Abstract
Keywords
Introduction
Advance care planning (ACP) aims to respect patient autonomy, improve quality of care, strengthen care relationships, prepare for end-of-life care and reduce overtreatment. 1 ACP could be initiated regardless of age or disease stage 2 or as soon as a patient is confronted with illness or frailty. 3 For effective ACP, structured documentation is recommended.4,5 This prevents poor communication between healthcare professionals, and improves availability of information in the electronic patient record (EPR).6,7 Healthcare professionals can regularly review and actualize preferences when circumstances change. 5
Current end-of-life care often does not meet the patient's preferences.8–10 Some patients receive aggressive treatment in the last months of life. This may lead to high symptom burden11,12 and unnecessary care transitions at the end of life. 8 Internationally, patients often do not die at their preferred place of death.9,10,13,14 In oncology, indicators of aggressive end-of-life treatment include chemotherapy, multiple emergency room (ER) visits, hospital admission including the intensive care unit (ICU), prolonged hospitalization and in-hospital deaths.15–17 Previous studies found that discussing end-of-life preferences with oncology patients was related to less aggressive end-of-life treatment.11,18 In addition, discussing ACP preferences may improve concordance between preferred and delivered care, the quality of communication,19,20 and may lead to increased home deaths. 21
Several studies showed that oncology patients want to talk about end-of-life care.22–24 ACP discussions are also valuable for patients with a curative treatment intention, 2 especially when they are frail or have comorbidities. Timely discussing and documenting preferences in a structured manner may contribute to more appropriate care in oncology. 25 Yet, structured ACP documentation is performed in only a small percentage of oncology patients.22,23–25 Our pilot study among older oncology patients (≥80 year) suggested that timely documentation of the preferred place of care or death in an ACP tool (ACPT) within the EPR was associated with fewer diagnostic tests, more deaths at home and less in-hospital deaths, compared to patients without an ACPT. However, this pilot involved a small and specific sample which was insufficient to generalize conclusions. 25
The current study describes the frequency of ACPT implementation in oncology practice and to what extent relevant information regarding ACP is described in the EPR. Next, this study assessed the association of complete ACPT(cACPT) registration with a) hospital care consumption, and b) the match between preferred and actual place of death, among adult oncology patients within a larger cohort and a broader age range. We hypothesized that structured documentation of patients’ preferences in an ACPT regardless of patients’ age or disease stage, is associated with less hospital care consumption, especially regarding the use of chemotherapy in the last month of life, and more deaths at the preferred place.
Methods
Design
A retrospective observational cohort study was conducted among deceased oncology patients aged 18 years and above who received treatment in a Dutch teaching hospital. Patients with a timely initiated ACPT including preferred place of care or death (ie, cACPT) were selected and compared with a similar number of randomly selected patients without ACPT.
Study Population
The study population was selected from the EPR, using the following criteria: (1) diagnosed with cancer, (2) aged 18 years and above, (3) treated by a (hemato-) oncologist, and (4) died between 1-1-2019 and 31-10-2021. Patients who registered an objection to using their data for research were excluded. Patients who were not registered as deceased in the EPR were checked for death by using municipality data. Patients who met the inclusion criteria were divided based on the presence of a cACPT, the presence of an incomplete ACPT and the absence of an ACPT. Patients with a cACPT were classified as patients having an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was initiated at least three months before death. Patients with an ACPT without the criteria timely or documented preferred place of care or death were classified as patients with an incomplete ACPT. These definitions were based on the results of our pilot study. 25
The inclusion period was selected to meet the required number of patients with a cACPT (see statistical analyses). For all patients without an ACPT during the period of inclusion, a random number was generated using Excel, and thereafter ordered on the assigned number (from low to high). The required number of patients was included top-down. The remaining patients were excluded.
Advance Care Planning Tool
The ACPT (see Supplemental Material) was developed by field experts of 15 Dutch hospitals and national palliative care network partners. The ACPT is based on the national quality framework for palliative care, 26 and integrated in the EPR in many Dutch hospitals. It covers the four dimensions of palliative care addressing physical, psychological, social, and spiritual needs. It contains information on the social context (ie, living situation, marital status) and values of patients (ie, what is important). Particularly, the ACPT focuses on the organization of care (ie, patients’ preferred place of care and death) and decision-making (eg, wanting to be readmitted to hospital).
Data Collection and Outcome Measures
The percentage of patients for whom an ACPT was initiated, regardless of the items documented, was calculated among all deceased patients. Data were extracted from the EPR. Relevant information about social context (eg, marital status, residence, presence of an informal caregiver) was also extracted from the ACPT, and, in case of patients without an ACPT, from correspondence letters or reports written in the last three months of life. Data were coded as ‘yes’ or ‘no’ and if relevant further specified.
Types of hospital care consumption variables at the end of life were determined on relevant literature.8,27–29 The primary endpoint was receiving systemic anti-tumor treatment. This variable was coded as ‘yes’ or ‘no’ and specified if relevant. Secondary endpoints were: treatment with radiotherapy, surgery requiring anesthesia, contacts within the hospital setting, diagnostic tests, palliative interventions and the match between the preferred and actual place of death. These variables were coded as ‘yes’ or ‘no’ and if relevant, specified with frequencies, length of stay, type of diagnostic tests and palliative interventions.
All hospital care consumption variables were manually collected for the last three months of life and the last month of life, respectively. To obtain a reliable dataset part of the care consumption variables were also retrieved digitally and checked for consistency. In case of inconsistency the EPR was consulted again (CB). The outcomes gathered manually were considered leading because digital extraction of variables sometimes was not feasible. For example, endocrine therapy was not always registered digitally by the healthcare professional, while the EPR showed that the patient used it.
Demographics and other baseline characteristics were gathered and were specified as age in years, gender, type of cancer, time treated by the (hemato) oncologist and treatment intention.
Statistical Analyses
Based on the pilot study, 25 we expected 10% of patients to receive systemic anti-tumor treatment in the last month of life in the cACPT group versus 20% in the group without an ACPT. To detect this difference a sample size of 214 patients per group was calculated (power > 80%, alpha < 0.05). A maximum of eight independent variables was added to the logistic regression model based on the rule of thumb to include at least 10 events per variable. 30 A P-value < .05 was considered statistically significant. Analyses were conducted in SPSS version 27.
The categorical variables were described using frequency tables and percentages, and numerical variables by using medians and (interquartile) ranges. Differences between patients with a cACPT and without an ACPT were tested with Chi-square tests for categorical variables, and with Mann-Whitney U tests for numerical variables. To assess the association between the use of a cACPT and systemic anti-tumor treatment in end-of-life care, multivariable logistic regression analysis was carried out. Adjusted Odd's Ratio (OR) with 95% confidence interval (C.I.) was calculated with systemic anti-tumor treatment, and for all secondary hospital care consumption variables, three and one month(s) before death, as dependent variable and the presence of a cACPT as independent variable. Confounders were added to each model separately. Age was modelled as a continuous variable and gender, type of cancer, time treated by the (hemato) oncologists as categorical variables. Variables were kept in the model as confounding factor, if the regression-coefficient of the association between the presence of a cACPT and outcome changed by more than 10%. 30
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki (version 7, October 2013) and approved by the Institutional Review Board of the Jeroen Bosch Hospital (2020.04.22.02, approved at June 10th 2020).
Results
Participants
In total, 793 patients had an ACPT of whom 216 patients had a cACPT (group 1) and 577 patients an incomplete ACPT. 216 patients were randomly selected from the group patients without an ACPT (n = 437, group 2) (Figure 1). Thirty patients were referred to another hospital in their last months of life (n = 2 in group 1, n = 28 in group 2). For a few of these patients, information about systemic anti-tumor treatment three months before death (n = 14 in group 2) and one month before death (n = 1 in group 1, n = 16 in group 2) was missing. For none of the referred patients (n = 30), information could be gathered about secondary hospital care consumption.
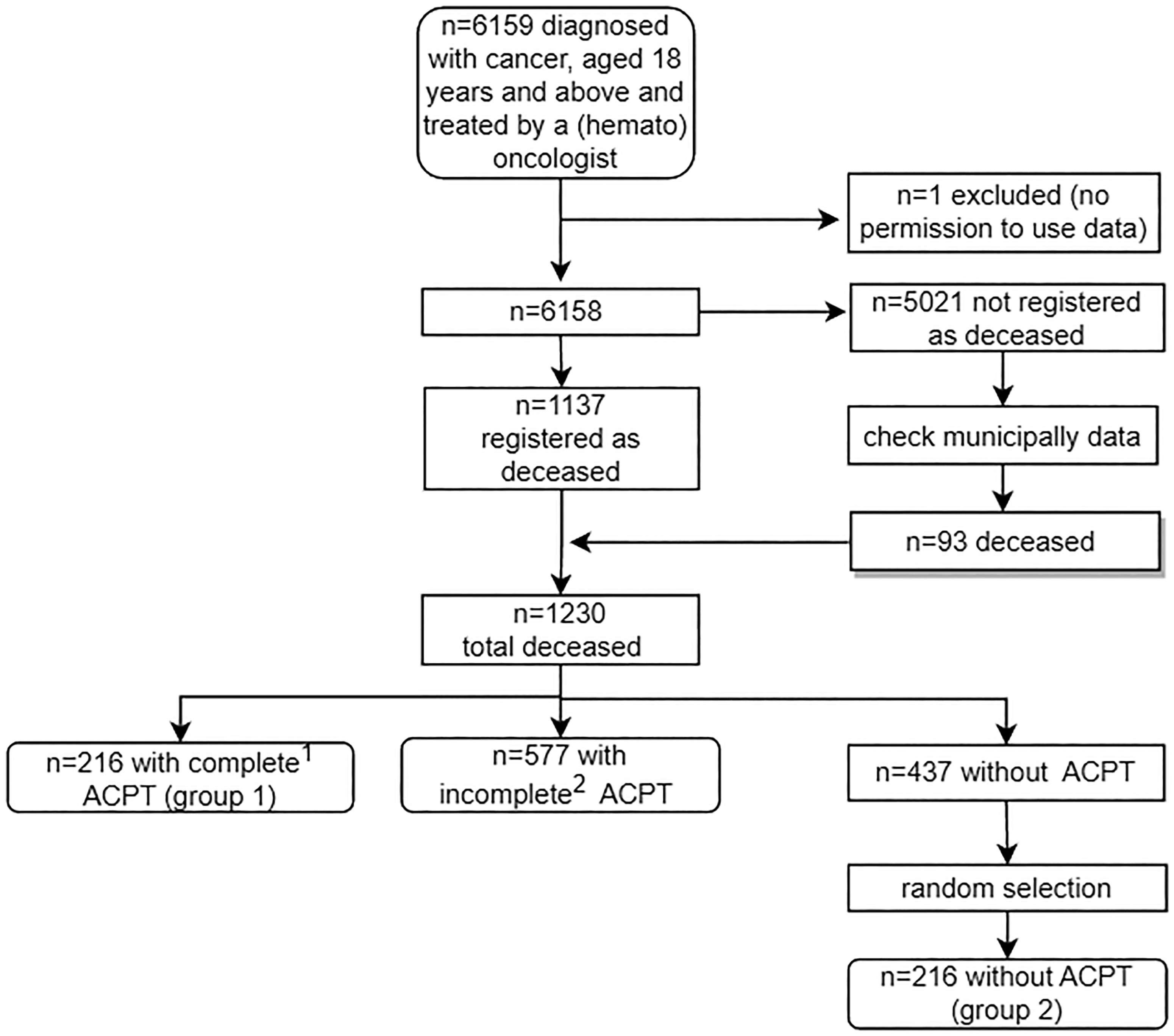
Patient Inclusion Procedure. Note: ACPT = advance care planning tool. 1complete ACPT: an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was initiated at least three months before death. 2incomplete ACPT: an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was not initiated at least three months before death or not documented at all.
ACPT Implementation and Relevant Information Regarding ACP Documented
The ACPT was initiated in 64.5% (n = 793) of all deceased patients (n = 1230) (Figure 1). In 17.6% (n = 216), the patients’ preferred place of care or death was documented at least three months before death (cACPT). The ACPT was often initiated in an outpatients’ clinic setting (n = 176, 81.5%), which also applied for documentation of the preferred place of care or death (n = 175, 81.0%).
Regarding the two study groups (cACPT and no ACPT), information about the social context was well documented in patients with a cACPT. Information about the presence of an informal caregiver and treatment intention were frequently not documented in both groups (Table 1).
Information Documented in the EPR During the Last Three Months of Life of Patients with Complete ACPT and Without ACPT.

Note: Group 1: patients with an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was initiated at least three months before death; Group 2: patients without an ACPT.
(ACPT = advance care planning tool).
(Residence: not at home is defined as living with family/friends, nursing home, hospital, hospice or elsewhere).
Table 2 reports patient demographics for both groups. Groups differed based on age and tumor type.
Patient Demographics.
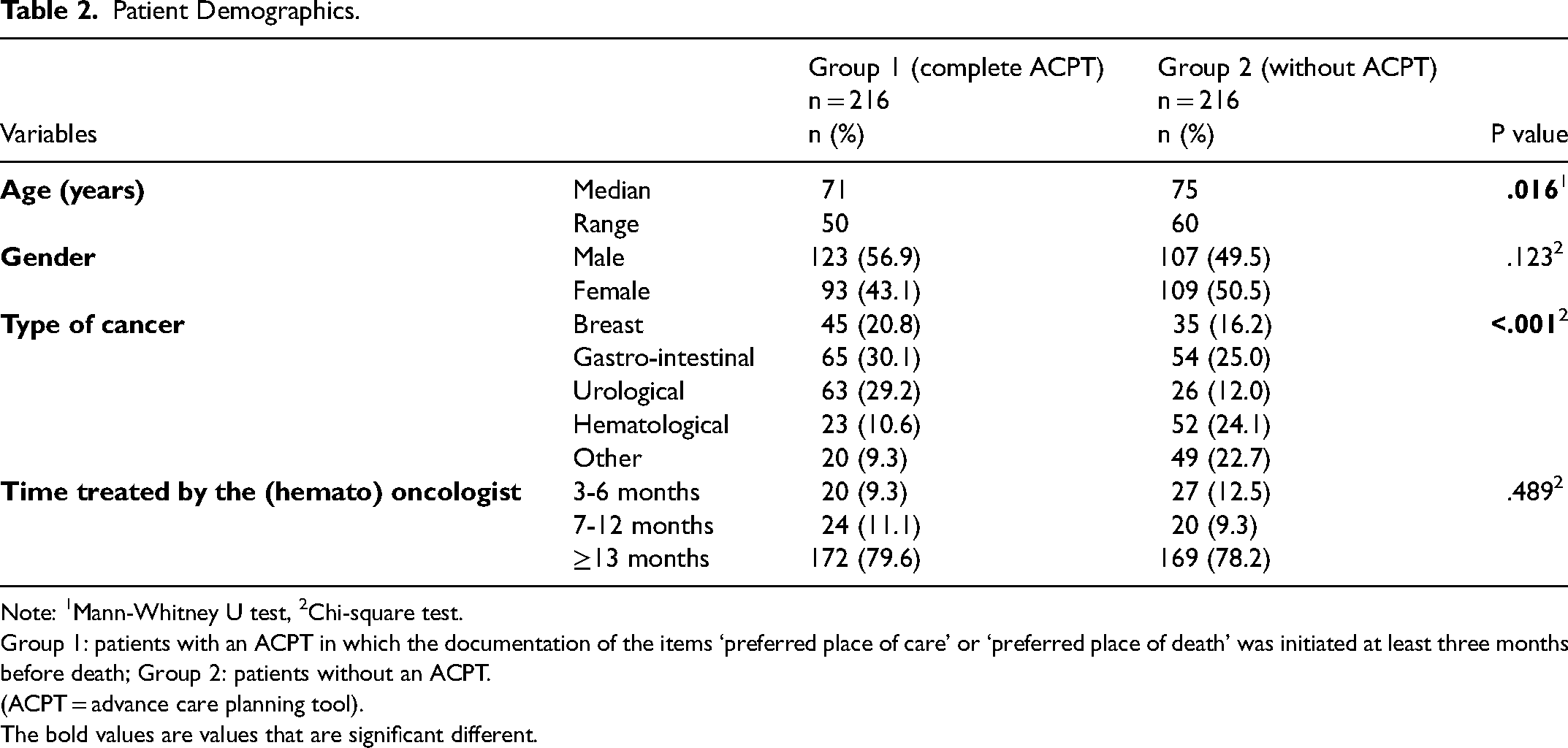
Note: 1Mann-Whitney U test, 2Chi-square test.
Group 1: patients with an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was initiated at least three months before death; Group 2: patients without an ACPT.
(ACPT = advance care planning tool).
The bold values are values that are significant different.
Confounding Factors
All statistical analyses investigating the association between the presence of a cACPT and the outcomes showed that age and tumor type were confounding factors (regression-coefficient changed by more than 10%), unlike gender and the time treated by a (hemato) oncologist.
Systemic Anti-tumor Treatment
No association was found between a cACPT and systemic anti-tumor treatment, three (Table 3) and one month(s) before death (Table 4). However, when specified for type of systemic anti-tumor treatment, patients with a cACPT received significantly less endocrine therapy three months before death than patients without an ACPT (Table 3).
Comparison Between Patients with a Complete ACPT and Without an ACPT of Hospital Care Consumption During the Last Three Months of Life.
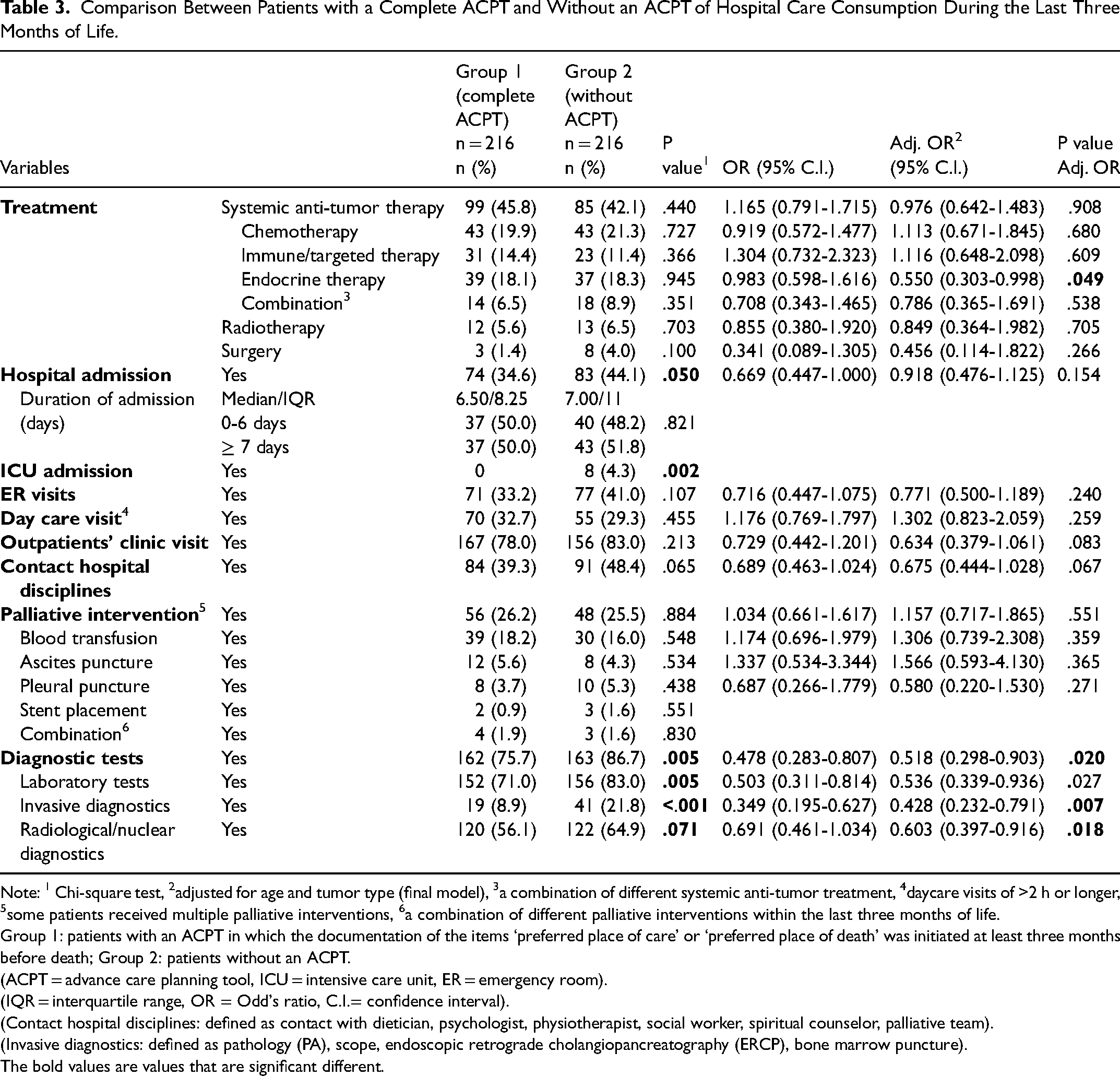
Note: 1 Chi-square test, 2adjusted for age and tumor type (final model), 3a combination of different systemic anti-tumor treatment, 4daycare visits of >2 h or longer, 5some patients received multiple palliative interventions, 6a combination of different palliative interventions within the last three months of life.
Group 1: patients with an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was initiated at least three months before death; Group 2: patients without an ACPT.
(ACPT = advance care planning tool, ICU = intensive care unit, ER = emergency room).
(IQR = interquartile range, OR = Odd's ratio, C.I.= confidence interval).
(Contact hospital disciplines: defined as contact with dietician, psychologist, physiotherapist, social worker, spiritual counselor, palliative team).
(Invasive diagnostics: defined as pathology (PA), scope, endoscopic retrograde cholangiopancreatography (ERCP), bone marrow puncture).
The bold values are values that are significant different.
Comparison Between Patients with a Complete ACPT and Without an ACPT of Hospital Care Consumption During the Last Month of Life.
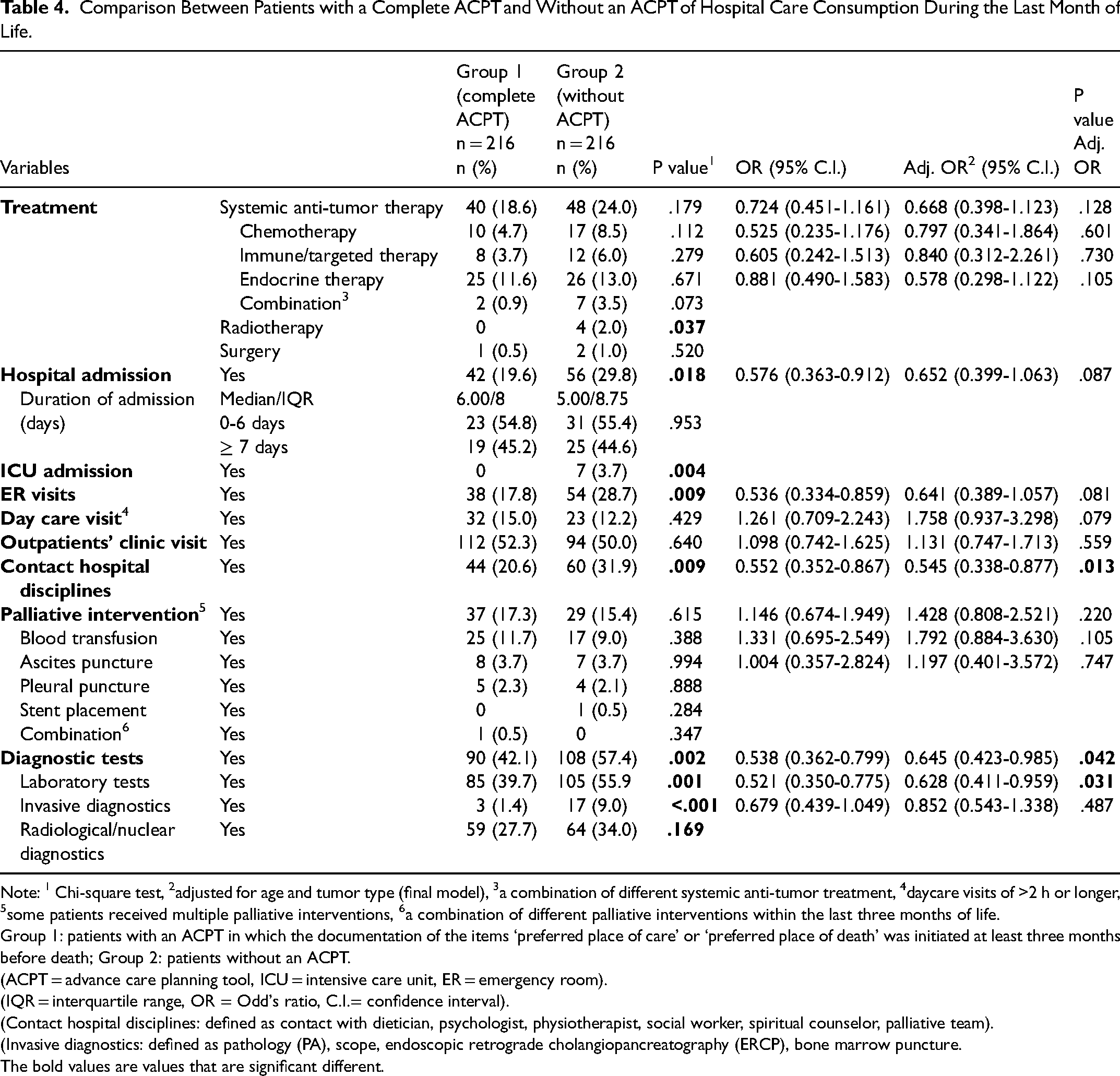
Note: 1 Chi-square test, 2adjusted for age and tumor type (final model), 3a combination of different systemic anti-tumor treatment, 4daycare visits of >2 h or longer, 5some patients received multiple palliative interventions, 6a combination of different palliative interventions within the last three months of life.
Group 1: patients with an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was initiated at least three months before death; Group 2: patients without an ACPT.
(ACPT = advance care planning tool, ICU = intensive care unit, ER = emergency room).
(IQR = interquartile range, OR = Odd's ratio, C.I.= confidence interval).
(Contact hospital disciplines: defined as contact with dietician, psychologist, physiotherapist, social worker, spiritual counselor, palliative team).
(Invasive diagnostics: defined as pathology (PA), scope, endoscopic retrograde cholangiopancreatography (ERCP), bone marrow puncture.
The bold values are values that are significant different.
Other Hospital Care Consumption
Patients with a cACPT less frequently underwent diagnostic tests in general; specifically, significantly fewer laboratory tests and invasive diagnostics, three and one month(s) before death (Tables 3 and 4), and fewer radiological/nuclear diagnostics three months before death (Table 3). They had also significantly less contacts with hospital disciplines one month before death compared to patients without an ACPT (Table 4).
Place of Death
In patients with a cACPT, the preferred and actual place of death were known more often compared to patients without an ACPT. If the preferred place of death was known for patients without an ACPT, this information was not documented in a structured way into the EPR. Among patients with documented actual place of death, patients with a cACPT died at home more often (67.8% vs 50.5%) and had less in-hospital deaths (11.0 vs 35.5%) compared to patients without an ACPT (Table 5). In patients with a cACPT, a match between preferred and actual place of death was found for 83% of the patients for whom the relevant information was available. In patients without ACPT, this information was mostly missing.
Documented Information About Preferred and Actual Place of Death in the EPR.
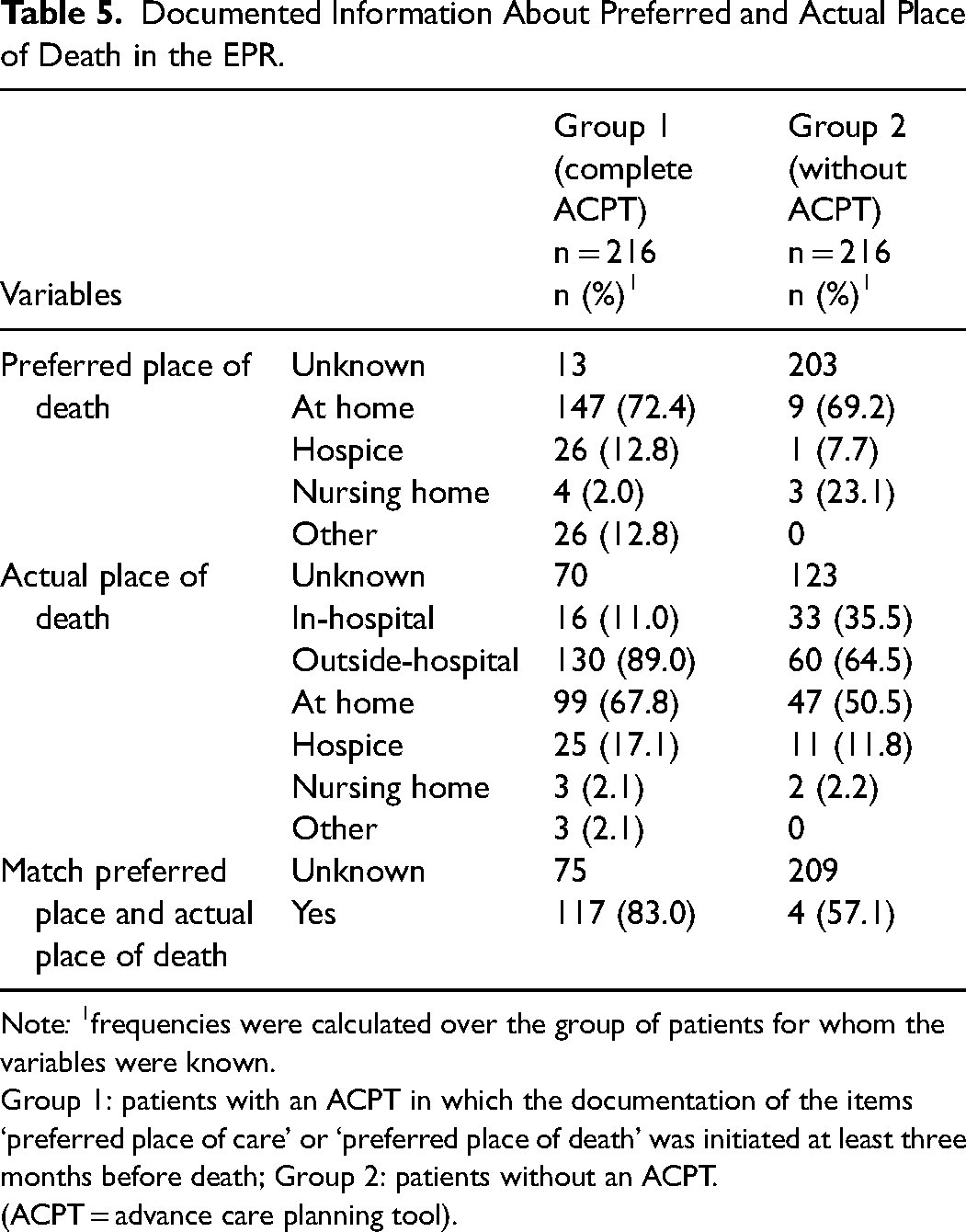
Note: 1frequencies were calculated over the group of patients for whom the variables were known.
Group 1: patients with an ACPT in which the documentation of the items ‘preferred place of care’ or ‘preferred place of death’ was initiated at least three months before death; Group 2: patients without an ACPT.
(ACPT = advance care planning tool).
Discussion
This study presents the ACPT implementation frequency, documentation of relevant information, the associations of a cACPT with systemic anti-tumor treatment and other hospital care consumption three and one month(s) before death, and place of death, among adult oncology patients. The ACPT had been implemented in the majority of all deceased patients but most often the ACPT was incomplete, ie, without documentation of the preferred place of care or death. No association was found between a cACPT and systemic anti-tumor treatments but a cACPT was associated with less frequent use of diagnostic tests during the last three months and the last month of life.
Three years after the start of the ACPT implementation, the tool had been initiated in almost 65% of the deceased patients. Our pilot study suggested that documentation of preferred place of care or death at least three months before death may contribute to more appropriate end-of-life care. 25 Yet, in the current study, the documentation of these specific preferences was found in a minority of the deceased patients (17.6%). The literature has identified some barriers to initiating ACP conversations from the perspectives of professionals, eg, prognostic uncertainty about life expectancy and patients’ (perceived) lack of willingness to discuss end-of-life issues, concerns for depriving patients’ hope, consequences for the relationship with patients, limited available time and support facilities.7,31 From the patients’ perspective, it is known that patients are open for discussions about end-of-life issues when these are conducted in a sensitive way. 32 In addition, a reason for the large number of incomplete ACPT may be that the discussions were conducted but not documented in the ACPT. In general, ACP is started late during the disease trajectory. 33 Initiating ACP early in the disease trajectory is not sufficiently integrated into routine practice yet. Future research may further focus on the experiences of patients and professionals with ACP discussions and their perspectives about the timing of these discussions. This may help to understand why elements of ACP, such as preferred place of care or death, remain undiscussed and undocumented.
No association was found between a cACPT and systemic anti-tumor treatments, except endocrine therapy, three months before death. Comparing these findings with results from other studies is difficult, because time frames used for these measurements largely vary, ie, from 180 to 14 days before patient's death.34–38 However, some studies showed that 3%-22% of the patients received chemotherapy in the last one month of life,34–36,38 which is comparable to our results. The low percentages of chemotherapy use in the last months of life in both groups may be explained that in recent years, many national campaigns were held to apply more appropriate end-of-life care. 39 This may have resulted in reluctance among Dutch healthcare professionals to provide systemic anti-tumor therapy at the end of life or in case of frailty. In addition, in our study, the use of chemotherapy in the last months of life is already quite low in both groups, which may explain why no differences were observed between the patients with cACPT and without ACPT. Although professionals consider anti-tumor treatment in the last months of life as an aggressive therapy,18,40 it should be emphasized that types of systemic anti-tumor treatment can differ in intensity and toxicity. It is unknown if ACP conversations change the patients’ choice or attitude for undergoing (less intensive) systemic anti-tumor treatment. This needs exploration in future research.
In patients with a cACPT, hospital care consumption was less than in patients without an ACPT, which was also found in our pilot-study. 25 These findings are similar to the results of another Dutch study, in which structured ACP documentation was associated with significantly fewer diagnostic tests and less contacts with hospital disciplines in the last months of life. 33 In both the current and pilot study, 25 more daycare visits were observed among patients with a cACPT, which matches the observation of Groenewoud and colleagues. 41 An explanation for these results may be the timely end-of-life care discussion in patients with a cACPT, contributing to the delivery of palliative interventions in an outpatient setting. None of the patients with a cACPT was admitted to the ICU, in contrast to eight patients without an ACPT. Caution may be needed in drawing conclusions. Both groups may not have been completely comparable with regard to the disease stage. Unfortunately, documentation of the treatment intent was often lacking. It is known for a small number of patients without ACPT they were treated with a curative intention. The unexpected finding that significantly less hormonal therapy was used among patients with a cACPT could also indicate differences regarding treatment intent between both groups, because hormonal therapy is mainly used early in the disease process, suggesting that some patients without ACPT were in an earlier disease stage. The presented results only indicate whether the treatment intention was documented in the EPR. Nevertheless, treatment intention might be a confounding factor which could not controlled for in our analyses due to the high number of not documented items.
The association between ACPT and the match between preferred and actual place of death could not be analyzed due to the lack of documentation on both items. However, of patients with a cACPT whose actual place of death was known (n = 136), most had an out-hospital death (89.0%) and died at home (67.8%). These percentages are higher than the national percentages for out-hospital deaths (84.4%) and home-deaths (54.5%). 42 In line with earlier studies,19,25,43–46 our results show that a cACPT seems to support death at the preferred place. In patients without an ACPT, information about the preferred place of death was frequently missing. This does not necessarily mean that it had not been discussed, but this information could not be retrieved. Information regarding the preferred place of death might get lost if it is not well documented in the EPR, limiting usability for the discussion and planning of the care around death.
Timely initiated ACPT including documentation of the preferred place of care or death was related to more appropriate end-of-life care. After all, timely knowing the patient's preferences enables more time to discuss and prepare appropriate care in line with the preferences. 47 As a result, patients may spend more time at home in the last months of life. Relatives may experience benefits of timely initiated ACP as well, and may feel more satisfied with the provided care. 48 In addition to the quality-of-life benefits for patients and relatives, the presented results of less hospital care consumption are important given the current and future healthcare staff shortages and the rising healthcare costs. However, we do did not study whether and how hospital care was transferred to the home situation.
Limitations
As mentioned, there may have been differences between the two groups due to the retrospective and observational design. Knowledge of WHO-performance status and the presence of comorbidities contribute to a better assessment of whether a systemic anti-tumor treatment should be started. 49 This information was often lacking in both groups and therefore, not presented. Missing values were considered inappropriate to impute, because they were expected not to be missing at random. For example, when the patient lives alone, the professional who experiences benefits of the ACPT, will be more triggered to document this information into the ACPT than the professional who has less affinity with the ACPT. As a result, the professional could be seen as a possible confounder, which should be considered in future research. Lastly, bias may have occurred in the presented ‘social context’ results: when extracted from the ACPT, no limit was set on the time of documentation, as the ACPT remains easily accessible in the EPR; for the patients without an ACPT, however, the EPR report had to be checked, which was, for practical reasons, only feasible for the documentation regarding the last three months of life. Some information about the social context may indeed have been recorded earlier than three months before death but could not be retrieved.
Conclusion
Although the ACPT is implemented in two thirds of patients, timely documentation of preferred place of care or death is often missing. Yet, timely documentation of these preferences may promote out-hospital-death and save hospital care consumption.
Footnotes
Acknowledgements
Anna van der Zalm1, Marianna Van Vugt1, Jorgen van der Zanden2. 1Team Management Information, Jeroen Bosch Hospital, 2Patient registration, Jeroen Bosch Hospital.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
