urgent need for interventions to help older adults “age in place,” aiming to maintain quality of life and minimize acute care utilization, particularly as healthcare systems struggle to maintain pace from a combination of rising service demands and shrinking clinical workforces.4,5
The interRAI suite (www.interrai.org) consists of clinical information systems designed to support integrated health and social care of older adults across care settings (i.e., home, acute, and long-term)6-10 by identifying needs, planning care, and allocating resources equitably. 11 The interRAI network comprises more than 150 researchers and clinicians, and interRAI assessments are conducted regularly in over 35 countries, with some national governments mandating their use.12-17 When combined with secondary physiological measures, such as anatomical and functional cerebral imaging,18,19 inflammatory biomarkers, 20 and cardiac profiling, 21 interRAI information systems can facilitate a more holistic understanding of aging and, consequently, inform strategies to mitigate functional decline. Ultimately, interRAI clinical information systems support data-driven decision-making and continuity of care, supporting their utility as population aging progresses.
The interRAI information systems have been the focus of many scientific studies exploring aging-related outcomes, such as falls, 22 cognitive function, 23 comorbidities and mortality, 24 and health service utilization. 25 The systems have also been used to promote care planning in the older adult’s support network 26 and improve healthcare delivery. 27 Several reviews have synthesized and interpreted the use of interRAI instruments to assess clinical outcomes28-30 or compared them to other assessment batteries.31-34 A 2014 systematic review examined the dual role of interRAI instruments both as evaluation instruments for interventions and as interventions to support integrated care planning in home care. 35 A broader mapping of how interRAI information systems can be deployed, either as clinical interventions or in the evaluation of novel interventions across different healthcare settings, would elucidate the full potential of interRAI instruments to support clinical care for older adults.
To this end, we conducted a scoping review of studies involving older adults (i.e., 65+ years of age) that: (1) implement an interRAI system as an intervention to guide care management and/or (2) use an interRAI system to measure an outcome. The research questions were how have interRAI information systems been used: (1) as interventions and (2) to evaluate intervention efficacy in older adults across diverse healthcare settings?
Methods
Study Design and Search Strategy
We conducted the review in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) guidelines.36,37 We systematically searched the following 6 databases, without data or language restrictions, for all relevant literature published prior to January 2025: PubMed, Scopus, Excerpta Medica (Embase), Cumulative Index to Nursing and Allied Health Literature (CINAHL), Academic Search Premier, and PsycInfo. Our search combined subject headings and (title and abstract) keywords for “older adults” and “interRAI.” We pre-registered this scoping review in Open Science Forum (https://osf.io/bgjkp), and the detailed protocol paper was published in June 2025. 38
Study Selection
We uploaded all records identified through the database search to Covidence (Melbourne, Australia),39,40 where duplicates were automatically removed. Title, abstract, and full-text screening were conducted independently by two reviewers. We discussed any discrepancies during a consensus meeting, with unresolved disagreements settled by the corresponding author.
To be eligible for inclusion in this scoping review, manuscripts had to focus on older adults (aged 65+ years) and/or individuals (i.e., caregivers and hospital staff) providing some element of care to older adults, regardless of setting. Further, an interRAI instrument had to be a central component of a randomized or non-randomized prospective clinical intervention, examining either the: 1. Effectiveness of an intervention (i.e., physical exercise or medication changes) on outcome(s) (i.e., muscle strength or fall prevention) derived from an interRAI instrument—“efficacy” category. 2. Effectiveness of an interRAI instrument as an intervention in itself to inform care management—“implementation” category.
We also reviewed the reference list of all included studies to identify eligible sources not captured during the initial search and screening. We excluded studies published as editorials, opinions, reviews, meta-analyses, dissertations/theses, and/or conference abstracts. We also excluded grey literature because it is typically difficult to reliably determine whether an instrument was used as an intervention, to evaluate an intervention, or simply as part of routine data collection.
Data Extraction and Presentation
We extracted the following data from each included study: 1. Study ID (i.e., author and publication year). 2. Study design (i.e., randomized or non-randomized intervention). 3. Intervention target (i.e., older adults and/or formal care providers). 4. Study country of origin. 5. Characteristics (i.e., age and sample size) for older adults and/or their providers. 6. Study setting (i.e., home/community and acute). 7. interRAI instrument used. 8. Intervention characteristics (i.e., frequency, length, and type) and which interRAI instrument was used as part of the intervention (implementation category). 9. Outcome, including the specific measure and whether it was an interRAI instrument; if it was an interRAI instrument, we clarified the component of the system used (i.e., scale, individual item, quality indicator, and clinical assessment protocol) (efficacy category); in alignment with scoping review best practices, and because it would be methodologically unsound given the between-study heterogeneity stemming from our broad inclusion criteria, we refrained from between-study comparison in our narrative synthesis.36,37
Results are presented in narrative and graphical form. Detailed supplementary material is available from the corresponding author upon request.
Results
Search Results
In total, our study search yielded 11,446 manuscripts from the 6 databases searched (Figure 1—flowchart). Exactly 3,527 manuscripts underwent title and abstract screening, of which 147 proceeded to full-text screening. The top 3 reasons for exclusion following the full-text screening: age less than 65 years or participants’ age being unclear (n = 22), an ineligible version (i.e., v3.0) of the Minimum Data Set (n = 21), and an ineligible study design (n = 11). In the end, we selected 64 manuscripts for this review, including 43 and 21 in the efficacy and implementation categories, respectively.41–104 Flowchart of Study Selection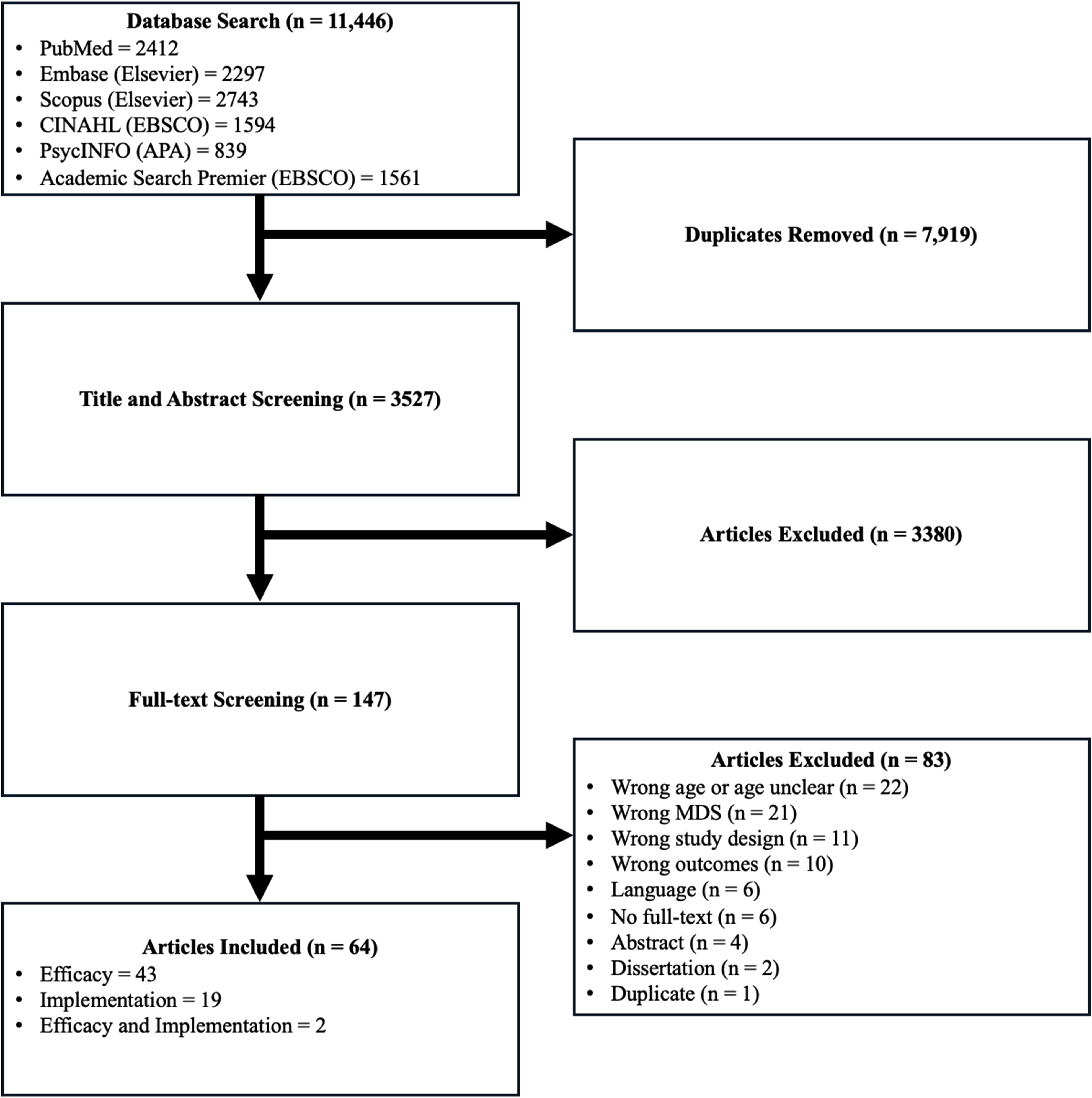
Efficacy Studies
Of the 43 efficacy studies included, 18 were randomized controlled trials. Across study characteristics, the United States was the most common study location (n = 17; 39.5%), long-term care was the most frequent setting (n = 32; 74.4%), version 2.0 of the Minimum Data Set was the most widely used tool (n = 28; 65.1%), and non-randomized designs predominated (n = 25; 58.1%) (Figure 2). The year of publication ranged from 1999 to 2024 and included studies from 13 different countries covering 6 continents—the missing inhabited continent being South America. For studies that reported mean age, the average participant age ranged from 69.0 to 91.8. The sample size ranged from 6 to 122,570 participants per group, with females comprising as few as 39.4% of the sample or as much as 89.9%; 7 studies (16.3%) did not report the percentage of their sample who identified as female in some or all groups. Characteristics of Efficacy Studies. MDS, Minimum Dataset; LTC, Long-Term Care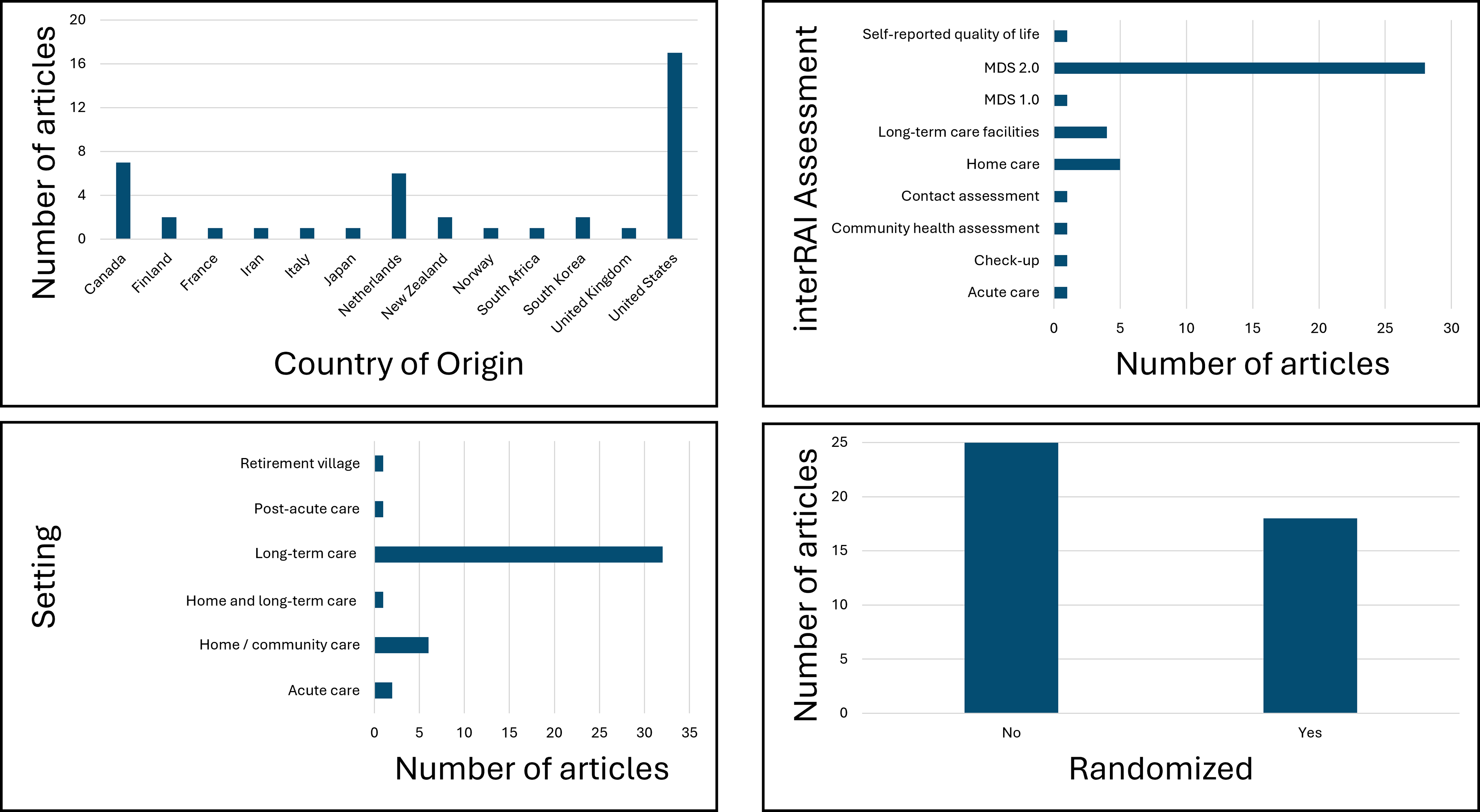
Interventions were as brief as 1 week and as long as 2 years. The frequency of the intervention (i.e., the number of times the intervention was deployed) varied considerably, with the most frequent being daily, and the fewest being just a one-time mobilization or a few times (i.e., quarterly) per year; frequency was not indicated in several studies (n = 29; 67.4%). The types of interventions and the objectives they aimed to accomplish also varied considerably. For example, several studies deployed novel programs (i.e., EDEN alternative,45,47 CHOICE, 50 AgeWell, 60 and PIECES 57 ) in which interRAI instruments or components were used to assess one or several outcomes; such programs were often overseen by a multidisciplinary team led by a physician or nurse. Other types of interventions included, but were not limited to: some form of therapy (i.e., geropsychological and behaviour-based ergonomics),44,52,79 education,42,50,61,63,70-72 and restorative43,54 care. Examples of intervention objectives include improving functional status and life quality, 43 preventing the use of physical restraints, 73 and reducing falls. 80 Three interventions primarily focused on testing medications/supplements.66,68,76 Control groups were more common in the studies that deployed randomization (n = 16/18; 88.9%), with some variation of “routine care” being the most common control intervention (n = 19/43; 44.2%). Outcomes measured varied considerably across studies, with the most common being participant-centred outcomes extracted from interRAI instruments (i.e., activities of daily living, n = 18, 41.9%; pain management, n = 16, 37.2%).
Implementation Studies
Of the 21 studies that focused on the implementation of an interRAI assessment, 10 (47.6%) were randomized controlled trials. Across study characteristics, the United States, Italy, and the Netherlands were the most common study locations (n = 3, 14.3%), home care was the most frequent setting (n = 11, 52.4%) and the most widely used tool (n = 12, 57.1%), and non-randomized designs predominated (n = 11, 52.4%) (Figure 3). Two of the 21 studies also had an “efficacy” component.94,100 Date of publication ranged from 1997 to 2018, and comprised 11 unique countries spanning Europe, North America, Asia, and Oceania. Participants’ average age ranged from 73.2 years per group to 85.8 years. Group size ranged from 14 to 2,118 participants; females represented anywhere from 50% of a sample to 83%. Eight studies did not report on sample sex balance. Characteristics of Implementation Studies. MDS, Minimum Dataset; LTC, Long-Term Care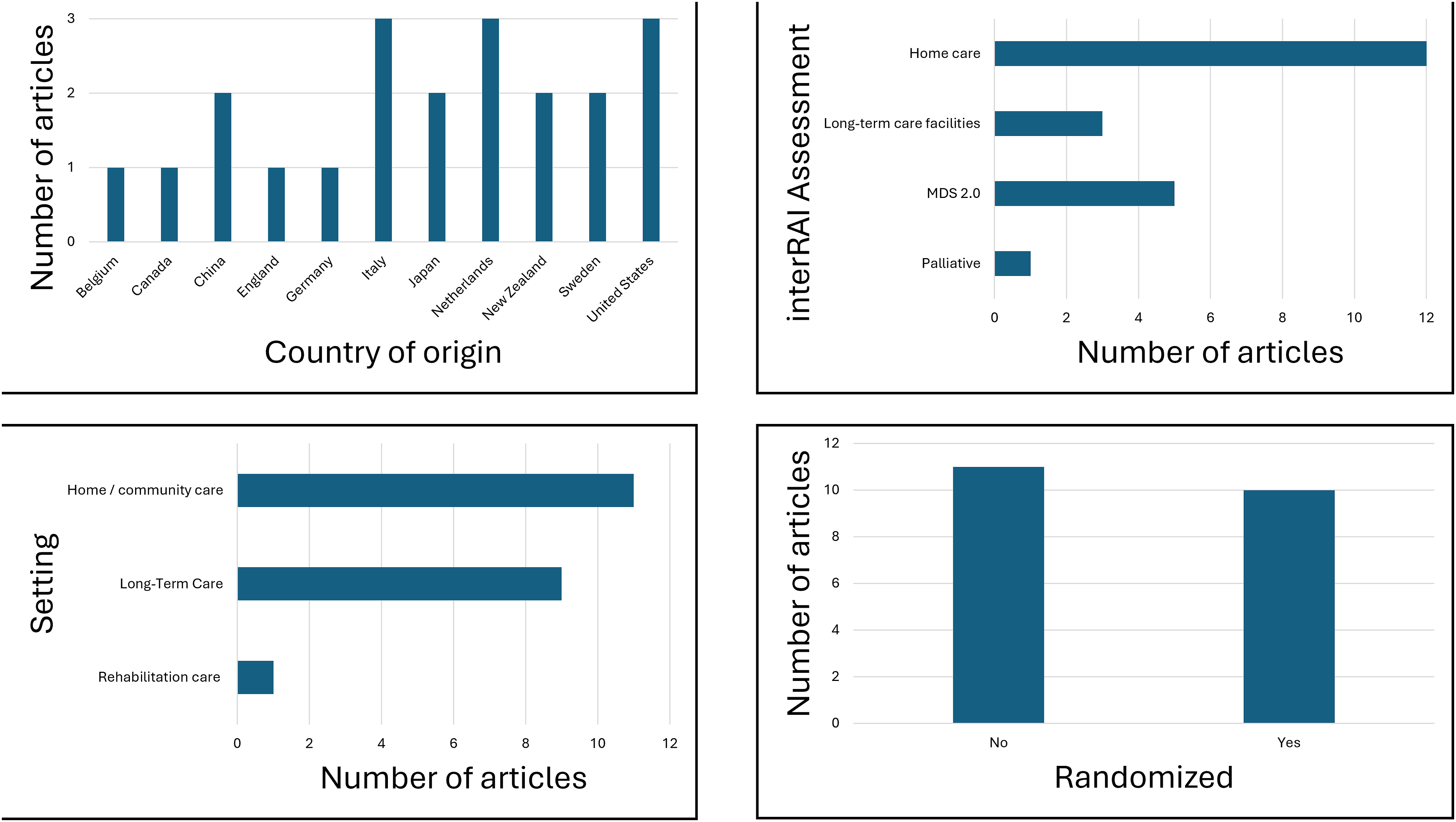
Interventions ranged from 6 to 156 weeks. Most interventions focused on comparing an interRAI instrument to another comprehensive geriatric assessment or routine care. Some interventions included a one-time assessment, with or without quarterly follow-up, while one study compared the success of an interRAI instrument to improve care management across distinct services (i.e., primary care vs. social). 94 Similar to the studies categorized as “efficacy,” participant-centred outcomes were the most commonly assessed.
Discussion
We conducted a scoping review to examine how studies of older adults (65+ years of age) deploy interRAI instruments either as intrinsic components of interventions (i.e., implementation) or as tools to evaluate interventions (i.e., efficacy) across healthcare settings (i.e., home, long-term, and acute). Among the 64 included manuscripts, we categorized more than two-thirds as “efficacy.” Regardless of categorization, we observed considerable heterogeneity across study elements, reflecting the broad applicability of interRAI instruments to patient assessment, care planning, and intervention delivery for diverse countries, populations, outcomes, and settings. Our findings indicate that interRAI instruments can effectively assess patient outcomes when evaluating interventions and serve as effective interventions for older adults across healthcare settings.
Seminal work from Gray and colleagues highlights the need to move toward a more integrated, person-centred assessment system. 105 Standardizing core data elements across care settings through interRAI information systems could reduce redundant assessments and improve continuity of care, ultimately creating a more efficient healthcare system. We found interRAI instruments to be successfully deployed as an intervention or to evaluate interventions in 17 different countries spanning 5 of the 6 inhabited continents. Further, interRAI was used across numerous settings, including long-term, home or community, acute, retirement village, and post-acute or rehabilitation care, with long-term and home/community emerging as the most common. Together, these findings suggest that the interRAI suite could form the foundation of an integrated clinical information system. In particular, the “core” interRAI items (i.e., those used in every interRAI instrument) would permit tracking of important outcomes for older adults as they move through healthcare sectors. Therefore, as the global population continues to age1,2 amid an already strained healthcare system,4,5 implementing an integrated clinical information system, facilitated by the mandated use of interRAI instruments, may represent the paradigm shift needed to enhance life quality while reducing healthcare resource utilization.
Stakeholders at all levels have a role in this paradigm shift, but policy-makers are uniquely positioned to drive it. Although many studies demonstrated pragmatic, local-level improvements, few provided evidence of system-wide scalability; systematically bridging from promising institutional gains to policy-level integration is a strength of interRAI information systems. Policy-makers can facilitate the paradigm shift by prioritizing the integration of interRAI into national health information strategies, establishing data sharing agreements within and across jurisdictions (however that is defined), and investing in training initiatives to build workforce capacity. Resources can be further optimized by emphasizing system metrics that are linked to interRAI information system outputs (e.g., quality indicators) and make a measurable difference, such as reduced/delayed institutionalization, improved life quality, and cost efficiencies. Despite the need for additional evidence, which will arguably always be a concern given interRAI’s versatility and the rapid evolution of the healthcare system,4,5 now is the time for change as the aging “grey wave” is upon us.
Numerous outcomes were assessed across the included studies, the majority being participant-centred; inherently, this makes sense, given interRAI’s overarching goal to maximize life quality. Consistent with the Geroscience hypothesis, 106 many of these outcomes are connected in that improving one often improves another. For example, previous work demonstrates that reducing pain can improve the ability to perform activities of daily living 107 and brain function. 108 Such relationships may, in part, reflect shared causal pathways, with oxidative stress, inflammaging, and mitochondrial dysfunction among the likely culprits. 109 As the population continues to age, interventions that simultaneously target and measure multiple participant-centred outcomes through robust information systems, like those supported by interRAI, will be essential for maximizing life quality while minimizing healthcare utilization.
A 2014 systematic review remains the only prior study to map the dual role of interRAI information systems as evaluation instruments for interventions and as interventions in their own right, but it focused exclusively on home care settings. 35 Other reviews have emphasized the utility of interRAI systems as outcome measures28-30 or compared interRAI to another established comprehensive geriatric assessment battery.31-34 Our findings align with the 2014 review in confirming that the interRAI information system is effective as an intervention and for evaluating interventions in home care settings. Yet, we extend the evidence base by highlighting interRAI systems across a broader range of older adult healthcare environments, thereby reinforcing their potential as the foundation for an integrated healthcare system.
Two major shortcomings emerged across the included studies, neither attributable to the instruments themselves, but rather to how and where they were deployed. First, a large proportion of studies did not clearly report the sex distribution of participants, and likely, even fewer reported sex-specific outcomes. A rapidly growing mass of literature suggests that males and females experience aging differently,18,20,110-113 with the latter often reporting worse outcomes.114,115 Thus, sex is an important consideration in the aging process. Second, all included studies originated from high- or upper-middle-income countries, with none from lower-middle or low-income countries. 116 Like all assessor-administered comprehensive geriatric assessments, interRAI information systems require substantial training and resources, barriers that limit feasibility in under-resourced settings. Some interRAI information systems, such as the “Check-Up Self Report,” have been used in low-income countries, 117 but there remains an urgent need to improve global equity in geriatric assessment. 118
This is the first scoping review to map how interRAI information systems have been used as, or within, interventions for older adults across healthcare settings; however, it is not without shortcomings. Our broad inclusion criteria contributed to the between-study heterogeneity, but in doing so, it emphasized a key takeaway: the interRAI information system is versatile enough to support an integrated healthcare system for the global aging community. Along these lines, search engines and strategies, despite being powerful, have inherent limits. Our initial search yield (n = 11,446) was inflated by “minimum dataset,” a broad sweeping term that captured many irrelevant studies. With only 64 studies meeting the review criteria, it suggests less than 1% of the sample size was included, when in reality, we faced the common challenge of every systematic search strategy: sensitivity vs specificity. Despite the large number of reports initially included, we cannot exclude the possibility that some literature was missed; this is especially true considering we excluded grey literature, which may provide particular insights for health policy. Likewise, we excluded studies that conducted a secondary or retrospective analysis of existing data, instead focusing on prospective intervention studies; anecdotally, this resulted in the exclusion of hundreds of studies. Finally, while our findings may inform the broader field of comprehensive geriatric assessments, they should not be generalized beyond interRAI instruments. In addition to addressing some of the noted limitations, future work should prioritize prospective studies examining the “implementation” and “efficacy” of interRAI, with a clear focus on how they deliver measurable improvements to the quadruple aim (i.e., better care, better health, lower costs, and happier providers).
This scoping review mapped how interRAI instruments have been used both to evaluate interventions and as intrinsic components of interventions. Our results emphasize the versatility of interRAI systems and their potential to advance efficient, person-centred care across countries and settings, particularly within home/community and long-term care in upper-middle and high-income nations. Accordingly, interRAI instruments can serve as the backbone of an integrated clinical information system. To do so, policy-makers should strengthen digital systems, ensure health records can connect across settings, and train staff to use interRAI effectively. This review serves as a valuable foundation for future research exploring novel intervention strategies with or through the interRAI information system—an urgent priority in the context of a rapidly aging population and shifting healthcare landscape.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to Alison Farrel for her invaluable assistance in developing the search strategy for this scoping review. Alison’s expertise and guidance were essential to the success of this project. We would also like to thank Drs. Bon Egbujie and Veronique Boscart for reviewing the manuscript prior to submission for publication.
Ethical Approval
Institutional review board approval was not required.
Author Contributions
NWB: conception and design, writing—original draft, and final approval of published version; SM: conception and design, writing—review and editing, and final approval of published version; NN: conception and design, writing—review and editing, and final approval of published version; JFY: conception and design, writing—review and editing, and final approval of published version; IB: conception and design, writing—review and editing, and final approval of published version; EB: conception and design, writing—review and editing, and final approval of published version; GH: conception and design, writing—review and editing, and final approval of published version; JAM: conception and design, writing—review and editing, and final approval of published version; KS: conception and design, writing—review and editing, and final approval of published version; CM: conception and design, writing—original draft, and final approval of published version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NWB is supported by a Canadian Institutes of Health Research Fellowship (FRN: 489847) and the Health Data Research Network Canada Pragmatic Trials Training Program—Postdoctoral Fellowship Stream. CM received funding from the Health Data Research Network Canada Pragmatic Trials Training Program and a Canadian Institutes of Health Research Project Grant.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
