Abstract
Introduction:
Radiofrequency (RF) microneedling requires minimal downtime; however, patients’ tolerance of the procedural pain can limit cosmetic results. The goal of this study was to determine the patient tolerance of RF microneedling with sufentanil sublingual tablet (SST) 30 μg and topical local anesthetic compared with topical local anesthetic alone.
Materials and Methods:
The study was a one-way crossover study of patients undergoing RF microneedling of the face or abdomen. Topical local anesthetic was applied 45 minutes prior to the initial procedure to one side of the face or abdomen (control side), followed by topical local anesthetic and SST administered 45 and 30 minutes, respectively, prior to the procedure on the opposite side (SST-treated side). The primary endpoint was patient completion of the procedure with additional endpoints of Richmond Agitation Sedation Scale scores, patient and provider satisfaction ratings, vital signs, oxygen saturation levels, and adverse events.
Results:
A total of 51 patients were enrolled averaging 44.8 ± 11.1 (SD) years of age. Forty-five percent of patients successfully completed the control side compared with 96% of patients successfully completing the SST-treated side (P < .001). Patients were less restless or agitated during the SST-treated side (6%) compared with the control side (51%; P < .001). Patient and provider satisfaction scores were significantly improved with SST (P < .001). Vital signs remained stable with SST treatment compared with the control side.
Discussion:
The SST is a safe and effective sublingual analgesic allowing patients to be awake and alert while increasing patient tolerance of RF microneedling as well as patient and provider satisfaction with the procedure.
Conclusion:
The SST is a novel analgesic that can be used in office-based settings with appropriate safety equipment available.
Introduction
Minimally invasive technologies to improve the cosmetic appearance of skin and the underlying tissues are becoming popular as they result in minimal downtime and can often produce results similar to more invasive surgical procedures. A factor that can limit optimal results from these treatments is patients’ tolerance of the procedural pain.1,2 Specifically regarding radiofrequency (RF) microneedling, the depth of needle placement and/or intensity of RF energy may need to be reduced due to patients experiencing moderate to severe pain.3,4 This may result in a less than optimal cosmetic result, requiring patients to endure repeated procedures or possibly be lost to follow-up due to lack of patient satisfaction.
In an effort to increase patient tolerance of the RF microneedling procedure, topical local anesthetic gel is applied (usually a combination of lidocaine and tetracaine with or without benzocaine), typically 30 to 45 minutes prior to the procedure. As this topical anesthetic alone is often not sufficient for the patient to successfully complete the procedure, additional more invasive methods of local anesthesia are often performed, such as local nerve blocks with lidocaine or a field block with tumescent anesthesia, to allow the optimal needle depth and energy level to be achieved comfortably.5,6 These local anesthetic injections require expertise to be performed effectively and safely, can be time-consuming, and can be painful and anxiety-provoking for the patient.
Oral benzodiazepines are an option to provide anxiolysis and amnestic effects but these drugs lack any analgesic properties. 7 Oral opioids have both analgesic and anxiolytic properties but the slow onset of effect requires administration 1 to 2 hours in advance of the procedure.8,9 Inhaled nitrous oxide is a fairly weak analgesic and is cumbersome, especially with facial procedures. The compounded sublingual lozenge containing midazolam, ketamine, and ondansetron (“MKO melt”) is not Food and Drug Administration (FDA)–approved and contains a subtherapeutic dose of the analgesic ketamine and, therefore, mainly has its effect via the benzodiazepine midazolam. 10
A novel sublingual opioid analgesic, sufentanil sublingual tablet (SST) 30 μg (DSUVIA; AcelRx Pharmaceuticals, Hayward, California; Figure 1), is a recently approved product for use in medically supervised settings. 11 The micro-sized tablet is prefilled in a plastic applicator for ease of sublingual dosing and has an analgesic onset in 15 minutes and provides significant analgesia in the perioperative setting with a single dose.10,12-14 At the peak plasma concentration (60 minutes) after a single dose, patients did not demonstrate cognitive impairment as measured using the Six-Item Screener test, which is an advantageous attribute in office-based procedures. 15 The purpose of the current study was to determine whether a preprocedural dose of SST combined with topical local anesthetic could increase the percent of patients successfully completing the RF microneedling protocol performed on either the face or body compared with topical local anesthetic alone.
Sufentanil sublingual tablet 30 μg in plastic applicator.
Materials and Methods
This study was approved by the Freeman Institutional Review Board on September 17, 2021. Following informed consent, patients who were 18 years of age or older and desired RF microneedling of either the face or abdomen were allowed to participate in this study that was conducted at 2 clinical sites (Maningas Cosmetic Surgery, Joplin, Missouri; Southern Oklahoma Women’s Health, Ardmore, Oklahoma) from October to November 2021. Exclusion criteria included patients with a known severe reaction to opioids, known allergy to lidocaine or tetracaine (used for the topical local anesthetic), opioid-tolerant patients taking more than 15 mg oral morphine equivalent per day, and patients requiring home oxygen therapy.
This was a one-way crossover design study using a split face/abdomen approach allowing patients to act as their own control. Patients received different analgesic treatments for each side of the face or body prior to the RF microneedling procedure (Figure 2). Initially, patients received a topical application of a compounded local anesthetic (lidocaine 23%/tetracaine 7%) in a plasticized topical gel (“control side”) 45 minutes prior to the initiation of RF microneedling (Morpheus8; InMode Aesthetic Solutions, Lake Forest, California; Figure 3) on one side of the face or abdomen.
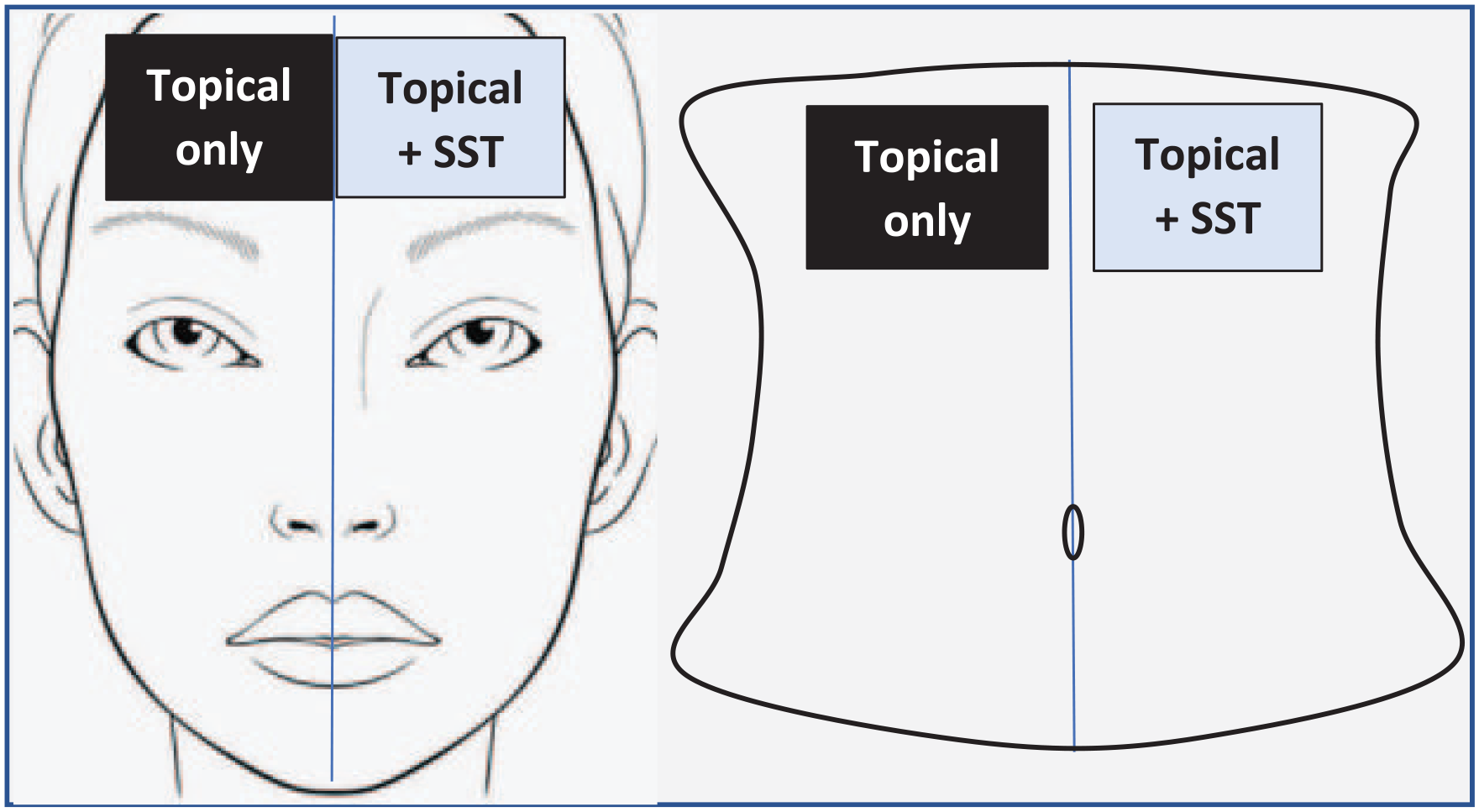
Split face/abdomen method.

Morpheus8 RF microneedling device.
The fractional RF device has programmable penetration depth and energy delivery and consists of 24 coated needles penetrating the subdermal tissue. The prespecified needle depth and energy settings for the study are shown in Table 1. The ability of the patient to tolerate the pain levels and either complete or terminate this control side was then recorded as the primary endpoint. At the end of this control side treatment, the Richmond Agitation Sedation Scale (RASS; Table 2) was used to determine the level of agitation or sedation that the patient was demonstrating. 16 This is an objective measurement scale with a score of 0 for an alert and calm patient, positive scores (eg, +1 to +4) for restless and agitated patients, and negative scores (eg, −1 to −5) for progressively greater levels of sedation. In addition, patient and provider satisfaction ratings were obtained for this control side by asking the patient to rate their satisfaction with pain control during the procedure as poor, fair, good, or excellent while the provider was also instructed to rate their satisfaction with the comfort level of the patient using this same scale.
RF Microneedling Settings.
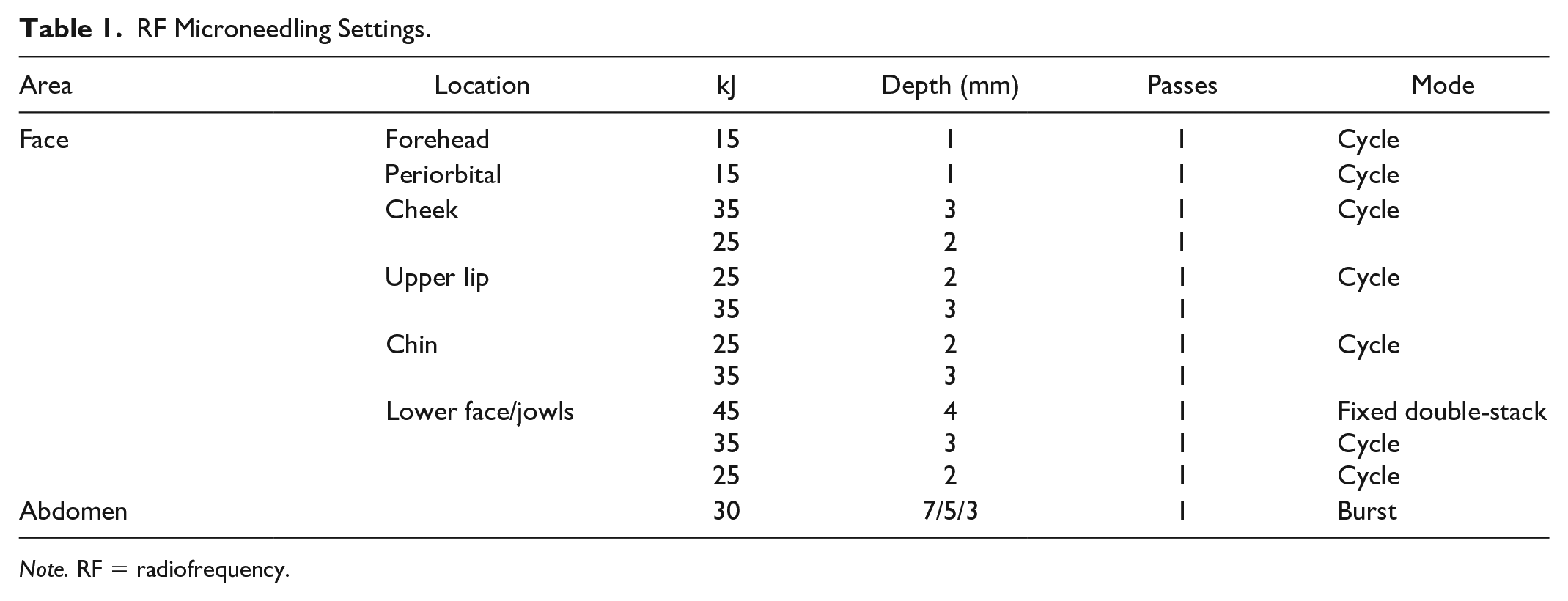
Note. RF = radiofrequency.
Richmond Agitation Sedation Scale Scoring.
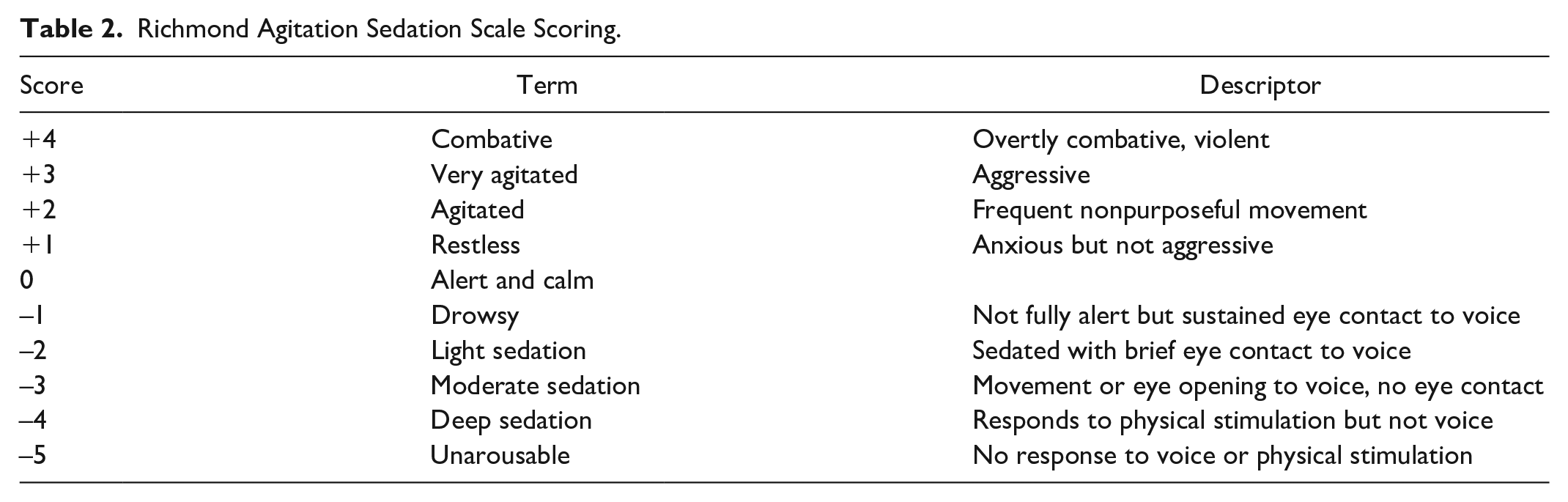
Following the control side treatment, the opposite side of the face or body was treated with the same topical local anesthetic gel, and 15 minutes after this application, a single dose of SST 30 μg was placed sublingually. To maximize sublingual absorption of the drug, patients were instructed to not speak and keep their tongue still for 5 to 10 minutes after dosing as the median erosion time of the tablet is 5 minutes. 17 The RF microneedling procedure was then started on this “SST-treated” side 30 minutes after SST dosing which was 45 minutes after the topical local anesthetic gel was applied. The same needle depth and energy settings as in Table 1 were utilized. The ability of the patient to complete this side of the treatment was then recorded, along with the RASS score and patient and provider satisfaction ratings.
Vital signs were measured and recorded every 15 minutes throughout the procedure and oxygen saturation values were measured continuously and recorded every 15 minutes throughout the procedure. Supplemental oxygen was available if a patient’s oxygen saturation dropped below 93% and did not immediately improve with a deep breath; otherwise, all patients were breathing room air. Any adverse events and medications required to treat these events were documented.
Data are reported as mean ± standard deviation (SD); the paired-sample McNemar test was utilized to compare the primary endpoint of the patient’s ability to complete the procedure on each side. A 2-sided Wilcoxon signed rank test was used to compare RASS scores and patient and provider satisfaction scores for each treatment. Procedure duration and vital signs were compared using the 2-sided t test.
Results
Patient Demographics and Primary Endpoint
A total of 51 patients were enrolled in the study and all were female with an average age of 44.8 ± 11.1 (range = 25-63) years and an average body mass index (BMI) of 27.2 ± 5.6 (range = 20.5-54.5) kg/m2. There was essentially an even split for treatment area, with 26 patients undergoing RF microneedling of the face and 25 patients receiving the procedure on the abdomen. The primary endpoint of successful completion at the original device settings was substantially improved by the use of SST. Less than half of the patients (23 of 51; 45%) successfully completed the control side (topical local anesthetic only) compared with 49 of 51 (96%) patients successfully completing the SST-treated side (P < .001). The 2 patients who did not successfully complete the SST-treated side also did not complete the control side. The average procedure duration was shorter for the control side (29.1 ± 14.5 minutes) compared with the SST-treated side (41.2 ± 16.7 minutes; P < .001) as significantly more patients aborted the procedure earlier on the control side due to uncontrolled pain.
Secondary Efficacy Endpoints
The RASS scores demonstrate that while the most common rating was “calm” (RASS score of 0) at the end of both treatments, more patients on the control side were rated as restless (RASS score of +1) or agitated (RASS score of +2) at the end of their treatment (or partial treatment for patients not tolerating the procedure), compared with the same patients following the procedure on the SST-treated side (Table 3). No patient scored a −1 or lower on the RASS, demonstrating there was no sedative effect with either treatment.
RASS Ratings Following Each Treatment.
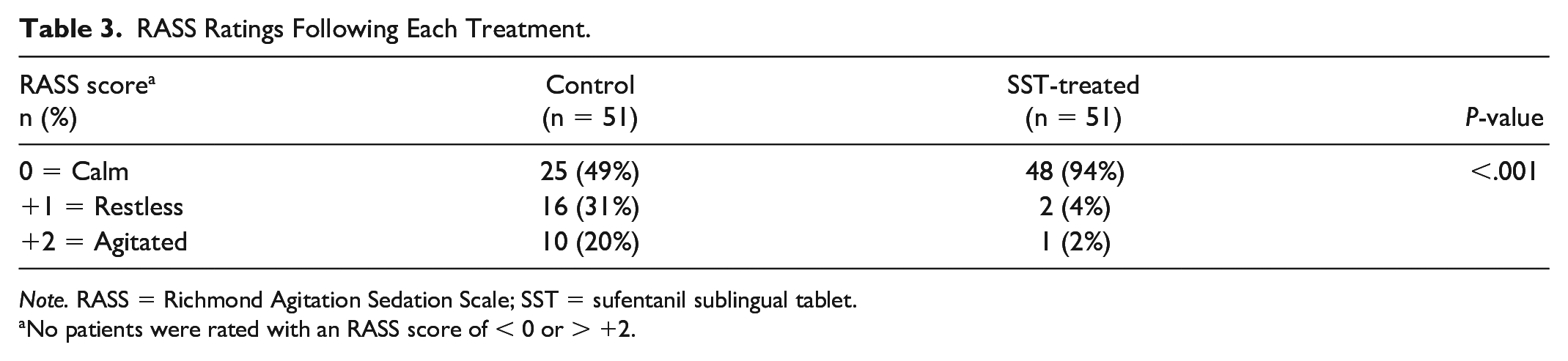
Note. RASS = Richmond Agitation Sedation Scale; SST = sufentanil sublingual tablet.
No patients were rated with an RASS score of < 0 or > +2.
Patient-reported satisfaction scores improved significantly with SST treatment compared with the control-treated side (P < .001; Figure 4). With topical local anesthetic only, patients mainly rated their satisfaction as fair, with no patient reporting excellent satisfaction with pain control. When RF microneedling was performed with both topical local anesthetic and SST treatment, more than 80% of patients rated their satisfaction with pain control as good or excellent. The 3 patients rating pain control as poor for the SST-treated side also reported a poor rating on the control side.
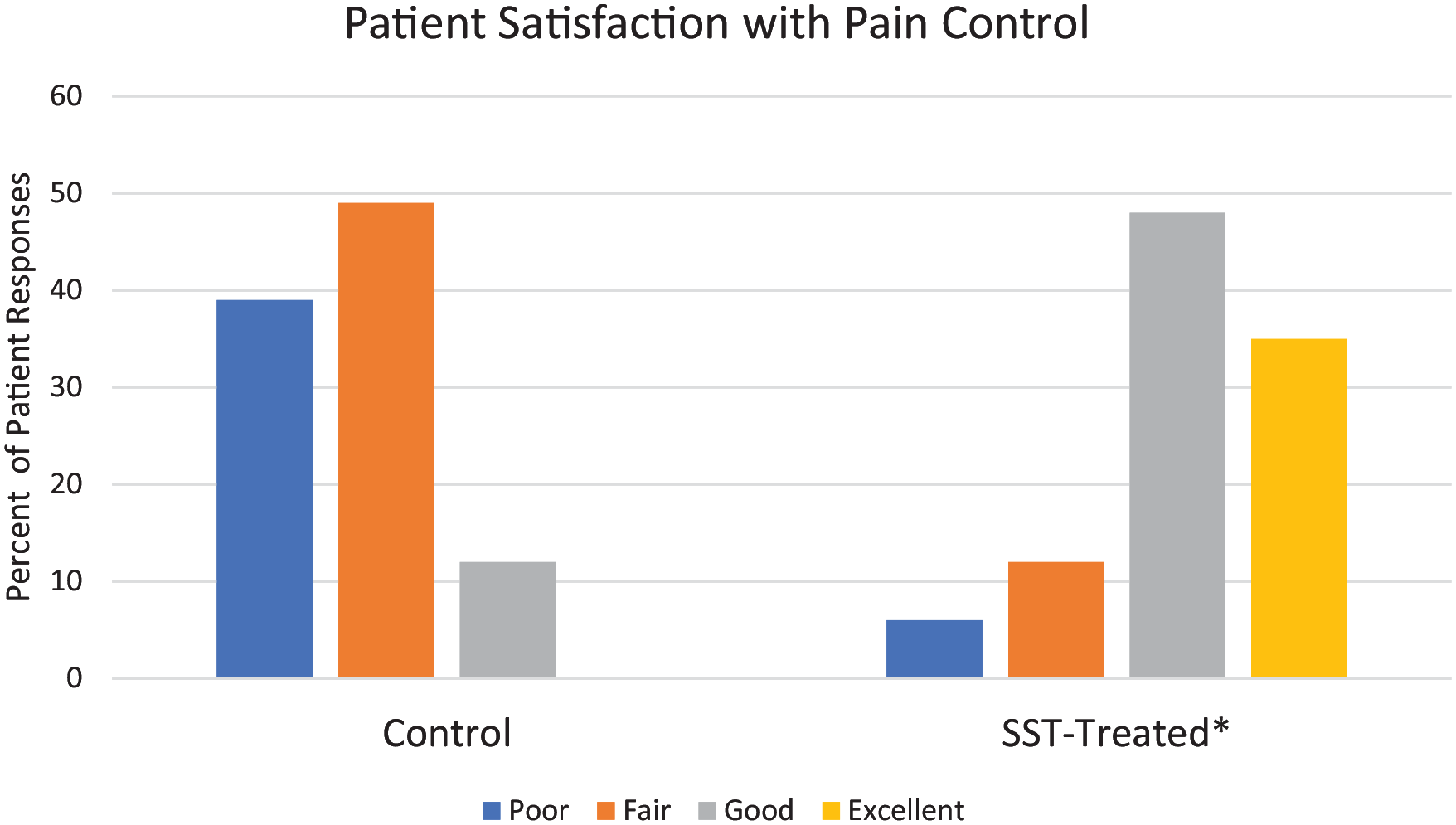
Patient-reported ratings of satisfaction with pain control for each treatment.
Provider satisfaction ratings with the comfort level of the patient also improved significantly for the SST-treated side compared with the control side (P < .001; Figure 5). Ninety percent of providers rated their satisfaction with the patient’s comfort level on the SST-treated side as good or excellent. Providers rated the patient comfort with the control side most commonly as poor and no patient received a rating of excellent. The 3 patients receiving provider ratings of poor for the SST-treated side also were rated as poor by the provider for the control side and these patients also rated themselves as having poor pain control for both sides.
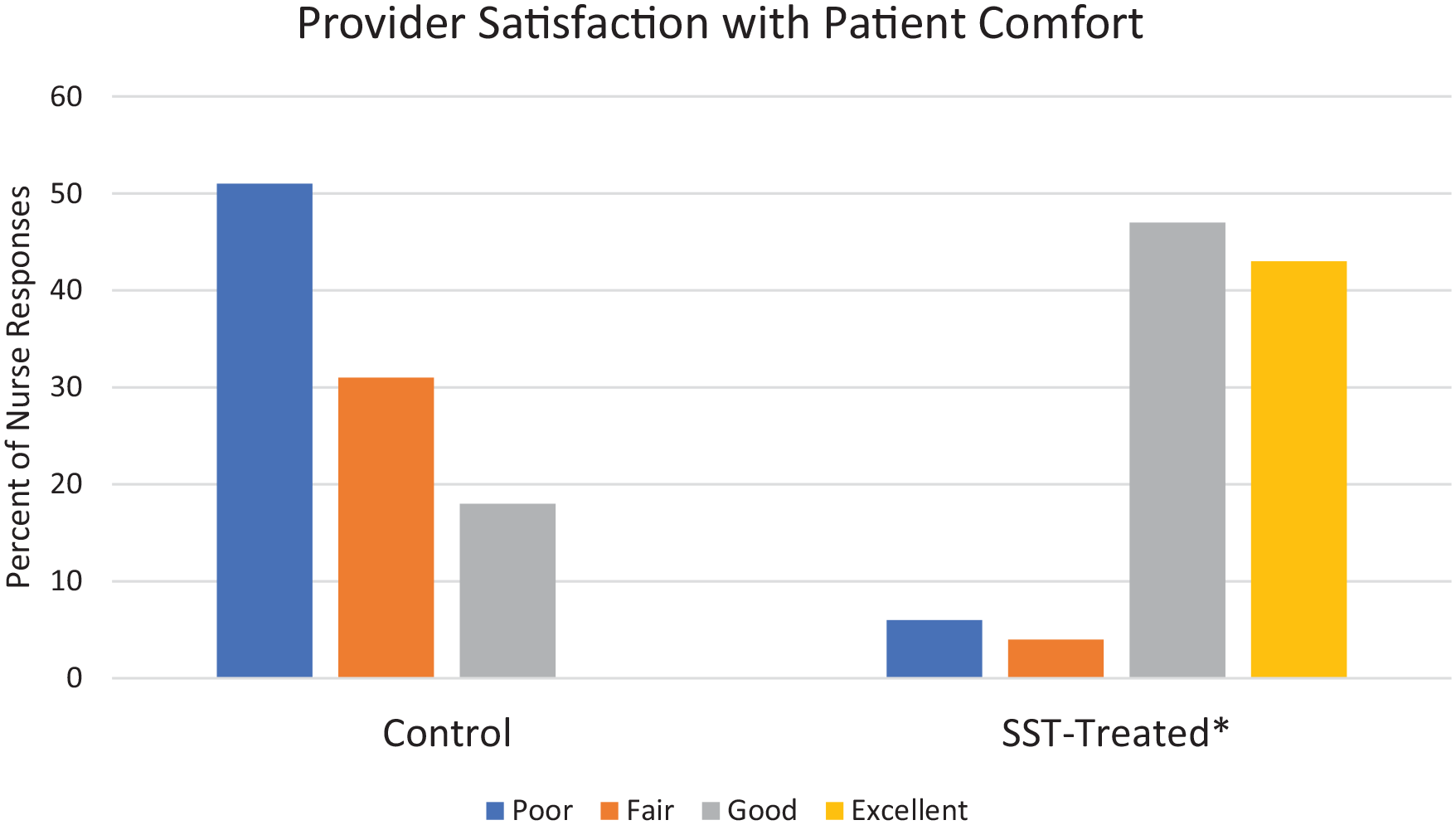
Provider-reported ratings of satisfaction with patient comfort for each treatment.
Safety
Vital signs and oxygen saturation during the control side treatment compared with baseline values are shown in Figure 6. Mean heart rate values increased from baseline 30 minutes after the start of the procedure. Mean systolic pressures increased at 15 and 30 minutes and diastolic pressures increased at 45 minutes after the start of the procedure. Mean respiratory rate increased at 15 and 30 minutes after the start of the procedure and mean oxygen saturation did not change.
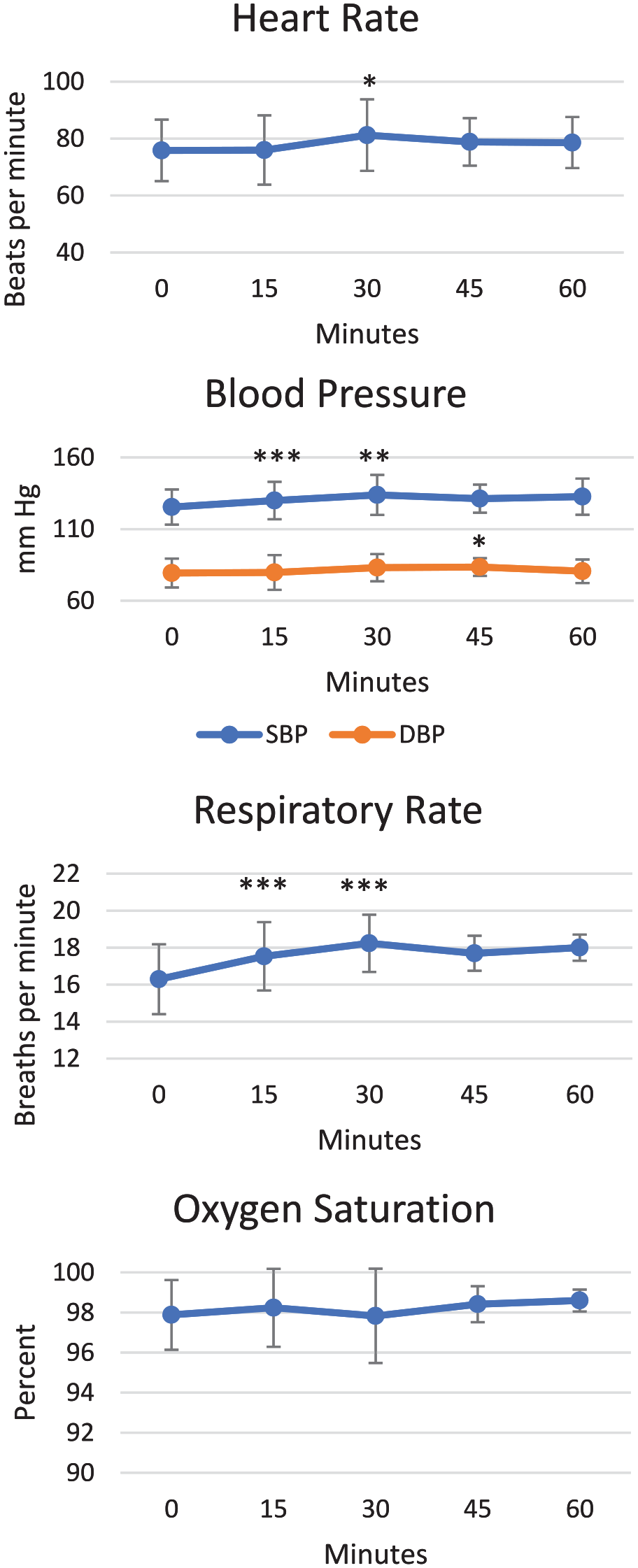
Vital signs and oxygen saturation for control side.
There was no effect on mean heart rate, systolic or diastolic pressure or oxygen saturation for the 30 minutes following SST administration compared with values recorded just prior to dosing with SST (at t = 0 minutes; Figure 7). Microneedling started 30 minutes following SST dosing and no changes in mean heart rate were observed throughout the procedure. Mean systolic pressures increased slightly at 45 minutes (15 minutes after the procedure began) but no changes in mean diastolic pressures occurred. No changes in mean respiratory rate occurred throughout the treatment and mean oxygen saturation values decreased by 1% at 45 minutes and by 0.7% at 60 minutes after SST dosing.
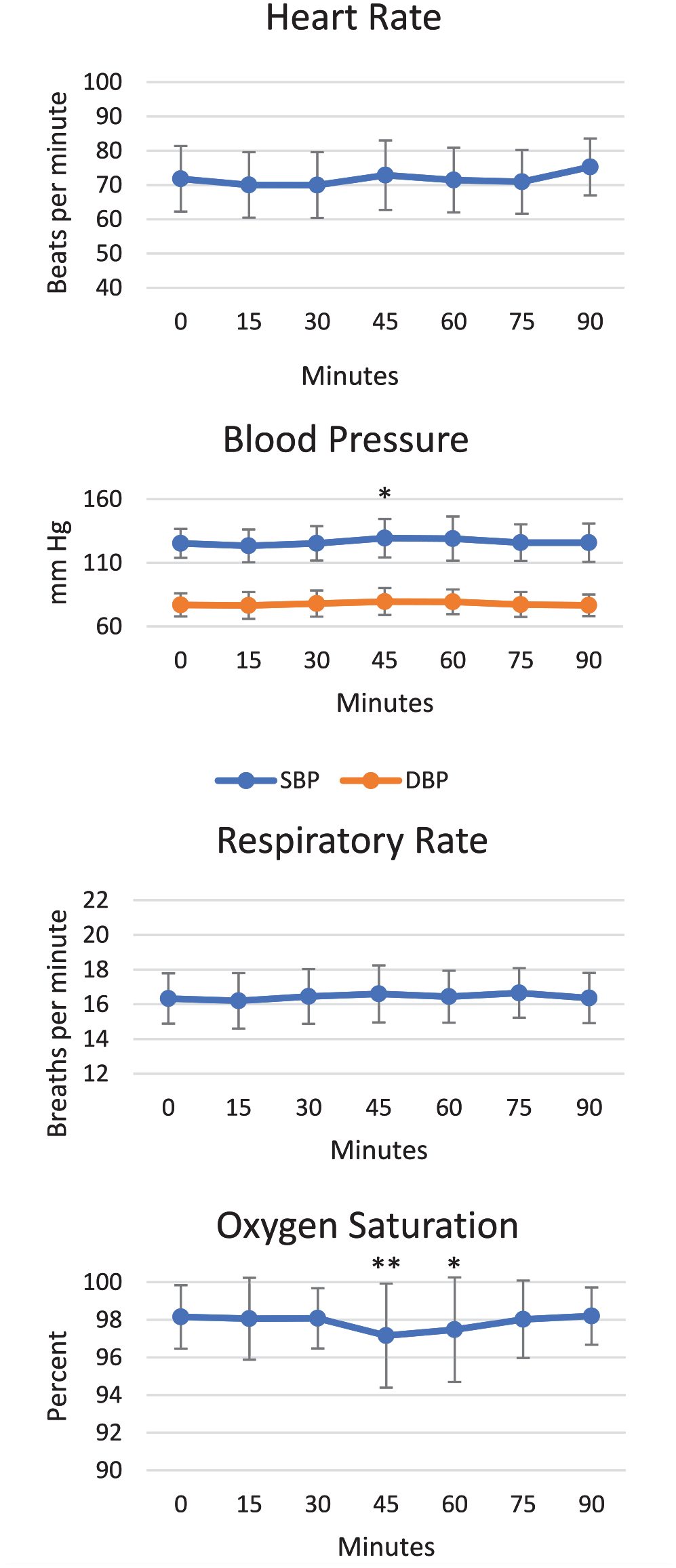
Vital signs and oxygen saturation for SST-treated side.
No patients throughout the study required supplemental oxygen. Adverse events were nausea (5.9%), vomiting (5.9%), and dizziness (2.0%), which all occurred after the entire treatment was performed. Patients with nausea/vomiting were treated with ondansetron oral disintegrating tablet 4 mg and resolution of their symptoms occurred prior to discharge. Of note, no patients had received antiemetic prophylaxis prior to the procedure. The case of dizziness was mild and transient and resolved without treatment.
Discussion
Although aesthetic procedures such as RF microneedling are minimally invasive from a needle penetration depth perspective, patients are often exposed to significant pain even with preprocedural topical application of local anesthetics. 4 This study demonstrated that less than half the patients were able to tolerate the standard initial needle depth and energy settings with a 45-minute preapplication of topical anesthetic gel to the first side of the face or abdomen. By adding SST 30 μg to the analgesic regimen 30 minutes prior to the second side of the procedure, patients more than doubled their completion rate for the procedure (from 45% to 96%).
Results of the secondary endpoint of the RASS score following the end of each treatment support the primary endpoint outcome of better patient tolerability of RF microneedling with SST. The SST together with the topical local anesthetic was able to decrease the percent of patients being scored as restless or agitated following the treatment from 51% down to 6%. Importantly, no patient was reported as drowsy (RASS score of −1) following either treatment side, supporting earlier studies that SST, while producing significant analgesia and patient comfort, does not affect cognitive function.
Both patient and provider satisfaction ratings of pain control and patient comfort, respectively, showed that more than 80% of patient and provider responses rated the first (control) side as poor or fair, with no respondent reporting an excellent rating. Therefore, while 45% of patients completed this side, they did so with relatively low satisfaction ratings. Interestingly, as the providers had observed many patients undergoing the RF microneedling procedure with SST, this likely resulted in their most prevalent score being in the poor category (more than 50%) for patient comfort on the control side. Patients, having not experienced the procedure with SST in the past, more frequently rated their pain control as fair on the control side. With the addition of SST, more than 80% of both the patient and provider satisfaction responses were good or excellent, a dramatic shift compared with the previous ratings.
Although vital signs are typically not measured during minimally invasive procedures such as RF microneedling, it is interesting to note the hemodynamic changes that do occur. The alterations in vital signs over the course of the procedure on the control side are clear objective evidence of the increased patient discomfort. Heart rate, systolic and diastolic blood pressure, and respiratory rate were all increased. For the 30-minute period following the administration of SST, there were no changes in any vital sign parameter, and with the initiation of the procedure, only a slight increase in systolic pressure at one timepoint was observed. This relative hemodynamic stability compared with the control side is further objective evidence of improved patient comfort and tolerance of the procedure. Importantly, the slight decrease in mean oxygen saturation levels following the initiation of the procedure was not clinically meaningful as no patient required supplemental oxygen and no corresponding respiratory rate decrease was noted.
Adverse events were minimal and not uncommon for opioid analgesics. The 6% incidence of nausea and vomiting was similar to previous reports from a study of SST use in an emergency department study for acute injury. 15 Similar to the current study, patients also did not receive prophylactic antiemetics. Likely, this low rate of nausea/vomiting at the end of the procedure could be further reduced with preprocedural oral ondansetron and this may be warranted for patients with a known history of gastrointestinal sensitivity to opioids. There were no central nervous system (CNS) side effects, with the exception of 1 patient experiencing dizziness. At no time were patients reported to have somnolence, which was further evidenced by the RASS scores remaining greater than or equal to 0. The lack of any respiratory adverse events in this study is likely due to the nature of the drug absorption from the sublingual tissues, which occurs over time, thereby significantly reducing the rate of rise and peak plasma concentrations (by 17-fold) compared with intravenous (IV) bolus administration of the same 30 μg dose of sufentanil. 18
These adverse event data, along with the RASS and vital sign data, support that the use of SST falls into the American Society of Anesthesiologists (ASA) minimal sedation (level 1) category, which is described as “a drug-induced state during which patients respond normally to verbal commands. Although cognitive function and physical coordination may be impaired, airway reflexes, and ventilatory and cardiovascular functions are unaffected.” 19 Importantly, prior to ordering SST, clinical sites must be certified under the product’s Risk Evaluation and Mitigation Strategies (REMS) program that ensures that basic monitoring and safety equipment, including supplemental oxygen, pulse oximetry, and naloxone, are available at sites prior to SST use. 20
Limitations of this study include that it was a one-way crossover design, as SST has an elimination half-life of 13 hours and the reverse order of treatments was not possible for a same-day procedure. An additional limitation is that patients and providers were not blinded to the treatments. For this reason, both subjective and objective endpoints were assessed regarding the patient comfort level.
Conclusion
Based on the results of this study, we have incorporated SST into our RF microneedling protocol, which has significantly improved our patients’ experience with this procedure. Being able to complete the procedure with the appropriate needle depth and energy levels to optimize the cosmetic result while maintaining patient comfort has led to greater patient satisfaction and more extensive treatment areas being able to be covered in one session.
Footnotes
Acknowledgements
Editorial assistance was provided by Pamela Palmer, MD PhD, of AcelRx Pharmaceuticals, Inc.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.M. and H.R. are paid consultants for InMode, Ltd; otherwise, they have no other disclosures. K.M. has no disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
