Abstract
Shared Care Models (SCMs), in which a team of clinicians share in patient care and resource utilization, represent an opportunity for surgeon-level system change. We aimed to identify the queues and stakeholders within a complex gastrointestinal surgical care pathway to demonstrate the implications of a SCM on system efficiency. A multidisciplinary group of surgeons and care navigators working in SCMs were asked to develop a patient encounter map through consensus to illustrate relevant queues and stakeholders within a SCM. Fifteen surgeon-related queues were identified, each representing a point of potential delay to care in the patient’s journey that could be addressed by shared care. A final patient encounter map was created, and advantages and challenges of SCMs were also described from multidisciplinary group discussions. The numerous queues identified in this map ultimately reflected opportunities for more efficient care navigation under a SCM through increased surgeon availability and shared resource utilization.
Introduction
Providing timely access to surgical care is an ongoing challenge for patients, providers, and healthcare leaders.1,2 Compounded by lockdowns and staff shortages, almost 600,000 fewer surgeries were performed in the first two years of the COVID-19 pandemic compared to 2019 in Canada. 3 One potential strategy to reduce the surgical backlog is Single Entry Models (SEMs), in which patients are triaged based on acuity and priority, and subsequently directed to the next available provider. 4 Although SEMs can improve timeliness, 5 wait times to consultation only represent the initial bottleneck within the continuum of surgical care. Other surgeon-related bottlenecks that patients queue for include those that depend on the surgeon availability and their allotted resources including clinic visits, endoscopy, and post-operative follow-up appointments. Outside the surgeon-related care pathway, bottlenecks such as queueing for imaging, consultations with other specialists, and adjuvant therapies also contribute to access issues that are beyond the individual surgeon’s control.
Surgical care manifests as a Complex Adaptive System (CAS) that is dynamic where pathways are not always predictable and simple to navigate. 6 In this context, large scale system change may seem out of reach, and a more practical consideration is to identify opportunities at a micro- (individual surgeons) or meso- (clinical care programs) level. Such an example is Shared Care Models (SCMs), or sometimes referred to as group practices, in which a team of clinicians with similar practice profiles collaborate to care for a target group of patients using shared resources. 7 While there is broad acceptance of care by a group of interchangeable clinicians in some disciplines (family medicine, obstetrics, and acute care surgery),8-12 experience in complex elective surgical practice is limited. Concerns around feasibility, patient experience, and erosion of patient trust remain firm and contribute to resistance to wider adoption of SCMs. 13
On the other hand, traditional solo surgeon models, in which each surgeon receives their own referrals and is solely responsible for the entirety of the patient’s surgical pathway, may cause delays in surgeon-related bottlenecks, increase provider isolation, and complicate care coordination.7,14 Nevertheless, many surgeons continue to work in solo practices, 15 and although SCMs exist sporadically across Canada, translational experience in transforming solo models into SCMs is limited. Similarly, clinical guidelines such as those from the National Comprehensive Cancer Network (NCCN) and Cancer Care Ontario (CCO) depict surgical care delivery as a linear series of evidence-based diagnostic and treatment steps which guide clinical decision-making, but they may omit a systems level view of the additional queues that complicate the patient’s care journey.16,17
If more SCMs are to be operationalized, it is essential to understand how centralization of resources and personnel can impact the patient journey, both positively and negatively, particularly in tertiary centres in which multiple sub-speciality providers are encountered. Previous studies have described one specific general surgery SCM at an academic tertiary centre that has not been formally mapped from the perspective of a CAS.7,18 Through evaluating this model, this study aimed to identify the queues and stakeholders within a complex Gastrointestinal (GI) surgical care pathway to demonstrate the potential implications of a functional SCM on system access and efficiency.
Methods
Setting: The shared care model
The study followed the Consolidated Criteria for Reporting Qualitative Research (COREQ) guidelines. 19 This study was conducted at a tertiary centre home to a colorectal surgery unit, a HPB surgery unit, and two affiliated community surgery programs, all functioning under a SCM. 7 Together, these sites serve a population of more than two million people living across both rural and urban communities and are part of an integrated regional cancer program in the province. Patients are referred through a SEM and are subsequently cared for by the next available surgeon at each encounter. This means that the surgeon who sees the patient in consultation may not be the same one who sees the patient for follow-up visits, operates on them, and so forth. These four units were used as a model to map the queues and stakeholders encountered in a SCM in complex elective GI surgery.
Mapping patient queues and stakeholders in a shared care models
Example case and preliminary list of queues and stakeholders encountered in a patient’s journey in a complex GI surgery SCM.

This case was used to develop a preliminary list of sequential, surgeon-related queues, each representing an encounter by the patient who enters a surgical SCM (Table 1). The initial list was generated based on the clinical experience of the research team, consisting of general surgeons who work in community, colorectal, and HPB SCMs. Alongside each queue, relevant stakeholders who were thought to be core members of the SCM were listed. The NCCN and CCO clinical guideline maps for rectal cancer informed the gold standard diagnostic and treatment steps that the example patient would undergo.16,17 These guidelines are developed from the best evidence available from research and expert opinion and are being used to guide management at the study centres.
Six individuals practicing in SCMs but outside of the research team were recruited via purposeful sampling to participate in three sessions over the course of two months to develop the map. This group included three surgeons (community, colorectal, and HPB surgery) and three patient care navigators (community surgery administrative assistant, colorectal, and HPB nurse navigators).
A total of three sessions with the multidisciplinary group were held. The first session was completed off-line, and participants were asked to review four different materials that were sent via e-mail on their own over a week time: (1) study rationale, (2) patient case of rectal cancer and liver metastases, (3) preliminary list of surgeon-related queues and stakeholders, and (4) NCCN and CCO maps. They were asked to reflect on the accuracy of the provided list of queues and stakeholders, while considering additional ones that participants thought were relevant as well as those that did not belong based on their experiences. The NCCN and CCO maps were provided to inform about the standard guidelines for clinical management.
The second session was held virtually on an on-line video-call platform (Microsoft Teams version 1.5.00.21551) in a group setting for an hour. A meeting facilitator, who was not directly involved in patient care in SCMs presented the project’s rationale and moderated the discussion. Each participant was serially asked to share their thoughts on the preliminary list and suggest edits until consensus was reached. They were also asked to provide their thoughts regarding how a SCM may impact a patient’s navigation through a surgical system. The video call was recorded with permission and transcribed for analysis.
A preliminary SCM patient encounter map was then created using process mapping based on the results from the second session (Microsoft PowerPoint version 16.65) and with guidance from previously described methodology.20,21
A third and final session was also held virtually with the multidisciplinary group. The map was reviewed to ensure final consensus around the appropriate surgeon-related queues and stakeholders, and suggestions for any changes were discussed. These also included changes to the visual aspects of the map and the level of detail appropriate to depict the SCM. The session was also recorded and transcribed for analysis. Changes were applied and used to build the final patient encounter map.
Transcriptions from both video calls were reviewed by two research members to confirm consensus and to gather themes regarding the stated benefits and challenges of SCMs. A list of quotes from the participants reflecting these benefits and challenges was developed.
Results
Session 1 and 2
Following the first session, three additional queues were recommended for a total of 15 queues (Figure 1). These included queues for a post-endoscopy follow-up visit, colorectal surgery Multidisciplinary Cancer Conference (MCC), and a HPB MCC. These were considered common and critical steps during the care for patients with rectal cancer and synchronous liver metastases. Additionally, participants expressed that patients may navigate extra queues from unplanned encounters (managing complications and completing insurance forms). To represent this, an overarching queue spanning the entire patient journey for unplanned surgical services was added. Participants also recognized that standard investigations were almost always ordered as part of the triaging process before surgical consultation. Thus, “ordering investigations” was combined with each “triaging consultation” queue. Lastly, stakeholders most responsible for triaging were identified as either administrative assistants in community hospitals or nurse navigators in larger academic centres, while surgeons may or may not be involved in this step. Thus, the term “queue navigator” was introduced for queues related to triaging consultations to refer to all of these stakeholders. Patient encounter map and final list of surgeon-related queues and stakeholders in a complex GI surgery SCM. BRBPR bright red blood per rectum; Gen Sx general surgery; Ix investigations; CR Sx colorectal surgery; HPB hepatopancreatobiliary surgery.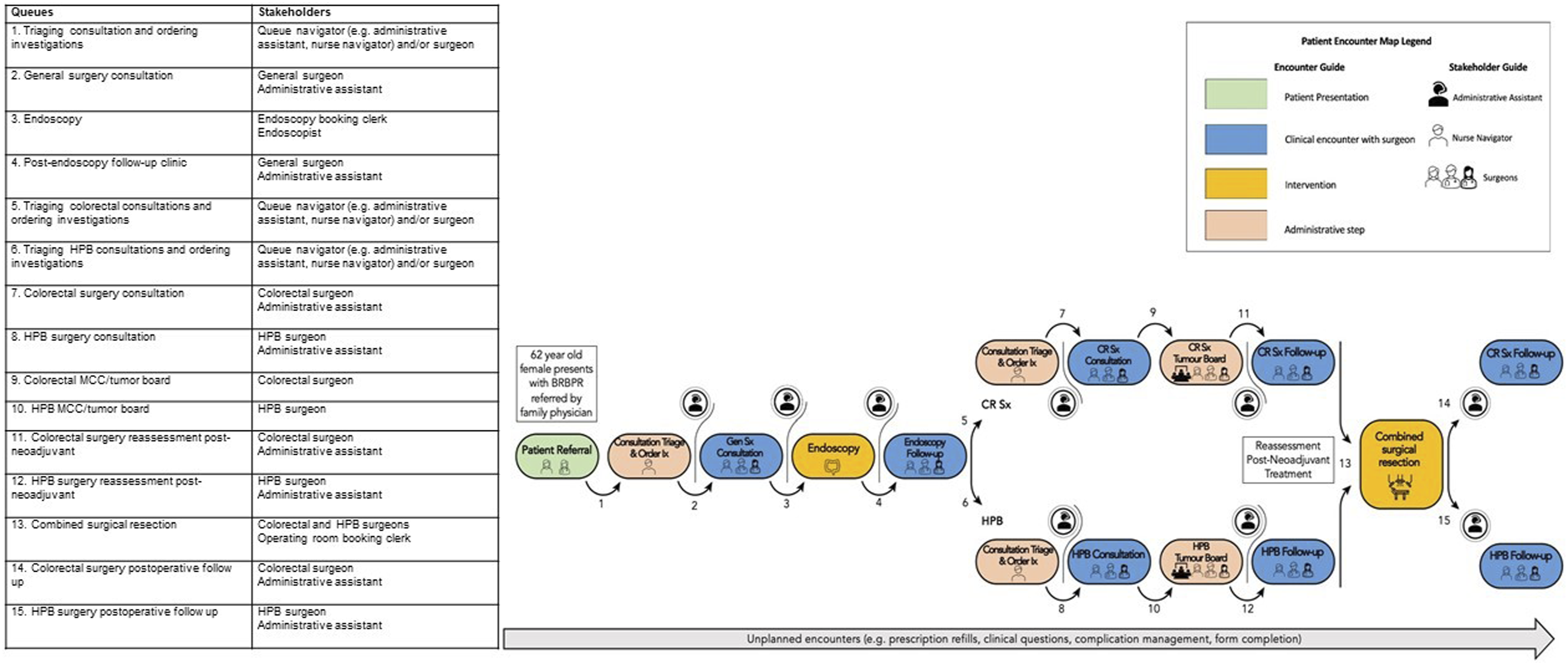
Session 3
In the third session, the final list of queues and stakeholders was unanimously approved. For the map, participants agreed that a visually engaging colour and pattern scheme to represent different types of queues (such as clinical encounters, administrative steps, and interventions) should be used. The importance of distinguishing the concept of queueing from actual encounters was raised. Whereas each box represented a surgeon-related encounter, queueing refers to the act of “waiting in line,” which contributes to access delays and inefficiencies. Queueing was therefore represented as the arrow preceding an encounter. Consensus for these edits was achieved, and the final patient encounter map was established (Figure 1).
Participant comments regarding benefits and challenges of shared care in surgery.

Discussion
This study evaluated a fully implemented SCM to illustrate the true number and nature of additional queues which a patient encounters in a CAS, and the potential benefits of SCMs in system efficiency. It follows that every stakeholder and additional queue has the potential to introduce bottlenecks if the patient is waiting for one individual rather than a team of individuals.
Fifteen individual queues were identified based on the example colorectal liver metastases patient case. Existing clinical guideline maps (e.g. NCCN and CCO) describe a useful pathway for diagnosis and treatment, yet may underestimate the complexity of navigating such care. For example, the CCO map for stage IV rectal cancer depicts only one wait time for management with curative intent. 17 Likewise, much of the literature around timeliness to surgery have focused on wait times to the initial consultation (“wait one”) or surgery (“wait two”).5,22,23 However, patients can experience wait times to be much more complicated and protracted, beginning with the onset of symptoms to the multiple preoperative assessments that increase time to definitive treatment. Accordingly, novel patient-centred wait times that take into consideration preoperative consultations and institutional factors such as surgical volume and geographic location may be able to better reflect the patient experience. 24
In their ideal form, SCMs create a consistently available point of contact (administrative and clinical), and therefore streamline these encounters, shortening, or potentially eliminating, bottlenecks. This is especially true for unpredictable queues (e.g. clinic visits for complications), illustrated in our model using the overarching queue arrow. The advantages of SCMs to timeliness and equity in care have theoretical and mathematical groundwork based on queueing theory. Specifically, solo surgeon practices represent a multiple line queueing model, and SCMs represent a single line queueing model, consisting of a single queue that feeds into many identical providers. 25 Compared to queueing under separate solo surgeons, the configuration of a SCM guarantees fairness while being the most efficient set-up for minimizing time in the queue. Given that a single list of patients wait under multiple collaborative surgeons, SCMs may also increase equity in resource use (e.g. operating room access) and referral patterns that reduces misallocation of patient demand and supply. 26 In practice, these systems have been proposed as a solution to address current healthcare disparities, such as in male vs. female surgeon referral patterns.27,28
A common challenge noted in the discussion of SCMs was the degree of trust and communication among the team and organization, which can especially be specific to the institution’s culture. A collaborative environment with supportive leadership results in a SCM in which a patient’s individual trust can instead be transferred from one surgeon to a team of surgeons. 7 Prior studies also suggest that SCMs have the potential to improve job satisfaction and work-life balance, decrease professional isolation, and improve knowledge sharing.12,29,30 This potential can best be met when a shared sense of ownership and vision is supported.11,31 This does not necessarily exclude variability among the team members such as in technical or nontechnical skills. In fact, we note that individual abilities were best supported in this institution when fostered as part of the team’s collective capacity. Quality of care can be improved through better knowledge sharing and collective adherence to guidelines, while creating a naturalistic mechanism for clinical second opinions.32,33 Other traditional concerns around SCMs revolve around loss of continuity of care and erosion of patient trust, yet there is evidence that patients accept shared care in the context of improved access while having confidence in any of the surgeons within a group.8,13,18
Limitations
To our knowledge, this is the first study to formally map a SCM in a CAS. Several limitations exist in our study. First, we used a specific case scenario that is not representative of all surgical cases, some of which may involve fewer queues. However, we aimed to evaluate a complex surgical journey, and the high-intensity and multidisciplinary nature of management for resectable metastatic rectal cancer was appropriate to demonstrate how SCMs may help patients effectively navigate a CAS. Second, discussions involved a focus group of multidisciplinary providers practicing in existing SCMs, which may bias some of the findings. To mitigate this, these sessions were moderated by research members outside of a SCM to gather more genuine perspectives around SCMs, and the study findings were anonymized. Lastly, the resulting patient encounter map does not illustrate queues that are outside the surgeon’s control, such as waiting for imaging and other specialist consultations. The aim, however, was to identify queues that are within the surgeon’s reach for system change and may nevertheless improve care coordination.
Conclusion
Although this study is not a direct comparison of SCMs vs. traditional solo surgeon practices, it provides insight into the potential advantages of a centralized queueing system along surgeon-related care pathways using a real-world complex surgical shared care practice. In order to extend translational experience in shared care, the process map will be used to help guide future studies that evaluate operational and clinical feasibility. Current studies underway include an outcome analysis of patients undergoing complex GI surgery as well as qualitative studies to evaluate the learner experience within a SCM. Likewise, we intend to study the patient experience spanning their preoperative, in-patient, and post-operative care beyond simple surveys which have thus far been performed. Ultimately, these efforts can help surgeons and patients navigate the complexity of surgical care through a more modern and collaborative approach.
Supplemental Material
Supplemental Material - Mapping a shared care model in complex gastrointestinal surgery: A qualitative study of queues and stakeholders within a Canadian general surgery practice
Supplemental Material for Mapping a shared care model in complex gastrointestinal surgery: A qualitative study of queues and stakeholders within a Canadian general surgery practice by Alex Lee, Janelle Rekman, Tyler Lamb, Morgann Reid, Luckshi Rajendran, Anastasia Turner, Husein Moloo and Fady Balaa in Healthcare Management Forum
Footnotes
Acknowledgements
We would like to thank all those who participated in our study to provide meaningful insight into SCMs.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the institution’s research ethics board (OHSN-REB 20210326-01H).
Supplemental material
Supplemental material for this article is available on-line.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
