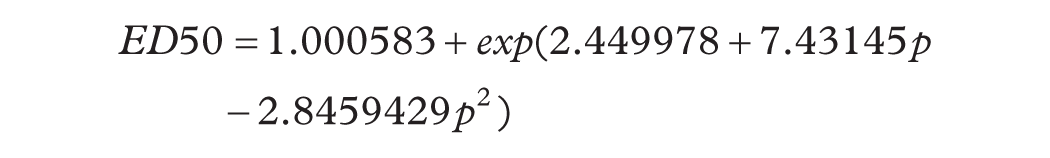Abstract
Light is the primary circadian time cue, but there are large interindividual differences in how sensitive the circadian system is to light. Currently, it is not well understood how individual differences in light sensitivity interact with real-world light environments to determine sleep and circadian timing. We used a validated computational model to simulate sleep and circadian timing (predicted dim light melatonin onset) under realistic assumptions about light and work schedules. Simulations were repeated varying light sensitivity (translated to equivalent ED50 values for interpretability), as well as evening, morning, and daytime illuminances. Brighter evening light led to later predicted circadian and sleep timing, with this effect being amplified by high light sensitivity. Reducing evening light was particularly beneficial for those with high light sensitivity or a long circadian period. Brighter morning light was beneficial for individuals with a long circadian period, or those with both high light sensitivity and high evening light. However, bright morning light could be maladaptive in individuals with a short circadian period or those with low light sensitivity and low evening light. Brighter daytime light attenuated the delaying effects of evening artificial light across conditions, indicating that increasing daytime light was the most universally beneficial lighting intervention. Our results demonstrate how circadian light sensitivity can be used to tailor individual-level solutions that support optimal sleep and circadian timing.
Keywords
Daily patterns of light exposure are the dominant time cue for the human circadian clock, enabling entrainment to the 24-h solar cycle (Roenneberg et al., 2013). The timing of circadian rhythms is influenced by light in a specific, phase-dependent fashion. Light exposure during the biological morning causes phase advance of circadian rhythms, whereas light exposure during the biological evening and early night causes phase delay of circadian rhythms (Khalsa et al., 2003). Technological advancements, including electric lighting, work schedules, and the built environments in which humans now spend the majority of their lives, have fundamentally altered light exposure patterns (Yetish et al., 2015). These changes have resulted in later and more variable circadian timing (Wright et al., 2013). As the circadian clock regulates sleep and many other areas of human physiology (Roenneberg and Merrow, 2016), disruption of circadian rhythms by modern light exposure patterns has important effects on physical and mental health (Burns et al., 2023; Fishbein et al., 2021; Windred et al., 2024a, 2024b).
General circadian principles have been used to inform the development of lighting design (Vetter et al., 2022), as well as light-based interventions such as morning light therapy to realign circadian rhythms (Gooley, 2008). However, it was recently discovered that the sensitivity of the circadian clock to light differs greatly between individuals (Chellappa, 2021). These interindividual differences in responses are particularly pronounced for intermediate levels of light exposure, which are typically experienced indoors (Phillips et al., 2019). Evening light exposure levels in real-world settings indicate a potentially strong interaction of circadian light sensitivity with an individual’s light environment in determining the effects on the circadian system (Cain et al., 2020). Consequently, the same pattern of daily light exposure could be beneficial for some individuals, while being harmful for others. These findings make it challenging to provide generic advice regarding lighting in the home and workplace. A fuller understanding of how an individual’s circadian light sensitivity determines optimal lighting choices is needed to tailor individual-level solutions that support optimal sleep and circadian timing.
Translating current understanding of circadian rhythms and their response to light from the lab to the real world presents several challenges. First, it is difficult to track circadian rhythms in real time. Second, light exposure patterns in the real world are variable and tied to an individual’s sleep-wake patterns, rather than being pre-defined stimuli as they are in laboratory settings. These challenges can potentially be addressed by computational modeling (Stone et al., 2020b). Physiologically based models have been developed to simulate human circadian rhythms and sleep-wake patterns (Asgari-Targhi and Klerman, 2019; Robinson et al., 2011), generating accurate predictions of circadian timing at the group-average level under a variety of real-world conditions (Cheng et al., 2021; Stone et al., 2019; Woelders et al., 2017). These models have been used to understand how light exposure patterns and work schedules affect sleep and circadian timing, taking into account known interactions between sleep, lighting, and the environment (Skeldon et al., 2017; Swaminathan et al., 2017). Furthermore, these models explicitly represent circadian light sensitivity in the form of model parameter values that can be systematically explored to examine differences in circadian physiology at the individual level (Stone et al., 2020a).
In this article, we used a validated computational model of human sleep and circadian rhythms to determine how an individual’s circadian light sensitivity interacts with their light environment on a day-working schedule. We specifically explored the roles of morning, daytime, and evening light levels in determining predicted sleep and circadian timing. In addition, we investigated how these relationships vary as a function of an individual’s tendency toward early versus late circadian timing due to their intrinsic circadian period.
Methods
The Model
To predict both sleep-wake times and circadian timing, we used a combined model of the sleep-wake switch (the Phillips-Robinson model; Phillips and Robinson, 2007; Robinson et al., 2011), coupled with a limit-cycle-oscillator model of the human circadian system and its response to light (Hilaire et al., 2007; Jewett et al., 1999). This combined model incorporates the circadian and homeostatic regulation of sleep, within a physiological framework. The model has been previously described in detail and shown to reproduce typical sleep-wake patterns under real-world assumptions about light patterns and work schedules (Skeldon et al., 2017). Full model equations can be found in the Supplementary Material. As shown in Figure 1, this model takes the following inputs: (1) time intervals when sleep is not allowed (e.g., corresponding to work shifts), and (2) assumed light levels, as a function of time of day and sleep-wake state. The model generates the following outputs: (1) daily predictions of sleep onset and offset times, and (2) daily predictions of circadian phase. The model includes parameter values that correspond to physiological properties of the sleep and circadian system, such as light sensitivity and intrinsic circadian period.
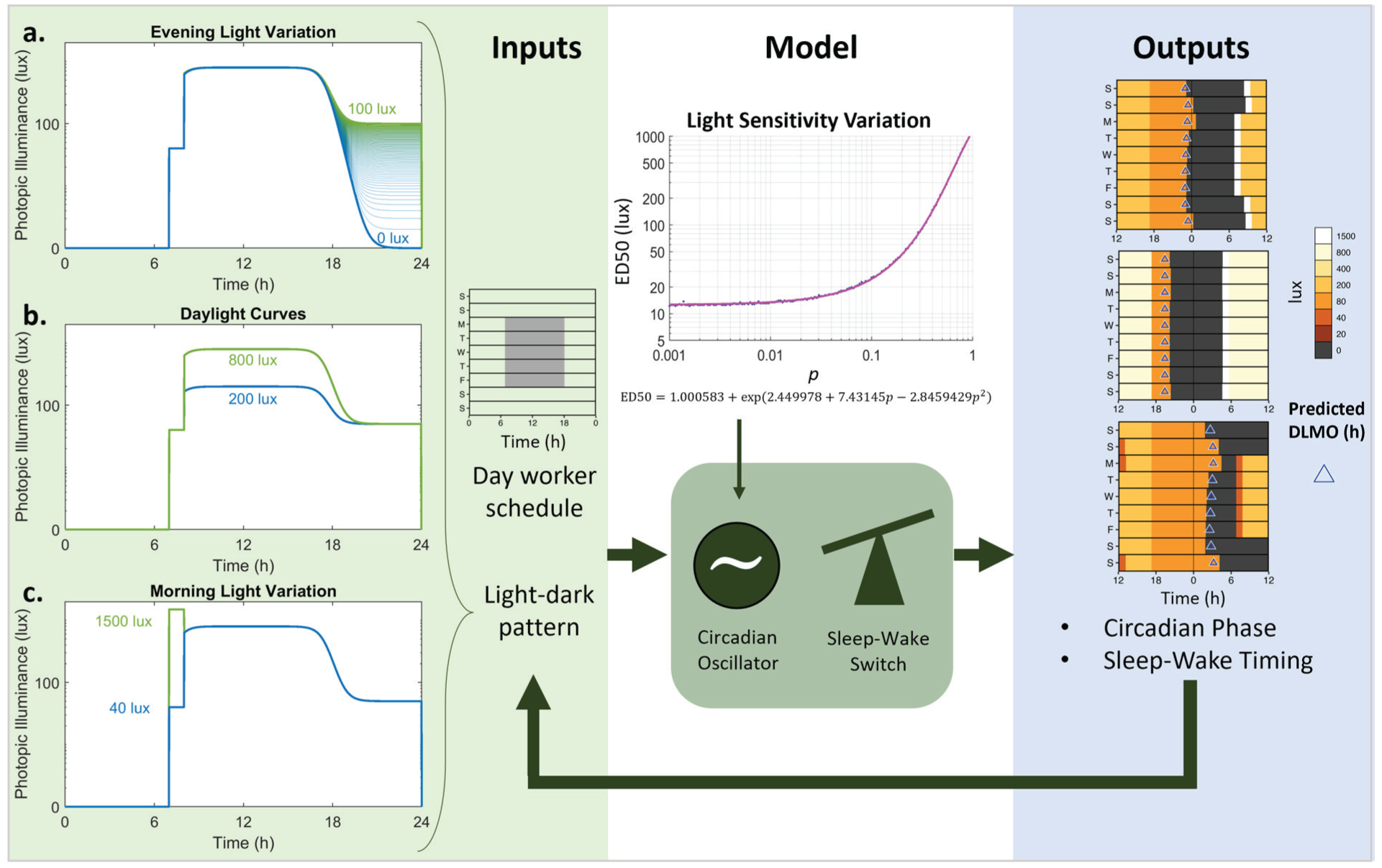
Schematic of simulations. Inputs to the model are daily light-dark patterns, on a day-work schedule with required wake between 7 a.m. and 6 p.m. on weekdays. Light patterns were varied across the following ranges: (a) evening light varied between 0 and 100 lux in 1 lux increments; (b) daytime light levels were either a maximum of 200 lux or 800 lux; and (c) morning light levels for the 1 hour after waking were either 40 lux or 1500 lux (representative of a bright light therapy intervention). The model is a combined circadian oscillator model of entrainment to light (Hilaire et al., 2007) and a sleep-wake switch model (Phillips and Robinson, 2007). Light sensitivity in the circadian model was varied to determine the shape of the dose-response curve to light. The light sensitivity parameter (p) was mapped to an equivalent ED50 using the equation indicated. Outputs of the model were the predicted circadian phase (represented as DLMO in triangles) and sleep-wake timing (represented as dark bars). Sleep-wake timing informed the light-dark pattern input each day, as light levels were set to 0 lux during sleep.
Simulations
We investigated the impact of varying circadian light sensitivity on sleep and circadian timing, across different lighting environments. We investigated light sensitivity via the parameter p in the model, which determines the shape of the dose-response curve to light (Stone et al., 2020a). Lower values of p increase sensitivity to lower light levels. Simulations were run with values of p ranging from 0.01 to 0.2 in 0.02 steps, and from 0.21 to 1 in 0.01 steps. For each value of p, we ran the simulation varying the evening light levels, defined as the light level during wakefulness from sunset time to sleep onset time, between 0 lux (total darkness) and 100 lux, in 1 lux increments. To aid in the interpretation of the model results, we converted the values of p into equivalent ED50 values (i.e., the light level required for 50% of the maximum response). To derive these values, we simulated a phase-shifting protocol. A 5-h light pulse was delivered to the model, timed close to the maximum phase delay response (see Supplementary Materials), with the model free-running in constant darkness before and after the stimulus. This simulation was performed across a range of p values from 0.001 to 1, and a range of light levels, from 0.0095 lux to 9500 lux, using the model’s reference light level of 9500 lux as the maximum value. For each value of the parameter p, the light level required for 50% of the maximum phase-shifting response was calculated as the equivalent ED50 value. This relationship was described by the following equation (R2 > 0.99):
To examine the impact of daytime light amplitude, we ran simulations with low daytime light amplitude (maximum value of 200 lux, representative of remaining indoors all day), and a summer day with higher daytime light amplitude (maximum value of 800 lux, representative of a typical average day with outdoor light exposure in young adults (Scheuermaier et al., 2010; Thorne et al., 2009).
To examine the impact of morning bright light therapy, we ran simulations either with 40 lux (representing moderate indoor lighting) or 1500 lux (representing bright light therapy) for the 1 h immediately post-wake each day. We chose 1500 lux as representative average light input to the retina for an individual using bright light therapy of the kind used in interventions that showed phase-advancing effects (Crowley and Eastman, 2015; Gooley, 2008; Lack et al., 2007; Rosenthal et al., 1990). To examine the impact of bright light therapy in a likely clinical scenario, such as an individual with extreme late chronotype and extreme early chronotype, we ran simulations with a default tau parameter (τ = 24.15 h), a long tau (τ = 24.5 h), and a short tau (τ = 23.8 h).
For all simulations, the model was initialized over a simulated period of 21 days to allow the model to entrain and was then run over a subsequent 28 days with constraints on sleep to simulate a 5-day work week. At the end of the 28 days, we simulated a 48-h constant routine, with no light input (0 lux) and forced wakefulness. We set the modeling conditions to have sunrise at 0700 h and sunset at 1730 h (similar to late winter/early spring in Melbourne, London, or New York). To simulate an individual working a standard daytime work schedule, the model was required to be awake between 0700 and 1800 h on weekdays, with no sleep timing constraints on weekends. The model predicted daily circadian timing, expressed as core body temperature minimum (CBTmin). Predicted dim light melatonin onset (DLMO) was obtained by subtracting 7 h from predicted CBTmin, consistent with prior applications of the model (e.g., Stone et al., 2019; Woelders et al., 2017). The last DLMO for each scenario was calculated from the 48-h constant routine. Sleep onset and offset timing was extracted daily and summarized for work and free days. The difference in midsleep time between free days and work days was calculated as an indicator of social jetlag.
We note that the circadian model’s light input was originally defined in terms of photopic illuminance (lux) based on experimental data where light was measured in these units, rather than the more biologically relevant unit of melanopic illuminance. We have therefore also calculated equivalent melanopic illuminance values for presenting model simulations (assuming a Melanopic:Photopic (M:P) ratio of 0.55 based on the light sources used in the experiments defining the model; see Supplementary Material).
Results
Model simulations were performed for a range of assumptions on light exposure patterns, as well as the model’s sensitivity to light. To aid interpretation, we present the model’s light sensitivity in terms of the equivalent ED50 value (i.e., the light level required to elicit 50% of the maximum response). This was achieved by mapping the model parameter
Evening Light and Light Sensitivity Interact to Determine Circadian Timing
We first examined scenarios where evening light levels and circadian light sensitivity were varied to understand interactions between these 2 parameters (Figure 2). These simulations were run in the context of a dim (40 lux) versus bright (1500 lux) morning, and a dimmer (max 200 lux) versus brighter day (max 800 lux). To aid interpretation for these scenarios, we considered DLMO between 2000 and 2200 h as the “target range” for suitable entrainment for the work roster simulated where the individual was required to be awake by 7 a.m. each work morning (shown in white on Figures 2 and 3).
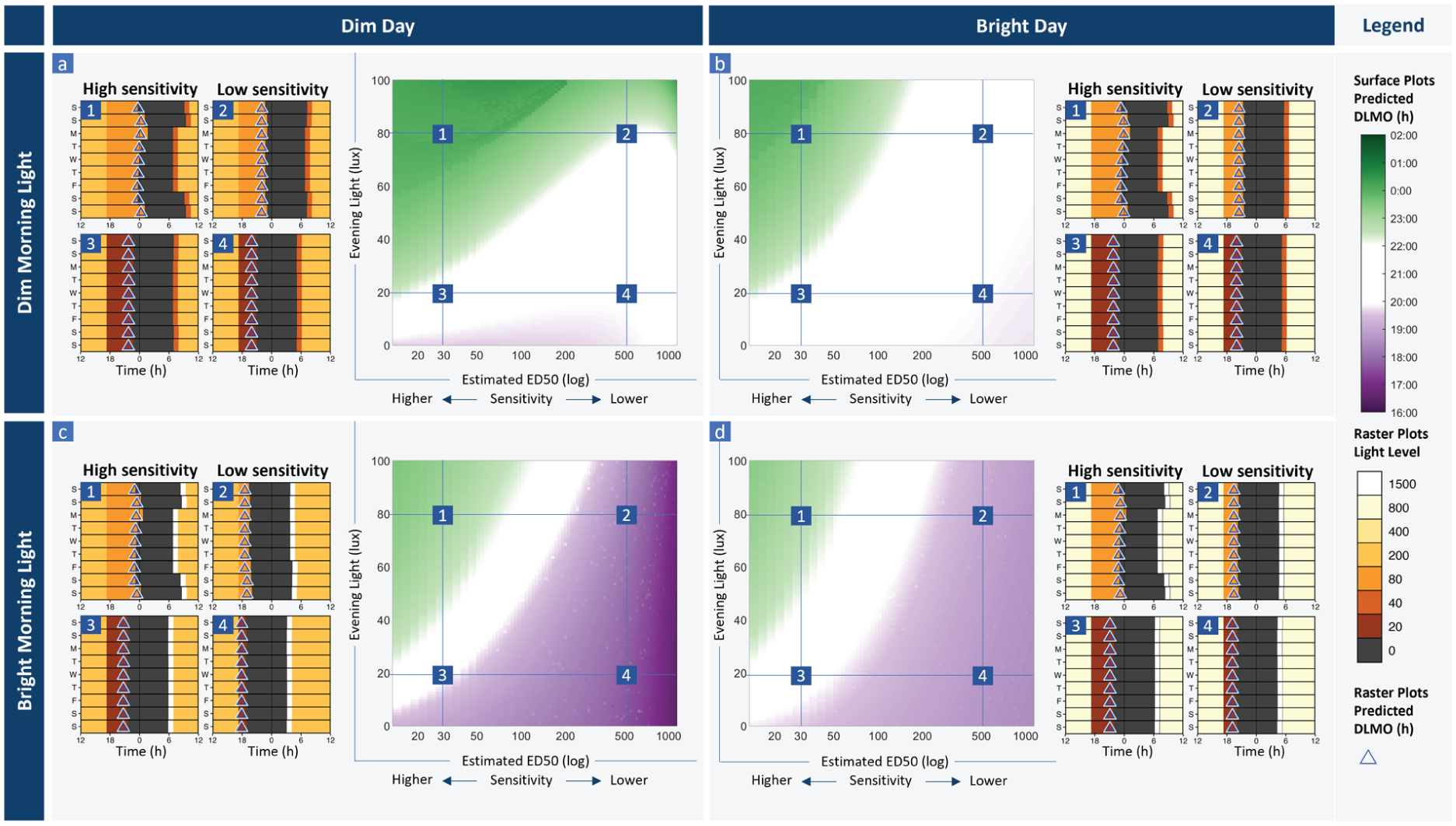
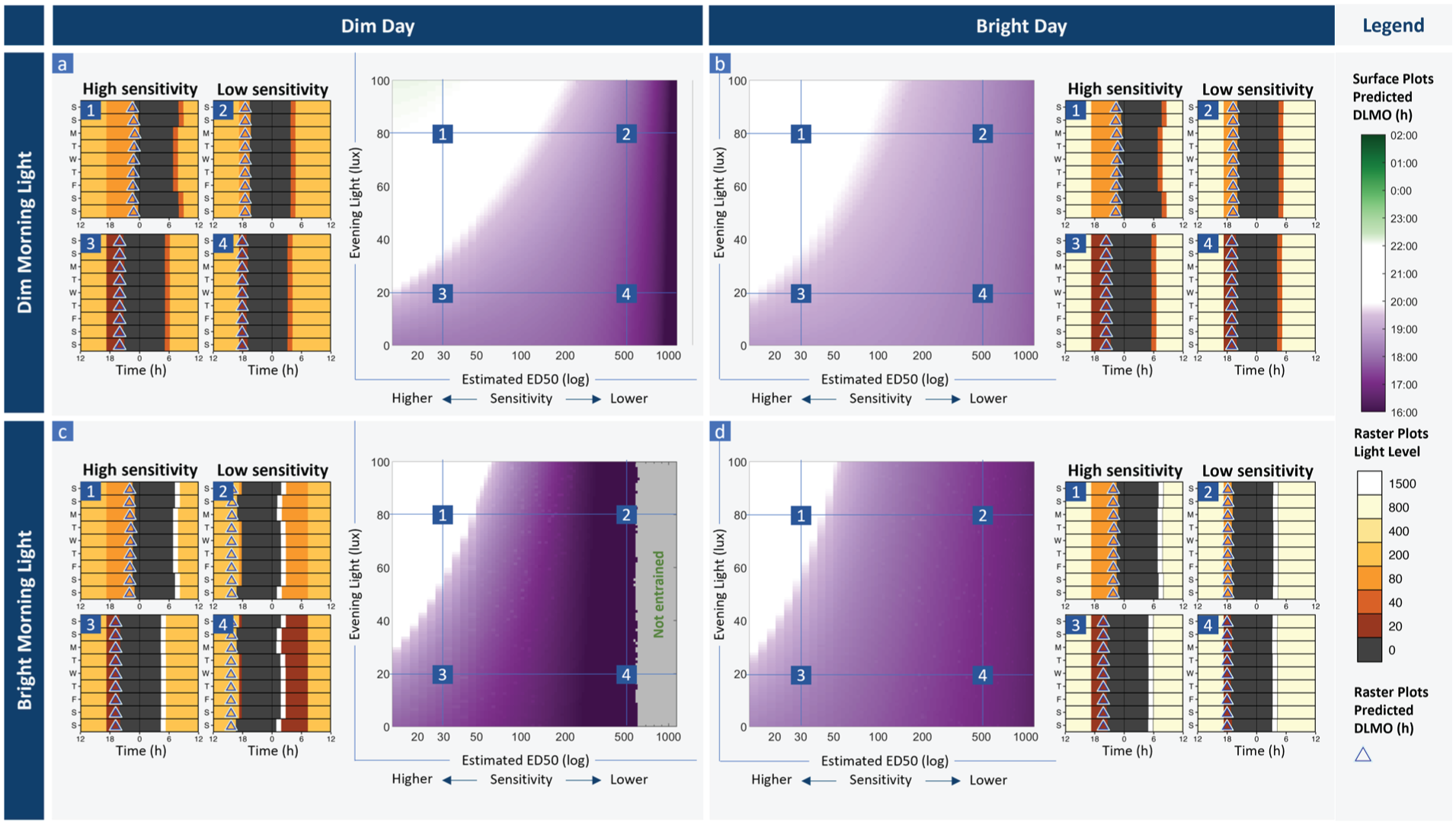
Model output representing entrainment profiles varying light sensitivity and evening light levels. Each panel shows a surface plot with predicted circadian phase (DLMO) for each combination of light sensitivity (modeled varying parameter p that modulates the shape of the dose response curve, reported as estimated ED50) and evening light level for (a) dim morning light (40 lux the hour after wake) and dim daytime light (max 200 lux), (b) dim morning light (40 lux) and bright daytime light (max 800 lux), (c) bright morning light (1500 lux) and dim daytime light (max 200 lux), and (d) bright morning light (1500 lux) and a bright daytime light (max 800 lux). In each case, we show raster plots for brighter (80 lux) evening light (1 and 2) and dimmer (20 lux) evening light (3 and 4), for a case with high circadian light sensitivity (ED50 = 30 lux) (1 and 3) and low circadian light sensitivity (ED50 = 500 lux) (2 and 4), corresponding to the marked points on each surface plot. Raster plots show the light inputs to the model for each scenario (with corresponding color legend), sleep timing (dark gray bars), and daily predicted DLMO (triangles) for the corresponding number on the surface plot. Here,
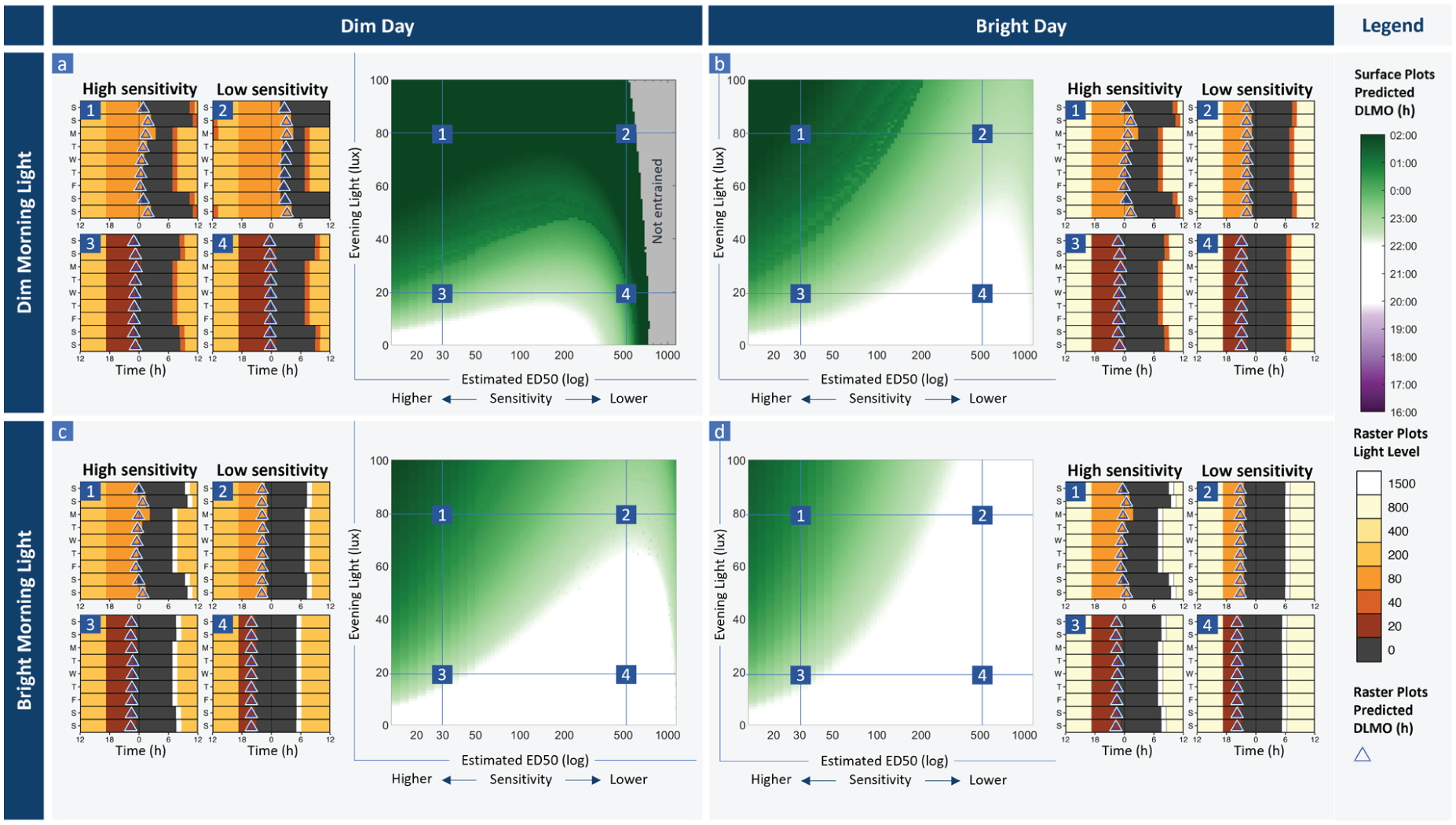
Simulations varying light sensitivity and evening light, for a late phenotype (tau = 24.5 h). Each panel shows a surface plot with predicted circadian phase (DLMO) for each combination of light sensitivity (modeled varying parameter p that modulates the shape of the dose-response curve, reported as estimated ED50) and evening light level for consistently (a) dim morning light (40 lux the hour after wake) and dimmer daytime light (max 200 lux), (b) dim morning light (40 lux) and a brighter day (max 800 lux), (c) bright morning light (1500 lux the hour after wake) and a dimmer day (max 200 lux), and (d) bright morning light (1500 lux) and a brighter day (max 800 lux). In each case, we show representative raster plots for bright evening light at 80 lux (1 and 2) and dimmer evening light at 20 lux (3 and 4), for a case with high circadian light sensitivity where ED50 = 30 lux (1 and 3) and low circadian light sensitivity where ED50 = 500 lux (2 and 4)), corresponding to the marked points on each surface plot. Raster plots show the light level input to the model each day, sleep timing (dark bars), and daily predicted circadian phase (DLMO) in triangles, over the last week of the modeled scenarios. Here tau is modeled as 24.50 h, representative of a late phenotype.
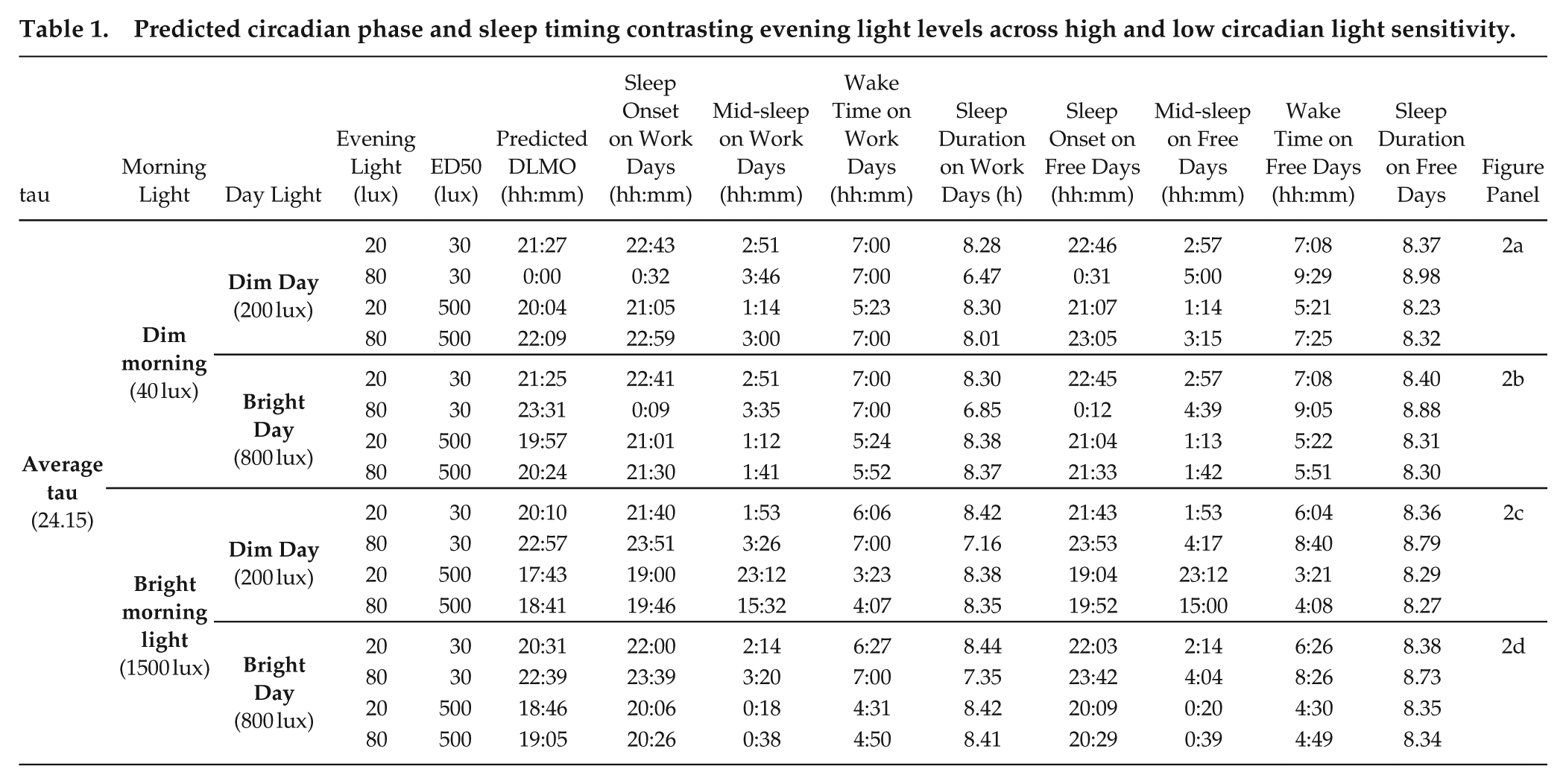
In general, increasing evening light levels led to later predicted circadian phase and later sleep timing. This effect was most pronounced for high levels of light sensitivity. Under dim morning conditions (Figure 2a and 2b) with low light sensitivity (ED50 = 500 lux), increasing evening light from 20 lux (DLMO = 20:04, Figure 2a4) to 80 lux (DLMO = 22:09, Figure 2a2) resulted in a 2 h 5 min delay in DLMO, but only a small change in weekday-weekend sleep timing (difference in midsleep time on weekdays vs weekends 0.01 vs 0.26 h, respectively; Table 1). Under dim morning conditions with high light sensitivity (ED50 = 30 lux), increasing evening light from 20 lux (DLMO = 21:27, Figure 2a3) to 80 lux (DLMO = 24:00, Figure 2a1) resulted in a 2 h 33 min delay in DLMO, with a substantial sleep restriction on weeknights (difference in midsleep time on weeknights vs weekends 0.10 vs 1.24 h, respectively; Table 1), including a late short sleep on the first night back at work (sleep onset 01:52 a.m., sleep duration 5 h 7 min), and shortened sleep duration persisting through the work week (mean worknight sleep duration 6 h 28 min).
Predicted circadian phase and sleep timing contrasting evening light levels across high and low circadian light sensitivity.
Bright Daytime Light Stabilizes Entrainment Across the Light Sensitivity Range
When we simulated brighter daytime light, the model displayed greater robustness to evening light levels, remaining within the target DLMO range across a wider range of light sensitivity and evening light combinations (Figure 2b). With brighter daytime light (max 800 lux), evening light up to 100 lux did not lead to DLMO delaying beyond 2200 h for lower light sensitivities (ED50 > 200 lux). As in the dim daytime light condition above, we observed a stronger relationship between evening light and DLMO with higher light sensitivity. Under bright daytime light conditions with low light sensitivity (ED50 = 500 lux), increasing evening light from 20 lux (DLMO = 19:57, Figure 2b4) to 80 lux (DLMO = 20:24, Figure 2b2) resulted in a 27 min delay in DLMO, compared with the 2 h 5 min delay observed for the same contrast with dim daytime light above. Under bright daytime, light conditions with high light sensitivity (ED50 = 30 lux), increasing evening light from 20 lux (DLMO = 21:25, Figure 2b3) to 80 lux (DLMO = 23:31, Figure 2b1) resulted in a 2 h 6 min delay in DLMO, compared with the 2 h 33 min delay observed for the same contrast with low daytime light above (Table 1).
Morning Bright Light Beneficial for Late Chronotypes, but Maladaptive in Some Cases
We simulated the effects of a 1-h bright (1500 lux) morning light pulse (Figure 2c and 2d), similar to bright light therapy used to aid circadian alignment (Gooley, 2008). In all scenarios, DLMO and sleep timing were earlier with the bright morning light, compared with 40 lux in the morning, as expected for a light pulse timed within the phase-advance region. This bright morning light was protective against delaying effects of bright evening light across a range of light sensitivities, with the DLMO target region spanning from ED50~90 lux to ED50~185 lux at an evening light level of 80 lux in Figure 2c. However, this range was narrower compared with the bright daytime light intervention (Figure 2b), where the DLMO target region was achieved from ED50~130 lux to ED50~1000 lux.
Interestingly, for a broad range of mid to low light sensitivity values (ED50 > 100 lux), the phase advances generated by bright morning light were large and potentially maladaptive, with DLMO occurring between ~17:00 and 19:00 and wake occurring in the early morning well before sunrise (~3-5 am). This advancing effect was particularly noticeable in the dim day condition (Figure 2c). In these cases, the day and evening light inputs were not sufficient to outweigh the strong morning light signal. In the case of low light sensitivity (Figure 2c2 and 2c4), the morning light pulse was essentially the dominant light signal to the pacemaker. In cases of high light sensitivity, the bright morning light was moderately protective against the phase delays induced by bright evening light seen in Figure 2a and 2b. For example, with high sensitivity (ED50 = 30 lux) and 80 lux evening light, DLMO was approximately 1 h earlier (22:57 Figure 2a1 vs 24:00 Figure 2b1, and 22:39 Figure 2c1 vs 23:31 Figure 2d1), with bright morning light versus dim morning light (52 min advance with a bright day, Figure 2b1 vs 2d1; 1 h 3 min advance with a dim day, Figures 2a1 and 2c1; Table 1), though predicted DLMO was still later than the target range (all > 22:39).
We also investigated the potential beneficial effects of bright morning light in the context of individuals with later chronotype, which we simulated using a long tau (24.5 h) (Figure 3). In this case, with dim daytime light and a dim morning (Figure 3a), we observed very delayed DLMO for all scenarios except very dim evening light levels (< 10 lux). With very low light sensitivity values (ED50 > 500 lux), the model was very delayed (DLMO between ~2 and 5 a.m.; Figure 3a2 and 3a4) or non-entrained. Both brighter daytime light (Figure 3b) and bright morning light (Figure 3c) were beneficial in phase advancing the model with a late chronotype, though the magnitude of this effect differed by light sensitivity. For low light sensitivity (ED50 = 500 lux), all interventions (dimmer evening light, brighter daytime light, bright morning light) led to circadian timing within the target range for this particular work schedule; only the combination of dim morning and dim daytime light (Figure 3a) had very late phase (DLMO = 00:36 at 20 evening lux, and 04:18 at 80 evening lux). For high light sensitivity (ED50 = 30 lux), all interventions led to a phase advance, but it was not always sufficient to achieve the target DLMO range. Bright morning light was more effective at bringing circadian timing into the target range compared with the bright day alone. This was particularly evident in cases with high light sensitivity (ED50 = 30 lux) and high evening light levels (80 lux). For example, brighter daytime light generated a 47 min phase advance in this scenario, compared with a 1 h 26 min advance for bright morning light, or a 1 h 56 min advance for the combination of brighter daytime light and bright morning light (Figure 3d, Table 2).
Predicted circadian phase and sleep timing contrasting evening light levels across high and low circadian light sensitivity for long and short tau.
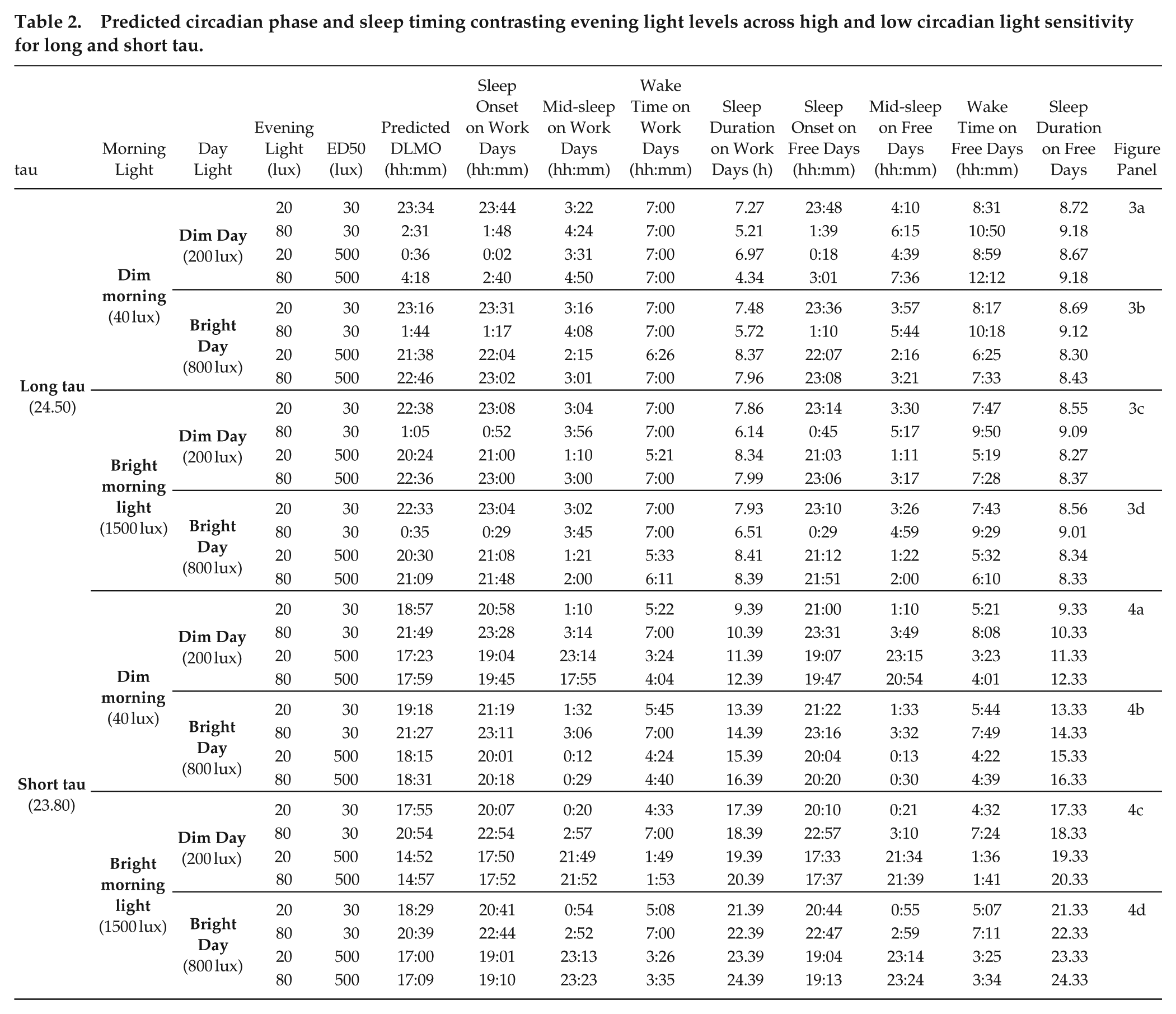
We also explored the pattern of results for an early chronotype, using short tau (23.8 h) (Figure 4). In this case, we saw moderate-to-extreme phase advances in most cases in the context of both a dim and bright day, particularly for moderate-to-low light sensitivity (ED50s 50-1000 lux). Brighter evening light was required to delay circadian timing into the target range, with evening light between 30 and 100 lux required for the high to moderate light sensitivity range, and greater than 100 lux needed for low light sensitivity (Figure 4a and 4b). The bright morning light pulse was particularly maladaptive for the early chronotype, leading to circadian timing much earlier than the target range in most cases (Figure 4c and 4d). In the context of a dim day, the bright morning light pulse led to very early DLMO times (e.g., DLMO = 14:52 at 20 evening lux for ED50 = 500 lux; Fig4c2, Table 2), and non-entrainment for cases with low light sensitivity (ED50 > 600 lux; Figure 4c). The extreme phase advances produced by the bright morning light were ameliorated somewhat in the context of a bright day, particularly for high light sensitivity (e.g., DLMO = 20:39 at 80 evening lux for ED50 = 30 lux; Figure 4d1, Table 2), though circadian timing remained earlier than the target range in most cases, particularly for low light sensitivity (e.g., DLMO = 17:00 at 20 evening lux for ED50 = 500 lux; Figure 4d2, Table 2).
Simulations varying light sensitivity and evening light, for an early phenotype (tau = 23.8 h). Each panel shows a surface plot with predicted circadian phase (DLMO) for each combination of light sensitivity (modeled varying parameter p that modulates the shape of the dose-response curve, reported as estimated ED50) and evening light level for consistently (a) dim morning light (40 lux the hour after wake) and dim daytime light (max 200 lux), (b) dim morning light (40 lux) and a bright day (max 800 lux), (c) bright morning light (1500 lux the hour after wake) and a dim day (max 200 lux), and (d) bright morning light (1500 lux) and a bright day (max 800 lux). In each case, we show representative raster plots for brighter evening light at 80 lux (1 and 2) and dimmer evening light at 20 lux (3 and 4), for a case with high circadian light sensitivity where ED50 = 30 lux (1 and 3) and low circadian light sensitivity where ED50 = 500 lux (2 and 4), corresponding to the marked points on each surface plot. Raster plots show the light level input to the model each day, sleep timing (dark bars), and daily predicted circadian phase (DLMO) in triangles, over the last week of the modeled scenarios. Here tau is modeled as 23.80 h, representative of an early phenotype.
Summary of Key Findings
Brighter evening light leads to more delayed phase and greater sleep restriction on work nights, whereas brighter morning light leads to more advanced phase.
Evening light interacts with circadian light sensitivity, and greater light sensitivity amplifies the effects of evening light.
More daytime light exposure led to being less sensitive to the phase delaying effects of evening light, particularly for lower light sensitivities.
Bright morning light was of specific benefit to late phenotypes, whereas daytime light was more beneficial for other phenotypes.
A summary of the predicted circadian phase and sleep variables contrasting low and high sensitivity at low and high evening light levels, across scenarios are presented in Tables 1 and 2.
Discussion
In this study, we investigated how circadian light sensitivity and light-dark patterns interact to determine sleep and circadian timing, using a computational model of the circadian system. We found that small changes in light sensitivity led to meaningful changes in sleep and circadian timing under realistic lighting assumptions. On a diurnal schedule, increasing light sensitivity amplified the effects of evening light, leading to later sleep and circadian timing in typical home lighting environments. We found that increasing daytime light was the most universally beneficial lighting intervention, reducing the delaying effects of evening light exposure. Bright morning light was of specific benefit to late chronotypes.
We found that brighter evening light led to later sleep and circadian timing, as well as more irregular sleep patterns. These effects were particularly pronounced with high circadian light sensitivity. From these model predictions, we can derive real-world recommendations. Converting photopic illuminance (the model’s original input) to the more biologically relevant melanopic illuminance (see Suppl. Table S1; Papatsimpa et al., 2021; Phillips et al., 2019; Tekieh et al., 2020), maintaining evening light below 8 to 11 melanopic equivalent daytime light illuminance (EDI) lux was broadly beneficial and was particularly important in cases where there was low daytime light, high circadian light sensitivity, and/or a late chronotype (long tau). This is consistent with recent lighting recommendations of a 10 melanopic EDI lux maximum for at least 3 h before bedtime (Brown et al., 2022), which is lower than the 18 melanopic EDI lux seen in actual homes (Cain et al., 2020). We consider this light level to be achievable (i.e., not prohibitively dim) with modern lighting design that independently adjusts melanopic illuminance while maintaining visual brightness. Notably, there were cases where dim evening light was not beneficial, particularly for short tau (Figure 4) or when bright morning light therapy was applied for the lower light sensitivity range (Figure 2). In these cases, even the brighter evening light levels tested (up to 100 photopic lux) were insufficient to counterbalance the phase-advancing effects of morning/daytime light exposures, leading to maladaptive phase advances. This has implications for older adults, who likely have lower light sensitivity (Duffy et al., 2007) and earlier chronotype (Fischer et al., 2017), where brighter evening light may actually be important for stabilizing rhythms. It may also be important with respect to sex differences in intrinsic circadian period, given findings that 35% of women, compared with 14% of men, have a tau less than 24 h (Duffy et al., 2011). These differential indications for evening light highlight the potential pitfalls of applying lighting interventions with a one-size-fits-all approach.
Our simulations revealed broadly beneficial effects of brighter daytime light exposure for stabilizing sleep and circadian timing, as well as mitigating delaying effects of evening light exposure across a wide range of light sensitivity values. This finding is consistent with prior modeling studies, which showed brighter daytime light levels led to earlier circadian timing and reduced interindividual variability (Papatsimpa et al., 2021; Skeldon et al., 2017). This model prediction also aligns with experimental findings that increasing exposure to natural light through the day and reducing evening light exposure leads to earlier circadian timing and less interindividual variability (Wright et al., 2013). Similar patterns are found examining sleep-based chronotype, whereby more time spent in outdoor daytime light is related to earlier chronotype (Roenneberg and Merrow, 2007). The beneficial effects of daytime light exposure on sleep and circadian timing are likely due to a combination of two factors: (1) light history effects of bright daytime light exposure reducing sensitivity to the evening light levels, which occurs due to conversion of the photoreceptor pool in the model from ready to activated, and (2) strengthening of circadian amplitude by light exposure during the circadian day, leading to more robust rhythms. Light history is known to influence the magnitude of both phase shifting (Chang et al., 2011) and melatonin suppression (Hébert et al., 2002), such that prior dim light enhances sensitivity to subsequent light.
Daytime light exposure has been linked to a wide range of health benefits. Experimental and epidemiological work find that consistent bright daytime light exposure is broadly beneficial for sleep (Mottram et al., 2011), mental health (Burns et al., 2023; Wirz-Justice et al., 1996), and health outcomes (Aries et al., 2015; Bodis et al., 2009). The beneficial effects of daytime light do not appear to be tied closely to the timing of the light. This runs contrary to a direct phase-shifting explanation, since light would have either phase-advancing or phase-delaying effects, depending on its specific timing. However, it is consistent with an amplitude-based explanation, as light exposure throughout the circadian day has an amplitude-boosting effect for the central circadian clock (Walch et al., 2025). Enhanced central circadian amplitude may help to organize peripheral rhythms, due to stronger output signals from the central clock (Windred et al., 2024a). Our model predictions showing universal benefits of brighter daytime light provide a potential explanation for daytime light’s broad health benefits and support enhanced daytime light as a promising intervention to stabilize sleep and circadian timing, and consequently improve health.
There is strong evidence for the potential benefits of circadian-informed lighting in the home (Papatsimpa and Linnartz, 2020; Vetter et al., 2022). Our results indicate that circadian-informed lighting could be optimized by accounting for an individual’s circadian light sensitivity. We simulated differences in light sensitivity that have applications to a wide range of scenarios where we would expect differences in light sensitivity. For example, there is accumulating evidence of differences in light sensitivity across age groups (Crowley et al., 2015; Eto and Higuchi, 2023; Hartstein et al., 2022; Lee et al., 2018; Sletten et al., 2009), in clinical populations including sleep/circadian rhythm disorders (Abbott et al., 2021; Aoki et al., 2001; Watson et al., 2018) and mood disorders (Bullock et al., 2019; Lewy et al., 1985; McGlashan et al., 2019; Roecklein et al., 2013), and across different medication classes (Hallam et al., 2005a, 2005b; McGlashan et al., 2018). Our results demonstrate the importance of careful consideration of the potential circadian side effects of medications, as altering light sensitivity, without modifying the light-dark cycle, can have significant implications for entrainment. In some cases, altering sensitivity could push an individual into a very delayed, very advanced, or even non-entrained condition. On the other hand, it may also be possible to use pharmacological interventions to reduce circadian sensitivity in cases of higher light sensitivity where modifications to the light environment are insufficient, such as individuals with a very long intrinsic circadian period.
Interactions between human circadian physiology (circadian light sensitivity) and environmental conditions cannot easily be explored systematically in the real world. Using a modeling approach enabled us to test complex interactions, yielding important insights into how these factors interact, and identifying candidate strategies to improve entrainment. Similar model-based approaches have previously been taken to examine effects of artificial light and social constraints on sleep and circadian outcomes (Skeldon et al., 2017), explore light considerations for the adaptation to daylight savings times (Xu et al., 2024), and the role of light patterns on chronotype distributions (Papatsimpa et al., 2021). Our work adds to these findings by highlighting the important role of light sensitivity, in addition to lighting conditions and other physiological factors such as intrinsic circadian period. In our investigation, we modified a parameter that specifically controls the shape of the model’s dose-response curve to light. There are other photic parameters in the model, including a parameter that modulates the shape of the phase-response curve (Stone et al., 2020a). However, individual differences in this factor have not been characterized to date. Therefore, we selected a parameter that is most analogous to the variations demonstrated in human physiology (Phillips et al., 2019; Zeitzer et al., 2000). In addition, we mapped this model parameter to an estimated ED50 (a physiologically measurable analog), enabling more direct comparisons between experimental work and model-based predictions.
Understanding how light sensitivity interacts with light exposure is important for understanding sleep phenotypes (e.g., differences in light sensitivity explaining how an individual’s light pattern/behavior has led to circadian disruption), and for designing personalized lighting strategies to promote stable entrainment. Our modeling approach could be used to simulate the expected distribution/range of responses of a given population such as in the context of mood disorders, or for a medication class, and use these to inform lighting design or light interventions. Similarly, light design for groups of individuals could draw from this approach to forecast expected interactions and use them to develop robust lighting strategies, including in groups where phase shifts are required to adapt to a work roster, such as shift workers, or to mitigate jetlag. We note that for these scenarios we set a target DLMO range of 2000 to 2200 h, but this target is context-dependent and may be adjusted to suit the individual situation. This type of modeling could also be used to explore other physiological outputs, such as circadian amplitude or melatonin suppression across different contexts. A key innovation presented here is linking the dose-response curve parameter (p) to an estimated ED50 value. This enables model calibration to a well-established biological construct that is measurable, albeit via a reasonably intensive experimental paradigm such as a melatonin suppression protocol. Ultimately, translation of this approach to a widely applicable tool will likely require development of low-burden proxies for light sensitivity.
Some limitations should be noted. In these simulations, we varied circadian parameters (light sensitivity, intrinsic period) and environmental parameters (lighting), while using default parameters for sleep homeostatic components of the model. This approach enables exploration of the physiological drivers of sleep timing, but it does not account for day-to-day changes in behavior due to psychological or social reasons. Future work could consider how other physiological variability (e.g., homeostatic pressure) or age-related changes interact with variations in circadian light sensitivity. Similarly, we have not accounted for different photoperiods or work rosters. It is likely that more irregular day-to-day light inputs, such as seen in shift workers, could lead to greater instability in circadian timing (Postnova et al., 2012, 2014, 2013), presenting a greater challenge for entrainment particularly for individuals who are hyper/hypo-sensitive. Future work could generalize our approach to these scenarios.
Our approach shows how modeling can be used to map the nuanced interactions between light environments and circadian light sensitivity, across different real-world scenarios. Our results contribute to understanding of how circadian light sensitivity can be used to tailor individual-level solutions that support optimal sleep and circadian timing.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304251391268 – Supplemental material for Who Needs Bright Light and When? Mapping the Interactions of Lighting Environments and Individual Differences in Circadian Light Sensitivity
Supplemental material, sj-docx-1-jbr-10.1177_07487304251391268 for Who Needs Bright Light and When? Mapping the Interactions of Lighting Environments and Individual Differences in Circadian Light Sensitivity by Julia E. Stone, Dorothee Steven, Weiqi Cheng, Sean W. Cain and Andrew J. K. Phillips in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank Eliah Castiello for his graphical design support. This work was supported by the Australian Research Council via grants awarded to AJKP (FT240100815, DP210102924, and DP220102812). JES was supported by an NHMRC Fellowship (2025333). WC was supported by a Monash Graduate Scholarship and Monash International Tuition Scholarship.
Author Contributions
JES, AJKP, SC, and DF contributed to the conception of the project. AJKP wrote the initial code, JES carried out the simulations. JES and WC prepared the data visualizations. JES and AJKP wrote the paper with input from all authors.
Ethical Considerations
Ethical approval was not required.
Consent To Participate
Not applicable.
Consent For Publication
Not applicable.
Conflict of Interest Statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JES, DS, and WC have no conflicts of interest with respect to the research, authorship and/or publication of this article. SWC and AJKP have received research funding from Delos, Beacon Lighting, and Versalux. SWC and AJKP are co-founders and co-directors of Circadian Health Innovations PTY LTD. SWC has consulted for Dyson.
Data Availability
Data is available upon reasonable request.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.